Effect of Circadian Rhythm Disturbance on the Human Musculoskeletal System and the Importance of Nutritional Strategies
Abstract
1. Introduction
2. Methods
3. Human Circadian Rhythm
3.1. The Biological Clock
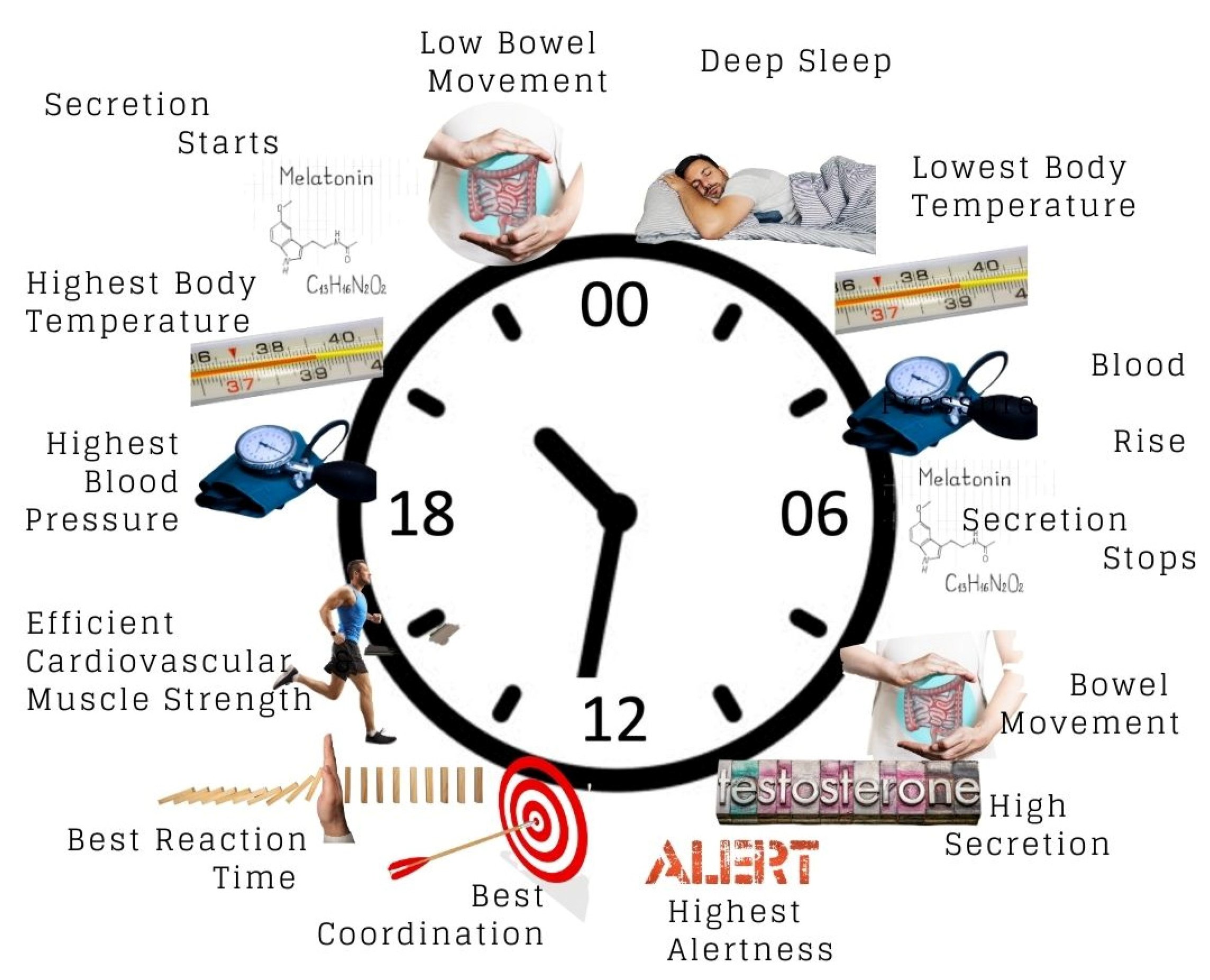
3.2. How Is Circadian Rhythm Measured?
3.3. Effect of Circadian Rhythm on the Body
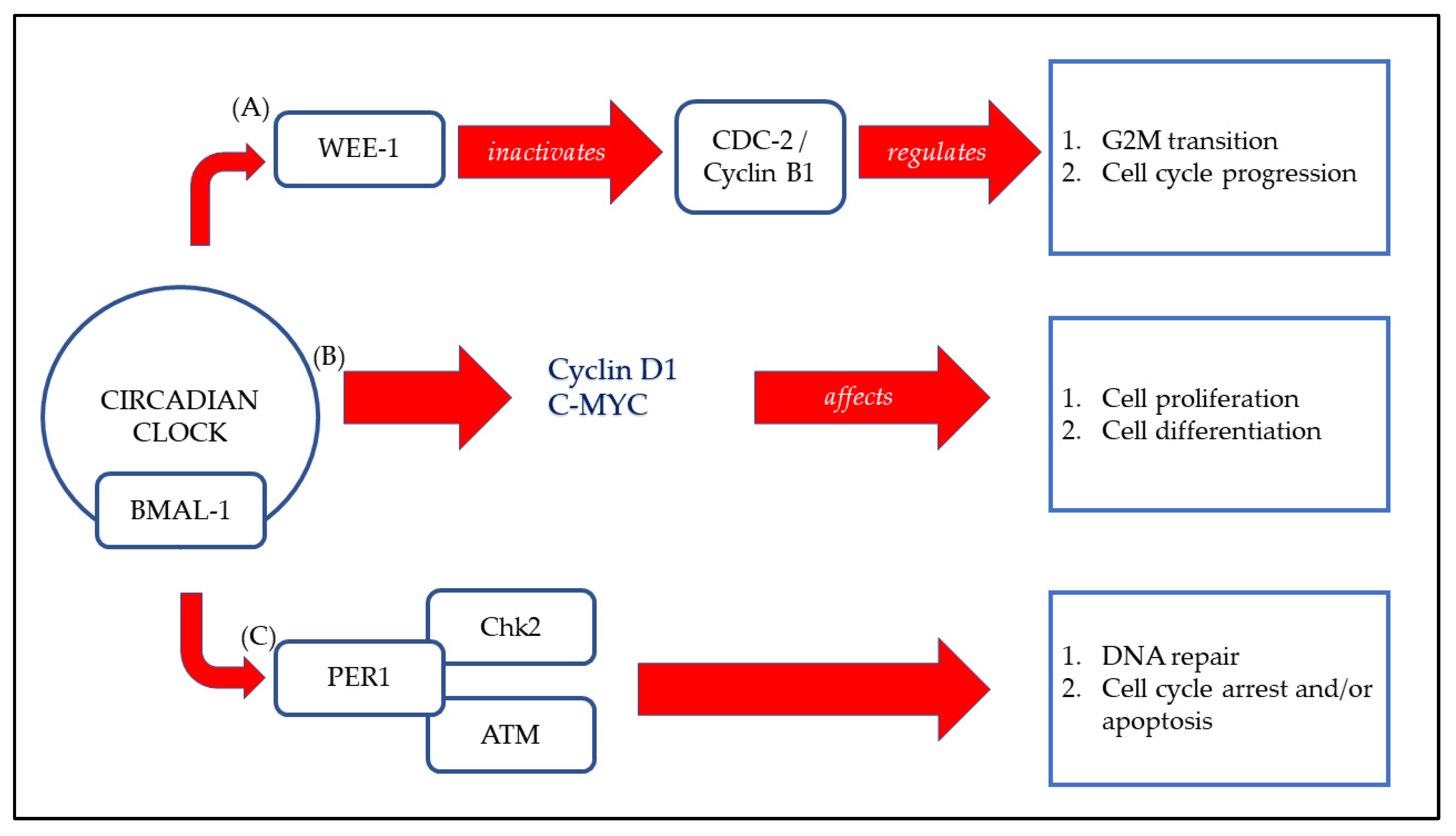
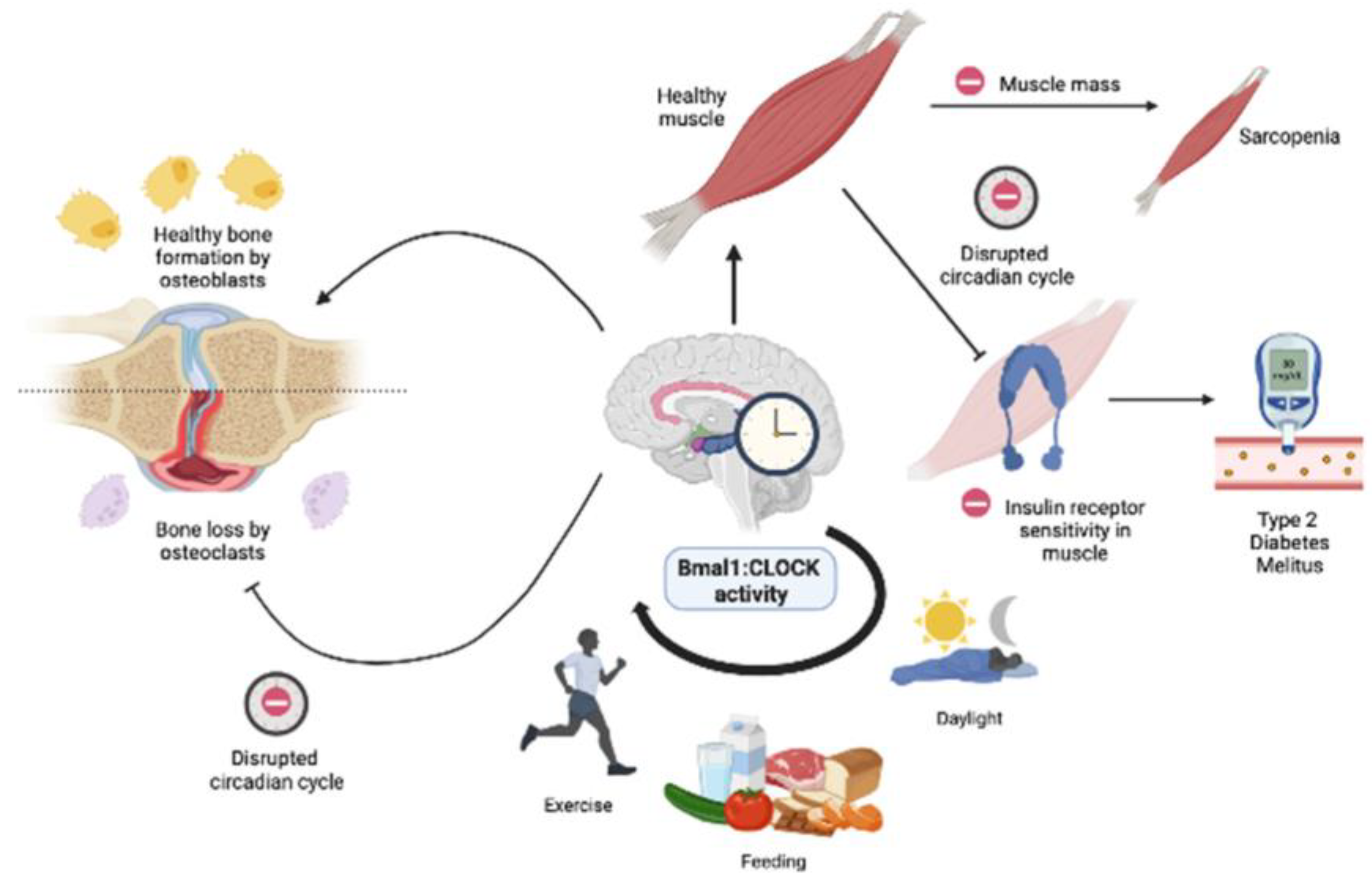
3.4. Effect of Circadian Rhythm on the Musculoskeletal System
3.5. Role of Skeletal Muscle Circadian Clock in Lipid and Glucose Metabolism
4. Circadian Rhythm and Bone Health
Less Number of Hours of Sleep and Associated Bone Defects and Osteoporosis
5. Nutritional Strategies for Musculoskeletal Health
6. Conclusions and Future Directions of Research
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mohd Azmi, N.A.S.; Juliana, N.; Azmani, S.; Mohd Effendy, N.; Abu, I.F.; Mohd Fahmi Teng, N.I.; Das, S. Cortisol on Circadian Rhythm and Its Effect on Cardiovascular System. Int. J. Environ. Res. Public Health 2021, 18, 676. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, S.; Shibata, S. The Role of Circadian Rhythms in Muscular and Osseous Physiology and Their Regulation by Nutrition and Exercise. Front. Neurosci. 2017, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lahens, N.F.; Balance, H.I.; Hughes, M.E.; Hogenesch, J.B. A circadian gene expression atlas in mammals: Implications for biology and medicine. Proc. Natl. Acad. Sci. USA 2014, 111, 16219–16224. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Monreal, M.A.; Harmsen, J.F.; Schrauwen, P.; Esser, K.A. Ticking for Metabolic Health: The Skeletal-Muscle Clocks. Obesity 2020, 28 (Suppl. 1), S46–S54. [Google Scholar] [CrossRef] [PubMed]
- Mansingh, S.; Handschin, C. Time to Train: The Involvement of the Molecular Clock in Exercise Adaptation of Skeletal Muscle. Front. Physiol. 2022, 13, 902031. [Google Scholar] [CrossRef] [PubMed]
- Mayeuf-Louchart, A.; Staels, B.; Duez, H. Skeletal muscle functions around the clock. Diabetes Obes. Metab. 2015, 17 (Suppl. 1), 39–46. [Google Scholar] [CrossRef] [PubMed]
- Tahara, Y.; Aoyama, S.; Shibata, S. The mammalian circadian clock and its entrainment by stress and exercise. J. Physiol. Sci. 2017, 67, 1–10. [Google Scholar] [CrossRef]
- Luo, B.; Zhou, X.; Tang, Q.; Yin, Y.; Feng, G.; Li, S.; Chen, L. Circadian rhythms affect bone reconstruction by regulating bone energy metabolism. J. Transl. Med. 2021, 19, 410. [Google Scholar] [CrossRef]
- Tian, Y.; Ming, J. The role of circadian rhythm in osteoporosis; a review. Front. Cell Dev. Biol. 2022, 10, 960456. [Google Scholar] [CrossRef]
- Mohd Azmi, N.A.S.; Juliana, N.; Mohd Fahmi Teng, N.I.; Azmani, S.; Das, S.; Effendy, N. Consequences of Circadian Disruption in Shift Workers on Chrononutrition and their Psychosocial Well-Being. Int. J. Environ. Res. Public Health 2020, 17, 2043. [Google Scholar] [CrossRef]
- Mohd Fuad, S.H.; Juliana, N.; Mohd Azmi, N.A.S.; Mohd Fahmi Teng, N.I.; Azmani, S.; Abu, I.F.; Das, S. Circadian Disruption and Occupational Toxicants Exposure Affecting the Immunity of Shift Workers During SARS CoV-2 Pandemic. Front. Public Health 2022, 10, 829013. [Google Scholar] [CrossRef] [PubMed]
- Potter, G.D.; Skene, D.J.; Arendt, J.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Circadian rhythm and sleep disruption: Causes, metabolic consequences, and countermeasures. Endocr. Rev. 2016, 37, 584–608. [Google Scholar] [CrossRef] [PubMed]
- Stothard, E.R.; McHill, A.W.; Depner, C.M.; Birks, B.R.; Moehlman, T.M.; Ritchie, H.K.; Guzzetti, J.R.; Chinoy, E.D.; LeBourgeois, M.K.; Axelsson, J.; et al. Circadian Entrainment to the Natural Light-Dark Cycle across Seasons and the Weekend. Curr. Biol. 2017, 27, 508–513. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Mojón, A.; Fernández, J.R. Influence of Circadian Time of Hypertension Treatment on Cardiovascular Risk: Results of the MAPEC Study. Chronobiol. Int. 2010, 27, 1629–1651. [Google Scholar] [CrossRef]
- Ohashi, N.; Shinsuke, I.; Sayaka, I.; Hideo, Y. Circadian Rhythm of Blood Pressure and the Renin-Angiotensin System in the Kidney. Hypertension Res. 2017, 40, 413–422. [Google Scholar] [CrossRef]
- Geneva, I.I.; Javaid, W. Disruption of the Body Temperature Circadian Rhythm in Hospitalized Patients. Am. J. Med. Sci. 2021, 362, 578–585. [Google Scholar] [CrossRef]
- Refinetti, R. The Circadian Rhythm of Body Temperature. Front. Biosc. 2010, 15, 564–594. [Google Scholar] [CrossRef]
- Gnocchi, D.; Bruscalupi, G. Circadian Rhythms and Hormonal Homeostasis: Pathophysiological Implications. Biology 2017, 6, 10. [Google Scholar] [CrossRef]
- Scheer, F.A.J.L.; Steven, A.S. Human Circadian System Causes a Morning Peak in Prothrombotic Plasminogen Activator Inhibitor-1 (PAI-1) Independent of the Sleep/Wake Cycle. Blood 2014, 123, 590–593. [Google Scholar] [CrossRef]
- Boege, H.L.; Mehreen, Z.B.; Marie, P.S. Circadian Rhythms and Meal Timing: Impact on Energy Balance and Body Weight. Curr. Opin. Biotechnol. 2021, 70, 1–6. [Google Scholar] [CrossRef]
- Morrison, M.; Shona, L.H.; Jonathon, W.; John, A.H. Sleep, Circadian Biology and Skeletal Muscle Interactions: Implications for Metabolic Health. Sleep Med. Rev. 2022, 66, 101700. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Ribelayga, C.P.; Mangel, S.C. A Circadian Clock in the Retina Regulates Rod-Cone Gap Junction Coupling and Neuronal Light Responses via Activation of Adenosine A2A Receptors. Front. Cell. Neurosci. 2021, 14, 605067. [Google Scholar] [CrossRef] [PubMed]
- Saini, C.; Suter, D.M.; Liani, A.; Gos, P.; Schibler, U. The mammalian circadian timing system: Synchronization of peripheral clocks. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Brainard, J.; Gobel, M.; Scott, B.; Koeppen, M.; Eckle, T. Health implications of disrupted circadian rhythms and the potential for daylight as therapy. Anesthesiol 2015, 122, 1170–1175. [Google Scholar] [CrossRef] [PubMed]
- Gale, J.E.; Cox, H.I.; Qian, J.; Block, G.D.; Colwell, C.S.; Matveyenko, A.V. Disruption of circadian rhythms accelerates development of diabetes through pancreatic beta-cell loss and dysfunction. J. Biol. Rhythm. 2011, 26, 423–433. [Google Scholar] [CrossRef]
- Yao, X.; Heidebrecht, B.L.; Chen, J.; Tyson, J.J. Mathematical Analysis of Robustness of Oscillations in Models of the Mammalian Circadian Clock. PLoS Comput. Biol. 2022, 18, e1008340. [Google Scholar] [CrossRef]
- Yoshitane, H.; Asano, Y.; Sagami, A.; Sakai, S.; Suzuki, Y.; Okamura, H.; Iwasaki, W.; Ozaki, H.; Fukada, Y. Functional D-box sequences reset the circadian clock and drive mRNA rhythms. Commun. Biol. 2019, 2, 300. [Google Scholar] [CrossRef]
- Fagiani, F.; Di Marino, D.; Romagnoli, A.; Travelli, C.; Voltan, D.; Di Cesare Mannelli, L.; Racchi, M.; Govoni, S.; Lanni, C. Molecular regulations of circadian rhythm and implications for physiology and diseases. Signal Transduct. Target. Ther. 2022, 7, 41. [Google Scholar] [CrossRef]
- Vitaterna, M.H.; King, D.P.; Chang, A.M.; Kornhauser, J.M.; Lowrey, P.L.; McDonald, J.D.; Dove, W.F.; Pinto, L.H.; Turek, F.W.; Takahashi, J.S. Mutagenesis and mapping of a mouse gene, Clock, essential for circadian behavior. Science 1994, 264, 719–725. [Google Scholar] [CrossRef]
- Rijo-Ferreira, F.; Joseph, S.T. Genomics of Circadian Rhythms in Health and Disease. Genome Med. 2019, 11, 82. [Google Scholar] [CrossRef]
- Robles, M.S.; Cox, J.; Mann, M. In-vivo quantitative proteomics reveals a key contribution of post-transcriptional mechanisms to the circadian regulation of liver metabolism. PLoS Genet. 2014, 10, e1004047. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Marhon, S.A.; Zhang, Y.; Steger, D.J.; Won, K.J.; Lazar, M.A. Rev-erbα dynamically modulates chromatin looping to control circadian gene transcription. Science 2018, 359, 1274–1277. [Google Scholar] [CrossRef] [PubMed]
- Dumont, M.; Paquet, J. Progressive decrease of melatonin production over consecutive days of simulated night work. Chronobiol. Int. 2014, 31, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Zisapel, N. New Perspectives on the Role of Melatonin in Human Sleep, Circadian Rhythms and Their Regulation. Br. J. Pharmacol. 2018, 175, 3190–3199. [Google Scholar] [CrossRef]
- Govindarajulu, M.; Patel, M.Y.; Wilder, D.M.; Long, J.B.; Arun, P. Blast Exposure Dysregulates Nighttime Melatonin Synthesis and Signaling in the Pineal Gland: A Potential Mechanism of Blast-Induced Sleep Disruptions. Brain Sci. 2022, 12, 1340. [Google Scholar] [CrossRef]
- Smith, K.A.; Martin, W.S.; Charles, A.C. Adaptation of Human Pineal Melatonin Suppression by Recent Photic History. J. Clin. Endocrinol. Metabol. 2004, 89, 3610–3614. [Google Scholar] [CrossRef]
- Vetter, C.; Pattison, P.M.; Houser, K.; Herf, M.; Phillips, A.J.; Wright, K.P.; Skene, D.J.; Brainard, G.C.; Boivin, D.B.; Glickman, G. A review of human physiological responses to light: Implications for the development of integrative lighting solutions. Leukos 2022, 18, 387–414. [Google Scholar] [CrossRef]
- Burgess, H.J.; Wyatt, J.K.; Park, M.; Fogg, L.F. Home Circadian Phase Assessments with Measures of Compliance Yield Accurate Dim Light Melatonin Onsets. Sleep 2015, 38, 889–897. [Google Scholar] [CrossRef]
- Reid, K.J. Assessment of circadian rhythms. Neurol. Clin. 2019, 37, 505–526. [Google Scholar] [CrossRef]
- Reddy, S.; Reddy, V.; Sharma, S. Physiology, Circadian Rhythm. [Updated 2022 May 8]. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Hasselberg, M.J.; McMahon, J.; Parker, K. The validity, reliability, and utility of the iButton(R) for measurement of body temperature circadian rhythms in sleep/wake research. Sleep Med. 2013, 14, 5–11. [Google Scholar] [CrossRef]
- Boubekri, M.; Cheung, I.N.; Reid, K.J.; Wang, C.H.; Zee, P.C. Impact of windows and daylight exposure on overall health and sleep quality of office workers: A case-control pilot study. J. Clin. Sleep Med. 2014, 10, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Cheung, I.N.; Zee, P.C.; Shalman, D.; Malkani, R.G.; Kang, J.; Reid, K.J. Morning and Evening Blue Enriched Light Exposure Alters Metabolic Function in Normal Weight Adults. PLoS ONE 2016, 11, e0155601. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, J.; Folkard, S.; Dongen, H.V.; Minors, D.; Owens, D.; Kerkhof, G.; Weinert, D.; Nevill, A.; Macdonald, I.; Sytnik, N.; et al. Temperature profiles, and the effect of sleep on them, in relation to morningness-eveningness in healthy female subjects. Chronobiol. Int. 2001, 18, 227–247. [Google Scholar] [CrossRef] [PubMed]
- Juda, M.; Vetter, C.; Roenneberg, T. The Munich ChronoType Questionnaire for Shift-Workers (MCTQShift). J. Boil. Rhythm. 2013, 28, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Logan, R.W.; McClung, C.A. Rhythms of life: Circadian disruption and brain disorders across the lifespan. Nat. Rev. Neurosc. 2018, 20, 49–65. [Google Scholar] [CrossRef]
- Lunsford-Avery, J.R.; Kollins, S.H. Delayed Circadian Rhythm Phase: A Cause of Late-Onset ADHD among Adolescents? J. Child Psychol. Psychiatry Allied Disciplines 2018, 59, 1248. [Google Scholar] [CrossRef]
- Van Andel, E.; Bijlenga, D.; Vogel, S.W.; Beekman, A.T.; Kooij, J.S. Effects of chronotherapy on circadian rhythm and ADHD symptoms in adults with attention-deficit/hyperactivity disorder and delayed sleep phase syndrome: A randomized clinical trial. Chronobiol. Int. 2021, 38, 260–269. [Google Scholar] [CrossRef]
- Rahmouni, K. Leptin-Induced Sympathetic Nerve Activation: Signaling Mechanisms and Cardiovascular Consequences in Obesity. Curr. Hyperten. Rev. 2010, 6, 104. [Google Scholar] [CrossRef]
- Buonfiglio, D.; Parthimos, R.; Dantas, R.; Cerqueira, S.R.; Gomes, G.; Andrade-Silva, J.; Ramos-Lobo, A.; Amaral, F.G.; Matos, R.; Sinésio, J., Jr.; et al. Melatonin Absence Leads to Long-Term Leptin Resistance and Overweight in Rats. Front. Endocrinol. 2018, 9, 122. [Google Scholar] [CrossRef]
- Puchalski, S.S.; Green, J.N.; Rasmussen, D.D. Melatonin effect on rat body weight regulation in response to high-fat diet at middle age. Endocrine 2003, 21, 163–167. [Google Scholar] [CrossRef]
- Walecka-Kapica, E.; Klupińska, G.; Chojnacki, J.; Tomaszewska-Warda, K.; Błońska, A.; Chojnacki, C. The effect of melatonin supplementation on the quality of sleep and weight status in postmenopausal women. Przegląd Menopauzalny = Menopause Rev. 2014, 13, 334. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.X.; Li, X.N.; Yang, G.Y.; Zhang, X.; Li, W.X.; Zhang, Q.Q.; Pan, H.X.; Zhang, H.H.; Zhou, M.Y.; Wang, Y.D.; et al. Circadian misalignment alters insulin sensitivity during the light phase and shifts glucose tolerance rhythms in female mice. PLoS ONE 2019, 14, e0225813. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.Q.; Ansari, T.S.; McGuinness, O.P.; Wasserman, D.H.; Johnson, C.H. Circadian Disruption Leads to Insulin Resistance and Obesity. Curr. Biol. 2013, 23, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Su, S.; McCall, W.V.; Isales, C.; Snieder, H.; Wang, X. Rest-activity circadian rhythm and impaired glucose tolerance in adults: An analysis of NHANES 2011–2014. BMJ Open Diabetes Res. Care 2022, 10, e002632. [Google Scholar] [CrossRef] [PubMed]
- Mazzoccoli, G.; de Cosmo, S.; Mazza, T. The biological clock: A pivotal hub in non-alcoholic fatty liver disease pathogenesis. Front. Physiol. 2018, 9, 193. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Kim, J.H. Impact of Skeletal Muscle Mass on Metabolic Health. Endocrinol. Metabol. 2020, 35, 1. [Google Scholar] [CrossRef]
- Dudek, M.; Gossan, N.; Yang, N.; Im, H.J.; Ruckshanthi, J.P.; Yoshitane, H.; Li, X.; Jin, D.; Wang, P.; Boudiffa, M.; et al. The chondrocyte clock gene Bmal1 controls cartilage homeostasis and integrity. J. Clin. Investig. 2016, 126, 365. [Google Scholar] [CrossRef]
- Xu, C.; Ochi, H.; Fukuda, T.; Sato, S.; Sunamura, S.; Takarada, T.; Hinoi, E.; Okawa, A.; Takeda, S. Circadian Clock Regulates Bone Resorption in Mice. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2016, 31, 1344–1355. [Google Scholar] [CrossRef]
- Yuan, G.; Hua, B.; Yang, Y.; Xu, L.; Cai, T.; Sun, N.; Yan, Z.; Lu, C.; Qian, R. The Circadian Gene Clock Regulates Bone Formation Via PDIA3. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2017, 32, 861–871. [Google Scholar] [CrossRef]
- Zhang, J.; Cohen, A.; Shen, B.; Du, L.; Tasdogan, A.; Zhao, Z.; Shane, E.J.; Morrison, S.J. The effect of parathyroid hormone on osteogenesis is mediated partly by osteolectin. Proc. Natl. Acad. Sci. USA 2021, 118, e2026176118. [Google Scholar] [CrossRef]
- Chiavistelli, S.; Giustina, A.; Mazziotti, G. Parathyroid hormone pulsatility: Physiological and clinical aspects. Bone Res. 2015, 3, 14049. [Google Scholar] [CrossRef] [PubMed]
- Giustina, A.; Berardelli, R.; Gazzaruso, C.; Mazziotti, G. Insulin and GH–IGF-I axis: Endocrine pacer or endocrine disruptor? Acta Diabetol. 2015, 52, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Hand, L.E.; Dickson, S.H.; Freemont, A.J.; Ray, D.W.; Gibbs, J.E. The circadian regulator Bmal1 in joint mesenchymal cells regulates both joint development and inflammatory arthritis. Arthritis Res. Ther. 2019, 21, 5. [Google Scholar] [CrossRef] [PubMed]
- Dyar, K.A.; Hubert, M.J.; Mir, A.A.; Ciciliot, S.; Lutter, D.; Greulich, F.; Quagliarini, F.; Kleinert, M.; Fischer, K.; Eichmann, T.O.; et al. Transcriptional programming of lipid and amino acid metabolism by the skeletal muscle circadian clock. PLoS Biol. 2018, 16, e2005886. [Google Scholar] [CrossRef]
- Miller, B.H.; McDearmon, E.L.; Panda, S.; Hayes, K.R.; Zhang, J.; Andrews, J.L.; Antoch, M.P.; Walker, J.R.; Esser, K.A.; Hogenesch, J.B.; et al. Circadian and CLOCK-controlled regulation of the mouse transcriptome and cell proliferation. Proc. Natl. Acad. Sci. USA 2007, 104, 3342–3347. [Google Scholar] [CrossRef]
- Basse, A.L.; Dalbram, E.; Larsson, L.; Gerhart-Hines, Z.; Zierath, J.R.; Treebak, J.T. Skeletal muscle insulin sensitivity show circadian rhythmicity which is independent of exercise training status. Front. Physiol. 2018, 9, 1198. [Google Scholar] [CrossRef]
- Harriet Wallberg-Henriksson, J.R.Z. GLUT4: A key player regulating glucose homeostasis? Insights from transgenic and knockout mice. Mol. Membr. Biol. 2001, 18, 205–211. [Google Scholar] [CrossRef]
- Abdul-Ghani, M.A.; Defronzo, R.A. Pathogenesis of insulin resistance in skeletal muscle. J. Biomed. Biotechnol. 2010, 2010, 476279. [Google Scholar] [CrossRef]
- Gurley, J.M.; Griesel, B.A.; Olson, A.L. Increased Skeletal Muscle GLUT4 Expression in Obese Mice After Voluntary Wheel Running Exercise Is Posttranscriptional. Diabetes 2016, 65, 2911. [Google Scholar] [CrossRef]
- Aoyama, S.; Nakahata, Y.; Shinohara, K. Chrono-Nutrition Has Potential in Preventing Age-Related Muscle Loss and Dysfunction. Front. Neurosc. 2021, 15, 16. [Google Scholar] [CrossRef]
- Nakata, M.; Kumari, P.; Kita, R.; Katsui, N.; Takeuchi, Y.; Kawaguchi, T.; Yamazaki, T.; Zhang, B.; Shimba, S.; Yada, T. Circadian Clock Component BMAL1 in the Paraventricular Nucleus Regulates Glucose Metabolism. Nutrients 2021, 13, 4487. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Jeong, J.H.; Hong, S.C. The impact of sleep and circadian disturbance on hormones and metabolism. Int. J. Endocrinol. 2015, 2015, 591729. [Google Scholar] [CrossRef]
- Doi, R.; Oishi, K.; Ishida, N. CLOCK regulates circadian rhythms of hepatic glycogen synthesis through transcriptional activation of Gys2. J. Biol. Chem. 2010, 285, 22114–22121. [Google Scholar] [CrossRef]
- Lamia, K.A.; Storch, K.F.; Weitz, C.J. Physiological significance of a peripheral tissue circadian clock. Proc. Natl. Acad. Sci. USA 2008, 105, 15172–15177. [Google Scholar] [CrossRef]
- Zhang, E.E.; Liu, Y.; Dentin, R.; Pongsawakul, P.Y.; Liu, A.C.; Hirota, T.; Nusinow, D.A.; Sun, X.; Landais, S.; Kodama, Y.; et al. Cryptochrome mediates circadian regulation of cAMP signaling and hepatic gluconeogenesis. Nat. Med. 2010, 16, 1152–1156. [Google Scholar] [CrossRef]
- Schiaffino, S.; Blaauw, B.; Dyar, K.A. The functional significance of the skeletal muscle clock: Lessons from Bmal1 knockout models. Skelet. Muscle 2016, 6, 33. [Google Scholar] [CrossRef]
- Dyar, K.A.; Ciciliot, S.; Wright, L.E.; Biensø, R.S.; Tagliazucchi, G.M.; Patel, V.R.; Forcato, M.; Paz, M.I.; Gudiksen, A.; Solagna, F.; et al. Muscle insulin sensitivity and glucose metabolism are controlled by the intrinsic muscle clock. Mol. Metabol. 2014, 3, 29–41. [Google Scholar] [CrossRef]
- Harmsen, J.F.; van Polanen, N.; van Weeghel, M.; Wefers, J.; Hoeks, J.; Vaz, F.M.; Pras-Raves, M.L.; van Kampen, A.H.C.; Schaart, G.; van Moorsel, D.; et al. Circadian misalignment disturbs the skeletal muscle lipidome in healthy young men. FASEB J. 2021, 35, e21611. [Google Scholar] [CrossRef]
- Wefers, J.; van Moorsel, D.; Hansen, J.; Connell, N.J.; Havekes, B.; Hoeks, J.; van Marken, L.W.D.; Duez, H.; Phielix, E.; Kalsbeek, A.; et al. Circadian misalignment induces fatty acid metabolism gene profiles and compromises insulin sensitivity in human skeletal muscle. Proc. Natl. Acad. Sci. USA 2018, 115, 7789–7794. [Google Scholar] [CrossRef]
- Poggiogalle, E.; Jamshed, H.; Peterson, C.M. Circadian regulation of glucose, lipid, and energy metabolism in humans. Metabolism 2018, 84, 11–27. [Google Scholar] [CrossRef]
- Schilperoort, M.; Bravenboer, N.; Lim, J.; Mletzko, K.; Busse, B.; van Ruijven, L.; Kroon, J.; Rensen, P.C.N.; Kooijman, S.; Winter, E.M. Circadian disruption by shifting the light-dark cycle negatively affects bone health in mice. FASEB J. 2020, 34, 1052–1064. [Google Scholar] [CrossRef]
- Lucassen, E.A.; Coomans, C.P.; van Putten, M.; de Kreij, S.R.; van Genugten, J.H.L.T.; Sutorius, R.P.M.; de Rooij, K.E.; van der Velde, M.; Verhoeve, S.L.; Smit, J.W.A.; et al. Environmental 24-hr Cycles Are Essential for Health. Curr. Biol. 2016, 26, 1843–1853. [Google Scholar] [CrossRef]
- Takarada, T.; Xu, C.; Ochi, H.; Nakazato, R.; Yamada, D.; Nakamura, S.; Kodama, A.; Shimba, S.; Mieda, M.; Fukasawa, K.; et al. Bone Resorption Is Regulated by Circadian Clock in Osteoblasts. J. Bone Min. Res. 2017, 32, 872–881. [Google Scholar] [CrossRef]
- Shao, P.; Ohtsuka-Isoya, M.; Shinoda, H. Circadian rhythms in serum bone markers and their relation to the effect of etidronate in rats. Chronobiol. Int. 2009, 20, 325–336. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, C.H.; Meng, Y. MicroRNA-1297 promotes the progression of osteoporosis through regulation of osteogenesis of bone marrow mesenchymal stem cells by targeting WNT5A. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 4541–4550. [Google Scholar] [CrossRef]
- Li, T.; Zhang, S.; Yang, Y.; Zhang, L.; Yuan, Y.; Zou, J. Co-regulation of circadian clock genes and microRNAs in bone metabolism. J. Zhejiang Univ.-SCIENCE B 2022, 23, 529–546. [Google Scholar] [CrossRef]
- Samsa, W.E.; Vasanji, A.; Midura, R.J.; Kondratov, R.V. Deficiency of circadian clock protein BMAL1 in mice results in a low bone mass phenotype. Bone 2016, 84, 194–203. [Google Scholar] [CrossRef]
- Kc, R.; Li, X.; Voigt, R.M.; Ellman, M.B.; Summa, K.C.; Vitaterna, M.H.; Keshavarizian, A.; Turek, F.W.; Meng, Q.J.; Stein, G.S.; et al. Environmental disruption of circadian rhythm predisposes mice to osteoarthritis-like changes in knee joint. J. Cell. Physiol. 2015, 230, 2174–2183. [Google Scholar] [CrossRef]
- Swanson, C.M.; Shea, S.A.; Wolfe, P.; Cain, S.W.; Munch, M.; Vujovic, N.; Czeisler, C.A.; Buxton, O.M.; Orwoll, E.S. Bone Turnover Markers After Sleep Restriction and Circadian Disruption: A Mechanism for Sleep-Related Bone Loss in Humans. J. Clin. Endocrinol. Metab. 2017, 102, 3722–3730. [Google Scholar] [CrossRef]
- Stone, K.L.; Ewing, S.K.; Lui, L.Y.; Ensrud, K.E.; Ancoli-Israel, S.; Bauer, D.C.; Cauley, J.A.; Hillier, T.A.; Cummings, S.R. Self-reported sleep and nap habits and risk of falls and fractures in older women: The study of osteoporotic fractures. J. Am. Geriatr. Soc. 2006, 54, 1177–1183. [Google Scholar] [CrossRef]
- Everson, C.A.; Folley, A.E.; Toth, J.M. Chronically Inadequate Sleep Results in Abnormal Bone Formation and Abnormal Bone Marrow in Rats. Exp. Biol. Med. 2012, 237, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Choi, Y.J.; Chung, Y.S. Other than daytime working is associated with lower bone mineral density: The Korea National Health and Nutrition Examination Survey 2009. Calcif. Tissue Int. 2013, 93, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Santhanam, P.; Khthir, R.; Dial, L.; Driscoll, H.K.; Gress, T.W. Femoral Neck Bone Mineral Density in Persons Over 50 Years Performing Shiftwork: An Epidemiological Study. J. Occup. Environ. Med. 2016, 58, e63–e65. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.; Li, Y.; Zong, G.; Guo, Y.; Li, J.; Manson, J.E.; Hu, F.B.; Willett, W.C.; Schernhammer, E.S.; Bhupathiraju, S.N. Rotating night shift work and adherence to unhealthy lifestyle in predicting risk of type 2 diabetes: Results from two large US cohorts of female nurses. BMJ 2018, 363, k4641. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, I. Interaction between bone and glucose metabolism [Review]. Endocr. J. 2017, 64, 1043–1053. [Google Scholar] [CrossRef] [PubMed]
- Swanson, C.M.; Kohrt, W.M.; Wolfe, P.; Wright, K.P.; Shea, S.A.; Cain, S.W.; Munch, M.; Vujović, N.; Czeisler, C.A.; Orwoll, E.S.; et al. Rapid suppression of bone formation marker in response to sleep restriction and circadian disruption in men. Osteoporos. Int. 2019, 30, 2485–2493. [Google Scholar] [CrossRef]
- Hettiarachchi, M.; Cooke, R.; Norton, C.; Jakeman, P. Temporal Change in Biomarkers of Bone Turnover Following Late Evening Ingestion of a Calcium-Fortified, Milk-Based Protein Matrix in Postmenopausal Women with Osteopenia. Nutrients 2019, 11, 1413. [Google Scholar] [CrossRef]
- Depner, C.M.; Rice, J.D.; Tussey, E.J.; Eckel, R.H.; Bergman, B.C.; Higgins, J.A.; Melanson, E.L.; Kohrt, W.M.; Wright, K.P., Jr.; Swanson, C.M. Bone turnover marker responses to sleep restriction and weekend recovery sleep. Bone 2021, 152, 116096. [Google Scholar] [CrossRef]
- Ochs-Balcom, H.M.; Hovey, K.M.; Andrews, C.; Cauley, J.A.; Hale, L.; Li, W.; Bea, J.W.; Sarto, G.E.; Stefanick, M.L.; Stone, K.L.; et al. Short Sleep is Associated with Low Bone Mineral Density and Osteoporosis in the Women’s Health Initiative. J. Bone Min. Res. 2020, 35, 261–268. [Google Scholar] [CrossRef]
- Norton, C.; Hettiarachchi, M.; Cooke, R.; Kozior, M.; Kontro, H.; Daniel, R.; Jakeman, P. Effect of 24-Week, Late-Evening Ingestion of a Calcium-Fortified, Milk-Based Protein Matrix on Biomarkers of Bone Metabolism and Site-Specific Bone Mineral Density in Postmenopausal Women with Osteopenia. Nutrients 2022, 14, 3486. [Google Scholar] [CrossRef]
- McElderry, J.D.; Zhao, G.; Khmaladze, A.; Wilson, C.G.; Franceschi, R.T.; Morris, M.D. Tracking circadian rhythms of bone mineral deposition in murine calvarial organ cultures. J. Bone Min. Res. 2013, 28, 1846–1854. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, A.L.; Dsouza, C.; Julien, C.; Rummler, M.; Gaumond, M.H.; Cermakian, N.; Willie, B.M. Bone adaptation to mechanical loading in mice is affected by circadian rhythms. Bone 2022, 154, 116218. [Google Scholar] [CrossRef] [PubMed]
- Hirai, T.; Tanaka, K.; Togari, A. β-adrenergic receptor signaling regulates Ptgs2 by driving circadian gene expression in osteoblasts. J. Cell Sci. 2014, 127 Pt 17, 3711–3719. [Google Scholar] [CrossRef] [PubMed]
- Fujihara, Y.; Kondo, H.; Noguchi, T.; Togari, A. Glucocorticoids mediate circadian timing in peripheral osteoclasts resulting in the circadian expression rhythm of osteoclast-related genes. Bone 2022, 61, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Kinoshita, S.; Shimba, S.; Ozono, K.; Michigami, T. Sympathetic activation induces skeletal Fgf23 expression in a circadian rhythm-dependent manner. J. Biol. Chem. 2014, 289, 1457–1466. [Google Scholar] [CrossRef] [PubMed]
- Maronde, E.; Schilling, A.F.; Seitz, S.; Schinke, T.; Schmutz, I.; Horst, G.; Van Der, A.M.; Albrecht, U. The Clock Genes Period 2 and Cryptochrome 2 Differentially Balance Bone Formation. PLoS ONE 2010, 5, e11527. [Google Scholar] [CrossRef]
- Kondratov, R.V.; Kondratova, A.A.; Gorbacheva, V.Y.; Vykhovanets, O.V.; Antoch, M.P. Early aging and age-related pathologies in mice deficient in BMAL1, the core component of the circadian clock. Genes Dev. 2006, 20, 1868–1873. [Google Scholar] [CrossRef]
- Li, X.; Liu, N.; Gu, B.; Hu, W.; Li, Y.; Guo, B.; Zhang, D. BMAL1 regulates balance of osteogenic—Osteoclastic function of bone marrow mesenchymal stem cells in type 2 diabetes mellitus through the NF-κB pathway. Mol. Biol. Rep. 2018, 45, 1691–1704. [Google Scholar] [CrossRef]
- Noguchi, T.; Hussein, A.I.; Horowitz, N.; Carroll, D.; Gower, A.C.; Demissie, S.; Gerstenfeld, L.C. Hypophosphatemia Regulates Molecular Mechanisms of Circadian Rhythm. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Dudek, M.; Yang, N.; Ruckshanthi, J.P.; Williams, J.; Borysiewicz, E.; Wang, P.; Adamson, A.; Li, J.; Bateman, J.F.; White, M.R.; et al. The intervertebral disc contains intrinsic circadian clocks that are regulated by age and cytokines and linked to degeneration. Ann. Rheum. Dis. 2017, 76, 576–584. [Google Scholar] [CrossRef]
- Karpouzos, A.; Diamantis, E.; Farmaki, P.; Savvanis, S.; Troupis, T. Nutritional aspects of bone health and fracture healing. J. Osteoporos. 2017, 2017, 4218472. [Google Scholar] [CrossRef]
- Elma, Ö.; Yilmaz, S.T.; Deliens, T.; Clarys, P.; Nijs, J.; Coppieters, I.; Polli, A.; Malfliet, A. Chronic Musculoskeletal Pain and Nutrition: Where Are We and Where Are We Heading? J. Inj. Funct. Rehab. 2020, 12, 1268–1278. [Google Scholar] [CrossRef]
- Arjmandi, B.H.; Foley, E.M. Evidence for the Effectiveness of Soy in Aging and Improving Quality of Life. In Aging—Life Span and Life Expectancy; Reynolds, R.J., Day, S.M., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Lian, N.; Luo, K.; Xie, H.; Kang, Y.; Tang, K.; Lu, P.; Li, T. Obesity by High-Fat Diet Increases Pain Sensitivity by Reprogramming Branched-Chain Amino Acid Catabolism in Dorsal Root Ganglia [Original Research]. Front. Nutr. 2022, 9, 902635. [Google Scholar] [CrossRef]
- Fischer, V.; Haffner-Luntzer, M.; Amling, M.; Ignatius, A. Calcium and vitamin D in bone fracture healing and post-traumatic bone turnover. Eur. Cell Mater 2018, 35, 365–385. [Google Scholar] [CrossRef]
- Zhu, K.; Prince, R.L. Calcium and bone. Clin. Biochem. 2012, 45, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Winzenberg, T.; Shaw, K.; Fryer, J.; Jones, G. Effects of calcium supplementation on bone density in healthy children: Meta-analysis of randomised controlled trials. BMJ 2006, 333, 775. [Google Scholar] [CrossRef] [PubMed]
- Huncharek, M.; Muscat, J.; Kupelnick, B. Impact of dairy products and dietary calcium on bone-mineral content in children: Results of a meta-analysis. Bone 2008, 43, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Masood, T.; Kushwaha, R.S.; Singh, R.; Sailwal, S.; Pandey, H.; Varma, A.; Singh, R.K.; Cornelissen, G. Circadian rhythm of serum 25 (OH) vitamin D, calcium and phosphorus levels in the treatment and management of type-2 diabetic patients. Drug Discov. Ther. 2015, 9, 70–74. [Google Scholar] [CrossRef]
- Walton, J.C.; Walker, W.H., 2nd; Bumgarner, J.R.; Meléndez-Fernández, O.H.; Liu, J.A.; Hughes, H.L.; Kaper, A.L.; Nelson, R.J. Circadian Variation in Efficacy of Medications. Clin. Pharmacol. Ther. 2021, 109, 1457–1488. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D and bone health. J. Nutr. 1996, 126 (Suppl. 4), 1159S–1164S. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.S.; Yu, S.; Kim, C.; Lee, H.J.; Yoon, I.Y.; Kim, T. Lower Serum Calcium Levels Associated with Disrupted Sleep and Rest–Activity Rhythm in Shift Workers. Nutrients 2022, 14, 3021. [Google Scholar] [CrossRef] [PubMed]
- Maurya, V.K.; Aggarwal, M. Factors influencing the absorption of vitamin D in GIT: An overview. J. Food Sci. Technol. 2017, 54, 3753–3765. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhai, Y.; Zhang, J.; Chen, J.Y.; Liu, D.; Zhao, W.H. Combined effects of physical activity and calcium on bone health in children and adolescents: A systematic review of randomized controlled trials. World J. Pediatrics 2020, 16, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.H. School-based nutrition interventions can improve bone health in children and adolescents. Osteoporos. Sarcopenia 2021, 7, 1–5. [Google Scholar] [CrossRef]
- Hidayat, K.; Chen, J.S.; Wang, T.C.; Liu, Y.J.; Shi, Y.J.; Su, H.W.; Liu, B.; Qin, L.Q. The Effects of Milk Supplementation on Bone Health Indices in Adults: A Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2022, 13, 1186–1199. [Google Scholar] [CrossRef]
- Golem, D.L.; Martin-Biggers, J.T.; Koenings, M.M.; Davis, K.F.; Byrd-Bredbenner, C. An integrative review of sleep for nutrition professionals. Adv. Nutr. 2014, 5, 742–759. [Google Scholar] [CrossRef]
- Doherty, R.; Madigan, S.; Warrington, G.; Ellis, J. Sleep and Nutrition Interactions: Implications for Athletes. Nutrients 2019, 11, 822. [Google Scholar] [CrossRef]
- Potter, G.D.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Nutrition and the circadian system. Br. J. Nutr. 2016, 116, 434–442. [Google Scholar] [CrossRef]
- Blume, C.; Garbazza, C.; Spitschan, M. Effects of light on human circadian rhythms, sleep and mood. Somnologie 2019, 23, 147–156. [Google Scholar] [CrossRef]
| Bone Disorders/Abnormalities | Study Population/In Vivo/In Vitro/Ex Vivo | Circadian Disorders/Disturbance |
|---|---|---|
| Bone loss and suppression of bone formation marker | Young adult men [97] | Sleep restriction with concurrent circadian disruption induced a relatively rapid decline in bone formation marker; N-terminal propeptide of type 1 procollagen (P1NP) and levels remained lower with ongoing exposure. |
| Post-menopausal osteopenia | Postmenopausal women [98] | Short-term sleep restriction and circadian disruption can adversely affect bone metabolism, and it was concluded that there is no P1NP recovery with ongoing exposure that, taken together, could lead to lower bone density over time. |
| Bone turnover marker (BTM) level | Young adult men and women [99] | The diurnal rhythm of bone remodelling is affected by nocturnal dietary patterns. |
| Femoral neck bone mineral density and content | Shift workers (men and women) age 50 years and above [94] | Intake of a dairy-based protein supplement fortified with calcium at bedtime potentiates nocturnal bone resorption rates in osteopenic postmenopausal women. |
| Total bone mineral density | Postmenopausal women [100] | N-terminal propeptide of type 1 procollagen (P1NP), osteocalcin, and C-telopeptide of type 1 collagen (CTX) have no significant effects on sleep restriction, either with or without the opportunity for recovery sleep. |
| Post-menopausal osteopenia | Postmenopausal women [101] | The effect of sleep restriction on bone metabolism potentially changed with other contributing factors such as age, sex, weight change, and circadian disruption. |
| Bone misconstruction, early osteoporosis | In vivo animal model; long-term continuous light exposure to mice strain C57BL/6 [83] | Middle-aged male shift workers have a significantly higher total femur and femoral bone mineral content (BMC) than women. Despite having physiological disturbances and hormonal changes due to shift work for years, there are no differences in history of fracture prevalence in men and women. |
| Bone mineralisation/bone formation | Animal model and ex vivo organ culture; neonatal murine calvarial organ cultures [102] | Sleeping 5 h or less per night was found to have significantly higher odds of low bone mass when compared to 7 h of night sleep. The chances of having osteoporosis of the total hip, spine, and whole body were at higher risk with short sleep duration. |
| Bone resorption and bone density | Cell and gene knockdown model; osteoblast-specific Bmal1-knockout mice and Bmal1 deficiency osteoblast cell culture [84] | Intake of dietary calcium daily at bedtime results in a significant reduction in biomarkers on homeostatic bone remodelling. The intervention, however, did not change the site-specific bone mineral density or trabecular bone score. |
| Bone formation in response to mechanical loading | Animal model; sham-loaded mice (in vivo mechanical loading) [103] | Continuous exposure to artificial light disrupts circadian rhythm and affects trabecular density. Data showed that the central circadian rhythm of SCN and the trabecular density decreased, and an inflammatory response was induced. As SCN neurons rebound to normal circadian rhythms, the early osteoporosis gradually recovered. |
| Implications of circadian oscillators in response to sympathetic nervous system activation | Cell and light–dark cycle animal model; gene knockdown (siRNA transfection) in MC3T3-E1 osteoblastic cells [104] | Bone mineralisation is regulated by the local circadian oscillator signalling pathway, and Per1 expression is involved in the process. However, the regulation of circadian signalling in this mechanism remains vague. Data suggest that circadian signalling plays a role in intramembranous ossification, particularly in the nucleation of apatite minerals. However, the roles of circadian rhythm in mineral propagation and crystallinity are still unclear. Due to this limitation, the current data suggest no influence. |
| Bone resorption | Cell; osteoclast derived from RAW264.7 [105] | Bone mass and bone resorption are regulated by the osteoblastic circadian clock system. Bmal1-expressed osteoblast, a core component of the circadian clock systems, inhibits the mechanism of bone resorption. Osteoblast-specific deficient Bmal1 results in low bone mass, specifically the lower BMD in the femur and tibia. This shows that Bmal1 inhibits osteoclastogenesis through its expression in osteoblasts. RANKL and osteoclasts increase in Bmal1 knockout mice, while in vitro study shows Bmal1 deficiency does not affect the osteoblast differentiation and maturation. In summary, the deletion of Bmal1 in osteoblasts recapitulates the deletion phenotype. Thus, higher bone formation, higher bone resorption, and lower bone mass. In vitro study shows Bmal1 regulates osteoclastogenesis and bone resorption via RANKL expression in osteoblasts. |
| Osteoblast regulation | Cell and gene knockdown models; Bmal1 knockout mice with light and dark cycle exposure. [106] | Circadian clocks influence mechanoresponse. This study shows that daily rhythms of clock genes are displayed in bone tissue, and mechanical loading affects circadian rhythm and bone response, and that time of day at which loading is applied affects this mechanism as well. |
| Osteoclast differentiation and bone mass | Cell and gene knockdown models; Bmal1-specific knockout mice and in vitro osteoclast cell differentiation [59] | Osteocyte mechanoresponsive genes Sost and Dkk1 are differentially regulated based on the time of day, as it moderately affects the response of bone formation in response to the in vivo mechanical loading in mice. This study shows the involvement of circadian genes, and from the zeitgeber data (ZT) of the mechanical loading, ZT14 showed greater endocortical bone formation compared to ZT2. The finding showed that loading time played a role in bone formation. |
| Osteoblast regulation and bone formation | Cell and gene knockout models; Per2 and Cry2 knockout mice and osteoclast cells [107] | Osteoblast and sympathetic nervous system are involved in bone formation by modulating the core clock genes via the β-adrenergic receptor (β-AR) in osteoblast. This study shows that a transcriptional factor, Nfil3 (a non-selective β-AR agonist) regulates the expression of Ptgs2 involving circadian clock genes Per2 and Bmal2 expression. |
| Bone formation and osteoblast formation abnormalities | Cell and gene knockout model; Bmal1 knockout mice and L929 cell [88,108] | These data suggest that glucocorticoids are involved in the transmission of the circadian timing from the SCN to peripheral osteoclasts. The osteoclast peripheral clock plays a role in the circadian rhythm and may be regulated by CTSK and NFATc1 expression. |
| Diabetes mellitus and bone metabolism | Cell and gene knockout model; Goto–Kakizaki rats and bone marrow mesenchymal stem cell [109] | Phosphate metabolism and sympathetic tone are activated during food intake. Phosphate metabolism is regulated by circadian rhythm, and phosphate is an important metabolite for bone metabolism. Data show that fibroblast growth factor 23 (Fgf23) regulates phosphorus levels in osteoblasts. Additionally, skeletal Fgf23 expression levels are high, consistent with urine epinephrine (marker for sympathetic tone) level. In addition, Fgf23 serum levels from mice are low in the daytime but elevated at night. All these data show that circulating Fgf23 level influences the excretion of phosphate in urine, and phosphate metabolism depends on circadian clock network and is relevant to food intake-associated sympathetic activation. |
| Low phosphate intake and bone healing | Cell and low phosphate-fed animal model [110] | Osteoclast differentiation decreased and bone mass increased in a Bmal1-specific knockout mouse. Cell-based assay and animal study showed that BMAL1 upregulated the calcineurin-dependent 1 (NCATc1) expression by binding to the E-box element of the NFATc1 promoter incorporation to the CLOCK genes. In addition, the steroid receptor coactivator (SRS) family was also involved in the interaction and regulation of BMAL:CLOCK1 transcriptional activity. These findings show that BMAL is highly involved in osteoclast and bone resorption molecular mechanism. |
| Low back pain, intervertebral disc degeneration | Cell and gene knockdown model; ex vivo intervertebral disc (IVD) explants from (PER2: luciferase (LUC) reporter mice) and human disc cells [111] | Bone formation is significantly increased in Per2 and Cry2 knockout mouse. Bone formation rate and osteoblast biological process are regulated by these genes in distinct pathways, Cry2 predominantly influencing mostly the osteoclastic and Per2 predominantly on the osteoblastic activity. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juliana, N.; Azmi, L.; Effendy, N.M.; Mohd Fahmi Teng, N.I.; Abu, I.F.; Abu Bakar, N.N.; Azmani, S.; Yazit, N.A.A.; Kadiman, S.; Das, S. Effect of Circadian Rhythm Disturbance on the Human Musculoskeletal System and the Importance of Nutritional Strategies. Nutrients 2023, 15, 734. https://doi.org/10.3390/nu15030734
Juliana N, Azmi L, Effendy NM, Mohd Fahmi Teng NI, Abu IF, Abu Bakar NN, Azmani S, Yazit NAA, Kadiman S, Das S. Effect of Circadian Rhythm Disturbance on the Human Musculoskeletal System and the Importance of Nutritional Strategies. Nutrients. 2023; 15(3):734. https://doi.org/10.3390/nu15030734
Chicago/Turabian StyleJuliana, Norsham, Liyana Azmi, Nadia Mohd Effendy, Nur Islami Mohd Fahmi Teng, Izuddin Fahmy Abu, Nur Nabilah Abu Bakar, Sahar Azmani, Noor Anisah Abu Yazit, Suhaini Kadiman, and Srijit Das. 2023. "Effect of Circadian Rhythm Disturbance on the Human Musculoskeletal System and the Importance of Nutritional Strategies" Nutrients 15, no. 3: 734. https://doi.org/10.3390/nu15030734
APA StyleJuliana, N., Azmi, L., Effendy, N. M., Mohd Fahmi Teng, N. I., Abu, I. F., Abu Bakar, N. N., Azmani, S., Yazit, N. A. A., Kadiman, S., & Das, S. (2023). Effect of Circadian Rhythm Disturbance on the Human Musculoskeletal System and the Importance of Nutritional Strategies. Nutrients, 15(3), 734. https://doi.org/10.3390/nu15030734







