The Chain-Mediating Effect of Obesity, Depressive Symptoms on the Association between Dietary Quality and Cardiovascular Disease Risk
Abstract
1. Introduction
2. Materials and Methods
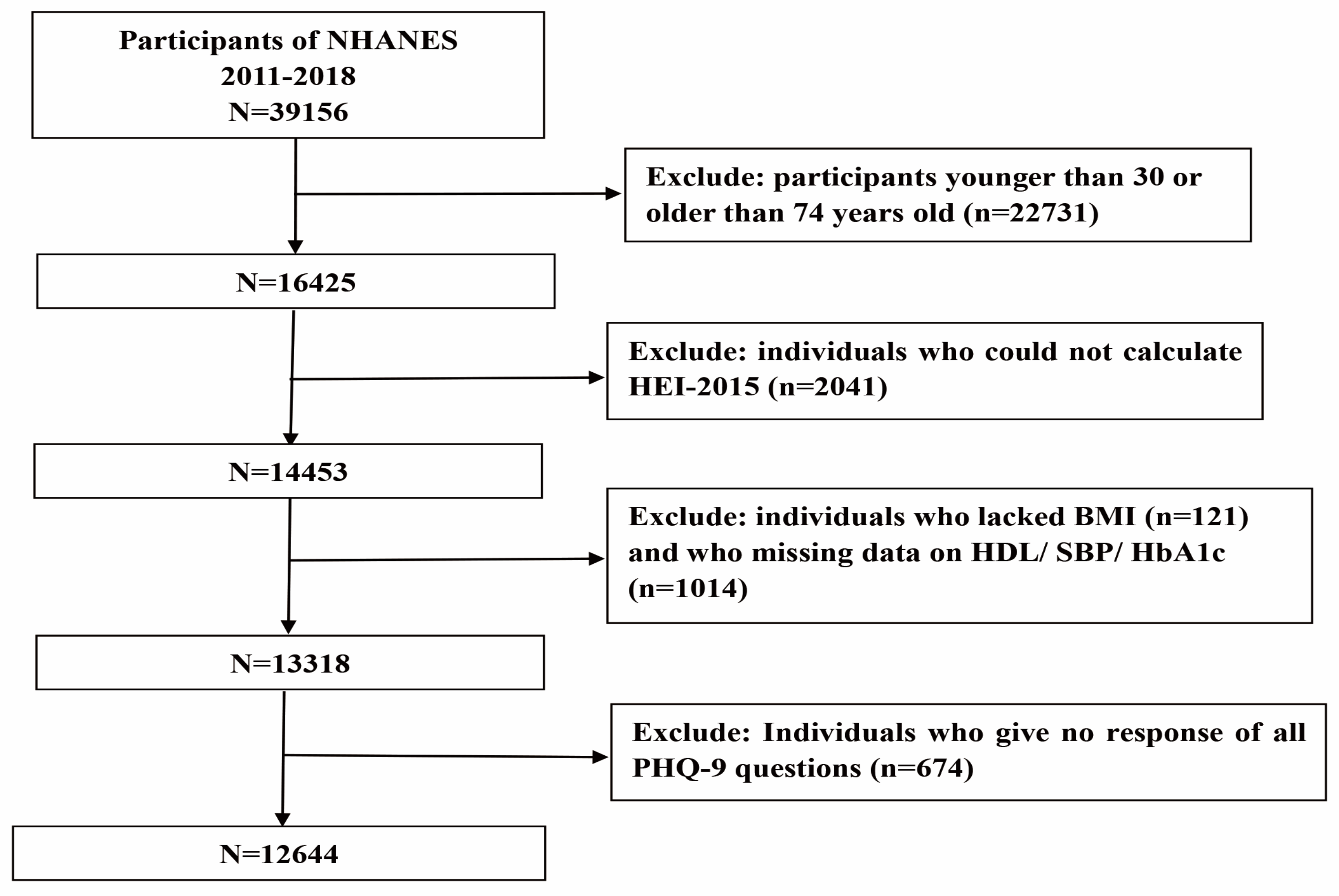
2.1. Data Source and Study Sample
2.2. Dietary Quality
2.3. Depressive Symptoms
2.4. Obesity
2.5. Cardiovascular Disease
2.6. Covariates
2.7. Sensitivity Analysis
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef] [PubMed]
- Virani, S.S.; Alonso, A.; Aparicio, H.J.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics—2021 Update: A Report from the American Heart Association. Circulation 2021, 143, e254–e743. [Google Scholar] [CrossRef] [PubMed]
- Stefanadis, C.; Antoniou, C.K.; Tsiachris, D.; Pietri, P. Coronary Atherosclerotic Vulnerable Plaque: Current Perspectives. J. Am. Heart Assoc. 2017, 6, e005543. [Google Scholar] [CrossRef] [PubMed]
- Hu, E.A.; Steffen, L.M.; Coresh, J.; Appel, L.J.; Rebholz, C.M. Adherence to the Healthy Eating Index-2015 and Other Dietary Patterns May Reduce Risk of Cardiovascular Disease, Cardiovascular Mortality, and All-Cause Mortality. J. Nutr. 2020, 150, 312–321. [Google Scholar] [CrossRef]
- Mozaffarian, D. Dietary and Policy Priorities for Cardiovascular Disease, Diabetes, and Obesity: A Comprehensive Review. Circulation 2016, 133, 187–225. [Google Scholar] [CrossRef]
- Yu, E.; Malik, V.S.; Hu, F.B. Cardiovascular Disease Prevention by Diet Modification: JACC Health Promotion Series. J. Am. Coll. Cardiol. 2018, 72, 914–926. [Google Scholar] [CrossRef]
- Rehm, C.D.; Penalvo, J.L.; Afshin, A.; Mozaffarian, D. Dietary Intake among US Adults, 1999–2012. JAMA—J. Am. Med. Assoc. 2016, 315, 2542–2553. [Google Scholar] [CrossRef]
- Wang, D.D.; Leung, C.W.; Li, Y.P.; Ding, E.L.; Chiuve, S.E.; Hu, F.B.; Willett, W.C. Trends in Dietary Quality among Adults in the United States, 1999 through 2010. JAMA Intern. Med. 2014, 174, 1587–1595. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef]
- Golovaty, I.; Tien, P.C.; Price, J.C.; Sheira, L.; Seligman, H.; Weiser, S.D. Food Insecurity May Be an Independent Risk Factor Associated with Nonalcoholic Fatty Liver Disease among Low-Income Adults in the United States. J. Nutr. 2020, 150, 91–98. [Google Scholar] [CrossRef]
- Brauer, P.; Royall, D.; Rodrigues, A. Use of the Healthy Eating Index in Intervention Studies for Cardiometabolic Risk Conditions: A Systematic Review. Adv. Nutr. 2021, 12, 1317–1331. [Google Scholar] [CrossRef]
- Xu, Z.; Steffen, L.M.; Selvin, E.; Rebholz, C.M. Diet quality, change in diet quality and risk of incident CVD and diabetes. Public Health Nutr. 2020, 23, 329–338. [Google Scholar] [CrossRef]
- Park, L.T.; Zarate, C.A., Jr. Depression in the Primary Care Setting. N. Engl. J. Med. 2019, 380, 559–568. [Google Scholar] [CrossRef]
- Health Centra, Anxiety & Depression Association of American. What is Depression? Available online: https://adaa.org/understanding-anxiety/depression#Types%20of%20Depression (accessed on 10 October 2022).
- Smith, K.B.; Smith, M.S. Obesity Statistics. Primary Care 2016, 43, 121. [Google Scholar] [CrossRef]
- Sanchez-Villegas, A.; Henriquez-Sanchez, P.; Ruiz-Canela, M.; Lahortiga, F.; Molero, P.; Toledo, E.; Martinez-Gonzalez, M.A. A longitudinal analysis of diet quality scores and the risk of incident depression in the SUN Project. BMC Med. 2015, 13, 197. [Google Scholar] [CrossRef]
- Akbaraly, T.N.; Sabia, S.; Shipley, M.J.; Batty, G.D.; Kivimaki, M. Adherence to healthy dietary guidelines and future depressive symptoms: Evidence for sex differentials in the Whitehall II study. Am. J. Clin. Nutr. 2013, 97, 419–427. [Google Scholar] [CrossRef]
- Asghari, G.; Mirmiran, P.; Yuzbashian, E.; Azizi, F. A systematic review of diet quality indices in relation to obesity. Br. J. Nutr. 2017, 117, 1055–1065. [Google Scholar] [CrossRef]
- Case, S.M.; Sawhney, M.; Stewart, J.C. Atypical depression and double depression predict new-onset cardiovascular disease in US adults. Depress. Anxiety 2018, 35, 10–17. [Google Scholar] [CrossRef]
- Park, S.J.; Lee, M.G.; Jo, M.; Kim, G.; Park, S. Joint effect of depression and health behaviors or conditions on incident cardiovascular diseases: A Korean population-based cohort study. J. Affect. Disord. 2020, 276, 616–622. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Li, X.; Chan, E.; Luo, H.; Chan, S.S.M.; Wong, G.H.Y.; Wong, I.C.K.; Lum, T.Y.S. Depression duration and risk of incident cardiovascular disease: A population-based six-year cohort study. J. Affect. Disord. 2022, 305, 188–195. [Google Scholar] [CrossRef]
- Harris, T.B.; Ballardbarbasch, R.; Madans, J.; Makuc, D.M.; Feldman, J.J. Overweight, Weight-Loss, and Risk of Coronary Heart-Disease in Older Women—The Nhanes-I Epidemiologic Follow-up-Study. Am. J. Epidemiol. 1993, 137, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Dhana, K.; Berghout, M.A.; Peeters, A.; Ikram, M.A.; Tiemeier, H.; Hofman, A.; Nusselder, W.; Kavousi, M.; Franco, O.H. Obesity in older adults and life expectancy with and without cardiovascular disease. Int. J. Obes. 2016, 40, 1535–1540. [Google Scholar] [CrossRef] [PubMed]
- Oktay, A.A.; Lauie, C.J.; Kokkinos, P.F.; Parto, P.; Pandey, A.; Ventura, H.O. The Interaction of Cardiorespiratory Fitness with Obesity and the Obesity Paradox in Cardiovascular Disease. Prog. Cardiovasc. Dis. 2017, 60, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Faith, M.S.; Butryn, M.; Wadden, T.A.; Fabricatore, A.; Nguyen, A.M.; Heymsfield, S.B. Evidence for prospective associations among depression and obesity in population-based studies. Obes. Rev. 2011, 12, e438–e453. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Visualizing and Interpreting HEI Scores. Available online: https://epi.grants.cancer.gov/hei/interpret-visualize-hei-scores.html. (accessed on 12 October 2022).
- Manea, L.; Gilbody, S.; McMillan, D. Optimal cut-off score for diagnosing depression with the Patient Health Questionnaire (PHQ-9): A meta-analysis. Can. Med. Assoc. J. 2012, 184, E191–E196. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, R.B.; Vasan, R.S.; Pencina, M.J.; Wolf, P.A.; Cobain, M.; Massaro, J.M.; Kannel, W.B. General cardiovascular risk profile for use in primary care: The Framingham heart study. Circulation 2008, 118, E86. [Google Scholar] [CrossRef]
- Parikh, N.S.; Chatterjee, A.; Diaz, I.; Merkler, A.E.; Murthy, S.B.; Iadecola, C.; Navi, B.B.; Kamel, H. Trends in Active Cigarette Smoking among Stroke Survivors in the United States, 1999 to 2018. Stroke 2020, 51, 1656–1661. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. NCfHS National Health and Nutrition Examination Survey. 2011–2012 Data Documentation, Codebook, and Frequencies Cholesterol—HDL (HDL_G). Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2011-2012/labmethods/hdl_g_met_hdl.pdf (accessed on 10 October 2022).
- Centers for Disease Control and Prevention. NCfHS National Health and Nutrition Examination Survey. Measurement Method of Nhanes Serum Total Cholesterol. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2011-2012/labmethods/tchol_g_met.pdf (accessed on 10 October 2022).
- Xu, Z.; Du, J.; Wang, J.J.; Jiang, C.X.; Ren, Y. Satellite Image Prediction Relying on GAN and LSTM Neural Networks. In Proceedings of the IEEE International Conference on Communications (IEEE ICC), Shanghai, China, 20–24 May 2019. [Google Scholar]
- Harmon, B.E.; Boushey, C.J.; Shvetsov, Y.B.; Ettienne, R.; Reedy, J.; Wilkens, L.R.; Le Marchand, L.; Henderson, B.E.; Kolonel, L.N. Associations of key diet-quality indexes with mortality in the Multiethnic Cohort: The Dietary Patterns Methods Project. Am. J. Clin. Nutr. 2015, 101, 587–597. [Google Scholar] [CrossRef]
- Liese, A.D.; Krebs-Smith, S.M.; Subar, A.F.; George, S.M.; Harmon, B.E.; Neuhouser, M.L.; Boushey, C.J.; Schap, T.E.; Reedy, J. The Dietary Patterns Methods Project: Synthesis of Findings across Cohorts and Relevance to Dietary Guidance. J. Nutr. 2015, 145, 393–402. [Google Scholar] [CrossRef]
- Alissa, E.M.; Ferns, G.A. Dietary fruits and vegetables and cardiovascular diseases risk. Crit. Rev. Food Sci. Nutr. 2017, 57, 1950–1962. [Google Scholar] [CrossRef]
- Zhuang, P.; Zhang, Y.; He, W.; Chen, X.Q.; Chen, J.N.; He, L.L.; Mao, L.; Wu, F.; Jiao, J.J. Dietary Fats in Relation to Total and Cause-Specific Mortality in a Prospective Cohort of 521,120 Individuals with 16 Years of Follow-up. Circ. Res. 2019, 124, 757–768. [Google Scholar] [CrossRef]
- Firth, J.; Marx, W.; Dash, S.; Carney, R.; Teasdale, S.B.; Solmi, M.; Stubbs, B.; Schuch, F.B.; Carvalho, A.F.; Jacka, F.; et al. The Effects of Dietary Improvement on Symptoms of Depression and Anxiety: A Meta-Analysis of Randomized Controlled Trials. Psychosom. Med. 2019, 81, 265–280. [Google Scholar] [CrossRef]
- Ginty, A.T.; Carroll, D.; Roseboom, T.J.; Phillips, A.C.; de Rooij, S.R. Depression and anxiety are associated with a diagnosis of hypertension 5 years later in a cohort of late middle-aged men and women. J. Hum. Hypertens. 2013, 27, 187–190. [Google Scholar] [CrossRef]
- Filippini, T.; Malavolti, M.; Whelton, P.K.; Vinceti, M. Sodium Intake and Risk of Hypertension: A Systematic Review and Dose-Response Meta-analysis of Observational Cohort Studies. Curr. Hypertens. Rep. 2022, 24, 133–144. [Google Scholar] [CrossRef]
- Dash, S.; Clarke, G.; Berk, M.; Jacka, F.N. The gut microbiome and diet in psychiatry: Focus on depression. Curr. Opin. Psychiatry 2015, 28, 1–6. [Google Scholar] [CrossRef]
- Kennedy, E. Dietary diversity, diet quality, and body weight regulation. Nutr. Rev. 2004, 62, S78–S81. [Google Scholar] [CrossRef]
- Bray, G.A.; Popkin, B.M. Dietary Sugar and Body Weight: Have We Reached a Crisis in the Epidemic of Obesity and Diabetes? Diabetes Care 2014, 37, 950–956. [Google Scholar] [CrossRef]
- Rogers, P.J.; Hogenkamp, P.S.; de Graaf, C.; Higgs, S.; Lluch, A.; Ness, A.R.; Penfold, C.; Perry, R.; Putz, P.; Yeomans, M.R.; et al. Does low-energy sweetener consumption affect energy intake and body weight? A systematic review, including meta-analyses, of the evidence from human and animal studies. Int. J. Obes. 2016, 40, 381–394. [Google Scholar] [CrossRef]
- Shong, K.E.; Oh, C.M.; Namkung, J.; Park, S.; Kim, H. Serotonin Regulates De Novo Lipogenesis in Adipose Tissues through Serotonin Receptor 2A. Endocrinol. Metab. 2020, 35, 470–479. [Google Scholar] [CrossRef]
- Comai, S.; Bertazzo, A.; Bailoni, L.; Zancato, M.; Costa, C.V.L.; Allegri, G. Protein and non-protein (free and protein-bound) tryptophan in legume seeds. Food Chem. 2007, 103, 657–661. [Google Scholar] [CrossRef]
- Patrick, R.P.; Ames, B.N. Vitamin D and the omega-3 fatty acids control serotonin synthesis and action, part 2: Relevance for ADHD, bipolar disorder, schizophrenia, and impulsive behavior. FASEB J. 2015, 29, 2207–2222. [Google Scholar] [CrossRef] [PubMed]
- Saghafian, F.; Malmir, H.; Saneei, P.; Milajerdi, A.; Larijani, B.; Esmaillzadeh, A. Fruit and vegetable consumption and risk of depression: Accumulative evidence from an updated systematic review and meta-analysis of epidemiological studies. Br. J. Nutr. 2018, 119, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Laudisio, D.; Frias-Toral, E.; Barrea, L.; Muscogiuri, G.; Savastano, S.; Colao, A.; on behalf of the Obesity Programs of Nutrition, Education, Research and Assessment (OPERA) Group. Anti-Inflammatory Nutrients and Obesity-Associated Metabolic-Inflammation: State of the Art and Future Direction. Nutrients 2022, 14, 1137. [Google Scholar] [CrossRef] [PubMed]
- Preiss, K.; Brennan, L.; Clarke, D. A systematic review of variables associated with the relationship between obesity and depression. Obes. Rev. 2013, 14, 906–918. [Google Scholar] [CrossRef]
- Bradley, S.M.; Rumsfeld, J.S. Depression and cardiovascular disease. Trends Cardiovasc. Med. 2015, 25, 614–622. [Google Scholar] [CrossRef]
- Adair, T.; Lopez, A.D. The role of overweight and obesity in adverse cardiovascular disease mortality trends: An analysis of multiple cause of death data from Australia and the USA. BMC Med. 2020, 18, 199. [Google Scholar] [CrossRef]
- McConnell, S.; Jacka, F.N.; Williams, L.J.; Dodd, S.; Berk, M. The relationship between depression and cardiovascular disease. Int. J. Psychiatry Clin. Pract. 2005, 9, 157–167. [Google Scholar] [CrossRef]
- Hamieh, N.; Meneton, P.; Wiernik, E.; Limosin, F.; Zins, M.; Goldberg, M.; Melchior, M.; Lemogne, C. Depression, treatable cardiovascular risk factors and incident cardiac events in the Gazel cohort. Int. J. Cardiol. 2019, 284, 90–95. [Google Scholar] [CrossRef]
- Iwamoto, S.J.; Abushamat, L.A.; Zaman, A.; Millard, A.J.; Cornier, M.A. Obesity Management in Cardiometabolic Disease: State of the Art. Curr. Atheroscler. Rep. 2021, 23, 59. [Google Scholar] [CrossRef]

| Characteristics | All Participants | Quartile of HEI-2015 | ||||
|---|---|---|---|---|---|---|
| Q1(0–44.12) | Q2(44.12–53.58) | Q3(53.58–63.50) | Q4(63.50–100) | p Value | ||
| No. of participants | 12,644 | 3161 | 3161 | 3161 | 3161 | |
| Age (mean±SD) a | 51.46 ± 12.61 | 49.25 ± 12.56 | 50.99 ± 12.51 | 51.72 ± 12.52 | 53.86 ± 12.44 | 0.961 |
| Gender (%) b | <0.001 | |||||
| Men | 6219(49.2) | 1752(55.6) | 1597(50.5) | 1505(47.6) | 1365(43.2) | |
| Women | 6425(50.8) | 1409(44.6) | 1564(49.5) | 1656(52.4) | 1796(56.8) | |
| Race/ethnicity (%) b | <0.001 | |||||
| Mexican American | 1813(14.3) | 426(13.5) | 478(15.1) | 494(15.6) | 415(13.1) | |
| Other Hispanic | 1405(11.1) | 273(8.6) | 311(9.8) | 371(11.7) | 450(14.2) | |
| Non-Hispanic White | 4683(37.0) | 1372(43.4) | 1160(36.7) | 1115(35.3) | 1036(32.8) | |
| Non-Hispanic Black | 2858(22.6) | 793(25.1) | 793(25.1) | 685(21.7) | 587(18.6) | |
| Other Race | 1885(14.9) | 297(9.4) | 419(13.3) | 496(15.7) | 673(21.3) | |
| Degree of education (%) b | <0.001 | |||||
| Less than high school | 2668(21.1) | 768(24.3) | 706(22.3) | 664(21.0) | 530(16.8) | |
| High school | 2772(21.9) | 848(26.8) | 772(24.4) | 653(20.7) | 499(15.8) | |
| More than high school | 7200(57.0) | 1544(48.9) | 1682(53.2) | 1844(58.3) | 2130(67.4) | |
| Ratio of family income to poverty (PIR) (mean ± SD) c | 2.62 ± 1.66 | 2.22 ± 1.53 | 2.48 ± 1.63 | 2.69 ± 1.66 | 3.08 ± 1.69 | <0.001 |
| Marital status (%) b | <0.001 | |||||
| Married | 8176(64.7) | 1907(60.4) | 1995(63.2) | 2096(66.3) | 2178(68.9) | |
| Other | 4462(35.3) | 1252(39.6) | 1163(36.8) | 1065(33.7) | 983(31.1) | |
| Smoke (%) b | <0.001 | |||||
| Nonsmokers | 6915(54.7) | 1419(44.9) | 1629(51.6) | 1800(56.9) | 2067(65.5) | |
| Former smoker | 2740(21.7) | 554(17.5) | 684(21.7) | 740(23.4) | 762(24.1) | |
| Current smoker | 2979(23.6) | 1185(37.5) | 844(26.7) | 621(19.6) | 329(10.4) | |
| Work physical activity (%) b | <0.001 | |||||
| Vigorous activity | 2733(21.6) | 884(28.0) | 736(23.3) | 598(18.9) | 515(16.3) | |
| Moderate activity | 2644(20.9) | 659(20.8) | 672(21.3) | 675(21.4) | 638(20.2) | |
| Low activity | 7267(57.5) | 1618(51.2) | 1753(55.5) | 1888(59.7) | 2008(63.5) | |
| Recreational physical activity (%) b | <0.001 | |||||
| Vigorous activity | 2723(21.5) | 486(15.4) | 584(18.5) | 715(22.6) | 938(29.7) | |
| Moderate activity | 3416(27.0) | 716(22.7) | 782(24.7) | 870(27.5) | 1048(33.2) | |
| Low activity | 6505(51.4) | 1959(62.0) | 1795(56.8) | 1576(49.9) | 1175(37.2) | |
| Obesity (%) b | <0.001 | |||||
| No | 7219(57.1) | 1578(49.9) | 1746(55.2) | 1819(57.5) | 2076(65.7) | |
| Yes | 5425(42.9) | 1583(50.1) | 1415(44.8) | 1342(42.5) | 1085(34.3) | |
| Diabetes (%) b | <0.001 | |||||
| No | 9971(78.9) | 2484(78.6) | 2507(79.3) | 2457(77.7) | 2523(79.8) | |
| Yes | 2673(21.1) | 677(21.4) | 654(20.7) | 704(22.3) | 638(20.2) | |
| Depressive symptoms (%) b | <0.001 | |||||
| No | 11447(90.5) | 2757(87.2) | 2836(89.7) | 2883(91.2) | 2971(94.0) | |
| Yes | 1197(9.5) | 404(12.8) | 325(10.3) | 278(8.8) | 190(6.0) | |
| Cardiovascular risk (%) b | <0.001 | |||||
| Low | 9727(76.9) | 2382(75.4) | 2409(76.2) | 2445(77.3) | 2491(78.8) | |
| High | 2917(23.1) | 779(24.6) | 752(23.8) | 716(22.7) | 670(21.2) | |
| Total cholesterol (%) b | 0.087 | |||||
| No | 7210(57.0) | 1864(59.0) | 1776(56.2) | 1786(56.5) | 1784(56.4) | |
| Yes | 5434(43.0) | 1297(41.0) | 1385(43.8) | 1375(43.5) | 1377(43.6) | |
| HDL cholesterol (%) b | <0.001 | |||||
| No | 10166(80.4) | 2345(74.2) | 2542(80.4) | 2585(81.8) | 2694(85.2) | |
| Yes | 2478(19.6) | 816(25.8) | 619(19.6) | 576(18.2) | 467(14.8) | |
| Hypertension (%) b | <0.001 | |||||
| No | 5135(40.6) | 1239(39.2) | 1221(38.6) | 1301(41.2) | 1374(76.9) | |
| Yes | 7509(59.4) | 1922(60.8) | 1940(61.4) | 1860(58.8) | 1787(56.5) | |
| Crude a | Model 1 a | Model 2 a | |||||||
|---|---|---|---|---|---|---|---|---|---|
| t | p Value | OR b (95% CI) | t | p Value | OR (95% CI) | t | p Value | OR (95% CI) | |
| HEI-2015 | |||||||||
| Q1(0–44.12) | Ref. | Ref. | Ref. | ||||||
| Q2(44.12–53.58) | −0.46 | 0.648 | 0.963(0.815, 1.137) | −2.49 | 0.015 | 0.774(0.630, 0.950) | −1.97 | 0.054 | 0.810(0.653, 1.003) |
| Q3(53.58–63.50) | −2.83 | 0.006 | 0.769(0.639, 0.926) | −4.29 | <0.001 | 0.591(0.462, 0.755) | −3.01 | 0.004 | 0.690(0.539, 0.882) |
| Q4(63.50–100) | −2.71 | 0.009 | 0.783(0.654, 0.938) | −6.92 | <0.001 | 0.456(0.363, 0.572) | −3.96 | <0.001 | 0.632(0.501, 0.797) |
| Depressive symptoms | 2.05 | 0.044 | 1.230(1.005, 1.506) | 5.32 | <0.001 | 1.925(1.505, 2.642) | 2.28 | 0.026 | 1.369(1.039, 1.803) |
| Obesity | 6.94 | <0.001 | 1.590(1.391, 1.816) | 10.02 | <0.001 | 2.035(1.766, 2.345) | 8.16 | <0.001 | 1.914(1.632, 2.244) |
| Crude a | Model 1 a | Model 2 a | |||||||
|---|---|---|---|---|---|---|---|---|---|
| t | p Value | OR b (95%CI) | t | p Value | OR (95%CI) | t | p Value | OR (95%CI) | |
| Depressive symptoms HEI−2015 | |||||||||
| Q1(0–44.12) | Ref. | Ref. | Ref. | ||||||
| Q2(44.12–53.58) | −2.07 | 0.043 | 0.802(0.648, 0.993) | −2.30 | 0.025 | 0.780(0.629, 0.968) | −1.08 | 0.285 | 0.884(0.703, 1.111) |
| Q3(53.58–63.50) | −4.55 | <0.001 | 0.555(0.429, 0.719) | −4.89 | <0.001 | 0.521(0.399, 0.680) | −2.67 | 0.009 | 0.682(0.512, 0.908) |
| Q4(63.50–100) | −7.25 | <0.001 | 0.392(0.303, 0.508) | −7.53 | <0.001 | 0.361(0.276, 0.473) | −4.18 | <0.001 | 0.553(0.417, 0.735) |
| Obesity HEI−2015 | |||||||||
| Q1(0–44.12) | Ref. | Ref. | Ref. | ||||||
| Q2(44.12–53.58) | −3.11 | 0.003 | 0.788(0.676, 0.918) | −3.32 | 0.002 | 0.774(0.663, 0.903) | −2.79 | 0.007 | 0.806(0.691, 0.941) |
| Q3(53.58–63.50) | −7.40 | <0.001 | 0.618(0.542, 0.704) | −7.51 | <0.001 | 0.600(0.523, 0.687) | −5.87 | <0.001 | 0.676(0.591, 0.772) |
| Q4(63.50–100) | −12.44 | <0.001 | 0.432(0.377, 0.494) | −12.62 | <0.001 | 0.413(0.359, 0.475) | −8.12 | <0.001 | 0.519(0.442, 0.610) |
| Cardiovascular Disease a | ||||||
|---|---|---|---|---|---|---|
| Crude Model b | Model 1 c | Model 2 d | ||||
| OR(95% CI) | p Value | OR(95% CI) | p Value | OR(95% CI) | p Value | |
| Total vegetables | 0.952(0.908, 0.998) | 0.043 | 0.903(0.854, 0.954) | <0.001 | 0.975(0.916, 1.037) | 0.412 |
| Greens and beans | 0.938(0.912, 0.965) | <0.001 | 0.933(0.900, 0.966) | <0.001 | 0.963(0.929, 0.999) | 0.042 |
| Total fruits | 0.981(0.950, 1.014) | 0.245 | 0.923(0.887, 0.962) | <0.001 | 0.959(0.918, 1.001) | 0.058 |
| Whole fruits | 0.988(0.959, 1.018) | 0.426 | 0.924(0.888, 0.961) | <0.001 | 0.964(0.921, 1.009) | 0.116 |
| Whole grains | 1.015(0.998, 1.032) | 0.087 | 0.971(0.949, 0.994) | 0.014 | 0.991(0.968, 1.014) | 0.416 |
| Dairy | 0.964(0.942, 0.988) | 0.003 | 0.981(0.951, 1.012) | <0.216 | 1.006(0.975, 1.039) | 0.691 |
| Total protein foods | 1.075(1.009, 1.146) | 0.025 | 0.970(0.896, 1.051) | 0.452 | 0.997(0.917, 1.084) | 0.950 |
| Seafood and plant proteins | 0.957(0.931, 0.985) | 0.003 | 0.909(0.878, 0.941) | <0.001 | 0.956(0.923, 0.989) | 0.011 |
| Fatty acid | 0.975(0.958, 0.992) | 0.005 | 0.960(0.937, 0.984) | 0.001 | 0.964(0.942, 0.986) | 0.002 |
| Sodium e | 0.978(0.959, 0.997) | 0.022 | 0.967(0.946, 0.988) | 0.003 | 0.962(0.939, 0.986) | 0.003 |
| Refined grains e | 0.998(0.983, 1.014) | 0.845 | 0.958(0.939, 0.977) | <0.001 | 0.974(0.952, 0.997) | 0.026 |
| Saturated fats e | 0.969(0.953, 0.986) | <0.001 | 0.978(0.954, 1.002) | 0.073 | 0.972(0.951, 0.994) | 0.014 |
| Added sugars e | 1.016(0.991, 1.040) | 0.208 | 0.961(0.933, 0.990) | 0.010 | 0.990(0.957, 1.023) | 0.521 |
| Model Pathways | Mediating Effect | |
|---|---|---|
| β (95%CI) | Proportion Mediated (%) | |
| Total effect | −0.0014(−0.0019, −0.0009) *** | 100 |
| Direct effect | −0.0012(−0.0017, −0.0008) *** | 88.74 |
| Indirect effect via obesity | −0.00013(−0.00020, −0.00006) *** | 9.03 |
| Indirect effect via depressive symptoms | −0.00003(−0.00005, −0.00001) * | 2.23 |
| Indirect effect via obesity and depressive symptoms | −0.000003(−0.000006, −0.0000009) ** | 0.25 |
| Model Pathways | Mediating Effect | |
|---|---|---|
| β (95%CI) | Proportion Mediated (%) | |
| Total effect | −0.0014(−0.0018, −0.0010) *** | 100 |
| Direct effect | −0.0013(−0.0016, −0.0009) *** | 89.64 |
| Indirect effect via obesity | −0.00012(−0.00016, −0.00007) *** | 8.40 |
| Indirect effect via depressive symptoms | −0.00003(−0.00005, −0.00001) * | 1.96 |
| Indirect effect via obesity and depressive symptoms | −0.000004(−0.000008, −0.0000008) ** | 0.30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; E, L.; Lu, Z.; Yu, Y.; Yang, X.; Chen, Y.; Jiang, X. The Chain-Mediating Effect of Obesity, Depressive Symptoms on the Association between Dietary Quality and Cardiovascular Disease Risk. Nutrients 2023, 15, 629. https://doi.org/10.3390/nu15030629
Zhang S, E L, Lu Z, Yu Y, Yang X, Chen Y, Jiang X. The Chain-Mediating Effect of Obesity, Depressive Symptoms on the Association between Dietary Quality and Cardiovascular Disease Risk. Nutrients. 2023; 15(3):629. https://doi.org/10.3390/nu15030629
Chicago/Turabian StyleZhang, Shuai, Limei E, Zhonghai Lu, Yingying Yu, Xuebin Yang, Yao Chen, and Xiubo Jiang. 2023. "The Chain-Mediating Effect of Obesity, Depressive Symptoms on the Association between Dietary Quality and Cardiovascular Disease Risk" Nutrients 15, no. 3: 629. https://doi.org/10.3390/nu15030629
APA StyleZhang, S., E, L., Lu, Z., Yu, Y., Yang, X., Chen, Y., & Jiang, X. (2023). The Chain-Mediating Effect of Obesity, Depressive Symptoms on the Association between Dietary Quality and Cardiovascular Disease Risk. Nutrients, 15(3), 629. https://doi.org/10.3390/nu15030629






