Effects of Nut Consumption on Blood Lipids and Lipoproteins: A Comprehensive Literature Update
Abstract
1. Introduction
2. Effects of Nuts on Blood Lipids
2.1. Dose-Response Effects of Nuts on Blood Lipids
2.2. Subgroup Analyses: Effects of Nuts on Blood Lipids
2.2.1. Sex
2.2.2. Age
2.2.3. BMI
2.2.4. Baseline Lipids/Lipoprotein Concentrations
2.2.5. Health Status
2.3. Effects of Nut Processing on Blood Lipid Profile
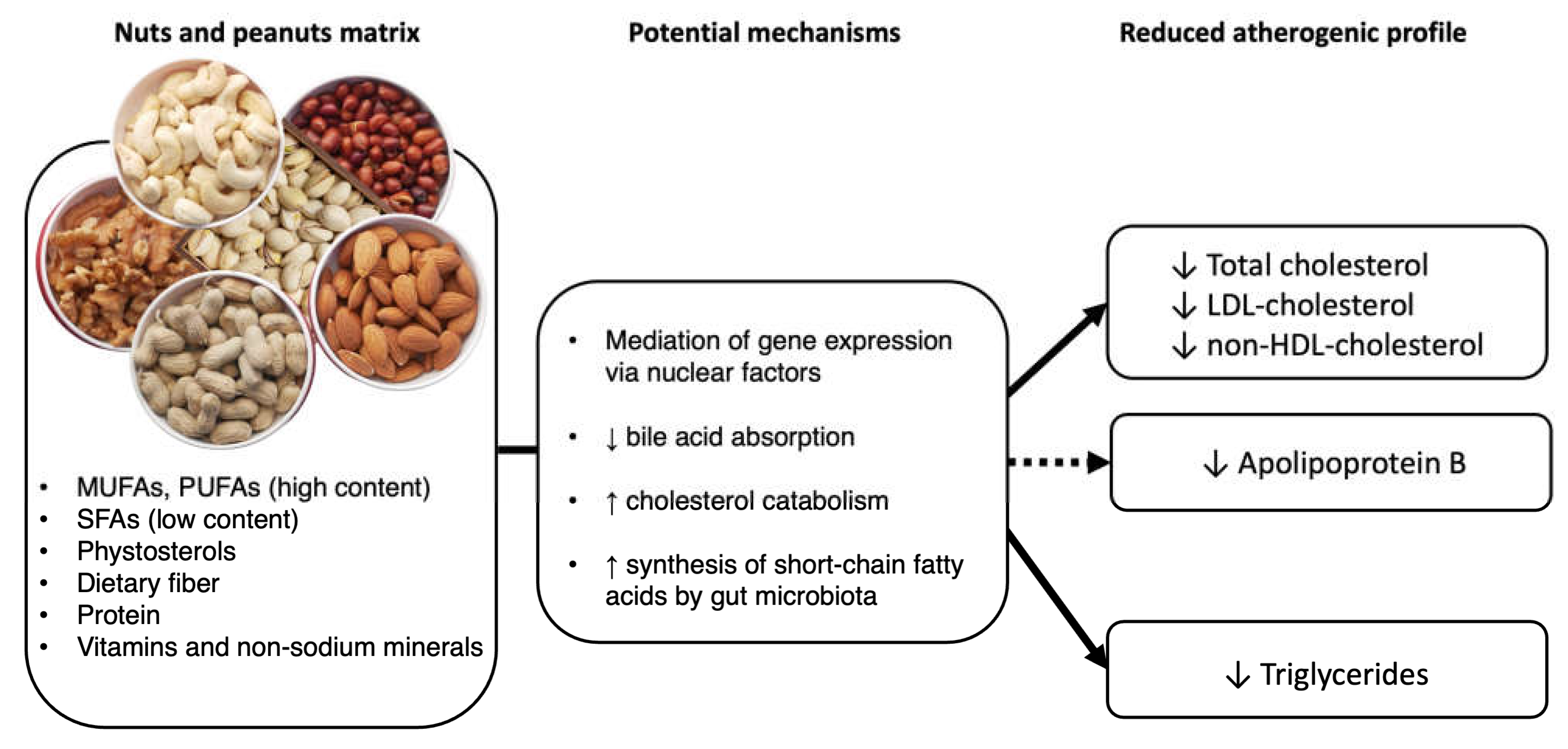
3. Proposed Mechanisms of Action of Cholesterol-Lowering by Nuts
3.1. Emerging Evidence of the Effects of Nuts on Lipoprotein Particle Size
3.2. Effects of Nut Consumption on Adiposity
4. Future Directions for Research on Nut Consumption and Blood Lipids
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, Regional, and National Age-Sex-Specific Mortality for 282 Causes of Death in 195 Countries and Territories, 1980–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 139, E1082–E1143. [Google Scholar] [CrossRef] [PubMed]
- Gakidou, E.; Afshin, A.; Abajobir, A.A.; Abate, K.H.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abdulle, A.M.; Abera, S.F.; Aboyans, V.; et al. Global, Regional, and National Comparative Risk Assessment of 84 Behavioural, Environmental and Occupational, and Metabolic Risks or Clusters of Risks, 1990–2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.L.; Mokdad, A.H.; Ballestros, K.; Echko, M.; Glenn, S.; Olsen, H.E.; Mullany, E.; Lee, A.; Khan, A.R.; Ahmadi, A.; et al. The State of US Health, 1990–2016: Burden of Diseases, Injuries, and Risk Factors among US States. JAMA J. Am. Med. Assoc. 2018, 319, 1444–1472. [Google Scholar] [CrossRef]
- Micha, R.; Peñalvo, J.L.; Cudhea, F.; Imamura, F.; Rehm, C.D.; Mozaffarian, D. Association between Dietary Factors and Mortality from Heart Disease, Stroke, and Type 2 Diabetes in the United States. JAMA J. Am. Med. Assoc. 2017, 317, 912–924. [Google Scholar] [CrossRef]
- Ros, E.; Tapsell, L.C.; Sabate, J. Nuts and Berries for Heart Health. Curr. Atheroscler. Rep. 2010, 12, 397–406. [Google Scholar] [CrossRef]
- Li, L.; Tsao, R.; Yang, R.; Kramer, J.K.G.; Hernandez, M. Fatty Acid Profiles, Tocopherol Contents, and Antioxidant Activities of Heartnut (Juglans Ailanthifolia Var. Cordiformis) and Persian Walnut (Juglans Regia L.). J. Agric. Food Chem. 2007, 55, 1164–1169. [Google Scholar] [CrossRef]
- Ros, E.; Hu, F.B. Consumption of Plant Seeds and Cardiovascular Health: Epidemiological and Clinical Trial Evidence. Circulation 2013, 128, 553–565. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. New Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Fito, M.; Guxens, M.; Corella, D.; Saez, G.; Estruch, R.; de la Torre, R.; Frances, F.; Cabezas, C.; Lopez-Sabater Mdel, C.; Marrugat, J.; et al. Effect of a Traditional Mediterranean Diet on Lipoprotein Oxidation: A Randomized Controlled Trial. Arch. Intern. Med. 2007, 167, 1195–1203. [Google Scholar] [CrossRef]
- Xia, J.; Yu, J.; Xu, D.; Yang, C.; Xia, H.; Sun, G. The Effects of Peanuts and Tree Nuts on Lipid Profile in Type 2 Diabetic Patients: A Systematic Review and Meta-Analysis of Randomized, Controlled-Feeding Clinical Studies. Front. Nutr. 2021, 8, 765571. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J. Nut Consumption and Blood Lipid Levels. Arch. Intern. Med. 2010, 170, 821. [Google Scholar] [CrossRef] [PubMed]
- Phung, O.J.; Makanji, S.S.; White, C.M.; Coleman, C.I. Almonds Have a Neutral Effect on Serum Lipid Profiles: A Meta-Analysis of Randomized Trials. J. Am. Diet Assoc. 2009, 109, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Parilli-Moser, I.; Hurtado-Barroso, S.; Guasch-Ferré, M.; Lamuela-Raventós, R.M. Effect of Peanut Consumption on Cardiovascular Risk Factors: A Randomized Clinical Trial and Meta-Analysis. Front. Nutr. 2022, 9, 853378. [Google Scholar] [CrossRef] [PubMed]
- Moosavian, S.P.; Rahimlou, M.; Rezaei Kelishadi, M.; Moradi, S.; Jalili, C. Effects of Almond on Cardiometabolic Outcomes in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Phytother. Res. 2022, 36, 1839–1853. [Google Scholar] [CrossRef]
- Blanco Mejia, S.; Kendall, C.W.C.; Viguiliouk, E.; Augustin, L.S.; Ha, V.; Cozma, A.I.; Mirrahimi, A.; Maroleanu, A.; Chiavaroli, L.; Leiter, L.A.; et al. Effect of Tree Nuts on Metabolic Syndrome Criteria: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. BMJ Open 2014, 4, e004660. [Google Scholar] [CrossRef]
- Mateș, L.; Popa, D.-S.; Rusu, M.E.; Fizeșan, I.; Leucuța, D. Walnut Intake Interventions Targeting Biomarkers of Metabolic Syndrome and Inflammation in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Antioxidants 2022, 11, 1412. [Google Scholar] [CrossRef]
- Liu, K.; Hui, S.; Wang, B.; Kaliannan, K.; Guo, X.; Liang, L. Comparative Effects of Different Types of Tree Nut Consumption on Blood Lipids: A Network Meta-Analysis of Clinical Trials. Am. J. Clin. Nutr. 2020, 111, 219–227. [Google Scholar] [CrossRef]
- Lee-Bravatti, M.A.; Wang, J.; Avendano, E.E.; King, L.; Johnson, E.J.; Raman, G. Almond Consumption and Risk Factors for Cardiovascular Disease: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2019, 10, 1076–1088. [Google Scholar] [CrossRef]
- Jalali, M.; Karamizadeh, M.; Ferns, G.A.; Zare, M.; Moosavian, S.P.; Akbarzadeh, M. The Effects of Cashew Nut Intake on Lipid Profile and Blood Pressure: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Complement Ther. Med. 2020, 50, 102387. [Google Scholar] [CrossRef]
- Eslami, O.; Khorramrouz, F.; Sohouli, M.; Bagheri, N.; Shidfar, F.; Fernandez, M.L. Effect of Nuts on Components of Metabolic Syndrome in Healthy Adults with Overweight/Obesity: A Systematic Review and Meta-Analysis. Nutr. Metab. Cardiovasc. Dis. 2022, 32, 2459–2469. [Google Scholar] [CrossRef]
- del Gobbo, L.C.; Falk, M.C.; Feldman, R.; Lewis, K.; Mozaffarian, D. Effects of Tree Nuts on Blood Lipids, Apolipoproteins, and Blood Pressure: Systematic Review, Meta-Analysis, and Dose-Response of 61 Controlled Intervention Trials. Am. J. Clin. Nutr. 2015, 102, 1347–1356. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Li, J.; Hu, F.B.; Salas-Salvadó, J.; Tobias, D.K. Effects of Walnut Consumption on Blood Lipids and Other Cardiovascular Risk Factors: An Updated Meta-Analysis and Systematic Review of Controlled Trials. Am. J. Clin. Nutr. 2018, 108, 174–187. [Google Scholar] [CrossRef]
- Banel, D.K.; Hu, F.B. Effects of Walnut Consumption on Blood Lipids and Other Cardiovascular Risk Factors: A Meta-Analysis and Systematic Review. Am. J. Clin. Nutr. 2009, 90, 56–63. [Google Scholar] [CrossRef]
- Asbaghi, O.; Moodi, V.; Hadi, A.; Eslampour, E.; Shirinbakhshmasoleh, M.; Ghaedi, E.; Miraghajani, M. The Effect of Almond Intake on Lipid Profile: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Food Funct. 2021, 12, 1882–1896. [Google Scholar] [CrossRef]
- Arabi, S.M.; Bahrami, L.S.; Milkarizi, N.; Nematy, M.; Kalmykov, V.; Sahebkar, A. Impact of Walnut Consumption on Cardio Metabolic and Anthropometric Parameters in Metabolic Syndrome Patients: GRADE-Assessed Systematic Review and Dose-Response Meta-Analysis of Data from Randomized Controlled Trials. Pharmacol. Res. 2022, 178, 106190. [Google Scholar] [CrossRef]
- Hadi, A.; Asbaghi, O.; Kazemi, M.; Haghighian, H.K.; Pantovic, A.; Ghaedi, E.; Abolhasani Zadeh, F. Consumption of Pistachio Nuts Positively Affects Lipid Profiles: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Crit. Rev. Food Sci. Nutr. 2021, 21, 1–14. [Google Scholar] [CrossRef]
- Morvaridzadeh, M.; Sepidarkish, M.; Farsi, F.; Akbari, A.; Mostafai, R.; Omidi, A.; Potter, E.; Heshmati, J. Effect of Cashew Nut on Lipid Profile: A Systematic Review and Meta-Analysis. Complement Med. Res. 2020, 27, 348–356. [Google Scholar] [CrossRef]
- Wang, P.; Sheng, Y.; Samadi, M. Effects of Almond Consumption on Lipid Profile in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Arch. Physiol. Biochem. 2021, 126, 1987477. [Google Scholar] [CrossRef]
- The Effects of Lowering LDL Cholesterol with Statin Therapy in People at Low Risk of Vascular Disease: Meta-Analysis of Individual Data from 27 Randomised Trials. Lancet 2012, 380, 581–590. [CrossRef]
- Efficacy and Safety of More Intensive Lowering of LDL Cholesterol: A Meta-Analysis of Data from 170000 Participants in 26 Randomised Trials. Lancet 2010, 376, 1670–1681. [CrossRef]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; Summerbell, C.D.; Worthington, H.V.; Song, F.; et al. Omega-3 Fatty Acids for the Primary and Secondary Prevention of Cardiovascular Disease. Cochrane Database Syst. Rev. 2020, 3, CD003177. [Google Scholar] [CrossRef]
- Ryan, E.; Galvin, K.; O’Connor, T.P.; Maguire, A.R.; O’Brien, N.M. Fatty Acid Profile, Tocopherol, Squalene and Phytosterol Content of Brazil, Pecan, Pine, Pistachio and Cashew Nuts. Int. J. Food Sci. Nutr. 2006, 57, 219–228. [Google Scholar] [CrossRef]
- Maguire, L.S.; O’Sullivan, S.M.; Galvin, K.; O’Connor, T.P.; O’Brien, N.M. Fatty Acid Profile, Tocopherol, Squalene and Phytosterol Content of Walnuts, Almonds, Peanuts, Hazelnuts and the Macadamia Nut. Int. J. Food Sci. Nutr. 2004, 55, 171–178. [Google Scholar] [CrossRef]
- Caldas, A.P.S.; Rocha, D.M.U.P.; Dionísio, A.P.; Hermsdorff, H.H.M.; Bressan, J. Brazil and Cashew Nuts Intake Improve Body Composition and Endothelial Health in Women at Cardiometabolic Risk (Brazilian Nuts Study): A Randomized Controlled Trial. Br. J. Nutr. 2022, 128, 1747–1757. [Google Scholar] [CrossRef]
- Hyson, D.A.; Schneeman, B.O.; Davis, P.A. Almonds and Almond Oil Have Similar Effects on Plasma Lipids and LDL Oxidation in Healthy Men and Women. J. Nutr. 2002, 132, 703–707. [Google Scholar] [CrossRef]
- Spiller, G.A.; Miller, A.; Olivera, K.; Reynolds, J.; Miller, B.; Morse, S.J.; Dewell, A.; Farquhar, J.W. Effects of Plant-Based Diets High in Raw or Roasted Almonds, or Roasted Almond Butter on Serum Lipoproteins in Humans. J. Am. Coll. Nutr. 2003, 22, 195–200. [Google Scholar] [CrossRef]
- Tey, S.L.; Robinson, T.; Gray, A.R.; Chisholm, A.W.; Brown, R.C. Do Dry Roasting, Lightly Salting Nuts Affect Their Cardioprotective Properties and Acceptability? Eur. J. Nutr. 2017, 56, 1025–1036. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Pearson, T.A.; Wan, Y.; Hargrove, R.L.; Moriarty, K.; Fishell, V.; Etherton, T.D. High–Monounsaturated Fatty Acid Diets Lower Both Plasma Cholesterol and Triacylglycerol Concentrations. Am. J. Clin. Nutr. 1999, 70, 1009–1015. [Google Scholar] [CrossRef]
- McKiernan, F.; Lokko, P.; Kuevi, A.; Sales, R.L.; Costa, N.M.B.; Bressan, J.; Alfenas, R.C.G.; Mattes, R.D. Effects of Peanut Processing on Body Weight and Fasting Plasma Lipids. Br. J. Nutr. 2010, 104, 418–426. [Google Scholar] [CrossRef]
- Mensink, R.P.; Zock, P.L.; Kester, A.D.; Katan, M.B. Effects of Dietary Fatty Acids and Carbohydrates on the Ratio of Serum Total to HDL Cholesterol and on Serum Lipids and Apolipoproteins: A Meta-Analysis of 60 Controlled Trials. Am. J. Clin. Nutr. 2003, 77, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Ros, E.; Singh, A.; O’Keefe, J.H. Nuts: Natural Pleiotropic Nutraceuticals. Nutrients 2021, 13, 3269. [Google Scholar] [CrossRef] [PubMed]
- Woollett, L.A.; Spady, D.K.; Dietschy, J.M. Saturated and Unsaturated Fatty Acids Independently Regulate Low Density Lipoprotein Receptor Activity and Production Rate. J. Lipid Res. 1992, 33, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Baccouch, R.; Shi, Y.; Vernay, E.; Mathelié-Guinlet, M.; Taib-Maamar, N.; Villette, S.; Feuillie, C.; Rascol, E.; Nuss, P.; Lecomte, S.; et al. The Impact of Lipid Polyunsaturation on the Physical and Mechanical Properties of Lipid Membranes. Biochim. Biophys. Acta (BBA) Biomembr. 2023, 1865, 184084. [Google Scholar] [CrossRef]
- Sampath, H.; Ntambi, J.M. Polyunsaturated fatty acid regulation of genes of lipid metabolism. Annu. Rev. Nutr. 2005, 25, 317–340. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Bulló, M.; Pérez-Heras, A.; Ros, E. Dietary Fibre, Nuts and Cardiovascular Diseases. Br. J. Nutr. 2006, 96, S45–S51. [Google Scholar] [CrossRef]
- Brown, L.; Rosner, B.; Willett, W.W.; Sacks, F.M. Cholesterol-Lowering Effects of Dietary Fiber: A Meta-Analysis. Am. J. Clin. Nutr. 1999, 69, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Zhang, G.; Qian, S.; Zhang, Q.; Tan, M. Associations between Dietary Fiber Intake and Cardiovascular Risk Factors: An Umbrella Review of Meta-Analyses of Randomized Controlled Trials. Front. Nutr. 2022, 9, 972399. [Google Scholar] [CrossRef]
- Moreau, R.A.; Nyström, L.; Whitaker, B.D.; Winkler-Moser, J.K.; Baer, D.J.; Gebauer, S.K.; Hicks, K.B. Phytosterols and Their Derivatives: Structural Diversity, Distribution, Metabolism, Analysis, and Health-Promoting Uses. Prog. Lipid Res. 2018, 70, 35–61. [Google Scholar] [CrossRef]
- del Gobbo, L.C.; Falk, M.C.; Feldman, R.; Lewis, K.; Mozaffarian, D. Are Phytosterols Responsible for the Low-Density Lipoprotein–Lowering Effects of Tree Nuts? J. Am. Coll. Cardiol. 2015, 65, 2765–2767. [Google Scholar] [CrossRef]
- Cofán, M.; Ros, E. Use of Plant Sterol and Stanol Fortified Foods in Clinical Practice. Curr. Med. Chem. 2019, 26, 6691–6703. [Google Scholar] [CrossRef]
- Ferguson, J.J.A.; Stojanovski, E.; MacDonald-Wicks, L.; Garg, M.L. Fat Type in Phytosterol Products Influence Their Cholesterol-Lowering Potential: A Systematic Review and Meta-Analysis of RCTs. Prog. Lipid Res. 2016, 64, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 Richest Dietary Sources of Polyphenols: An Application of the Phenol-Explorer Database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Colletti, A. Polyphenols Effect on Circulating Lipids and Lipoproteins: From Biochemistry to Clinical Evidence. Curr. Pharm. Des. 2018, 24, 178–190. [Google Scholar] [CrossRef]
- Meisinger, C.; Baumert, J.; Khuseyinova, N.; Loewel, H.; Koenig, W. Plasma Oxidized Low-Density Lipoprotein, a Strong Predictor for Acute Coronary Heart Disease Events in Apparently Healthy, Middle-Aged Men from the General Population. Circulation 2005, 112, 651–657. [Google Scholar] [CrossRef]
- Matsuura, E.; Hughes, G.R.V.; Khamashta, M.A. Oxidation of LDL and Its Clinical Implication. Autoimmun. Rev. 2008, 7, 558–566. [Google Scholar] [CrossRef] [PubMed]
- von Eckardstein, A.; Nordestgaard, B.G.; Remaley, A.T.; Catapano, A.L. High-Density Lipoprotein Revisited: Biological Functions and Clinical Relevance. Eur. Heart J. 2022; ahead of print. [Google Scholar] [CrossRef]
- Hernáez, Á.; Castañer, O.; Elosua, R.; Pintó, X.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Arós, F.; Serra-Majem, L.; Fiol, M.; et al. Mediterranean Diet Improves High-Density Lipoprotein Function in High-Cardiovascular-Risk Individuals. Circulation 2017, 135, 633–643. [Google Scholar] [CrossRef]
- Holligan, S.D.; West, S.G.; Gebauer, S.K.; Kay, C.D.; Kris-Etherton, P.M. A Moderate-Fat Diet Containing Pistachios Improves Emerging Markers of Cardiometabolic Syndrome in Healthy Adults with Elevated LDL Levels. Br. J. Nutr. 2014, 112, 744–752. [Google Scholar] [CrossRef]
- Berryman, C.E.; Grieger, J.A.; West, S.G.; Chen, C.-Y.O.; Blumberg, J.B.; Rothblat, G.H.; Sankaranarayanan, S.; Kris-Etherton, P.M. Acute Consumption of Walnuts and Walnut Components Differentially Affect Postprandial Lipemia, Endothelial Function, Oxidative Stress, and Cholesterol Efflux in Humans with Mild Hypercholesterolemia. J. Nutr. 2013, 143, 788–794. [Google Scholar] [CrossRef]
- Yvan-Charvet, L.; Wang, N.; Tall, A.R. Role of HDL, ABCA1, and ABCG1 Transporters in Cholesterol Efflux and Immune Responses. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 139–143. [Google Scholar] [CrossRef]
- Reyes-Soffer, G.; Ginsberg, H.N.; Berglund, L.; Duell, P.B.; Heffron, S.P.; Kamstrup, P.R.; Lloyd-Jones, D.M.; Marcovina, S.M.; Yeang, C.; Koschinsky, M.L.; et al. Lipoprotein(a): A Genetically Determined, Causal, and Prevalent Risk Factor for Atherosclerotic Cardiovascular Disease: A Scientific Statement From the American Heart Association. Arterioscler. Thromb. Vasc. Biol. 2022, 42, e48–e60. [Google Scholar] [CrossRef]
- Krauss, R.M. Lipoprotein Subfractions and Cardiovascular Disease Risk. Curr. Opin. Lipidol. 2010, 21, 305–311. [Google Scholar] [CrossRef]
- Qiao, Y.-N.; Zou, Y.-L.; Guo, S.-D. Low-Density Lipoprotein Particles in Atherosclerosis. Front. Physiol. 2022, 13, 931931. [Google Scholar] [CrossRef]
- Mora, S.; Otvos, J.D.; Rifai, N.; Rosenson, R.S.; Buring, J.E.; Ridker, P.M. Lipoprotein Particle Profiles by Nuclear Magnetic Resonance Compared With Standard Lipids and Apolipoproteins in Predicting Incident Cardiovascular Disease in Women. Circulation 2009, 119, 931–939. [Google Scholar] [CrossRef]
- Valkama, A.J.; Meinilä, J.M.; Koivusalo, S.B.; Lindström, J.; Rönö, K.; Stach-Lempinen, B.; Eriksson, J.G. Diet Quality as Assessed by the Healthy Food Intake Index and Relationship with Serum Lipoprotein Particles and Serum Fatty Acids in Pregnant Women at Increased Risk for Gestational Diabetes. Br. J. Nutr. 2018, 120, 914–924. [Google Scholar] [CrossRef]
- Phillips, C.M.; Harrington, J.M.; Perry, I.J. Relationship between Dietary Quality, Determined by DASH Score, and Cardiometabolic Health Biomarkers: A Cross-Sectional Analysis in Adults. Clin. Nutr. 2019, 38, 1620–1628. [Google Scholar] [CrossRef]
- Millar, S.R.; Navarro, P.; Harrington, J.M.; Shivappa, N.; Hébert, J.R.; Perry, I.J.; Phillips, C.M. Comparing Dietary Score Associations with Lipoprotein Particle Subclass Profiles: A Cross-Sectional Analysis of a Middle-to Older-Aged Population. Clin. Nutr. 2021, 40, 4720–4729. [Google Scholar] [CrossRef]
- García-Gavilán, J.F.; Connelly, M.A.; Babio, N.; Mantzoros, C.S.; Ros, E.; Salas-Salvadó, J. Nut Consumption Is Associated with a Shift of the NMR Lipoprotein Subfraction Profile to a Less Atherogenic Pattern among Older Individuals at High CVD Risk. Cardiovasc. Diabetol. 2022, 21, 189. [Google Scholar] [CrossRef]
- Rajaram, S.; Cofán, M.; Sala-Vila, A.; Haddad, E.; Serra-Mir, M.; Bitok, E.; Roth, I.; Freitas-Simoes, T.M.; Kaur, A.; Valls-Pedret, C.; et al. Effects of Walnut Consumption for 2 Years on Lipoprotein Subclasses Among Healthy Elders. Circulation 2021, 144, 1083–1085. [Google Scholar] [CrossRef]
- Lee, Y.; Berryman, C.E.; West, S.G.; Chen, C.-Y.O.; Blumberg, J.B.; Lapsley, K.G.; Preston, A.G.; Fleming, J.A.; Kris-Etherton, P.M. Effects of Dark Chocolate and Almonds on Cardiovascular Risk Factors in Overweight and Obese Individuals: A Randomized Controlled-Feeding Trial. J. Am. Heart Assoc. 2017, 6, e005162. [Google Scholar] [CrossRef]
- Tindall, A.M.; Kris-Etherton, P.M.; Petersen, K.S. Replacing Saturated Fats with Unsaturated Fats from Walnuts or Vegetable Oils Lowers Atherogenic Lipoprotein Classes Without Increasing Lipoprotein(a). J. Nutr. 2020, 150, 818–825. [Google Scholar] [CrossRef]
- Hernández-Alonso, P.; Salas-Salvadó, J.; Baldrich-Mora, M.; Mallol, R.; Correig, X.; Bulló, M. Effect of Pistachio Consumption on Plasma Lipoprotein Subclasses in Pre-Diabetic Subjects. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 396–402. [Google Scholar] [CrossRef]
- Xepapadaki, E.; Nikdima, I.; Sagiadinou, E.C.; Zvintzou, E.; Kypreos, K.E. HDL and Type 2 Diabetes: The Chicken or the Egg? Diabetologia 2021, 64, 1917–1926. [Google Scholar] [CrossRef]
- Fernández-Rodríguez, R.; Mesas, A.E.; Garrido-Miguel, M.; Martínez-Ortega, I.A.; Jiménez-López, E.; Martínez-Vizcaíno, V. The Relationship of Tree Nuts and Peanuts with Adiposity Parameters: A Systematic Review and Network Meta-Analysis. Nutrients 2021, 13, 2251. [Google Scholar] [CrossRef]
- Guarneiri, L.L.; Cooper, J.A. Intake of Nuts or Nut Products Does Not Lead to Weight Gain, Independent of Dietary Substitution Instructions: A Systematic Review and Meta-Analysis of Randomized Trials. Adv. Nutr. 2021, 12, 384–401. [Google Scholar] [CrossRef]
- Nishi, S.K.; Viguiliouk, E.; Blanco Mejia, S.; Kendall, C.W.C.; Bazinet, R.P.; Hanley, A.J.; Comelli, E.M.; Salas Salvadó, J.; Jenkins, D.J.A.; Sievenpiper, J.L. Are Fatty Nuts a Weighty Concern? A Systematic Review and Meta-Analysis and Dose-Response Meta-Regression of Prospective Cohorts and Randomized Controlled Trials. Obes. Rev. 2021, 22, 13330. [Google Scholar] [CrossRef]
- Fernández-Rodríguez, R.; Martínez-Vizcaíno, V.; Garrido-Miguel, M.; Martínez-Ortega, I.A.; Álvarez-Bueno, C.; Eumann Mesas, A. Nut Consumption, Body Weight, and Adiposity in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutr. Rev. 2022, 80, 645–655. [Google Scholar] [CrossRef]
- McArthur, B.M.; Mattes, R.D. Energy Extraction from Nuts: Walnuts, Almonds and Pistachios. Br. J. Nutr. 2020, 123, 361–371. [Google Scholar] [CrossRef]
- Tapsell, L.C.; Neale, E.P.; Satija, A.; Hu, F.B. Foods, Nutrients, and Dietary Patterns: Interconnections and Implications for Dietary Guidelines. Adv. Nutr. 2016, 7, 445–454. [Google Scholar] [CrossRef]
- Yi, S.Y.; Steffen, L.M.; Zhou, X.; Shikany, J.M.; Jacobs, D.R. Association of Nut Consumption with CVD Risk Factors in Young to Middle-Aged Adults: The Coronary Artery Risk Development in Young Adults (CARDIA) Study. Nutr. Metab. Cardiovasc. Dis. 2022, 32, 2321–2329. [Google Scholar] [CrossRef]
- Gebauer, S.K.; Novotny, J.A.; Bornhorst, G.M.; Baer, D.J. Food Processing and Structure Impact the Metabolizable Energy of Almonds. Food Funct. 2016, 7, 4231–4238. [Google Scholar] [CrossRef] [PubMed]
- Creedon, A.C.; Hung, E.S.; Berry, S.E.; Whelan, K. Nuts and Their Effect on Gut Microbiota, Gut Function and Symptoms in Adults: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2020, 12, 2347. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, E.; Lambert, K.; Stanford, J.; Neale, E.P. The Effect of Nut Consumption (Tree Nuts and Peanuts) on the Gut Microbiota of Humans: A Systematic Review. Br. J. Nutr. 2021, 125, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Hernández-Alonso, P.; Drouin-Chartier, J.-P.; Ruiz-Canela, M.; Razquin, C.; Toledo, E.; Li, J.; Dennis, C.; Wittenbecher, C.; Corella, D.; et al. Walnut Consumption, Plasma Metabolomics, and Risk of Type 2 Diabetes and Cardiovascular Disease. J. Nutr. 2021, 151, 303–311. [Google Scholar] [CrossRef]
| Publication | Search Dates | Population | Study Design | Sample Size | Duration of Intervention | Intervention | Control | Outcome Measures | Results Mean Change in mmol/L (95%CI) |
|---|---|---|---|---|---|---|---|---|---|
| Phung, 2009 [13] | through Jul 2008 | Non-specified | RCTs with parallel or crossover design | 5 RCTs 142 participants | 4 weeks | Almonds 25–168 g/d | NCEP step II, usual diet, NCEP step I, high-fat diet, low-fat diet | Lipid profile: TC, LDL-C, HDL-C, TG, LDL/HDL; ApoA-I and apoB Lp(a) | ↓ LDL-C −0.18 (−0.34, −0.02) (5 RCTs) |
| Banel, 2009 [24] | through May 2008 | All patient populations and age groups | RCTs with parallel or crossover design | 13 RCTs 365 participants | 4–24 weeks | Walnuts 15–108 g/d | Controlled diet, Western diet, Med diet, modified low-fat diet, habitual diet, low-fat diet, cholesterol lowering meals | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TC −0.27 (−0.38, −0.15) ↓ LDL-C −0.24 (−0.34, −0.14) (11 RCTs) |
| Sabaté, 2010 [12] | 1992–2004 | No recent exposure to lipid-lowering medications | Controlled trials; duration of intervention ≥ 3 weeks; no body weight change between diets at the end of intervention | 25 trials 583 participants (pooled analysis with individual participant data) | 3–8 weeks | Tree nuts and peanuts 34–100 g/d | Western diet, Med diet, low total, and saturated fat | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TC −0.28 (−0.36, −0.2) ↓ LDL-C −0.26 (−0.34, −0.19) (25 trials) |
| Mejia, 2014 [16] | through Apr 2014 | Non-specified | RCTs; duration of intervention ≥ 3 weeks | 47 RCTs 2211 participants | 3 weeks–18 months | Tree nuts (almonds, Brazil nuts, cashews, hazelnuts, macadamia nuts, pecans, pine nuts, pistachios, walnuts, and mixed nuts) 30–85.5 g/d | Habitual diet, diet for diabetes, Western diet, low-fat diet, muffin, NCEP step I diet, AHA step 1 diet, ad libitum diet, NCEP step II diet, NCEP step II diet + muffin, cheese | At least one criterion of MetS (waist circumference, TG, HDL-C, blood pressure, glycemic control) | ↓ TG −0.06 (−0.09, −0.03) (43 RCTs) |
| Del Gobbo, 2015 [22] | through Mar 2013 | Free of known CVD; Not receiving medication for diabetes, obesity, MetS, hypertension or hyperlipidemia; ≥18 yo | Randomized and nonrandomized controlled trials with parallel or crossover design | 42 RCTs and 18 nonrandomized trials 2582 participants | 3–26 weeks | Tree nuts 5–100 g/d | Habitual diet, healthy diet, low-fat diet, high-CHO diet, olive oil diet, habitual diet + red meat, low saturated fat diet with cereals and canola oil, AHA step 1 diet, American diet, isocaloric controlled diet, NCEP step 1 or 2 diet, salted pretzels, isocaloric high cholesterol diet, NCEP step 1, Med diet, AHA step 1 diet, ADA diet (with and without nuts) | Lipid profile: TC, LDL-C, HDL-C, TG; apolipoproteins | ↓ TC −0.09 (−0.11, −0.07) ↓ LDL-C −0.11 (−0.13, −0.09) (38 RCTs) ↓ ApoB (g/L) −0.042 (−0.065, −0.026) (13 RCTs) |
| Guasch-Ferré, 2018 [23] | through Jan 2018 | Adults | RCTs with a parallel or crossover design; Duration of intervention ≥ 3 weeks | 26 RCTs 1059 participants | 4 weeks–1 year | Walnuts 15–108 g/d | ad libitum control diet, Med diet, ADA diet, low-fat diet, habitual diet, controlled diet (walnut-free) | At least one of the lipid markers: TC, LDL-C, HDL-C, TG apolipoproteins | ↓ TC −0.18 (−0.24, −0.12) ↓ LDL-C −0.14 (−0.2, −0.09) ↓ TG −0.05 (−0.1, −0.01) (23 RCTs) |
| Lee-Bravatti, 2019 [19] | 2015-June 2017 for lipid outcomes | Healthy or with CVD risk factors; ≥18 yo | RCTs; Duration of intervention ≥ 3 weeks | 15 RCTs 534 participants | 4–16 weeks | Almonds 37–113 g/d | NCEP step II diet, low-fat diet, high-fat diet custom diet, Med diet, NCEP step I diet, ADA diet | Lipid profile: TC, LDL-C, HDL-C, TG, TC/HDL, HDL/LDL; apolipoproteins, Lp(a) | ↓ TC −0.28 (−0.43, −0.12) ↓ LDL-C −0.15 (−0.26, −0.05) (13 RCTs) |
| Morvaridzadeh, 2020 [28] | through June 2019 | Non-specified | RCTs with a parallel or crossover design | 3 RCTs 384 participants | 4–12 weeks | Cashews 28–108 g | Isocaloric diet, baked potato chips | Lipid profile: TC, LDL-C, HDL-C, TG | No change |
| Liu, 2020 [18] | through June 2019 | ≥18 yo | RCTs; duration of intervention ≥ 3 weeks | 34 RCTs 1677 participants | 3–24 weeks | Tree nuts (walnuts, pistachios, hazelnuts, cashews, or almonds) 15–168 g/d | Control diet (nut-free) | Lipid profile: TC, LDL-C, HDL-C, TG | Walnut-enriched ↓ LDL-C −0.09 (−0.12, −0.07) ↓ TG −0.09 (−0.11, −0.07) (16 RCTs) Pistachio-enriched ↓ LDL-C −0.17 (−0.28, −0.06) Hazelnut-enriched No change Almond-enriched No change |
| Jalali, 2020 [20] | through Nov 2019 | ≥18 yo | RCTs | 3 RCTs 392 participants | 4–12 weeks | Cashews 30–42 g/d | Diet for diabetes, isocaloric controlled diet (nut-free) | Lipid profile: TC, LDL-C, HDL-C, TG | No change |
| Hadi, 2021 [27] | through June 2019 | ≥18 yo | RCTs; duration of intervention ≥ 3 weeks | 12 RCTs 771 participants | 3–24 weeks | Pistachios 32–126 g/d | Control diet | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TC −0.19 (−0.33, −0.06) (10 RCTs) ↓ LDL-C −0.1 (−0.14, −0.06) (12 RCTs) ↓ TG −0.13 (−0.16, −0.09) (10 RCTs) |
| Asbaghi, 2021 [25] | through Sept 2020 | Healthy or otherwise; ≥18 yo | RCTs with a parallel or crossover design; Duration of intervention ≥ 3 weeks | 27 RCTs 2049 participants | 3–77 weeks | Almonds 10–168 g/d | No almond consumption or dietary substitutions containing no almond were used | At least one of the lipid markers: TC, LDL-C, HDL-C, TG | ↓ TC −0.13 (−0.2, −0.05) ↓ TG −0.08 (−0.13, −0.02) (27 RCTs) ↓ LDL-C −0.15 (−0.23, −0.07) (26 RCTs) |
| Wang, 2021 [29] | through Jan 2020 | Adults with T2DB | RCTs with a parallel or crossover design; Duration of intervention ≥ 2 weeks | 5 RCTs 120 participants | 3–12 weeks | Almonds 30–60 g/d | Control diet, NCEP step II diet, peanuts, sunflower kernels | Lipid profile: TC, LDL-C, HDL-C, TG | No change |
| Xia, 2021 [11] | through June 2021 | Patients with T2DB | RCTs | 16 RCTs 1041 participants | 6–52 weeks | Peanuts and tree nuts (walnuts, pistachios, macadamia nuts, pecans, cashews, almonds, hazelnuts, pine nuts, and Brazil nuts) 6–128 g/d | High-fat diet, low-fat diet, normal-fat diet, habitual diet, diet for diabetes, ADA meal plan (nut-free) | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TC −0.14 (−0.26, −0.02) (14 RCTs) ↓ TG −0.1 (−0.17, −0.02) (12 RCTs) |
| Moosavian, 2022 [15] | through Mar 2021 | Patients with T2DB; ≥18 yo | RCTs; Duration of intervention ≥ 3 weeks | 9 RCTs 264 participants | 4–12 weeks | Almonds 29–113g/d | NCEP step II diet, cheese, raw peanut with low carbohydrate diet, high-fat diet, low-fat diet, sunflower kernels with diabetic diet, custom diet (almond-free) | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ LDL −0.14 (−0.26, −0.02) (8 RCTs) |
| Arabi, 2022 [26] | through Dec 2021 | Diagnosed with MetS; ≥18 yo | RCTs with a parallel or crossover design | 8 RCTs 506 participants | 4–112 days | Walnuts (all forms, plain, or walnut-fortified food) 30 g–108 g/d | Standardized shakes, control diet, isocaloric white bread, ad libitum diet without walnuts, lifestyle counseling | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TG −0.17 (−0.32, −0.03) (5 RCTs) |
| Mates, 2022 [17] | through Nov 2021 | Middle-aged and older adults ≥40 yo or mean age ≥50 yo | RCTs with a parallel or crossover design; Duration of intervention ≥ 3 weeks | 17 RCTs 2466 participants | 4 weeks-2 years | Walnuts (including plain or walnut-fortified food) 19.3–75 g/d | Med diet, modified low-fat diet, Western-type diet, habitual diet, CKD patients’ diet (walnut-free) | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TC −0.13 (−0.2, −0.07) ↓ LDL-C −0.15 (−0.2, −0.11) (12 RCTs) ↓ TG −0.08 (−0.12, −0.04) (13 RCTs) |
| Eslami, 2022 [21] | through Apr 2021 | Overweight/obese (BMI: 25–40 kg/m2); free of chronic diseases; ≥18 yo | RCTs with a parallel or crossover design; Duration of intervention ≥ one week | 10 RCTS 711 participants | 4–72 weeks | Peanuts and tree nuts (almonds, walnuts, hazelnuts, pistachios, cashews, macadamia nuts, Brazil nuts, pine nuts, pecans, mixed nuts) 20–60 g/d | Isocaloric nut-free diet | At least one of the following: Serum lipid profile: TC, LDL-C, HDL-C, TG | ↓ TG −0.15 (−0.29, −0.01) (9 RCTs) |
| Parilli-Moser, 2022 [14] | through July 2021 | Healthy or with MetS or at high risk of MetS | RCTs | 9 RCTs 643 participants | 2–24 weeks | Peanuts, peanut butter or high oleic acid peanuts 25–200 g/d | Hypocaloric diet, habitual diet, ADA meal plan, substitute snack (grain bar, white rice bar, candy, or almonds) (peanut-free) | Lipid profile: TC, LDL-C, HDL-C, TG | ↓ TG −0.13 (−0.2, −0.07) (9 RCTs) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guasch-Ferré, M.; Tessier, A.-J.; Petersen, K.S.; Sapp, P.A.; Tapsell, L.C.; Salas-Salvadó, J.; Ros, E.; Kris-Etherton, P.M. Effects of Nut Consumption on Blood Lipids and Lipoproteins: A Comprehensive Literature Update. Nutrients 2023, 15, 596. https://doi.org/10.3390/nu15030596
Guasch-Ferré M, Tessier A-J, Petersen KS, Sapp PA, Tapsell LC, Salas-Salvadó J, Ros E, Kris-Etherton PM. Effects of Nut Consumption on Blood Lipids and Lipoproteins: A Comprehensive Literature Update. Nutrients. 2023; 15(3):596. https://doi.org/10.3390/nu15030596
Chicago/Turabian StyleGuasch-Ferré, Marta, Anne-Julie Tessier, Kristina S. Petersen, Philip A. Sapp, Linda C. Tapsell, Jordi Salas-Salvadó, Emilio Ros, and Penny M. Kris-Etherton. 2023. "Effects of Nut Consumption on Blood Lipids and Lipoproteins: A Comprehensive Literature Update" Nutrients 15, no. 3: 596. https://doi.org/10.3390/nu15030596
APA StyleGuasch-Ferré, M., Tessier, A.-J., Petersen, K. S., Sapp, P. A., Tapsell, L. C., Salas-Salvadó, J., Ros, E., & Kris-Etherton, P. M. (2023). Effects of Nut Consumption on Blood Lipids and Lipoproteins: A Comprehensive Literature Update. Nutrients, 15(3), 596. https://doi.org/10.3390/nu15030596








