High Extra Virgin Olive Oil Consumption Is Linked to a Lower Prevalence of NAFLD with a Prominent Effect in Obese Subjects: Results from the MICOL Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. The MICOL Study
2.2. Outcome, Clinical, and Dietary Data
2.3. Statistical Analysis
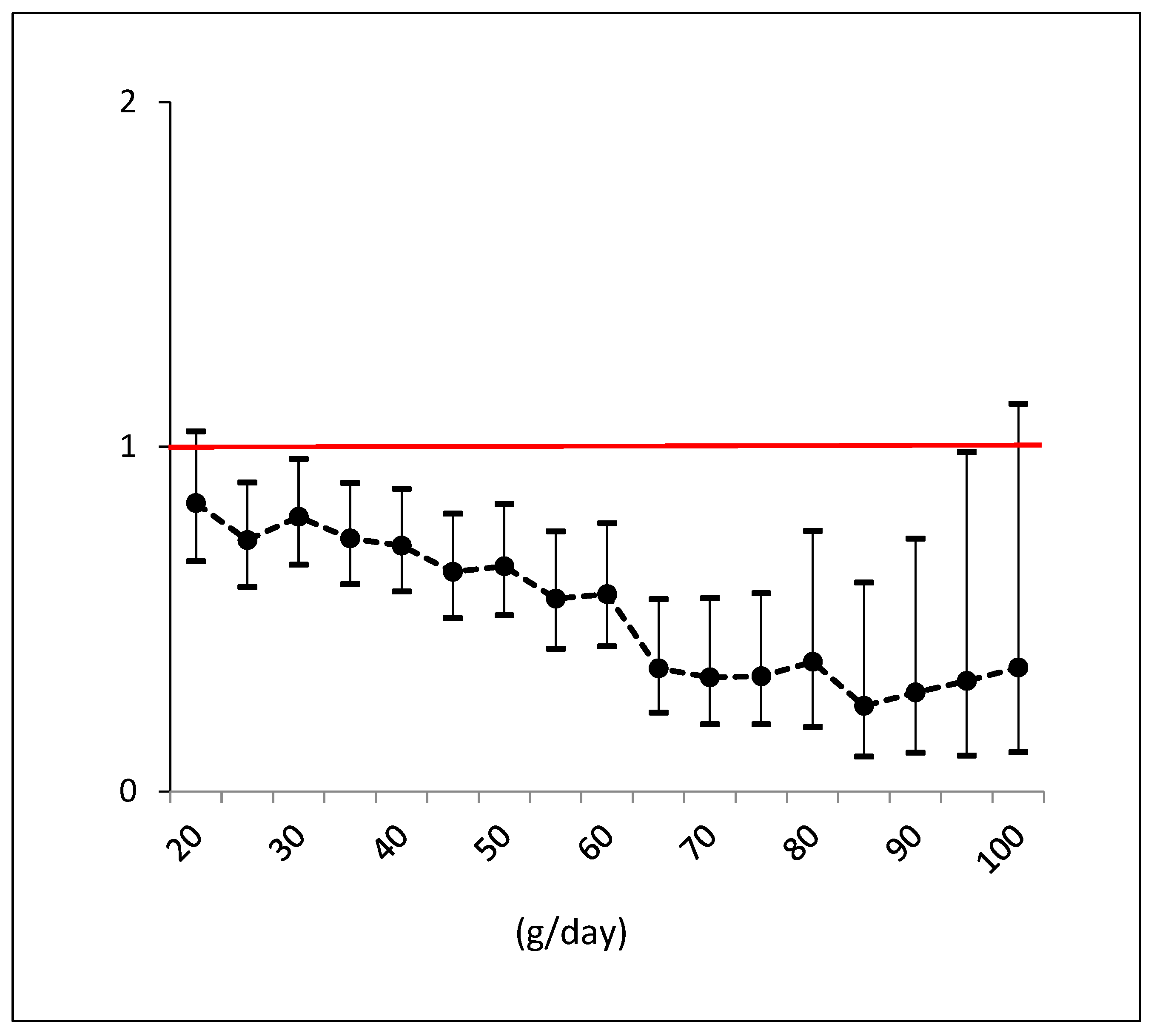
3. Results
3.1. Role of Mediterranean Food Items as NAFLD Risk Factors
3.2. Differential Effect of EVOO on Body Weight
3.3. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| NAFLD | Non-alcoholic fatty liver disease |
| EVOO | Extra virgin olive oil |
| MD | Mediterranean diet |
| BMI | Body mass index |
| NASH | Non-alcoholic steatohepatitis |
| CVD | Cardiovascular disease |
| GWAS | Genome-wide association studies |
| T2DM | Type 2 diabetes mellitus |
| SBP | Systolic blood pressure |
| DBP | Diastolic blood pressure |
| GGT | Gamma-glutamyl transferase |
| AST | Aspartate aminotransferase |
| ALT | Alanine aminotransferase |
| HDL-C | High-density lipoprotein cholesterol |
| LDL-C | Low-density lipoprotein cholesterol |
| BMR | Basal metabolic rate |
| ORs | Odds ratios |
| SD | Standard deviation |
| C.I. | Confidence interval |
| RRR | Relative risk reduction |
| p | Alpha level |
| RCT | Randomized controlled trial |
References
- Powell, E.E.; Wong, V.W.S.; Rinella, M. Non-alcoholic fatty liver disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- Pierantonelli, I.; Svegliati-Baroni, G. Nonalcoholic Fatty Liver Disease: Basic Pathogenetic Mechanisms in the Progression From NAFLD to NASH. Transplantation 2019, 103, e1–e13. [Google Scholar] [CrossRef] [PubMed]
- Thoma, C.; Day, C.P.; Trenell, M.I. Lifestyle interventions for the treatment of non-alcoholic fatty liver disease in adults: A systematic review. J. Hepatol. 2012, 56, 255–266. [Google Scholar] [CrossRef]
- Koutoukidis, D.A.; Koshiaris, C.; Henry, J.A.; Noreik, M.; Morris, E.; Manoharan, I.; Tudor, K.; Bodenham, E.; Dunnigan, A.; Jebb, S.A.; et al. The effect of the magnitude of weight loss on non-alcoholic fatty liver disease: A systematic review and meta-analysis. Metabolism 2020, 115, 154455. [Google Scholar] [CrossRef]
- Promrat, K.; Kleiner, D.E.; Niemeier, H.M.; Jackvony, E.; Kearns, M.; Wands, J.R.; Fava, J.L.; Wing, R.R. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010, 51, 121–129. [Google Scholar] [CrossRef]
- Marchesini, G.; Day, C.P.; Dufour, J.F.; Canbay, A.; Nobili, V.; Ratziu, V.; Tilg, H.; Roden, M.; Gastaldelli, A.; Yki-Järvinen, H.; et al. EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef]
- Haigh, L.; Kirk, C.; El Gendy, K.; Gallacher, J.; Errington, L.; Mathers, J.C.; Anstee, Q.M. The effectiveness and acceptability of Mediterranean diet and calorie restriction in non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis. Clin. Nutr. 2022, 41, 1913–1931. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Salomone, F.; Mlynarsky, L. The Mediterranean dietary pattern as the diet of choice for non-alcoholic fatty liver disease: Evidence and plausible mechanisms. Liver Int. 2017, 37, 936–949. [Google Scholar] [CrossRef]
- Jensen, T.; Abdelmalek, M.F.; Sullivan, S.; Nadeau, K.J.; Green, M.; Roncal, C.; Nakagawa, T.; Kuwabara, M.; Sato, Y.; Kang, D.-H.; et al. Fructose and sugar: A major mediator of non-alcoholic fatty liver disease. J. Hepatol. 2018, 68, 1063–1075. [Google Scholar] [CrossRef]
- Yki-Järvinen, H.; Luukkonen, P.K.; Hodson, L.; Moore, J.B. Dietary carbohydrates and fats in nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 770–786. [Google Scholar] [CrossRef]
- Berná, G.; Romero-Gomez, M. The role of nutrition in non-alcoholic fatty liver disease: Pathophysiology and management. Liver Int. 2020, 40, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Perdomo, C.M.; Frühbeck, G.; Escalada, J. Impact of Nutritional Changes on Nonalcoholic Fatty Liver Disease. Nutrients 2019, 11, 677. [Google Scholar] [CrossRef] [PubMed]
- Alferink, L.J.M.; Erler, N.S.; de Knegt, R.J.; Janssen, H.L.A.; Metselaar, H.J.; Murad, S.D.; Jong, J.C.K.-D. Adherence to a plant-based, high-fibre dietary pattern is related to regression of non-alcoholic fatty liver disease in an elderly population. Eur. J. Epidemiology 2020, 35, 1069–1085. [Google Scholar] [CrossRef] [PubMed]
- Assy, N.; Nassar, F.; Nasser, G.; Grosovski, M. Olive oil consumption and non-alcoholic fatty liver disease. World J. Gastroenterol. 2009, 15, 1809–1815. [Google Scholar] [CrossRef] [PubMed]
- Soto-Alarcon, S.A.; Valenzuela, R.; Valenzuela, A.; Videla, L.A. Liver Protective Effects of Extra Virgin Olive Oil: Interaction between Its Chemical Composition and the Cell-signaling Pathways Involved in Protection. Endocr. Metab. Immune Disord. -Drug Targets 2017, 18, 75–84. [Google Scholar] [CrossRef]
- Hussein, O.; Grosovski, M.; Lasri, E.; Svalb, S.; Ravid, U.; Assy, N. Monounsaturated fat decreases hepatic lipid content in non-alcoholic fatty liver disease in rats. World J. Gastroenterol. 2007, 13, 361–368. [Google Scholar] [CrossRef]
- Li, Z.Z.; Berk, M.; McIntyre, T.M.; Feldstein, A.E. Hepatic lipid partitioning and liver damage in nonalcoholic fatty liver disease: Role of stearoyl-Coa desaturase. J. Biol. Chem. 2009, 284, 5637–5644. [Google Scholar] [CrossRef]
- Ricchi, M.; Odoardi, M.R.; Carulli, L.; Anzivino, C.; Ballestri, S.; Pinetti, A.; Fantoni, L.I.; Marra, F.; Bertolotti, M.; Banni, S.; et al. Differential effect of oleic and palmitic acid on lipid accumulation and apoptosis in cultured hepatocytes. J. Gastroenterol. Hepatol. 2009, 24, 830–840. [Google Scholar] [CrossRef]
- Misciagna, G.; Leoci, C.; Elba, S.; Petruzzi, J.; Guerra, V.; Mossa, A.; Noviello, M.R.; Coviello, A.; Capece-Minutolo, M.; Giorgiot, I. The epidemiology of cholelithiasis in southern Italy. Eur. J. Gastroenterol. Hepatol. 1994, 6, 937–942. [Google Scholar] [CrossRef]
- Osella, A.R.; Misciagna, G.; Guerra, V.M.; Chiloiro, M.; Cuppone, R.; Cavallini, A.; Di Leo, A. Hepatitis C virus (HCV) infection and liver-related mortality: A population-based cohort study in southern Italy. Int. J. Epidemiol. 2000, 29, 922–927. [Google Scholar] [CrossRef]
- Saadeh, S.; Younossi, Z.M.; Remer, E.M.; Gramlich, T.; Ong, J.P.; Hurley, M.; Mullen, K.D.; Cooper, J.N.; Sheridan, M.J. The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002, 123, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. Br. Med. J. 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- Schofield, W.N. Predicting basal metabolic rate, new standards and review of previous work. Hum. Nutr. Clin. Nutr. 1985, 39 (Suppl. S1), 5–41. [Google Scholar]
- Zelber-Sagi, S.; Ivancovsky-Wajcman, D.; Isakov, N.F.; Webb, M.; Orenstein, D.; Shibolet, O.; Kariv, R. High red and processed meat consumption is associated with non-alcoholic fatty liver disease and insulin resistance. J. Hepatol. 2018, 68, 1239–1246. [Google Scholar] [CrossRef]
- Semmler, G.; Bachmayer, S.; Wernly, S.; Wernly, B.; Niederseer, D.; Huber-Schönauer, U.; Stickel, F.; Aigner, E.; Datz, C. Nut consumption and the prevalence and severity of non-alcoholic fatty liver disease. PLoS ONE 2020, 15, e0244514. [Google Scholar] [CrossRef]
- Kim, S.-A.; Shin, S. Fruit and vegetable consumption and non-alcoholic fatty liver disease among Korean adults: A prospective cohort study. J. Epidemiol. Community Health 2020, 74, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Pintó, X.; Fanlo-Maresma, M.; Corbella, E.; Corbella, X.; Mitjavila, M.T.; Moreno, J.J.; Casas, R.; Estruch, R.; Corella, D.; Bulló, M.; et al. A Mediterranean Diet Rich in Extra-Virgin Olive Oil Is Associated with a Reduced Prevalence of Nonalcoholic Fatty Liver Disease in Older Individuals at High Cardiovascular Risk. J. Nutr. 2019, 149, 1920–1929. [Google Scholar] [CrossRef]
- Dorosti, M.; Jafary Heidarloo, A.; Bakhshimoghaddam, F.; Alizadeh, M. Whole-grain consumption and its effects on hepatic steatosis and liver enzymes in patients with non-alcoholic fatty liver disease: A randomised controlled clinical trial. Br. J. Nutr. 2020, 123, 328–336. [Google Scholar] [CrossRef]
- He, K.; Li, Y.; Guo, X.; Zhong, L.; Tang, S. Food groups and the likelihood of non-alcoholic fatty liver disease: A systematic review and meta-analysis. Br. J. Nutr. 2020, 124, 1–13. [Google Scholar] [CrossRef]
- Tajima, R.; Kimura, T.; Enomoto, A.; Saito, A.; Kobayashi, S.; Masuda, K.; Iida, K. No association between fruits or vegetables and non-alcoholic fatty liver disease in middle-aged men and women. Nutrition 2019, 61, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, S.; Akhlaghi, M.; Sasani, M.R.; Boldaji, R.B. Olive oil lessened fatty liver severity independent of cardiometabolic correction in patients with non-alcoholic fatty liver disease: A randomized clinical trial. Nutrition 2019, 57, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Rodas, M.C.; Valenzuela, R.; Echeverría, F.; Rincón-Cervera, M.Á.; Espinosa, A.; Illesca, P.; Muñoz, P.; Corbari, A.; Romero, N.; Gonzalez-Mañan, D.; et al. Supplementation with Docosahexaenoic Acid and Extra Virgin Olive Oil Prevents Liver Steatosis Induced by a High-Fat Diet in Mice through PPAR-α and Nrf2 Upregulation with Concomitant SREBP-1c and NF-kB Downregulation. Mol. Nutr. Food Res. 2017, 61, 1700479. [Google Scholar] [CrossRef]
- Alferink, L.J.; Jong, J.C.K.-D.; Erler, N.S.; Veldt, B.J.; Schoufour, J.D.; de Knegt, R.J.; Ikram, M.A.; Metselaar, H.J.; LA Janssen, H.; Franco, O.H.; et al. Association of dietary macronutrient composition and non-alcoholic fatty liver disease in an ageing population: The Rotterdam Study. Gut 2018, 68, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Choi, Y.; Um, S.-J.; Yoon, S.K.; Park, T. Oleuropein attenuates hepatic steatosis induced by high-fat diet in mice. J. Hepatol. 2010, 54, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, Y.M.; Bermúdez, B.; López, S.; Abia, R.; Villar, J.; Muriana, F.J.G. Minor compounds of olive oil have postprandial anti-inflammatory effects. Br. J. Nutr. 2007, 98, 260–263. [Google Scholar] [CrossRef]
- Covas, M.I.; Nyyssönen, K.; Poulsen, H.E.; Kaikkonen, J.; Zunft, H.J.F.; Kiesewetter, H.; Gaddi, A.; de la Torre, R.; Mursu, J.; Bäumler, H. The effect of polyphenols in olive oil on heart disease risk factors: A randomized trial. Ann. Intern. Med. 2006, 145, 333–341. [Google Scholar] [CrossRef]

| No NAFLD | NAFLD | ||
|---|---|---|---|
| n = 1628 | n = 808 | p-Value | |
| Demographics | |||
| Age (years) | 53.8 ± 16 | 55.6 ± 14 | 0.004 |
| Male n (%) | 804 (49) | 468 (58) | <0.0001 |
| Educational levels | |||
| Illiterate | 59 (3.6) | 30 (3.7) | |
| Primary School | 469 (28.9) | 254 (31.4) | |
| Secondary School | 495 (30.5) | 247 (30.6) | 0.6 |
| High School | 401 (24.7) | 191 (23.6) | |
| Graduated | 200 (12.3) | 86 (10.6) | |
| Living together yes n (%) | 1215 (74) | 647 (80) | 0.003 |
| Work classes | |||
| Jobless or Pensioneers | 550 (33.8) | 298 (36.9) | |
| Elementary Occupations | 584 (35.9) | 293 (36.3) | 0.47 |
| Craft, Agriculture, and Sales | 421 (25.9) | 185 (22.9) | |
| Managers and Professionals | 73 (4.5) | 32 (4) | |
| Smoking habit yes n (%) | 274 (17) | 122 (15) | 0.27 |
| Anthropometrics | |||
| BMI classes | |||
| Normo-weight | 587 (36.1) | 47 (5.8) | |
| Overweight | 722 (44.4) | 287 (35.5) | <0.0001 |
| Obese | 319 (19.6) | 474 (58.7) | |
| Waist (cm) | 87 ± 12 | 100 ± 11.5 | <0.0001 |
| Hip (cm) | 100 ± 9.2 | 109 ± 11 | <0.0001 |
| Food data | |||
| Extra virgin olive oil (g/day) | 31.4 (20.1; 41.8) | 31.4 (20.7; 41.8) | 0.90 |
| Fruits (g/day) | 517.8 (301.3; 928.2) | 499.6 (286.4; 895.7) | 0.25 |
| Vegetables (g/day) | 202.4 (125.8; 310) | 196.3 (118.7; 306.2) | 0.23 |
| Legumes (g/day) | 30.2 (17.1; 45.6) | 27.6 (17.1; 45.4) | 0.34 |
| Cereals (g/day) | 239.2 (164.7; 363.6) | 230.8 (151.6; 350.2) | 0.08 |
| Fresh Fish (g/day) | 30.7 (17.1; 47.2) | 29.7 (16.6; 49) | 0.64 |
| Total Meat (g/day) | 66.1 (43.1; 99) | 65.6 (43.4; 100.8) | 0.67 |
| Dairy Products (g/day) | 201 (110.3; 287.5) | 188.7 (104; 279.5) | 0.17 |
| Biochemistry | |||
| ALT (U/L) | 12 (10; 16) | 17 (13; 24) | <0.0001 |
| AST (U/L) | 10 (9; 12) | 12 (10; 14) | <0.0001 |
| GGT (U/L) | 11 (8; 15) | 14 (11; 19) | <0.0001 |
| Bilirubin (mg/dL) | 0.82 (0.67; 0.99) | 0.79 (0.64; 1) | 0.08 |
| Glucose (mg/dL) | 100 (94; 108) | 107 (99; 118) | <0.0001 |
| HDL-C (mg/dL) | 52 (44; 63) | 44 (39; 53) | <0.0001 |
| LDL-C (mg/dL) | 119 (99; 141) | 124 (101; 146) | 0.008 |
| Triglycerides (mg/dL) | 88 (65; 125) | 137 (93; 188) | <0.0001 |
| Comorbidities | |||
| Hypertension yes n (%) | 470 (29) | 334 (41) | <0.0001 |
| SBP (mm Hg) | 120.8 ± 20 | 126.8 ± 18.6 | <0.0001 |
| DBP (mm Hg) | 73.2 ± 9.8 | 76.8 ± 9.9 | <0.0001 |
| Diabetes yes n (%) | 112 (7) | 116 (14) | <0.0001 |
| Units | OR * | C.I. | p-Value | ||
|---|---|---|---|---|---|
| BMI (kg/m2) | (1 kg/m2) | 1.257 | 1.228 | 1.287 | <0.0001 |
| EVOO (g/day) | 1 sd ≈ 20 g | 0.867 | 0.761 | 0.988 | 0.032 |
| Fruits (g/day) | 1 sd ≈ 570 g | 1.114 | 0.959 | 1.294 | 0.16 |
| Vegetables (g/day) | 1 sd ≈ 190 g | 0.961 | 0.857 | 1.078 | 0.49 |
| Legumes (g/day) | 1 sd ≈ 25 g | 0.928 | 0.831 | 1.035 | 0.18 |
| Cereals (g/day) | 1 sd ≈ 150 g | 1.325 | 1.044 | 1.681 | 0.02 |
| Fresh Fish (g/day) | 1 sd ≈ 30 g | 1.035 | 0.927 | 1.155 | 0.54 |
| Total Meat (g/day) | 1 sd ≈ 54 g | 1.062 | 0.937 | 1.204 | 0.34 |
| Milk and Dairy (g/day) | 1 sd ≈ 150 g | 1.063 | 0.945 | 1.197 | 0.30 |
| Total kilocalories | (unit 500 kcal) | 0.810 | 0.655 | 1.002 | 0.052 |
| Model 1 * | Model 2 ** | |||||||
|---|---|---|---|---|---|---|---|---|
| OR | C.I. | p-Value | OR | C.I. | p-Value | |||
| continuous EVOO § | 0.792 | 0.710 | 0.880 | <0.0001 | 0.833 | 0.743 | 0.934 | 0.0018 |
| low consumption (ref.) | 1 | - | - | - | 1 | - | - | - |
| moderate consumption | 0.835 | 0.660 | 1.060 | 0.139 | 0.891 | 0.690 | 1.151 | 0.37 |
| high consumption | 0.658 | 0.510 | 0.860 | 0.002 | 0.735 | 0.555 | 0.974 | 0.032 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tedesco, C.C.; Bonfiglio, C.; Notarnicola, M.; Rendina, M.; Castellaneta, A.; Di Leo, A.; Giannelli, G.; Fontana, L. High Extra Virgin Olive Oil Consumption Is Linked to a Lower Prevalence of NAFLD with a Prominent Effect in Obese Subjects: Results from the MICOL Study. Nutrients 2023, 15, 4673. https://doi.org/10.3390/nu15214673
Tedesco CC, Bonfiglio C, Notarnicola M, Rendina M, Castellaneta A, Di Leo A, Giannelli G, Fontana L. High Extra Virgin Olive Oil Consumption Is Linked to a Lower Prevalence of NAFLD with a Prominent Effect in Obese Subjects: Results from the MICOL Study. Nutrients. 2023; 15(21):4673. https://doi.org/10.3390/nu15214673
Chicago/Turabian StyleTedesco, Calogero Claudio., Caterina Bonfiglio, Maria Notarnicola, Maria Rendina, Antonino Castellaneta, Alfredo Di Leo, Gianluigi Giannelli, and Luigi Fontana. 2023. "High Extra Virgin Olive Oil Consumption Is Linked to a Lower Prevalence of NAFLD with a Prominent Effect in Obese Subjects: Results from the MICOL Study" Nutrients 15, no. 21: 4673. https://doi.org/10.3390/nu15214673
APA StyleTedesco, C. C., Bonfiglio, C., Notarnicola, M., Rendina, M., Castellaneta, A., Di Leo, A., Giannelli, G., & Fontana, L. (2023). High Extra Virgin Olive Oil Consumption Is Linked to a Lower Prevalence of NAFLD with a Prominent Effect in Obese Subjects: Results from the MICOL Study. Nutrients, 15(21), 4673. https://doi.org/10.3390/nu15214673








