A Randomized Controlled Trial of Changes in Fluid Distribution across Menstrual Phases with Creatine Supplementation
Abstract
1. Introduction
2. Materials and Methods
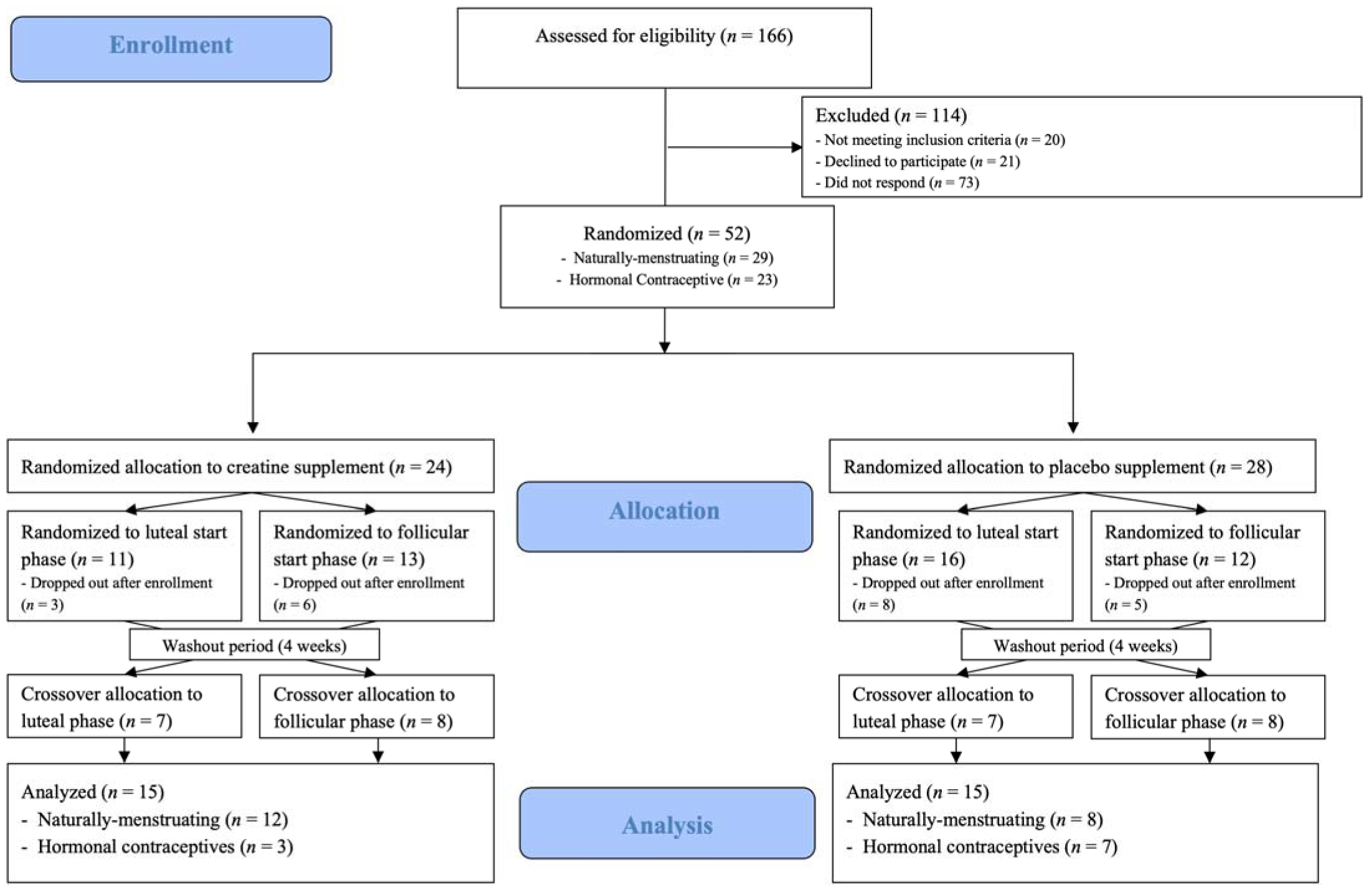
2.1. Subjects
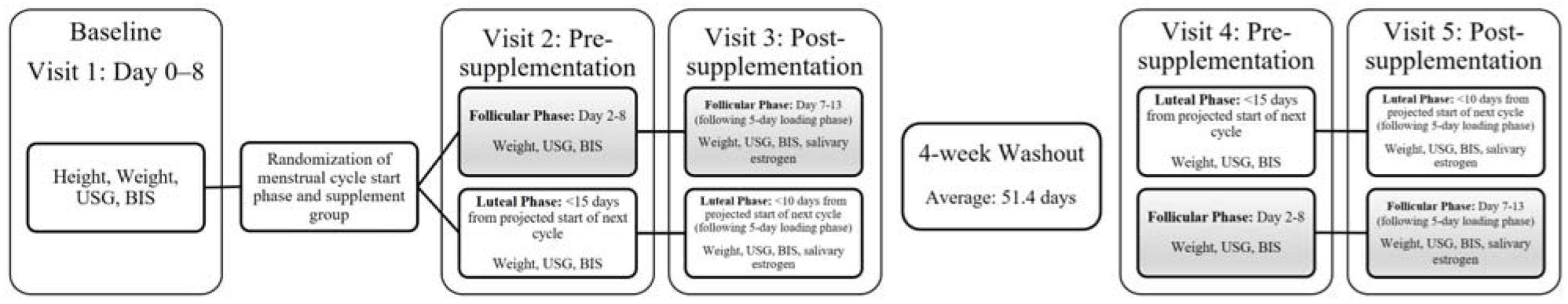
2.2. Experimental Design
2.3. Bioelectrical Impedance Spectroscopy (BIS)
2.4. Hormonal Status
2.5. Supplementation
2.6. Statistical Analysis
3. Results
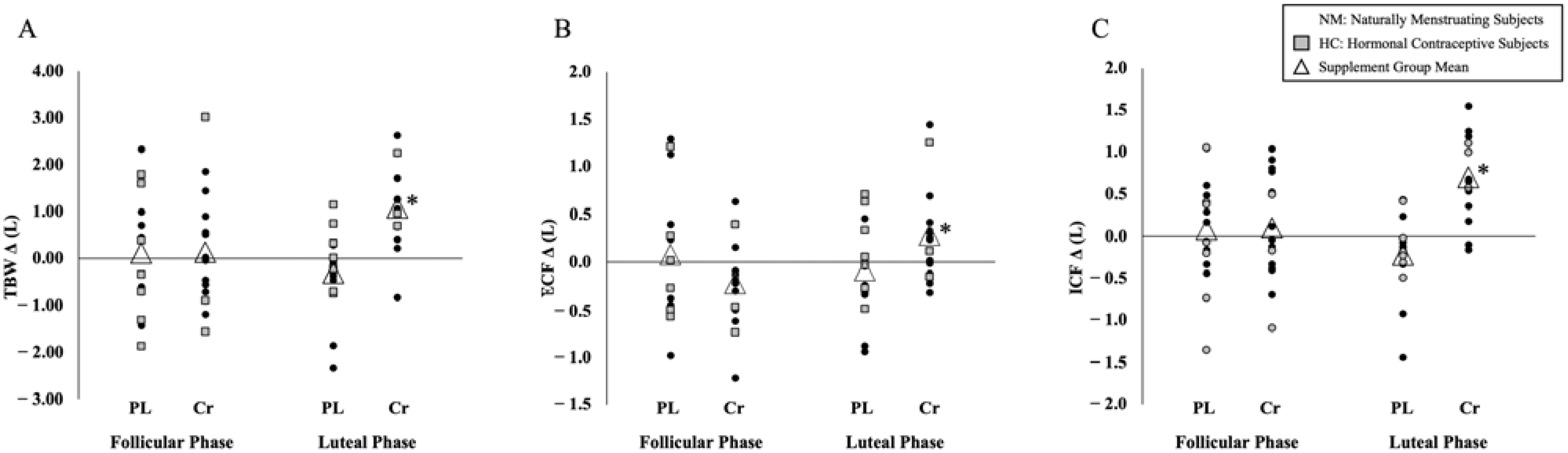
3.1. TBW
3.2. ECF
3.3. ICF
4. Discussion
4.1. TBW
4.2. ECF
4.3. ICF
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smith, E.S.; McKay, A.K.A.; Kuikman, M.; Ackerman, K.E.; Harris, R.; Elliott-Sale, K.J.; Stellingwerff, T.; Burke, L.M. Auditing the Representation of Female Versus Male Athletes in Sports Science and Sports Medicine Research: Evidence-Based Performance Supplements. Nutrients 2022, 14, 953. [Google Scholar] [CrossRef] [PubMed]
- Antonio, J.; Candow, D.G.; Forbes, S.C.; Gualano, B.; Jagim, A.R.; Kreider, R.B.; Rawson, E.S.; Smith-Ryan, A.E.; VanDusseldorp, T.A.; Willoughby, D.S.; et al. Common questions and misconceptions about creatine supplementation: What does the scientific evidence really show? J. Int. Soc. Sports Nutr. 2021, 18, 13. [Google Scholar] [CrossRef]
- Smith-Ryan, A.; Cabre, H.; Eckerson, J.; Candow, D. Creatine Supplementation in Women’s Health: A Lifespan Perspective. Nutrients 2021, 13, 877. [Google Scholar] [CrossRef] [PubMed]
- Benton, M.J.; Spicher, J.M.; McCormick, S. Community-Based Survey Exploring Use of the Dietary Supplement Creatine by Adult Non-Athletes. Nutrients 2021, 13, 2529. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 18. [Google Scholar] [CrossRef]
- Naderi, A.; Earnest, C.P.; Lowery, R.P.; Wilson, J.M.; Willems, M.E.T. Co-ingestion of Nutritional Ergogenic Aids and High-Intensity Exercise Performance. Sport. Med. 2016, 46, 1407–1418. [Google Scholar] [CrossRef]
- Lyoo, I.K.; Yoon, S.; Kim, T.-S.; Hwang, J.; Kim, J.E.; Won, W.; Bae, S.; Renshaw, P.F. A Randomized, Double-Blind Placebo-Controlled Trial of Oral Creatine Monohydrate Augmentation for Enhanced Response to a Selective Serotonin Reuptake Inhibitor in Women with Major Depressive Disorder. Am. J. Psychiatry 2012, 169, 937–945. [Google Scholar] [CrossRef]
- Avgerinos, K.I.; Spyrou, N.; Bougioukas, K.I.; Kapogiannis, D. Effects of creatine supplementation on cognitive function of healthy individuals: A systematic review of randomized controlled trials. Exp. Gerontol. 2018, 108, 166–173. [Google Scholar] [CrossRef]
- Benton, D.; Donohoe, R. The influence of creatine supplementation on the cognitive functioning of vegetarians and omnivores. Br. J. Nutr. 2010, 105, 1100–1105. [Google Scholar] [CrossRef]
- Handy, A.B.; Greenfield, S.F.; Yonkers, K.A.; Payne, L.A. Psychiatric Symptoms across the Menstrual Cycle in Adult Women: A Comprehensive Review. Harv. Rev. Psychiatry 2022, 30, 100–117. [Google Scholar] [CrossRef]
- Ali, S.A.; Begum, T.; Reza, F. Hormonal Influences on Cognitive Function. Malays. J. Med Sci. 2018, 25, 31–41. [Google Scholar] [CrossRef]
- Dalbo, V.J.; Roberts, M.D.; Stout, J.R.; Kerksick, C. Putting to rest the myth of creatine supplementation leading to muscle cramps and dehydration. Br. J. Sport. Med. 2008, 42, 567–573. [Google Scholar] [CrossRef]
- Chang, C.-T.; Wu, C.-H.; Yang, C.-W.; Huang, J.-Y.; Wu, M.-S. Creatine Monohydrate Treatment Alle-viates Muscle Cramps Associated with Haemodialysis. Nephrol. Dial. Transpl. 2002, 17, 1978–1981. [Google Scholar] [CrossRef] [PubMed]
- Sobolewski, E.J.; Thompson, B.J.; Smith, A.E.; Ryan, E.D. The Physiological Effects of Creatine Supple-mentation on Hydration: A Review. Am. J. Lifestyle Med. 2011, 5, 320–327. [Google Scholar] [CrossRef]
- Deminice, R.; Rosa, F.T.; Pfrimer, K.; Ferrioli, E.; Jordao, A.A.; Freitas, E. Creatine Supplementation Increases Total Body Water in Soccer Players: A Deuterium Oxide Dilution Study. Int. J. Sport. Med. 2015, 37, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.W.; Chuter, V.H.; Silk, L.N.; Thom, J.M. Exercise Performance over the Menstrual Cycle in Temperate and Hot, Humid Conditions. Med. Sci. Sport. Exerc. 2012, 44, 2190–2198. [Google Scholar] [CrossRef]
- Lopez, R.M.; Casa, D.J.; McDermott, B.P.; Ganio, M.S.; Armstrong, L.E.; Maresh, C.M. Does Creatine Supplementation Hinder Exercise Heat Tolerance or Hydration Status? A Systematic Review with Meta-Analyses. J. Athl. Train. 2009, 44, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Powers, M.E.; Arnold, B.L.; Weltman, A.L.; Perrin, D.H.; Mistry, D.; Kahler, D.M.; Kraemer, W.; Volek, J. Creatine Supplementation Increases Total Body Water without Altering Fluid Distribution. J. Athl. Train. 2003, 38, 44–50. [Google Scholar]
- Rosene, J.M.; Whitman, S.A.; Fogarty, T.D. A Comparison of Thermoregulation with Creatine Sup-plementation between the Sexes in a Thermoneutral Environment. J. Athl. Train. 2004, 39, 50–55. [Google Scholar]
- Elliott-Sale, K.J.; Minahan, C.L.; De Jonge, X.A.K.J.; Ackerman, K.E.; Sipilä, S.; Constantini, N.W.; Lebrun, C.M.; Hackney, A.C. Methodological Considerations for Studies in Sport and Exercise Science with Women as Participants: A Working Guide for Standards of Practice for Research on Women. Sport. Med. 2021, 51, 843–861. [Google Scholar] [CrossRef]
- Weiss, C.; Powers, M. Creatine Supplementation Does Not Impair the Thermoregulatory Response during a Bout of Exercise in the Heat. J. Sport. Med. Phys. Fit. 2006, 46, 555. [Google Scholar]
- Kutz, M.R.; Gunter, M.J. Creatine Monohydrate Supplementation on Body Weight and Percent Body Fat. Natl. Strength Cond. Assoc. J. Strength Cond. Res. 2003, 17, 817–821. [Google Scholar] [CrossRef]
- Francaux, M.; Poortmans, J.R. Effects of training and creatine supplement on muscle strength and body mass. Eur. J. Appl. Physiol. 1999, 80, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Kamal, E.M.; Hafez, A.M. Effect of copper intrauterine device vs. injectable contraceptive on serum hormone levels and cell mitotic activity in endometrium. Middle East Fertil. Soc. J. 2013, 18, 142–146. [Google Scholar] [CrossRef]
- Almeida, D.; Colombini, A.; Machado, M. Creatine supplementation improves performance, but is it safe? Double-blind placebo-controlled study. J. Sport. Med. Phys. Fit. 2020, 60, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Giersch, G.E.; Morrissey, M.C.; Katch, R.K.; Colburn, A.T.; Sims, S.T.; Stachenfeld, N.S.; Casa, D.J. Menstrual cycle and thermoregulation during exercise in the heat: A systematic review and meta-analysis. J. Sci. Med. Sport 2020, 23, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Kilduff, L.; Georgiades, E.; James, N.; Minnion, R.; Mitchell, M.; Kiingsmore, D.; Hadjicharlambous, M.; Pitsiladis, Y. The Effects of Creatine Supplementation on Cardiovascular, Metabolic, and Thermoreg-ulatory Responses during Exercise in the Heat in Endurance-Trained Humans. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 443–460. [Google Scholar] [CrossRef]
- Mendel, R.W.; Blegen, M.; Cheatham, C.; Antonio, J.; Ziegenfuss, T. Effects of creatine on thermoregulatory responses while exercising in the heat. Nutrition 2005, 21, 301–307. [Google Scholar] [CrossRef]
- Hetherington-Rauth, M.; Baptista, F.; Sardinha, L. BIA-assessed cellular hydration and muscle performance in youth, adults, and older adults. Clin. Nutr. 2019, 39, 2624–2630. [Google Scholar] [CrossRef]
- Ribeiro, A.S.; Avelar, A.; Kassiano, W.; Nunes, J.P.; Schoenfeld, B.J.; Aguiar, A.F.; Trindade, M.C.; Silva, A.M.; Sardinha, L.B.; Cyrino, E.S. Creatine Supplementation Does Not Influence the Ratio between Intracellular Water and Skeletal Muscle Mass in Resistance-Trained Men. Int. J. Sport Nutr. Exerc. Metab. 2020, 30, 405–411. [Google Scholar] [CrossRef]
- Ziegenfuss, T.N.; Lowery, L.M.; Lemon, P.W.R. Nutrition and Exercise Acute Fluid Volume Changes in Men during Three Days of Creatine Supplementation. An International Electronic. J. Exerc. Physiol. 1998, 1, 1–9. [Google Scholar]
- Stachenfeld, N.S. Sex Hormone Effects on Body Fluid Regulation. Exerc. Sport Sci. Rev. 2008, 36, 152–159. [Google Scholar] [CrossRef]
- Gould, L.M.; Cabre, H.E.; Brewer, G.J.; Hirsch, K.R.; Blue, M.N.; Smith-Ryan, A.E. Impact of Follicular Menstrual Phase on Body Composition Measures and Resting Metabolism. Med. Sci. Sport. Exerc. 2021, 53, 2396–2404. [Google Scholar] [CrossRef]
- Mitchell, C.; Rose, J.; Familoni, B.; Winters, S.; Ling, F. The Use of Multifrequency Bioelectrical Im-pedance Analysis to Estimate Fluid Volume Changes as a Function of the Menstrual Cycle. Basic Life Sci. 1993, 60, 189–191. [Google Scholar]
- Stachenfeld, N.S.; Taylor, H.S. Effects of estrogen and progesterone administration on extracellular fluid. J. Appl. Physiol. 2004, 96, 1011–1018. [Google Scholar] [CrossRef]
- Wickham, K.A.; McCarthy, D.G.; Spriet, L.L.; Cheung, S.S. Sex differences in the physiological responses to exercise-induced dehydration: Consequences and mechanisms. J. Appl. Physiol. 2021, 131, 504–510. [Google Scholar] [CrossRef]
- González-Alonso, J.; Mora-Rodríguez, R.; Below, P.R.; Coyle, E.F. Dehydration Markedly Impairs Cardiovascular Function in Hyperthermic Endurance Athletes during Exercise. J. Appl. Physiol. 1997, 82, 1229–1236. [Google Scholar] [PubMed]
- Sawka, M.N.; Noakes, T.D. Does Dehydration Impair Exercise Performance? Med. Sci. Sport. Exerc. 2007, 39, 1209–1217. [Google Scholar] [CrossRef] [PubMed]

| Supplement Group | Age (years) | Height (cm) | Weight (kg) | BMI (kg/m2) |
|---|---|---|---|---|
| PL (n = 15) | 24.5 ± 4.6 | 167.4 ± 4.6 | 65.0 ± 8.2 | 23.1 ± 1.8 |
| Cr (n = 15) | 25.4 ± 7.2 | 164.1 ± 5.8 | 65.6 ± 9.2 | 23.6 ± 2.9 |
| FP Pre | FP Post | LP Pre | LP Post | |
|---|---|---|---|---|
| BM (kg) | PL: 66.14 ± 6.29 Cr: 65.52 ± 9.42 | 66.27 ± 6.30 65.23 ± 9.41 | 66.32 ± 6.19 65.21 ± 9.38 | 66.05 ± 6.16 65.76 ± 9.41 |
| TBW (L) | PL: 34.80 ± 3.36 Cr: 35.27 ± 5.95 | 34.91 ± 3.09 35.89 ± 5.41 | 34.85 ± 2.87 35.22 ± 5.29 | 34.54 ± 2.85 36.30 ± 5.35 * |
| ECF (L) | PL: 13.98 ± 1.23 Cr: 14.40 ± 1.92 | 13.39 ± 2.71 14.49 ± 2.28 | 14.00 ± 0.99 14.08 ± 2.07 | 13.91 ± 0.98 14.36 ± 2.02 * |
| ICF (L) | PL: 20.85 ± 2.23 Cr: 21.65 ± 3.51 | 20.93 ± 2.07 21.71 ± 3.49 | 20.85 ± 2.01 21.14 ± 3.28 | 20.62 ± 1.95 21.84 ± 3.32 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moore, S.R.; Gordon, A.N.; Cabre, H.E.; Hackney, A.C.; Smith-Ryan, A.E. A Randomized Controlled Trial of Changes in Fluid Distribution across Menstrual Phases with Creatine Supplementation. Nutrients 2023, 15, 429. https://doi.org/10.3390/nu15020429
Moore SR, Gordon AN, Cabre HE, Hackney AC, Smith-Ryan AE. A Randomized Controlled Trial of Changes in Fluid Distribution across Menstrual Phases with Creatine Supplementation. Nutrients. 2023; 15(2):429. https://doi.org/10.3390/nu15020429
Chicago/Turabian StyleMoore, Sam R., Amanda N. Gordon, Hannah E. Cabre, Anthony C. Hackney, and Abbie E. Smith-Ryan. 2023. "A Randomized Controlled Trial of Changes in Fluid Distribution across Menstrual Phases with Creatine Supplementation" Nutrients 15, no. 2: 429. https://doi.org/10.3390/nu15020429
APA StyleMoore, S. R., Gordon, A. N., Cabre, H. E., Hackney, A. C., & Smith-Ryan, A. E. (2023). A Randomized Controlled Trial of Changes in Fluid Distribution across Menstrual Phases with Creatine Supplementation. Nutrients, 15(2), 429. https://doi.org/10.3390/nu15020429






