Abstract
Arterial stiffness, an age-dependent phenomenon, is improved with exercise, which in turn may prevent cardiovascular diseases in women. However, there is a lack of consolidated information on the impact of exercise on arterial stiffness among healthy women. The aim of this review was to (i) analyse the effect of exercise on arterial stiffness in healthy young, middle-aged, and older women, and (ii) recommend types, intensity, and frequency for each age group. Database searches on PubMed, ScienceDirect, Web of Science, and Scopus were conducted using PRISMA guidelines until September 2022. The keywords were: exercise, women/female, and arterial stiffness. The inclusion criteria were: healthy women, supervised exercise, and arterial stiffness measures. Study quality and bias were assessed using the PEDro scale. Fifty-one papers were classified into young (n = 15), middle-aged (n = 14), and older (n = 22) women. Improvements in arterial stiffness were observed among: young women (Pulse Wave Velocity, PWV: 4.9–6.6 m/s), following an 8-week high-intensity aerobic (3 days/week) or hypoxic high-intensity interval training; middle-aged women (PWV: 5.1–7.9 m/s), aerobic exercise with moderate intensity or stretching exercise at “moderate to heavy” (Borg Scale), 20–30 s per site, 10 s of rest interval for 30 min; and for older women (PWV: 7.9–15.6 m/s), resistance training at light intensity, aerobic exercise at any intensity, or a combination of the two exercises. This review shows that arterial stiffness increases with age in healthy women and has an inverse relationship with exercise intensity. Therefore, when prescribing exercise to improve arterial stiffness, age and arterial stiffness measures should be accounted for.
1. Introduction
Arterial stiffness describes rigidity in the arterial wall, which has been accepted as an independent risk factor for cardiovascular morbidity and mortality [1]. Increased arterial stiffness is also highly associated with an increased risk of cardiovascular diseases (CVDs), such as hypertension [2], chronic kidney disease [3], and stroke [4]. The risk of CVDs in women is often underestimated due to the misconception that women are more “protected” than men [5] and is, hence, understudied, under-recognised, underdiagnosed, undertreated, and under-represented [6]. The Lancet reported that 35% of deaths in women worldwide are caused by CVDs [6], with a staggering 275 million women diagnosed, while 8.9 million died from CVDs in 2019. The prevalence of high arterial stiffness was 10.6% for ages under 29 years, 40.1% for ages 30–49 years, and 91.5% for ages over 50 years [7]. Thus, to delay the development of atherosclerotic CVDs, prevention or containment of risk factors that accelerate arterial stiffness should be in place [8], considering that the etiological factors contributing to arterial stiffness among women in different age groups may differ [9]. Exercise is the most common modality in treating and preventing arterial stiffness [10]. Therefore, exercise prescriptions should consider the etiological factors of women at different ages to improve arterial stiffness more effectively.
Arterial stiffness is an unavoidable physiological process. It is difficult to determine which factors contribute to the progression of arterial stiffness. Therefore, the current interventions for arterial stiffness suffer from a lack of targeting. This may be the reason why medical treatment for the progression of arterial stiffness is modestly effective [3]. In fact, specific populations, such as young women, middle-aged women, and older women, have specific factors associated with arterial stiffness. Oestrogen is thought to play an important role in the female-specific changes in arterial stiffness [9]. In adult women, overall oestrogen levels reach a peak in the late 20s. Then oestrogen levels begin to decline at 30 years of age until menopause. Menopause is a milestone in female ageing- it means that oestrogen levels will remain at low levels [11]. Under this premise, young women are in a period of oestrogen upswing, middle-aged women are mainly in a period of oestrogen downturn, and older women are in a period of low oestrogen levels. Oestrogen could reduce arterial stiffness through increased nitric oxide (NO) [12]. In addition, norepinephrine, which can drive vascular smooth cell (VSMC) contraction, is positively correlated with age in women [13]. VSMCs are most abundant cell types in vessel walls and have emerged as key players in arterial stiffness [14]. Furthermore, the advanced glycation end product (AGE) accumulation hastens multisystem functional decline with age [15]. AGEs are a non-enzymatic process involving glycation of sugars or reactive oxoaldehyde with proteins, lipids, or nucleic acids. AGEs promote arterial stiffness through the upregulation of inflammation and collagen cross-linking [16], which, in turn, leads to endothelial dysfunction and subsequently increases vascular stiffness. Exercise can modulate these etiological factors and, thus, improve arterial stiffness. However, due to the different characteristics of these etiological factors in women of different ages, it is possible that similar exercise prescriptions may have different effects on arterial stiffness. Incidentally, there are studies that have tentatively confirmed this speculation. For example, an incremental exercise program to exhaustion using Bruce protocol does not modify arterial stiffness in young women [17] but increases arterial stiffness in middle-aged women [18]. In addition, resistance training at 80% 1-RM showed positive effects on arterial stiffness in young women [19] but no change in arterial stiffness in middle-aged and older women [20,21]. Considering the etiological factors and impact of exercise (types, intensity, and frequency), however, to date, no review has been conducted on the effects of exercise on arterial stiffness in healthy young, middle-aged, and older women.
The purpose of this review was to explore the current literature on the effects of exercise on arterial stiffness in healthy young, middle-aged, and older women, to provide guidance on exercise for the prevention of cardiovascular disease in women and propose suggestions for further research.
2. Methods
2.1. Data Selection
This systemic review was conducted in accordance with the Preferred Reporting Items for Systemic Reviews and Meta-analyses (PRISMA) Guideline [22]. The journal articles were searched electronically using the following databases: PubMed, ScienceDirect, Web of Science, and Scopus. Peer-reviewed articles published in English until 30th September 2022 were reviewed. No contact with the studies’ authors was made. The search strategy used in Web of Science is displayed in Table 1, and a similar strategy was used to search the other databases.

Table 1.
Search strategy for Web of Science.
2.2. Studies Selection
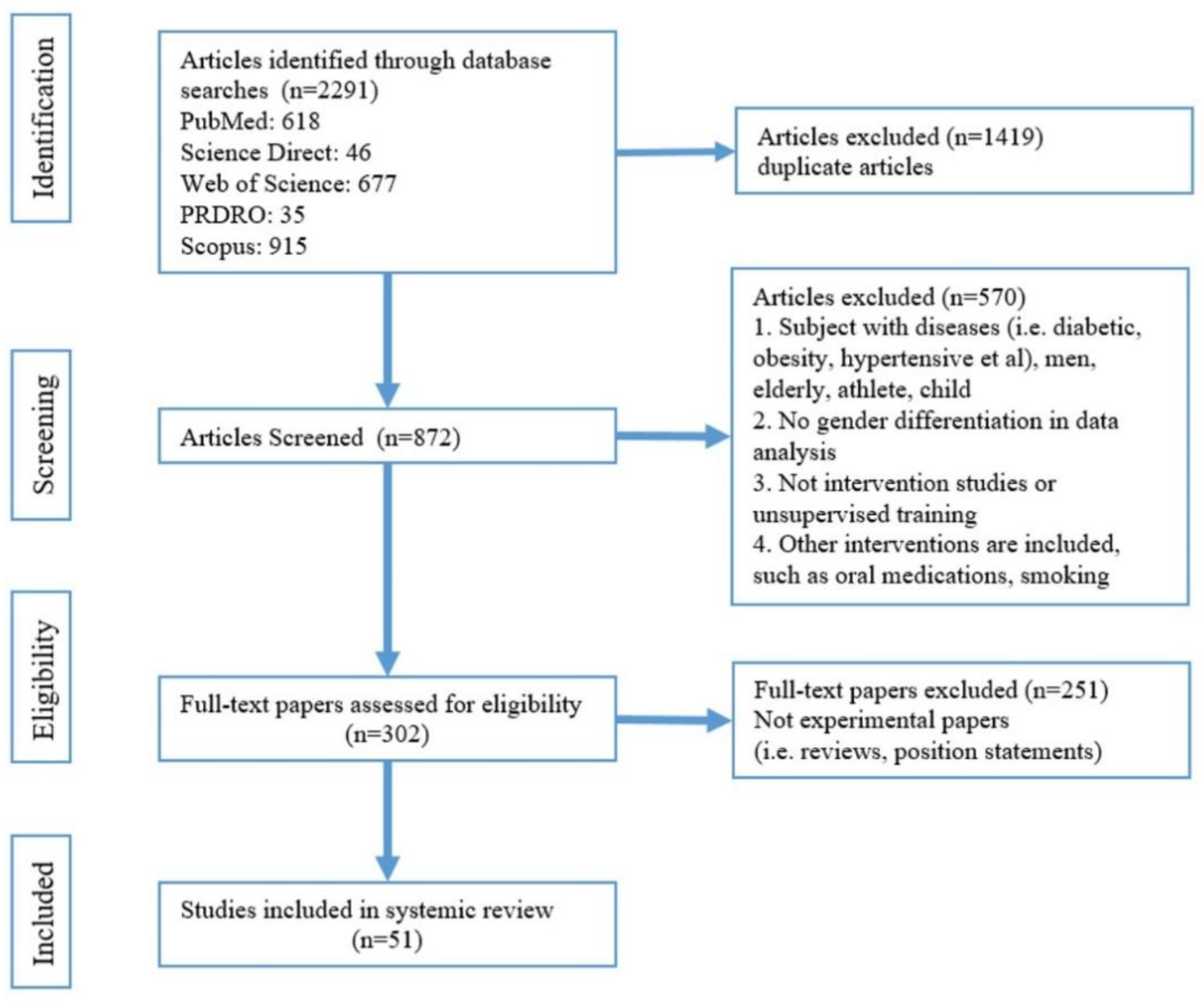
Figure 1 shows a flow diagram of the search. The articles were selected for review after thorough screening and exclusion of ineligible articles. The inclusion criteria were as follows: exercise was the only intervention and must be under supervision; arterial stiffness assessment must be included; subjects were healthy women, without hypertension, diabetes, obesity, or pregnancy; statistics must be gender-specific and a research paper. Then, all the selected papers were categorised into a young-women group (Table 2), a middle-aged-women group (Table 3), and older-women group (Table 4). Women between 15 and 24 years of age are defined as young age [23], those aged more than 60 years or postmenopausal are defined as older women [24]. Women between the ages of young and older are middle-aged women.
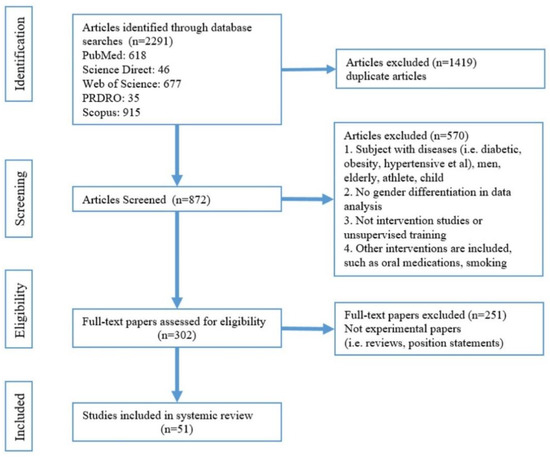
Figure 1.
Flowchart for study selection.

Table 2.
Characteristics of selected experimental trials on young women (n = 15).

Table 3.
Characteristics of selected experimental trials in middle-aged women (n = 14).

Table 4.
Characteristics of selected experimental trials in old women (n = 22).
2.3. Data Extraction and Studies Methodological Quality
The titles and/or abstracts of all articles in the database were reviewed to assess the requirement for full text based on the study selection criteria. Each full-text paper was evaluated systematically according to: (a) subject characteristics, (b) exercise volume (frequency, intensity, time, mode, etc.), and (c) main findings. Characteristics of the included studies for young, middle-aged, and old women are presented in Table 2, Table 3 and Table 4, respectively.
The quality assessment was performed using the PEDro Scale. Two reviewers independently assessed the articles and average score from both assessments was used. The 51 studies averaged a score of 7.96, with four studies scoring less than 7, mainly due to the absence of a double-blind design.
3. Results and Discussions
This systematic review aims at elucidating the effect of exercise on arterial stiffness measures (PWV, elastance) among young (18–24 years), middle-aged (25–49 years), and older (50 and above) women. Evidence from the retrieved studies (n = 51) suggests that exercise can be an effective way to improve arterial stiffness in women. In young and middle-aged women, high-intensity aerobic exercise and stretching at “somewhat heavy” to “heavy” based on the Borg Scale are recommended, while in older women, aerobic exercise at any intensity, resistance exercise at low intensity, combined aerobic and resistance exercise, and stretching and vibration training can all reduce arterial stiffness. In addition, this review also confirms that arterial stiffness is age-dependent in women. The pulse wave velocity (PWV), a wide measure of arterial stiffness, at rest ranges from 4.9–6.6 m/s and 5.1–7.9 m/s to 7.9–15.6 m/s in young, middle-aged, and elderly women, respectively. The values seem to increase slightly in middle-aged women whilst showing a sharp increase in older women compared to younger. These may influence the effect of exercise on arterial stiffness, where improvements in PWV are 7 and 8.7% in young and middle-aged women, respectively [35,42], while 12.5% in older women [51]. It appears that the effect of exercise is dependent on the severity of the arterial stiffness, which is, in turn, age-dependent. Factors, such as exercise prescription, i.e., type, intensity, duration, and frequency, are also major determinants for the said effects.
3.1. Defining Exercise Intensity
Exercise intensity is the primary factor in exercise prescription. Exercise intensities in this review are classified using HRmax, HRR, RM, METS, and VO2max. For HRmax: 50–63% is classified as light intensity; 64–76% as moderate intensity; and 77–93% as high intensity [70]. For HRR, light intensity and moderate intensity are defined as 30 and 50% HRR, respectively, while high intensity is defined as more than 50% HRR [71]. For RM, less than 67% of 1 RM or more than 12 RM is classified as low intensity [72], 67–85% of 1 RM or 6–12 RM as moderate intensity, while more than 85% of 1 RM or less than 6 RM is classified as high intensity. For METS, light intensity is less than 3 METs; moderate intensity ranges from 3 to 6 METs; and high intensity is more than 6 METs [73]. For VO2max, less than 51% is defined as light intensity; between 52 and 67% is defined as moderate intensity; higher than 67% is defined as high intensity [74].
3.2. Effect of Exercise on Arterial Stiffness in Young Women
Among young women, five different types of exercises, aerobic, HIIT, resistance training (RT), anaerobic, and stretching exercises have been reported in 15 studies. However, only two studies have shown the positive effects of aerobic (running, cycling, or elliptical) [35] and hypoxic HIIT-running [25] exercises on arterial stiffness (about 7% improvements). In these studies, moderate–high intensity was based on HRmax and HRR respectively. The lack of changes in other studies [17,29,34], which also employed aerobic exercises, could be due to (i) vascular physiology, (ii) the acute nature of exercise intervention compared to 3 days/week for 8 weeks in Lane et al. [35] and 6 weeks in Park et al. [25], and (iii) environment (hypoxic) [25]. On the other hand, while RT is the most widely studied type of exercise [21,28,30,31,32,33,37], no changes in arterial stiffness were reported. In these studies, moderate–high intensity based on RM was employed. Adversely, acute-effect moderate–high intensity RT has shown a transient increase in PWV [28,31,32,33]. The increase in arterial stiffness due to resistance training may be due to increased levels of catecholamines and activation of the sympathetic nervous system [75]. Catecholamine levels and the sympathetic nervous system can increase vascular smooth muscle tone and, thus, vascular stiffness through humoral and neural regulation, respectively [75].
Based on the evidence presented, to observed changes in arterial stiffness among young women, at least 3 days/week of 8-week high-intensity aerobic (running or cycling) or hypoxic HIIT training are recommended, while acute aerobic or resistance-type exercise is not recommended.
3.3. Effect of Exercise on Arterial Stiffness in Middle-Aged Women
Among middle-aged women, four different types of exercises, including aerobic, RT, stretching exercise, and myofascial release exercises, are reported in 14 studies. For aerobic exercise, out of four, three studies [18,40,46,47] showed positive effects on arterial stiffness (about 23.27% improvement). It appears that acute high-intensity running exercise based on HR and HRR [18,40] and prolonged moderate cycling exercise based on VO2max with 2 days/week for 12 weeks [46] are beneficial for middle-aged women. One study [47] did not observe any change following a 14-week high-intensity cycling exercise based on HRR, which could be due to (i) study design (cross-over with insufficient wash-out period and small sample size), (ii) excess intensity, and (iii) low mean age. The three studies which showed a positive effect from aerobic exercise on arterial stiffness in middle-aged women are at moderate–high intensities. In addition, no adverse effect of aerobic exercise on arterial stiffness was reported in middle-aged women.
Meanwhile, for RT, which is the most commonly studied type of exercise among middle-aged women, there are two studies [41,45] that reported positive effects on arterial stiffness in middle-aged women (about 10.97% improvement), while five others do not [20,38,39,46,48]. The lack of changes following RT in this age group could be due to multifactorial reasons, (i) exercise intensity, (ii) mode of contraction, and (iii) muscle group tested. It seems that acute low–moderate RM (Augustine et al., 2018) and prolonged training (2 days/week for 10-week; in Okamoto et al. 2009), involving major muscle groups (upper limb) and using isotonic contraction, are important factors to consider in prescribing exercise to middle-aged women.
Although low–moderate intensity RT based on RM and body weight showed a positive effect on arterial stiffness [41,45], several other studies reported that light-intensity RT based on RM has either a detrimental effect or no effect on vascular health [38,46,48], while high-intensity RT with maximal isokinetic knee extension/flexion also has a negative effect on arterial stiffness [39] in middle-aged women. In addition, to a lesser extent, the menstrual cycle could also influence the effect of exercise on arterial stiffness among middle-aged women [41].
While for stretching exercise, two studies [42,43] showed positive results (about 9.2% improvement), one study does not report positive changes [44]. The lack of change could be associated with low stretching activity, stretching duration per site, and interval of stretching may be a key factor influencing the change in arterial stiffness. Based on the positive findings [42,43], stretching intensity of “somewhat heavy” to “heavy” in Borg Scale 7 days/week for 6 months [43], stretching exercise with 20–30 s per site, and 10 s of rest interval for 30 min [42] may be helpful for vascular health among middle-aged women.
In brief, the most preferable type of exercise is aerobic exercise at moderate–high-intensity running or cycling, acute or prolonged (2 days/week for 12 weeks), while for RT, low–moderate-intensity acute or prolonged (2 days/week for 10 weeks) involving major muscle groups and using the isotonic mode of contraction, and for stretching exercise, acute 30 min or prolonged at “somewhat heavy” to “heavy” based on Borg Scale seem beneficial to improve arterial stiffness among middle-aged women.
3.4. Effect of Exercise on Arterial Stiffness in Older Women
Among older women, there are a total of 22 studies on the effect of exercise on arterial stiffness, the highest number compared to young women and middle-aged women. The most common types of exercise reported are aerobic exercises (walking, running, cycling, aquarobics, Taichi, steps), prolonged aerobic [51,52,53,56,57,58,60,62,63,64,66,68,69] and acute aerobic [56,64], resistance training [21,50,54,55,61,68], combined resistance training and aerobic exercise [49,59,65,67], flexibility training [61], and vibration training [50]. The majority of the studies (17) showed a positive effect of exercise on arterial stiffness in older women. More importantly, no adverse effect of exercise was reported in the studies. Hence, the types of exercises listed (excluding vibration training) can be recommended for older women depending on their ability.
For aerobic exercise, there are 16 studies listed, with all reporting positive changes in arterial stiffness among older women (about 18.46% improvement). Out of these, 14 studies prescribed prolonged exercises, while 2 prescribed acute types. The intensity of the aerobic exercise ranges from low to moderate to high based on HRmax (60–80%), HRR (40–80%), METS (1.5–3.0), VO2peak (50%), and at lactate threshold, while the duration of the intervention ranges from 8 to 18 weeks with 1–3 days/week in most studies. Meanwhile, two studies reported positive changes following acute aerobic exercise. The intensity of these exercises ranges from moderate to high based on self-paced walk (1 h walk) [56] and anaerobic threshold cycling (90%) [64]. Based on the evidence presented, it is suggested that for a long-term effect, any intensity, 8–18 weeks, 1–3 days/week, while for the acute effect of aerobic exercise, 20–90 min moderate to high intensity can be considered as the ideal type of aerobic exercise (walking, running, cycling) to be prescribed for older women.
For RT, vascular stiffness improvements in older women were observed following low-intensity exercises based on RM (about 0.85% improvement) [50,61]. What’s more, no studies have found a detrimental effect of low-intensity resistance training on the vascular health of older women as typically seen in middle-aged women. Therefore, older women can consider low-resistance training based on their ability. In addition, some researchers found that combining low-resistance training and aerobic exercise also showed a beneficial effect on vascular stiffness in older women [49,50].
For stretching exercise, 2 days/week for 16 weeks performing a whole-body static stretching program may be helpful for vascular health among middle-aged women. For vibration exercise, dynamic leg exercises (full squats, high squats, wide squats, and calf raises) on a vibrating platform with 24–40 Hertz intensity for 12 weeks showed no change in arterial stiffness. Therefore, it is difficult to provide this exercise program for this age group.
In conclusion, the most preferable type of exercise is aerobic exercise, such as prolonged swimming, rotating exercise, chair-based exercise, walking, cycling, Taichi, steps with low–moderate–high intensity (1–3 days/week, 8–18 weeks), or acute walking/cycling with moderate–high intensity (20–90 min). For RT, low intensity or low-intensity RT combined with aerobic exercise, and for stretching exercise, 2 days/week for 16 weeks involving a whole-body static stretching program seem beneficial to improve arterial stiffness among older women.
3.5. Possible Mechanisms Underlying the Effect of Exercise on Arterial Stiffness in Women
Acute and long-term exercises have shown beneficial effects on arterial stiffness in women in general. However, the changes following acute exercise may only be temporary, as arterial stiffness decreased significantly after acute exercise and would return to basal levels 24 h later [76,77,78]. These temporary changes may be exclusive to improved vascular function rather than structural adaptations [79,80]. Acute exercise increases blood flow and shear stress, which leads to an increased release of endothelial NO, further causing the vascular smooth muscle to relax in response to the sustained stress caused by increased blood flow [81] and sympathetic excitability [82], which may account for the decrease in arterial stiffness. There are also other mechanisms by which acute exercise alters vascular stiffness, such as reduced release of vasodilator mediators, vasoconstrictor factors, or vascular modifications [83].
On the other hand, long-term exercise causes and an adaptive response in the arteries. This could be due to the adaptation of the improved endothelial function [84], upregulation of endothelial NO synthase protein expression and phosphorylation, reduction in matrix metalloproteinases [85], improvement in insulin resistance [86], a decrease in proinflammatory cytokines [87], and improvement in the ability of glucose tolerance and insulin sensitivity [87]. Another important biomarker is the glycation index, which increases with age. The products of glycation, AGEs, have negative effects on vascular media and adventitia layers, which will cause the vasculature to be stiffer. High AGE levels may be a biomarker contributing to reduced physical activity in an older population [88]. Hence, regular exercise may improve arterial stiffness by reducing the production of glycation in females [89].
3.6. General Guidelines and Direction for Further Studies Investigating the Effect of Exercise on Arterial Stiffness in Women
It is clear that, while the effects of exercise on arterial stiffness in older women have been studied in more detail, relatively less research has been carried out on its effects in young and middle-aged women. Therefore, fewer exercise prescriptions could be presented to young and middle-aged women compared with older women. However, it is important to know that arterial stiffness in older women is unlikely to be restored to the level of young women, even when significant improvements have been made through exercise. Therefore, it should be highlighted that the greatest chance of eradicating cardiovascular disease in the future may be in primary and elementary prevention that begins at a young age [90].
Based on this review, it seems that arterial stiffness is more challenging to regulate through exercise in young and middle-aged women compared to older women. This may be attributed to the fact that older women tend to have stiffer arteries and a greater range of adjustment. Each of the principles of exercise prescription, including intensity, frequency, and time, can individually influence the effect of exercise on arterial stiffness (see What are the new findings for some practical applications). Future research should conduct dose-dependent intervention studies between each principle of exercise prescription and arterial stiffness in young and middle-aged women, especially focusing on exercise intensity due to its greater criticality than other principles. Indicators to assess intensity should be matched to the type of exercise, such as HRmax, HRR, and VO2max can assess aerobic exercise and HIIT intensity, while RM can assess resistance training intensity. In addition, both acute and regular exercise interventions should be performed to reveal the effect of exercise more precisely on arterial stiffness. Acute exercise may only cause functional changes in the arteries, whereas regular exercise may trigger alterations in the structure of the arteries. For acute exercise, arterial stiffness measurements should be taken at multiple time points, including before exercise, 15 min after exercise, and 30 min after exercise, to reflect the dynamic changes in arterial stiffness. Similarly, for regular exercise, multiple time-point measurements of arterial stiffness are also necessary. However, a sufficient period of intervention is more crucial and, according to this review, at least 8 weeks of exercise intervention are required. For older women, there are more exercise prescriptions available to improve arterial stiffness than for young and middle-aged women. Therefore, future research could focus more on developing types of exercise that are preferred by older women. In addition, arterial stiffness may be influenced by some confounding factors, such as somatotype, lifestyle, and diet. Stiffer arteries are found in physically inactive and obese people [91], while high sodium intake contributes to arterial stiffening [92]. Further research could take these factors into account to provide more effective guidelines for improving arterial stiffness in women.
4. Summary
4.1. What Is Already Known?
- Arterial stiffness is an independent predictor of cardiovascular disease.
- Arterial stiffness increases with age in women.
- Exercise can improve arterial stiffness in pathological states.
4.2. What Are the New Findings?
- The effect of exercise is dependent on age and arterial stiffness measure.
- Exercise intensity is inversely related to age and arterial stiffness measure.
- For young women, prolonged high-intensity aerobic exercise is recommended.
- For middle-aged women, moderate-intensity aerobic or stretching exercises are recommended.
- For older women, any intensity of aerobic exercise, such as daily walking and cycling, or light-intensity resistance training are recommended.
- To summarise, structured exercises can influence arterial stiffness positively. In addition, aerobic exercise is consistently found to be beneficial for woman, with an inverse relationship between age and recommended aerobic exercise intensity.
Author Contributions
Y.S.L. wrote the first draft of the manuscript, conducted the systematic search, and gained funding for the publication. Y.S.L. and T.K.K. independently extracted the data from each study and consolidated it in Table 2, Table 3 and Table 4. A.Y. conceptualised the review idea. T.K.K. and A.Y. interpreted the data analysis. All authors have read and agreed to the published version of the manuscript.
Funding
This article is funded by Humanity and Social Sciences Research Grant of Jilin Provincial Education Department (Grant number: JJKH20220808SK) and the Natural Science Foundation of Changchun Normal University (Grant number: 2021004).
Institutional Review Board Statement
This systematic review does not require an ethical approval.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zieman, S.J.; Melenovsky, V.; Kass, D.A. Mechanisms, Pathophysiology, and Therapy of Arterial Stiffness. Arter. Thromb. Vasc. Biol. 2005, 25, 932–943. [Google Scholar] [CrossRef]
- Safar, M.E.; Asmar, R.; Benetos, A.; Blacher, J.; Boutouyrie, P.; Lacolley, P.; Laurent, S.; London, G.; Pannier, B.; Protogerou, A.; et al. Interaction Between Hypertension and Arterial Stiffness. Hypertension 2018, 72, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Hsu, B.-G.; Tsai, J.-P. Arterial stiffness: A brief review. Tzu-Chi Med. J. 2021, 33, 115–121. [Google Scholar] [CrossRef]
- Mancia, G.; Fagard, R.; Narkiewicz, K.; Redán, J.; Zanchetti, A.; Böhm, M.; Christiaens, T.; Cifkova, R.; De Backer, G.; Dominiczak, A.; et al. 2013 Practice guidelines for the management of arterial hypertension of the European Society of Hypertension (ESH) and the European Society of Cardiology (ESC): ESH/ESC Task Force for the Management of Arterial Hypertension. J. Hypertens. 2013, 31, 1925–1938. [Google Scholar] [CrossRef] [PubMed]
- Maas, A.; Appelman, Y. Gender differences in coronary heart disease. Neth. Heart J. 2010, 18, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Vogel, B.; Acevedo, M.; Appelman, Y.; Merz, C.N.B.; Chieffo, A.; Figtree, G.A.; Guerrero, M.; Kunadian, V.; Lam, C.S.P.; Maas, A.H.E.M.; et al. The Lancet women and cardiovascular disease Commission: Reducing the global burden by 2030. Lancet 2021, 397, 2385–2438. [Google Scholar] [CrossRef] [PubMed]
- Strong, J.P.; Malcom, G.T.; Mcmahan, C.A.; Tracy, R.E.; Newman, W.P., III; Herderick, E.E.; Cornhill, J.F. Prevalence and extent of atherosclerosis in adolescents and young adults: Implications for prevention from the Pathobiological Determinants of Atherosclerosis in Youth Study. JAMA 1999, 281, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Panchangam, C.; Merrill, E.D.; Raghuveer, G. Utility of arterial stiffness assessment in children. Cardiol. Young 2018, 28, 362–376. [Google Scholar] [CrossRef]
- DuPont, J.J.; Kenney, R.M.; Patel, A.R.; Jaffe, I.Z. Sex differences in mechanisms of arterial stiffness. Br. J. Pharmacol. 2019, 176, 4208–4225. [Google Scholar] [CrossRef]
- Pierce, D.R.; Doma, K.; Leicht, A.S. Acute Effects of exercise mode on arterial stiffness and wave reflection in healthy young adults: A systematic review and meta-analysis. Front. Physiol. 2018, 9, 73. [Google Scholar] [CrossRef]
- Lephart, E.D.; Naftolin, F. Menopause and the skin: Old favorites and new innovations in cosmeceuticals for estrogen-deficient skin. Dermatol. Ther. 2021, 11, 53–69. [Google Scholar] [CrossRef] [PubMed]
- Hart, E.C.; Joyner, M.J. The curse of the sympathetic nervous system: Are men or women more unfortunate? J. Physiol. 2010, 588 Pt 22, 4345–4346. [Google Scholar] [CrossRef] [PubMed]
- Gerlo, E.A.M.; Schoors, D.F.; Dupont, A.G. Age- and sex-related differences for the urinary excretion of norepinephrine, epinephrine, and dopamine in adults. Clin. Chem. 1991, 37, 875–878. [Google Scholar] [CrossRef] [PubMed]
- Sorokin, V.; Vickneson, K.; Kofidis, T.; Woo, C.C.; Lin, X.Y.; Foo, R.; Shanahan, C.M. Role of vascular smooth muscle cell plasticity and interactions in vessel wall inflammation. Front. Immunol. 2020, 11, 599415. [Google Scholar] [CrossRef]
- Semba, R.D.; Nicklett, E.J.; Ferrucci, L. Does accumulation of advanced glycation end products contribute to the aging phenotype? J. Gerontol. Ser. A 2010, 65A, 963–975. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ling, W.; Teng, X.; Quan, C.; Cai, S.; Hu, S. Effect of advanced glycation end products, extracellular matrix metalloproteinase inducer and matrix metalloproteinases on type-I collagen metabolism. Biomed. Rep. 2016, 4, 691–693. [Google Scholar] [CrossRef] [PubMed]
- Doonan, R.J.; Mutter, A.; Egiziano, G.; Gomez, Y.-H.; Daskalopoulou, S.S. Differences in arterial stiffness at rest and after acute exercise between young men and women. Hypertens. Res. 2013, 36, 226–231. [Google Scholar] [CrossRef]
- Li, Z.-J.; Du, L.-F.; Luo, X.-H. Gender differences in ventricular-vascular coupling following exercise. Chin. Med. Sci. J. 2015, 30, 231–238. [Google Scholar] [CrossRef]
- Kioi, K.; Yamamoto, R.; Mori, K.; Nomura, T. Acute effect of resistance exercise on arterial stiffness in healthy young women. J. Allied Health Sci. 2018, 9, 1–5. [Google Scholar] [CrossRef]
- Fjeldstad, A.S.; Bemben, M.G.; Bemben, D.A. Resistance training effects on arterial compliance in premenopausal women. Angiology 2009, 60, 750–756. [Google Scholar] [CrossRef]
- Rossow, L.M.; Fahs, C.A.; Thiebaud, R.S.; Loenneke, J.P.; Kim, D.; Mouser, J.G.; Shore, E.A.; Beck, T.W.; Bemben, D.A.; Bemben, M.G. Arterial stiffness and blood flow adaptations following eight weeks of resistance exercise training in young and older women. Exp. Gerontol. 2014, 53, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- The Society for Adolescent Health and Medicine. Young adult health and well-being: A position statement of the Society for Adolescent Health and Medicine. J. Adolesc. Health 2017, 60, 758–759. [Google Scholar] [CrossRef] [PubMed]
- Kowal, P.; Dowd, J.E. Definition of an Older Person. Proposed Working Definition of an Older Person in Africa for the MDS Project; World Health Organization: Geneva, Switzerland, 2001; Volume 2, pp. 5188–9286.
- Park, H.-Y.; Jung, W.-S.; Kim, S.-W.; Lim, K. Effects of interval training under hypoxia on the autonomic nervous system and arterial and hemorheological function in healthy women. Int. J. Women’s Health 2022, 14, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Merchant, N.; Bajdek, N. Racial differences in arterial stiffness following acute bouts of anaerobic exercise. Circulation. 2021, 144, A10585. [Google Scholar] [CrossRef]
- Lee, C.; Lee, S.; Lee, C.; Lee, S. Acute effects of foam rolling exercises on arterial stiffness, flexibility and autonomic nervous system function in young and middle-aged women. Exerc. Sci. 2021, 30, 491–500. [Google Scholar] [CrossRef]
- Marshall, E.M.; Parks, J.C.; Singer, T.J.; Tai, Y.L.; DeBord, A.R.; Humm, S.M.; Kingsley, J.D. Vascular responses to high-intensity battling rope exercise between the sexes. J. Sport. Sci. Med. 2021, 20, 349–356. [Google Scholar] [CrossRef]
- Lim, J.; Kim, H.; Hwang, C.; Yoo, J.; Perez, H.; Handberg, E.M.; Christou, D. Acute Effects of Aerobic Exercise on Arterial Stiffness and Wave Reflection in Young Men and Young Premenopausal Women. FASEB J. 2018, 32, 587. [Google Scholar] [CrossRef]
- Tomschi, F.; Köster, P.; Predel, H.-G.; Lay, D.; Bloch, W.; Grau, M. Acute effects of lower and upper body-resistance training on arterial stiffness, peripheral, and central blood pressure in young normotensive women. Sport Sci. Health 2018, 14, 357–363. [Google Scholar] [CrossRef]
- Augustine, J.A.; Lefferts, W.K.; Heffernan, K.S. Sex differences in aortic stiffness following acute resistance exercise. Artery Res. 2018, 23, 52–55. [Google Scholar] [CrossRef]
- Okamoto, T.; Kobayashi, R.; Sakamaki-Sunaga, M. Effect of resistance exercise on arterial stiffness during the follicular and luteal phases of the menstrual cycle. Int. J. Sport. Med. 2017, 38, 347–352. [Google Scholar] [CrossRef]
- Kingsley, J.D.; Tai, Y.L.; Mayo, X.; Glasgow, A.; Marshall, E. Free-weight resistance exercise on pulse wave reflection and arterial stiffness between sexes in young, resistance-trained adults. Eur. J. Sport Sci. 2017, 17, 1056–1064. [Google Scholar] [CrossRef]
- Perdomo, S.J.; Moody, A.M.; McCoy, S.M.; Barinas-Mitchell, E.; Jakicic, J.M.; Gibbs, B.B. Effects on carotid–femoral pulse wave velocity 24 h post exercise in young healthy adults. Hypertens. Res. 2016, 39, 435–439. [Google Scholar] [CrossRef]
- Lane, A.D.; Yan, H.; Ranadive, S.M.; Kappus, R.M.; Sun, P.; Cook, M.D.; Harvey, I.; Woods, J.; Wilund, K.; Fernhall, B. Sex differences in ventricular–vascular coupling following endurance training. Eur. J. Appl. Physiol. 2014, 114, 2597–2606. [Google Scholar] [CrossRef]
- Harris, E.; Rakobowchuk, M.; Birch, K.M. Sprint interval and sprint continuous training increases circulating CD34+ cells and cardio-respiratory fitness in young healthy women. PloS ONE 2014, 9(9), e108720. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Masuhara, M.; Ikuta, K. Effects of eccentric and concentric resistance training on arterial stiffness. J. Hum. Hypertens. 2006, 20, 348–354. [Google Scholar] [CrossRef]
- Cebrowska, K.; Minczykowski, A.; Krauze, T.; Guzik, P.; Wykrętowicz, A. Arterial stiffness increases in response to an acute arterial load challenge induced by an isometric handgrip in healthy individuals. Kardiol. Pol. 2022, 80, 342–345. [Google Scholar] [CrossRef] [PubMed]
- Grigoriadis, G.; Rosenberg, A.J.; Lefferts, W.K.; Wee, S.O.; Schroeder, E.C.; Baynard, T. similar effects of acute resistance exercise on carotid stiffness in males and females. Int. J. Sport. Med. 2020, 41, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Chen, X.; Zeng, Z.; Li, S.; Wang, J.; Yu, F.; Liu, S.; Li, H.; Fernhall, B. Sex differences in lower-limb arterial stiffness following acute aerobic exercise. Sci. Sport. 2020, 35, e39–e48. [Google Scholar] [CrossRef]
- Augustine, N.K.N.; Heffernan, K.S. Menstrual phase and the vascular response to acute resistance exercise. Eur. J. Appl. Physiol. 2018, 118, 937–946. [Google Scholar] [CrossRef]
- Logan, J.G.; Kim, S.-S.; Lee, M.; Byon, H.D.; Yeo, S. Effects of static stretching exercise on lumbar flexibility and central arterial stiffness. J. Cardiovasc. Nurs. 2018, 33, 322–328. [Google Scholar] [CrossRef]
- Shinno, H.; Kurose, S.; Yamanaka, Y.; Higurashi, K.; Fukushima, Y.; Tsutsumi, H.; Kimura, Y. Evaluation of a static stretching intervention on vascular endothelial function and arterial stiffness. Eur. J. Sport Sci. 2017, 17, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Bemben, M.G.; Bemben, D.A. Effects of an 8-month yoga intervention on arterial compliance and muscle strength in premenopausal women. J. Sport. Sci. Med. 2012, 11, 322–330. [Google Scholar]
- Okamoto, T.; Masuhara, M.; Ikuta, K. Home-based resistance training improves arterial stiffness in healthy premenopausal women. Eur. J. Appl. Physiol. 2009, 107, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, M.; Maeda, S.; Miyaki, A.; Misono, M.; Saito, Y.; Tanabe, K.; Kuno, S.; Ajisaka, R. Effect of 12 weeks of moderate-intensity resistance training on arterial stiffness: A randomised controlled trial in women aged 32-59 years. Br. J. Sport. Med. 2009, 43, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Sabatier, M.J.; Schwark, E.H.; Lewis, R.; Sloan, G.; Cannon, J.; McCully, K. Femoral artery remodeling after aerobic exercise training without weight loss in women. Dyn. Med. 2008, 7, 13. [Google Scholar] [CrossRef]
- Cortez-Cooper, M.Y.; Devan, A.E.; Anton, M.M.; Farrar, R.P.; Beckwith, K.A.; Todd, J.S.; Tanaka, H. Effects of high intensity resistance training on arterial stiffness and wave reflection in women. Am. J. Hypertens. 2005, 18, 930–934. [Google Scholar] [CrossRef]
- Pekas, E.J.; Shin, J.; Son, W.-M.; Headid, R.J.; Park, S.-Y. Habitual combined exercise protects against age-associated decline in vascular function and lipid profiles in elderly postmenopausal women. Int. J. Environ. Res. Public Health 2020, 17, 3893. [Google Scholar] [CrossRef]
- Jaime, S.J.; Maharaj, A.; Alvarez-Alvarado, S.; Figueroa, A. Impact of low-intensity resistance and whole-body vibration training on aortic hemodynamics and vascular function in postmenopausal women. Hypertens. Res. 2019, 42, 1979–1988. [Google Scholar] [CrossRef]
- Molisz, A.; Schmederer, Z.; Siebert, J.; Kadamani, T.; Glasner, P.; Rosłonkiewicz, K.; Nowicka-Sauer, K.; Gutknecht, P.; Trzeciak, B.; Suchanowski, A. Haemodynamic parameters in postmenopausal women—Beneficial effect of moderate continuous exercise training. Ann. Agric. Environ. Med. 2019, 26, 425–428. [Google Scholar] [CrossRef]
- Kim, J.H.; Jung, Y.S.; Kim, J.W.; Ha, M.S.; Ha, S.M.; Kim, D.Y. Effects of aquatic and land-based exercises on amyloid beta, heat shock protein 27, and pulse wave velocity in elderly women. Exp. Gerontol. 2018, 108, 62–68. [Google Scholar] [CrossRef]
- Nishiwaki, M.; Fujibayashi, M.; Nanayama, C.; Ogawa, N.; Itakura, I.; Matsumoto, N. Increasing levels of daily physical activity for arterial stiffness reduction in older women: A community-based pilot study. J. Sport. Med. Phys. Fit. 2018, 58, 1701–1709. [Google Scholar] [CrossRef]
- Yasuda, T.; Fukumura, K.; Tomaru, T.; Nakajima, T. Thigh muscle size and vascular function after blood flow-restricted elastic band training in older women. Oncotarget 2016, 7, 33595–33607. [Google Scholar] [CrossRef]
- Yasuda, T.; Fukumura, K.; Iida, H.; Nakajima, T. Effects of detraining after blood flow-restricted low-load elastic band training on muscle size and arterial stiffness in older women. Springerplus 2015, 4, 348. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Lee, D.-C. Cardiac and pulmonary benefits of forest walking versus city walking in elderly women: A randomised, controlled, open-label trial. Eur. J. Integr. Med. 2014, 6, 5–11. [Google Scholar] [CrossRef]
- Matsubara, T.; Miyaki, A.; Akazawa, N.; Choi, Y.; Ra, S.-G.; Tanahashi, K.; Kumagai, H.; Oikawa, S.; Maeda, S. Aerobic exercise training increases plasma Klotho levels and reduces arterial stiffness in postmenopausal women. Am. J. Physiol.-Heart Circ. Physiol. 2014, 306, H348–H355. [Google Scholar] [CrossRef] [PubMed]
- Tanahashi, K.; Akazawa, N.; Miyaki, A.; Choi, Y.; Ra, S.-G.; Matsubara, T.; Kumagai, H.; Oikawa, S.; Maeda, S. aerobic exercise training decreases plasma asymmetric dimethylarginine concentrations with increase in arterial compliance in postmenopausal women. Am. J. Hypertens. 2014, 27, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Corrick, K.L.; Hunter, G.R.; Fisher, G.; Glasser, S.P. Changes in vascular hemodynamics in older women following 16 weeks of combined aerobic and resistance training. J. Clin. Hypertens. (Greenwich) 2013, 15, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Hui-Chan, C.W.Y.; Tsang, W.W.N. Effects of Tai Chi training on arterial compliance and muscle strength in female seniors: A randomized clinical trial. Eur. J. Prev. Cardiol. 2013, 20, 238–245. [Google Scholar] [CrossRef]
- Williams, A.D.; Ahuja, K.; Almond, J.B.; Robertson, I.K.; Ball, M.J. Progressive resistance training might improve vascular function in older women but not in older men. J. Sci. Med. Sport 2013, 16, 76–81. [Google Scholar] [CrossRef]
- Miyaki, A.; Maeda, S.; Choi, Y.; Akazawa, N.; Tanabe, Y.; Ajisaka, R. Habitual aerobic exercise increases plasma pentraxin 3 levels in middle-aged and elderly women. Appl. Physiol. Nutr. Metab. 2012, 37, 907–911. [Google Scholar] [CrossRef] [PubMed]
- Ohta, M.; Hirao, N.; Mori, Y.; Takigami, C.; Eguchi, M.; Tanaka, H.; Ikeda, M.; Yamato, H. Effects of bench step exercise on arterial stiffness in post-menopausal women: Contribution of IGF-1 bioactivity and nitric oxide production. Growth Horm. IGF Res. 2012, 22, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.M.O.; Sales, M.M.; Moraes, J.F.V.N.; Asana, R.Y.; Neto, W.B.; Santana, H.A.P.; Silva, C.B.; Moreira, S.R.; Simões, H.G.; Lima, R.M.; et al. Influence of the I/D polymorphism of the angiotensin converting enzyme gene and acute aerobic exercise in the ambulatory arterial stiffness index of elderly women. J. Exerc. Physiol. Online 2011, 14, 1–9. [Google Scholar]
- Figueroa, A.; Park, S.Y.; Seo, D.Y.; Sanchez-Gonzalez, M.A.; Baek, Y.H. Combined resistance and endurance exercise training improves arterial stiffness, blood pressure, and muscle strength in postmenopausal women. Menopause 2011, 18, 980–984. [Google Scholar] [CrossRef]
- Nishiwaki, M.; Kawakami, R.; Saito, K.; Tamaki, H.; Takekura, H.; Ogita, F. Vascular adaptations to hypobaric hypoxic training in postmenopausal women. J. Physiol. Sci. 2011, 61, 83–91. [Google Scholar] [CrossRef]
- Miura, H.; Nakagawa, E.; Takahashi, Y. Influence of group training frequency on arterial stiffness in elderly women. Eur. J. Appl. Physiol. 2008, 104, 1039–1044. [Google Scholar] [CrossRef]
- Casey, D.P.; Pierce, G.L.; Howe, K.S.; Mering, M.C.; Braith, R.W. Effect of resistance training on arterial wave reflection and brachial artery reactivity in normotensive postmenopausal women. Eur. J. Appl. Physiol. 2007, 100, 403–408. [Google Scholar] [CrossRef]
- Sugawara, J.; Inoue, H.; Hayashi, K.; Yokoi, T.; Kono, I. Effect of low-intensity aerobic exercise training on arterial compliance in postmenopausal women. Hypertens. Res. 2004, 27, 897–901. [Google Scholar] [CrossRef]
- Canning, K.L.; Brown, R.E.; Jamnik, V.K.; Salmon, A.; Ardern, C.I.; Kuk, J.L. Individuals underestimate moderate and vigorous intensity physical activity. PLoS ONE 2014, 9, e97927. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Day, S.; Deming, R. Acute exercise intensity and memory function: Evaluation of the transient hypofrontality hypothesis. Medicina 2019, 55, 445. [Google Scholar] [CrossRef]
- Haff, G.G.; Triplett, N.T. Essentials of Strength Training and Conditioning, 4th ed.; Human Kinetics: Champaign, IL, USA, 2015. [Google Scholar]
- Larson-Meyer, D.E. A systematic review of the energy cost and metabolic intensity of yoga. Med. Sci. Sport. Exerc. 2016, 48, 1558–1569. [Google Scholar] [CrossRef] [PubMed]
- Kuehl, R.; Scharhag-Rosenberger, F.; Schommer, K.; Schmidt, M.E.; Dreger, P.; Huber, G.; Bohus, M.; Ulrich, C.M.; Wiskemann, J. Exercise intensity classification in cancer patients undergoing allogeneic HCT. Med. Sci. Sport. Exerc. 2015, 47, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Yoon, E.S.; Jung, S.J.; Cheun, S.K.; Oh, Y.S.; Kim, S.H.; Jae, S.Y. Effects of acute resistance exercise on arterial stiffness in young men. Korean Circ. J. 2010, 40, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Saz-Lara, A.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Notario-Pacheco, B.; Ruiz-Grao, M.C.; Martínez-Vizcaíno, V. The acute effect of exercise on arterial stiffness in healthy subjects: A meta-analysis. J. Clin. Med. 2021, 10, 291. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, T.; Zhu, W.; Wu, H.; Yan, S. Acute effects of continuous and interval low-intensity exercise on arterial stiffness in healthy young men. Eur. J. Appl. Physiol. 2014, 114, 1385–1392. [Google Scholar] [CrossRef]
- Yamato, Y.; Hasegawa, N.; Sato, K.; Hamaoka, T.; Ogoh, S.; Iemitsu, M. Acute effect of static stretching exercise on arterial stiffness in healthy young adults. Am. J. Phys. Med. Rehabil. 2016, 95, 764–770. [Google Scholar] [CrossRef]
- Green, D.J.; Smith, K.J. Effects of exercise on vascular function, structure, and health in humans. Cold Spring Harb. Perspect. Med. 2018, 8, a029819. [Google Scholar] [CrossRef]
- Tomoto, T.; Sugawara, J.; Hirasawa, A.; Imai, T.; Maeda, S.; Ogoh, S. Impact of short-term training camp on arterial stiffness in endurance runners. J. Physiol. Sci. 2015, 65, 445–449. [Google Scholar] [CrossRef]
- Asano, R.Y.; Sales, M.M.; Coelho, J.M.; de Moraes, J.F.V.N.; Pereira, L.A.; Campbell, C.S.G.; Simoes, H.G. Exercise, nitric oxide, and endothelial dysfunction: A brief review. J. Exerc. Physiol. Online 2012, 15, 76–86. [Google Scholar]
- Houssiere, A.; Najem, B.; Ciarka, A.; Velez-Roa, S.; Naeije, R.; van de Borne, P. Chemoreflex and metaboreflex control during static hypoxic exercise. Am. J. Physiol.-Heart Circ. Physiol. 2005, 288, H1724–H1729. [Google Scholar] [CrossRef] [PubMed]
- Siasos, G.; Athanasiou, D.; Terzis, G.; Stasinaki, A.; Oikonomou, E.; Tsitkanou, S.; Kolokytha, T.; Spengos, K.; Papavassiliou, A.G.; Tousoulis, D. Acute effects of different types of aerobic exercise on endothelial function and arterial stiffness. Eur. J. Prev. Cardiol. 2016, 23, 1565–1572. [Google Scholar] [CrossRef]
- Mestek, M.L.; Westby, C.M.; Van Guilder, G.P.; Greiner, J.J.; Stauffer, B.; DeSouza, C.A. Regular aerobic exercise, without weight loss, improves endothelium-dependent vasodilation in overweight and obese adults. Obesity 2010, 18, 1667–1669. [Google Scholar] [CrossRef] [PubMed]
- Donley, D.A.; Fournier, S.B.; Reger, B.L.; DeVallance, E.; Bonner, D.E.; Olfert, I.M.; Frisbee, J.C.; Chantler, P.D. Aerobic exercise training reduces arterial stiffness in metabolic syndrome. J. Appl. Physiol. 2014, 116, 1396–1404. [Google Scholar] [CrossRef]
- Yokoyama, H.; Emoto, M.; Fujiwara, S.; Motoyama, K.; Morioka, T.; Koyama, H.; Shoji, T.; Inaba, M.; Nishizawa, Y. Short-term aerobic exercise improves arterial stiffness in type 2 diabetes. Diabetes Res. Clin. Pract. 2004, 65, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, N.T.; Martin, J.S.; Laughlin, M.H.; Padilla, J. Exercise-induced signals for vascular endothelial adaptations: Implications for cardiovascular disease. Curr. Cardiovasc. Risk Rep. 2012, 6, 331–346. [Google Scholar] [CrossRef]
- Drenth, M.H.; Zuidema, S.U.; Krijnen, W.P.; Bautmans, I.; Smit, A.J.; Van Der Schans, C.; Hobbelen, H. Advanced glycation end products are associated with physical activity and physical functioning in the older population. J. Gerontol. Ser. A 2018, 73, 1545–1551. [Google Scholar] [CrossRef]
- Ebert, H.; Lacruz, M.E.; Kluttig, A.; Simm, A.; Greiser, K.H.; Tiller, D.; Kartschmit, N.; Mikolajczyk, R. Advanced glycation end products and their ratio to soluble receptor are associated with limitations in physical functioning only in women: Results from the CARLA cohort. BMC Geriatr. 2019, 19, 299. [Google Scholar] [CrossRef]
- Gooding, H.C.; Gidding, S.S.; Moran, A.E.; Redmond, N.; Allen, N.B.; Bacha, F.; Burns, T.L.; Catov, J.M.; Grandner, M.A.; Harris, K.M.; et al. Challenges and opportunities for the prevention and treatment of cardiovascular disease among young adults: Report from a national heart, lung, and blood institute working group. J. Am. Heart Assoc. 2020, 9, e016115. [Google Scholar] [CrossRef]
- Xiong, J.; Qian, Y.; Yu, S.; Ji, H.; Teliewubai, J.; Chi, C.; Lu, Y.; Zhou, Y.; Fan, X.; Li, J.; et al. Somatotype and Its Impact on Asymptomatic Target Organ Damage in the Elderly Chinese: The Northern Shanghai Study. Clin. Interv. Aging 2021, 16, 887–895. [Google Scholar] [CrossRef]
- D’Elia, L.; Galletti, F.; La Fata, E.; Sabino, P.; Strazzullo, P. Effect of dietary sodium restriction on arterial stiffness: Systematic review and meta-analysis of the randomized controlled trials. J. Hypertens. 2018, 36, 734–743. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).