Fermented Oats as a Novel Functional Food
Abstract
1. Introduction
2. Fermented Oat Products on the Market
3. Methods Employed to Produce Fermented Oats
3.1. Bacteria
3.2. Fungi
3.3. Yeasts
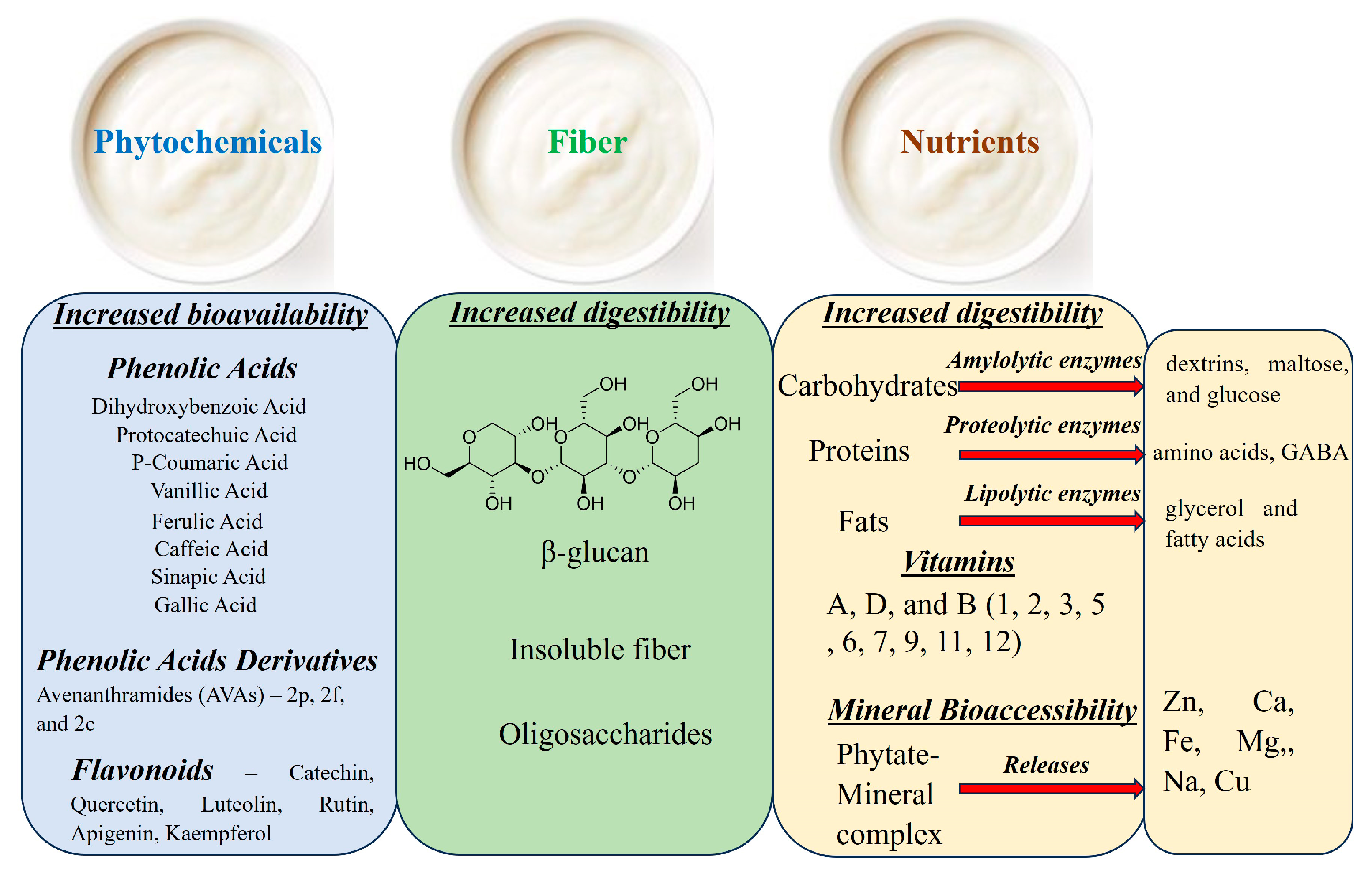
4. Effects of Fermentation on the Chemical Composition of Oats
4.1. Effect of Fermentation on Macronutrients in Oats
4.1.1. Carbohydrates
4.1.2. Proteins
4.1.3. Fats
4.2. Effect of Fermentation on Micronutrients in Oats
4.2.1. Vitamins
4.2.2. Minerals
4.3. Effect of Fermentation on Fiber Content in Oats
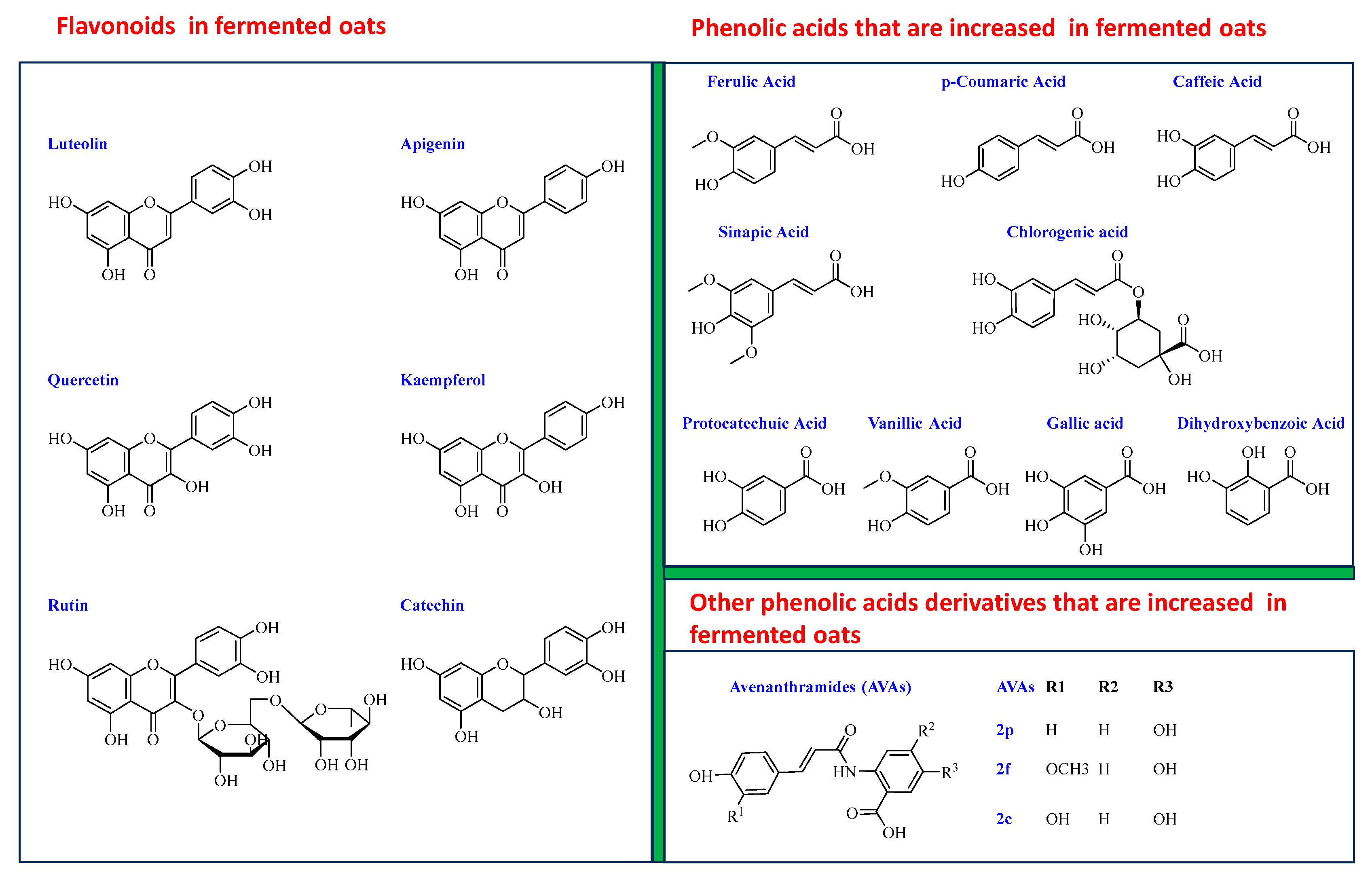
4.4. Effect of Fermentation on Phytochemicals in Oats
4.4.1. TPC
4.4.2. Phenolic Acids
4.4.3. AVAs
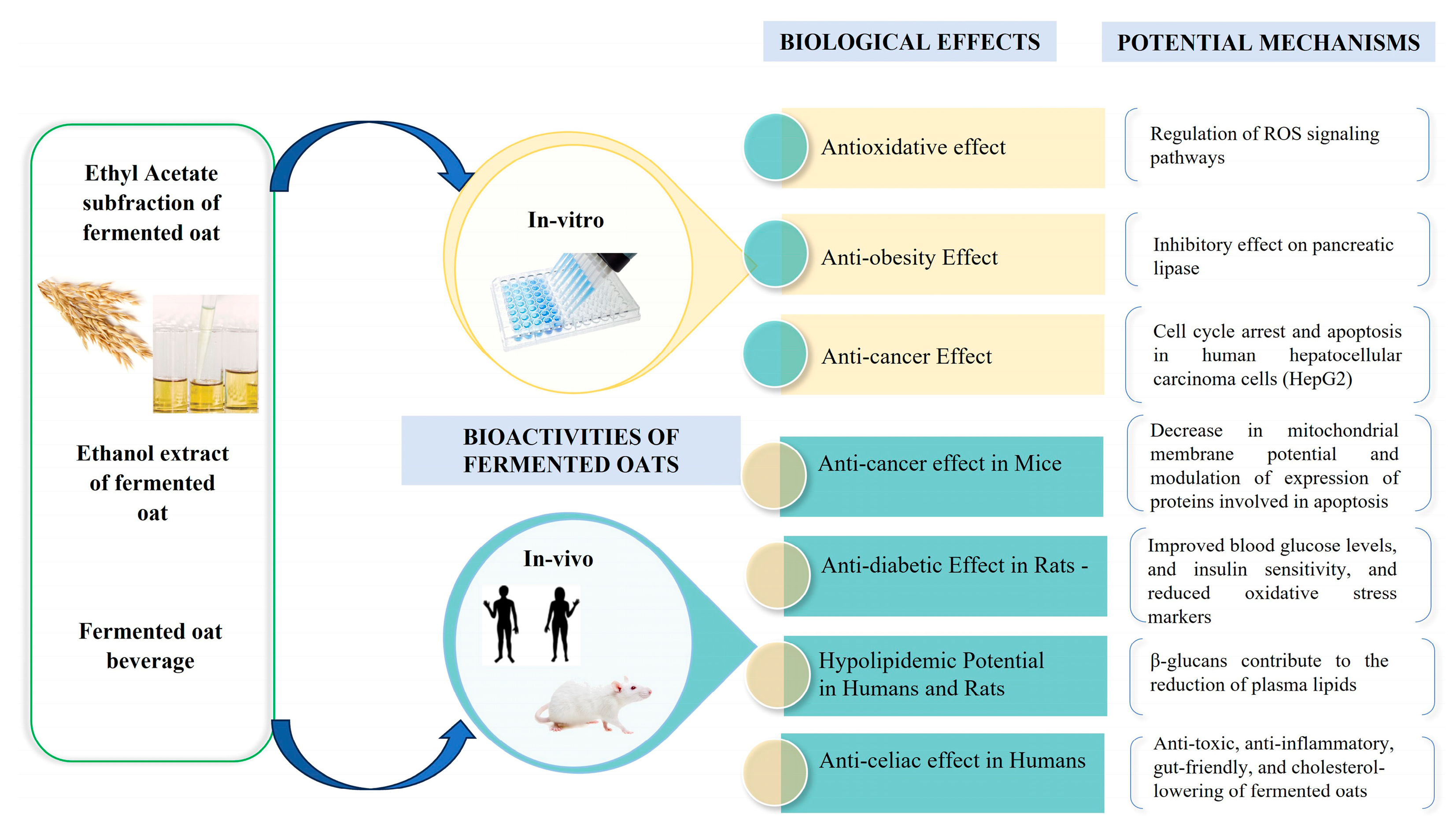
5. Biological Function of Fermented Oats
5.1. Antidiabetic Effects of Fermented Oats
5.2. Anti-Obesity Effects of Fermented Oats
5.3. Anti-Cholesterol Effects of Fermented Oats
5.4. Anti-Cancer Effects of Fermented Oats
5.5. Anti-Celiac Effects of Fermented Oats
6. Future Perspectives for Fermented Oats
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dimidi, E.; Cox, S.R.; Rossi, M.; Whelan, K. Fermented Foods: Definitions and Characteristics, Impact on the Gut Microbiota and Effects on Gastrointestinal Health and Disease. Nutrients 2019, 11, 1806. [Google Scholar] [CrossRef] [PubMed]
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527. [Google Scholar] [CrossRef] [PubMed]
- Sivamaruthi, B.S.; Kesika, P.; Prasanth, M.I.; Chaiyasut, C. A Mini Review on Antidiabetic Properties of Fermented Foods. Nutrients 2018, 10, 1973. [Google Scholar] [CrossRef] [PubMed]
- Gotcheva, V.; Hristozova, E.; Hristozova, T.; Guo, M.; Roshkova, Z.; Angelov, A. Assessment of Potential Probiotic Properties of Lactic Acid Bacteria and Yeast Strains. Food Biotechnol. 2002, 16, 211–225. [Google Scholar] [CrossRef]
- Angelov, A.; Gotcheva, V.; Kuncheva, R.; Hristozova, T. Development of a new oat-based probiotic drink. Int. J. Food Microbiol. 2006, 112, 75–80. [Google Scholar] [CrossRef]
- Govender, M.; Choonara, Y.E.; Kumar, P.; du Toit, L.C.; van Vuuren, S.; Pillay, V. A review of the advancements in probiotic delivery: Conventional vs. non-conventional formulations for intestinal flora supplementation. AAPS PharmSciTech 2014, 15, 29–43. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N. Probiotic fermentation of plant based products: Possibilities and opportunities. Crit. Rev. Food Sci. Nutr. 2012, 52, 183–199. [Google Scholar] [CrossRef]
- Verspreet, J.; Damen, B.; Broekaert, W.F.; Verbeke, K.; Delcour, J.A.; Courtin, C.M. A Critical Look at Prebiotics Within the Dietary Fiber Concept. Annu. Rev. Food Sci. Technol. 2016, 7, 167–190. [Google Scholar] [CrossRef]
- Frías, J.; Martínez-Villaluenga, C.; Peñas, E. Fermented Foods in Health and Disease Prevention, 1st ed.; Academic Press: Boston, MA, USA, 2016; pp. 1–762. [Google Scholar]
- Herrera-Ponce, A.; Nevárez-Morillón, G.; Ortega-Rívas, E.; Pérez-Vega, S.; Salmerón, I. Fermentation adaptability of three probiotic Lactobacillus strains to oat, germinated oat and malted oat substrates. Lett. Appl. Microbiol. 2014, 59, 449–456. [Google Scholar] [CrossRef]
- Soycan, G.; Schär, M.Y.; Kristek, A.; Boberska, J.; Alsharif, S.N.S.; Corona, G.; Shewry, P.R.; Spencer, J.P.E. Composition and content of phenolic acids and avenanthramides in commercial oat products: Are oats an important polyphenol source for consumers? Food Chem. X 2019, 3, 100047. [Google Scholar] [CrossRef]
- Aparicio-García, N.; Martínez-Villaluenga, C.; Frias, J.; Perez, L.C.; Fernández, C.F.; Alba, C.; Rodríguez, J.M.; Peñas, E. A Novel Sprouted Oat Fermented Beverage: Evaluation of Safety and Health Benefits for Celiac Individuals. Nutrients 2021, 13, 2522. [Google Scholar] [CrossRef]
- Angelov, A.; Yaneva-Marinova, T.; Gotcheva, V. Oats as a matrix of choice for developing fermented functional beverages. J. Food Sci. Technol. 2018, 55, 2351–2360. [Google Scholar] [CrossRef] [PubMed]
- Products ZOE Concentrates of Live (Active) Bacteria. Available online: https://ekozoe.lv/en/ (accessed on 28 July 2023).
- Öste Triantafyllou, A.A.; Carina Martensson, O. Fermented Product Based on an Oat Suspension. U.S. Patent 7160564B2, 9 November 2001. [Google Scholar]
- Vasudha, S.; Mishra, H.N. Non dairy probiotic beverages. Int. Food Res. J. 2013, 20, 7–15. [Google Scholar]
- ProViva Active® by Probi. Available online: https://www.probi.com/ (accessed on 1 August 2023).
- Mårtensson, O.; Andersson, C.; Andersson, K.; Öste, R.; Holst, O. Formulation of an oat-based fermented product and its comparison with yoghurt. J. Sci. Food Agric. 2001, 81, 1314–1321. [Google Scholar] [CrossRef]
- Jooste, R.N.a.P.J. Cereal-Based Functional Foods. In Probiotics, 3rd ed.; IntechOpen: Rijeka, Croatia, 2012; pp. 161–196. [Google Scholar]
- Oatmilk Non-Dairy Yogurt. Available online: https://nancysyogurt.com/products/oatmilk-non-dairy-yogurt/ (accessed on 28 July 2023).
- Tchekessi, C.K.C.; Bokossa, I.Y.; Azokpota, P.; Agbangla, C.; Daube, G.; Scippo, M.-L.; Korsak Koulagenko, N.; Angelov, A. Isolation and Quantification of Lactic Acid Bacteria from Traditional Fermented Products in Benin. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 1–8. [Google Scholar]
- Organic Oatmeal Cups. Available online: https://bakeryonmain.com/collection/oatmeal-cups/ (accessed on 18 July 2023).
- Lifeway Oats. Available online: https://lifewaykefir.com/lifewayoat/ (accessed on 28 July 2023).
- Collins, Y.; Dohnalek, M.; Kleinbach-Sauter, H.; Mcdonagh, M.D.; Omarov, M.N. Co-Fermented Food Product From Dairy and Grain. U.S. Patent 11464248B2, 2 November 2018. [Google Scholar]
- Goncerzewicz, A.; Misiewicz, A.; Owczarek, L.; Jasińska, U.; Skąpska, S. The Effect of a Newly Developed Oat-Banana Fermented Beverage with a Beta-glucan Additive on ldhL Gene Expression in Streptococcus thermophilus T(K)M(3) KKP 2030p. Curr. Microbiol. 2016, 73, 773–780. [Google Scholar] [CrossRef]
- Yu, Q.; Qian, J.; Guo, Y.; Qian, H.; Yao, W.; Cheng, Y. Applicable Strains, Processing Techniques and Health Benefits of Fermented Oat Beverages: A Review. Foods 2023, 12, 1708. [Google Scholar] [CrossRef]
- Ahmed, A.; Aslam, M.; Ashraf, M.; Nasim, F.u.-H.; Batool, K.; Bibi, A. Microbial ¦Â-Glucosidases: Screening, Characterization, Cloning and Applications. J. Appl. Environ. Microbiol. 2017, 5, 57–73. [Google Scholar] [CrossRef]
- Wronkowska, M.; Rostek, D.; Lenkiewicz, M.; Kurantowicz, E.; Yaneva, T.G.; Starowicz, M. Oat flour fermented by Lactobacillus strains—Kinetics of volatile compound formation and antioxidant capacity. J. Cereal Sci. 2022, 103, 103392. [Google Scholar] [CrossRef]
- Gupta, M.; Bajaj, B.K. Development of fermented oat flour beverage as a potential probiotic vehicle. Food Biosci. 2017, 20, 104–109. [Google Scholar] [CrossRef]
- Asadzadeh, A.; Jalali, H.; Azizi, M.H.; Mohammadi Nafchi, A. Production of oat bran functional probiotic beverage using Bifidobacterium lactis. J. Food Meas. Charact. 2021, 15, 1301–1309. [Google Scholar] [CrossRef]
- Ruiz, L.; Ruas-Madiedo, P.; Gueimonde, M.; de Los Reyes-Gavilán, C.G.; Margolles, A.; Sánchez, B. How do bifidobacteria counteract environmental challenges? Mechanisms involved and physiological consequences. Genes. Nutr. 2011, 6, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Klajn, V.M.; Ames, C.W.; da Cunha, K.F.; Lorini, A.; Hackbart, H.; Filho, P.J.S.; Cruxen, C.; Fiorentini, Â.M. Probiotic fermented oat dairy beverage: Viability of Lactobacillus casei, fatty acid profile, phenolic compound content and acceptability. J. Food Sci. Technol. 2021, 58, 3444–3452. [Google Scholar] [CrossRef]
- Blandino, A.; Al-Aseeri, M.E.; Pandiella, S.S.; Cantero, D.; Webb, C. Cereal-based fermented foods and beverages. Food Res. Int. 2003, 36, 527–543. [Google Scholar] [CrossRef]
- Herrera-Ponce, A.L.; Salmeron-Ochoa, I.; Rodriguez-Figueroa, J.C.; Santellano-Estrada, E.; Garcia-Galicia, I.A.; Alarcon-Rojo, A.D. High-intensity ultrasound as pre-treatment in the development of fermented whey and oat beverages: Effect on the fermentation, antioxidant activity and consumer acceptance. J. Food Sci. Technol. 2022, 59, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Hole, A.S.; Rud, I.; Grimmer, S.; Sigl, S.; Narvhus, J.; Sahlstrøm, S. Improved bioavailability of dietary phenolic acids in whole grain barley and oat groat following fermentation with probiotic Lactobacillus acidophilus, Lactobacillus johnsonii, and Lactobacillus reuteri. J. Agric. Food Chem. 2012, 60, 6369–6375. [Google Scholar] [CrossRef]
- Kalpa, R.E.; Sreejit, V.; Preetha, R.; Nagamaniammai, G. Synbiotic microencapsulation of Lactobacillus brevis and Lactobacillus delbrueckii subsp. lactis using oats/oats brans as prebiotic for enhanced storage stability. J. Food Sci. Technol. 2023, 60, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Tan, Z.; Wang, X.; Cui, M.; Wang, Y.; Jiao, Z. Impact inoculum dosage of lactic acid bacteria on oat and wheat silage fermentation at ambient and low temperatures. Crop Pasture Sci. 2018, 69, 1225–1236. [Google Scholar] [CrossRef]
- Deen, B.; Diez-Gonzalez, F. Assessment of Pediococcus acidilactici ATCC 8042 as potential Salmonella surrogate for thermal treatments of toasted oats cereal and peanut butter. Food Microbiol. 2019, 83, 187–192. [Google Scholar] [CrossRef]
- Chen, L.; Bai, S.; You, M.; Xiao, B.; Li, P.; Cai, Y. Effect of a low temperature tolerant lactic acid bacteria inoculant on the fermentation quality and bacterial community of oat round bale silage. Anim. Feed. Sci. Technol. 2020, 269, 114669. [Google Scholar] [CrossRef]
- Carabajal Torrez, J.A.; Llebeili, Y.; Rodriguez de Olmos, A.; Gerez, C.L. [Lactic starter cultures to improve the oat bioactive compounds]. Rev. Argent. Microbiol. 2021, 53, 333–342. [Google Scholar] [CrossRef]
- Heo, S.J.; Kim, A.J.; Park, M.J.; Kang, K.; Soung, D.Y. Nutritional and Functional Properties of Fermented Mixed Grains by Solid-State Fermentation with Bacillus amyloliquefaciens 245. Foods 2020, 9, 1693. [Google Scholar] [CrossRef] [PubMed]
- Bei, Q.; Liu, Y.; Wang, L.; Chen, G.; Wu, Z. Improving free, conjugated, and bound phenolic fractions in fermented oats (Avena sativa L.) with Monascus anka and their antioxidant activity. J. Funct. Foods 2017, 32, 185–194. [Google Scholar] [CrossRef]
- Bei, Q.; Wu, Z.; Chen, G. Dynamic changes in the phenolic composition and antioxidant activity of oats during simultaneous hydrolysis and fermentation. Food Chem. 2020, 305, 125269. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Gao, F.; Zhang, X.; Wang, O.; Wu, W.; Zhu, S.; Zhang, D.; Zhou, F.; Ji, B. Evaluation of γ-aminobutyric acid, phytate and antioxidant activity of tempeh-like fermented oats (Avena sativa L.) prepared with different filamentous fungi. J. Food Sci. Technol. 2014, 51, 2544–2551. [Google Scholar] [CrossRef]
- Cai, S.; Wang, O.; Wu, W.; Zhu, S.; Zhou, F.; Ji, B.; Gao, F.; Zhang, D.; Liu, J.; Cheng, Q. Comparative study of the effects of solid-state fermentation with three filamentous fungi on the total phenolics content (TPC), flavonoids, and antioxidant activities of subfractions from oats (Avena sativa L.). J. Agric. Food Chem. 2012, 60, 507–513. [Google Scholar] [CrossRef]
- Song, T.; Zhang, Z.; Jin, Q.; Feng, W.; Shen, Y.; Fan, L.; Cai, W. Nutrient profiles, functional compositions, and antioxidant activities of seven types of grain fermented with Sanghuangporus sanghuang fungus. J. Food Sci. Technol. 2021, 58, 4091–4101. [Google Scholar] [CrossRef]
- Espinosa-Páez, E.; Alanis-Guzmán, M.G.; Hernández-Luna, C.E.; Báez-González, J.G.; Amaya-Guerra, C.A.; Andrés-Grau, A.M. Increasing Antioxidant Activity and Protein Digestibility in Phaseolus vulgaris and Avena sativa by Fermentation with the Pleurotus ostreatus Fungus. Molecules 2017, 22, 2275. [Google Scholar] [CrossRef]
- Wu, H.; Liu, H.-N.; Ma, A.-M.; Zhou, J.-Z.; Xia, X.-D. Synergetic effects of Lactobacillus plantarum and Rhizopus oryzae on physicochemical, nutritional and antioxidant properties of whole-grain oats (Avena sativa L.) during solid-state fermentation. LWT 2022, 154, 112687. [Google Scholar] [CrossRef]
- Grzegorz, D.; Aurelija, P.; Aldona, B.; Lucyna, K.; Sylwester, C.; Iwona, K. Changes in Selected Quality Indices in Microbially Fermented Commercial Almond and Oat Drinks. Appl. Sci. 2022, 12, 9983. [Google Scholar] [CrossRef]
- Rossi, M.; Amaretti, A.; Raimondi, S. Folate production by probiotic bacteria. Nutrients 2011, 3, 118–134. [Google Scholar] [CrossRef] [PubMed]
- Kariluoto, S.; Edelmann, M.; Nyström, L.; Sontag-Strohm, T.; Salovaara, H.; Kivelä, R.; Herranen, M.; Korhola, M.; Piironen, V. In situ enrichment of folate by microorganisms in beta-glucan rich oat and barley matrices. Int. J. Food Microbiol. 2014, 176, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Călinoiu, L.F.; Cătoi, A.F.; Vodnar, D.C. Solid-State Yeast Fermented Wheat and Oat Bran as A Route for Delivery of Antioxidants. Antioxidants 2019, 8, 372. [Google Scholar] [CrossRef] [PubMed]
- Gokavi, S.; Zhang, L.; Huang, M.-K.; Zhao, X.; Guo, M. Oat-based Symbiotic Beverage Fermented by Lactobacillus plantarum, Lactobacillus paracasei ssp. casei, and Lactobacillus acidophilus. J. Food Sci. Technol. 2005, 70, M216–M223. [Google Scholar] [CrossRef]
- Gargi Das, J.M.M. Nutrient Profile of Fermented Oats. J. Food Sci. Technol. 2017, 2, 69–71. [Google Scholar]
- Östman, E.M.; Nilsson, M.; Liljeberg Elmståhl, H.G.M.; Molin, G.; Björck, I.M.E. On the Effect of Lactic Acid on Blood Glucose and Insulin Responses to Cereal Products: Mechanistic Studies in Healthy Subjects and In Vitro. J. Cereal Sci. 2002, 36, 339–346. [Google Scholar] [CrossRef]
- Ruxton Carrie, C.R. The Role of Oats and Oat Product. J. Nutr. 2014, 14, 55–57. [Google Scholar]
- Omoba, O.S.; Isah, L.R. Influence of Sourdough Fermentation on Amino Acids Composition, Phenolic Profile, and Antioxidant Properties of Sorghum Biscuits. Prev. Nutr. Food Sci. 2018, 23, 220–227. [Google Scholar] [CrossRef]
- Matthews, A.; Grimaldi, A.; Walker, M.; Bartowsky, E.; Grbin, P.; Jiranek, V. Lactic acid bacteria as a potential source of enzymes for use in vinification. Appl. Environ. Microbiol. 2004, 70, 5715–5731. [Google Scholar] [CrossRef]
- Svanberg, U.; Lorri, W. Fermentation and nutrient availability. Food Control 1997, 8, 319–327. [Google Scholar] [CrossRef]
- Mao, H.; Xu, M.; Ji, J.; Zhou, M.; Li, H.; Wen, Y.; Wang, J.; Sun, B. The utilization of oat for the production of wholegrain foods: Processing technology and products. Food Front. 2022, 3, 28–45. [Google Scholar] [CrossRef]
- Markowiak-Kopeć, P.; Śliżewska, K. The Effect of Probiotics on the Production of Short-Chain Fatty Acids by Human Intestinal Microbiome. Nutrients 2020, 12, 1107. [Google Scholar] [CrossRef]
- Capozzi, V.; Russo, P.; Dueñas, M.T.; López, P.; Spano, G. Lactic acid bacteria producing B-group vitamins: A great potential for functional cereals products. Appl. Microbiol. Biotechnol. 2012, 96, 1383–1394. [Google Scholar] [CrossRef]
- Adebo, J.A.; Njobeh, P.B.; Gbashi, S.; Oyedeji, A.B.; Ogundele, O.M.; Oyeyinka, S.A.; Adebo, O.A. Fermentation of Cereals and Legumes: Impact on Nutritional Constituents and Nutrient Bioavailability. Fermentation 2022, 8, 63. [Google Scholar] [CrossRef]
- Bohn, T.; Davidsson, L.; Walczyk, T.; Hurrell, R.F. Phytic acid added to white-wheat bread inhibits fractional apparent magnesium absorption in humans. Am. J. Clin. Nutr. 2004, 79, 418–423. [Google Scholar] [CrossRef]
- Fujita, J.; Shigeta, S.; Yamane, Y.; Fukuda, H.; Kizaki, Y.; Wakabayashi, S.; Ono, K. Production of two types of phytase from Aspergillus oryzae during industrial koji making. J. Biosci. Bioeng. 2003, 95, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2015, 52, 676–684. [Google Scholar] [CrossRef]
- Prasadi, V.P.N.; Joye, I.J. Dietary Fibre from Whole Grains and Their Benefits on Metabolic Health. Nutrients 2020, 12, 3045. [Google Scholar] [CrossRef]
- Joyce, S.A.; Kamil, A.; Fleige, L.; Gahan, C.G.M. The Cholesterol-Lowering Effect of Oats and Oat Beta Glucan: Modes of Action and Potential Role of Bile Acids and the Microbiome. Front. Nutr. 2019, 6, 171. [Google Scholar] [CrossRef]
- Gupta, S.; Cox, S.; Abu-Ghannam, N. Process optimization for the development of a functional beverage based on lactic acid fermentation of oats. Biochem. Eng. J. 2010, 52, 199–204. [Google Scholar] [CrossRef]
- Bocchi, S.; Rocchetti, G.; Elli, M.; Lucini, L.; Lim, C.-Y.; Morelli, L. The combined effect of fermentation of lactic acid bacteria and in vitro digestion on metabolomic and oligosaccharide profile of oat beverage. Food Res. Int. 2021, 142, 110216. [Google Scholar] [CrossRef] [PubMed]
- Bernat, N.; Chafer, M.; Gonzalez-Martinez, C.; Rodriguez-Garcia, J.; Chiralt, A. Optimisation of oat milk formulation to obtain fermented derivatives by using probiotic Lactobacillus reuteri microorganisms. Food Sci. Technol. Int. 2015, 21, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Mårtensson, O.; Öste, R.; Holst, O. The effect of yoghurt culture on the survival of probiotic bacteria in oat-based, non-dairy products. Food Res. Int. 2002, 35, 775–784. [Google Scholar] [CrossRef]
- Jaskari, J.; Kontula, P.; Siitonen, A.; Jousimies-Somer, H.; Mattila-Sandholm, T.; Poutanen, K. Oat beta-glucan and xylan hydrolysates as selective substrates for Bifidobacterium and Lactobacillus strains. Appl. Microbiol. Biotechnol. 1998, 49, 175–181. [Google Scholar] [CrossRef]
- Bhanja Dey, T.; Chakraborty, S.; Jain, K.K.; Sharma, A.; Kuhad, R.C. Antioxidant phenolics and their microbial production by submerged and solid state fermentation process: A review. Trends Food Sci. Technol. 2016, 53, 60–74. [Google Scholar] [CrossRef]
- Jeng, W.Y.; Wang, N.C.; Lin, M.H.; Lin, C.T.; Liaw, Y.C.; Chang, W.J.; Liu, C.I.; Liang, P.H.; Wang, A.H. Structural and functional analysis of three β-glucosidases from bacterium Clostridium cellulovorans, fungus Trichoderma reesei and termite Neotermes koshunensis. J. Struct. Biol. 2011, 173, 46–56. [Google Scholar] [CrossRef]
- Hitayezu, R.; Baakdah, M.M.; Kinnin, J.; Henderson, K.; Tsopmo, A. Antioxidant activity, avenanthramide and phenolic acid contents of oat milling fractions. J. Cereal Sci. 2015, 63, 35–40. [Google Scholar] [CrossRef]
- Peterson, D.M.; Emmons, C.L.; Hibbs, A.H. Phenolic Antioxidants and Antioxidant Activity in Pearling Fractions of Oat Groats. J. Cereal Sci. 2001, 33, 97–103. [Google Scholar] [CrossRef]
- Tong, L.-t.; Liu, L.-y.; Zhong, K.; Wang, Y.; Guo, L.-n.; Zhou, S.-m. Effects of Cultivar on Phenolic Content and Antioxidant Activity of Naked Oat in China. J. Integr. Agric. 2014, 13, 1809–1816. [Google Scholar] [CrossRef]
- Hu, C.; Sang, S. Triterpenoid Saponins in Oat Bran and Their Levels in Commercial Oat Products. J. Agric. Food Chem. 2020, 68, 6381–6389. [Google Scholar] [CrossRef]
- Hu, C.; Tang, Y.; Zhao, Y.; Sang, S. Quantitative Analysis and Anti-inflammatory Activity Evaluation of the A-Type Avenanthramides in Commercial Sprouted Oat Products. J. Agric. Food Chem. 2020, 68, 13068–13075. [Google Scholar] [CrossRef]
- Yang, J.; Wang, P.; Wu, W.; Zhao, Y.; Idehen, E.; Sang, S. Steroidal Saponins in Oat Bran. J. Agric. Food Chem. 2016, 64, 1549–1556. [Google Scholar] [CrossRef]
- He, Z.; Zhang, H.; Wang, T.; Wang, R.; Luo, X. Effects of Five Different Lactic Acid Bacteria on Bioactive Components and Volatile Compounds of Oat. Foods 2022, 11, 3230. [Google Scholar] [CrossRef] [PubMed]
- Skoglund, M.; Peterson, D.M.; Andersson, R.; Nilsson, J.; Dimberg, L.H. Avenanthramide content and related enzyme activities in oats as affected by steeping and germination. J. Cereal Sci. 2008, 48, 294–303. [Google Scholar] [CrossRef]
- Gohari, S.T.; El-Batawy, O.I.; Mahdy, S.M. Biological Attributes of Unfermented and Fermented Oat Milk Compared to Cow Milk in Alloxan-Induced Diabetic Rats. J. Food Dairy. Sci. 2019, 10, 61–69. [Google Scholar] [CrossRef]
- Alharbi, H.F.; Algonaiman, R.; Barakat, H. Ameliorative and Antioxidative Potential of Lactobacillus plantarum-Fermented Oat (Avena sativa) and Fermented Oat Supplemented with Sidr Honey against Streptozotocin-Induced Type 2 Diabetes in Rats. Antioxidants 2022, 11, 1122. [Google Scholar] [CrossRef] [PubMed]
- Algonaiman, R.; Alharbi, H.F.; Barakat, H. Antidiabetic and Hypolipidemic Efficiency of Lactobacillus plantarum Fermented Oat (Avena sativa) Extract in Streptozotocin-Induced Diabetes in Rats. Fermentation 2022, 8, 267. [Google Scholar] [CrossRef]
- Lowe, M.E. Structure and function of pancreatic lipase and colipase. Annu. Rev. Nutr. 1997, 17, 141–158. [Google Scholar] [CrossRef]
- Thomson, A.B.R.; De Pover, A.; Keelan, M.; Jarocka-Cyrta, E.; Clandinin, M.T. Inhibition of lipid absorption as an approach to the treatment of obesity. Meth. Enzymol. 1997, 286, 3–44. [Google Scholar] [CrossRef]
- Cai, S.; Wang, O.; Wang, M.; He, J.; Wang, Y.; Zhang, D.; Zhou, F.; Ji, B. In vitro inhibitory effect on pancreatic lipase activity of subfractions from ethanol extracts of fermented Oats (Avena sativa L.) and synergistic effect of three phenolic acids. J. Agric. Food Chem. 2012, 60, 7245–7251. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, M.; Guo, X.; Bai, X.; Zhang, J.; Huo, R.; Zhang, Y. Improving the adsorption characteristics and antioxidant activity of oat bran by superfine grinding. Food Sci. Nutr. 2023, 11, 216–227. [Google Scholar] [CrossRef]
- Whitehead, A.; Beck, E.J.; Tosh, S.; Wolever, T.M. Cholesterol-lowering effects of oat β-glucan: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 100, 1413–1421. [Google Scholar] [CrossRef] [PubMed]
- Mårtensson, O.; Biörklund, M.; Lambo, A.M.; Dueñas-Chasco, M.; Irastorza, A.; Holst, O.; Norin, E.; Welling, G.; Öste, R.; Önning, G. Fermented, ropy, oat-based products reduce cholesterol levels and stimulate the bifidobacteria flora in humans. Nutr. Res. 2005, 25, 429–442. [Google Scholar] [CrossRef]
- Zhang, N.; Zhao, L.; Cai, S.; Zeng, X.; Wu, W.; Ji, B.; Zhou, F. Ethyl acetate subfractions from ethanol extracts of fermented oats (Avena sativa L.) exert anti-cancer properties in vitro and in vivo through G2/M and S Phase arrest and apoptosis. J. Cancer 2021, 12, 1853–1866. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Fasano, A. Celiac disease. Curr. Opin. Gastroenterol. 2008, 24, 687–691. [Google Scholar] [CrossRef]
- Arentz-Hansen, H.; Fleckenstein, B.; Molberg, Ø.; Scott, H.; Koning, F.; Jung, G.n.; Roepstorff, P.; Lundin, K.E.A.; Sollid, L.M.; Londei, M. The Molecular Basis for Oat Intolerance in Patients with Celiac Disease. PLoS Med. 2004, 1, e1. [Google Scholar] [CrossRef] [PubMed]
- Lundin, K.E.; Nilsen, E.M.; Scott, H.G.; Løberg, E.M.; Gjøen, A.; Bratlie, J.; Skar, V.; Mendez, E.; Løvik, A.; Kett, K. Oats induced villous atrophy in coeliac disease. Gut 2003, 52, 1649–1652. [Google Scholar] [CrossRef]
- Kaukinen, K.; Collin, P.; Huhtala, H.; Mäki, M. Long-term consumption of oats in adult celiac disease patients. Nutrients 2013, 5, 4380–4389. [Google Scholar] [CrossRef]
- Koskinen, O.; Villanen, M.; Korponay-Szabo, I.; Lindfors, K.; Mäki, M.; Kaukinen, K. Oats do not induce systemic or mucosal autoantibody response in children with coeliac disease. J. Pediatr. Gastroenterol. Nutr. 2009, 48, 559–565. [Google Scholar] [CrossRef]
- Lionetti, E.; Gatti, S.; Galeazzi, T.; Caporelli, N.; Francavilla, R.; Cucchiara, S.; Roggero, P.; Malamisura, B.; Iacono, G.; Tomarchio, S.; et al. Safety of Oats in Children with Celiac Disease: A Double-Blind, Randomized, Placebo-Controlled Trial. J. Pediatr. 2018, 194, 116–122. [Google Scholar] [CrossRef]
- Sey, M.S.; Parfitt, J.; Gregor, J. Prospective study of clinical and histological safety of pure and uncontaminated Canadian oats in the management of celiac disease. J. Parenter. Enteral Nutr. 2011, 35, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Hernando, A.; Mujico, J.R.; Juanas, D.; Méndez, E. Confirmation of the cereal type in oat products highly contaminated with gluten. J. Am. Diet. Assoc. 2006, 106, 665–666. [Google Scholar] [CrossRef] [PubMed]
- Hernando, A.; Mujico, J.R.; Mena, M.C.; Lombardía, M.; Méndez, E. Measurement of wheat gluten and barley hordeins in contaminated oats from Europe, the United States and Canada by Sandwich R5 ELISA. Eur. J. Gastroenterol. Hepatol. 2008, 20, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Thompson, T. Gluten contamination of commercial oat products in the United States. N. Engl. J. Med. 2004, 351, 2021–2022. [Google Scholar] [CrossRef]
- Koerner, T.B.; Cléroux, C.; Poirier, C.; Cantin, I.; Alimkulov, A.; Elamparo, H. Gluten contamination in the Canadian commercial oat supply. Food Addit. Contam. 2011, 28, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Vora, H.; McIntire, J.; Kumar, P.; Deshpande, M.; Khosla, C. A scaleable manufacturing process for pro-EP-B2, a cysteine protease from barley indicated for celiac sprue. Biotechnol. Bioeng. 2007, 98, 177–185. [Google Scholar] [CrossRef]
- Hu, C.; Tang, Y.; Snooks, H.D.; Sang, S. Novel Steroidal Saponins in Oat Identified by Molecular Networking Analysis and Their Levels in Commercial Oat Products. J. Agric. Food Chem. 2021, 69, 7084–7092. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djorgbenoo, R.; Hu, J.; Hu, C.; Sang, S. Fermented Oats as a Novel Functional Food. Nutrients 2023, 15, 3521. https://doi.org/10.3390/nu15163521
Djorgbenoo R, Hu J, Hu C, Sang S. Fermented Oats as a Novel Functional Food. Nutrients. 2023; 15(16):3521. https://doi.org/10.3390/nu15163521
Chicago/Turabian StyleDjorgbenoo, Richmond, Juanjuan Hu, Changling Hu, and Shengmin Sang. 2023. "Fermented Oats as a Novel Functional Food" Nutrients 15, no. 16: 3521. https://doi.org/10.3390/nu15163521
APA StyleDjorgbenoo, R., Hu, J., Hu, C., & Sang, S. (2023). Fermented Oats as a Novel Functional Food. Nutrients, 15(16), 3521. https://doi.org/10.3390/nu15163521





