Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development
Abstract
:1. Introduction
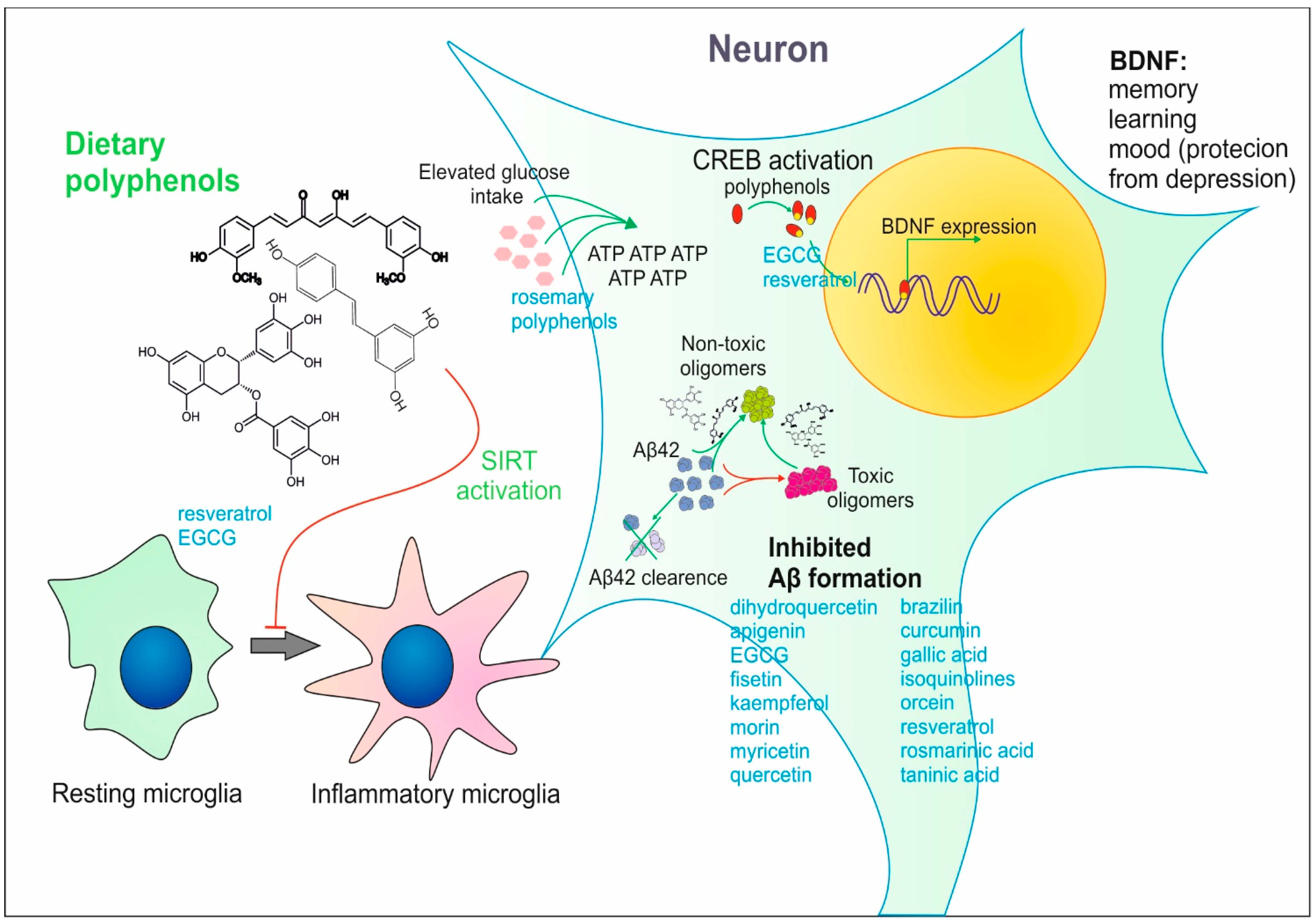
2. Effect of Dietary Polyphenols on Brain Activity
2.1. Impact of Various Polyphenols on Brain Functions—Animal Models
2.2. The Impact of Plant-Derived Polyphenols on the Brain and Cognitive Functions
| Study Objective | Study Group | Study Description | Main Results | References | |
|---|---|---|---|---|---|
| Interventional studies | To investigate the acute and subjective effects of cocoa flavanol (CF) consumption during mental demand | Healthy adults during mental demand | Randomized, controlled, double-blinded, balanced, three-period crossover trial; groups consuming 520 mg, 994 mg cocoa flavonol drinks, or matched control (0 mg) | Cognitive function improvement, attenuated mental fatigue | [14] |
| To investigate the effect of acute cocoa flavonol consumption in dark chocolate on visual and cognitive functions | Healthy young adults | Randomized, single-blinded, order counterbalanced, crossover study; participants intake 720 mg of cocoa flavonols in dark chocolate or a matched amount of white chocolate, one-week interval between sessions | CF improved visual contrast sensitivity and reduced the time required to detect motion direction, but had no statistically reliable effect on the minimum proportion of coherent motion that could be detected; in terms of cognitive performance, CF improved spatial memory and performance on some aspects of the choice reaction time task | [13] | |
| Evaluation of the long-term association between polyphenol intake and cognitive performance | 35–60-year-old women 45–60-year-old men | Participants in the SU.VI.MAX study (randomized, double-blind, placebo-controlled 8-year trial) were invited to perform 24-h dietary records every 2 months, records were randomly distributed across 2 weekend days and 4 weekdays in the year with all seasons and days covered, data collected with computerized questionnaires | Association between language and verbal memory improvement and high total polyphenol intake; catechins, theaflavins, flavonols, and hydroxybenzoic acids intake corresponded with better language and verbal memory performance, especially with episodic memory; negative association between executive functioning and dihydrochalcones, catechins, proanthocyanidins, and flavonols was observed | [15] | |
| Impact of chocolate, wine and tea flavonoids intake on cognitive performance | 70–74-year-old participants, 55% women | Cross-sectional study | Study participants consuming chocolate, tea, or wine had significantly better mean scores in cognitive tests, lower prevalence of weak test scores; dose-dependent effects with a maximum of 10 g/day (chocolate) 100 mL/day (wine), linear for tea | [16] | |
| Interventional studies | Evaluation of the effect of polyphenol extracts from grapes and blueberries on memory functions | 60–70-year-old healthy participants | Bicentric, randomized, double-blind, placebo-controlled; study group received 258 mg of flavonols/day or placebo for 6 months | Improvement in verbal episodic and recognition memory tests; improved test results in groups with advanced cognitive decline in response to polyphenols intake | [17] |
| Analysis of the short-term efficacy of cranberry juice intake on neuropsychologic functions in the elderly without cognitive deficits | 50 participants, >60 years old, without cognitive deficits or dementia | Randomized, double-blind, placebo-controlled, conducted in parallel groups, 6-week study; participants randomly allocated to study groups obtained 245 g of cranberry juice (n = 25) or placebo (n = 25) daily; neuropsychologic tests before the interventions and after 6 weeks of the study | No significant actions were reported | [18] | |
| Impact of concord grape juice on neurocognition. | 12 older adults with memory decline, dementia excluded | Randomized, placebo-controlled, double-blind trial, 12 weeks concord juice supplementation | Significantly improved verbal learning Increased verbal and spatial recall No effect on depressive symptoms or weight | [20] | |
| Impact of daily consumption of wild blueberry juice on neurocognition. | 9 adults with early memory changes | Randomized, placebo-controlled, double-blind trial, 12 weeks of blueberry juice supplementation | Improved paired associate learning, word list recall, Trends suggesting reduced depressive symptoms (not significant) | [19] | |
| Effect of Pycnogenol on ADHD symptoms | 61 children | Randomized, placebo-controlled, double-blind trial, duration 4 weeks, daily supplementation of pycnogenol in dose of 1 mg/kg; children examined before supplementation, 1 month after start, 1 month after end of the trial | 1 month after starting, significant reduction of hyperreactivity in ADHD-affected children; improved attention, motoric coordination, and concentration 1 month after the end of supplementation: relapse of the symptoms | [45] | |
| Assessing safety, tolerability, and efficacy of green tea polyphenols | 410 untreated participants with Parkinson’s disease, disease duration no longer than 5 years | Multicenter, double-blind, randomized, placebo-controlled, 12 month study, tests at the beginning, after 6 months and at the end of the study; not heavy tea drinkers were randomly allocated to groups obtaining 400, 800, or 1200 mg of green tea polyphenols daily, given orally in two doses | Contradictory results; beneficial effect observed after 6 months of the intervention was abolished after 12 months | [23] | |
| Observational studies | Role of polyphenols intake profile in dementia development | 1329 participants, mean age 78 | Observational, nonintervention, 12-year follow-up study | Lower dementia and Alzheimer’s disease development risk was associated with a diet containing polyphenols from nuts, citrus, berries, leafy vegetables, soy, cereals, and olive oil accompanied by tea and red wine | [56] |
| Examination of the role of flavonoids (and their subclasses) intake on the risk of PD development | 49,281 men 80,336 women participants from the NHS (Nurses’ Health Study) and HPFS (Health Professionals Follow-up Study) programs | Progressive, nonintervention 20–22-year follow-up study Questionnaires among the studied groups | In the men’s group high flavonoids intake was linked with a 40% lower PD risk as compared with participants consuming low flavonoids quantities. Anthocyanins from berry consumption lower the risk of PD development | [60] |
2.3. The Impact on Cognitive Functions in Humans
3. Direct and Indirect Action of Plant Polyphenols
4. Availability of Polyphenols in the Brain Tissue
4.1. BBB Reduces the Direct Impact of Dietary Polyphenols on Neurons
4.2. Polyphenol Transport across the Blood-Brain Barrier
4.3. Evidence from Human Studies on the Passage of Polyphenols through the BBB
5. Nanotechnological Solutions to Improve the Bioavailability of Polyphenols in the Brain
5.1. Nanoencapsulation
5.2. Natural Nano-Carriers
5.3. Solid Lipid Nanoparticles (SLNs)
5.4. Polymeric Nanoparticles
5.5. Liposomes
6. Plausible Adverse Effects of Polyphenol Supplementation
7. Factors That May Influence the Efficacy of Polyphenols in the Prevention of Neurodegenerative Diseases
8. Conclusions and Perspectives
- Indications and possible contraindications
- Optimal composition of the polyphenol mixture
- Dose and duration of treatment
- Optimal bioavailability of polyphenols
- Nanoformulation to increase phytochemical bioavailability and delivery to the CNS, including transportation through the BBB
- Effectiveness of preparations composed of postbiotic polyphenol metabolites or polyphenols pretreated with hydrolytic enzymes
- Evaluation of possible side effects
- Pharmacokinetics in human blood and brain tissue—dependence between plasma concentrations and concentrations in brain tissue or cerebrospinal fluid.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 6-OHDA | 6-hydroxydopamine |
| Aβ | beta-amyloid |
| ABC | ATP-binding cassette |
| AD | Alzheimer’s disease |
| ADHD | attention deficit hyperactivity disorder |
| AlCl3 | aluminium chloride |
| ALM | amyotrophic lateral sclerosis |
| α-M | α-mangostin |
| APP | amyloid precursor protein |
| APP/PS1 | double transgenic mice expressing a chimeric mouse/human amyloid precursor protein and a mutant human presenilin 1 |
| AUC | the area under the plasma concentration time curve |
| AVLT | auditory verbal learning test |
| AWR | abdominal withdrawal reflex |
| BAX | Bcl-2-associated X protein |
| BBB | blood-brain barrier |
| BCRP | breast cancer resistance protein |
| BCL2 | B-cell lymphoma 2 |
| BDNF | brain-derived neurotrophic factor |
| bEnd.3 | brain endothelium cell line, cells isolated from brain tissue derived from a mouse with endothelioma |
| BMI | body mass index |
| CA | caffeic acid |
| CANTAB | the Cambridge neuropsychological test automated battery |
| CAT | catalase |
| CACS | chronic acute combining stress |
| CF | cocoa flavanol |
| CNS | central nervous system |
| CREB | cAMP response element-binding protein |
| CSF | cerebrospinal fluid |
| C-SLNs | curcumin-loaded solid lipid nanoparticles |
| DND | delayed neuronal cell death |
| EGCG | epigallocatechin-3-gallate |
| ERK ½ | extracellular signal-regulated kinase ½ |
| ETC | electron-transported chain |
| FDA | the Food and Drug Administration |
| GMO | glyceryl monooleate |
| GPX1 | glutathione peroxidase 1 |
| 3GRES | 3-O-glucuronidated-resveratrol |
| 4GRES | 4-O-glucuronidated-resveratrol |
| GSH | glutathione |
| HD | Huntington’s disease |
| HPLC-MS2 | High-performance liquid chromatography-tandem mass spectrometry |
| 5-HT | 5-hydroxytryptamine |
| 6-OHDA | 6-hydroxydopamine |
| IBS | irritable bowel syndrome |
| ITM | intestinal tract motility |
| JNK | c-Jun N-terminal kinase |
| LPS | lipopolysaccharide |
| LSP | lychee seed polyphenols |
| MANT | modified attention network task |
| MAO | monoamine oxidase |
| MCAO | middle cerebral artery occlusion |
| MCP-1 | monocyte chemoattractant protein-1 |
| MMSE | mini-mental state examination |
| MRP2 | multidrug-resistant protein type 2 |
| MWM | Moris water maze |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NanoCurc | nanoparticle-encapsulated curcumin |
| NFkB | nuclear factor kappa-light chain enhancer of activated B cells |
| NSC | neural stem cells |
| NO | nitric oxide |
| OX26 | antitransferrin receptor antibody |
| PACA | poly (alkyl cyanoacrylate) |
| PAL | paired associate learning |
| PD | Parkinson’s disease |
| PEGB | polyphenol-rich grape and blueberry extract |
| PGA | poly (glycolic acid) |
| P-gp | P-glycoprotein |
| PLA | poly (lactic acid) |
| PLGA | poly (D, L-lactide-co-glycolic acid) |
| PLGA@QT | PLGA-functionalized quercetin nanoparticles |
| PSA | polysaccharide A |
| QNPs | quercetin nanoparticles |
| QT-SPIONs | quercetin-conjugated superparamagnetic iron oxide nanoparticles |
| RAWM | radial arm water maze |
| RCTs | randomized control trials |
| ROS | reactive oxygen species |
| SCFA | short-chain fatty acids |
| SH-SY5Y | a thrice-subcloned cell line derived from the SK-N-SH neuroblastoma cell line. It serves as a model for neurodegenerative disorders |
| SIRT1 | Sirtuin 1 |
| SK-N-SH | neuroblastoma cell line |
| SLNs | solid lipid nanoparticles |
| SOD1 | superoxide dismutase 1 |
| SRES | 3-sulfated-resveratrol |
| Tet-1 | ten-eleven translocation methylcytosine dioxygenase 1 |
| Tf(α-M) | α-M liposomes modified with transferrin |
| TfR | transferrin receptor |
| TLR4/NFkB | toll-like receptor 4/nuclear factor kappa B |
| TNF-alpha | tumor necrosis factor–alpha |
| TOWRE-2 | Test of Word Reading Efficiency—second edition |
| WBB | flavonoid rich wild blueberry |
References
- Chen, X.; Guo, C.; Kong, J. Oxidative stress in neurodegenerative diseases. Neural Regen. Res. 2012, 7, 376–385. [Google Scholar] [PubMed]
- Berr, C. Cognitive impairment and oxidative stress in the elderly: Results of epidemiological studies. BioFactors 2000, 13, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Head, E. Oxidative Damage and Cognitive Dysfunction: Antioxidant Treatments to Promote Healthy Brain Aging. Neurochem. Res. 2009, 34, 670–678. [Google Scholar] [CrossRef] [Green Version]
- Uttara, B.; Singh, V.A.; Zamboni, P.; Mahajan, T.R. Oxidative Stress and Neurodegenerative Diseases: A Review of Upstream and Downstream Antioxidant Therapeutic Options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Małkiewicz, M.A.; Szarmach, A.; Sabisz, A.; Cubała, W.J.; Szurowska, E.; Winklewski, P.J. Blood-brain barrier permeability and physical exercise. J. Neuroinflamm. 2019, 16, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, G.H.; Kim, J.E.; Rhie, S.J.; Yoon, S. The Role of Oxidative Stress in Neurodegenerative Diseases. Exp. Neurobiol. 2015, 24, 325–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Lorenzo, C.; Colombo, F.; Biella, S.; Stockley, C.; Restani, P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients 2021, 13, 273. [Google Scholar] [CrossRef]
- Figueira, I.; Garcia, G.; Pimpão, R.C.; Terrasso, A.P.; Costa, I.; Almeida, A.F.; Tavares, L.; Pais, T.F.; Pinto, P.; Ventura, M.R.; et al. Polyphenols journey through blood-brain barrier towards neuronal protection. Sci. Rep. 2017, 7, 11456. [Google Scholar] [CrossRef] [Green Version]
- Faria, A.; Mateus, N.; Calhau, C. Flavonoid transport across blood-brain barrier: Implication for their direct neuroprotective actions. Nutr. Aging 2012, 1, 89–97. [Google Scholar] [CrossRef]
- Cortes-Canteli, M.; Iadecola, C. Alzheimer’s Disease and Vascular Aging: JACC Focus Seminar. J. Am. Coll. Cardiol. 2020, 75, 942–951. [Google Scholar] [CrossRef]
- Niccoli, T.; Partridge, L. Ageing as a Risk Factor for Disease. Curr. Biol. 2012, 22, R741–R752. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.-K. Antiaging agents: Safe interventions to slow aging and healthy life span extension. Nat. Prod. Bioprospect. 2022, 12, 18. [Google Scholar] [CrossRef] [PubMed]
- Field, D.T.; Williams, C.M.; Butler, L.T. Consumption of cocoa flavanols results in an acute improvement in visual and cognitive functions. Physiol. Behav. 2011, 103, 255–260. [Google Scholar] [CrossRef] [Green Version]
- Scholey, A.B.; French, S.J.; Morris, P.J.; Kennedy, D.O.; Milne, A.L.; Haskell, C.F. Consumption of cocoa flavanols results in acute improvements in mood and cognitive performance during sustained mental effort. J. Psychopharmacol. 2009, 24, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Fezeu, L.; Andreeva, V.A.; Touvier, M.; Scalbert, A.; Hercberg, S.; Galan, P. Total and Specific Polyphenol Intakes in Midlife Are Associated with Cognitive Function Measured 13 Years Later. J. Nutr. 2012, 142, 76–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nurk, E.; Refsum, H.; Drevon, C.A.; Tell, G.S.; Nygaard, H.A.; Engedal, K.; Smith, A.D. Intake of Flavonoid-Rich Wine, Tea, and Chocolate by Elderly Men and Women Is Associated with Better Cognitive Test Performance. J. Nutr. 2009, 139, 120–127. [Google Scholar] [CrossRef] [Green Version]
- Bensalem, J.; Dudonné, S.; Etchamendy, N.; Pellay, H.; Amadieu, C.; Gaudout, D.; Dubreuil, S.; Paradis, M.-E.; Pomerleau, S.; Capuron, L.; et al. Polyphenols From Grape and Blueberry Improve Episodic Memory in Healthy Elderly with Lower Level of Memory Performance: A Bicentric Double-Blind, Randomized, Placebo-Controlled Clinical Study. J. Gerontol. Ser. A 2019, 74, 996–1007. [Google Scholar] [CrossRef]
- Crews, W.D.; Harrison, D.W.; Griffin, M.L.; Addison, K.; Yount, A.M.; Giovenco, M.A.; Hazell, J. A Double-Blinded, Placebo-Controlled, Randomized Trial of the Neuropsychologic Efficacy of Cranberry Juice in a Sample of Cognitively Intact Older Adults: Pilot Study Findings. J. Altern. Complement. Med. 2005, 11, 305–309. [Google Scholar] [CrossRef] [Green Version]
- Krikorian, R.; Shidler, M.D.; Nash, T.A.; Kalt, W.; Vinqvist-Tymchuk, M.R.; Shukitt-Hale, B.; Joseph, J.A. Blueberry Supplementation Improves Memory in Older Adults. J. Agric. Food Chem. 2010, 58, 3996–4000. [Google Scholar] [CrossRef] [Green Version]
- Krikorian, R.; Nash, T.A.; Shidler, M.D.; Shukitt-Hale, B.; Joseph, J.A. Concord grape juice supplementation improves memory function in older adults with mild cognitive impairment. Br. J. Nutr. 2010, 103, 730–734. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.; Yamashita, T.; Hu, X.; Bian, Z.; Hu, X.; Feng, T.; Tadokoro, K.; Morihara, R.; Abe, K. Protective and anti-oxidative effects of curcumin and resveratrol on Aβ-oligomer-induced damage in the SH-SY5Y cell line. J. Neurol. Sci. 2022, 441, 120356. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Liu, M.; Yao, W.; Du, K.; He, M.; Jin, X.; Jiao, L.; Ma, G.; Wei, B.; Wei, M. Epigallocatechin-3-Gallate Attenuates Microglial Inflammation and Neurotoxicity by Suppressing the Activation of Canonical and Noncanonical Inflammasome via TLR4/NF-κB Pathway. Mol. Nutr. Food Res. 2019, 63, 1801230. [Google Scholar] [CrossRef]
- Chan, P.; Qin, Z.; Zheng, Z.; Zhang, L.; Fang, X.; Sun, F.; Gu, Z.; Chen, S.; Ma, J.; Meng, C.; et al. P2.204 A randomized, double-blind, placebo-controlled, delayed start study to assess safty, tolerability and efflcacy of green tea polyphenols in Parkinson’s disease. Park. Relat. Disord. 2009, 15, S145. [Google Scholar] [CrossRef]
- Kobayashi, H.; Murata, M.; Kawanishi, S.; Oikawa, S. Polyphenols with Anti-Amyloid β Aggregation Show Potential Risk of Toxicity Via Pro-Oxidant Properties. Int. J. Mol. Sci. 2020, 21, 3561. [Google Scholar] [CrossRef] [PubMed]
- Pandareesh, M.D.; Mythri, R.B.; Srinivas Bharath, M.M. Review. Neurochem. Int. 2015, 89, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Figueira, I.; Menezes, R.; Macedo, D.; Costa, I.; dos Santos, N.C. Polyphenols Beyond Barriers: A Glimpse into the Brain. Curr. Neuropharmacol. 2017, 15, 562–594. [Google Scholar] [CrossRef] [Green Version]
- Gomez-Pinilla, F.; Nguyen, T.T.J. Natural mood foods: The actions of polyphenols against psychiatric and cognitive disorders. Nutr. Neurosci. 2012, 15, 127–133. [Google Scholar] [CrossRef] [Green Version]
- Finsterwald, C.; Fiumelli, H.; Cardinaux, J.-R.; Martin, J.-L. Regulation of Dendritic Development by BDNF Requires Activation of CRTC1 by Glutamate. J. Biol. Chem. 2010, 285, 28587–28595. [Google Scholar] [CrossRef] [Green Version]
- Casadesus, G.; Shukitt-Hale, B.; Stellwagen, H.M.; Zhu, X.; Lee, H.-G.; Smith, M.A.; Joseph, J.A. Modulation of Hippocampal Plasticity and Cognitive Behavior by Short-term Blueberry Supplementation in Aged Rats. Nutr. Neurosci. 2004, 7, 309–316. [Google Scholar] [CrossRef]
- Karuppagounder, S.S.; Madathil, S.K.; Pandey, M.; Haobam, R.; Rajamma, U.; Mohanakumar, K.P. Quercetin up-regulates mitochondrial complex-I activity to protect against programmed cell death in rotenone model of Parkinson’s disease in rats. Neuroscience 2013, 236, 136–148. [Google Scholar] [CrossRef]
- Kulkarni, S.K.; Bhutani, M.K.; Bishnoi, M. Antidepressant activity of curcumin: Involvement of serotonin and dopamine system. Psychopharmacology 2008, 201, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Ku, B.S.; Tie, L.; Yao, H.Y.; Jiang, W.G.; Ma, X.; Li, X.J. Curcumin reverses the effects of chronic stress on behavior, the HPA axis, BDNF expression and phosphorylation of CREB. Brain Res. 2006, 1122, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.M.; El Mohsen, M.A.; Vauzour, D.; Rendeiro, C.; Butler, L.T.; Ellis, J.A.; Whiteman, M.; Spencer, J.P.E. Blueberry-induced changes in spatial working memory correlate with changes in hippocampal CREB phosphorylation and brain-derived neurotrophic factor (BDNF) levels. Free Radic. Biol. Med. 2008, 45, 295–305. [Google Scholar] [CrossRef]
- Mai, C.; Qiu, L.; Zeng, Y.; He, Y. Yunnan Black Tea Flavonoids Can Improve Cognitive Dysfunction in Septic Mice by Activating SIRT1. Evid.-Based Complement. Altern. Med. 2021, 2021, 5775040. [Google Scholar] [CrossRef]
- Chan, Y.-C.; Hosoda, K.; Tsai, C.-J.; Yamamoto, S.; Wang, M.-F. Favorable Effects of Tea on Reducing the Cognitive Deficits and Brain Morphological Changes in Senescence-Accelerated Mice. J. Nutr. Sci. Vitaminol. 2006, 52, 266–273. [Google Scholar] [CrossRef] [Green Version]
- Hamaguchi, T.; Ono, K.; Murase, A.; Yamada, M. Phenolic Compounds Prevent Alzheimer’s Pathology through Different Effects on the Amyloid-β Aggregation Pathway. Am. J. Pathol. 2009, 175, 2557–2565. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Xu, J.; Rottinghaus, G.E.; Simonyi, A.; Lubahn, D.; Sun, G.Y.; Sun, A.Y. Resveratrol protects against global cerebral ischemic injury in gerbils. Brain Res. 2002, 958, 439–447. [Google Scholar] [CrossRef]
- Tang, X.l.; Wang, X.; Fang, G.; Zhao, Y.L.; Yan, J.; Zhou, Z.; Sun, R.; Luo, A.I.; Li, S.Y. Resveratrol ameliorates sevoflurane-induced cognitive impairment by activating the SIRT1/NF-κB pathway in neonatal mice. J. Nutr. Biochem. 2021, 90, 108579. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, Y.; Zhao, Z.; Xue, Y. Effects of green tea polyphenols on caveolin-1 of microvessel fragments in rats with cerebral ischemia. Neurol. Res. 2010, 32, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Bian, Y.; Yamashita, T.; Taira, Y.; Shang, J.; Tsunoda, K.; Feng, T.; Sasaki, R.; Liu, X.; Shi, X.; Tadokoro, K.; et al. A Polyphenolic Complex Attenuates Inflammatory Response and Blood- Brain Barrier Disruption. Curr. Neurovasc. Res. 2020, 17, 286–293. [Google Scholar] [CrossRef]
- Xiong, R.; Zhou, X.-G.; Tang, Y.; Wu, J.-M.; Sun, Y.-S.; Teng, J.-F.; Pan, R.; Law, B.Y.-K.; Zhao, Y.; Qiu, W.-Q.; et al. Lychee seed polyphenol protects the blood–brain barrier through inhibiting Aβ(25–35)-induced NLRP3 inflammasome activation via the AMPK/mTOR/ULK1-mediated autophagy in bEnd.3 cells and APP/PS1 mice. Phyther. Res. 2021, 35, 954–973. [Google Scholar] [CrossRef]
- Yu, Y.-C.; Li, J.; Zhang, M.; Pan, J.-C.; Yu, Y.; Zhang, J.-B.; Zheng, L.; Si, J.; Xu, Y. Resveratrol Improves Brain-Gut Axis by Regulation of 5-HT-Dependent Signaling in the Rat Model of Irritable Bowel Syndrome. Front. Cell. Neurosci. 2019, 13, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinheiro Fernandes, F.D.; Fontenele Menezes, A.P.; de Sousa Neves, J.C.; Fonteles, A.A.; da Silva, A.T.A.; de Araújo Rodrigues, P.; Santos do Carmo, M.R.; de Souza, C.M.; de Andrade, G.M. Caffeic acid protects mice from memory deficits induced by focal cerebral ischemia. Behav. Pharmacol. 2014, 25, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hamada, H.; Gerk, P.M. Selectivity of Dietary Phenolics for Inhibition of Human Monoamine Oxidases A and B. Biomed Res. Int. 2019, 2019, 8361858. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trebatická, J.; Kopasová, S.; Hradečná, Z.; Činovský, K.; Škodáček, I.; Šuba, J.; Muchová, J.; Žitňanová, I.; Waczulíková, I.; Rohdewald, P.; et al. Treatment of ADHD with French maritime pine bark extract, Pycnogenol®. Eur. Child Adolesc. Psychiatry 2006, 15, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Carbone, M.; Lulli, D.; Passarelli, F.; Pastore, S. Topical Plant Polyphenols Prevent Type I Interferon Signaling in the Skin and Suppress Contact Hypersensitivity. Int. J. Mol. Sci. 2018, 19, 2652. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Suwanwela, N.C.; Patumraj, S. Curcumin by down-regulating NF-kB and elevating Nrf2, reduces brain edema and neurological dysfunction after cerebral I/R. Microvasc. Res. 2016, 106, 117–127. [Google Scholar] [CrossRef]
- Hou, W.; Hu, S.; Su, Z.; Wang, Q.; Meng, G.; Guo, T.; Zhang, J.; Gao, P. Myricetin attenuates LPS-induced inflammation in RAW 264.7 macrophages and mouse models. Future Med. Chem. 2018, 10, 2253–2264. [Google Scholar] [CrossRef]
- Testa, G.; Gamba, P.; Badilli, U.; Gargiulo, S.; Maina, M.; Guina, T.; Calfapietra, S.; Biasi, F.; Cavalli, R.; Poli, G.; et al. Loading into Nanoparticles Improves Quercetin’s Efficacy in Preventing Neuroinflammation Induced by Oxysterols. PLoS ONE 2014, 9, e96795. [Google Scholar] [CrossRef] [Green Version]
- Ishisaka, A.; Mukai, R.; Terao, J.; Shibata, N.; Kawai, Y. Specific localization of quercetin-3-O-glucuronide in human brain. Arch. Biochem. Biophys. 2014, 557, 11–17. [Google Scholar] [CrossRef]
- Wang, J.; Du, X.-X.; Jiang, H.; Xie, J.-X. Curcumin attenuates 6-hydroxydopamine-induced cytotoxicity by anti-oxidation and nuclear factor-kappaB modulation in MES23.5 cells. Biochem. Pharmacol. 2009, 78, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Velander, P.; Wu, L.; Henderson, F.; Zhang, S.; Bevan, D.R.; Xu, B. Natural product-based amyloid inhibitors. Biochem. Pharmacol. 2017, 139, 40–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamada, M.; Ono, K.; Hamaguchi, T.; Noguchi-Shinohara, M. Natural Phenolic Compounds as Therapeutic and Preventive Agents for Cerebral Amyloidosis. In Natural Compounds as Therapeutic Agents for Amyloidogenic Diseases; Vassallo, N., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 79–94. ISBN 978-3-319-18365-7. [Google Scholar]
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; et al. Curcumin Inhibits Formation of Amyloid β Oligomers and Fibrils, Binds Plaques, and Reduces Amyloid in Vivo. J. Biol. Chem. 2005, 280, 5892–5901. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rane, J.S.; Bhaumik, P.; Panda, D. Curcumin Inhibits Tau Aggregation and Disintegrates Preformed Tau Filaments in vitro. J. Alzheimer’s Dis. 2017, 60, 999–1014. [Google Scholar] [CrossRef] [PubMed]
- Lefèvre-Arbogast, S.; Gaudout, D.; Bensalem, J.; Letenneur, L.; Dartigues, J.-F.; Hejblum, B.P.; Féart, C.; Delcourt, C.; Samieri, C. Pattern of polyphenol intake and the long-term risk of dementia in older persons. Neurology 2018, 90, e1979–e1988. [Google Scholar] [CrossRef] [Green Version]
- Aryal, S.; Skinner, T.; Bridges, B.; Weber, J.T. The Pathology of Parkinson’s Disease and Potential Benefit of Dietary Polyphenols. Molecules 2020, 25, 4382. [Google Scholar] [CrossRef]
- Bianchi, V.E.; Rizzi, L.; Somaa, F. The role of nutrition on Parkinson’s disease: A systematic review. Nutr. Neurosci. 2022, 26, 605–628. [Google Scholar] [CrossRef]
- Lu, Z.; Nie, G.; Belton, P.S.; Tang, H.; Zhao, B. Structure–activity relationship analysis of antioxidant ability and neuroprotective effect of gallic acid derivatives. Neurochem. Int. 2006, 48, 263–274. [Google Scholar] [CrossRef]
- Gao, X.; Cassidy, A.; Schwarzschild, M.A.; Rimm, E.B.; Ascherio, A. Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology 2012, 78, 1138–1145. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.-H.; Liu, Y.; Xue, N.-J.; Zheng, R.; Yan, Y.-Q.; Wang, Z.-X.; Li, Y.-L.; Ying, C.-Z.; Song, Z.; Tian, J.; et al. Quercetin Protects against MPP+/MPTP-Induced Dopaminergic Neuron Death in Parkinson’s Disease by Inhibiting Ferroptosis. Oxid. Med. Cell. Longev. 2022, 2022, 7769355. [Google Scholar] [CrossRef]
- Barfoot, K.L.; May, G.; Lamport, D.J.; Ricketts, J.; Riddell, P.M.; Williams, C.M. The effects of acute wild blueberry supplementation on the cognition of 7–10-year-old schoolchildren. Eur. J. Nutr. 2019, 58, 2911–2920. [Google Scholar] [CrossRef] [Green Version]
- Whyte, A.R.; Cheng, N.; Butler, L.T.; Lamport, D.J.; Williams, C.M. Flavonoid-Rich Mixed Berries Maintain and Improve Cognitive Function Over a 6 h Period in Young Healthy Adults. Nutrients 2019, 11, 2685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Godos, J.; Caraci, F.; Castellano, S.; Currenti, W.; Galvano, F.; Ferri, R.; Grosso, G. Association Between Dietary Flavonoids Intake and Cognitive Function in an Italian Cohort. Biomolecules 2020, 10, 1300. [Google Scholar] [CrossRef]
- Dias, I.R.; de Sousa Santos, C.; e Magalhães, C.O.D.; de Oliveira, L.R.S.; Peixoto, M.F.D.; De Sousa, R.A.L.; Cassilhas, R.C. Does calorie restriction improve cognition? IBRO Rep. 2020, 9, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Carmona-Gutierrez, D.; Hofer, S.J.; Kroemer, G. Caloric Restriction Mimetics against Age-Associated Disease: Targets, Mechanisms, and Therapeutic Potential. Cell Metab. 2019, 29, 592–610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bensalem, J.; Dudonné, S.; Gaudout, D.; Servant, L.; Calon, F.; Desjardins, Y.; Layé, S.; Lafenetre, P.; Pallet, V. Polyphenol-rich extract from grape and blueberry attenuates cognitive decline and improves neuronal function in aged mice. J. Nutr. Sci. 2018, 7, e19. [Google Scholar] [CrossRef] [Green Version]
- Grabska-Kobylecka, I.; Kaczmarek-Bak, J.; Figlus, M.; Prymont-Przyminska, A.; Zwolinska, A.; Sarniak, A.; Wlodarczyk, A.; Glabinski, A.; Nowak, D. The Presence of Caffeic Acid in Cerebrospinal Fluid: Evidence That Dietary Polyphenols Can Cross the Blood-Brain Barrier in Humans. Nutrients 2020, 12, 1531. [Google Scholar] [CrossRef]
- Lee, B.; Shim, I.; Lee, H.; Hahm, D.-H. Effects of Epigallocatechin Gallate on Behavioral and Cognitive Impairments, Hypothalamic–Pituitary–Adrenal Axis Dysfunction, and Alternations in Hippocampal BDNF Expression Under Single Prolonged Stress. J. Med. Food 2018, 21, 979–989. [Google Scholar] [CrossRef]
- Rothenberg, D.O.; Zhang, L. Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption. Nutrients 2019, 11, 1361. [Google Scholar] [CrossRef] [Green Version]
- Zaletel, I.; Filipović, D.; Puškaš, N. Hippocampal BDNF in physiological conditions and social isolation. Rev. Neurosci. 2017, 28, 675–692. [Google Scholar] [CrossRef]
- Baron, D.C.; Marko, D.M.; Tsiani, E.; MacPherson, R.E.K. Rosemary extract increases neuronal cell glucose uptake and activates AMPK. Appl. Physiol. Nutr. Metab. 2020, 46, 141–147. [Google Scholar] [CrossRef]
- D’Angelo, S.; Mele, E.; Di Filippo, F.; Viggiano, A.; Meccariello, R. Sirt1 Activity in the Brain: Simultaneous Effects on Energy Homeostasis and Reproduction. Int. J. Environ. Res. Public Health 2021, 18, 1243. [Google Scholar] [CrossRef] [PubMed]
- Karunaweera, N.; Raju, R.; Gyengesi, E.; Münch, G. Plant polyphenols as inhibitors of NF-κB induced cytokine production—A potential anti-inflammatory treatment for Alzheimer’s disease? Front. Mol. Neurosci. 2015, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Sonawane, S.K.; Uversky, V.N.; Chinnathambi, S. Baicalein inhibits heparin-induced Tau aggregation by initializing non-toxic Tau oligomer formation. Cell Commun. Signal. 2021, 19, 16. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Li, Z.; Rizak, J.D.; Wu, S.; Wang, Z.; He, R.; Su, M.; Qin, D.; Wang, J.; Hu, X. Resveratrol Attenuates Formaldehyde Induced Hyperphosphorylation of Tau Protein and Cytotoxicity in N2a Cells. Front. Neurosci. 2017, 10, 598. [Google Scholar] [CrossRef] [Green Version]
- Shen, X.; Luo, T.; Li, S.; Ting, O.; He, F.; Xu, J.; Wang, H. Quercetin inhibits okadaic acid-induced tau protein hyperphosphorylation through the Ca2+-calpain-p25-CDK5 pathway in HT22 cells. Int. J. Mol. Med. 2018, 41, 1138–1146. [Google Scholar] [CrossRef] [Green Version]
- Tahir, M.S.; Almezgagi, M.; Zhang, Y.; Bashir, A.; Abdullah, H.M.; Gamah, M.; Wang, X.; Zhu, Q.; Shen, X.; Ma, Q.; et al. Mechanistic new insights of flavonols on neurodegenerative diseases. Biomed. Pharmacother. 2021, 137, 111253. [Google Scholar] [CrossRef]
- Haskell-Ramsay, C.F.; Schmitt, J.; Actis-Goretta, L. The Impact of Epicatechin on Human Cognition: The Role of Cerebral Blood Flow. Nutrients 2018, 10, 986. [Google Scholar] [CrossRef] [Green Version]
- Fraga, C.G.; Litterio, M.C.; Prince, P.D.; Calabró, V.; Piotrkowski, B.; Galleano, M. Cocoa flavanols: Effects on vascular nitric oxide and blood pressure. J. Clin. Biochem. Nutr. 2010, 48, 63–67. [Google Scholar] [CrossRef] [Green Version]
- Rendeiro, C.; Rhodes, J.S.; Spencer, J.P.E. The mechanisms of action of flavonoids in the brain: Direct versus indirect effects. Neurochem. Int. 2015, 89, 126–139. [Google Scholar] [CrossRef]
- Marunaka, Y.; Marunaka, R.; Sun, H.; Yamamoto, T.; Kanamura, N.; Inui, T.; Taruno, A. Actions of Quercetin, a Polyphenol, on Blood Pressure. Molecules 2017, 22, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, Y.-C.; Sheen, J.-M.; Hu, W.L.; Hung, Y.-C. Polyphenols and Oxidative Stress in Atherosclerosis-Related Ischemic Heart Disease and Stroke. Oxid. Med. Cell. Longev. 2017, 2017, 8526438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez-Mateos, A.; Rendeiro, C.; Bergillos-Meca, T.; Tabatabaee, S.; George, T.W.; Heiss, C.; Spencer, J.P.E. Intake and time dependence of blueberry flavonoid–induced improvements in vascular function: A randomized, controlled, double-blind, crossover intervention study with mechanistic insights into biological activity. Am. J. Clin. Nutr. 2013, 98, 1179–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Widlansky, M.E.; Gokce, N.; Keaney, J.F.; Vita, J.A. The clinical implications of endothelial dysfunction. J. Am. Coll. Cardiol. 2003, 42, 1149–1160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serra, D.; Almeida, L.M.; Dinis, T.C.P. Chapter One—Polyphenols in the management of brain disorders: Modulation of the microbiota-gut-brain axis. In Advances in Food and Nutrition Research; Toldrá, F., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 91, pp. 1–27. ISBN 1043-4526. [Google Scholar]
- Ramakrishna, C.; Kujawski, M.; Chu, H.; Li, L.; Mazmanian, S.K.; Cantin, E.M. Bacteroides fragilis polysaccharide A induces IL-10 secreting B and T cells that prevent viral encephalitis. Nat. Commun. 2019, 10, 2153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, Y.-C.; Ching, Y.-H.; Chiu, C.-C.; Liu, J.-Y.; Hung, S.-W.; Huang, W.-C.; Huang, Y.-T.; Chuang, H.-L. TLR2 and interleukin-10 are involved in Bacteroides fragilis-mediated prevention of DSS-induced colitis in gnotobiotic mice. PLoS ONE 2017, 12, e0180025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ochoa-Repáraz, J.; Mielcarz, D.W.; Wang, Y.; Begum-Haque, S.; Dasgupta, S.; Kasper, D.L.; Kasper, L.H. A polysaccharide from the human commensal Bacteroides fragilis protects against CNS demyelinating disease. Mucosal Immunol. 2010, 3, 487–495. [Google Scholar] [CrossRef] [Green Version]
- Tan, H.-E.; Sisti, A.C.; Jin, H.; Vignovich, M.; Villavicencio, M.; Tsang, K.S.; Goffer, Y.; Zuker, C.S. The gut–brain axis mediates sugar preference. Nature 2020, 580, 511–516. [Google Scholar] [CrossRef]
- Lv, W.; Wu, X.; Chen, W.; Li, Y.; Zhang, G.; Chao, L.; Zhou, J.; Guo, A.; Liu, C.; Guo, S. The Gut Microbiome Modulates the Changes in Liver Metabolism and in Inflammatory Processes in the Brain of Chronic Unpredictable Mild Stress Rats. Oxid. Med. Cell. Longev. 2019, 2019, 7902874. [Google Scholar] [CrossRef] [Green Version]
- Koudoufio, M.; Desjardins, Y.; Feldman, F.; Spahis, S.; Delvin, E.; Levy, E. Insight into Polyphenol and Gut Microbiota Crosstalk: Are Their Metabolites the Key to Understand Protective Effects against Metabolic Disorders? Antioxidants 2020, 9, 982. [Google Scholar] [CrossRef]
- Filosa, S.; Di Meo, F.; Crispi, S. Polyphenols-gut microbiota interplay and brain neuromodulation. Neural Regen. Res. 2018, 13, 2055–2059. [Google Scholar] [CrossRef]
- Lu, J.F.; Zhu, M.Q.; Zhang, H.; Liu, H.; Xia, B.; Wang, Y.L.; Shi, X.; Peng, L.; Wu, J.W. Neohesperidin attenuates obesity by altering the composition of the gut microbiota in high-fat diet-fed mice. FASEB J. 2020, 34, 12053–12071. [Google Scholar] [CrossRef]
- Guirro, M.; Gual-Grau, A.; Gibert-Ramos, A.; Alcaide-Hidalgo, J.M.; Canela, N.; Arola, L.; Mayneris-Perxachs, J. Metabolomics Elucidates Dose-Dependent Molecular Beneficial Effects of Hesperidin Supplementation in Rats Fed an Obesogenic Diet. Antioxidants 2020, 9, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Estruel-Amades, S.; Massot-Cladera, M.; Pérez-Cano, F.J.; Franch, À.; Castell, M.; Camps-Bossacoma, M. Hesperidin Effects on Gut Microbiota and Gut-Associated Lymphoid Tissue in Healthy Rats. Nutrients 2019, 11, 324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, Y.; Sun, J.; Xia, S.; Tang, X.; Shi, Y.; Le, G. Effects of resveratrol on gut microbiota and fat storage in a mouse model with high-fat-induced obesity. Food Funct. 2014, 5, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Juan, M.E.; Alfaras, I.; Planas, J.M. Determination of Dihydroresveratrol in Rat Plasma by HPLC. J. Agric. Food Chem. 2010, 58, 7472–7475. [Google Scholar] [CrossRef]
- Brandt, N.; Kotowska, D.; Kristensen, C.M.; Olesen, J.; Lützhøft, D.O.; Halling, J.F.; Hansen, M.; Al-Soud, W.A.; Hansen, L.; Kiilerich, P.; et al. The impact of exercise training and resveratrol supplementation on gut microbiota composition in high-fat diet fed mice. Physiol. Rep. 2018, 6, e13881. [Google Scholar] [CrossRef] [Green Version]
- Basholli-Salihu, M.; Schuster, R.; Mulla, D.; Praznik, W.; Viernstein, H.; Mueller, M. Bioconversion of piceid to resveratrol by selected probiotic cell extracts. Bioprocess Biosyst. Eng. 2016, 39, 1879–1885. [Google Scholar] [CrossRef] [Green Version]
- Youdim, K.A.; Dobbie, M.S.; Kuhnle, G.; Proteggente, A.R.; Abbott, N.J.; Rice-Evans, C. Interaction between flavonoids and the blood–brain barrier: In vitro studies. J. Neurochem. 2003, 85, 180–192. [Google Scholar] [CrossRef]
- Abbott, N.J.; Friedman, A. Overview and introduction: The blood–brain barrier in health and disease. Epilepsia 2012, 53, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte–endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Ziberna, L.; Fornasaro, S.; Čvorović, J.; Tramer, F.; Passamonti, S. Chapter 37—Bioavailability of Flavonoids: The Role of Cell Membrane Transporters. In Polyphenols in Human Health and Disease; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Academic Press: San Diego, CA, USA, 2014; pp. 489–511. ISBN 978-0-12-398456-2. [Google Scholar]
- Youdim, K.A.; Qaiser, M.Z.; Begley, D.J.; Rice-Evans, C.A.; Abbott, N.J. Flavonoid permeability across an in situ model of the blood–brain barrier. Free Radic. Biol. Med. 2004, 36, 592–604. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, S.; Halliwell, B. Do polyphenols enter the brain and does it matter? Some theoretical and practical considerations. Genes Nutr. 2012, 7, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaidyanathan, J.B.; Walle, T. Transport and Metabolism of the Tea Flavonoid (–)-Epicatechin by the Human Intestinal Cell Line Caco-2. Pharm. Res. 2001, 18, 1420–1425. [Google Scholar] [CrossRef]
- Shukla, S.; Zaher, H.; Hartz, A.; Bauer, B.; Ware, J.A.; Ambudkar, S. V Curcumin Inhibits the Activity of ABCG2/BCRP1, a Multidrug Resistance-Linked ABC Drug Transporter in Mice. Pharm. Res. 2009, 26, 480–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, K.W.; Li, Y.; Paxton, J.W.; Birch, N.P.; Scheepens, A. Identification of novel dietary phytochemicals inhibiting the efflux transporter breast cancer resistance protein (BCRP/ABCG2). Food Chem. 2013, 138, 2267–2274. [Google Scholar] [CrossRef]
- Alvarez, A.I.; Real, R.; Pérez, M.; Mendoza, G.; Prieto, J.G.; Merino, G. Modulation of the activity of ABC transporters (P-glycoprotein, MRP2, BCRP) by flavonoids and drug response. J. Pharm. Sci. 2010, 99, 598–617. [Google Scholar] [CrossRef]
- Vingtdeux, V.; Giliberto, L.; Zhao, H.; Chandakkar, P.; Wu, Q.; Simon, J.E.; Janle, E.M.; Lobo, J.; Ferruzzi, M.G.; Davies, P.; et al. AMP-activated Protein Kinase Signaling Activation by Resveratrol Modulates Amyloid-β Peptide Metabolism. J. Biol. Chem. 2010, 285, 9100–9113. [Google Scholar] [CrossRef] [Green Version]
- Suominen, T.; Piepponen, T.P.; Kostiainen, R. Permeation of Dopamine Sulfate through the Blood-Brain Barrier. PLoS ONE 2015, 10, e0133904. [Google Scholar]
- Janle, E.M.; Lila, M.A.; Grannan, M.; Wood, L.; Higgins, A.; Yousef, G.G.; Rogers, R.B.; Kim, H.; Jackson, G.S.; Ho, L.; et al. Pharmacokinetics and Tissue Distribution of 14C-Labeled Grape Polyphenols in the Periphery and the Central Nervous System Following Oral Administration. J. Med. Food 2010, 13, 926–933. [Google Scholar] [CrossRef] [Green Version]
- Suganuma, M.; Okabe, S.; Oniyama, M.; Tada, Y.; Ito, H.; Fujiki, H. Wide distribution of [3H](-)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis 1998, 19, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Abd El Mohsen, M.M.; Kuhnle, G.; Rechner, A.R.; Schroeter, H.; Rose, S.; Jenner, P.; Rice-Evans, C.A. Uptake and metabolism of epicatechin and its access to the brain after oral ingestion. Free Radic. Biol. Med. 2002, 33, 1693–1702. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.W.; Cheng, F.C.; Huang, Y.T.; Chen, C.F.; Tsai, T.H. Determination of naringenin and its glucuronide conjugate in rat plasma and brain tissue by high-performance liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1998, 714, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S.S.R. Influence of Piperine on the Pharmacokinetics of Curcumin in Animals and Human Volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef]
- Lao, C.D.; Ruffin, M.T.; Normolle, D.; Heath, D.D.; Murray, S.I.; Bailey, J.M.; Boggs, M.E.; Crowell, J.; Rock, C.L.; Brenner, D.E. Dose escalation of a curcuminoid formulation. BMC Complement. Altern. Med. 2006, 6, 10. [Google Scholar] [CrossRef] [Green Version]
- Szwajgier, D.; Borowiec, K.; Pustelniak, K. The Neuroprotective Effects of Phenolic Acids: Molecular Mechanism of Action. Nutrients 2017, 9, 477. [Google Scholar] [CrossRef] [Green Version]
- Zini, A.; Rio, D.D.; Stewart, A.J.; Mandrioli, J.; Merelli, E.; Sola, P.; Nichelli, P.; Serafini, M.; Brighenti, F.; Edwards, C.A.; et al. Do flavan-3-ols from green tea reach the human brain? Nutr. Neurosci. 2006, 9, 57–61. [Google Scholar] [CrossRef]
- Turner, R.S.; Thomas, R.G.; Craft, S.; van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S.; et al. A randomized, double-blind, placebo-controlled trial of resveratrol for Alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef]
- Ringman, J.M.; Frautschy, S.A.; Teng, E.; Begum, A.N.; Bardens, J.; Beigi, M.; Gylys, K.H.; Badmaev, V.; Heath, D.D.; Apostolova, L.G.; et al. Oral curcumin for Alzheimer’s disease: Tolerability and efficacy in a 24-week randomized, double blind, placebo-controlled study. Alzheimer’s Res. Ther. 2012, 4, 43. [Google Scholar] [CrossRef] [Green Version]
- Ishisaka, A.; Ichikawa, S.; Sakakibara, H.; Piskula, M.K.; Nakamura, T.; Kato, Y.; Ito, M.; Miyamoto, K.; Tsuji, A.; Kawai, Y.; et al. Accumulation of orally administered quercetin in brain tissue and its antioxidative effects in rats. Free Radic. Biol. Med. 2011, 51, 1329–1336. [Google Scholar] [CrossRef]
- Cichon, N.; Saluk-Bijak, J.; Gorniak, L.; Przyslo, L.; Bijak, M. Flavonoids as a Natural Enhancer of Neuroplasticity—An Overview of the Mechanism of Neurorestorative Action. Antioxidants 2020, 9, 1035. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Wightman, E.L.; Reay, J.L.; Lietz, G.; Okello, E.J.; Wilde, A.; Haskell, C.F. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: A double-blind, placebo-controlled, crossover investigation. Am. J. Clin. Nutr. 2010, 91, 1590–1597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witte, A.V.; Kerti, L.; Margulies, D.S.; Flöel, A. Effects of Resveratrol on Memory Performance, Hippocampal Functional Connectivity, and Glucose Metabolism in Healthy Older Adults. J. Neurosci. 2014, 34, 7862–7870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thaung Zaw, J.J.; Howe, P.R.C.; Wong, R.H.X. Long-term effects of resveratrol on cognition, cerebrovascular function and cardio-metabolic markers in postmenopausal women: A 24-month randomised, double-blind, placebo-controlled, crossover study. Clin. Nutr. 2021, 40, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, S.J.; Ryan, L.; Clegg, M.E. The Effects of a Functional Food Breakfast on Gluco-Regulation, Cognitive Performance, Mood, and Satiety in Adults. Nutrients 2020, 12, 2974. [Google Scholar] [CrossRef]
- Köbe, T.; Witte, A.V.; Schnelle, A.; Tesky, V.A.; Pantel, J.; Schuchardt, J.-P.; Hahn, A.; Bohlken, J.; Grittner, U.; Flöel, A. Impact of Resveratrol on Glucose Control, Hippocampal Structure and Connectivity, and Memory Performance in Patients with Mild Cognitive Impairment. Front. Neurosci. 2017, 11, 105. [Google Scholar]
- Gu, J.; Li, Z.; Chen, H.; Xu, X.; Li, Y.; Gui, Y. Neuroprotective Effect of Trans-Resveratrol in Mild to Moderate Alzheimer Disease: A Randomized, Double-Blind Trial. Neurol. Ther. 2021, 10, 905–917. [Google Scholar] [CrossRef]
- Ye, S.; Hou, Z.; Zhong, L.; Zhang, Q. Effect of curcumin on the induction of glutathione S-transferases and NADP(H):quinone oxidoreductase and its possible mechanism of action. Yao Xue Xue Bao 2007, 42, 376–380. [Google Scholar]
- Abdolahi, M.; Jafarieh, A.; Sarraf, P.; Sedighiyan, M.; Yousefi, A.; Tafakhori, A.; Abdollahi, H.; Salehinia, F.; Djalali, M. The Neuromodulatory Effects of ω-3 Fatty Acids and Nano-Curcumin on the COX-2/iNOS Network in Migraines: A Clinical Trial Study from Gene Expression to Clinical Symptoms. Endocr. Metab. Immune Disord.-Drug Targets 2019, 19, 874–884. [Google Scholar]
- Djalali, M.; Abdolahi, M.; Hosseini, R.; Miraghajani, M.; Mohammadi, H.; Djalali, M. The effects of nano-curcumin supplementation on Th1/Th17 balance in migraine patients: A randomized controlled clinical trial. Complement. Ther. Clin. Pract. 2020, 41, 101256. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Kopin, I.J.; Goldstein, D.S. Catecholamine Metabolism: A Contemporary View with Implications for Physiology and Medicine. Pharmacol. Rev. 2004, 56, 331–349. [Google Scholar] [CrossRef] [Green Version]
- Peaston, R.T.; Weinkove, C. Measurement of catecholamines and their metabolites. Ann. Clin. Biochem. 2004, 41, 17–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, K.; Koide, Y.; Shohmori, T. Determination of p-hydroxyphenylacetic acid in cerebrospinal fluid by high-performance liquid chromatography with electrochemical detection. Clin. Chim. Acta 1982, 123, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Squillaro, T.; Cimini, A.; Peluso, G.; Giordano, A.; Melone, M.A.B. Nano-delivery systems for encapsulation of dietary polyphenols: An experimental approach for neurodegenerative diseases and brain tumors. Biochem. Pharmacol. 2018, 154, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Augustin, M.A.; Hemar, Y. Nano- and micro-structured assemblies for encapsulation of food ingredients. Chem. Soc. Rev. 2009, 38, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Conte, R. Polyphenols nanoencapsulation for therapeutic applications. Biomol. Res. Ther. 2016, 5, 139. [Google Scholar]
- Caruana, M.; Cauchi, R.; Vassallo, N. Putative Role of Red Wine Polyphenols against Brain Pathology in Alzheimer’s and Parkinson’s Disease. Front. Nutr. 2016, 3, 31. [Google Scholar]
- Sabogal-Guáqueta, A.M.; Muñoz-Manco, J.I.; Ramírez-Pineda, J.R.; Lamprea-Rodriguez, M.; Osorio, E.; Cardona-Gómez, G.P. The flavonoid quercetin ameliorates Alzheimer’s disease pathology and protects cognitive and emotional function in aged triple transgenic Alzheimer’s disease model mice. Neuropharmacology 2015, 93, 134–145. [Google Scholar] [CrossRef] [Green Version]
- Cai, X.; Fang, Z.; Dou, J.; Yu, A.; Zhai, G. Bioavailability of quercetin: Problems and promises. Curr. Med. Chem. 2013, 20, 2572–2582. [Google Scholar] [CrossRef]
- Esfanjani, A.F.; Jafari, S.M. Biopolymer nano-particles and natural nano-carriers for nano-encapsulation of phenolic compounds. Colloids Surf. B Biointerfaces 2016, 146, 532–543. [Google Scholar]
- Aluani, D.; Tzankova, V.; Yordanov, Y.; Kondeva-Burdina, M.; Yoncheva, K. In vitro protective effects of encapsulated quercetin in neuronal models of oxidative stress injury. Biotechnol. Biotechnol. Equip. 2017, 31, 1055–1063. [Google Scholar] [CrossRef] [Green Version]
- Rifaai, R.A.; Mokhemer, S.A.; Saber, E.A.; El-Aleem, S.A.A.; El-Tahawy, N.F.G. Neuroprotective effect of quercetin nanoparticles: A possible prophylactic and therapeutic role in alzheimer’s disease. J. Chem. Neuroanat. 2020, 107, 101795. [Google Scholar] [CrossRef] [PubMed]
- Amanzadeh Jajin, E.; Esmaeili, A.; Rahgozar, S.; Noorbakhshnia, M. Quercetin-Conjugated Superparamagnetic Iron Oxide Nanoparticles Protect AlCl3-Induced Neurotoxicity in a Rat Model of Alzheimer’s Disease via Antioxidant Genes, APP Gene, and miRNA-101. Front. Neurosci. 2021, 14, 598617. [Google Scholar]
- Kaur, I.P.; Bhandari, R.; Bhandari, S.; Kakkar, V. Potential of solid lipid nanoparticles in brain targeting. J. Control. Release 2008, 127, 97–109. [Google Scholar]
- Naqvi, S.; Panghal, A.; Flora, S.J.S. Nanotechnology: A promising approach for delivery of neuroprotective drugs. Front. Neurosci. 2020, 14, 494. [Google Scholar]
- Loureiro, J.A.; Andrade, S.; Duarte, A.; Neves, A.R.; Queiroz, J.F.; Nunes, C.; Sevin, E.; Fenart, L.; Gosselet, F.; Coelho, M.A.N.; et al. Resveratrol and Grape Extract-loaded Solid Lipid Nanoparticles for the Treatment of Alzheimer’s Disease. Molecules 2017, 22, 277. [Google Scholar] [CrossRef] [Green Version]
- Sandhir, R.; Yadav, A.; Mehrotra, A.; Sunkaria, A.; Singh, A.; Sharma, S. Curcumin Nanoparticles Attenuate Neurochemical and Neurobehavioral Deficits in Experimental Model of Huntington’s Disease. NeuroMol. Med. 2014, 16, 106–118. [Google Scholar] [CrossRef]
- Kundu, P.; Das, M.; Tripathy, K.; Sahoo, S.K. Delivery of Dual Drug Loaded Lipid Based Nanoparticles across the Blood–Brain Barrier Impart Enhanced Neuroprotection in a Rotenone Induced Mouse Model of Parkinson’s Disease. ACS Chem. Neurosci. 2016, 7, 1658–1670. [Google Scholar] [CrossRef]
- Abdul Rahim, R.; Jayusman, P.A.; Muhammad, N.; Ahmad, F.; Mokhtar, N.; Naina Mohamed, I.; Mohamed, N.; Shuid, A.N. Recent Advances in Nanoencapsulation Systems Using PLGA of Bioactive Phenolics for Protection against Chronic Diseases. Int. J. Environ. Res. Public Health 2019, 16, 4962. [Google Scholar] [CrossRef] [Green Version]
- Del Amo, L.; Cano, A.; Ettcheto, M.; Souto, E.B.; Espina, M.; Camins, A.; García, M.L.; Sánchez-López, E. Surface functionalization of PLGA nanoparticles to increase transport across the BBB for Alzheimer’s disease. Appl. Sci. 2021, 11, 4305. [Google Scholar]
- Patel, M.M.; Patel, B.M. Crossing the blood–brain barrier: Recent advances in drug delivery to the brain. CNS Drugs 2017, 31, 109–133. [Google Scholar]
- FDA Current Good Manufacturing Practice (CGMP) Regulations. 2023. Available online: https://www.fda.gov/drugs/pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations (accessed on 1 May 2023).
- Zhi, K.; Raji, B.; Nookala, A.R.; Khan, M.M.; Nguyen, X.H.; Sakshi, S.; Pourmotabbed, T.; Yallapu, M.M.; Kochat, H.; Tadrous, E. PLGA nanoparticle-based formulations to cross the blood–brain barrier for drug delivery: From R&D to cGMP. Pharmaceutics 2021, 13, 500. [Google Scholar] [PubMed]
- Mathew, A.; Aravind, A.; Fukuda, T.; Hasumura, T.; Nagaoka, Y.; Yoshida, Y.; Maekawa, T.; Venugopal, K.; Kumar, D.S. Curcumin nanoparticles- a gateway for multifaceted approach to tackle Alzheimer’s disease. In Proceedings of the 2011 11th IEEE International Conference on Nanotechnology, Portland, OR, USA, 15–18 August 2011; pp. 833–836. [Google Scholar]
- Mathew, A.; Fukuda, T.; Nagaoka, Y.; Hasumura, T.; Morimoto, H.; Yoshida, Y.; Maekawa, T.; Venugopal, K.; Kumar, D.S. Curcumin Loaded-PLGA Nanoparticles Conjugated with Tet-1 Peptide for Potential Use in Alzheimer’s Disease. PLoS ONE 2012, 7, e32616. [Google Scholar]
- Tiwari, S.K.; Agarwal, S.; Seth, B.; Yadav, A.; Nair, S.; Bhatnagar, P.; Karmakar, M.; Kumari, M.; Chauhan, L.K.S.; Patel, D.K.; et al. Curcumin-Loaded Nanoparticles Potently Induce Adult Neurogenesis and Reverse Cognitive Deficits in Alzheimer’s Disease Model via Canonical Wnt/β-Catenin Pathway. ACS Nano 2014, 8, 76–103. [Google Scholar] [CrossRef]
- Ray, B.; Bisht, S.; Maitra, A.; Maitra, A.; Lahiri, D.K. Neuroprotective and Neurorescue Effects of a Novel Polymeric Nanoparticle Formulation of Curcumin (NanoCurcTM) in the Neuronal Cell Culture and Animal Model: Implications for Alzheimer’s disease. J. Alzheimer’s Dis. 2011, 23, 61–77. [Google Scholar] [CrossRef]
- Ghosh, A.; Sarkar, S.; Mandal, A.K.; Das, N. Neuroprotective role of nanoencapsulated quercetin in combating ischemia-reperfusion induced neuronal damage in young and aged rats. PLoS ONE 2013, 8, e57735. [Google Scholar]
- Sun, D.; Li, N.; Zhang, W.; Zhao, Z.; Mou, Z.; Huang, D.; Liu, J.; Wang, W. Design of PLGA-functionalized quercetin nanoparticles for potential use in Alzheimer’s disease. Colloids Surf. B Biointerfaces 2016, 148, 116–129. [Google Scholar]
- Yang, B.; Dong, Y.; Wang, F.; Zhang, Y. Nanoformulations to enhance the bioavailability and physiological functions of polyphenols. Molecules 2020, 25, 4613. [Google Scholar] [PubMed]
- Priprem, A.; Watanatorn, J.; Sutthiparinyanont, S.; Phachonpai, W.; Muchimapura, S. Anxiety and cognitive effects of quercetin liposomes in rats. Nanomed. Nanotechnol. Biol. Med. 2008, 4, 70–78. [Google Scholar] [CrossRef]
- Wang, Y.; Xia, Z.; Xu, J.-R.; Wang, Y.-X.; Hou, L.-N.; Qiu, Y.; Chen, H.-Z. α-Mangostin, a polyphenolic xanthone derivative from mangosteen, attenuates β-amyloid oligomers-induced neurotoxicity by inhibiting amyloid aggregation. Neuropharmacology 2012, 62, 871–881. [Google Scholar]
- Chen, Z.-L.; Huang, M.; Wang, X.-R.; Fu, J.; Han, M.; Shen, Y.-Q.; Xia, Z.; Gao, J.-Q. Transferrin-modified liposome promotes α-mangostin to penetrate the blood–brain barrier. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Mennen, L.I.; Walker, R.; Bennetau-Pelissero, C.; Scalbert, A. Risks and safety of polyphenol consumption. Am. J. Clin. Nutr. 2005, 81, 326S–329S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, K.R.; Appel, C.L. Polyphenols as dietary supplements: A double-edged sword. Nutr. Diet. Suppl. 2009, 2, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Weisburg, J.H.; Weissman, D.B.; Sedaghat, T.; Babich, H. In vitro cytotoxicity of epigallocatechin gallate and tea extracts to cancerous and normal cells from the human oral cavity. Basic Clin. Pharmacol. Toxicol. 2004, 95, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Are polyphenols antioxidants or pro-oxidants? What do we learn from cell culture and in vivo studies? Arch. Biochem. Biophys. 2008, 476, 107–112. [Google Scholar] [CrossRef]
- Lambert, J.D.; Kwon, S.-J.; Hong, J.; Yang, C.S. Salivary hydrogen peroxide produced by holding or chewing green tea in the oral cavity. Free Radic. Res. 2007, 41, 850–853. [Google Scholar] [CrossRef] [PubMed]
- Bhalli, J.A.; Neft, R.; Noteboom, J.; Tebbe, C.C.; Chan, M.; Kuhn, K.; Pearce, G.; Jordan, L.; Beevers, C. Caffeic acid genotoxicity: Correlation of the pig-a assay with regulatory genetic toxicology in vivo endpoints. Environ. Mol. Mutagen. 2019, 60, 837–844. [Google Scholar] [CrossRef]
- Sugimoto, K.; Araki, N.; Ohmori, M.; Harada, K.; Cui, Y.; Tsuruoka, S.; Kawaguchi, A.; Fujimura, A. Interaction between grapefruit juice and hypnotic drugs: Comparison of triazolam and quazepam. Eur. J. Clin. Pharmacol. 2006, 62, 209–215. [Google Scholar] [CrossRef]
- Temme, E.H.M.; Van Hoydonck, P.G.A. Tea consumption and iron status. Eur. J. Clin. Nutr. 2002, 56, 379–386. [Google Scholar] [CrossRef] [Green Version]
- Gabrielli, G.B.; De Sandre, G. Excessive tea consumption can inhibit the efficacy of oral iron treatment in iron-deficiency anemia. Haematologica 1995, 80, 518–520. [Google Scholar]
- Nelson, M.; Poulter, J. Impact of tea drinking on iron status in the UK: A review. J. Hum. Nutr. Diet. 2004, 17, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, A.B.; Klein, S.L.; Lakshmanan, Y.; Gearhart, J.P. Exposure to genistein during gestation and lactation demasculinizes the reproductive system in rats. J. Urol. 2003, 169, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Bingham, S.; Setchell, K.D. Biological effects of a diet of soy protein rich in isoflavones on the menstrual cycle of premenopausal women. Am. J. Clin. Nutr. 1994, 60, 333–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gentile, M.T.; Camerino, I.; Ciarmiello, L.; Woodrow, P.; Muscariello, L.; De Chiara, I.; Pacifico, S. Neuro-Nutraceutical Polyphenols: How Far Are We? Antioxidants 2023, 12, 539. [Google Scholar] [CrossRef]
- Marín Fernández, L.; Miguélez González, E.M.; Villar Granja, C.J.; Lombó Brugos, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. BioMed Res. Int. 2015, 2015, 905215. [Google Scholar]
- Polia, F.; Pastor-Belda, M.; Martínez-Blázquez, A.; Horcajada, M.-N.; Tomás-Barberán, F.A.; García-Villalba, R. Technological and biotechnological processes to enhance the bioavailability of dietary (poly) phenols in humans. J. Agric. Food Chem. 2022, 70, 2092–2107. [Google Scholar] [CrossRef]
- Carecho, R.; Carregosa, D.; dos Santos, C.N. Low Molecular Weight (poly)Phenol Metabolites Across the Blood-Brain Barrier: The Underexplored Journey. Brain Plast. 2020, 6, 193–214. [Google Scholar] [CrossRef]

| Objectives | Study Description | Main Results | References |
|---|---|---|---|
| To assess the impact of blueberry flavonoids on long-term memory function. | Male Lister-hooded rats were divided into three groups—young group approximately 6 months old, aged group approximately 18 months old and aged group approximately 18 months old blueberry-supplemented. Supplementation with the blueberry diet (2% w/w) was conducted for 12 weeks. Rats were tested in a cross-maze apparatus. Hippocampal and cortical regions were analyzed. | Blueberry supplementation results in improvement in spatial working memory tasks in aged animals. This correlated with the activation of CREB and increased BDNF levels in hippocampus. Additionally, increased phosphorylation level of ERK1/2 was observed. | [33] |
| To assess changes in hippocampal plasticity parameters (hippocampal neurogenesis, extracellular receptor kinase activation, IGF-1 and IGF-1R levels) after blueberry supplementation. | 19-months old male F344 rats were divided into two groups—control diet and blueberry extract diet. The dose of blueberry extract was 20 g/kg. Rats were fed with those diets for 8 weeks. Animals were tested in radial arm water maze (RAWM) test. | All tested parameters of hippocampal neuronal plasticity were elevated after blueberry extract supplementation. Observed changes in those parameters correlated with improvements in spatial memory. | [29] |
| To assess neuroprotective effect of quercetin in rotenone-induced hemiparkinsonian rats. | Adult Sprague-Dawley rats were infused with rotenone into the substantia nigra. Quercetin (in doses 25, 50 or 75 mg/kg) was administered at 12 h intervals for 4 days. Mitochondrial dysfunction, oxidative stress, programmed cell death and dopamine neuronal demise were assessed. | Quercetin up-regulates electron transport chain (ETC) complex-I activity in damaged or normal dopaminergic neurons. It also effectively scavenge OH generated in the mitochondria, and completely reverse striatal dopamine loss and nigral glutathione depletion. Quercetin has also blocked programmed cell death in nigral neurons. The effect of quercetin was dose-dependent. | [30] |
| To assess the effects of green tea polyphenols (GTPs) on the permeability of blood-brain barrier (BBB) and expression of caveolin-1 and ERK1/2 after cerebral ischemia. | Cerebral ischemia was induced in rats by middle cerebral artery occlusion (MCAO). Animals were divided into control and GTP group, in each four time points were included: MCA occluded for 0 h, 1 h, 2 h and 4 h. The infarct volume and neurological deficits were assessed. The BBB permeability, caveolin-1 and ERK1/2 levels were determined. | GTPs significantly reduced infarct volume, ameliorated the neurological deficit, and reduced the permeability of BBB. Both caveolin-1 and phosphorylated ERK1/2 expression levels were reduced by GTPs. | [39] |
| To investigate the effect of Neumentix (containing, among others, rosmarinic acid) on inflammation and BBB disruption in transient MCAO (tMCAO). | Mice were treated with vehicle or Neumentix (134 mg/kg/d) for 14 days. Then animals were subjected to tMCAO for 1 h. After this procedure mice kept receiving vehicle or Neumentix for next 5 days. | Neumentix reduced infarct volume, inhibited expression of Iba-1, TNF-α, MCP-1 and improve the integrity of BBB. Moreover, it ameliorated neurobehavioral impairment observed in the corner test. | [40] |
| To assess protective effect of lychee seed polyphenols (LSP) on BBB integrity. | bEnd.3 cell cultures and APP/PS1 mice were utilized. Expression of tight junction proteins (TJs), activation of Aβ(25–35)-induced NLRP3 inflammasome, permeability of bEnd.3 monolayer and cognitive function of mice were examined. | LSP protects BBB integrity through inhibiting Aβ(25–35)-induced NLRP3 inflammasome activation via the AMPK/mTOR/ULK1-mediated autophagy. LSP significantly reduced the permeability of bEnd.3 monolayer. It also improved the spatial learning and memory function in tested mice and increased the expression of TJs. | [41] |
| To investigate the effect of resveratrol on stress-related depression, anxiety, intestinal and visceral dysfunction in rat model of irritable bowel syndrome (IBS). | Rats received chronic acute combining stress (CACS) for 22 days. Resveratrol was administered at doses of 10, 20 and 40 mg/kg 50 min before CACS procedure. Changes in behavior, visceral sensitivity and intestinal motility were measured by the forced swimming, marble bury, abdominal withdrawal reflex (AWR) and intestinal tract motility (ITM) tests. | Resveratrol dose-dependently normalized CACS-induced dysfunctions. This polyphenol alleviate IBS-like effects on depression, anxiety, visceral hypersensitivity and intestinal motility abnormality through regulation of 5-HT1A-dependent PKA-CREB-BDNF signaling in the brain-gut axis. | [42] |
| To investigate the therapeutic potential of resveratrol on sevoflurane-induced cognitive impairment. | 6 day-old mice received anesthesia with 3% sevoflurane 2 h daily on postnatal days (P) 6, P7 and P8. Resveratrol (100 mg/kg) was administered for 6 consecutive days to neonatal mice before anesthesia. Animals were tested on mouse Morris water maze (MWM) test. | Resveratrol reversed the effect of repeated sevoflurane exposure and modulated SIRT1-NF-κB pathway in microglia leading to amelioration of cognitive impairment in neonatal mice. | [38] |
| To assess the potential of resveratrol to ameliorate ischemia-induced neuronal cell death. | Mongolian gerbils were divided into three groups: sham control, ischemia and ischemia treated with resveratrol. Transient global cerebral ischemia was induced for 5 min. Resveratrol was injected (30 mg/kg), either during or shortly after occlusion, and again 24 h after ischemia. | Resveratrol significantly decreased delayed neuronal cell death (DND) and glial cell activation. The analysis of resveratrol’s bioavailability indicated that this compound can cross the blood-brain barrier. | [37] |
| To investigate neuroprotective potential of caffeic acid (CA). | Mice were subjected to a permanent middle cerebral artery occlusion. Mice were pretreated and post-treated with CA (2, 20, 60 mg/kg) at 24, 48, 72, 96 or 120 h after ischemia. Animals were evaluated for brain infarction, neurological deficit score, locomotor activity, working memory, short-term aversive memory, long-term aversive memory and spatial memory. | CA reduced the infarcted area and improved neurological deficit scores. CA also alleviate working, spatial and long-term aversive memory deficits. It also inhibited the ischemia-induced reduction in the synaptophysin level. | [43] |
| Study Objective | Study Group | Study Description | Main Results | Reference |
|---|---|---|---|---|
| Determination of the effect of a blueberry smoothie containing 253 g of anthocyanins on particular aspects of executive functions | Randomized, single-blind, parallel-group study, 54 healthy schoolchildren (aged 7–10) | Study group received a 200 mL WBB drink (253 mg anthocyanins) or a matched placebo. Verbal memory (AVLT), executive function (MANT), and reading efficiency (TOWRE-2) were evaluated before and after 2 h of consumption | Consumption of blueberries significantly improved the memory and attentional aspects without affecting reading efficiency | [62] |
| Determination of the acute effect of a smoothie of berries on the improvement of executive functions and mood | Single-blind, randomized, placebo-controlled, between-subjects study, 40 healthy subjects (aged 20–30) | The study group received a smoothie of berries (blueberries, raspberries, and strawberries) containing 14.3 g of polyphenols or placebo. Executive function (MANT, switching tasks) and mood (positive and negative affect schedule) were assessed at baseline and 2, 4, and 6 h after consumption | Smoothie consumption maintained cognitive performance during the working day compared to placebo, while not influencing the mood | [63] |
| Determination of the influence of polyphenol-rich grape and blueberry extract (PEGB) on memory | A bicentric double-blind, randomized, placebo-controlled clinical study, 215 healthy elderly people (aged 60–70 years, BMI 20–30, 26 < MMSE score ≤ 29) | The study group received 600 mg/day of PEGB or placebo for 6 months. The Cambridge neuropsychological test automated battery (CANTAB) was assessed at baseline and after 24 weeks of supplementation | Supplementation with PEGB had no effect on memory performance in the whole cohort. However, PEGB supplementation significantly enhanced cognitive performance in elderly people with significant cognitive impairment | [17] |
| Dietary flavonoids have been studied for cognitive performance | Cohort study: 2044 adult men and women from southern Italy | The demographics and dietary habits of 808 adults living in southern Italy were analyzed. Food frequency questionnaires (FFQs) were used to assess dietary intake. Data on the polyphenol content of foods were estimated using the Phenol-Explorer database. The Short Portable Mental Status Questionnaire was used as a screening tool for cognitive status | Diet rich in flavonoids negatively correlates with cognitive dysfunction in the adult population. It has been shown that anthocyanins, flavan-3-ols, flavonols, and catechins significantly improve cognitive performance | [64] |
| Sample | Study Group | Analyzed Polyphenols | Polyphenols Detected in CNS | References |
|---|---|---|---|---|
| CSF *, plasma * | Twenty-eight patients (age 46 ± 16 years; 18 women and 10 men) with neurological diseases (multiple sclerosis, polyneuropathy, mononeuropathy, meningitis, brain tumor, epilepsy, amyotrophic lateral sclerosis, an Friedreich’s ataxia) on a “western” diet |
|
| [68] |
| CNS * | Six patients (aged 34–61, 3 women and 3 men) with suspicion of multiple sclerosis acute ingestion of green tea |
| undetectable in the CSF | [120] |
| Brain MRI * CSF * | One hundred and nineteen patients with mild-to-moderate Alzheimer’s disease 52-week supplementation with resveratrol (500 mg/day, with a dose increase of 500 mg every 13 weeks, final dose 2000 mg/day) |
|
| [121] |
| CSF * | Thirty-six patients with mild-to-moderate Alzheimer’s disease oral curcumin supplementation at a dose of 2 g/day, 4 g/day, or placebo for 24 weeks, then for the next 24 weeks |
| undetectable in the CSF | [122] |
| human brain tissue # |
|
| [123] |
| Mechanism | Predisposing Factors | Example | Reference |
|---|---|---|---|
| Pro-oxidant activity | High polyphenol concentration, presence of iron and H2O2, and a high pH, auto-oxidation-induced generation of ROS | Epigallocatechin gallate | [169,170,171] |
| Genotoxicity | High concentration and long-term consumption of high doses | Caffeic acid | [172] |
| Interaction with pharmaceuticals | Changes in bioavailability and pharmacokinetics of benzodiazepines by inhibition of cytochrome P450 3A4 (CYP3A4) | Naringenin (grapefruit juice) | [173] |
| Antinutritional effect: inhibition of iron absorption | Long-term consumption of large amounts of tea and coffee | Tannins | [174,175,176] |
| Interaction with the endocrine system | Large consumption of isoflavones with estrogenic activity | Genistein, daidzein, and glycitein | [177,178] |
| Factor | Main Mechanism |
|---|---|
| Class of polyphenols | The bioavailability is determined by the class of polyphenols and ranks as follows: phenolic acids > isoflavones > flavonols > catechins > flavanones > proanthocyanidins > anthocyanins. |
| Differences in chemical structure within the polyphenol class | Changes in physicochemical properties: solubility, ability to polymerize, and ability to enter reactions of conjugation. |
| Small intestine | Hydrolysis of most glycosides—aglycone absorbed by enterocytes, conjugation reactions, methylation, glucuronidation, and sulfation. |
| Liver | Aglycone-conjugation reactions: methylation, glucuronidation, and sulfation; part of the products goes back to the intestine with bile; part is excreted with urine; the majority enter other organs and tissues through the bloodstream. |
| Colon | Metabolism by bacterial enzymes to fewer complex compounds, absorption by colonocytes, and part of the ingested polyphenol dose are excreted with feces. |
| Food matrix | Can protect polyphenols from degradation. The food matrix changes caused by diet modification can affect gut microbiota and improve or decrease bioavailability depending on their composition. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grabska-Kobyłecka, I.; Szpakowski, P.; Król, A.; Książek-Winiarek, D.; Kobyłecki, A.; Głąbiński, A.; Nowak, D. Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development. Nutrients 2023, 15, 3454. https://doi.org/10.3390/nu15153454
Grabska-Kobyłecka I, Szpakowski P, Król A, Książek-Winiarek D, Kobyłecki A, Głąbiński A, Nowak D. Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development. Nutrients. 2023; 15(15):3454. https://doi.org/10.3390/nu15153454
Chicago/Turabian StyleGrabska-Kobyłecka, Izabela, Piotr Szpakowski, Aleksandra Król, Dominika Książek-Winiarek, Andrzej Kobyłecki, Andrzej Głąbiński, and Dariusz Nowak. 2023. "Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development" Nutrients 15, no. 15: 3454. https://doi.org/10.3390/nu15153454
APA StyleGrabska-Kobyłecka, I., Szpakowski, P., Król, A., Książek-Winiarek, D., Kobyłecki, A., Głąbiński, A., & Nowak, D. (2023). Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development. Nutrients, 15(15), 3454. https://doi.org/10.3390/nu15153454






