Effects of Long-Chain Polyunsaturated Fatty Acids in Combination with Lutein and Zeaxanthin on Episodic Memory in Healthy Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design of Trial 1
2.2. Study Design of Trial 2
2.3. Participants in Trials 1 and 2
2.4. Experimental Supplements
2.5. Outcome Assessments
2.6. Memory Function Assessments
- Verbal memory: First, the participants learned 15 words. Immediately after that, from the 30 words (15 learned and 15 unlearned words) that were randomly presented, participants recognized the words that were previously learned. After 30 min or more, again, from the 30 words (15 learned and 15 unlearned words) that were randomly presented, participants recognized the words learned previously. The number of correct responses to 60 trials, including immediate and delayed recognition, was calculated as a verbal memory score.
- Visual memory: First, participants learned 15 meaningless visual images. Immediately after that, from the 30 images (15 learned and 15 unlearned images) that were randomly presented, participants recognized the images that were previously learned. After 30 min or more, again, from the 30 images (15 learned and 15 unlearned images) that were randomly presented, participants recognized previously learned images. The number of correct responses to 60 trials, including immediate and delayed recognition, was calculated as a visual memory score.
2.7. Dietary Assessment and Study Diary
2.8. Fatty Acid Analysis
2.9. Lutein and Zeaxanthin Analysis
2.10. Sample Size
2.11. Randomization, Allocation, and Blinding
2.12. Statistical Analysis
3. Results
3.1. Trial 1
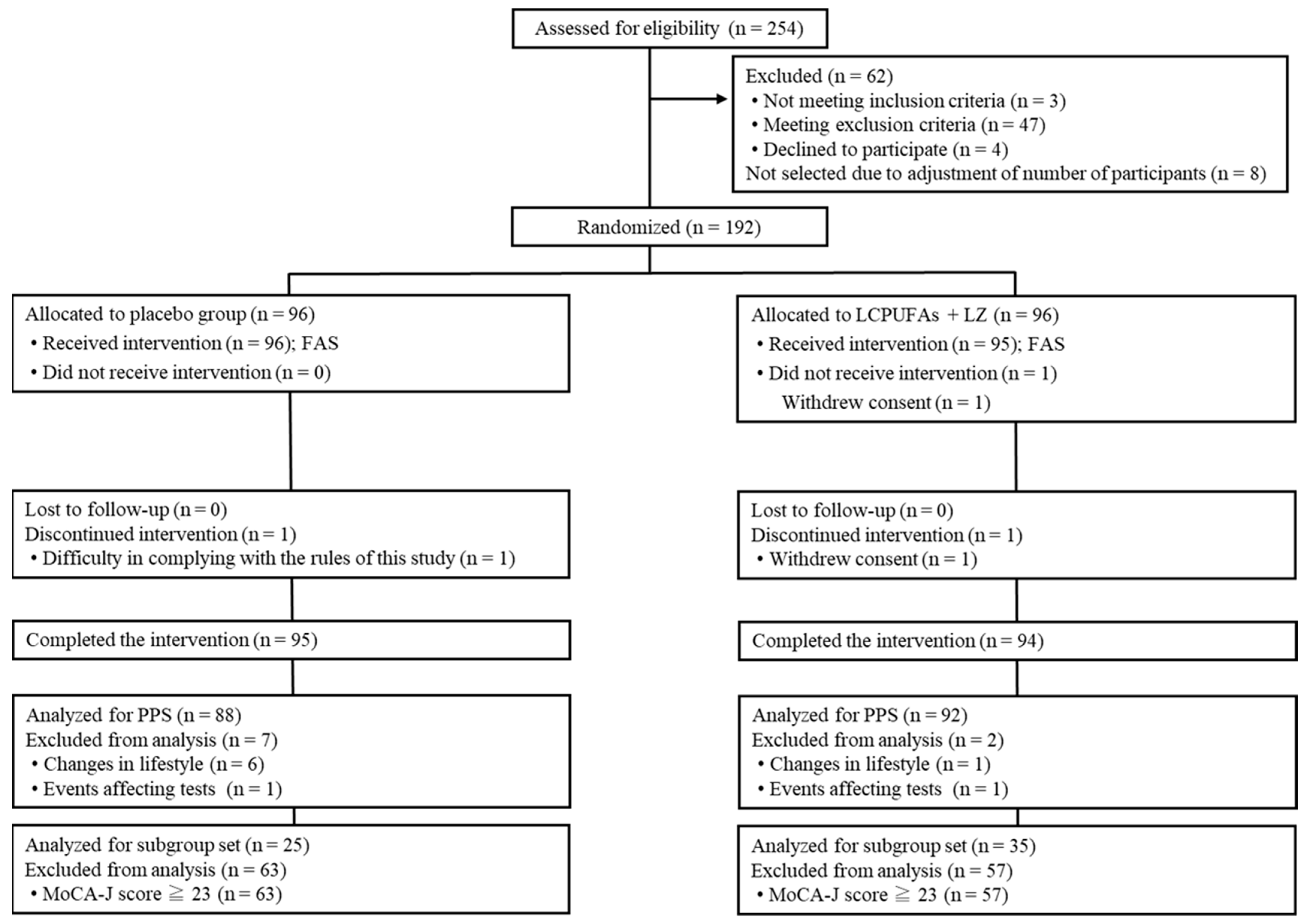
3.1.1. Flow of the Participants and Baseline Characteristics in Trial 1
3.1.2. PPS Analysis in Trial 1
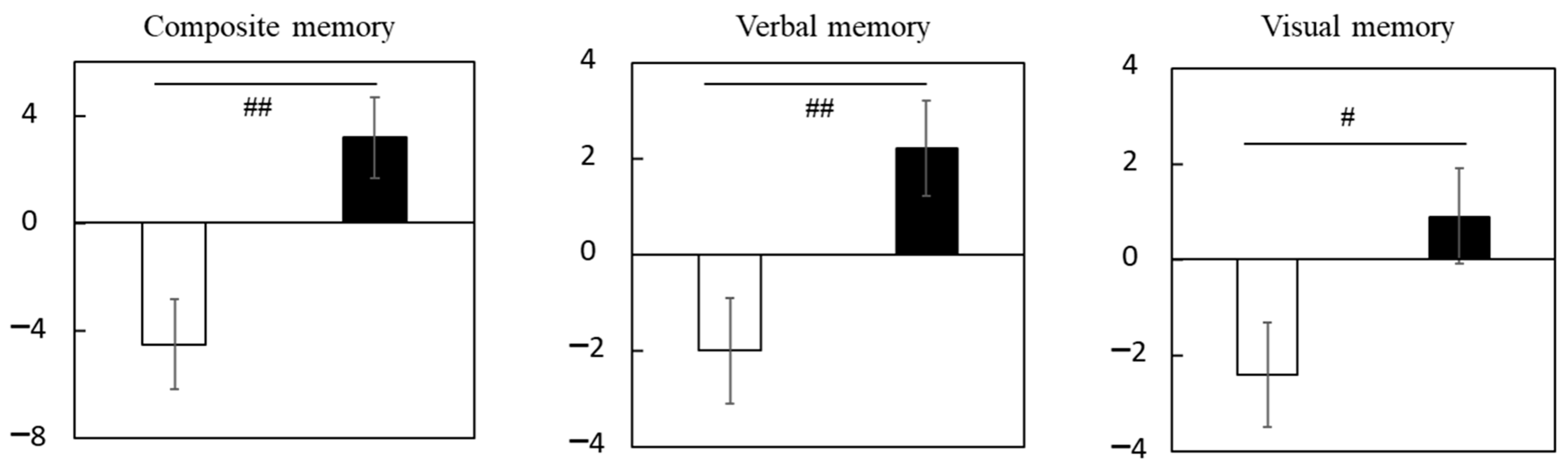
3.1.3. Subgroup Analysis of Participants with Cognitive Decline in Trial 1
3.1.4. Safety in Trial 1
3.2. Trial 2
3.2.1. Flow of the Participants and Baseline Characteristics in Trial 2
3.2.2. PPS Analysis in Trial 2
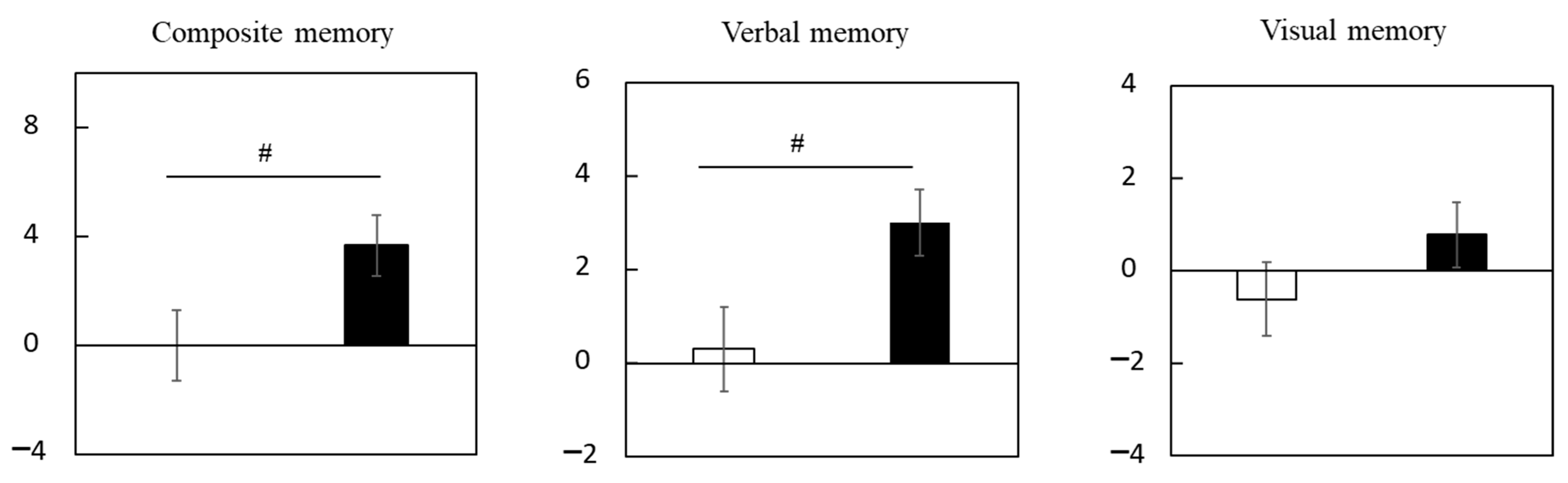
3.2.3. Subgroup Analysis of Participants with Cognitive Decline in Trial 2
3.2.4. Safety in Trial 2
3.3. Combined Analysis of the Two Trials
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Otsuka, R.; Nishita, Y.; Tange, C.; Tomida, M.; Kato, Y.; Nakamoto, M.; Imai, T.; Ando, F.; Shimokata, H. Dietary diversity decreases the risk of cognitive decline among Japanese older adults. Geriatr. Gerontol. Int. 2017, 17, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Soderberg, M.; Edlund, C.; Kristensson, K.; Dallner, G. Fatty acid composition of brain phospholipids in aging and in Alzheimer’s disease. Lipids 1991, 26, 421–425. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Liu, Y.; Jandacek, R.; Rider, T.; Tso, P. The aging human orbitofrontal cortex: Decreasing polyunsaturated fatty acid composition and associated increases in lipogenic gene expression and stearoyl-CoA desaturase activity. Prostagland. Leukot. Essent. Fat. Acids 2008, 78, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, H.; Kontani, M.; Kawashima, H.; Kiso, Y.; Shibata, H.; Osumi, N. Differential effect of arachidonic acid and docosahexaenoic acid on age-related decreases in hippocampal neurogenesis. Neurosci. Res. 2014, 88, 58–66. [Google Scholar] [CrossRef]
- Yurko-Mauro, K.; McCarthy, D.; Rom, D.; Nelson, E.B.; Ryan, A.S.; Blackwell, A.; Salem, N., Jr.; Stedman, M.; Midas Investigators. Beneficial effects of docosahexaenoic acid on cognition in age-related cognitive decline. Alzheimers Dement. 2010, 6, 456–464. [Google Scholar] [CrossRef]
- Lee, L.K.; Shahar, S.; Chin, A.V.; Yusoff, N.A. Docosahexaenoic acid-concentrated fish oil supplementation in subjects with mild cognitive impairment (MCI): A 12-month randomised, double-blind, placebo-controlled trial. Psychopharmacology 2013, 225, 605–612. [Google Scholar] [CrossRef]
- Yabuzaki, J. Carotenoids Database: Structures, chemical fingerprints and distribution among organisms. Database 2017, 2017, bax004. [Google Scholar] [CrossRef]
- Fernández-García, E.; Carvajal-Lérida, I.; Jarén-Galán, M.; Garrido-Fernández, J.; Pérez-Gálvez, A.; Hornero-Méndez, D. Carotenoids bioavailability from foods: From plant pigments to efficient biological activities. Food Res. Int. 2012, 46, 438–450. [Google Scholar] [CrossRef]
- Granado-Lorencio, F.; Blanco-Navarro, I.; Perez-Sacristan, B.; Hernandez-Alvarez, E. Biomarkers of carotenoid bioavailability. Food Res. Int. 2017, 99, 902–916. [Google Scholar] [CrossRef]
- Olmedilla, B.; Granado, F.; Southon, S.; Wright, A.J.; Blanco, I.; Gil-Martinez, E.; Berg, H.; Corridan, B.; Roussel, A.M.; Chopra, M.; et al. Serum concentrations of carotenoids and vitamins A, E, and C in control subjects from five European countries. Br. J. Nutr. 2001, 85, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Johra, F.T.; Bepari, A.K.; Bristy, A.T.; Reza, H.M. A Mechanistic Review of beta-Carotene, Lutein, and Zeaxanthin in Eye Health and Disease. Antioxidants 2020, 9, 1046. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Y.; Fu, Z.J.; Ma, H.; Jang, W.C.; So, K.F.; Wong, D.; Lo, A.C. Effect of lutein on retinal neurons and oxidative stress in a model of acute retinal ischemia/reperfusion. Investig. Ophthalmol. Vis. Sci. 2009, 50, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Y.; Fung, F.K.; Fu, Z.J.; Wong, D.; Chan, H.H.; Lo, A.C. Anti-inflammatory effects of lutein in retinal ischemic/hypoxic injury: In vivo and in vitro studies. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5976–5984. [Google Scholar] [CrossRef]
- Li, S.Y.; Lo, A.C. Lutein protects RGC-5 cells against hypoxia and oxidative stress. Int. J. Mol. Sci. 2010, 11, 2109–2117. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Green, R.C.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between Serum and Brain Carotenoids, alpha-Tocopherol, and Retinol Concentrations and Cognitive Performance in the Oldest Old from the Georgia Centenarian Study. J. Aging Res. 2013, 2013, 951786. [Google Scholar] [CrossRef]
- Pap, R.; Pandur, E.; Janosa, G.; Sipos, K.; Nagy, T.; Agocs, A.; Deli, J. Lutein Decreases Inflammation and Oxidative Stress and Prevents Iron Accumulation and Lipid Peroxidation at Glutamate-Induced Neurotoxicity. Antioxidants 2022, 11, 2269. [Google Scholar] [CrossRef]
- Zeni, A.L.B.; Camargo, A.; Dalmagro, A.P. Lutein prevents corticosterone-induced depressive-like behavior in mice with the involvement of antioxidant and neuroprotective activities. Pharmacol. Biochem. Behav. 2019, 179, 63–72. [Google Scholar] [CrossRef]
- Orhan, C.; Erten, F.; Er, B.; Tuzcu, M.; Sahin, N.; Durmaz Kursun, O.E.; Juturu, V.; Sahin, K. Lutein/zeaxanthin isomers regulate neurotrophic factors and synaptic plasticity in trained rats. Turk. J. Med. Sci. 2021, 51, 2167–2176. [Google Scholar] [CrossRef]
- Santocono, M.; Zurria, M.; Berrettini, M.; Fedeli, D.; Falcioni, G. Lutein, zeaxanthin and astaxanthin protect against DNA damage in SK-N-SH human neuroblastoma cells induced by reactive nitrogen species. J. Photochem. Photobiol. B 2007, 88, 1–10. [Google Scholar] [CrossRef]
- Renzi-Hammond, L.M.; Bovier, E.R.; Fletcher, L.M.; Miller, L.S.; Mewborn, C.M.; Lindbergh, C.A.; Baxter, J.H.; Hammond, B.R. Effects of a Lutein and Zeaxanthin Intervention on Cognitive Function: A Randomized, Double-Masked, Placebo-Controlled Trial of Younger Healthy Adults. Nutrients 2017, 9, 1246. [Google Scholar] [CrossRef]
- Hammond, B.R., Jr.; Miller, L.S.; Bello, M.O.; Lindbergh, C.A.; Mewborn, C.; Renzi-Hammond, L.M. Effects of Lutein/Zeaxanthin Supplementation on the Cognitive Function of Community Dwelling Older Adults: A Randomized, Double-Masked, Placebo-Controlled Trial. Front. Aging Neurosci. 2017, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Smith, S.J.; Drummond, P.D. The Effects of Lutein and Zeaxanthin Supplementation on Cognitive Function in Adults With Self-Reported Mild Cognitive Complaints: A Randomized, Double-Blind, Placebo-Controlled Study. Front. Nutr. 2022, 9, 843512. [Google Scholar] [CrossRef]
- Dangour, A.D.; Allen, E.; Elbourne, D.; Fasey, N.; Fletcher, A.E.; Hardy, P.; Holder, G.E.; Knight, R.; Letley, L.; Richards, M.; et al. Effect of 2-y n-3 long-chain polyunsaturated fatty acid supplementation on cognitive function in older people: A randomized, double-blind, controlled trial. Am. J. Clin. Nutr. 2010, 91, 1725–1732. [Google Scholar] [CrossRef]
- van de Rest, O.; Geleijnse, J.M.; Kok, F.J.; van Staveren, W.A.; Dullemeijer, C.; Olderikkert, M.G.; Beekman, A.T.; de Groot, C.P. Effect of fish oil on cognitive performance in older subjects: A randomized, controlled trial. Neurology 2008, 71, 430–438. [Google Scholar] [CrossRef]
- Hisatsune, T.; Kaneko, J.; Kurashige, H.; Cao, Y.; Satsu, H.; Totsuka, M.; Katakura, Y.; Imabayashi, E.; Matsuda, H. Effect of Anserine/Carnosine Supplementation on Verbal Episodic Memory in Elderly People. J. Alzheimers Dis. 2016, 50, 149–159. [Google Scholar] [CrossRef]
- Calapai, G.; Bonina, F.; Bonina, A.; Rizza, L.; Mannucci, C.; Arcoraci, V.; Lagana, G.; Alibrandi, A.; Pollicino, C.; Inferrera, S.; et al. A Randomized, Double-Blinded, Clinical Trial on Effects of a Vitis vinifera Extract on Cognitive Function in Healthy Older Adults. Front. Pharmacol. 2017, 8, 776. [Google Scholar] [CrossRef]
- Wechsler, D. Wechsler Memory Scale—Revised Japanese Version; Nihon Bunka Kagaku-sha: Tokyo, Japan, 2001. [Google Scholar]
- Fujiwara, Y.; Suzuki, H.; Yasunaga, M.; Sugiyama, M.; Ijuin, M.; Sakuma, N.; Inagaki, H.; Iwasa, H.; Ura, C.; Yatomi, N.; et al. Brief screening tool for mild cognitive impairment in older Japanese: Validation of the Japanese version of the Montreal Cognitive Assessment. Geriatr. Gerontol. Int. 2010, 10, 225–232. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.; the CONSORT Group. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef]
- Bergeron, D.; Flynn, K.; Verret, L.; Poulin, S.; Bouchard, R.W.; Bocti, C.; Fulop, T.; Lacombe, G.; Gauthier, S.; Nasreddine, Z.; et al. Multicenter Validation of an MMSE-MoCA Conversion Table. J. Am. Geriatr. Soc. 2017, 65, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, H.; Sueyasu, T.; Kontani, M.; Kawashima, H.; Shibata, H.; Koga, Y. Low Doses of Long-chain Polyunsaturated Fatty Acids Affect Cognitive Function in Elderly Japanese Men: A Randomized Controlled Trial. J. Oleo Sci. 2015, 64, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, C.T.; Johnson, L.G. Reliability and validity of a computerized neurocognitive test battery, CNS Vital Signs. Arch. Clin. Neuropsychol. 2006, 21, 623–643. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Nakajima, T.; Wada, T.; Tarui, N.; Ito, S.; Yuasa, Y.; Maoka, T. Analysis of Carotenoids in Human Serum and Erythrocyte by Ultra Performance Liquid Chromatography (UPLC). Carotenoid Sci. 2009, 14, 46–49. [Google Scholar]
- Fujiwara, Y.; Suzuki, H.; Kawai, H.; Hirano, H.; Yoshida, H.; Kojima, M.; Ihara, K.; Obuchi, S. Physical and sociopsychological characteristics of older community residents with mild cognitive impairment as assessed by the Japanese version of the Montreal Cognitive Assessment. J. Geriatr. Psychiatry Neurol. 2013, 26, 209–220. [Google Scholar] [CrossRef]
- Narazaki, K.; Nofuji, Y.; Honda, T.; Matsuo, E.; Yonemoto, K.; Kumagai, S. Normative data for the montreal cognitive assessment in a Japanese community-dwelling older population. Neuroepidemiology 2013, 40, 23–29. [Google Scholar] [CrossRef]
- Carson, N.; Leach, L.; Murphy, K.J. A re-examination of Montreal Cognitive Assessment (MoCA) cutoff scores. Int. J. Geriatr. Psychiatry 2018, 33, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Akema, S.; Mameno, T.; Nakagawa, T.; Inagaki, H.; Fukutake, M.; Hatta, K.; Murotani, Y.; Tsujioka, Y.; Hagino, H.; Higashi, K.; et al. Relationship between occlusal force and psychological frailty in Japanese community-dwelling older adults: The Septuagenarians, Octogenarians, Nonagenarians Investigation with Centenarians study. J. Am. Geriatr. Soc. 2023, 71, 1819–1828. [Google Scholar] [CrossRef]
- Fukutake, M.; Ogawa, T.; Ikebe, K.; Mihara, Y.; Inomata, C.; Takeshita, H.; Matsuda, K.; Hatta, K.; Gondo, Y.; Masui, Y.; et al. Impact of cognitive function on oral perception in independently living older people. Clin. Oral Investig. 2019, 23, 267–271. [Google Scholar] [CrossRef] [PubMed]
- RStudio Team. RStudio: Integrated Development Environment for R; RStudio, PBC: Boston, MA, USA, 2022; Available online: http://www.rstudio.com/ (accessed on 31 March 2023).
- Balduzzi, S.; Rucker, G.; Schwarzer, G. How to perform a meta-analysis with R: A practical tutorial. Evid. Based Ment. Health 2019, 22, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, H.; Ito, M.; Sueyasu, T.; Sasaki, H.; Morita, S.; Kaneda, Y.; Rogi, T.; Kondo, S.; Kouzaki, M.; Tsukiura, T.; et al. Effects of combining exercise with long-chain polyunsaturated fatty acid supplementation on cognitive function in the elderly: A randomised controlled trial. Sci. Rep. 2020, 10, 12906. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, R.; Kato, Y.; Imai, T.; Ando, F.; Shimokata, H. Higher serum EPA or DHA, and lower ARA compositions with age independent fatty acid intake in Japanese aged 40 to 79. Lipids 2013, 48, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Kakutani, S.; Ishikura, Y.; Tateishi, N.; Horikawa, C.; Tokuda, H.; Kontani, M.; Kawashima, H.; Sakakibara, Y.; Kiso, Y.; Shibata, H.; et al. Supplementation of arachidonic acid-enriched oil increases arachidonic acid contents in plasma phospholipids, but does not increase their metabolites and clinical parameters in Japanese healthy elderly individuals: A randomized controlled study. Lipids Health Dis. 2011, 10, 241. [Google Scholar] [CrossRef] [PubMed]
- Ogura, T.; Takada, H.; Okuno, M.; Kitade, H.; Matsuura, T.; Kwon, M.; Arita, S.; Hamazaki, K.; Itomura, M.; Hamazaki, T. Fatty acid composition of plasma, erythrocytes and adipose: Their correlations and effects of age and sex. Lipids 2010, 45, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Sasaki, S.; Kawabata, T.; Hasegawa, K.; Akabane, M.; Tsugane, S. Single measurement of serum phospholipid fatty acid as a biomarker of specific fatty acid intake in middle-aged Japanese men. Eur. J. Clin. Nutr. 2001, 55, 643–650. [Google Scholar] [CrossRef]
- Shiojima, Y.; Takahashi, M.; Takahashi, R.; Moriyama, H.; Bagchi, D.; Bagchi, M.; Akanuma, M. Effect of Dietary Pyrroloquinoline Quinone Disodium Salt on Cognitive Function in Healthy Volunteers: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study. J. Am. Nutr. Assoc. 2022, 41, 796–809. [Google Scholar] [CrossRef]
- Asama, T.; Hiraoka, T.; Ohkuma, A.; Okumura, N.; Yamaki, A.; Urakami, K. Cognitive Improvement and Safety Assessment of a Dietary Supplement Containing Propolis Extract in Elderly Japanese: A Placebo-Controlled, Randomized, Parallel-Group, Double-Blind Human Clinical Study. Evid. Based Complement. Alt. Med. 2021, 2021, 6664217. [Google Scholar] [CrossRef]
- Sekikawa, T.; Kizawa, Y.; Li, Y.; Takara, T. Cognitive function improvement with astaxanthin and tocotrienol intake: A randomized, double-blind, placebo-controlled study. J. Clin. Biochem. Nutr. 2020, 67, 307–316. [Google Scholar] [CrossRef]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008, 11, 75–83. [Google Scholar] [CrossRef]
- McGahon, B.M.; Martin, D.S.; Horrobin, D.F.; Lynch, M.A. Age-related changes in synaptic function: Analysis of the effect of dietary supplementation with omega-3 fatty acids. Neuroscience 1999, 94, 305–314. [Google Scholar] [CrossRef]
- Kelly, L.; Grehan, B.; Chiesa, A.D.; O’Mara, S.M.; Downer, E.; Sahyoun, G.; Massey, K.A.; Nicolaou, A.; Lynch, M.A. The polyunsaturated fatty acids, EPA and DPA exert a protective effect in the hippocampus of the aged rat. Neurobiol. Aging 2011, 32, 2318.e1–2318.e15. [Google Scholar] [CrossRef]
- Kotani, S.; Nakazawa, H.; Tokimasa, T.; Akimoto, K.; Kawashima, H.; Toyoda-Ono, Y.; Kiso, Y.; Okaichi, H.; Sakakibara, M. Synaptic plasticity preserved with arachidonic acid diet in aged rats. Neurosci. Res. 2003, 46, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Park, S.J.; Tamura, M.; Ando, S. Effect of the long-term feeding of dietary lipids on the learning ability, fatty acid composition of brain stem phospholipids and synaptic membrane fluidity in adult mice: A comparison of sardine oil diet with palm oil diet. Mech. Ageing Dev. 1998, 101, 119–128. [Google Scholar] [CrossRef]
- Fukaya, T.; Gondaira, T.; Kashiyae, Y.; Kotani, S.; Ishikura, Y.; Fujikawa, S.; Kiso, Y.; Sakakibara, M. Arachidonic acid preserves hippocampal neuron membrane fluidity in senescent rats. Neurobiol. Aging 2007, 28, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C.; Michael, G.J.; Whelpton, R.; Scott, A.G.; Michael-Titus, A.T. Dietary enrichment with omega-3 polyunsaturated fatty acids reverses age-related decreases in the GluR2 and NR2B glutamate receptor subunits in rat forebrain. Neurobiol. Aging 2007, 28, 424–439. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.H.; Shi, Y.; Wang, L.S.; Yang, Z.R. The influence of orally administered docosahexaenoic acid on cognitive ability in aged mice. J. Nutr. Biochem. 2009, 20, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.Z.; Wang, J.; Sheridan, S.D.; Perlis, R.H.; Rasenick, M.M. N-3 polyunsaturated fatty acids promote astrocyte differentiation and neurotrophin production independent of cAMP in patient-derived neural stem cells. Mol. Psychiatry 2021, 26, 4605–4615. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, E.; Hashimoto, M.; Shido, O. Docosahexaenoic acid promotes neurogenesis in vitro and in vivo. Neuroscience 2006, 139, 991–997. [Google Scholar] [CrossRef]
- Mohn, E.S.; Erdman, J.W., Jr.; Kuchan, M.J.; Neuringer, M.; Johnson, E.J. Lutein accumulates in subcellular membranes of brain regions in adult rhesus macaques: Relationship to DHA oxidation products. PLoS ONE 2017, 12, e0186767. [Google Scholar] [CrossRef]
- Ademowo, O.S.; Dias, I.H.K.; Diaz-Sanchez, L.; Sanchez-Aranguren, L.; Stahl, W.; Griffiths, H.R. Partial Mitigation of Oxidized Phospholipid-Mediated Mitochondrial Dysfunction in Neuronal Cells by Oxocarotenoids. J. Alzheimers Dis. 2020, 74, 113–126. [Google Scholar] [CrossRef]
- Wu, W.; Li, Y.; Wu, Y.; Zhang, Y.; Wang, Z.; Liu, X. Lutein suppresses inflammatory responses through Nrf2 activation and NF-kappaB inactivation in lipopolysaccharide-stimulated BV-2 microglia. Mol. Nutr. Food Res. 2015, 59, 1663–1673. [Google Scholar] [CrossRef]
- Hajizadeh-Sharafabad, F.; Zahabi, E.S.; Malekahmadi, M.; Zarrin, R.; Alizadeh, M. Carotenoids supplementation and inflammation: A systematic review and meta-analysis of randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2022, 62, 8161–8177. [Google Scholar] [CrossRef] [PubMed]
- Ginguay, A.; Regazzetti, A.; Laprevote, O.; Moinard, C.; De Bandt, J.P.; Cynober, L.; Billard, J.M.; Allinquant, B.; Dutar, P. Citrulline prevents age-related LTP decline in old rats. Sci. Rep. 2019, 9, 20138. [Google Scholar] [CrossRef]
- Marefati, N.; Beheshti, F.; Memarpour, S.; Rezaei, M.; Hosseini, M. The effects of pre-treatment with olibanum and its constituent, boswellic acid on synaptic plasticity impairments induced by lipopolysaccharide in rats. Avicenna J. Phytomed. 2021, 11, 68–78. [Google Scholar]
- Moghimi, M.; Parvardeh, S.; Zanjani, T.M.; Ghafghazi, S. Protective effect of alpha-terpineol against impairment of hippocampal synaptic plasticity and spatial memory following transient cerebral ischemia in rats. Iran J. Basic Med. Sci. 2016, 19, 960–969. [Google Scholar]
- Yin, S.T.; Tang, M.L.; Su, L.; Chen, L.; Hu, P.; Wang, H.L.; Wang, M.; Ruan, D.Y. Effects of Epigallocatechin-3-gallate on lead-induced oxidative damage. Toxicology 2008, 249, 45–54. [Google Scholar] [CrossRef]
- Murray, C.A.; Lynch, M.A. Dietary supplementation with vitamin E reverses the age-related deficit in long term potentiation in dentate gyrus. J. Biol. Chem. 1998, 273, 12161–12168. [Google Scholar] [CrossRef]
- Frakes, A.E.; Ferraiuolo, L.; Haidet-Phillips, A.M.; Schmelzer, L.; Braun, L.; Miranda, C.J.; Ladner, K.J.; Bevan, A.K.; Foust, K.D.; Godbout, J.P.; et al. Microglia induce motor neuron death via the classical NF-kappaB pathway in amyotrophic lateral sclerosis. Neuron 2014, 81, 1009–1023. [Google Scholar] [CrossRef]
- Manochkumar, J.; Doss, C.G.P.; El-Seedi, H.R.; Efferth, T.; Ramamoorthy, S. The neuroprotective potential of carotenoids in vitro and in vivo. Phytomedicine 2021, 91, 153676. [Google Scholar] [CrossRef]
- Baierle, M.; Nascimento, S.N.; Moro, A.M.; Brucker, N.; Freitas, F.; Gauer, B.; Durgante, J.; Bordignon, S.; Zibetti, M.; Trentini, C.M.; et al. Relationship between inflammation and oxidative stress and cognitive decline in the institutionalized elderly. Oxid. Med. Cell Longev. 2015, 2015, 804198. [Google Scholar] [CrossRef] [PubMed]
- Hosotani, K.; Kitagawa, M. Measurement of individual differences in intake of green and yellow vegetables and carotenoids in young unmarried subjects. J. Nutr. Sci. Vitaminol. 2007, 53, 207–212. [Google Scholar] [CrossRef] [PubMed]


| Placebo | LCPUFAs + LZ | p | |
|---|---|---|---|
| Age (years) a | 65.0 ± 0.8 | 65.7 ± 1.0 | 0.586 |
| Sex (M/F) b | 17/19 | 15/20 | 0.711 |
| BMI (kg/m2) a | 22.4 ± 0.4 | 22.7 ± 0.5 | 0.682 |
| Education (years) a | 13.9 ± 0.3 | 14.6 ± 0.4 | 0.157 |
| MoCA-J a | 23.3 ± 0.4 | 22.8 ± 0.5 | 0.472 |
| Composite memory a | 95.2 ± 1.2 | 95.1 ± 1.0 | 0.925 |
| Verbal memory a | 50.1 ± 0.8 | 50.8 ± 0.8 | 0.547 |
| Visual memory a | 45.1 ± 0.8 | 44.3 ± 0.6 | 0.409 |
| ARA in plasma PL (%) a | 9.0 ± 0.2 | 9.5 ± 0.2 | 0.159 |
| DHA in plasma PL (%) a | 6.5 ± 0.3 | 6.6 ± 0.2 | 0.821 |
| EPA in plasma PL (%) a | 1.8 ± 0.2 | 1.9 ± 0.2 | 0.583 |
| Group | Baseline | 12 Weeks | Δ | Δ Adjusted | |
|---|---|---|---|---|---|
| Episodic memory | |||||
| Composite memory | Placebo | 95.2 ± 1.2 | 94.7 ± 1.6 | −0.5 ± 1.5 | −0.5 ± 1.3 |
| LCPUFAs + LZ | 95.1 ± 1.0 | 97.7 ± 1.3 * | 2.6 ± 1.2 | 2.6 ± 1.3 | |
| Verbal memory | Placebo | 50.1 ± 0.8 | 49.8 ± 1.0 | −0.3 ± 0.9 | −0.4 ± 0.7 |
| LCPUFAs + LZ | 50.8 ± 0.8 | 52.5 ± 0.8 * | 1.7 ± 0.7 | 1.8 ± 0.7 # | |
| Visual memory | Placebo | 45.1 ± 0.8 | 44.9 ± 0.9 | −0.2 ± 1.0 | 0.1 ± 0.8 |
| LCPUFAs + LZ | 44.3 ± 0.6 | 45.3 ± 0.7 | 1.0 ± 0.9 | 0.7 ± 0.8 |
| Placebo | LCPUFAs + LZ | p | |
|---|---|---|---|
| Age (years) a | 65.1 ± 0.6 | 65.1 ± 0.6 | 0.972 |
| Sex (M/F) b | 48/40 | 48/44 | 0.749 |
| Residential area (Tokyo/Osaka) b | 39/49 | 41/51 | 0.973 |
| BMI (kg/m2) a | 22.8 ± 0.3 | 22.7 ± 0.3 | 0.729 |
| Education (years) a | 14.5 ± 0.2 | 14.9 ± 0.2 | 0.135 |
| MoCA-J a | 23.7 ± 0.3 | 23.8 ± 0.3 | 0.899 |
| Composite memory a | 96.0 ± 0.8 | 95.9 ± 0.8 | 0.948 |
| Verbal memory a | 50.4 ± 0.5 | 50.5 ± 0.5 | 0.938 |
| Visual memory a | 45.4 ± 0.5 | 45.4 ± 0.4 | 0.980 |
| ARA in plasma PL (%) a | 9.8 ± 0.2 | 9.9 ± 0.2 | 0.867 |
| DHA in plasma PL (%) a | 6.6 ± 0.2 | 6.8 ± 0.2 | 0.289 |
| EPA in plasma PL (%) a | 2.1 ± 0.2 | 2.0 ± 0.1 | 0.392 |
| Group | Baseline | 12 Weeks | ∆ | ∆ Adjusted | |
|---|---|---|---|---|---|
| Episodic memory | |||||
| Composite memory | Placebo | 96.0 ± 0.8 | 96.3 ± 0.9 | 0.3 ± 0.8 | 0.3 ± 0.7 |
| LCPUFAs + LZ | 95.9 ± 0.8 | 96.4 ± 0.7 | 0.5 ± 0.8 | 0.5 ± 0.7 | |
| Verbal memory | Placebo | 50.4 ± 0.5 | 51.5 ± 0.5 * | 1.1 ± 0.5 | 1.1 ± 0.5 |
| LCPUFAs + LZ | 50.5 ± 0.5 | 52.0 ± 0.5 ** | 1.5 ± 0.6 | 1.6 ± 0.5 | |
| Visual memory | Placebo | 45.4 ± 0.5 | 44.5 ± 0.6 | −0.8 ± 0.5 | −0.8 ± 0.4 |
| LCPUFAs + LZ | 45.4 ± 0.4 | 44.3 ± 0.4 * | −1.1 ± 0.5 | −1.1 ± 0.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sueyasu, T.; Yasumoto, K.; Tokuda, H.; Kaneda, Y.; Obata, H.; Rogi, T.; Izumo, T.; Kondo, S.; Saito, J.; Tsukiura, T.; et al. Effects of Long-Chain Polyunsaturated Fatty Acids in Combination with Lutein and Zeaxanthin on Episodic Memory in Healthy Older Adults. Nutrients 2023, 15, 2825. https://doi.org/10.3390/nu15132825
Sueyasu T, Yasumoto K, Tokuda H, Kaneda Y, Obata H, Rogi T, Izumo T, Kondo S, Saito J, Tsukiura T, et al. Effects of Long-Chain Polyunsaturated Fatty Acids in Combination with Lutein and Zeaxanthin on Episodic Memory in Healthy Older Adults. Nutrients. 2023; 15(13):2825. https://doi.org/10.3390/nu15132825
Chicago/Turabian StyleSueyasu, Toshiaki, Keisuke Yasumoto, Hisanori Tokuda, Yoshihisa Kaneda, Hidenori Obata, Tomohiro Rogi, Takayuki Izumo, Sumio Kondo, Jiro Saito, Takashi Tsukiura, and et al. 2023. "Effects of Long-Chain Polyunsaturated Fatty Acids in Combination with Lutein and Zeaxanthin on Episodic Memory in Healthy Older Adults" Nutrients 15, no. 13: 2825. https://doi.org/10.3390/nu15132825
APA StyleSueyasu, T., Yasumoto, K., Tokuda, H., Kaneda, Y., Obata, H., Rogi, T., Izumo, T., Kondo, S., Saito, J., Tsukiura, T., & Nakai, M. (2023). Effects of Long-Chain Polyunsaturated Fatty Acids in Combination with Lutein and Zeaxanthin on Episodic Memory in Healthy Older Adults. Nutrients, 15(13), 2825. https://doi.org/10.3390/nu15132825





