Early Life Nutrition and the Role of Complementary Feeding on Later Adherence to the Mediterranean Diet in Children up to 3 Years of Age
Abstract
:1. Introduction
2. Materials and Methods
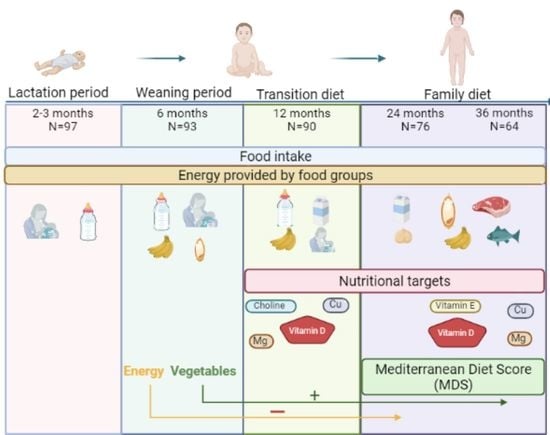
2.1. Sample Recruitment and Study Design
2.2. General Characteristics
2.3. Nutritional Assessment
2.4. Statistical Analyses
3. Results
3.1. Description of the Sample and Mediterranean Diet Score Calculation
3.2. Dietary Intake
3.3. Nutritional Status
3.4. Dietary Quality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beluska-Turkan, K.; Korczak, R.; Hartell, B.; Moskal, K.; Maukonen, J.; Alexander, D.E.; Salem, N.; Harkness, L.; Ayad, W.; Szaro, J.; et al. Nutritional gaps and supplementation in the first 1000 days. Nutrients 2019, 11, 2891. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verduci, E.; Banderali, G.; Montanari, C.; Berni Canani, R.; Cimmino Caserta, L.; Corsello, G.; Mosca, F.; Piazzolla, R.; Rescigno, M.; Terracciano, L.; et al. Childhood dietary intake in Italy: The epidemiological “MY FOOD DIARY” survey. Nutrients 2019, 11, 1129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Health and Medical Research Council. Australian Government Nutrient Reference Values for Australia and New Zealand. What Are Nutrient Reference Values? Available online: https://www.nrv.gov.au/introduction (accessed on 4 February 2022).
- World Health Organisation (WHO). Infant and Young Child Feeding. Available online: https://www.who.int/news-room/fact-sheets/detail/infant-and-young-child-feeding (accessed on 17 January 2022).
- Birch, L.; Doub, A. Learning to eat: Birth to age 2 y. Am. J. Clin. Nutr. 2014, 99, 723–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Emmett, P.M. Dietary patterns during complementary feeding and later outcomes. Nestle Nutr. Inst. Workshop Ser. 2016, 85, 145–154. [Google Scholar] [CrossRef]
- Green, R.; Sutherland, J.; Dangour, A.D.; Shankar, B.; Webb, P. Global dietary quality, undernutrition and non-communicable disease: A longitudinal modelling study. BMJ Open 2016, 6, e009331–e009340. [Google Scholar] [CrossRef] [Green Version]
- United Nations Educational Scientific and Cultural Organization (UNESCO). Evaluation of Nominations for Inscription in 2010 on the Representative List of the Intangible Cultural Heritage of Humanity. Available online: https://ich.unesco.org/en/decisions/5.COM/6.41 (accessed on 10 February 2022).
- Bach-faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef] [Green Version]
- Funtikova, A.N.; Benítez-Arciniega, A.A.; Gomez, S.F.; Fitó, M.; Elosua, R.; Schröder, H. Mediterranean diet impact on changes in abdominal fat and 10-year incidence of abdominal obesity in a Spanish population. Br. J. Nutr. 2014, 111, 1481–1487. [Google Scholar] [CrossRef] [Green Version]
- Tognon, G.; Hebestreit, A.; Lanfer, A.; Moreno, L.A.; Pala, V.; Siani, A.; Tornaritis, M.; De Henauw, S.; Veidebaum, T.; Molnár, D.; et al. Mediterranean diet, overweight and body composition in children from eight European countries: Cross-sectional and prospective results from the IDEFICS study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 205–213. [Google Scholar] [CrossRef]
- Sanmarchi, F.; Esposito, F.; Marini, S.; Masini, A.; Scrimaglia, S.; Capodici, A.; Arrichiello, F.; Ferretti, F.; Rangone, M.; Celenza, F.; et al. Children’s and families’ determinants of health-related behaviors in an italian primary school sample: The “seven days for my health” project. Int. J. Environ. Res. Public Health 2022, 19, 460. [Google Scholar] [CrossRef]
- Scaglioni, S.; De Cosmi, V.; Ciappolino, V.; Parazzini, F.; Brambilla, P.; Agostoni, C. Factors influencing children’s eating behaviours. Nutrients 2018, 10, 706. [Google Scholar] [CrossRef] [Green Version]
- Ocké, M.; de Boer, E.; Brants, H.; van der Laan, J.; Niekerk, M.; van Rossum, C.; Temme, L.; Freisling, H.; Nicolas, G.; Casagrande, C.; et al. PANCAKE—Pilot study for the Assessment of Nutrient intake and food Consumption Among Kids in Europe. EFSA Support. Publ. 2012, 9, 339. [Google Scholar] [CrossRef]
- Slimani, N.; Fahey, M.; Welch, A.; Wirfält, E.; Stripp, C.; Bergström, E.; Linseisen, J.; Schulze, M.; Bamia, C.; Chloptsios, Y.; et al. Diversity of dietary patterns observed in the European Prospective Investigation into Cancer and Nutrition (EPIC) project. Public Health Nutr. 2002, 5, 1311–1328. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Martín, M.; Arboleya, S.; Gueimonde, M.; González, S. Nutritional composition of processed baby foods targeted at infants from 0–12 months. J. Food Compos. Anal. 2019, 79, 55–62. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA) Guidance on the EU Menu methodology. EFSA J. 2014, 12, 3944. [CrossRef] [Green Version]
- Devaney, B.; Kalb, L.; Briefel, R.; Zavitsky-Novak, T.; Clusen, N.; Ziegler, P. Feeding infants and toddlers study: Overview of the study design. J. Am. Diet. Assoc. 2004, 104, 8–13. [Google Scholar] [CrossRef]
- Heinig, M.J.; Nommsen, L.A.; Peerson, J.M.; Lonnerdal, B.; Dewey, K.G. Energy and protein intakes of breast-fed and formula-fed infants during the first year of life and their association with growth velocity: The DARLING Study. Am. J. Clin. Nutr. 1993, 58, 152–161. [Google Scholar] [CrossRef] [Green Version]
- Centro de Ensenanza Superior de Nutricion Humana y Dietetica (CESNID). Tablas de Composicion de Alimentos por Medidas Caseras de Consumo Habitual en España; McGrawHill, Ed.; Publicaciones y Ediciones de la Universidad de Barcelona: Barcelona, Spain, 2008. [Google Scholar]
- Dalmau Serra, J.; Moreno Villares, J.M. Leches de crecimiento en la alimentación infantil. Acta Pediatr. Esp. 2011, 69, 373–378. [Google Scholar]
- United States Department of Agriculture (USDA). Food Composition Databases. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 12 January 2022).
- Trichopoulou, A.; Kouris-blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulou, A. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef] [Green Version]
- European Food Safety Authority (EFSA). DRV Finder. Available online: https://multimedia.efsa.europa.eu/drvs/index.htm (accessed on 17 January 2022).
- Dirección General de Salud Pública; Servicio de Promoción de la Salud; Gobierno de La Rioja. Alimentación de 1-3 años; Gobierno de la Rioja: La Rioja, Spain, 2019. [Google Scholar]
- Roess, A.A.; Jacquier, E.F.; Catellier, D.J.; Carvalho, R.; Lutes, A.C.; Anater, A.S.; Dietz, W.H. Food consumption patterns of infants and toddlers: Findings from the feeding infants and toddlers study (FITS) 2016. J. Nutr. 2018, 148, 1525S–1535S. [Google Scholar] [CrossRef] [Green Version]
- Bailey, R.L.; Catellier, D.J.; Jun, S.; Dwyer, J.T.; Jacquier, E.F.; Anater, A.S.; Eldridge, A.L. Total usual nutrient intakes of US children (under 48 months): Findings from the Feeding Infants and Toddlers Study (FITS) 2016. J. Nutr. 2018, 148, 1557S–1566S. [Google Scholar] [CrossRef]
- Netting, M.J.; Moumin, N.A.; Knight, E.J.; Golley, R.K.; Makrides, M.; Green, T.J. The Australian Feeding Infants and Toddler Study (OzFITS 2021): Breastfeeding and early feeding Practices. Nutrients 2022, 14, 206. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, E.E. Growth of breast-fed and formula-fed infants. Nestlé Nutr. Work. Pediatr. Progr. 2006, 58, 51–63. [Google Scholar] [CrossRef] [Green Version]
- Bell, K.A.; Wagner, C.L.; Feldman, H.A.; Shypailo, R.J.; Belfort, M.B. Associations of infant feeding with trajectories of body composition and growth. Am. J. Clin. Nutr. 2017, 106, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.; Dalrymple, K.V.; Briley, A.L.; Pasupathy, D.; Seed, P.T.; Flynn, A.C.; Poston, L. Mode of infant feeding, eating behaviour and anthropometry in infants at 6-months of age born to obese women—A secondary analysis of the UPBEAT trial. BMC Pregnancy Childbirth 2018, 18, 355. [Google Scholar] [CrossRef]
- Butte, N.F.; Wong, W.W.; Hopkinson, J.M.; Smith, E.O.; Ellis, K.J. Infant feeding mode affects early growth and body composition. Pediatrics 2000, 106, 1355–1366. [Google Scholar] [CrossRef]
- DiSantis, K.I.; Collins, B.N.; Fisher, J.O.; Davey, A. Do infants fed directly from the breast have improved appetite regulation and slower growth during early childhood compared with infants fed from a bottle? Int. J. Behav. Nutr. Phys. Act. 2011, 8, 89–100. [Google Scholar] [CrossRef] [Green Version]
- Switkowski, K.M.; Aris, I.M.; Gingras, V.; Oken, E.; Young, J.G. Estimated causal effects of complementary feeding behaviors on early childhood diet quality in a US cohort. Am. J. Clin. Nutr. 2022, 115, 1105–1114. [Google Scholar] [CrossRef]
- Agostoni, C.; Decsi, T.; Fewtrell, M.; Goulet, O.; Kolacek, S.; Koletzko, B.; Michaelsen, K.F.; Moreno, L.; Puntis, J.; Rigo, J.; et al. Complementary Feeding: A Commentary by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2008, 46, 99–110. [Google Scholar] [CrossRef] [Green Version]
- Giovannini, M.; Riva, E.; Banderali, G.; Scaglioni, S.; Veehof, S.H.E.; Sala, M.; Radaelli, G.; Agostoni, C. Feeding practices of infants through the first year of life in Italy. Acta Paediatr. Int. J. Paediatr. 2004, 93, 492–497. [Google Scholar] [CrossRef]
- Wright, C.M.; Parkinson, K.N.; Drewett, R.F. Why are babies weaned early? Data from a prospective population based cohort study. Arch. Dis. Child. 2004, 89, 813–816. [Google Scholar] [CrossRef] [Green Version]
- Alvisi, P.; Brusa, S.; Alboresi, S.; Amarri, S.; Bottau, P.; Cavagni, G.; Corradini, B.; Landi, L.; Loroni, L.; Marani, M.; et al. Recommendations on complementary feeding for healthy, full-term infants. Ital. J. Pediatr. 2015, 41, 36–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleischer, D.M.; Spergel, J.M.; Assa’ad, A.H.; Pongracic, J.A. Primary prevention of allergic disease through nutritional interventions. J. Allergy Clin. Immunol. Pract. 2013, 1, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Caffarelli, C.; Di Mauro, D.; Mastrorilli, C.; Bottau, P.; Cipriani, F.; Ricci, G. Solid food introduction and the development of food allergies. Nutrients 2018, 10, 1790. [Google Scholar] [CrossRef] [Green Version]
- Schack-Nielsen, L.; Sørensen, T.I.; Mortensen, E.L.; Michaelsen, K.F. Late introduction of complementary feeding, rather than duration of breastfeeding, may protect against adult overweight. Am. J. Clin. Nutr. 2010, 91, 619–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreiras, O.; Carbajal, Á.; Cabrera, L.; Cuadrado, C. Tablas de composición de alimentos. In Pirámide de la Alimentacion Saludable Para la Población Spañola; Sociedad Española de Nutrición Comunitaria (SENC): Madrid, Spain, 2007. [Google Scholar]
- Comunidad de Madrid; Consejería de Sanidad. Recomendaciones Dietético Nutricionales; Madrid, España, 2013. Available online: http://www.sendimad.org/nuevasendimad/sites/default/files/Recomendaciones%20Dietetico%20Nut%202015%20SERMAS.pdf (accessed on 14 March 2022).
- Fox, M.K.; Pac, S.; Devaney, B.; Jankowski, L. Feeding Infants and Toddlers study: What foods are infants and toddlers eating? J. Am. Diet. Assoc. 2004, 104, 22–30. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farinetti, A.; Zurlo, V.; Manenti, A.; Coppi, F.; Mattioli, A.V. Mediterranean diet and colorectal cancer: A systematic review. Nutrition 2017, 43–44, 83–88. [Google Scholar] [CrossRef]
- Dalmau, J.; Moráis, A.; Martínez, V.; Peña-Quintana, L.; Varea, V.; Martínez, M.J.; Soler, B. Evaluación de la alimentación y consumo de nutrientes en menores de 3 años. Estudio piloto ALSALMA. An. Pediatr. 2014, 81, 22–31. [Google Scholar] [CrossRef]
- Dalmau, J.; Peña-Quintana, L.; Moráis, A.; Martínez, V.; Varea, V.; Martínez, M.J.; Soler, B. Análisis cuantitativo de la ingesta de nutrientes en niños menores de 3 años. Estudio ALSALMA. An. Pediatr. 2015, 82, 255–266. [Google Scholar] [CrossRef]
- Gómez-Martín, M.; Domínguez, B.; Gueimonde, M.; González, S. Identification of nutritional targets in spanish children belonging to the laydi cohort for the development of health promotion strategies in the first two years of life. Int. J. Environ. Res. Public Health 2021, 18, 939. [Google Scholar] [CrossRef]
- Foterek, K.; Hilbig, A.; Kersting, M.; Alexy, U. Age and time trends in the diet of young children: Results of the DONALD study. Eur. J. Nutr. 2016, 55, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, 1080–1086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manzano, A.S.; Rodriguez, B.; Piñeiro, R.; Nuñez, E.; Escrig, R.; Cabrera, L.; Calvo, C. Utilización de Vitamina D en Pediatria; Asociación Española de Pediatría: Madrid, Spain, 2019. [Google Scholar]
- Rodríguez-Dehli, A.C.; Riaño-Galán, I.; Fernández-Somoano, A.; Navarrete-Muñoz, E.M.; Espada, M.; Vioque, J.; Tardón, A. Hipovitaminosis D y factores asociados a los 4 años en el norte de España. An. Pediatr. 2017, 86, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Martínez, E.; Llull, R.; del Mar Bibiloni, M.; Pons, A.; Tur, J.A. Adherence to the Mediterranean dietary pattern among Balearic Islands adolescents. Br. J. Nutr. 2010, 103, 1657–1664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grosso, G.; Marventano, S.; Buscemi, S.; Scuderi, A.; Matalone, M.; Platania, A.; Giorgianni, G.; Rametta, S.; Nolfo, F.; Galvano, F.; et al. Factors associated with adherence to the Mediterranean diet among adolescents living in Sicily, southern Italy. Nutrients 2013, 5, 4908–4923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lazarou, C.; Panagiotakos, D.B.; Matalas, A.-L. Level of adherence to the Mediterranean diet among children from Cyprus: The CYKIDS study. Public Health Nutr. 2009, 12, 991–1000. [Google Scholar] [CrossRef] [Green Version]
- Da Rocha, B.R.S.; Rico-Campà, A.; Romanos-Nanclares, A.; Ciriza, E.; Barbosa, K.B.F.; Martínez-González, M.Á.; Martín-Calvo, N. Adherence to Mediterranean diet is inversely associated with the consumption of ultra-processed foods among Spanish children: The SENDO project. Public Health Nutr. 2021, 24, 3294–3303. [Google Scholar] [CrossRef]



| 24 Months | 36 Months | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Boys (n = 44) | Girls (n = 32) | Boys (n = 37) | Girls (n = 27) | ||||||
| Mean ± SD | Median | Mean ± SD | Median | Mean ± SD | Median | Mean ± SD | Median | Score | |
| MUFA/SFA ratio a | 1.19 ± 0.35 | <1.07 ≥1.08 | 1.26 ± 0.40 | <1.13 ≥1.14 | 1.23 ± 0.30 | <1.19 ≥1.20 | 1.48 ± 0.45 | <1.35 ≥1.36 | 0 1 |
| Legumes (g/day) | 34.85 ± 21.35 | <29.93 ≥29.94 | 27.34 ± 16.13 | <24.28 ≥24.29 | 36.97 ± 17.73 | <35.70 ≥35.71 | 37.50 ± 28.02 | <34.28 ≥34.29 | 0 1 |
| Cereals and potatoes (g/day) | 121.07 ± 55.94 | <115.34 ≥115.35 | 105.12 ± 56.91 | <98.47 ≥98.48 | 147.85 ± 52.66 | <132.12 ≥132.13 | 130.24 ± 71.61 | <116.42 ≥116.43 | 0 1 |
| Vegetables (g/day) | 101.24 ± 75.13 | <85.60 ≥85.61 | 107.38 ± 98.27 | <89.04 ≥89.05 | 106.83 ± 91.60 | <84.85 ≥84.86 | 114.40 ± 143.73 | <64.28 ≥64.29 | 0 1 |
| Fruits (g/day) | 173.95 ± 86.96 | <167.23 ≥167.24 | 186.63 ± 121.79 | <140.16 ≥140.17 | 212.68 ± 99.61 | <202.28 ≥202.29 | 171.52 ± 104.47 | <145.07 ≥145.08 | 0 1 |
| Dairy products b | 438.48 ± 206.20 | ≥465.63 <465.62 | 397.31 ± 208.15 | ≥377.94 <377.93 | 470.34 ± 224.86 | ≥462.50 <462.49 | 274.05 ± 170.06 | ≥275.0 <274.9 | 0 1 |
| Meat (g/day) | 41.20 ± 23.95 | ≥39.29 <39.28 | 39.30 ± 21.06 | ≥39.64 <39.63 | 50.19 ± 35.13 | ≥45.71 <45.70 | 48.00 ± 31.93 | ≥38.57 <38.56 | 0 1 |
| Lactation Period | Weaning Period | Transition Diet | Family Diet | |||
|---|---|---|---|---|---|---|
| 2–3 Months | 6 Months | 12 Months | 24 Months | 36 Months | ||
| Subjects (n) | 97 | 93 | 90 | 76 | 64 | |
| Gender | Male | 56 (57.7) | 53 (57.0) | 51 (56.7) | 44 (57.9) | 37 (57.8) |
| Female | 41 (42.3) | 40 (43.0) | 39 (43.3) | 32 (42.1) | 27 (42.2) | |
| Weight (kg) | 5.82 ± 0.83 | 7.76 ± 0.84 * | 9.97 ± 1.52 * | 12.66 ± 1.76 * | 15.22 ± 2.52 * | |
| Height (cm) | 59.98 ± 2.88 | 67.73 ± 2.36 * | 75.89 ± 3.37 * | 88.46 ± 3.84 * | 96.27 ± 4.58 * | |
| Lactation | BF | 52 (53.6) | 39 (41.9) | 23 (25.6) * | 10 (13.3) | 5 (7.8) |
| IF | 45 (46.4) | 54 (58.1) | 56 (62.2) | 9 (12.0) * | 4 (6.3) | |
| Other | 0 | 0 | 11 (12.2) | 56 (74.7) * | 55 (85.9) | |
| Supplementation | No | 7 (7.3) | 6 (6.5) | 10 (11.1) | 75 (98.7) * | 64 (100) |
| Yes | 89 (92.7) | 87 (93.5) | 80 (88.9) | 1 (1.3) * | 0 | |
| Food texture | Mashed food | 0 | 80 (87.9) | 8 (8.9) * | 0 | 0 |
| Semi-solid | 0 | 9 (9.9) | 60 (66.7) * | 1 (1.3) * | 1 (1.6) | |
| Regular | 0 | 2 (2.2) | 22 (24.4) * | 75 (98.7) * | 63 (98.4) | |
| Special diet | No | 0 | 88 (94.6) | 87 (96.7) | 73 (96.1) | 63 (98.4) |
| Yes | 0 | 5 (5.4) | 3 (3.3) | 3 (3.9) | 1 (1.6) | |
| Delivery type | Vaginal | 75 (77.3) | 71 (76.3) | 68 (75.6) | 58 (76.3) | 48 (75.0) |
| C-section | 22 (22.7) | 22 (23.7) | 22 (24.4) | 18 (23.7) | 16 (25.0) | |
| DRV | 12 Months n= 90 | DRV Compliance 12 Months (%) | 24 Months n = 76 | DRV Compliance 24 Months (%) | 36 Months n = 64 | DRV Compliance 36 Months (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| AI | AR | Median (IR) | >AI | <AR | Median (IR) | >AI | <AR | Median (IR) | >AI | <AR | |
| Energy (kcal/day) | - | - | 1019.0 (925.0–1146.1) a | - | - | 1147.4 (941.9–1363.8) a | - | - | 1253.0 (1016.8–1470.0) a | - | - |
| Macronutrients | - | - | - | - | - | - | |||||
| Fat (g/day) | - | - | 36.2 (32.5–40.6) a | - | - | 41.1 (35.1–47.5) a | - | - | 46.2 (36.0–54.8) a | - | - |
| SFA (g/day) | ALAP | - | 10.7 (7.4–12.8) a | - | - | 15.2 (11.5–18.3) b | - | - | 15.7 (12.0–19.6) b | - | - |
| MUFA (g/day) | - | - | 10.2 (9.0–12.4) a | - | - | 16.5 (13.7–19.7) b | - | - | 17.9 (14.5–26.1) b | - | - |
| PUFA (g/day) | - | - | 2.8 (2.2–3.4) a | - | - | 4.3 (3.6–5.1) a,b | - | - | 5.5 (4.1–6.7) b | - | - |
| Carbohydrate (g/day) | - | - | 130.1 (119.5–151.9) a | - | - | 139.5 (109.0–168.4) a | - | - | 148.5 (122.5–172.4) a | - | - |
| Dietary fiber (g/day) | 10 | 13.6 (11.4–16.3) a | 83.3 | - | 14.3 (11.2–16.8) a | 84.2 | - | 15.8 (12.1–18.8) a | 79.7 | - | |
| Protein (g/day) | - | - | 36.8 (30.7–43.1) a | 52.2 (44.1–63.0) a | 55.7 (47.6–65.9) a | - | - | ||||
| Animal protein (g/day) | - | - | 17.0 (12.4–23.6) a | 32.2 (24.0–39.2) b | 33.9 (28.4–40.9) b | - | - | ||||
| Vegetal protein (g/day) | - | - | 11.8 (9.2–15.3) a | 17.1 (13.2–22.0) a,b | 20.8 (15.8–25.3) b | - | - | ||||
| Micronutrients | - | - | |||||||||
| Vitamin A (μg RAE/day) | - | 205 | 959.7 (737.7–1282.4) a | - | 0 | 564.8 (388.7–841.3) b | - | 6.6* | 564.1 (364.1–861.0) b | - | 6.3 |
| Thiamin (mg/day) | - | 0.072 | 1.0 (0.8–1.2) a | - | 0 | 0.9 (0.8–1.1) a | - | 0 | 1.1 (0.8–1.3) a | - | 0 |
| Riboflavin (mg/day) | - | 0.5 | 1.4 (1.1–1.6) a | - | 1.1 | 1.4 (1.1–1.7) a | - | 2.6 | 1.5 (1.1–1.8) a | - | 0 |
| Niacin (mg/day) | - | 1.3 | 9.3 (8.0–12.1) a | - | 0 | 10.8 (8.2–13.4) a | - | 0 | 11.4 (9.3–14.9) a | - | 0 |
| Vitamin B-6 (mg/day) | - | 0.5 | 1.4 (1.2–1.7) a | - | 1.1 | 1.4 (1.1–1.8) a | - | 0 | 1.6 (1.2–1.9) a | - | 0 |
| Folate (μg DFE/day) | - | 90 | 359.0 (249.2–462.7) a | - | 1.1 | 446.2 (342.4–570.3) a | - | 1.3 | 566.7 (413.9–676.2) a | - | 0 |
| Vitamin B-12 (μg/day) | 1.5 | 2.4 (1.8–3.4) a | 88.9 | - | 3.0 (2.2–4.2) a | 92.1 | - | 3.0 (2.3–3.8) a | 92.2 | - | |
| Vitamin C (mg/day) | 15 | 162.3 (122.0–224.8) a | - | 0 | 93.5 (57.7–129.3) b | - | 1.3 | 88.5 (59.6–149.0) b | - | 1.6 | |
| Vitamin D (μg/day) | 15 | - | 6.0 (3.8–7.3) a | 0 | - | 2.8 (0.9–4.1) b | 1.3 | - | 2.3 (1.1–4.0) b | 0 | - |
| Vitamin E (mg/day) | 6 $ | - | 8.7 (6.7–11.0) a | 80.0 | - | 5.1 (4.0–6.7) b | 38.2 * | - | 5.7 (4.5–7.5) b | 12.5 * | - |
| Vitamin K (μg/day) | 12 | - | 45.0 (27.4–82.1) a | 94.4 | - | 31.7 (19.9–75.4) a | 94.7 | - | 38.8 (21.1–116.5) a | 90.6 | - |
| Choline (mg/day) | 140 | - | 112.4 (88.2–155.8) a | 27.8 | - | 234.8 (184.4–286.5) b | 88.2 * | - | 266.3 (187.0–299.4) b | 89.1 | - |
| Calcium (mg/day) | - | 390 | 643.3 (525.3–736.1) a | - | 8.9 | 730.0 (577.8–975.8) a | - | 7.9 | 650.1 (527.4–889.1) a | - | 7.8 |
| Copper (mg/day) | 0.7 ‡ | - | 0.6 (0.4–0.7) a | 27.8 | - | 0.9 (0.7–1.0) a | 73.7 * | - | 1.0 (0.7–1.2) a | 48.4 * | - |
| Phosphorus (mg/day) | 250 | - | 631.0 (551.8–782.9) a | 100 | - | 930.0 (777.1–1121.5) a | 100 | - | 948.9 (774.4–1115.4) a | 100 | - |
| Potassium (mg/day) | 800 | - | 2070.7 (1712.9–2542.4) a | 98.9 | - | 2365.8 (1836.7–2657.4) a | 98.7 | - | 2506.2 (1941.0–2739.1) a | 100 | - |
| Iron (mg/day) | - | 5 | 9.4 (7.6–11.3) a | - | 2.2 | 7.6 (6.3–8.8) a | - | 13.2 * | 8.3 (6.6–9.9) a,b | - | 6.4 |
| Magnesium (mg/day) | 170 & | - | 147.7 (123.0–186.9) a | 35.6 | - | 194.8 (155.8–232.1) a | 65.8* | - | 203.7 (166.6–240.1) a | 28.1 * | - |
| Selenium (μg/day) | 15 | - | 30.9 (21.5–46.1) a | 95.6 | - | 66.4 (49.5–79.7) b | 100 | - | 68.5 (50.1–81.5) b | 100 | - |
| Zinc (mg/day) | - | 3.6 | 5.8 (4.5–6.8) a | - | 10.0 | 6.4 (5.3–7.9) a | - | 3.9 | 6.6 (5.6–7.8) a | - | 4.7 |
| MDS | ||||
|---|---|---|---|---|
| Predictors | R2 | β | p | |
| MDS at 24 months | ||||
| Model | Energy | 0.069 | −0.286 | 0.013 |
| MDS at 36 months | ||||
| Model | Vegetables | 0.105 | 0.365 | 0.006 |
| Fruits | 0.105 | −0.265 | 0.044 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Martín, M.; Herrero-Morín, D.; Arboleya, S.; Gueimonde, M.; González, S. Early Life Nutrition and the Role of Complementary Feeding on Later Adherence to the Mediterranean Diet in Children up to 3 Years of Age. Nutrients 2022, 14, 1664. https://doi.org/10.3390/nu14081664
Gómez-Martín M, Herrero-Morín D, Arboleya S, Gueimonde M, González S. Early Life Nutrition and the Role of Complementary Feeding on Later Adherence to the Mediterranean Diet in Children up to 3 Years of Age. Nutrients. 2022; 14(8):1664. https://doi.org/10.3390/nu14081664
Chicago/Turabian StyleGómez-Martín, María, David Herrero-Morín, Silvia Arboleya, Miguel Gueimonde, and Sonia González. 2022. "Early Life Nutrition and the Role of Complementary Feeding on Later Adherence to the Mediterranean Diet in Children up to 3 Years of Age" Nutrients 14, no. 8: 1664. https://doi.org/10.3390/nu14081664
APA StyleGómez-Martín, M., Herrero-Morín, D., Arboleya, S., Gueimonde, M., & González, S. (2022). Early Life Nutrition and the Role of Complementary Feeding on Later Adherence to the Mediterranean Diet in Children up to 3 Years of Age. Nutrients, 14(8), 1664. https://doi.org/10.3390/nu14081664