Interplay of Walnut Consumption, Changes in Circulating miRNAs and Reduction in LDL-Cholesterol in Elders
Abstract
:1. Introduction
2. Materials and Methods
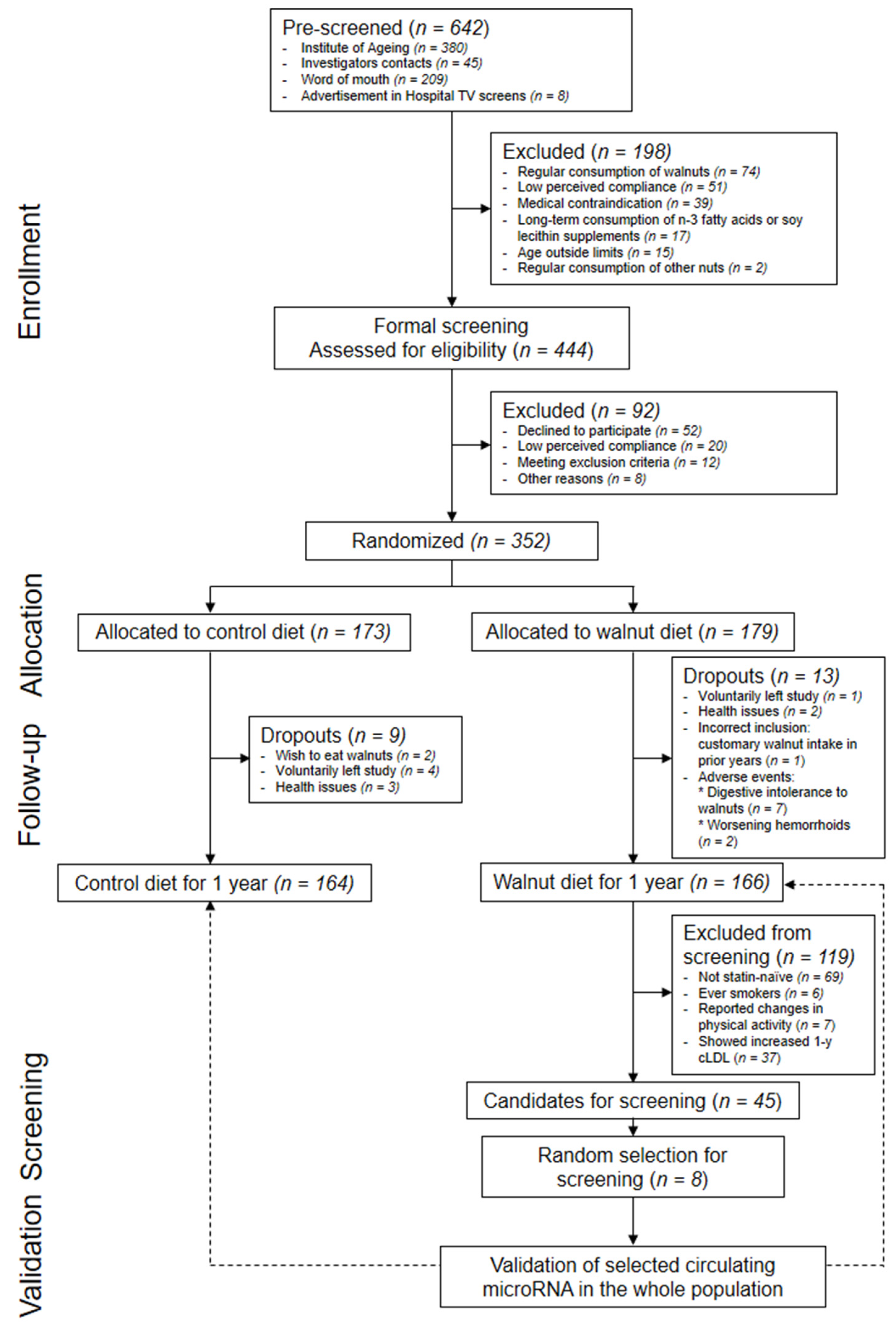
2.1. Study Design and Participants
2.2. Laboratory Determinations
2.3. Nutritional Examination
2.4. Statistical Analyses
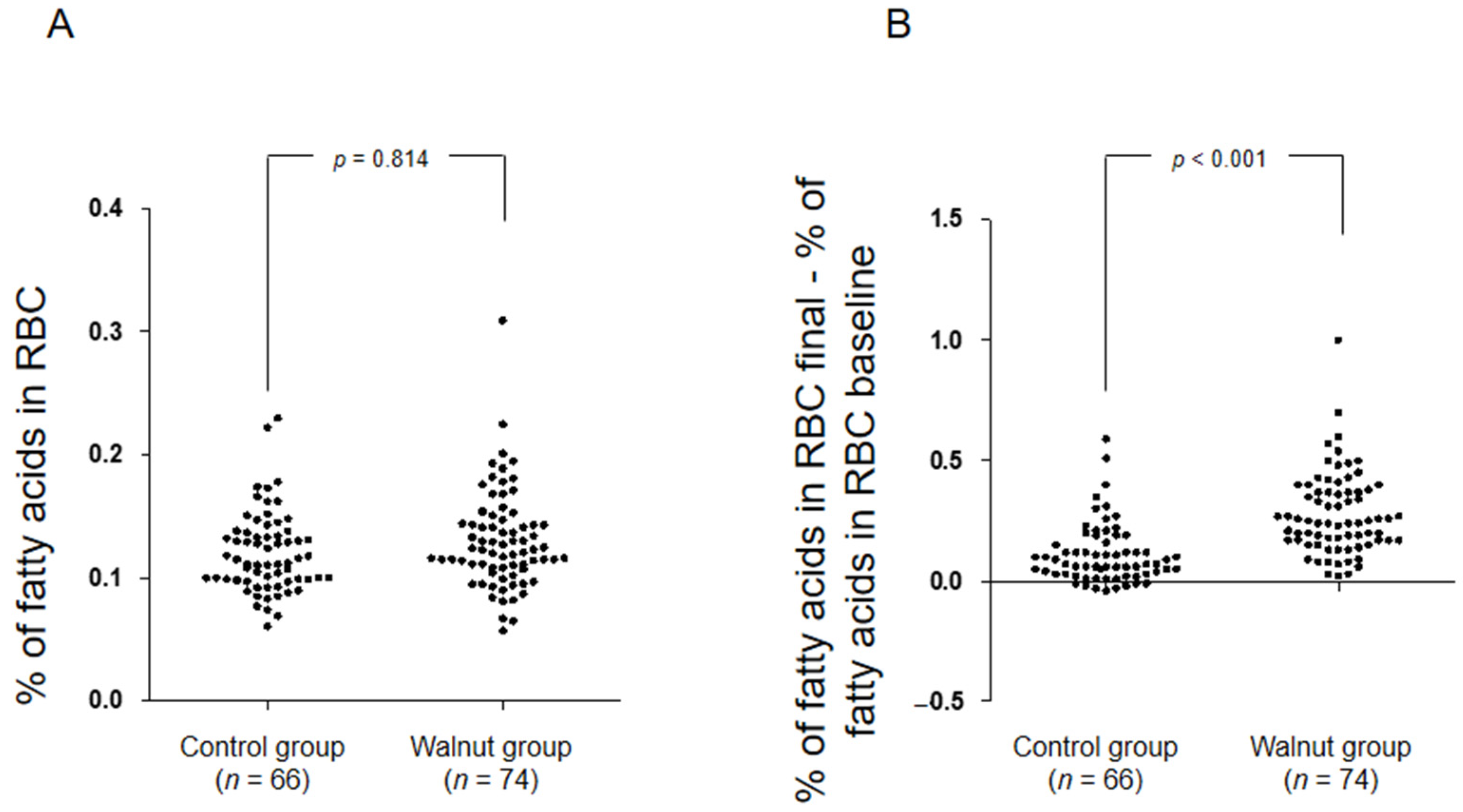
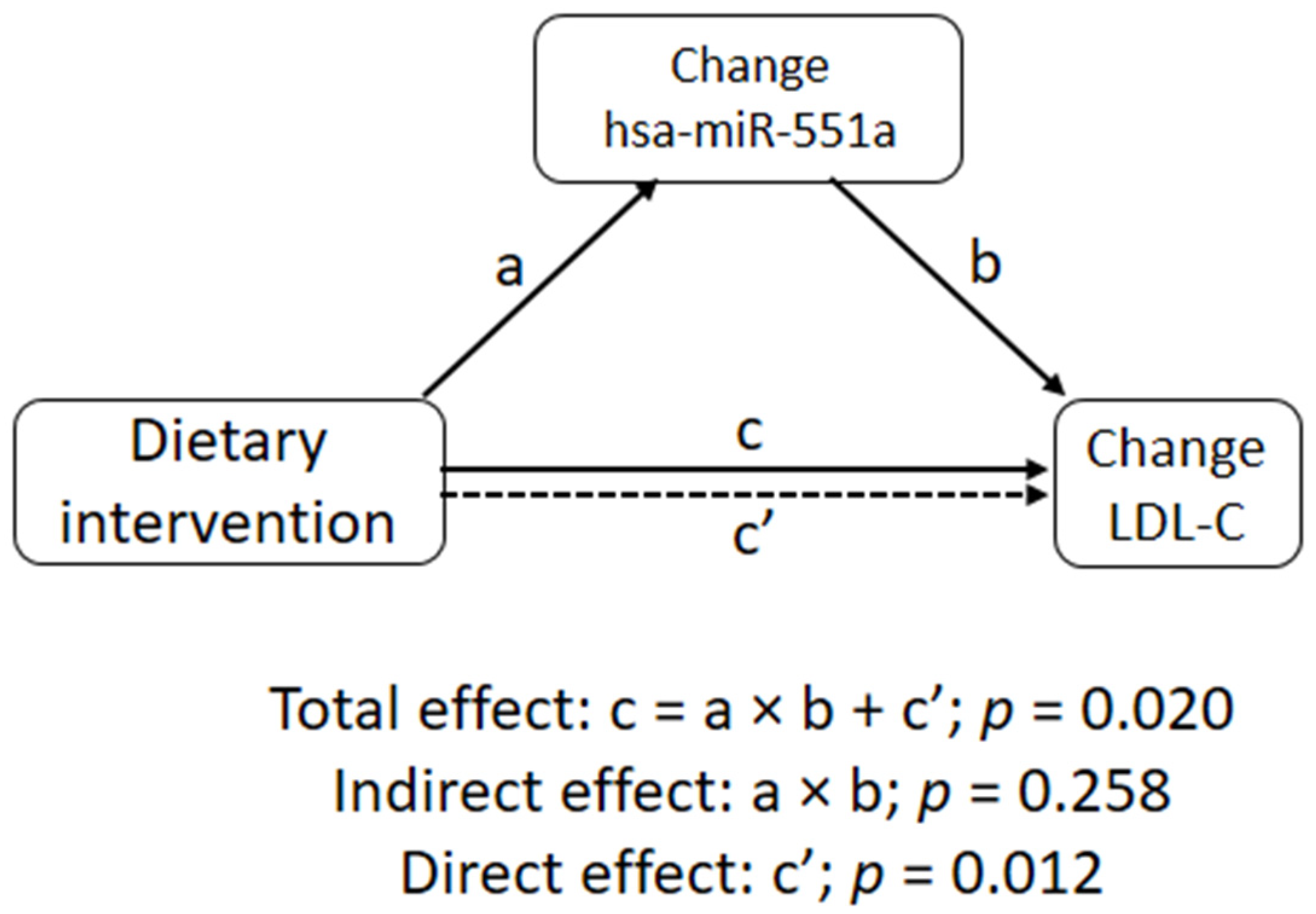
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, K.; Hui, S.; Wang, B.; Kaliannan, K.; Guo, X.; Liang, L. Comparative effects of different types of tree nut consumption on blood lipids: A network meta-analysis of clinical trials. Am. J. Clin. Nutr. 2020, 111, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Oda, K.; Ros, E. Nut consumption and blood lipid levels: A pooled analysis of 25 intervention trials. Arch. Intern. Med. 2010, 170, 821–827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kris-Etherton, P.M.; Yu-Poth, S.; Sabaté, J.; Ratcliffe, H.E.; Zhao, G.; Etherton, T.D. Nuts and their bioactive constituents: Effects on serum lipids and other factors that affect disease risk. Am. J. Clin. Nutr. 1999, 70, 504S–511S. [Google Scholar] [CrossRef] [Green Version]
- Ros, E. Nuts and CVD. Brit. J. Nutr. 2015, 113, S111–S120. [Google Scholar] [CrossRef] [Green Version]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef] [Green Version]
- Zampetaki, A.; Willeit, P.; Drozdov, I.; Kiechl, S.; Mayr, M. Profiling of circulating microRNAs: From single biomarkers to re-wired networks. Cardiovasc. Res. 2012, 93, 555–562. [Google Scholar] [CrossRef] [Green Version]
- Rotllan, N.; Price, N.; Pati, P.; Goedeke, L.; Fernández-Hernando, C. microRNAs in lipoprotein metabolism and cardiometabolic disorders. Atherosclerosis 2016, 246, 352–360. [Google Scholar] [CrossRef] [Green Version]
- Fung, E.C.; Butt, A.N.; Eastwood, J.; Swaminathan, R.; Sodi, R. Circulating microRNA in cardiovascular disease. Adv. Clin. Chem. 2019, 91, 99–122. [Google Scholar]
- Mantilla-Escalante, D.C.; López de las Hazas, M.C.; Gil-Zamorano, J.; del Pozo-Acebo, L.; Crespo, M.C.; Martín-Hernández, R.; del Saz, A.; Tomé-Carneiro, J.; Cardona, F.; Cornejo-Pareja, I.; et al. Postprandial Circulating miRNAs in response to a dietary fat challenge. Nutrients 2019, 11, 1326. [Google Scholar] [CrossRef] [Green Version]
- Aganzo, M.; Montojo, M.T.; López de Las Hazas, M.C.; Martínez-Descals, A.; Ricote-Vila, M.; Sanz, R.; González-Peralta, I.; Martín-Hernández, R.; de Dios, O.; Garcés, C.; et al. Customized dietary intervention avoids unintentional weight loss and modulates circulating mirnas footprint in Huntington’s disease. Mol. Nutr. Food Res. 2018, 62, e1800619. [Google Scholar] [CrossRef]
- Lin, Q.; Ma, L.; Liu, Z.; Yang, Z.; Wang, J.; Liu, J.; Jiang, G. Targeting microRNAs: A new action mechanism of natural compounds. Oncotarget 2017, 8, 15961–15970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quintanilha, B.J.; Reis, B.Z.; Duarte, G.B.S.; Cozzolino, S.M.F.; Rogero, M.M. Nutrimiromics: Role of microRNAs and nutrition in modulating inflammation and chronic diseases. Nutrients 2017, 9, 1168. [Google Scholar] [CrossRef] [PubMed]
- López de las Hazas, M.C.; Gil-Zamorano, J.; Cofán, M.; Mantilla-Escalante, D.C.; García-Ruiz, A.; del Pozo-Acebo, L.; Pastor, O.; Yañez-Mo, M.; Mazzeo, C.; Serra-Mir, M.; et al. One-year dietary supplementation with walnuts modifies exosomal miRNA in elderly subjects. Eur. J. Nutr. 2021, 60, 1999–2011. [Google Scholar] [CrossRef] [PubMed]
- Ortega, F.J.; Cardona-Alvarado, M.I.; Mercader, J.M.; Moreno-Navarrete, J.M.; Moreno, M.; Sabater, M.; Fuentes-Batllevell, N.; Ramírez-Chávez, E.; Ricart, W.; Molina-Torres, J.; et al. Circulating profiling reveals the effect of a polyunsaturated fatty acid-enriched diet on common microRNAs. J. Nutr. Biochem. 2015, 26, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Alonso, P.; Giardina, S.; Salas-Salvadó, J.; Arcelin, P.; Bulló, M. Chronic pistachio intake modulates circulating microRNAs related to glucose metabolism and insulin resistance in prediabetic subjects. Eur. J. Nutr. 2017, 56, 2181–2191. [Google Scholar] [CrossRef] [PubMed]
- Mantilla-Escalante, D.C.; López de Las Hazas, M.C.; Crespo, M.C.; Martín-Hernández, R.; Tomé-Carneiro, J.; Del Pozo-Acebo, L.; Salas-Salvadó, J.; Bulló, M.; Dávalos, A. Mediterranean diet enriched in extra-virgin olive oil or nuts modulates circulating exosomal non-coding RNAs. Eur. J. Nutr. 2021, 60, 4279–4293. [Google Scholar] [CrossRef]
- Rajaram, S.; Cofán, M.; Sala-Vila, A.; Haddad, E.; Serra-Mir, M.; Bitok, E.; Roth, I.; Freitas-Simoes, T.M.; Kaur, A.; Valls-Pedret, C.; et al. Effects of walnut consumption for 2 years on lipoprotein subclasses among healthy elders. Circulation 2021, 144, 1083–1085. [Google Scholar] [CrossRef]
- Rajaram, S.; Valls-Pedret, C.; Cofán, M.; Sabaté, J.; Serra-Mir, M.; Pérez-Heras, A.M.; Arechiga, A.; Casaroli-Marano, R.P.; Alforja, S.; Sala-Vila, A.; et al. The Walnuts and Healthy Aging Study (WAHA): Protocol for a nutritional intervention trial with walnuts on brain aging. Front. Aging Neurosci. 2017, 8, 333. [Google Scholar] [CrossRef] [Green Version]
- Sala-Vila, A.; Valls-Pedret, C.; Rajaram, S.; Coll-Padrós, N.; Cofán, M.; Serra-Mir, M.; Pérez-Heras, A.M.; Roth, I.; Freitas-Simoes, T.M.; Doménech, M.; et al. Effect of a 2-year diet intervention with walnuts on cognitive decline. The Walnuts and Healthy Aging (WAHA) study: A randomized controlled trial. Am. J. Clin. Nutr. 2020, 111, 590–600. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Li, Z.; Cao, Y.; Jie, Z.; Liu, Y.; Li, Y.; Li, J.; Zhu, G.; Liu, Z.; Tu, Y.; Peng, G.; et al. miR-495 and miR-551a inhibit the migration and invasion of human gastric cancer cells by directly interacting with PRL-3. Cancer Lett. 2012, 323, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Loo, J.M.; Scherl, A.; Nguyen, A.; Man, F.Y.; Weinberg, E.; Zeng, Z.; Saltz, L.; Paty, P.B.; Tavazoie, S.F. Extracellular metabolic energetics can promote cancer progression. Cell 2015, 160, 393–406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anuj; Arivazhagan, L.; Venkatraman, G.; Rayala, S.K. Increased expression of microRNA 551a by c-fos reduces focal adhesion kinase levels and blocks tumorigenesis. Mol. Cell Biol. 2019, 39, e00577-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, Z.; Sha, X. Demethoxycurcumin inhibited human epithelia ovarian cancer cells’ growth via up-regulating miR-551a. Tumour Biol. 2017, 3, 1010428317694302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, D.; Dai, C.; Zhou, L.; Li, Y.; Liu, K.; Deng, Y.J.; Li, N.; Zheng, Y.; Hao, Q.; Yang, S.; et al. Meta-analysis of the association between nut consumption and the risks of cancer incidence and cancer-specific mortality. Aging 2020, 12, 10772–10794. [Google Scholar] [CrossRef] [PubMed]
| Variable | Control Diet (n = 164) | Walnut Diet (n = 166) | p-Value a |
|---|---|---|---|
| Women—Number (%) | 111 (67.7) | 114 (68.7) | 0.847 |
| Age—year | 68.8 (68.3; 69.3) | 69.0 (68.5; 69.5) | 0.470 |
| Ever Smoking, Yes—Number (%) | 47 (28.7) | 46 (27.7) | 0.848 |
| Weight—kg | 71.0 (69.0; 73.0) | 69.9 (68.1; 71.7) | 0.405 |
| Body Mass Index—kg/m2 | 27.4 (26.8; 28.1) | 26.8 (26.2; 27.3) | 0.101 |
| Waist Circumference—cm | 99 (97; 101) | 97 (96; 99) | 0.128 |
| Hypertension—Number (%) | 94 (57.3) | 89 (53.6) | 0.499 |
| Type 2 Diabetes—Number (%) | 16 (9.8) | 20 (12.0) | 0.504 |
| Dyslipidemia—Number (%) | 86 (52.4) | 89 (53.6) | 0.831 |
| Physical Activity b | 2412 (1525; 3834) | 2629 (1770; 3909) | 0.340 |
| Variable | Visit | Control Diet (n = 164) | Walnut Diet (n = 166) | p-Value a |
|---|---|---|---|---|
| Total Cholesterol—mg/dL | Baseline | 207.5 (202.5; 212.4) | 203.0 (198.0; 207.9) | 0.206 |
| End of intervention | 204.8 (199.7; 209.9) | 195.1 (189.9; 200.4) | 0.010 | |
| Delta | −2.7 (−7.0; 1.6) | −7.9 (−12.1; −3.7) | 0.085 | |
| Adjusted change | −1.91 (−5.73; 1.91) | −9.07 (−12.87; −5.28) | 0.010 | |
| LDL-cholesterol—mg/dL | Baseline | 129.0 (124.6; 133.3) | 124.8 (120.6; 129.1) | 0.179 |
| End of intervention | 126.2 (121.6; 130.7) | 117.0 (112.5; 121.6) | 0.005 | |
| Delta | −2.81 (−6.91; 1.29) | −7.80 (−11.53; −4.07) | 0.076 | |
| Adjusted change | −1.97 (−5.45; 1.51) | −8.86 (−12.31; −5.40) | 0.006 | |
| HDL-cholesterol—mg/dL | Baseline | 58.3 (56.2; 60.5) | 57.6 (55.5; 59.8) | 0.648 |
| End of intervention | 58.8 (56.6; 61.0) | 58.1 (55.9; 60.3) | 0.667 | |
| Delta | 0.43 (−0.24; 1.10) | 0.46 (−0.25; 1.17) | 0.960 | |
| Adjusted change | 0.41 (−0.29; 1.11) | 0.40 (−0.29; 1.09) | 0.984 | |
| Triglycerides—mg/dL | Baseline | 99.9 (93.2; 106.6) | 101.8 (95.0; 108.6) | 0.693 |
| End of intervention | 99.3 (92.7; 106.6) | 101.8 (95.0; 108.6) | 0.974 | |
| Delta | −0.63 (−6.63; 5.37) | −2.39 (−7.19; 2.41) | 0.650 | |
| Adjusted change | 1.20 (−6.14; 3.75) | −2.40 (−7.31; 2.52) | 0.736 |
| Variables | Randomly Selected for c-miRNA Screening (n = 8) | Non-Selected (n = 37) | p-Value a |
|---|---|---|---|
| 1 y Change in LDL-cholesterol—mg/dL | −18.8 (−26.9; −10.6) | −19.2 (−25.7; −12.7) | 0.954 |
| Women—Number (%) | 5 (62.5) | 27 (73.0) | 0.553 |
| Baseline Age—y | 69.8 (66.4; 73.1) | 69.0 (67.9; 70.1) | 0.581 |
| 1 y Change in Weight—kg | 0.04 (−0.57; 0.65) | 0.33 (−0.41; 1.06) | 0.718 |
| 1 y Change in Body Mass Index—kg/m2 | 0.01 (−0.22; 0.23) | 0.09 (−0.19; 0.38) | 0.771 |
| 1 y Change in Waist Circumference—cm | 0.0 (−0.6; 0.6) | 0.1 (−0.9; 1.2) | 0.904 |
| 1 y Change in Physical Activity b | −56 (−300; 472) | 0 (−490; 0) | 0.456 |
| 1 y Change in Total cholesterol—mg/dL | −15.9 (−22.8; −8.9) | −20.6 (−28.0; −13.1) | 0.565 |
| 1 y Change in HDL-cholesterol—mg/dL | 1.3 (−0.7; 3.2) | −0.3 (−1.8; 1.3) | 0.375 |
| 1 y Change in Triacylglycerols—mg/dL | 8.1 (−8.2; 24.5) | −2.8 (−13.3; 7.7) | 0.355 |
| 1 y Change in Energy Intake—kcal/day | 251 (−90; 593) | 136 (13; 258) | 0.433 |
| 1 y Change in Protein Intake—% Energy | −1.3 (−3.1; 0.4) | −1.5 (−2.7; −0.4) | 0.875 |
| 1 y Change in Carbohydrate Intake—% Energy | −6.1 (−12.8; 0.6) | −2.4 (−5.0; 0.1) | 0.235 |
| 1 y Change in Total Fat Intake—% Energy | 8.1 (1.5; 14.7) | 5.4 (3.3; 7.5) | 0.304 |
| SFA—% Energy | 0.1 (−2.0; 2.3) | −0.4 (−1.1; 0.4) | 0.598 |
| MUFA—% Energy | −2.5 (−6.5; 1.6) | −1.8 (−3.4; −0.2) | 0.731 |
| PUFA—% Energy | 9.2 (6.6; 11.9) | 8.0 (7.0; 8.9) | 0.287 |
| ALA—% Energy | 2.00 (1.59; 2.41) | 1.66 (1.49; 1.83) | 0.095 |
| 1 y Change in Fiber Intake—g/day | 1.63 (−4.20; 7.46) | 1.99 (−0.28; 4.27) | 0.891 |
| 1 y Change in Alcohol Intake—g/day | 1.25 (−4.73; 7.22) | −1.29 (−4.02; 1.44) | 0.422 |
| 1 y Change in Cholesterol Intake—mg/day | −14.5 (−111.4; 82.4) | −22.8 (−59.3; 13.6) | 0.847 |
| miRNA (Relative Units) | Visit | Control Diet (n = 164) | Walnut Diet (n = 166) | p-Value a |
|---|---|---|---|---|
| hsa-miR-224-3p | Baseline | 0.384 (0.151; 0.617) | 0.593 (0.339; 0.846) | 0.128 |
| 1 year | 0.253 (−0.038; 0.545) | 0.722 (0.405; 1.040) | ||
| hsa-miR-551a | Baseline | 0.678 (0.502; 0.855) | 0.801 (0.632; 0.970) | 0.006 |
| 1 year | 0.496 (0.330; 0.662) | 0.956 (0.798; 1.115) | ||
| hsa-miR-181c-3p | Baseline | 0.016 (0.010; 0.021) | 0.024 (0.018; 0.029) | 0.272 |
| 1 year | 0.019 (0.014; 0.025) | 0.022 (0.017; 0.028) | ||
| hsa-miR-1271-5p | Baseline | 0.312 (0.167; 0.456) | 0.706 (0.560; 0.851) | 0.312 |
| 1 year | 0.432 (−0.121; 0.985) | 1.220 (0.665; 1.776) | ||
| hsa-miR-30d-3p | Baseline | 0.339 (0.215; 0.462) | 0.567 (0.447; 0.687) | 0.557 |
| 1 year | 0.408 (0.000; 0.816) | 0.810 (0.413; 1.208) | ||
| hsa-miR-493-3p | Baseline | 0.290 (−0.155; 0.735) | 1.154 (0.237; 0.685) | 0.366 |
| 1 year | 0.423 (−3.528; 4.375) | 3.948 (−0.218; 8.111) | ||
| hsa-miR-589-3p | Baseline | 0.284 (−0.089; 0.658) | 0.882 (0.526; 1.239) | 0.796 |
| 1 year | 0.239 (−0.073; 0.551) | 0.911 (0.614; 1.209) | ||
| hsa-miR-1227-3p | Baseline | 0.153 (−1.633; 1.939) | 1.616 (−0.139; 3.370) | 0.363 |
| 1 year | 0.186 (−0.021; 0.392) | 0.474 (0.271; 0.677) | ||
| hsa-miR-181c-5p | Baseline | 0.398 (0.222; 0.573) | 0.614 (0.447; 0.782) | 0.704 |
| 1 year | 0.408 (0.214; 0.602) | 0.678 (0.493; 0.863) | ||
| hsa-miR-130b-5p | Baseline | 0.476 (0.318; 0.634) | 0.708 (0.552; 0.865) | 0.854 |
| 1 year | 0.539 (0.341; 0.738) | 0.800 (0.603; 0.996) | ||
| hsa-miR-542-5p | Baseline | 0.497 (0.269; 0.724) | 0.427 (0.179; 0.674) | 0.187 |
| 1 year | 0.375 (0.177; 0.573) | 0.536 (0.321; 0.752) | ||
| hsa-miR-340-3p | Baseline | 0.399 (0.248; 0.550) | 0.624 (0.471; 0.778) | 0.501 |
| 1 year | 0.404 (0.206; 0.601) | 0.733 (0.531; 0.934) | ||
| hsa-miR-192-5p | Baseline | 1.509 (0.641; 2.376) | 2.875 (2.024; 3.727) | 0.708 |
| 1 year | 1.483 (−0.029; 2.995) | 3.298 (1.814; 4.782) | ||
| hsa-miR-330-3p | Baseline | 0.313 (0.112; 0.514) | 0.807 (0.085; 0.583) | 0.175 |
| 1 year | 0.334 (0.085; 0.853) | 1.006 (0.751; 1.260) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gil-Zamorano, J.; Cofán, M.; López de las Hazas, M.-C.; García-Blanco, T.; García-Ruiz, A.; Doménech, M.; Serra-Mir, M.; Roth, I.; Valls-Pedret, C.; Rajaram, S.; et al. Interplay of Walnut Consumption, Changes in Circulating miRNAs and Reduction in LDL-Cholesterol in Elders. Nutrients 2022, 14, 1473. https://doi.org/10.3390/nu14071473
Gil-Zamorano J, Cofán M, López de las Hazas M-C, García-Blanco T, García-Ruiz A, Doménech M, Serra-Mir M, Roth I, Valls-Pedret C, Rajaram S, et al. Interplay of Walnut Consumption, Changes in Circulating miRNAs and Reduction in LDL-Cholesterol in Elders. Nutrients. 2022; 14(7):1473. https://doi.org/10.3390/nu14071473
Chicago/Turabian StyleGil-Zamorano, Judit, Montserrat Cofán, María-Carmen López de las Hazas, Tatiana García-Blanco, Almudena García-Ruiz, Mónica Doménech, Mercè Serra-Mir, Irene Roth, Cinta Valls-Pedret, Sujatha Rajaram, and et al. 2022. "Interplay of Walnut Consumption, Changes in Circulating miRNAs and Reduction in LDL-Cholesterol in Elders" Nutrients 14, no. 7: 1473. https://doi.org/10.3390/nu14071473
APA StyleGil-Zamorano, J., Cofán, M., López de las Hazas, M.-C., García-Blanco, T., García-Ruiz, A., Doménech, M., Serra-Mir, M., Roth, I., Valls-Pedret, C., Rajaram, S., Sabaté, J., Ros, E., Dávalos, A., & Sala-Vila, A. (2022). Interplay of Walnut Consumption, Changes in Circulating miRNAs and Reduction in LDL-Cholesterol in Elders. Nutrients, 14(7), 1473. https://doi.org/10.3390/nu14071473











