Meta-Analysis of Randomized Clinical Trials Evaluating Effectiveness of a Multivitamin Supplementation against Oxidative Stress in Healthy Subjects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Samples
2.2. Study Collection and Variable Selection
2.3. Data Preprocessing and Transformation
2.4. Data Analyses
3. Results
3.1. Characteristics of Trials and Subjects
3.2. Variable Selection
3.3. Meta-Analysis Model Choice
3.4. Overall Effects of PMS on the Selected Variables
3.5. Subgroup Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ashoori, M.; Saedisomeolia, A. Riboflavin (vitamin B) and oxidative stress: A review. Br. J. Nutr. 2014, 111, 1985–1991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NIH State-of-the-Science Conference Statement on Multivitamin/Mineral Supplements and Chronic Disease Prevention. NIH Consens. State Sci. Statements 2006, 23, 1–30.
- Burnett-Hartman, A.N.; Fitzpatrick, A.L.; Gao, K.; Jackson, S.A.; Schreiner, P.J. Supplement use contributes to meeting recommended dietary intakes for calcium, magnesium, and vitamin C in four ethnicities of middle-aged and older Americans: The Multi-Ethnic Study of Atherosclerosis. J. Am. Diet. Assoc. 2009, 109, 422–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fairfield, K.M.; Fletcher, R.H. Vitamins for chronic disease prevention in adults: Scientific review. JAMA 2002, 287, 3116–3126. [Google Scholar] [CrossRef]
- Fletcher, R.H.; Fairfield, K.M. Vitamins for chronic disease prevention in adults: Clinical applications. JAMA 2002, 287, 3127–3129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, S.P.; White, K.K.; Park, S.-Y.; Sharma, S. Multivitamin-multimineral supplements’ effect on total nutrient intake. Am. J. Clin. Nutr. 2007, 85, 280S–284S. [Google Scholar] [PubMed]
- Mann, B.A.; Garry, P.J.; Hunt, W.C.; Owen, G.M.; Goodwin, J.S. Daily multivitamin supplementation and vitamin blood levels in the elderly: A randomized, double-blind, placebo-controlled trial. J. Am. Geriatr. Soc. 1987, 35, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Gaziano, J.M. Antioxidants in cardiovascular disease: Randomized trials. Nutr. Rev. 1996, 54, 175–177. [Google Scholar] [CrossRef]
- McKay, D.L.; Perrone, G.; Rasmussen, H.; Dallal, G.; Hartman, W.; Cao, G.; Blumberg, J.B. The effects of a multivitamin/mineral supplement on micronutrient status, antioxidant capacity and cytokine production in healthy older adults consuming a fortified diet. J. Am. Coll. Nutr. 2000, 19, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Nayak, B.K. Understanding the relevance of sample size calculation. Indian J. Ophthalmol. 2010, 58, 469–470. [Google Scholar] [CrossRef]
- Taylor, R.; Ivanov, O.; Page, A.; Brotherton, J.; Achat, H.; Close, G. Predictors of non-attendance from BreastScreen NSW in women who report current mammography screening. Aust. N. Z. J. Public Health 2003, 27, 581–587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hedges, L.V.; Olkin, I. Statistical Methods for Meta-Analysis; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Greco, T.; Zangrillo, A.; Biondi-Zoccai, G.; Landoni, G. Meta-analysis: Pitfalls and hints. Heart Lung Vessel. 2013, 5, 219–225. [Google Scholar] [PubMed]
- Isakov, V.A.; Bogdanova, A.A.; Bessonov, V.V.; Sentsova, T.B.; Tutelyan, V.A.; Lin, Y.; Kazlova, V.; Hong, J.; Velliquette, R.A. Effects of multivitamin, multimineral and phytonutrient supplementation on nutrient status and biomarkers of heart health risk in a russian population: A randomized, double blind, placebo controlled study. Nutrients 2018, 10, 120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, S.; Lim, Y.; Kim, Y.J.; Jung, E.S.; Suh, D.H.; Lee, C.H.; Park, E.; Hong, J.; Velliquette, R.A.; Kwon, O.; et al. Multivitamin and mineral supplementation containing phytonutrients scavenges reactive oxygen species in healthy subjects: A randomized, double-blinded, placebo-controlled trial. Nutrients 2019, 11, 101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.J.; Ahn, Y.H.; Lim, Y.; Kim, J.Y.; Kim, J.; Kwon, O. Daily nutritional dose supplementation with antioxidant nutrients and phytochemicals improves DNA and LDL stability: A double-blind, randomized, and placebo-controlled trial. Nutrients 2013, 5, 5218–5232. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.-Y. Statistical notes for clinical researchers: Assessing normal distribution (2) using skewness and kurtosis. Restor. Dent. Endod. 2013, 38, 52–54. [Google Scholar] [CrossRef]
- Soloman, S.R.; Sawilowsky, S.S. Impact of rank-based normalizing transformations on the accuracy of test scores. J. Mod. Appl. Stat. Methods 2009, 8, 9. [Google Scholar] [CrossRef]
- Cochran, W.G.J.B. The comparison of percentages in matched samples. Biometrika 1950, 37, 256–266. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.Y.; Yang, Y.K.; Oh, S.-Y.; Hong, Y.-C.; Lee, E.-K.; Kwon, O. Diet quality scores and oxidative stress in Korean adults. Eur. J. Clin. Nutr. 2011, 65, 1271–1278. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Gavaghan, D.; Egger, M. Publication and related bias in meta-analysis: Power of statistical tests and prevalence in the literature. J. Clin. Epidemiol. 2000, 53, 1119–1129. [Google Scholar] [CrossRef]
- Haidich, A.B. Meta-analysis in medical research. Hippokratia 2010, 14 (Suppl. 1), 29–37. [Google Scholar] [PubMed]
- Aronson, D.; Bartha, P.; Zinder, O.; Kerner, A.; Markiewicz, W.; Avizohar, O.; Brook, G.J.; Levy, Y. Obesity is the major determinant of elevated C-reactive protein in subjects with the metabolic syndrome. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 674–679. [Google Scholar] [CrossRef] [Green Version]
- Xu, W.; Huang, H.; Yu, L.; Cao, L. Meta-analysis of gene expression profiles indicates genes in spliceosome pathway are up-regulated in hepatocellular carcinoma (HCC). Med. Oncol. 2015, 32, 96. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, M.; Akhtar-Danesh, N.; McMillan, C.R.; Thabane, L. Is plasma vitamin C an appropriate biomarker of vitamin C intake? A systematic review and meta-analysis. Nutr. J. 2007, 6, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naidu, K.A. Vitamin C in human health and disease is still a mystery? An overview. Nutr. J. 2003, 2, 7. [Google Scholar] [CrossRef] [Green Version]
- Simmonds, M.; Stewart, G.; Stewart, L. A decade of individual participant data meta-analyses: A review of current practice. Contemp. Clin. Trials 2015, 45 Pt A, 76–83. [Google Scholar] [CrossRef]
- Nordmann, A.J.; Kasenda, B.; Briel, M. Meta-analyses: What they can and cannot do. Swiss Med. Wkly. 2012, 142, w13518. [Google Scholar] [CrossRef]
- Ferro, A.; Morais, S.; Rota, M.; Pelucchi, C.; Bertuccio, P.; Bonzi, R.; Galeone, C.; Zhang, Z.-F.; Matsuo, K.; Ito, H.; et al. Alcohol intake and gastric cancer: Meta-analyses of published data versus individual participant data pooled analyses (StoP Project). Cancer Epidemiol. 2018, 54, 125–132. [Google Scholar] [CrossRef] [Green Version]
- Bravata, D.M.; Olkin, I. Simple pooling versus combining in meta-analysis. Eval. Health Prof. 2001, 24, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Schober, P.; Vetter, T.R. Meta-Analysis in Clinical Research. Anesth. Analg. 2020, 131, 1090–1091. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.; Ahn, Y.H.; Yoo, J.K.; Park, K.S.; Kwon, O. Verifying Identities of Plant-Based Multivitamins Using Phytochemical Fingerprinting in Combination with Multiple Bioassays. Plant Foods Hum. Nutr. 2017, 72, 288–293. [Google Scholar] [CrossRef] [PubMed]
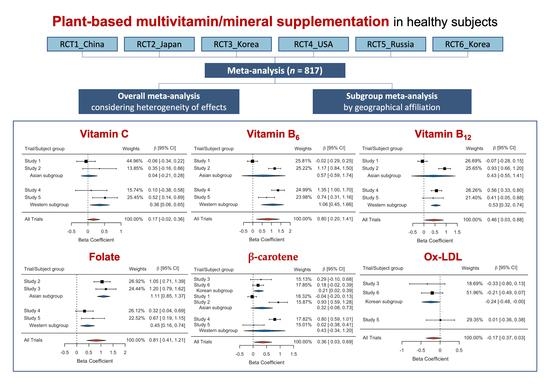
| Studies | Data Source | Country | Year | Period (wk) | RFS ‡ | PP § Subjects (ITT ¶ Subjects) | Age (SD #) | Sex (M/F) |
|---|---|---|---|---|---|---|---|---|
| Study 1 | Unpublished | China | 2004 | 8 | <12 (23) | 297 (334) | 33.2 (11.0) | 123/174 |
| Study 2 | Unpublished | Japan | 2007 | 8 | <12 (23) | 116 (126) | 43.0 (14.2) | 40/76 |
| Study 3 | Kim et al. [16] | Korea | 2013 | 8 | <37 (47) | 80 (90) | 43.2 (9.2) | 38/42 |
| Study 4 | Unpublished | USA | 2015 | 6 | <12 (23) | 120 (120) | 33.7 (12.1) | 57/63 |
| Study 5 | Isakov et al. [14] | Russia | 2018 | 8 | <12 (23) | 120 (120) | 49.2 (7.5) | 21/99 |
| Study 6 | Kang et al. [15] | Korea | 2019 | 8 | <37 (47) | 84 (96) | 39.9 (11.3) | 26/58 |
| Total | 817 (886) | 38.7 (12.6) | 305/512 | |||||
| Categories | Variables | Individual Studies | Total Count | |||||
|---|---|---|---|---|---|---|---|---|
| # 1 | # 2 | # 3 | # 4 | # 5 | # 6 | |||
| Chemical Scavenger | α-tocopherol, plasma | O | X | X | O | X | X | 2 |
| γ-tocopherol, plasma | O | X | X | O | X | X | 2 | |
| Vitamin C, plasma | O | O | X | O | O | X | 4 | |
| Vitamin B6, plasma | O | O | X | O | O | X | 4 | |
| Vitamin B12, plasma | O | O | X | O | O | X | 4 | |
| Folate, plasma | X | O | O | O | O | X | 4 | |
| Lutein, plasma | O | O | X | X | X | O | 3 | |
| Zeaxanthin, plasma | X | O | X | X | X | O | 2 | |
| α-carotene, plasma | O | X | X | X | X | O | 2 | |
| β-carotene, plasma | O | O | O | O | O | O | 6 | |
| Lycopene, plasma | O | X | X | X | X | O | 2 | |
| Oxidative Damage | Homocysteine, plasma | O | O | O | O | O | X | 5 |
| CRP †, plasma | O | X | O | O | X | O | 4 | |
| MDA ‡, plasma | X | X | O | X | X | O | 2 | |
| Ox-LDL §, plasma | X | X | O | X | O | O | 3 | |
| Comet assay, PBMC | O | X | O | X | X | O | 3 | |
| Comet assay, PBMC H2O2 challenge | X | X | O | X | X | O | 2 | |
| 8-OHdG #, urine | O | X | X | X | X | O | 2 | |
| QOL | Short Form 36 questionnaire | O | X | O | X | X | X | 2 |
| Categories | Variable (Optimal Transformation) | Meta-Analysis | Model | Heterogeneity | |
|---|---|---|---|---|---|
| Cochran’s Q (p-Value) | I2 (%) | ||||
| Chemical scavenger | Vitamin C (Square-root) | Overall | Fixed-effect | 6.37 (0.0950) | 52.00 |
| Asian sub | Fixed-effect | 1.90 (0.1682) | 47.35 | ||
| Western sub | Fixed-effect | 1.82 (0.1772) | 45.07 | ||
| Vitamin B6 (Square-root) | Overall | Random-effect | 47.97 (<0.0001) | 92.32 | |
| Asian sub | Random-effect | 29.81(<0.0001) | 96.65 | ||
| Western sub | Random-effect | 4.76 (0.0292) | 78.98 | ||
| Vitamin B12 (Inverse-normal) | Overall | Random-effect | 35.13 (<0.0001) | 89.58 | |
| Asian sub | Random-effect | 32.37 (<0.0001) | 96.91 | ||
| Western sub | Fixed-effect | 0.32 (0.5730) | <0.01 | ||
| Folate (Square-root) | Overall | Random-effect | 12.58 (0.0056) | 75.21 | |
| Asian sub | Fixed-effect | 0.33 (0.5648) | <0.01 | ||
| Western sub | Fixed-effect | 1.29 (0.2564) | 22.36 | ||
| β-carotene (Inverse-normal) | Overall | Random-effect | 54.70 (<0.0001) | 90.11 | |
| Korean sub | Fixed-effect | 0.22 (0.6357) | <0.01 | ||
| Asian sub | Random-effect | 25.38 (<0.0001) | 90.34 | ||
| Western sub | Random-effect | 11.82 (0.0006) | 91.54 | ||
| Oxidative Damage | Ox-LDL (Square-root) | Overall | Fixed-effect | 1.43 (0.4882) | <0.01 |
| Korean sub | Fixed-effect | 0.21 (0.6436) | <0.01 | ||
| Category | Variable | Relevant Trials | Meta-Analysis | |||
|---|---|---|---|---|---|---|
| Weights (%) | Beta Coefficient | Standard Error | p-Value | |||
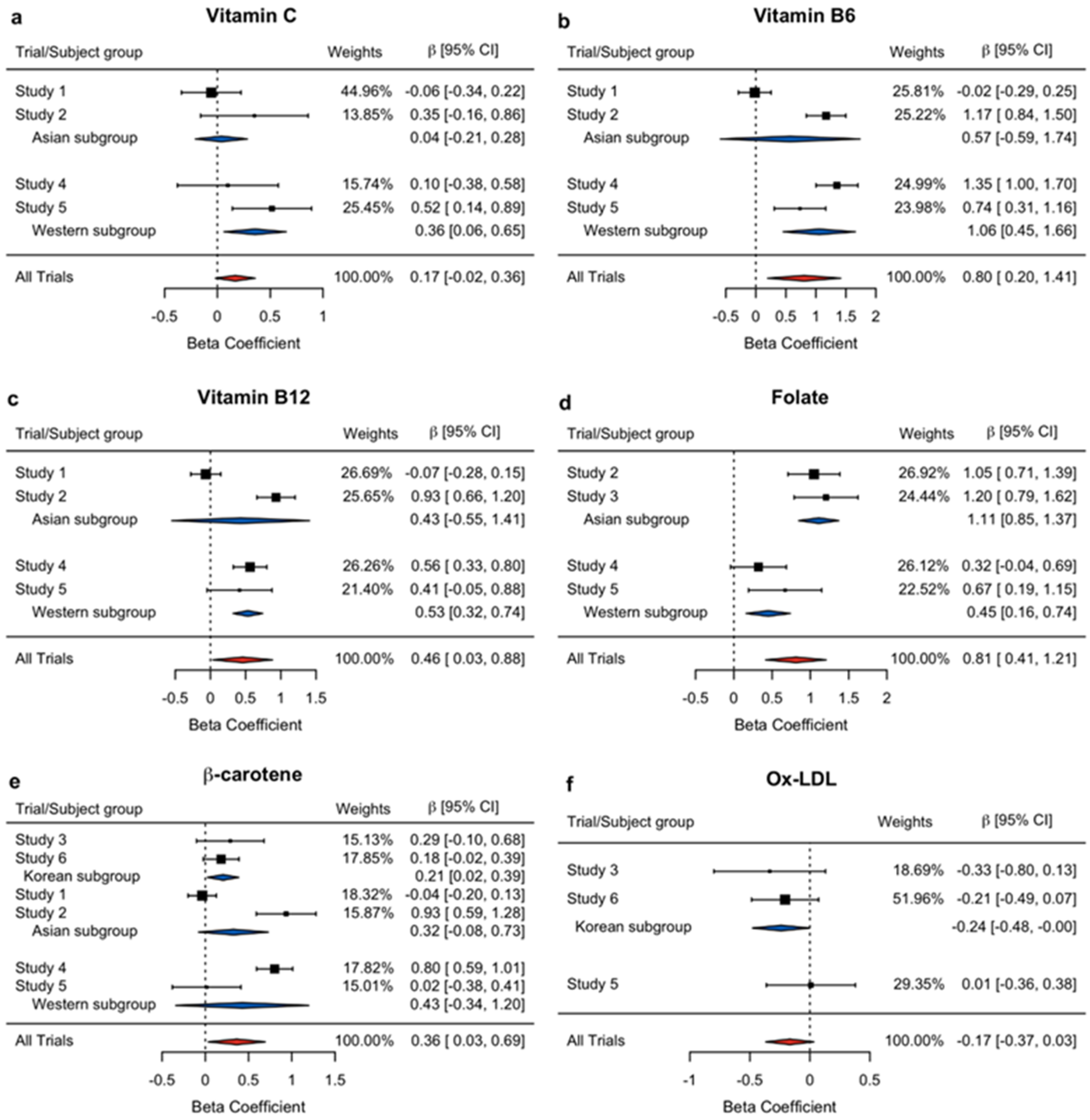
| Chemical Scavenger | Vitamin C | Study 1 | 44.96 | −0.0586 | 0.1440 | 0.6842 |
| Study 2 | 13.85 | 0.3504 | 0.2595 | 0.1784 | ||
| Study 4 | 15.74 | 0.0988 | 0.2434 | 0.6855 | ||
| Study 5 | 25.45 | 0.5166 | 0.1914 | 0.0080 | ||
| Overall | 100 | 0.1692 | 0.0966 | 0.0797 | ||
| Asian sub | - | 0.0377 | 0.1259 | 0.7647 | ||
| Western sub | - | 0.3570 | 0.1505 | 0.0177 | ||
| Vitamin B6 | Study 1 | 25.81 | −0.0182 | 0.1391 | 0.8959 | |
| Study 2 | 25.22 | 1.1703 | 0.1674 | <0.0001 | ||
| Study 4 | 24.99 | 1.3509 | 0.1778 | <0.0001 | ||
| Study 5 | 23.98 | 0.7359 | 0.2188 | 0.0010 | ||
| Overall | 100 | 0.8045 | 0.3110 | 0.0097 | ||
| Asian sub | - | 0.5724 | 0.5942 | 0.3354 | ||
| Western sub | - | 1.0566 | 0.3072 | 0.0006 | ||
| Vitamin B12 | Study 1 | 26.69 | −0.0662 | 0.1088 | 0.5432 | |
| Study 2 | 25.65 | 0.9322 | 0.1377 | <0.0001 | ||
| Study 4 | 26.26 | 0.5640 | 0.1213 | <0.0001 | ||
| Study 5 | 21.40 | 0.4148 | 0.2353 | 0.0805 | ||
| Overall | 100 | 0.4583 | 0.2170 | 0.0347 | ||
| Asian sub | - | 0.4294 | 0.4992 | 0.3896 | ||
| Western sub | - | 0.5327 | 0.1078 | <0.0001 | ||
| Folate | Study 2 | 26.92 | 1.0468 | 0.1728 | <0.0001 | |
| Study 3 | 24.44 | 1.2046 | 0.2128 | <0.0001 | ||
| Study 4 | 26.12 | 0.3212 | 0.1857 | 0.0869 | ||
| Study 5 | 22.52 | 0.6692 | 0.2440 | 0.0070 | ||
| Overall | 100 | 0.8108 | 0.2024 | 0.0001 | ||
| Asian sub | - | 1.1095 | 0.1341 | <0.0001 | ||
| Western sub | - | 0.4488 | 0.1478 | 0.0024 | ||
| β-carotene | Study 1 | 15.13 | −0.0354 | 0.0834 | 0.6718 | |
| Study 2 | 17.85 | 0.9339 | 0.1755 | <0.0001 | ||
| Study 3 | 18.32 | 0.2889 | 0.1985 | 0.1495 | ||
| Study 4 | 15.87 | 0.8014 | 0.1063 | <0.0001 | ||
| Study 5 | 17.82 | 0.0156 | 0.2023 | 0.9388 | ||
| Study 6 | 15.01 | 0.1826 | 0.1047 | 0.0851 | ||
| Overall | 100 | 0.3631 | 0.1681 | 0.0307 | ||
| Korean sub | - | 0.2057 | 0.0926 | 0.0263 | ||
| Asian sub | - | 0.3248 | 0.2063 | 0.1154 | ||
| Western sub | - | 0.4274 | 0.3924 | 0.2762 | ||
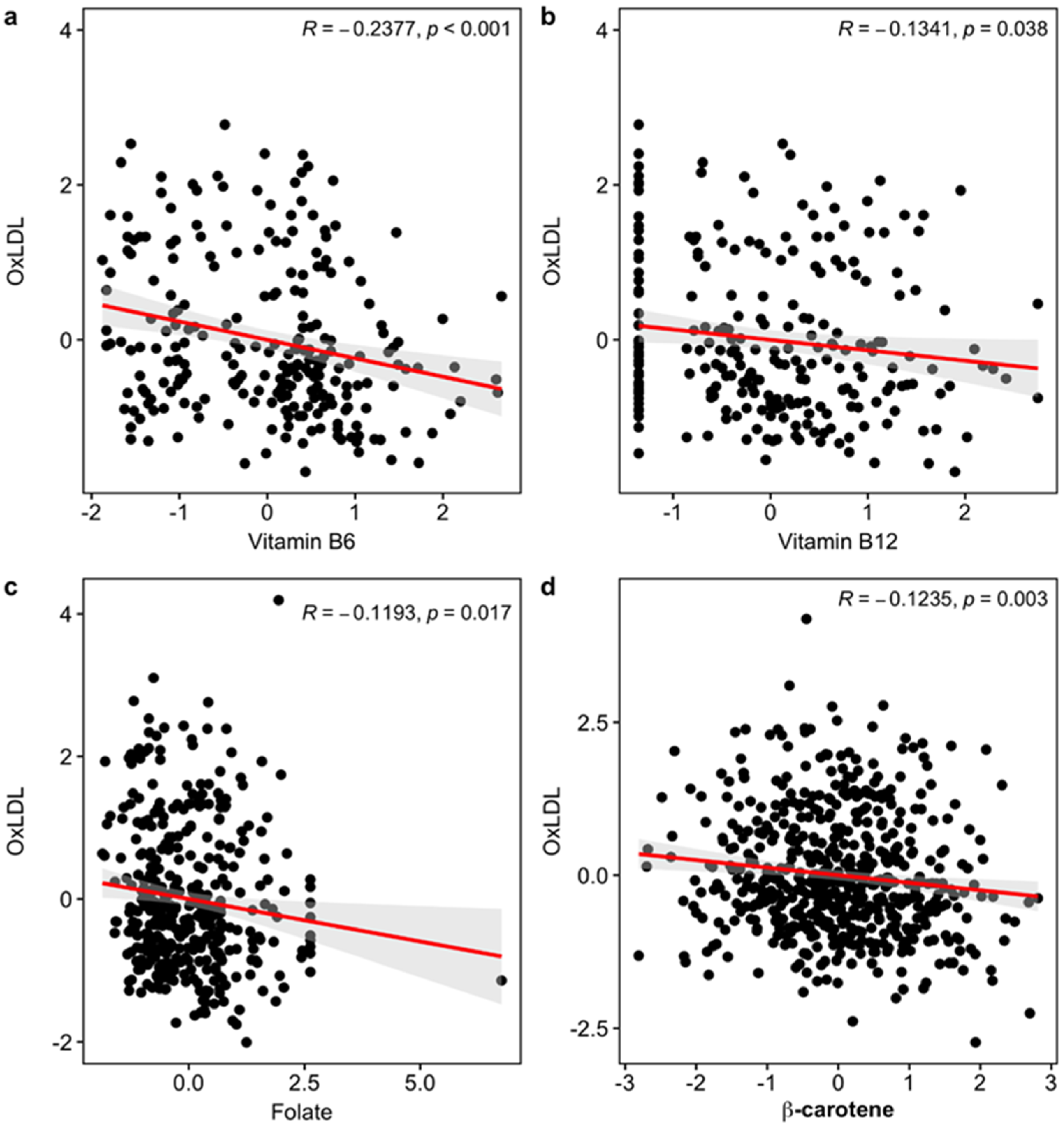
| Oxidative Damage | Ox-LDL (Square root) | Study 3 | 18.69 | −0.3342 | 0.2371 | 0.1627 |
| Study 5 | 51.96 | 0.0085 | 0.1892 | 0.9644 | ||
| Study 6 | 29.35 | −0.2063 | 0.1422 | 0.1507 | ||
| Overall | 100 | −0.1672 | 0.1025 | 0.1029 | ||
| Korean sub | - | −0.2401 | 0.1219 | 0.0489 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Huh, I.; Kang, S.; Nam, Y.-e.; Cho, Y.; Kamruzzaman, M.; Hong, J.; Kwon, O.; Park, T. Meta-Analysis of Randomized Clinical Trials Evaluating Effectiveness of a Multivitamin Supplementation against Oxidative Stress in Healthy Subjects. Nutrients 2022, 14, 1170. https://doi.org/10.3390/nu14061170
Lee S, Huh I, Kang S, Nam Y-e, Cho Y, Kamruzzaman M, Hong J, Kwon O, Park T. Meta-Analysis of Randomized Clinical Trials Evaluating Effectiveness of a Multivitamin Supplementation against Oxidative Stress in Healthy Subjects. Nutrients. 2022; 14(6):1170. https://doi.org/10.3390/nu14061170
Chicago/Turabian StyleLee, Seoyoung, Iksoo Huh, Seunghee Kang, Yea-eun Nam, Youngseo Cho, Md Kamruzzaman, Jina Hong, Oran Kwon, and Taesung Park. 2022. "Meta-Analysis of Randomized Clinical Trials Evaluating Effectiveness of a Multivitamin Supplementation against Oxidative Stress in Healthy Subjects" Nutrients 14, no. 6: 1170. https://doi.org/10.3390/nu14061170
APA StyleLee, S., Huh, I., Kang, S., Nam, Y.-e., Cho, Y., Kamruzzaman, M., Hong, J., Kwon, O., & Park, T. (2022). Meta-Analysis of Randomized Clinical Trials Evaluating Effectiveness of a Multivitamin Supplementation against Oxidative Stress in Healthy Subjects. Nutrients, 14(6), 1170. https://doi.org/10.3390/nu14061170