Providing the Best Parenteral Nutrition before and after Surgery for NEC: Macro and Micronutrients Intakes
Abstract
:1. Introduction
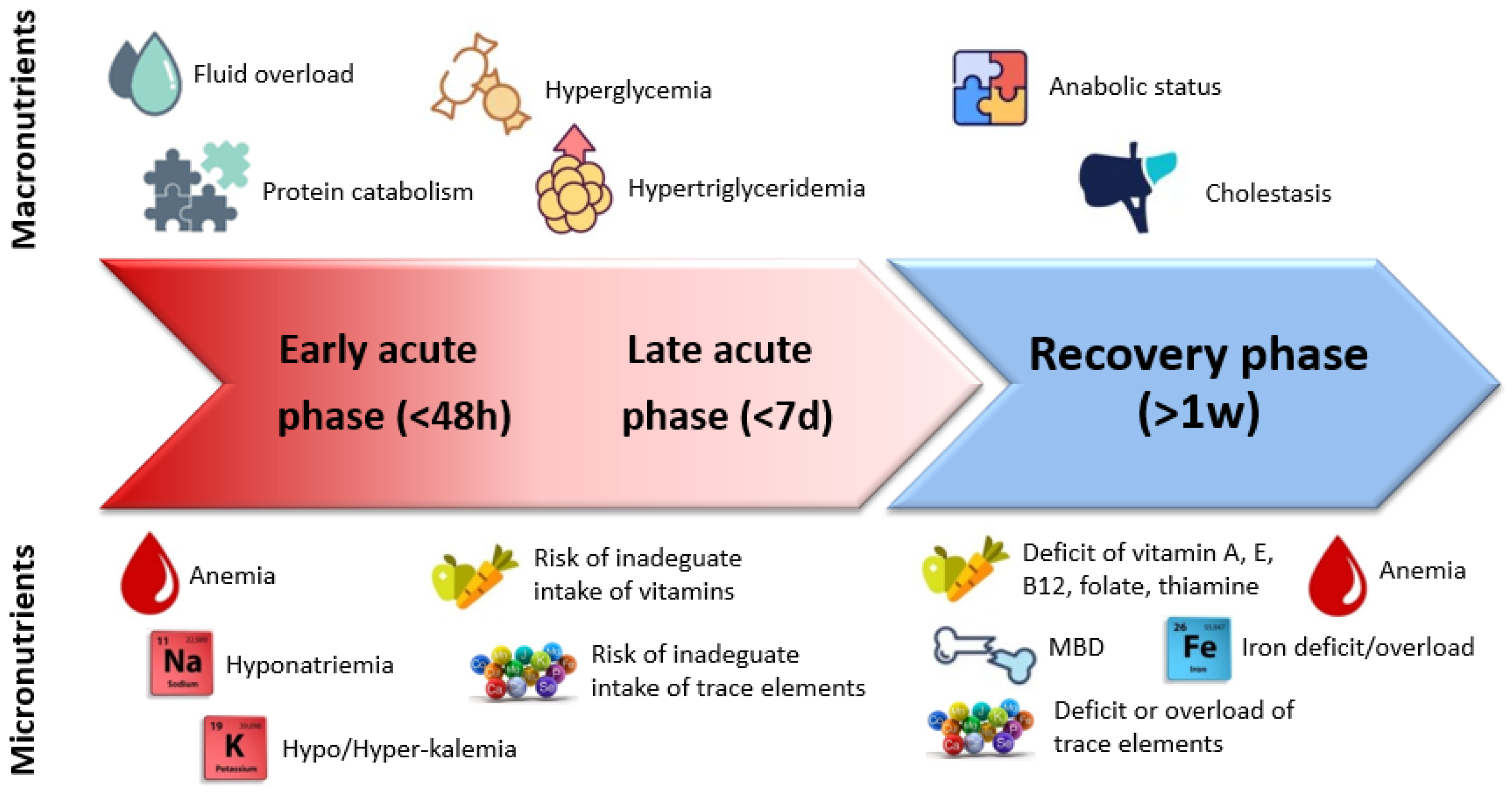
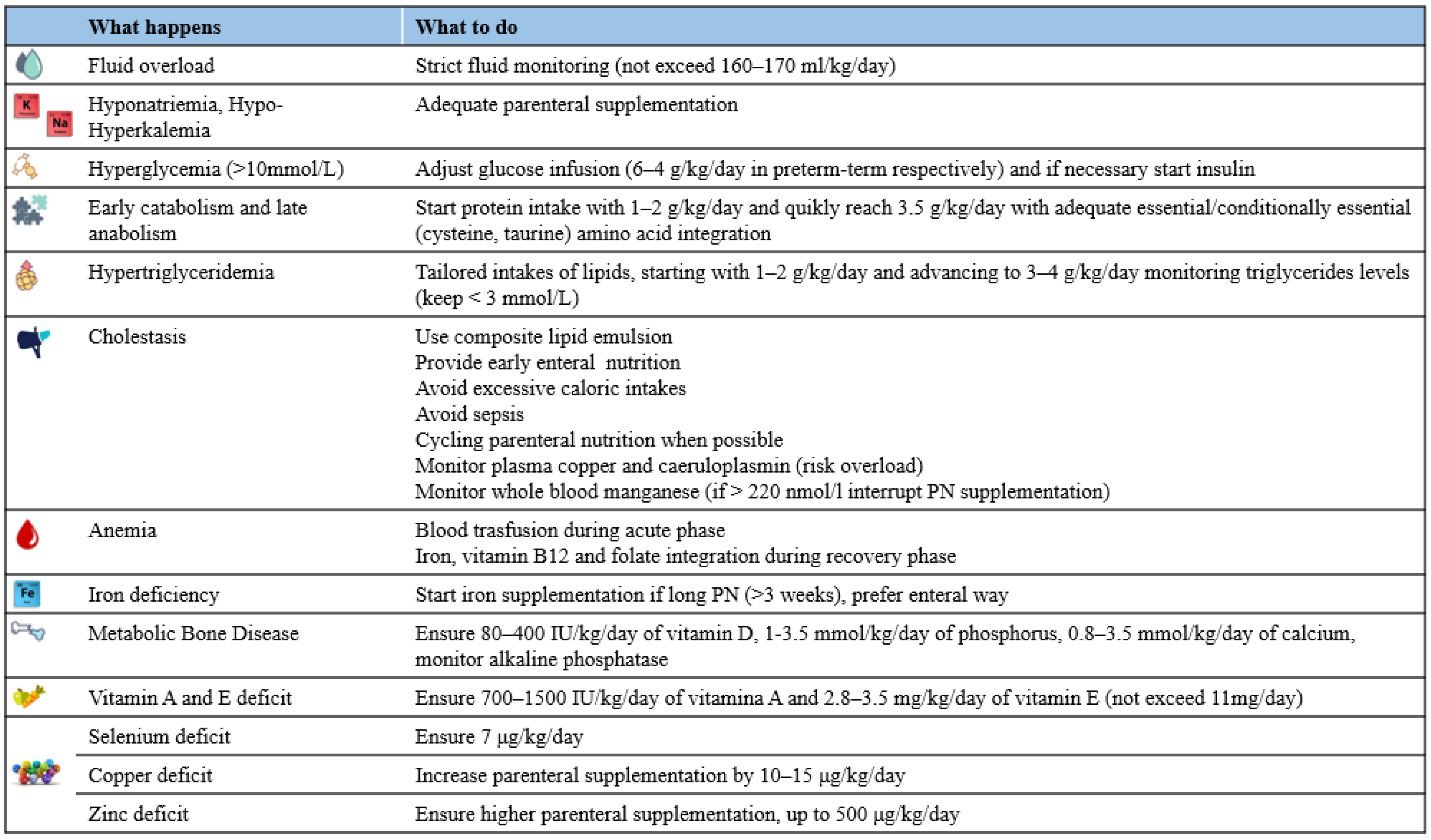
2. Before Surgery
3. At Surgery
3.1. Variability in Metabolic Demand
3.2. Hyperglycemia
3.3. A positive Immunological Response
4. After Surgery
4.1. Fluid Balance
4.2. Energy and Macronutrients Needs
4.2.1. Glucose
4.2.2. Amino Acids
- -
- Glutamine is the most abundant free amino acid in the body and it is the preferred respiratory fuel for rapidly proliferating cells. In case of metabolic stress, the endogenous production of glutamine could be insufficient, becoming a “conditionally essential amino acid”. Meta-analyses do not provide evidence that glutamine supplementation gives advantage for preterm infants [55]. Furthermore, studies focusing on surgical newborns also showed no benefit from glutamine supplementation during PN [56,57].
- -
- Taurine (synthesized from methionine and cysteine) deficiency may increase glyco-conjugates of bile acids resulting in cholestasis [58]. Studies showed that prolonged PN with a taurine-free parenteral solution resulted in reduced plasma taurine levels [59]. Although the cause of neonatal cholestasis and PNALD is probably multifactorial, there are few data indicating that adequate taurine may prevent cholestasis, in particular in patients with NEC [60]. The right dose of taurine supplementation is not yet known [58]: Spenser et al. demonstrated some benefits from taurine integration with a dose ranging from 6 to 21.6 mg/kg/day depending on the total protein delivery and formulation [60].
- -
4.2.3. Lipids
4.3. Micronutrients
4.3.1. Vitamins and Electrolytes
- -
- A total of 200–1000 IU/day (or 80–400 IU/kg/day) of vitamin D for preterm infants and 400 IU/day (or 40–150 IU/kg/day) for term infants up to 12 months of age;
- -
- A minimum of 0.8 mmol/kg/day up to 3.5 mmol/kg/day of calcium in preterm infants;
- -
- A minimum of 1 mmol/kg/day up to 3.5 mmol/kg/day of phosphorus in preterm infants.
4.3.2. Trace Elements
- -
- -
- Manganese should be supplied at a dose of no more than 1 μg/kg/day, however with blood levels regularly monitored as there is high risk of accumulation with neurotoxicity [130,131]. This risk is increased in neonates due to the evidence of significant quantities of Mn in neonatal PN as a contaminant [132,133]. Patients who develop PNALD are at a higher risk of Mn accumulation [79,134]: In case of PNALD whole blood manganese should be determined and if >220 nmol/L, parenteral supplementation should be discontinued [79,80].
- -
- Anemia often occurs during the acute phase due to several causes, especially bleeding and blood test monitoring. During this phase, no evidence suggests iron deficiency since multiple transfusions are needed and might give iron support. However, prolonged PN (particularly > 3 weeks) without iron supplementation may induce iron deficiency and anemia [22,101]. For this reason, it is important to plan iron supplementation that should preferentially be given enterally rather than with intermittent infusions due to adverse drug reactions, in particular anaphylaxis [80]. If patients develop SBS, the incidence of anemia is controversial [22,77,101,105,106] and it can be explained by blood draws, iron, or vitamin B12 deficiency, recurrent infection and intestinal anastomotic ulcers that can cause refractory anemia [77,81]. Therefore, the iron status, comprising serum ferritin and hemoglobin, should be regularly monitored in patients on long-term PN in order to prevent iron deficiency and iron overload [80].
- -
- Salivary, gastric, pancreatic, and intestinal juices contain a significant amount of zinc that normally is reabsorbed in the proximal small intestine, more specifically in the distal duodenum and proximal jejunum [135]. Newborns with elevated enteral fluid losses (diarrhea, steatorrhea, or stoma losses) are at a high risk for zinc deficiency [136]. Many case series document zinc deficiency in newborns with bowel damage, especially with jejunostomy or ileostomy [22,79,101,106,137]. Symptoms of zinc deficiency are well known, ranging from weight loss, failure to thrive, periorificial dermatitis, glossitis, and increased susceptibility to infections [138,139,140,141,142,143]. Intravenous zinc supplementation of 400 to 500 μg/kg/day in preterm infants is recommended, however no specific recommendations in infants with small intestinal stoma are available [80]. A monocentric study by D’Aniello et al. showed that a zinc deficit is prevented in newborns with a small bowel stoma if supplementation is 500 μg/kg/day parenterally [82]. Therefore, higher parenteral zinc supplementation should be planned early in infants after surgical NEC with jejunostomy or ileostomy.
- -
- Similarly, copper deficiency can be found in infants with jejunostomy or ileostomy or long-term PN [22,77,106,142]. Copper is primarily absorbed via the small intestine and patients who have increased copper losses through stool or ostomy require additional supplementation in PN solutions by 10–15 μg/kg [137]. However, there is no agreement on the best parenteral copper dose: The American Society for Clinical Nutrition and the ASPEN recommend 20 μg/kg/day [107,108], while the ESPGHAN guideline recommends 40 μg/kg/day in preterm infants and 20 μg/kg/day in term infants [80]. Additionally, there are no specific indications for surgical patients. Adler et al. found that 20 μg/kg/day of copper in PN of neonates with ostomies is insufficient to prevent Cu deficiency [83]. Moreover, the predominant pathway of copper excretion is through bile and it remains common practice to reduce or eliminate copper in the PN solutions of infants with PNALD because of the risk of hepatic toxicity.
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hall, N.J.; Eaton, S.; Pierro, A. Royal Australasia of Surgeons Guest Lecture. Necrotizing enterocolitis: Prevention, treatment, and outcome. J. Pediatr. Surg. 2013, 48, 2359–2367. [Google Scholar] [CrossRef]
- Lin, H.C.; Wu, S.F.; Underwood, M. Necrotizing enterocolitis. N. Engl. J. Med. 2011, 364, 1878–1879. [Google Scholar]
- Ellsbury, D.L.; Clark, R.H.; Ursprung, R.; Handler, D.L.; Dodd, E.D.; Spitzer, A.R. A Multifaceted Approach to Improving Outcomes in the NICU: The Pediatrix 100 000 Babies Campaign. Pediatrics 2016, 137, e20150389. [Google Scholar] [CrossRef] [Green Version]
- Stoll, B.J.; Hansen, N.I.; Bell, E.F.; Shankaran, S.; Laptook, A.R.; Walsh, M.C.; Hale, E.C.; Newman, N.S.; Schibler, K.; Carlo, W.A.; et al. Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 2010, 126, 443–456. [Google Scholar] [CrossRef] [Green Version]
- Battersby, C.; Santhalingam, T.; Costeloe, K.; Modi, N. Incidence of neonatal necrotising enterocolitis in high-income countries: A systematic review. Arch. Dis. Child.-Fetal Neonatal Ed. 2018, 103, F182–F189. [Google Scholar] [CrossRef] [PubMed]
- Jones, I.H.; Hall, N.J. Contemporary Outcomes for Infants with Necrotizing Enterocolitis—A Systematic Review. J. Pediatr. 2020, 220, 86–92.e3. [Google Scholar] [CrossRef] [PubMed]
- Papillon, S.; Castle, S.L.; Gayer, C.P.; Ford, H.R. Necrotizing Enterocolitis: Contemporary Management and Outcomes. Adv. Pediatr. 2013, 60, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Duro, D.; Kalish, L.A.; Johnston, P.; Jaksic, T.; McCarthy, M.; Martin, C.; Dunn, J.C.; Brandt, M.; Nobuhara, K.K.; Sylvester, K.G.; et al. Risk Factors for Intestinal Failure in Infants with Necrotizing Enterocolitis: A Glaser Pediatric Research Network Study. J. Pediatr. 2010, 157, 203–208.e1. [Google Scholar] [CrossRef] [Green Version]
- Bazacliu, C.; Neu, J. Necrotizing Enterocolitis: Long Term Complications. Curr. Pediatr. Rev. 2019, 15, 115–124. [Google Scholar] [CrossRef]
- Lin, G.C.; Robinson, D.T.; Olsen, S.; Reber, K.M.; Moallem, M.; DiGeronimo, R.; Mulroy, C.; Datta, A.; Murthy, K. Nutritional Practices and Growth in Premature Infants After Surgical Necrotizing Enterocolitis. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 111–116. [Google Scholar] [CrossRef]
- Ou, J.; Courtney, C.M.; Steinberger, A.E.; Tecos, M.E.; Warner, B.W. Nutrition in Necrotizing Enterocolitis and Following Intestinal Resection. Nutrients 2020, 12, 520. [Google Scholar] [CrossRef] [Green Version]
- Moltu, S.J.; Bronsky, J.; Embleton, N.; Gerasimidis, K.; Indrio, F.; Köglmeier, J.; de Koning, B.; Lapillonne, A.; Norsa, L.; Verduci, E.; et al. Nutritional Management of the Critically Ill Neonate: A Position Paper of the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2021, 73, 274–289. [Google Scholar] [CrossRef] [PubMed]
- Eaton, S.; De Coppi, P.; Pierro, A. Fluid, Electrolyte and Nutritional Support of the Surgical Neonate. In Rickham’s Neonatal Surgery; Losty, P., Flake, A., Rintala, R., Hutson, J., Lwai, N., Eds.; Springer: London, UK, 2018. [Google Scholar]
- Christian, V.J.; Polzin, E.; Welak, S. Nutrition Management of Necrotizing Enterocolitis. Nutr. Clin. Pract. 2018, 33, 476–482. [Google Scholar] [CrossRef]
- Pierro, A. Metabolism and nutritional support in the surgical neonate. J. Pediatr. Surg. 2002, 37, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Joosten, K.; Embleton, N.; Yan, W.; Senterre, T.; ESPGHAN/ESPEN/ESPR/CSPEN Working Group on Pediatric Parenteral Nutrition. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Energy. Clin. Nutr. 2018, 37, 2309–2314. [Google Scholar] [CrossRef] [PubMed]
- Jochum, F.; Moltu, S.J.; Senterre, T.; Nomayo, A.; Goulet, O.; Iacobelli, S.; The ESPGHAN/ESPEN/ESPR/CSPEN Working Group on Pediatric Parenteral Nutrition. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Fluid and electrolytes. Clin. Nutr. 2018, 37, 2344–2353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayer, O.; Kerner, J.A. Management of short bowel syndrome in postoperative very low birth weight infants. Semin. Fetal Neonatal Med. 2017, 22, 49–56. [Google Scholar] [CrossRef]
- Hall, N.J.; Peters, M.; Eaton, S.; Pierro, A. Hyperglycemia is associated with increased morbidity and mortality rates in neonates with necrotizing enterocolitis. J. Pediatr. Surg. 2004, 39, 898–901. [Google Scholar] [CrossRef] [PubMed]
- Lapillonne, A.; Mis, N.F.; Goulet, O.; van den Akker, C.; Wu, J.; Koletzko, B.; ESPGHAN/ESPEN/ESPR/CSPEN Working Group on Pediatric Parenteral Nutrition. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Lipids. Clin. Nutr. 2018, 37, 2324–2336. [Google Scholar] [CrossRef] [PubMed]
- Bronsky, J.; Campoy, C.; Braegger, C.; Cai, W.; Carnielli, V.; Darmaun, D.; Decsi, T.; Domellöf, M.; Embleton, N.; Fewtrell, M.; et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Vitamins. Clin. Nutr. 2018, 37, 2366–2378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Namjoshi, S.; Muradian, S.; Bechtold, H.; Reyen, L.; Venick, R.S.; Marcus, E.A.; Vargas, J.H.; Wozniak, L.J. Nutrition Deficiencies in Children With Intestinal Failure Receiving Chronic Parenteral Nutrition. J. Parenter. Enter. Nutr. 2017, 42, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, A.; Ahle, M.; Andersson, R.E.; Swolin-Eide, D.; Elfvin, A. Increased risk of rickets but not fractures during childhood and adolescence following necrotizing enterocolitis among children born preterm in Sweden. Pediatr. Res. 2019, 86, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Neu, J. Neonatal necrotizing enterocolitis: An update. Acta Paediatr. 2005, 94, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Neu, J.; Walker, W.A. Necrotizing enterocolitis. N. Engl. J. Med. 2011, 364, 255–264. [Google Scholar] [CrossRef] [Green Version]
- Duci, M.; Fascetti-Leon, F.; Erculiani, M.; Priante, E.; Cavicchiolo, M.E.; Verlato, G.; Gamba, P. Neonatal independent predictors of severe NEC. Pediatr. Surg. Int. 2018, 34, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Plank, L.D.; Connolly, A.B.; Hill, G.L. Sequential Changes in the Metabolic Response in Severely Septic Patients During the First 23 Days After the Onset of Peritonitis. Ann. Surg. 1998, 228, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Powis, M.R.; Smith, K.; Rennie, M.; Halliday, D.; Pierro, A. Characteristics of protein and energy metabolism in neonates with necrotizing enterocolitis—A pilot study. J. Pediatr. Surg. 1999, 34, 5–12. [Google Scholar] [CrossRef]
- Chwals, W.J.; Letton, R.W.; Jamie, A.; Charles, B. Stratification of injury severity using energy expenditure response in surgical infants. J. Pediatr. Surg. 1995, 30, 1161–1164. [Google Scholar] [CrossRef]
- Pierro, A.; Eaton, S. Metabolism and nutrition in the surgical neonate. Semin. Pediatr. Surg. 2008, 17, 276–284. [Google Scholar] [CrossRef]
- Powis, M.R.; Smith, K.; Rennie, M.; Halliday, D.; Pierro, A. Effect of major abdominal operations on energy and protein metabolism in infants and children. J. Pediatr. Surg. 1998, 33, 49–53. [Google Scholar] [CrossRef]
- Pereira-Da-Silva, L.; Barradas, S.; Moreira, A.C.; Alves, M.; Papoila, A.L.; Virella, D.; Cordeiro-Ferreira, G. Evolution of Resting Energy Expenditure, Respiratory Quotient, and Adiposity in Infants Recovering from Corrective Surgery of Major Congenital Gastrointestinal Tract Anomalies: A Cohort Study. Nutrients 2020, 12, 3093. [Google Scholar] [CrossRef]
- Feferbaum, R.; Leone, C.; Siqueira, A.A.; Valenti, V.E.; Gallo, P.R.; Reis, A.O.; Lopes, A.C.; Nascimento, V.G.; De Oliveira, A.G.; De Carvalho, T.D.; et al. Rest energy expenditure is decreased during the acute as compared to the recovery phase of sepsis in newborns. Nutr. Metab. 2010, 7, 63. [Google Scholar] [CrossRef]
- Bauer, J.; Hentschel, R.; Linderkamp, O. Effect of Sepsis Syndrome on Neonatal Oxygen Consumption and Energy Expenditure. Pediatrics 2002, 110, e69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, M.O.; Pierro, A.; Hammond, P.; Nunn, A.; Lloyd, D.A. Glucose utilization in the surgical newborn infant receiving total parenteral nutrition. J. Pediatr. Surg. 1993, 28, 1121–1125. [Google Scholar] [CrossRef]
- Anand, K.J.; Aynsley-Green, A. Measuring the severity of surgical stress in newborn infants. J. Pediatr. Surg. 1988, 23, 297–305. [Google Scholar] [CrossRef]
- Fivez, T.; Kerklaan, D.; Mesotten, D.; Verbruggen, S.; Wouters, P.J.; Vanhorebeek, I.; Debaveye, Y.; Vlasselaers, D.; Desmet, L.; Casaer, M.; et al. Early versus Late Parenteral Nutrition in Critically Ill Children. N. Engl. J. Med. 2016, 374, 1111–1122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mehta, N.M.; Jaksic, T. The critically ill child. In Nutrition in Pediatrics, 4th ed.; Duggan, C., Watkins, J., Walker, W.A., Eds.; BC Decker Inc.: Hamilton, ON, Canada, 2008; pp. 663–673. [Google Scholar]
- Heidegger, C.P.; Berger, M.M.; Graf, S.; Zingg, W.; Darmon, P.; Costanza, M.C.; Thibault, R.; Pichard, C. Optimization of energy provision with supplemental parenteral nutrition in critically ill patients: A randomized controlled clinical trial. Lancet 2013, 381, 385–393. [Google Scholar] [CrossRef]
- van Puffelen, E.; Hulst, J.M.; Vanhorebeek, I.; Dulfer, K.; Van Den Berghe, G.; Joosten, K.F.M.; Verbruggen, S.C.A.T. Effect of late versus early initiation of parenteral nutrition on weight deterioration during PICU stay: Secondary analysis of the PEPaNIC randomised controlled trial. Clin. Nutr. 2020, 39, 104–109. [Google Scholar] [CrossRef]
- Patel, J.J.; Martindale, R.G.; McClave, S.A. Controversies Surrounding Critical Care Nutrition: An Appraisal of Permissive Underfeeding, Protein, and Outcomes. J. Parenter. Enter. Nutr. 2018, 42, 508–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, S.J. Fluid therapy and outcome: Balance is best. J. Extra-Corpor. Technol. 2014, 46, 28–32. [Google Scholar]
- Xie, X.; Guo, S.; Deng, C.; Guo, C. Postoperative outcomes based on crystalloid administration in pediatric patients with necrotizing enterocolitis undergoing laparotomy. Medicine 2020, 99, e21987. [Google Scholar] [CrossRef] [PubMed]
- El Gkotmi, N.; Kosmeri, C.; Filippatos, T.D.; Elisaf, M.S. Use of intravenous fluids/solutions: A narrative review. Curr. Med. Res. Opin. 2017, 33, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Perel, P.; Roberts, I.; Ker, K. Colloids versus crystalloids for fluid resuscitation in critically ill patients. Cochrane Database Syst. Rev. 2013, 28, CD000567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moschino, L.; Duci, M.; Leon, F.F.; Bonadies, L.; Priante, E.; Baraldi, E.; Verlato, G. Optimizing Nutritional Strategies to Prevent Necrotizing Enterocolitis and Growth Failure after Bowel Resection. Nutrients 2021, 13, 340. [Google Scholar] [CrossRef] [PubMed]
- Dean, B.; England, R.; Morris, M.A.; Briars, G. Clinical Guideline for the Management of Stoma Output in Neonates and Infants; Norfolk and Norwich University Hospitals NHS Foundation Trust: Norwich, UK, 2020. [Google Scholar]
- Butte, N.F. Energy requirements of infants. Public Health Nutr. 2005, 8, 953–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srinivasan, V. Stress Hyperglycemia in Pediatric Critical Illness: The Intensive Care Unit Adds to the Stress! J. Diabetes Sci. Technol. 2012, 6, 37–47. [Google Scholar] [CrossRef] [Green Version]
- Farrag, H.M.; Cowett, R.M. Glucose homeostasis in the micropremie. Clin. Perinatol. 2000, 27, 1–22. [Google Scholar] [CrossRef]
- Mesotten, D.; Joosten, K.; van Kempen, A.; Verbruggen, S.; Braegger, C.; Bronsky, J.; Cai, W.; Campoy, C.; Carnielli, V.; Darmaun, D.; et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Carbohydrates. Clin. Nutr. 2018, 37, 2337–2343. [Google Scholar] [CrossRef] [Green Version]
- Hartman, C.; Shamir, R.; Simchowitz, V.; Lohner, S.; Cai, W.; Decsi, T.; Braegger, C.; Bronsky, J.; Campoy, C.; Carnielli, V.; et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Complications. Clin. Nutr. 2018, 37, 2418–2429. [Google Scholar] [CrossRef] [Green Version]
- Jakobsen, M.S.; Jørgensen, M.H.; Husby, S.; Andersen, L.; Jeppesen, P.B. Low-Fat, High-Carbohydrate Parenteral Nutrition (PN) May Potentially Reverse Liver Disease in Long-Term PN-Dependent Infants. Am. J. Dig. Dis. 2015, 60, 252–259. [Google Scholar] [CrossRef]
- Mosca, F. Nutritional management of surgical newborns and infants in the acute phase and during follow up. Med. Surg. Pediatr. 2018, 40, 197. [Google Scholar] [CrossRef] [PubMed]
- Moe-Byrne, T.; Brown, J.V.; McGuire, W. Glutamine supplementation to prevent morbidity and mortality in preterm infants. Cochrane Database Syst. Rev. 2016, 2016, CD001457. [Google Scholar] [CrossRef]
- Albers, M.J.I.J.; Steyerberg, E.W.; Hazebroek, F.W.J.; Mourik, M.; Borsboom, G.J.J.M.; Rietveld, T.; Huijmans, J.G.M.; Tibboel, D. Glutamine Supplementation of Parenteral Nutrition Does Not Improve Intestinal Permeability, Nitrogen Balance, or Outcome in Newborns and Infants Undergoing Digestive-Tract Surgery. Results from a Double-Blind, Randomized, Controlled Trial. Ann. Surg. 2005, 241, 599–606. [Google Scholar] [CrossRef]
- SIGN Trial Group; Ong, E.; Eaton, S.; Wade, A.M.; Horn, V.; Losty, P.D.; Curry, J.I.; Sugarman, I.D.; Klein, N.J.; Pierro, A. Randomized clinical trial of glutamine-supplemented versus standard parenteral nutrition in infants with surgical gastrointestinal disease. Br. J. Surg. 2012, 99, 929–938. [Google Scholar] [CrossRef]
- van Goudoever, J.B.; Carnielli, V.; Darmaun, D.; de Pipaon, M.S.; Braegger, C.; Bronsky, J.; Cai, W.; Campoy, C.; Decsi, T.; Domellöf, M.; et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Amino acids. Clin. Nutr. 2018, 37, 2315–2323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinton, N.E.; Laidlaw, S.A.; Ament, M.E.; Kopple, J.D. Taurine Concentrations in Plasma, Blood Cells, and Urine of Children Undergoing Long-Term Total Parenteral Nutrition. Pediatr. Res. 1987, 21, 399–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spencer, A.U.; Yu, S.; Tracy, T.F.; Aouthmany, M.M.; Llanos, A.; Brown, M.B.; Brown, M.; Shulman, R.J.; Hirschl, R.B.; DeRusso, P.A.; et al. Parenteral Nutrition–Associated Cholestasis in Neonates: Multivariate Analysis of the Potential Protective Effect of Taurine. J. Parenter. Enteral. Nutr. 2005, 29, 337–344. [Google Scholar] [CrossRef]
- Amin, H.J.; Zamora, S.A.; McMillan, D.D.; Fick, G.H.; Butzner, J.; Parsons, H.G.; Scott, R. Arginine supplementation prevents necrotizing enterocolitis in the premature infant. J. Pediatr. 2002, 140, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.; Lyttle, A.; Amin, H.; Shaireen, H.; Robertson, H.L.; Lodha, A.K. Arginine supplementation in prevention of necrotizing enterocolitis in the premature infant: An updated systematic review. BMC Pediatr. 2014, 14, 226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Küster, A.; Tea, I.; Ferchaud-Roucher, V.; Le Borgne, S.; Plouzennec, C.; Winer, N.; Rozé, J.-C.; Robins, R.; Darmaun, D. Cord Blood Glutathione Depletion in Preterm Infants: Correlation with Maternal Cysteine Depletion. PLoS ONE 2011, 6, e27626. [Google Scholar] [CrossRef] [PubMed]
- Stephens, B.E.; Walden, R.V.; Gargus, R.A.; Tucker, R.; McKinley, L.; Mance, M.; Nye, J.; Vohr, B.R. First-Week Protein and Energy Intakes Are Associated With 18-Month Developmental Outcomes in Extremely Low Birth Weight Infants. Pediatrics 2009, 123, 1337–1343. [Google Scholar] [CrossRef]
- Poindexter, B.B.; Langer, J.C.; Dusick, A.M.; Ehrenkranz, R.A. Early provision of parenteral amino acids in extremely low birth weight infants: Relation to growth and neurodevelopmental outcome. National Institute of Child Health and Human Development Neonatal Research Network. J. Pediatr. 2006, 148, 300–305.e1. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.H.; Chace, D.H.; Spitzer, A.R.; Pediatrix Amino Acid Study Group. Effects of Two Different Doses of Amino Acid Supplementation on Growth and Blood Amino Acid Levels in Premature Neonates Admitted to the Neonatal Intensive Care Unit: A Randomized, Controlled Trial. Pediatrics 2007, 120, 1286–1296. [Google Scholar] [CrossRef]
- Blanco, C.L.; Gong, A.K.; Schoolfield, J.; Green, B.K.; Daniels, W.; Liechty, E.A.; Ramamurthy, R. Impact of Early and High Amino Acid Supplementation on ELBW Infants at 2 Years. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Vlaardingerbroek, H.; Vermeulen, M.J.; Rook, D.; Van den Akker, C.H.P.; Dorst, K.; Wattimena, J.L.; Vermes, A.; Schierbeek, H.; van Goudoever, J.B. Safety and Efficacy of Early Parenteral Lipid and High-Dose Amino Acid Administration to Very Low Birth Weight Infants. J. Pediatr. 2013, 163, 638–644.e5. [Google Scholar] [CrossRef] [PubMed]
- Burattini, I.; Bellagamba, M.P.; Spagnoli, C.; D’Ascenzo, R.; Mazzoni, N.; Peretti, A.; Cogo, P.E.; Carnielli, V.P. Marche Neonatal Network. Targeting 2.5 versus 4 g/kg/day of Amino Acids for Extremely Low Birth Weight Infants: A Randomized Clinical Trial. J. Pediatr. 2013, 163, 1278–1282.e1. [Google Scholar] [CrossRef]
- Battaglia, F.C. Aminoacid oxidation and urea production rates in fetal like. Biol. Neonate 1995, 67, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Osborn, D.A.; Schindler, T.; Jones, L.J.; Sinn, J.K.H.; Bolisetty, S. Higher versus lower amino acid intake in parenteral nutrition for newborn infants. Cochrane Database Syst. Rev. 2018, 3, CD005949. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, G.; Burrin, D. Impact of Parenteral Lipid Emulsion Components on Cholestatic Liver Disease in Neonates. Nutrients 2021, 13, 508. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, V.; Malviya, M.N.; Soll, R. Lipid emulsions for parenterally fed preterm infants. Cochrane Database Syst. Rev. 2019, 6, 013163. [Google Scholar] [CrossRef] [PubMed]
- Teresa, C.; Antonella, D.; de Ville, D.G.J. New Nutritional and Therapeutical Strategies of NEC. Curr. Pediatr. Rev. 2019, 15, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Hill, S. Practical management of home parenteral nutrition in infancy. Early Hum. Dev. 2019, 138, 104876. [Google Scholar] [CrossRef] [PubMed]
- D’Antiga, L.; Goulet, O. Intestinal failure in children: The European view. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Zhang, T.; Yan, W.; Lu, L.; Tao, Y.; Cai, W.; Wang, Y. Micronutrient deficiencies in pediatric short bowel syndrome: A 10-year review from an intestinal rehabilitation center in China. Pediatr. Surg. Int. 2020, 36, 1481–1487. [Google Scholar] [CrossRef] [PubMed]
- Meneghelli, M.; Pasinato, A.; Salvadori, S.; Gaio, P.; Fantinato, M.; Vanzo, V.; De Terlizzi, F.; Verlato, G. Bone status in preterm infant: Influences of different nutritional regimens and possible markers of bone disease. J. Perinatol. 2016, 36, 394–400. [Google Scholar] [CrossRef]
- Burjonrappa, S.C.; Miller, M. Role of trace elements in parenteral nutrition support of the surgical neonate. J. Pediatr. Surg. 2012, 47, 760–771. [Google Scholar] [CrossRef]
- Domellof, M.; Szitanyi, P.; Simchowitz, W.; Franz, A.; Mimouni, F. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Iron and trace minerals. Clin. Nutr. 2018, 37, 2354–2359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fusaro, F.; Tambucci, R.; Romeo, E.; Bagolan, P.; Dall’Oglio, L.; Ceccarelli, S.; Francalanci, P.; Hermans, D.; Pietrobattista, A.; Diamanti, A.; et al. Anastomotic ulcers in short bowel syndrome: New suggestions from a multidisciplinary approach. J. Pediatr. Surg. 2018, 53, 483–488. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, R.; Terquem, E.L.; Poupon, J.; Assaf, Z.; Kermorvant-Duchemin, E.; Maggio, L.; Lapillonne, A. Parenteral Zinc Intake in Newborns With Jejunostomy or Ileostomy: Results of a Monocentric Cohort Study. J. Pediatr. Gastroenterol. Nutr. 2020, 70, 521–526. [Google Scholar] [CrossRef]
- Adler, A.; Worley, S.; Radhakrishnan, K. Increased Needs for Copper in Parenteral Nutrition for Children in the Neonatal Intensive Care Unit With an Ostomy. Nutr. Clin. Pract. 2020, 35, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Wang, H.; Amin, S.B. Copper Supplementation in Premature Infants with Parenteral Nutrition-Associated Cholestasis. Nutr. Clin. Pract. 2018, 33, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, B.; Goulet, O.; Hunt, J.; Krohn, K.; Shamir, R. Guidelines on Paediatric Parenteral Nutrition of the European Society of Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) and the European Society for Clinical Nutrition and Metabolism (ESPEN), supported by the European Society of Paediatric Research (ESPR). J. Pediatr. Gastroenterol. Nutr. 2005, 41, S1–S87. [Google Scholar] [PubMed] [Green Version]
- Satrom, K.; Gourley, G. Cholestasis in Preterm Infants. Clin. Perinatol. 2016, 43, 355–373. [Google Scholar] [CrossRef]
- Christensen, R.D.; Henry, E.; Wiedmeier, S.E.; Burnett, J.; Lambert, D.K. Identifying patients, on the first day of life, at high-risk of developing parenteral nutrition-associated liver disease. J. Perinatol. 2007, 27, 284–290. [Google Scholar] [CrossRef] [Green Version]
- Robinson, D.T.; Ehrenkranz, R.A. Parenteral nutrition-associated cholestasis in small for gestational age infants. J. Pediatr. 2008, 152, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Roth, B.; Fkelund, M.; Fan, B.G.; Hägerstrand, I.; Nilsson-Ehle, P. Lipid deposition in Kupffer cells after parenteral fat nutrition in rats: A biochemical and ultrastructural study. Intensive Care Med. 1996, 22, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Kurvinen, A.; Nissinen, M.J.; Andersson, S.; Korhonen, P.; Ruuska, T.; Taimisto, M.; Kalliomäki, M.; Lehtonen, L.; Sankilampi, U.; Arikoski, P.; et al. Parenteral plant sterols and intestinal failure-associated liver disease in neonates. J. Parenter. Enteral. Nutr. 2012, 54, 803–811. [Google Scholar] [CrossRef]
- Pupillo, D.; Correani, A.; Biagetti, C.; D’Ascenzo, R.; Simonato, M.; Verlato, G.; Cogo, P.; Rocchi, M.B.; Carnielli, V.P. Half-life of plasma phytosterols in very low birth weight preterm infants on routine parenteral nutrition with vegetable oil-based lipid emulsions. Clin. Nutr. 2018, 37, 262–269. [Google Scholar] [CrossRef]
- Correani, A.; Pignotti, A.; Marinelli, L.; Biagetti, C.; D’Ascenzo, R.; Vedovelli, L.; Verlato, G.; Cogo, P.; Rocchi, M.B.L.; Carnielli, V.P. Plasma Phytosterol Half-Life and Levels Are Increased in Very Low Birth Weight Preterm Infants with Parenteral Nutrition-Associated Cholestasis. Lipids 2018, 53, 717–725. [Google Scholar] [CrossRef]
- Savini, S.; D’Ascenzo, R.; Biagetti, C.; Serpentini, G.; Pompilio, A.; Bartoli, A.; Cogo, P.E.; Carnielli, V.P. The effect of 5 intravenous lipid emulsions on plasma phytosterols in preterm infants receiving parenteral nutrition: A randomized clinical trial. Am. J. Clin. Nutr. 2013, 98, 312–318. [Google Scholar] [CrossRef] [Green Version]
- Goulet, O.; Antébi, H.; Wolf, C.; Talbotec, C.; Alcindor, L.G.; Corriol, O.; Lamor, M.; Colomb-Jung, V. A new intravenous fat emulsion containing soybean oil, medium-chain triglycerides, olive oil, and fish oil: A single-center, double-blind randomized study on efficacy and safety in pediatric patients receiving home parenteral nutrition. J. Parenter. Enter. Nutr. 2010, 34, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, M.H.; Carter, B.A.; Hawthorne, K.; King, K.; Abrams, S. High Rates of Resolution of Cholestasis in Parenteral Nutrition-Associated Liver Disease with Fish Oil-Based Lipid Emulsion Monotherapy. J. Pediatr. 2013, 162, 793–798.e1. [Google Scholar] [CrossRef] [PubMed]
- Nehra, D.; Fallon, E.M.; Potemkin, A.K.; Voss, S.D.; Mitchell, P.D.; Valim, C.; Belfort, M.B.; Bellinger, D.C.; Duggan, C.; Gura, K.M.; et al. A comparison of 2 intravenous lipid emulsions: Interim analysis of a randomized controlled trial. J. Parenter. Enter. Nutr. 2014, 38, 693–701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.W.; Lee, N.M.; Kim, J.H.; Kim, K.S.; Kim, S.-N. Parenteral Fish Oil–Containing Lipid Emulsions May Reverse Parenteral Nutrition–Associated Cholestasis in Neonates: A Systematic Review and Meta-Analysis. J. Nutr. 2015, 145, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Vayalthrikkovil, S.; Rabi, Y.; Amin, H.; Spence, J.-M.; Robertson, H.L.; Lodha, A.; Bashir, R.A. Parenteral Fish-Oil Lipid Emulsions in the Prevention of Severe Retinopathy of Prematurity: A Systematic Review and Meta-Analysis. Am. J. Perinatol. 2017, 34, 705–715. [Google Scholar] [CrossRef]
- Kotiya, P.; Zhao, X.; Cheng, P.; Zhu, X.; Xiao, Z.; Wang, J. Fish oil- and soy oil-based lipid emulsions in neonatal parenteral nutrition: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2016, 70, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Skouroliakou, M.; Konstantinou, D.; Agakidis, C.; Kaliora, A.; Kalogeropoulos, N.; Massara, P.; Antoniadi, M.; Panagiotakos, D.; Karagiozoglou-Lampoudi, T. Parenteral MCT/omega-3 polyunsaturated fatty acidenriched intravenous fat emulsion is associated with cytokine and fatty acid profiles consistent with attenuated inflammatory response in preterm neonates: A randomized, double-blind clinical trial. Nutr. Clin. Pract. 2016, 31, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Ubesie, A.C.; Kocoshis, S.A.; Mezoff, A.G.; Henderson, C.J.; Helmrath, M.A.; Cole, C.R. Multiple Micronutrient Deficiencies among Patients with Intestinal Failure during and after Transition to Enteral Nutrition. J. Pediatr. 2013, 163, 1692–1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kocoshis, S.A. Medical management of pediatric intestinal failure. Sem. Pediatr. Surg. 2010, 19, 20–26. [Google Scholar] [CrossRef]
- Youssef, N.N.; Mezoff, A.G.; Carter, B.A.; Cole, C.R. Medical Update and Potential Advances in the Treatment of Pediatric Intestinal Failure. Curr. Gastroenterol. Rep. 2012, 14, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Vettleson, K.L.; Larson, H.J.; Mohamed, M.W.; Grindeland, C.J.; Williamson, J.D.M. Micronutrient Monitoring and Management in Parenteral Nutrition–Dependent Premature Neonates: A Case Series. J. Pediatr. Pharmacol. Ther. 2021, 26, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Tang, Q.; Feng, Y.; Huang, J.; Tao, Y.; Wang, Y.; Cai, W.; Shi, C. Nutrition assessment in children with short bowel syndrome weaned off parenteral nutrition: A long-term follow-up study. J. Pediatr. Surg. 2007, 42, 1372–1376. [Google Scholar] [CrossRef]
- Yang, C.-F.J.; Duro, D.; Zurakowski, D.; Lee, M.; Jaksic, T.; Duggan, C. High Prevalence of Multiple Micronutrient Deficiencies in Children with Intestinal Failure: A Longitudinal Study. J. Pediatr. 2011, 159, 39–44.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greene, H.L.; Hambidge, K.M.; Schanler, R.; Tsang, R.C. Guidelines for the use of vitamins, trace elements, calcium, magnesium, and phosphorus in infants and children receiving total parenteral nutrition: Report of the Subcommittee on Pediatric Parenteral Nutrient Requirements from the Committee on Clinical Practice Issues of the American Society for Clinical Nutrition. Am. J. Clin. Nutr. 1988, 48, 1324–1342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corkings, M.R. The ASPEN Pediatric Nutrition Support Core Curriculum, 2nd ed.; ASPEN: Silver Spring, MD, USA, 2015. [Google Scholar]
- Wozniak, L.J.; Bechtold, H.M.; Reyen, L.E.; Hall, T.R.; Vargas, J.H. Vitamin D Deficiency in Children With Intestinal Failure Receiving Home Parenteral Nutrition. J. Parenter. Enter. Nutr. 2015, 39, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Diamanti, A.; Bizzarri, C.; Basso, M.S.; Gambarara, M.; Cappa, M.; Daniele, A.; Noto, C.; Castro, M. How does long-term parenteral nutrition impact the bone mineral status of children with intestinal failure? J. Bone Miner. Metab. 2010, 28, 351–358. [Google Scholar] [CrossRef]
- Khan, F.A.; Fisher, J.G.; Bairdain, S.; Sparks, E.A.; Zurakowski, D.; Modi, B.P.; Duggan, C.; Jaksic, T. Metabolic bone disease in pediatric intestinal failure patients: Prevalence and risk factors. J. Pediatr. Surg. 2015, 50, 136–139. [Google Scholar] [CrossRef] [Green Version]
- Walters, J.R. The role of the intestine in bone homeostasis. Eur. J. Gastroenterol. Hepatol. 2003, 15, 845–849. [Google Scholar] [CrossRef] [PubMed]
- Cakir, M.; Mungan, I.; Karahan, C.; Can, G.; Okten, A. Necrotizing enterocolitis increases the bone resorption in premature infants. Early Hum. Dev. 2006, 82, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Dabezies, E.J.; Warren, P.D. Fractures in Very Low Birth Weight Infants With Rickets. Clin. Orthop. Relat. Res. 1997, 335, 233–239. [Google Scholar] [CrossRef]
- Betto, M.; Gaio, P.; Ferrini, I.; De Terlizzi, F.; Zambolin, M.; Scattolin, S.; Pasinato, A.; Verlato, G. Assessment of bone health in preterm infants through quantitative ultrasound and biochemical markers. J. Matern. Neonatal Med. 2014, 27, 1343–1347. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-L.; Lee, C.-L.; Tseng, H.-I.; Yang, S.-N.; Yang, R.-C.; Jao, H.-C. Assisted exercise improves bone strength in very low birthweight infants by bone quantitative ultrasound. J. Paediatr. Child Health 2010, 46, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Erdem, E.; Tosun, Ö.; Bayat, M.; Korkmaz, Z.; Halis, H.; Guneş, T. Daily physical activity in low-risk extremely low birth weight preterm infants: Positive impact on bone mineral density and anthropometric measurements. J. Bone Miner. Metab. 2015, 33, 329–334. [Google Scholar] [CrossRef]
- Eelloo, J.A.; Roberts, S.A.; Emmerson, A.J.B.; Ward, K.A.; Adams, J.E.; Mughal, M.Z. Bone status of children aged 5–8 years, treated with dexamethasone for chronic lung disease of prematurity. Arch. Dis. Child.-Fetal Neonatal Ed. 2008, 93, F222–F224. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, C.; McDevitt, H.; Ahmed, S.; White, M. Longitudinal changes in bone health as assessed by the speed of sound in very low birth weight preterm infants. J. Pediatr. 2006, 148, 450–455. [Google Scholar] [CrossRef]
- McDevitt, H.; Ahmed, S. Quantitative Ultrasound Assessment of Bone Health in the Neonate. Neonatology 2006, 91, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.A.; Committee on Nutrition. Calcium and vitamin D requirements of enterally fed preterm infants. Pediatrics 2013, 131, e1676–e1683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaio, P.; Verlato, G.; Daverio, M.; Cavicchiolo, M.E.; Nardo, D.; Pasinato, A.; De Terlizzi, F.; Baraldi, E. Incidence of metabolic bone disease in preterm infants of birth weight < 1250 g and in those suffering from bronchopulmonary dysplasia. Clin. Nutr. ESPEN 2018, 23, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, M.S.; Cole, T.J.; Bishop, N.J.; Lucas, A. Neonatal factors predicting childhood height in preterm infants: Evidence for a persisting effect of early metabolic bone disease? J. Pediatr. 2000, 137, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Cormack, B.E.; Jiang, Y.; Harding, J.E.; Crowther, C.A.; Bloomfield, F.M. Neonatal Refeeding Syndrome and Clinical Outcome in Extremely Low-Birth-Weight Babies: Secondary Cohort Analysis from the ProVIDe Trial. J. Parenter. Enteral. Nutr. 2021, 45, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.B.; Lee, Y.; Oh, A.; Yoo, H.-W.; Choi, J.-H. Efficacy and safety of parenteral vitamin D therapy in infants and children with vitamin D deficiency caused by intestinal malabsorption. Ann. Pediatr. Endocrinol. Metab. 2020, 25, 112–117. [Google Scholar] [CrossRef]
- Collins, J.E.; Rolles, C.J.; Sutton, H.; Ackery, D. Vitamin B12 absorption after necrotizing enterocolitis. Arch. Dis. Child. 1984, 59, 731–734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davies, B.; Abel, G.; Puntis, J.; Arthur, R.; Truscott, J.; Oldroyd, B.; Stringer, M. Limited ileal resection in infancy: The long-term consequences. J. Pediatr. Surg. 1999, 34, 583–587. [Google Scholar] [CrossRef]
- Roilides, I.; Vasilaki, K.; Xinias, I.; Iosifidis, E.; Antachopoulos, C.; Roilides, E. Thiamine Deficiency in a Child with Short Bowel Syndrome and Review. Pediatr. Gastroenterol. Hepatol. Nutr. 2019, 22, 493–499. [Google Scholar] [CrossRef]
- Rannem, T.; Hylander, E.; Ladefoged, K.; Staun, M.; Tjellesen, L.; Jarnum, S. The metabolism of [75Se]selenite in patients with short bowel syndrome. J. Parenter. Enter. Nutr. 1996, 20, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Hardya, I.J.; Gillandersb, L.; Hardya, G. Is manganese an essential supplement for parenteral nutrition? Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 289–296. [Google Scholar] [CrossRef]
- Aschner, J.L.; Anderson, A.W.; Slaughter, J.; Aschner, M.; Steele, S.D.; Beller, A.B.; Mouvery, A.; Furlong, H.M.; Maitre, N. Neuroimaging identifies increased manganese deposition in infants receiving parenteral nutrition. Am. J. Clin. Nutr. 2015, 102, 1482–1489. [Google Scholar] [CrossRef] [Green Version]
- Dickerson, R.N. Manganese intoxication and parenteral nutrition. Nutrition 2001, 17, 689–693. [Google Scholar] [CrossRef]
- Sauberan, B.J.; Katheria, A.C. Unintentional Manganese Delivery in Neonatal Parenteral Nutrition. J. Pediatr. Gastroenterol. Nutr. 2020, 71, 132–135. [Google Scholar] [CrossRef] [PubMed]
- McMillan, N.B.; Mulroy, C.; Mackay, M.W.; McDonald, C.M.; Jackson, W.D. Correlation of Cholestasis with Serum Copper and Whole-Blood Manganese Levels in Pediatric Patients. Nutr. Clin. Pract. 2008, 23, 161–165. [Google Scholar] [CrossRef]
- Finch, C.W. Review of Trace Mineral Requirements for Preterm Infants: What Are the Current Recommendations for Clinical Practice? Nutr. Clin. Pract. 2015, 30, 44–58. [Google Scholar] [CrossRef]
- Wulf, K.; Wilhelm, A.; Spielmann, M.; Wirth, S.; Jenke, A.C. Frequency of Symptomatic Zinc Deficiency in Very Low Birth Weight Infants. Klin. Pädiatrie 2012, 225, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Balay, K.S.; Hawthorne, K.M.; Hicks, P.D.; Chen, Z.; Griffin, I.J.; Abrams, S.A. Low Zinc Status and Absorption Exist in Infants with Jejunostomies or Ileostomies Which Persists after Intestinal Repair. Nutrients 2012, 4, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Srouji, M.N.; Balistreri, W.F.; Caleb, M.H.; South, M.A.; Starr, S. Zinc deficiency during parental nutrition: Skin manifestations and immune incompetence in a premature infant. J. Pediatr. Surg. 1978, 13, 570–575. [Google Scholar] [CrossRef]
- Arlette, J.P.; Johnston, M.M. Zinc deficiency dermatosis in premature infants receiving prolonged parenteral alimentation. J. Am. Acad. Dermatol. 1981, 5, 37–42. [Google Scholar] [CrossRef]
- Palma, P.A.; Conley, S.B.; Crandell, S.S.; Denson, S.E. Zinc Deficiency Following Surgery in Zinc-Supplemented Infants. Pediatrics 1982, 69, 801–803. [Google Scholar] [CrossRef] [PubMed]
- Harper, J.I.; Thompson, D.; Kovar, I.Z.; Copeman, P.W.; Barltrop, D. Zinc deficiency in a preterm neonate with necrotizing enterocolitis. J. R. Soc. Med. 1984, 77, 40–41. [Google Scholar] [PubMed]
- Suita, S.; Ikeda, K.; Hayashida, Y.; Naito, K.; Handa, N.; Doki, T. Zinc and copper requirements during parenteral nutrition in the newborn. J. Pediatr. Surg. 1984, 19, 126–130. [Google Scholar] [CrossRef]
- Barbarot, S.; Chantier, E.; Kuster, A.; Hello, M.; Roze, J.; Blouin, E.; Stalder, J.-F. Symptomatic Acquired Zinc Deficiency in At-Risk Premature Infants: High Dose Preventive Supplementation Is Necessary. Pediatr. Dermatol. 2010, 27, 380–383. [Google Scholar] [CrossRef]
- Marquardt, M.L.; Done, S.L.; Sandrock, M.; Berdon, W.E.; Feldman, K.W. Copper Deficiency Presenting as Metabolic Bone Disease in Extremely Low Birth Weight, Short-Gut Infants. Pediatrics 2012, 130, e695–e698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blackmer, A.B.; Bailey, E. Management of Copper Deficiency in Cholestatic Infants: Review of the Literature and a Case Series. Nutr. Clin. Pract. 2013, 28, 75–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sievers, E.; Schleyerbach, U.; Oldigs, H.D.; Arpe, T.; Schaub, J. Molybdenum in infancy: Balance studies in patients with intestinal stomata. Acta Pædiatrica 2001, 90, 1121–1126. [Google Scholar] [CrossRef]
| Studies | Early Acute | Late Acute | Recovery |
|---|---|---|---|
| Moltu et al., 2021 [12] | 40–55 | 60–80 | 90–120 |
| Joosten et al., 2018 [16] | 45–55 | 60–65 | 90–120 |
| Feferbaum et al., 2010 [33] | 49.4 +/− 13.1 | / | 68.3 +/− 10.9 |
| Bauer et al., 2002 [34] | 58 +/− 3 | 55 +/− 2 | 50 +/− 2 |
| Jones et al., 1993 [35] | 40.1–60.5 for 4–7 days post-surgery |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guiducci, S.; Duci, M.; Moschino, L.; Meneghelli, M.; Fascetti Leon, F.; Bonadies, L.; Cavicchiolo, M.E.; Verlato, G. Providing the Best Parenteral Nutrition before and after Surgery for NEC: Macro and Micronutrients Intakes. Nutrients 2022, 14, 919. https://doi.org/10.3390/nu14050919
Guiducci S, Duci M, Moschino L, Meneghelli M, Fascetti Leon F, Bonadies L, Cavicchiolo ME, Verlato G. Providing the Best Parenteral Nutrition before and after Surgery for NEC: Macro and Micronutrients Intakes. Nutrients. 2022; 14(5):919. https://doi.org/10.3390/nu14050919
Chicago/Turabian StyleGuiducci, Silvia, Miriam Duci, Laura Moschino, Marta Meneghelli, Francesco Fascetti Leon, Luca Bonadies, Maria Elena Cavicchiolo, and Giovanna Verlato. 2022. "Providing the Best Parenteral Nutrition before and after Surgery for NEC: Macro and Micronutrients Intakes" Nutrients 14, no. 5: 919. https://doi.org/10.3390/nu14050919
APA StyleGuiducci, S., Duci, M., Moschino, L., Meneghelli, M., Fascetti Leon, F., Bonadies, L., Cavicchiolo, M. E., & Verlato, G. (2022). Providing the Best Parenteral Nutrition before and after Surgery for NEC: Macro and Micronutrients Intakes. Nutrients, 14(5), 919. https://doi.org/10.3390/nu14050919







