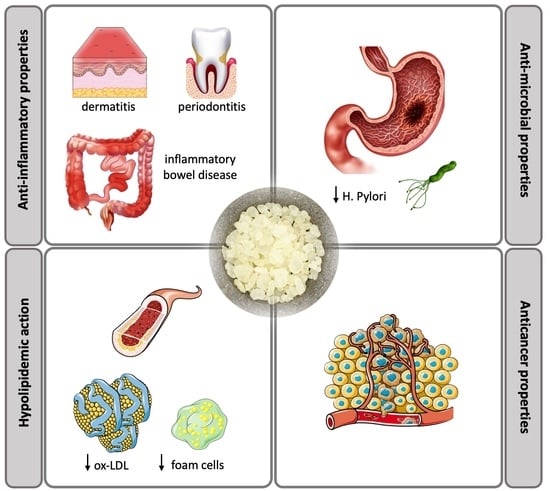
Overview of Chios Mastic Gum (Pistacia lentiscus) Effects on Human Health
Abstract
:1. Introduction
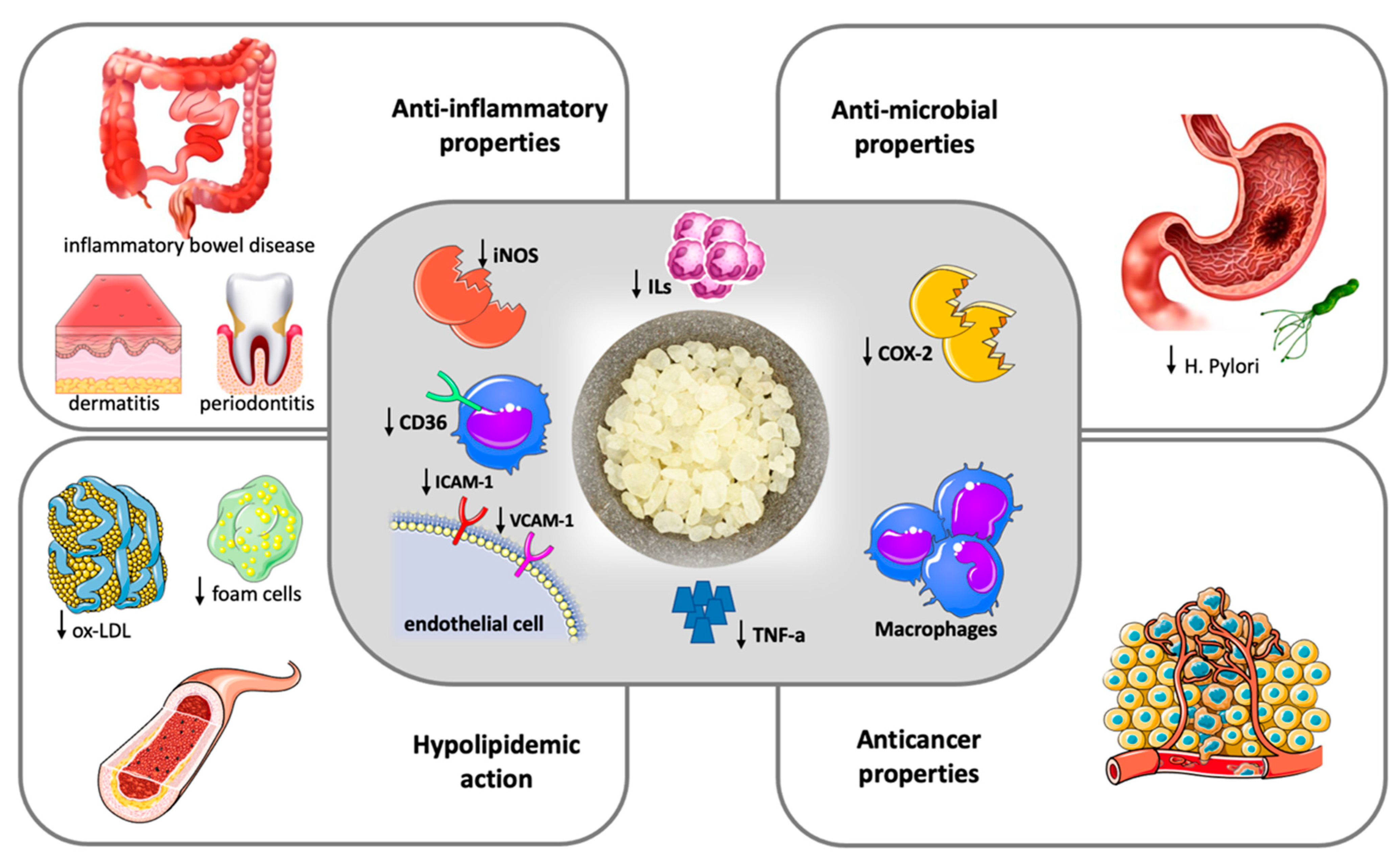
2. Anti-Inflammatory Properties
3. Anti-Oxidant Properties
4. Anti-Atherogenic Properties
5. Anticancer Properties
6. Antibacterial Properties
7. Adverse Effects—Toxicity
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ierapetritis, D. The Geography of the Chios Mastic Trade from the 17th through to the 19th Century. Ethnobot. Res. Appl. 2010, 8, 153–167. [Google Scholar] [CrossRef] [Green Version]
- Medicines Agency. E. EMA-Annual Report 2015; European Medicines Agency Press: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Paraschos, S.; Mitakou, S.; Skaltsounis, A.-L. Chios Gum Mastic: A Review of its Biological Activities. Curr. Med. Chem. 2012, 19, 2292–2302. [Google Scholar] [CrossRef] [PubMed]
- Assimopoulou, A.N.; Papageorgiou, V.P. GC-MS analysis of penta- and tetra-cyclic triterpenes from resins of Pistacia species. Part I. Pistacia lentiscus var. Chia. Biomed. Chromatogr. 2005, 19, 285–311. [Google Scholar] [CrossRef] [PubMed]
- Magiatis, P.; Melliou, E.; Skaltsounis, A.L.; Chinou, I.B.; Mitaku, S. Chemical composition and antimicrobial activity of the essential oils of Pistacia lentiscus var. chia. Planta Med. 1999, 65, 749–752. [Google Scholar] [CrossRef]
- Cronkite, D.A.; Strutt, T.M. The regulation of Inflammation by innate and adaptive lymphocytes. J. Immunol. Res. 2018, 2018, 1467538. [Google Scholar] [CrossRef]
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, S140–S146. [Google Scholar] [CrossRef]
- Zhou, L.; Satoh, K.; Takahashi, K.; Watanabe, S.; Nakamura, W.; Maki, J.; Hatano, H.; Takekawa, F.; Shimada, C.; Sakagami, H. Re-evaluation of anti-inflammatory activity of mastic using activated macrophages. Vivo 2009, 23, 583–590. [Google Scholar]
- Loizou, S.; Paraschos, S.; Mitakou, S.; Chrousos, G.P.; Lekakis, I.; Moutsatsou, P. Chios mastic gum extract and isolated phytosterol tirucallol exhibit anti-inflammatory activity in human aortic endothelial cells. Exp. Biol. Med. 2009, 234, 553–561. [Google Scholar] [CrossRef]
- Kaliora, A.C.; Stathopoulou, M.G.; Triantafillidis, J.K.; Dedoussis, G.V.; Andrikopoulos, N.K. Chios mastic treatment of patients with active Crohn’s disease clinical research. World J. Gastroenterol. 2007, 13, 748–753. [Google Scholar] [CrossRef]
- Kaliora, A.C.; Stathopoulou, M.G.; Triantafillidis, J.K.; Dedoussis, G.V.; Andrikopoulos, N.K. Alterations in the function of circulating mononuclear cells derived from patients with Crohn’s disease treated withmastic. World J. Gastroenterol. 2007, 13, 6031–6036. [Google Scholar] [CrossRef]
- Papada, E.; Gioxari, A.; Amerikanou, C.; Forbes, A.; Tzavara, C.; Smyrnioudis, I.; Kaliora, A.C. Regulation of faecal biomarkers in inflammatory bowel disease patients treated with oral mastiha (Pistacia lentiscus) supplement: A double-blind and placebo-controlled randomised trial. Phyther. Res. 2019, 33, 360–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papada, E.; Forbes, A.; Amerikanou, C.; Torović, L.; Kalogeropoulos, N.; Tzavara, C.; Triantafillidis, J.K.; Kaliora, A.C. Antioxidative efficacy of a Pistacia lentiscus supplement and its effect on the plasma amino acid profile in inflammatory bowel disease: A Randomised, double-blind, placebo-controlled trial. Nutrients 2018, 10, 1779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papada, E.; Amerikanou, C.; Torović, L.; Kalogeropoulos, N.; Tzavara, C.; Forbes, A.; Kaliora, A.C. Plasma free amino acid profile in quiescent Inflammatory Bowel Disease patients orally administered with Mastiha (Pistacia lentiscus); A randomised clinical trial. Phytomedicine 2019, 56, 40–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amerikanou, C.; Dimitropoulou, E.; Gioxari, A.; Papada, E.; Tanaini, A.; Fotakis, C.; Zoumpoulakis, P.; Kaliora, A.C. Linking the IL-17A immune response with NMR-based faecal metabolic profile in IBD patients treated with mastiha. Biomed. Pharmacother. 2021, 138, 111535. [Google Scholar] [CrossRef] [PubMed]
- Dabos, K.J.; Sfika, E.; Vlatta, L.J.; Frantzi, D.; Amygdalos, G.I.; Giannikopoulos, G. Is Chios mastic gum effective in the treatment of functional dyspepsia? A prospective randomised double-blind placebo controlled trial. J. Ethnopharmacol. 2010, 127, 205–209. [Google Scholar] [CrossRef]
- Kanoni, S.; Kumar, S.; Amerikanou, C.; Kurth, M.J.; Stathopoulou, M.G.; Bourgeois, S.; Masson, C.; Kannt, A.; Cesarini, L.; Kontoe, M.S.; et al. Nutrigenetic interactions might modulate the antioxidant and anti-inflammatory status in mastiha-supplemented patients with NAFLD. Front. Immunol. 2021, 12, 1688. [Google Scholar] [CrossRef]
- Moudi, Z.; Edozahi, M.; Emami, S.A.; Asili, J.; Pour, M.S. Effects of mastic oleoresin on wound healing and episiotomy pain: A mixed methods study. J. Ethnopharmacol. 2018, 214, 225–231. [Google Scholar] [CrossRef]
- Triantafyllou, A.; Bikineyeva, A.; Dikalova, A.; Nazarewicz, R.; Lerakis, S.; Dikalov, S. Anti-inflammatory activity of Chios mastic gum is associated with inhibition of TNF-alpha induced oxidative stress. Nutr. J. 2011, 10, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Kartalis, A.; Didagelos, M.; Georgiadis, I.; Benetos, G.; Smyrnioudis, N.; Marmaras, H.; Voutas, P.; Zotika, C.; Garoufalis, S.; Andrikopoulos, G. Effects of Chios mastic gum on cholesterol and glucose levels of healthy volunteers: A prospective, randomized, placebo-controlled, pilot study (chios-mastiha). Eur. J. Prev. Cardiol. 2016, 23, 722–729. [Google Scholar] [CrossRef]
- Kontogiannis, C.; Georgiopoulos, G.; Loukas, K.; Papanagnou, E.D.; Pachi, V.K.; Bakogianni, I.; Laina, A.; Kouzoupis, A.; Karatzi, K.; Trougakos, I.P.; et al. Chios mastic improves blood pressure haemodynamics in patients with arterial hypertension: Implications for regulation of proteostatic pathways. Eur. J. Prev. Cardiol. 2019, 26, 328–331. [Google Scholar] [CrossRef]
- Kottakis, F.; Kouzi-Koliakou, K.; Pendas, S.; Kountouras, J.; Choli-Papadopoulou, T. Effects of mastic gum Pistacia lentiscus var. Chia on innate cellular immune effectors. Eur. J. Gastroenterol. Hepatol. 2009, 21, 143–149. [Google Scholar] [CrossRef]
- Dabos, K.J.; Sfika, E.; Vlatta, L.J.; Giannikopoulos, G. The effect of mastic gum on Helicobacter pylori: A randomized pilot study. Phytomedicine 2010, 17, 296–299. [Google Scholar] [CrossRef]
- Bebb, J.R.; Bailey-Flitter, N.; Ala’Aldeen, D.; Atherton, J.C. Mastic gum has no effect on Helicobacter pylori load in vivo. J. Antimicrob. Chemother. 2003, 52, 522–523. [Google Scholar] [CrossRef] [Green Version]
- Zahouani, Y.; Ben Rhouma, K.; Kacem, K.; Sebai, H.; Sakly, M. Aqueous leaf extract of Pistacia lentiscus improves acute acetic acid-induced colitis in rats by reducing inflammation and oxidative stress. J. Med. Food 2021, 24, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Gioxari, A.; Kaliora, A.C.; Papalois, A.; Agrogiannis, G.; Triantafillidis, J.K.; Andrikopoulos, N.K. Pistacia lentiscus resin regulates intestinal damage and inflammation in trinitrobenzene sulfonic acid-induced colitis. J. Med. Food 2011, 14, 1403–1411. [Google Scholar] [CrossRef] [PubMed]
- Ostovan, M.; Fazljou, S.M.B.; Khazraei, H.; Khodaei, M.A.; Torbati, M. The anti-inflammatory effect of Pistacia lentiscus in a rat model of colitis. J. Inflamm. Res. 2020, 13, 369. [Google Scholar] [CrossRef] [PubMed]
- Kannt, A.; Papada, E.; Kammermeier, C.; D’Auria, G.; Jiménez-Hernández, N.; Stephan, M.; Schwahn, U.; Madsen, A.N.; Østergaard, M.V.; Dedoussis, G.; et al. Mastiha (Pistacia lentiscus) improves gut microbiota diversity, hepatic steatosis, and disease activity in a biopsy-confirmed mouse model of advanced non-alcoholic steatohepatitis and fibrosis. Mol. Nutr. Food Res. 2019, 63, 1688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Georgiadis, I.; Karatzas, T.; Korou, L.M.; Katsilambros, N.; Perrea, D. Beneficial health effects of chios gum mastic and peroxisome proliferator-activated receptors: Indications of common mechanisms. J. Med. Food 2015, 18, 1–10. [Google Scholar] [CrossRef]
- Kishimoto, R.; Kato, N.; Koike, M.; Iwashita, N.; Takagi, Y.; Fukuyama, T. Topical treatment with mastic (resin from Pistacia lentiscus) elicits anti-inflammatory and anti-pruritic responses by modulating keratinocyte activation in a mouse model of allergic dermatitis. Phytomedicine 2021, 91, 153679. [Google Scholar] [CrossRef]
- Khedir, S.B.; Bardaa, S.; Chabchoub, N.; Moalla, D.; Sahnoun, Z.; Rebai, T. The healing effect of Pistaciaï lentiscus fruit oil on laser burn. Pharm. Biol. 2017, 55, 1407–1414. [Google Scholar] [CrossRef] [Green Version]
- Sharifi-Rad, M.; Anil Kumar, N.V.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Tsouh Fokou, P.V.; Azzini, E.; Peluso, I.; et al. Lifestyle, oxidative stress, and antioxidants: Back and forth in the pathophysiology of chronic diseases. Front. Physiol. 2020, 11, 694. [Google Scholar] [CrossRef]
- Dayem, A.A.; Hossain, M.K.; Lee, S.B.; Kim, K.; Saha, S.K.; Yang, G.M.; Choi, H.Y.; Cho, S.G. The role of reactive oxygen species (ROS) in the biological activities of metallic nanoparticles. Int. J. Mol. Sci. 2017, 18, 120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poznyak, A.V.; Nikiforov, N.G.; Markin, A.M.; Kashirskikh, D.A.; Myasoedova, V.A.; Gerasimova, E.V.; Orekhov, A.N.; Pandey, A.K.; Woolard, M.D.; Eid, A.H. Overview of OxLDL and its impact on cardiovascular health: Focus on atherosclerosis. Front. Pharmacol. 2021, 11, 2248. [Google Scholar] [CrossRef] [PubMed]
- Andrikopoulos, N.K.; Kaliora, A.C.; Assimopoulou, A.N.; Papapeorgiou, V.P. Biological activity of some naturally occurring resins, gums and pigments against in vitro LDL oxidation. Phytother. Res. 2003, 17, 501–507. [Google Scholar] [CrossRef]
- Cosentino-Gomes, D.; Rocco-Machado, N.; Meyer-Fernandes, J.R. Cell signaling through protein kinase C oxidation and activation. Int. J. Mol. Sci. 2012, 13, 10697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dedoussis, G.V.Z.; Kaliora, A.C.; Psarras, S.; Chiou, A.; Mylona, A.; Papadopoulos, N.G.; Andrikopoulos, N.K. Antiatherogenic effect of Pistacia lentiscus via GSH restoration and downregulation of CD36 mRNA expression. Atherosclerosis 2004, 174, 293–303. [Google Scholar] [CrossRef]
- Feng, J.; Han, J.; Freida, S.; Pearce, A.; Silverstein, R.L.; Gotto, A.M.; Hajjar, D.P.; Nicholson, A.C.; Feng, J.; Han, J.; et al. Induction of CD36 expression by oxidized LDL and IL-4 by a common signaling pathway dependent on protein kinase C and PPAR-γ. J. Lipid Res. 2000, 41, 688–696. [Google Scholar] [CrossRef]
- Andreadou, I.; Mitakou, S.; Paraschos, S.; Efentakis, P.; Magiatis, P.; Kaklamanis, L.; Halabalaki, M.; Skaltsounis, L.; Iliodromitis, E.K. “Pistacia lentiscus L.” reduces the infarct size in normal fed anesthetized rabbits and possess antiatheromatic and hypolipidemic activity in cholesterol fed rabbits. Phytomedicine 2016, 23, 1220–1226. [Google Scholar] [CrossRef]
- Triantafyllou, A.; Chaviaras, N.; Sergentanis, T.N.; Protopapa, E.; Tsaknis, J. Chios mastic gum modulates serum biochemical parameters in a human population. J. Ethnopharmacol. 2007, 111, 43–49. [Google Scholar] [CrossRef]
- Balan, K.V.; Prince, J.; Han, Z.; Dimas, K.; Cladaras, M.; Wyche, J.H.; Sitaras, N.M.; Pantazis, P. Antiproliferative activity and induction of apoptosis in human colon cancer cells treated in vitro with constituents of a product derived from Pistacia lentiscus L. var. chia. Phytomedicine 2007, 14, 263–272. [Google Scholar] [CrossRef]
- Balan, K.V.; Demetzos, C.; Prince, J.; Dimas, K.; Cladaras, M.; Han, Z.; Wyche, J.H.; Pantazis, P. Induction of apoptosis in human colon cancer HCT116 cells treated with an extract of the plant product, chios mastic gum. Vivo 2005, 19, 93–102. [Google Scholar]
- He, M.L.; Yuan, H.Q.; Jiang, A.L.; Gong, A.Y.; Chen, W.W.; Zhang, P.J.; Young, C.Y.F.; Zhang, J.Y. Gum mastic inhibits the expression and function of the androgen receptor in prostate cancer cells. Cancer 2006, 106, 2547–2555. [Google Scholar] [CrossRef] [PubMed]
- He, M.-L.; Li, A.; Xu, C.-S.; Wang, S.-L.; Zhang, M.-J.; Gu, H.; Yang, Y.-Q.; Tao, H.-H. Mechanisms of antiprostate cancer by gum mastic: NF-κB signal as target 1. Acta Pharmacol. Sin. 2007, 28, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Loutrari, H.; Magkouta, S.; Pyriochou, A.; Koika, V.; Kolisis, F.N.; Papapetropoulos, A.; Roussos, C. Mastic oil from Pistacia lentiscus var. chia inhibits growth and survival of human K562 leukemia cells and attenuates angiogenesis. Nutr. Cancer 2006, 55, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Tassoua, C.C.; Nycha, G.J.E. Antimicrobial activity of the essential oil of mastic gum (Pistacia Zentiscus var. chia) on gram positive and gram negative bacteria in broth and in model food system. Int. Biodeterior. Biodegrad. 1995, 36, 411–420. [Google Scholar] [CrossRef]
- Di Pierro, F.; Sagheddu, V.; Galletti, S.; Forti, M.; Elli, M.; Bertuccioli, A.; Gaeta, S. Antibacterial activity of a fractionated Pistacia lentiscus oil against pharyngeal and ear pathogens, alone or in combination with antibiotics. Front. Microbiol. 2021, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Koychev, S.; Dommisch, H.; Chen, H.; Pischon, N. Antimicrobial effects of mastic extract against oral and periodontal pathogens. J. Periodontol. 2017, 88, 511–517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milia, E.; Usai, M.; Szotáková, B.; Elstnerová, M.; Králová, V.; D’Hallewin, G.; Spissu, Y.; Barberis, A.; Marchetti, M.; Bortone, A.; et al. The pharmaceutical ability of Pistacia lentiscus L leaves essential oil against periodontal bacteria and candida sp. and its anti-inflammatory potential. Antibiotics 2020, 9, 281. [Google Scholar] [CrossRef] [PubMed]
- Ezz Eldin, H.M.; Badawy, A.F. In vitro anti-trichomonas vaginalis activity of pistacia lentiscus mastic and ocimum basilicum essential oil. J. Parasit. Dis. 2015, 39, 465–473. [Google Scholar] [CrossRef] [Green Version]
- Partipilo, M.L.; Woster, P.S. The role of helicobacter pylori in peptic ulcer disease. Pharmacother. J. Hum. Pharmacol. Drug Ther. 1993, 13, 330–339. [Google Scholar] [CrossRef]
- Papastergiou, V.; Georgopoulos, S.; Karatapanis, S. Current and future insights in H. pylori eradication regimens: The need of tailoring therapy. Curr. Pharm. Des. 2014, 20, 4521–4532. [Google Scholar] [CrossRef] [PubMed]
- Savoldi, A.; Carrara, E.; Graham, D.Y.; Conti, M.; Tacconelli, E. Prevalence of antibiotic resistance in helicobacter pylori: A systematic review and meta-analysis in World Health Organization regions. Gastroenterology 2018, 155, 1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huwez, F.U.; Thirlwell, D.; Cockayne, A.; Ala’Aldeen, D.A.M. Mastic gum kills Helicobacter pylori. N. Engl. J. Med. 1998, 339, 1946. [Google Scholar] [CrossRef] [PubMed]
- Marone, P.; Bono, L.; Leone, E.; Bona, S.; Carretto, E.; Perversi, L. Bactericidal activity of Pistacia lentiscus mastic gum against Helicobacter Pylori. Journal of Chemotherapy 2013, 13, 611–614. [Google Scholar] [CrossRef]
- Shaw, D.W. Contact Dermatitis From Gum Mastic (Pistacia lentiscus) and Gum Storax (Liquidambar styraciflua) in Mastisol-Allergic Patients. Dermat. Contact. Atopic. Occup. Drug 2021, 32, 430–436. [Google Scholar] [CrossRef]
- Ostovan, M.; Anbardar, M.H.; Khazraei, H.; Fazljou, S.M.b.; Khodabandeh, Z.; Shamsdin, S.A.; Khodaei, M.A.; Torbati, M. The short-term effects of Pistacia lentiscus oil and sesame oil on liver and kidney pathology of rats and human cancer cell lines. Galen Med. J. 2020, 9, e2001. [Google Scholar] [CrossRef]
- Kang, J.S.; Wanibuchi, H.; Salim, E.I.; Kinoshita, A.; Fukushima, S. Evaluation of the toxicity of mastic gum with 13 weeks dietary administration to F344 rats. Food Chem. Toxicol. 2007, 45, 494–501. [Google Scholar] [CrossRef]
- Doi, K.; Wei, M.; Kitano, M.; Uematsu, N.; Inoue, M.; Wanibuchi, H. Enhancement of preneoplastic lesion yield by Chios Mastic Gum in a rat liver medium-term carcinogenesis bioassay. Toxicol. Appl. Pharmacol. 2009, 234, 135–142. [Google Scholar] [CrossRef]
- Janakat, S.; Al-Merie, H. Evaluation of hepatoprotective effect of Pistacia lentiscus, Phillyrea latifolia and Nicotiana glauca. J. Ethnopharmacol. 2002, 83, 135–138. [Google Scholar] [CrossRef]
| Compound |
|---|
| 1,4-poly-β-myrcene |
| 20(S)-3β-acetoxy-20-hydroxydammara-24-ene |
| 3-oxo-28-norlup-20 (29)-ene |
| 3β-hydroxy-28-norolean-12-ene |
| 3-oxo-28-norolean-12-ene |
| 3-oxo-dammara-20 (21),24-diene |
| (8R)-3-Oxo-8-hydroxy-polypoda13E,17E,21-triene |
| 3-oxo-malabarica-14(26),17E,21-triene |
| 3β-hydroxymalabarica-14(26),17E,21-triene |
| banillic acid |
| gallic acid trans-cinnamomic acid |
| isomasticadienonic acid |
| masticadienolic acid |
| moronic acid |
| oleanolic acid |
| oleanolic aldehyde |
| p-hydroxy-benzoic acid |
| p-hydroxy-phenylacetic acid |
| tirucallol |
| tyrosol |
| Study | Design | Effect |
|---|---|---|
| Kaliora et al. [10] | 10 pts with active CD and 8 healthy controls 2.2 g of mastic daily for 4 weeks |
|
| Kaliora et al. [11] | 10 pts with active CD and 8 healthy controls 2.2 g of mastic daily, 4 weeks |
|
| Papada et al. [12,13] | 60 pts with IBD randomized to either 2.8 g of mastic daily for 3 months or placebo |
|
| Papada et al. [14] | 68 pts with IBD randomized to either 2.8 g of mastic daily for 6 months or placebo |
|
| Amerikanou et al. [15] | 129 pts with IBD—68 randomized to mastic group (2.8 g daily for 6 months for pts in remission and for 3 months for pts in relapse) and 61 to placebo |
|
| Dabos et al. [16] | 148 pts with functional dyspepsia randomized to either mastic 350 mg tid or placebo for 3 weeks |
|
| Kanoni et al. [17] | 98 patients with obesity (BMI ≥ 30 kg/m2) and NAFLD and randomized to either mastic 2.1 g/day or placebo for 6 months |
|
| Moudi et al. [18] | 147 postpartum women randomized to topical application of 15 g mastic for 3 days on episiotomy wound or to placebo |
|
| Triantafyllou et al. [19] | 133 subjects were randomized to either 5 g mastic powder (high dose) or mastic solution for 18 months |
|
| Kartalis et al. [20] | 156 subjects received different doses of mastic for 8 weeks |
|
| Kontogiannis et al. [21] | 27 subject (13 hypertensive) randomized to receive one dose of 2.8 g mastic |
|
| Kottakis et al. [22] | 5 pts with H. Pylori infection and 3 controls treated with 1 g of mastic daily for 2 months |
|
| Dabos et al. [23] | 52 pts with H. Pylori randomized to receive either 350 mg tid of mastic for 14 days (Group A), or 1.05 g tid of mastic (Group B) for 14 days, or pantoprazole 20 mg bd plus mastic 350 mg tid for 14 days (Group C) or pantoprazole 20 mg bd plus amoxicillin 1 g bd plus clarithromycin 500 mg bd for 10 days (Group D) |
|
| Bebb et al. [24] | 8 pts with H. Pylori 1 g mastic four times daily for 14 days |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soulaidopoulos, S.; Tsiogka, A.; Chrysohoou, C.; Lazarou, E.; Aznaouridis, K.; Doundoulakis, I.; Tyrovola, D.; Tousoulis, D.; Tsioufis, K.; Vlachopoulos, C.; et al. Overview of Chios Mastic Gum (Pistacia lentiscus) Effects on Human Health. Nutrients 2022, 14, 590. https://doi.org/10.3390/nu14030590
Soulaidopoulos S, Tsiogka A, Chrysohoou C, Lazarou E, Aznaouridis K, Doundoulakis I, Tyrovola D, Tousoulis D, Tsioufis K, Vlachopoulos C, et al. Overview of Chios Mastic Gum (Pistacia lentiscus) Effects on Human Health. Nutrients. 2022; 14(3):590. https://doi.org/10.3390/nu14030590
Chicago/Turabian StyleSoulaidopoulos, Stergios, Aikaterini Tsiogka, Christina Chrysohoou, Emilia Lazarou, Konstantinos Aznaouridis, Ioannis Doundoulakis, Dimitra Tyrovola, Dimitris Tousoulis, Konstantinos Tsioufis, Charalambos Vlachopoulos, and et al. 2022. "Overview of Chios Mastic Gum (Pistacia lentiscus) Effects on Human Health" Nutrients 14, no. 3: 590. https://doi.org/10.3390/nu14030590