Taiwanese Vegetarians Are Associated with Lower Dementia Risk: A Prospective Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Research Design
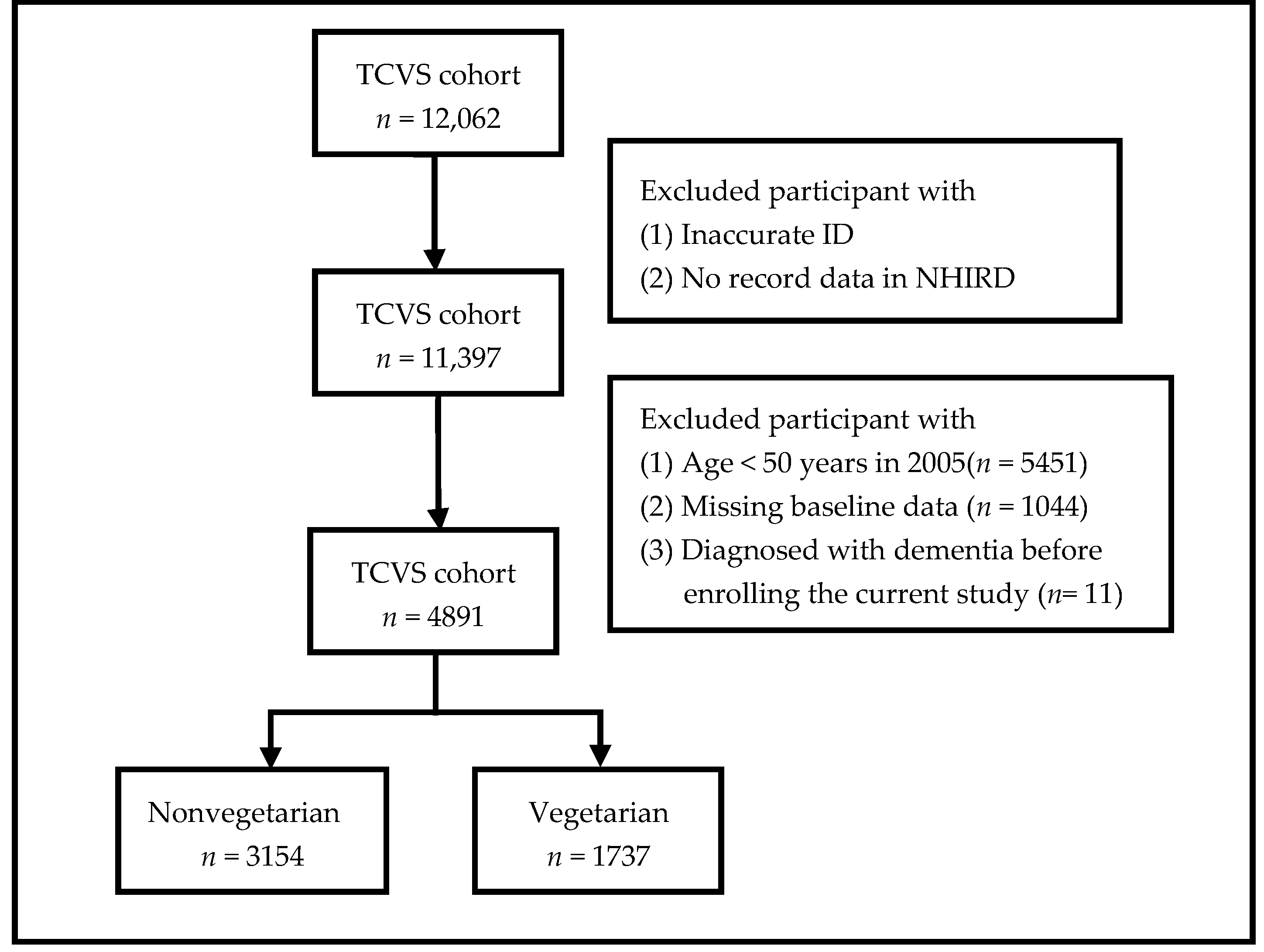
2.2. Criteria for Exclusion
2.3. Dementia Ascertainment
2.4. Statistical Analysis
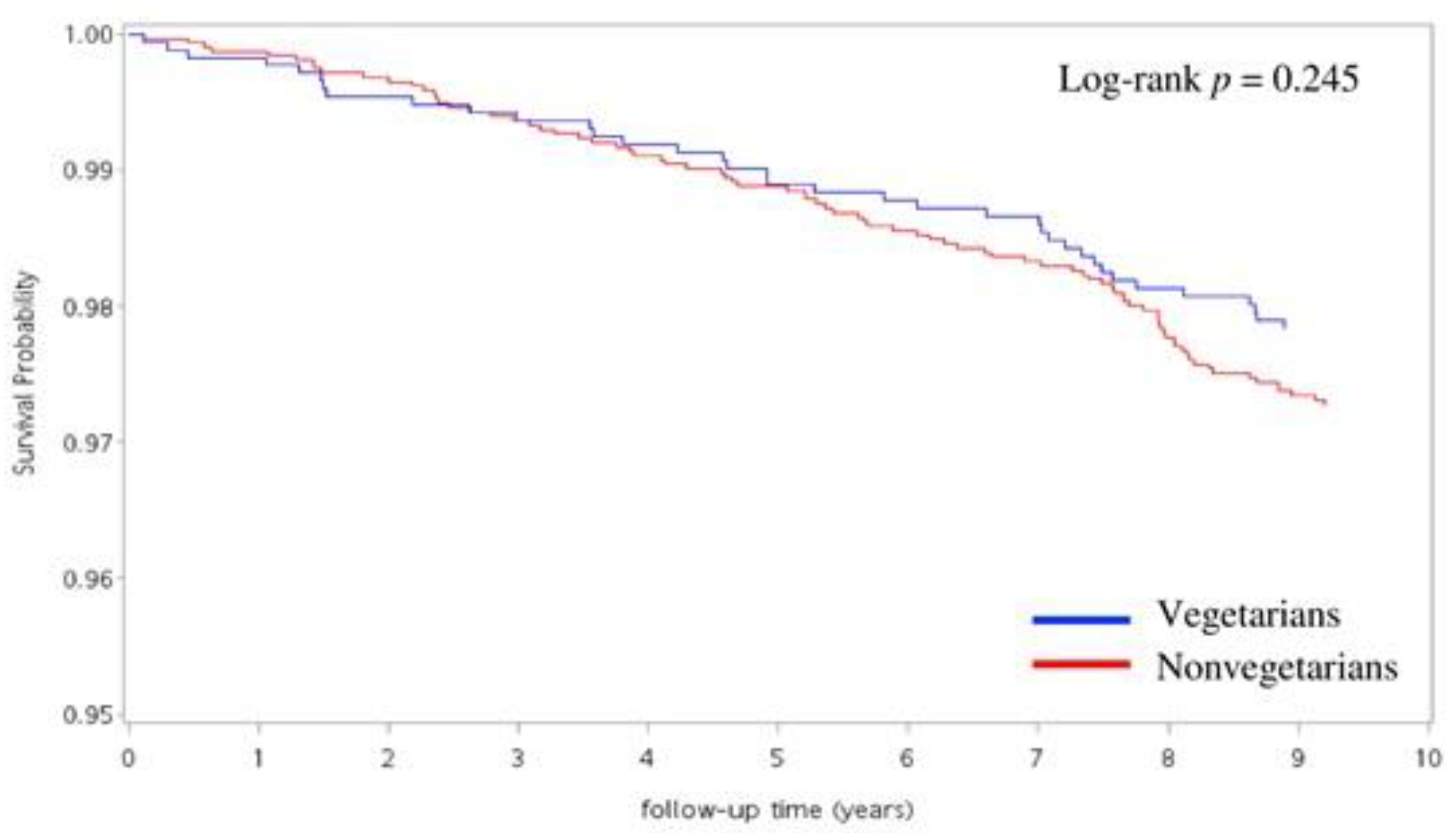
3. Results
4. Discussion
4.1. Cardiometabolic Risk Factors
4.2. Inflammation
4.3. Gut Microbiota (GM)
4.4. Dietary Pattern
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Matthew, B.; Heather, M.S.; Maria, C.C.; Sam, F.; Hye, K.; Harry, J. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimers Dement. 2015, 6, 718–726. [Google Scholar]
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafreda, S.G.; Huntley, J.; Ames, D.; Ballard, C.; Banerjee, S.; Burns, A.; Cohen-Mansfield, J.; et al. Dementia prevention, intervention, and care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef] [Green Version]
- Livingston, G.; Huntley, J.; Sommerlad, A.; Ames, D.; Ballard, C.; Banerjee, S.; Brayne, C.; Burns, A.; Cohen-Mansfield, J.; Cooper, C.; et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 2020, 396, 413–446. [Google Scholar] [CrossRef]
- Kalmijn, S.; Launer, L.J.; Ott, A.; Witteman, J.C.M.; Hofman, A.; Breteler, M.M.B. Dietary fat intake and the risk of incident dementia in the Rotterdam study. Ann. Neurol. 1997, 42, 776–782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lourida, I.; Soni, M.; Tompson-Coon, J.; Purandare, N.; Lang, I.A.; Ukoumunne, O.C.; Llewellyn, D.J. Mediterranean diet, cognitive function, and Dementia: A systematic review. Epidemiology 2013, 4, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Ninomiya, T.; Ohara, T.; Doi, Y.; Uchida, K.; Shirota, T.; Yonemoto, K.; Kitazono, T.; Kiyohara, Y. Dietary patterns and risk of dementia in an elderly Japanese population: The Hisayama Study. Am. J. Clin. Nutr. 2013, 97, 1076–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, C.-Y.; Sun, Y.; Lee, H.-J.; Chen, T.-F.; Wang, P.-N.; Lin, K.-N.; Tang, L.-Y.; Lin, C.-C.; Chiu, M.-J. Modest overweight and healthy dietary habits reduce risk of dementia: A nationwide survey in Taiwan. JPAD 2017, 4, 37–43. [Google Scholar] [CrossRef]
- Kahleova, H.; Levin, S.; Barnard, N. Cardio-Metabolic Benefits of Plant-Based Diets. Nutrient 2017, 9, 848. [Google Scholar] [CrossRef]
- Chiu, T.H.T.; Huang, H.-Y.; Chiu, Y.-F.; Pan, W.-H.; Kao, H.-Y.; Chiu, J.P.C.; Lin, M.-N.; Lin, C.-L. Taiwanese Vegetarians and Omnivores: Dietary Composition, Prevalence of Diabetes and IFG. PLoS ONE 2014, 9, e88547. [Google Scholar] [CrossRef] [Green Version]
- Chiu, T.H.; Chang, H.-R.; Wang, L.-Y.; Chang, C.-C.; Lin, M.-N.; Lin, C.-L. Vegetarian diet and incidence of total, ischemic, and hemorrhagic stroke in 2 cohorts in Taiwan. Neurology 2020, 94, e1112–e1121. [Google Scholar] [CrossRef] [Green Version]
- Shen, Y.-C.; Chang, C.-E.; Lin, M.-N.; Lin, C.-L. Vegetarian Diet Is Associated with Lower Risk of Depression in Taiwan. Nutrient 2021, 13, 1059. [Google Scholar] [CrossRef] [PubMed]
- National Health Insurance Research Database. Available online: https://nhird.nhri.org.tw/en/index.html (accessed on 7 October 2021).
- Niu, H.; Álvarez-Álvarez, I.; Guillén-Grima, F.; Aguinaga-Ontoso, I. Prevalence and incidence of Alzheimer’s disease in Europe: A meta-analysis. Neurología 2017, 32, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.K.; Lai, C.L.; Tai, C.T.; Lin, R.T.; Yen, Y.Y.; Howng, S.L. Incidence and subtypes of dementia in southern Taiwan Impact of socio-demographic factors. Neurology 1998, 50, 1572–1579. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Shen, Y.C.; Chen, C.H.; Zhau, Y.W.; Li, S.R.; Lu, M. A three-year follow-up study of age-related dementia in an urban area of Beijing. Acta Psychiatr. Scand. 1991, 83, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.-B.; Yiao, S.-Y.; Sun, Y.; Lee, H.-J.; Yang, S.-C.; Chiu, M.-J.; Chen, T.-F.; Lin, K.-N.; Tang, L.-Y.; Lin, C.-C.; et al. Comorbidity and dementia: A nationwide survey in Taiwan. PLoS ONE 2017, 12, e0175475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kivipelto, M.; Helkala, E.-L.; Laakso, M.; Hänninen, T.; Hallikainen, M.; Alhainen, K.; Soininen, H.; Tuomilehto, J.; Nissinen, A. Midlife vascular risk factors and Alzheimer’s disease in later life: Longitudinal, population based study. BMJ 2001, 322, 1447–1451. [Google Scholar] [CrossRef] [Green Version]
- Gottesman, R.; Schneider, A.L.C.; Zhou, Y.; Coresh, J.; Green, E.; Gupta, N.; Knopman, D.S.; Mintz, A.; Rahmim, A.; Sharrett, A.R.; et al. Association Between Midlife Vascular Risk Factors and Estimated Brain Amyloid Deposition. JAMA J. Am. Med. Assoc. 2017, 317, 1443–1450. [Google Scholar] [CrossRef]
- Rannveig, S.E.; Karin, P.; Maria, L.B.; Anne-Brita, K.; Lena, C.; Knut, E.; Geil, S.; Eva, S.; Ingvild, S. Association between vascular comorbidities and progression of Alzheimer’s disease: A two-year observational study in Norwegian memory clinics. BMC Geriatr. 2018, 18, 120–127. [Google Scholar]
- Corrada, M.M.; Hayden, K.M.; Paganini-Hill, A.; Bullain, S.S.; DeMoss, J.; Aguirre, C.; Brookmeyer, R.; Kawas, C.H. Age of onset of hypertension and risk of dementia in the oldest-old: The 90+ Study. Alzheimer’s Dement. 2017, 13, 103–110. [Google Scholar] [CrossRef] [Green Version]
- Heneka, M.T.; Carson, M.J.; El Khoury, J.; Landreth, G.E.; Brosseron, F.; Feinstein, D.L.; Jacobs, A.H.; Wyss-Coray, T.; Vitorica, J.; Ransohoff, R.M.; et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar] [CrossRef] [Green Version]
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Li, H.; Wang, Y.; Barnes, L.; Schneider, J.A.; Bennett, D.A.; Morris, M.C. Relation of DASH- and Mediterranean-like dietary patterns to cognitive decline in older persons. Neurology 2014, 83, 1410–1416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van De Rest, O.; Berendsen, A.A.M.; Haveman-Nies, A.; De Groot, L.C.P.G.M. Dietary Patterns, Cognitive Decline, and Dementia: A Systematic Review. Adv. Nutr. 2015, 6, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Farooqui, A.A.; Horrocks, L.A.; Farooqui, T. Modulation of inflammation in brain: A matter of fat. J. Neurochem. 2006, 101, 577–599. [Google Scholar] [CrossRef]
- Fisher, M.; Levine, P.H.; Weiner, B.; Ockene, I.S.; Johnson, B.; Johnson, M.H.; Natale, A.M.; Vaudreuil, C.H.; Hoogasian, J. The effect of vegetarian diets on plasma lipid and platelet levels. Arch. Intern. Med. 1986, 146, 1193–1197. [Google Scholar] [CrossRef]
- Li, D.; Ball, M.; Bartlett, M.; Sinclair, A. Lipoprotein(a), essential fatty acid status and lipoprotein lipids in female Australian veg-etarians. Clin. Sci. 1999, 97, 175. [Google Scholar] [CrossRef]
- Vogt, N.M.; Kerby, R.L.; Dill-McFarland, K.A.; Harding, S.J.; Merluzzi, A.P.; Johnson, S.C.; Carlsson, C.M.; Asthana, S.; Zetterberg, H.; Blennow, K.; et al. Gut microbiome alterations in Alzheimer’s disease. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Leblhuber, F.; Ehrlich, D.; Steiner, K.; Geisler, S.; Fuchs, D.; Lanser, L.; Kurz, K. The Immunopathogenesis of Alzheimer’s Disease Is Related to the Composition of Gut Microbiota. Nutrient 2021, 13, 361. [Google Scholar] [CrossRef]
- Liu, S.; Gao, J.; Zhu, M.; Liu, K.; Zhang, H.-L. Gut Microbiota and Dysbiosis in Alzheimer’s Disease: Implications for Pathogenesis and Treatment. Mol. Neurobiol. 2020, 57, 5026–5043. [Google Scholar] [CrossRef]
- Megur, A.; Baltriukienė, D.; Bukelskienė, V.; Burokas, A. The Microbiota–Gut–Brain Axis and Alzheimer’s Disease: Neuroinflammation Is to Blame? Nutrient 2020, 13, 37. [Google Scholar] [CrossRef]
- Sochocka, M.; Donskow-Łysoniewska, K.; Diniz, B.S.; Kurpas, D.; Brzozowska, E.; Leszek, J. The Gut Microbiome Alterations and Inflammation-Driven Pathogenesis of Alzheimer’s Disease—A Critical Review. Mol. Neurobiol. 2019, 56, 1841–1851. [Google Scholar] [CrossRef] [Green Version]
- Tomova, A.; Bukovsky, I.; Rembert, E.; Yonas, W.; Alwarith, J.; Barnard, N.D.; Kahleova, H. The Effects of Vegetarian and Vegan Diets on Gut Microbiota. Front. Nutr. 2019, 6, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, J.E.; Poles, J.; Shaw, D.P.; Karhu, E.; Khan, S.A.; Lyons, A.E.; Sacco, S.B.; McDaniel, H.R. The effects of twenty-one nutrients and phytonutrients on cognitive function: A narrative review. J. Clin. Transl. Res. 2021, 4, 575–620. [Google Scholar]
- Chauhan, A.; Chauhan, V. Beneficial Effects of Walnuts on Cognition and Brain Health. Nutrients 2020, 12, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grant, W.B. Trends in Diet and Alzheimer’s Disease During the Nutrition Transition in Japan and Developing Countries. J. Alzheimer’s Dis. 2013, 38, 611–620. [Google Scholar] [CrossRef]
- Grant, W.B. Using multicountry ecological and observational studies to determine dietary risk factors for Alzheimer’s disease. J. Am. Coll. Nutr. 2016, 35, 476–489. [Google Scholar] [CrossRef]
- Zhang, H.; Greenwood, D.C.; A Risch, H.; Bunce, D.; Hardie, L.J.; E Cade, J. Meat consumption and risk of incident dementia: Cohort study of 493,888 UK Biobank participants. Am. J. Clin. Nutr. 2021, 114, 175–184. [Google Scholar] [CrossRef]
- Giem, P.; Beeson, W.L.; Fraser, G.E. The incidence of dementia and intake of animal products: Preliminary findings from the Ad-ventist Health Study. Neuroepidemiology 1993, 12, 28–36. [Google Scholar] [CrossRef]
- Petersson, S.D.; Philippou, E. Mediterranean Diet, Cognitive Function, and Dementia: A Systematic Review of the Evidence. Adv. Nutr. Int. Rev. J. 2016, 7, 889–904. [Google Scholar] [CrossRef] [Green Version]
- Van den Brink, A.C.; Brouwer-Brolsma, E.M.; Berendsen, A.A.M.; Van De Rest, O. The Mediterranean, Dietary Approaches to Stop Hypertension (DASH), and Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) Diets Are Associated with Less Cognitive Decline and a Lower Risk of Alzheimer’s Disease—A Review. Adv. Nutr. 2019, 10, 1040–1065. [Google Scholar] [CrossRef]
- Zhang, H.; Hardie, L.; Bawajeeh, A.O.; Cade, J. Meat Consumption, Cognitive Function and Disorders: A Systematic Review with Narrative Synthesis and Meta-Analysis. Nutrient 2020, 12, 1528. [Google Scholar] [CrossRef] [PubMed]
| Nonvegetarians | Vegetarians | p-Value | |
|---|---|---|---|
| n (%) | n (%) | ||
| Total | 3154 (64.5) | 1737 (35.5) | |
| Sociodemographic characteristics | |||
| Age, Mean (SD) | 57.8 (6.3) | 58.1 (6.5) | 0.253 |
| Age group | |||
| <65 years | 2704 (85.7) | 1477 (85.0) | 0.525 |
| 65–74 years | 408 (12.9) | 221 (12.7) | |
| ≥75 years | 42 (1.3) | 39 (2.2) | |
| Sex | <0.01 | ||
| Male | 1345 (42.6) | 461 (26.5) | |
| Female | 1809 (57.4) | 1276 (73.5) | |
| Education level | <0.01 | ||
| ≤Elementary school | 1008(32.0) | 658 (37.9) | |
| Middle and high school | 1421 (45.1) | 744 (42.8) | |
| Higher education | 725 (23.0) | 335 (19.3) | |
| Marriage | <0.01 | ||
| Married | 2940 (93.2) | 1576 (90.7) | |
| Single/Divorced/Widowed | 214 (6.8) | 161 (9.3) | |
| Lifestyle habits | |||
| Regular exercise habit | 1470 (46.6) | 689 (39.7) | <0.01 |
| Smoking | 529 (16.8) | 173 (10.0) | <0.01 |
| Alcohol drinking | 507 (16.1) | 199 (11.5) | <0.01 |
| Nonvegetarians | Vegetarians | p-Value | |
|---|---|---|---|
| n (%) | n (%) | ||
| Hypertension | 554 (17.6%) | 282 (16.2%) | 0.250 |
| Diabetes mellitus | 449 (14.2%) | 190 (10.9%) | 0.001 * |
| Hyperlipidemia | 621 (19.7%) | 310 (17.8%) | 0.119 |
| COPD | 286 (9.1%) | 163 (9.4%) | 0.717 |
| Hyperthyroidism | 70 (2.2%) | 55 (3.2%) | 0.047 * |
| Hypothyroidism | 61 (1.9%) | 46 (2.6%) | 0.103 |
| Cerebrovascular disease | 393 (12.5%) | 172 (9.9%) | 0.008 * |
| Ischemic Heart disease | 454 (14.4%) | 221 (12.7%) | 0.109 |
| Cardiac arrhythmia | 290 (9.2%) | 198 (11.4%) | 0.015 * |
| Renal disease | 181 (5.7%) | 82 (4.7%) | 0.145 |
| Anxiety disease | 396 (12.6%) | 228 (13.1%) | 0.591 |
| Major depressive disorder | 69 (2.2%) | 31 (1.8%) | 0.398 |
| Substance use disorder | 15 (0.5%) | 3 (0.2%) | 0.041 * |
| Nonvegetarians | Vegetarians | |
|---|---|---|
| Cases/Person-year | 84/28,798.47 | 37/15,926.9 |
| Crude, HR (95% CI) | 1 | 0.796 (0.540–1.171) |
| Adjusted for age, sex | 1 | 0.688 (0.466– 1.018) |
| Adjusted for age, sex, education level, marriage | 1 | 0.676 (0.457–1.001) |
| Adjusted for age, sex, education level, marriage, regular exercise, smoking, drinking | 1 | 0.661 (0.446–0.979) * |
| Adjusted for age, sex, education level, marriage, regular exercise, smoking, drinking, baseline medical comorbidity | 1 | 0.671 (0.452– 0.996) * |
| Nonvegetarians | Vegetarians | Adjusted HR * | 95% CI | p-Value | |||
|---|---|---|---|---|---|---|---|
| N | Person-Years | N | Person-Years | ||||
| Stratification of Age Groups | |||||||
| 50–64 years | 36 | 24,906.05 | 12 | 13,678.64 | 0.601 | 0.309–1.166 | 0.422 |
| 65–74 years | 39 | 3560.74 | 15 | 1971.74 | 0.613 | 0.333–1.129 | 0.794 |
| ≥75 years | 9 | 331.69 | 10 | 276.51 | 1.089 | 0.335–3.543 | 0.233 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, J.-H.; Huang, C.-F.; Lin, M.-N.; Chang, C.-E.; Chang, C.-C.; Lin, C.-L. Taiwanese Vegetarians Are Associated with Lower Dementia Risk: A Prospective Cohort Study. Nutrients 2022, 14, 588. https://doi.org/10.3390/nu14030588
Tsai J-H, Huang C-F, Lin M-N, Chang C-E, Chang C-C, Lin C-L. Taiwanese Vegetarians Are Associated with Lower Dementia Risk: A Prospective Cohort Study. Nutrients. 2022; 14(3):588. https://doi.org/10.3390/nu14030588
Chicago/Turabian StyleTsai, Jui-Hsiu, Ching-Feng Huang, Ming-Nan Lin, Chiao-Erh Chang, Chia-Chen Chang, and Chin-Lon Lin. 2022. "Taiwanese Vegetarians Are Associated with Lower Dementia Risk: A Prospective Cohort Study" Nutrients 14, no. 3: 588. https://doi.org/10.3390/nu14030588
APA StyleTsai, J.-H., Huang, C.-F., Lin, M.-N., Chang, C.-E., Chang, C.-C., & Lin, C.-L. (2022). Taiwanese Vegetarians Are Associated with Lower Dementia Risk: A Prospective Cohort Study. Nutrients, 14(3), 588. https://doi.org/10.3390/nu14030588







