Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5
Abstract
Contents
| 1. Introduction | 2 |
| 2. Thiamine—Vitamin B1 | 2 |
| 2.1. Introduction and Properties | 2 |
| 2.2. Sources of Thiamine | 3 |
| 2.3. Pharmacokinetics of Thiamine | 7 |
| 2.4. Physiological Function of Thiamine | 9 |
| 2.5. Thiamine Deficiency | 10 |
| 2.6. Pharmacological Use of Thiamine | 12 |
| 2.7. Toxicity of Thiamine | 17 |
| 2.8. Drug-Vitamin Interactions Associated with Thiamine Deficiency | 17 |
| 3. Riboflavin—Vitamin B2 | 17 |
| 3.1. Introduction and Properties | 17 |
| 3.2. Sources of Riboflavin | 17 |
| 3.3. Pharmacokinetics of Riboflavin | 20 |
| 3.4. Physiological Functions of Riboflavin | 23 |
| 3.5. Riboflavin Deficiency | 24 |
| 3.6. Analytical Determination | 25 |
| 3.7. Pharmacological Use of Riboflavin | 26 |
| 3.8. Toxicity of Riboflavin | 26 |
| 3.9. Drug Interactions Affecting Pharmacokinetics and Interfering with Physiological Function of Riboflavin | 26 |
| 4. Niacin—Vitamin B3 | 27 |
| 4.1. Introduction and Properties | 27 |
| 4.2. Sources of Niacin | 27 |
| 4.3. Pharmacokinetics of Niacin | 31 |
| 4.4. Physiological Functions of Niacin | 33 |
| 4.5. Niacin Deficiency | 38 |
| 4.6. Pharmacological Use of Niacin | 38 |
| 4.7. Toxicity of Niacin | 44 |
| 5. Pantothenic Acid—Vitamin B5 | 45 |
| 5.1. Introduction and Properties | 45 |
| 5.2. Sources of Pantothenic Acid | 45 |
| 5.3. Physiological Function of Pantothenic Acid | 48 |
| 5.4. Pharmacokinetics of Pantothenic Acid | 49 |
| 5.5. Pantothenic Acid Deficiency | 53 |
| 5.6. Pharmacological Use of Pantothenic Acid | 55 |
| 6. Conclusions | 58 |
| References | 59 |
1. Introduction
2. Thiamine—Vitamin B1
2.1. Introduction and Properties
2.2. Sources of Thiamine
2.3. Pharmacokinetics of Thiamine
2.4. Physiological Function of Thiamine
2.5. Thiamine Deficiency
2.6. Pharmacological Use of Thiamine
2.7. Toxicity of Thiamine
2.8. Drug-Vitamin Interactions Associated with Thiamine Deficiency
3. Riboflavin—Vitamin B2
3.1. Introduction and Properties
3.2. Sources of Riboflavin
3.3. Pharmacokinetics of Riboflavin
3.3.1. Absorption
3.3.2. Metabolism
Uptake and Transport
Synthesis of Flavin Cofactors
Excretion
3.4. Physiological Functions of Riboflavin
3.4.1. Flavoproteins
3.4.2. Flavoproteins’ Importance in Human Health
3.5. Riboflavin Deficiency
3.6. Analytical Determination
3.7. Pharmacological Use of Riboflavin
3.8. Toxicity of Riboflavin
3.9. Drug Interactions Affecting Pharmacokinetics and Interfering with Physiological Function of Riboflavin
3.9.1. Boric Acid
3.9.2. Doxorubicin
3.9.3. Antipsychotics
3.9.4. Antidepressants
4. Niacin—Vitamin B3
4.1. Introduction and Properties
4.2. Sources of Niacin
4.3. Pharmacokinetics of Niacin
4.3.1. Absorption and Distribution
4.3.2. Metabolism
NAD Synthesis
NAD Recycling
Synthesis of NADP
4.3.3. Niacin Elimination
4.4. Physiological Functions of Niacin
4.4.1. Redox Reactions
4.4.2. NAD as Substrate
4.4.3. ADP-Ribosyl Cyclases
4.4.4. Poly(ADP-Ribose) Polymerases (PARP)
4.4.5. Sirtuins
4.5. Niacin Deficiency
4.6. Pharmacological Use of Niacin
4.6.1. Atherosclerosis, Dyslipidaemia and Cardiovascular Risk
Nicotinic Acid Receptor GPR109A
Mechanisms of Nicotinic Acid Action
4.6.2. Aging
4.6.3. Cancer and Cell Death
4.6.4. Neurological Disorders
Parkinson’s Disease
Multiple Sclerosis
Schizophrenia
4.6.5. Skin Cancer Prevention
4.7. Toxicity of Niacin
5. Pantothenic Acid—Vitamin B5
5.1. Introduction and Properties
5.2. Sources of Pantothenic Acid
5.3. Physiological Function of Pantothenic Acid
5.4. Pharmacokinetics of Pantothenic Acid
5.4.1. Absorption
5.4.2. Distribution
5.4.3. Metabolism
5.4.4. Excretion
5.5. Pantothenic Acid Deficiency
5.5.1. Symptoms of Vitamin B5 Deficiency in Animals
5.5.2. The Symptoms of Vitamin B5 Deficiency in Human Subjects
5.5.3. Mutation of Pantothenate Kinase 2
5.6. Pharmacological Use of Pantothenic Acid
5.6.1. Triacylglycerols, Cholesterol
5.6.2. Cystinosis
5.6.3. Skin Disorders
Acne
Topical Treatment (Eyes, Nose Mucosa, Skin)
5.7. Toxicity of Pantothenic Acid
5.7.1. Acute Toxicity
5.7.2. Chronic Toxicity
6. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Spedding, S. Vitamins are more Funky than Casimir thought. Australas. Med. J. 2013, 6, 104–106. [Google Scholar] [CrossRef]
- Tylicki, A.; Lotowski, Z.; Siemieniuk, M.; Ratkiewicz, A. Thiamine and selected thiamine antivitamins-biological activity and methods of synthesis. Biosci. Rep. 2018, 38, 1–23. [Google Scholar] [CrossRef]
- Goodman, L.S.; Brunton, L.L.; Chabner, B.; Knollmann, B.R.C. Goodman & Gilman’s Pharmacological Basis of Therapeutics; McGraw-Hill: New York, NY, USA, 2011. [Google Scholar]
- Brown, G. Defects of thiamine transport and metabolism. J. Inherit. Metab. Dis. 2014, 37, 577–585. [Google Scholar] [CrossRef]
- Manzetti, S.; Zhang, J.; Van der Spoel, D. Thiamin function, metabolism, uptake, and transport. Biochemistry 2014, 53, 821–835. [Google Scholar] [CrossRef]
- Lonsdale, D. A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives. Evid. Based Complement. Altern. Med. 2006, 3, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Bettendorff, L.; Wirtzfeld, B.; Makarchikov, A.F.; Mazzucchelli, G.; Frederich, M.; Gigliobianco, T.; Gangolf, M.; De Pauw, E.; Angenot, L.; Wins, P. Discovery of a natural thiamine adenine nucleotide. Nat. Chem. Biol. 2007, 3, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Jurgenson, C.T.; Begley, T.P.; Ealick, S.E. The structural and biochemical foundations of thiamin biosynthesis. Annu. Rev. Biochem. 2009, 78, 569–603. [Google Scholar] [CrossRef] [PubMed]
- Bocobza, S.E.; Aharoni, A. Switching the light on plant riboswitches. Trends Plant Sci. 2008, 13, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Wang, H.; Xie, J. Thiamin (vitamin B1) biosynthesis and regulation: A rich source of antimicrobial drug targets? Int. J. Biol. Sci. 2011, 7, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Wolak, N.; Zawrotniak, M.; Gogol, M.; Kozik, A.; Rapala-Kozik, M. Vitamins B1, B2, B3 and B9-Occurrence, Biosynthesis Pathways and Functions in Human Nutrition. Mini Rev. Med. Chem. 2017, 17, 1075–1111. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B.; Chapman, L.M. The importance of thiamine (vitamin B1) in plant health: From crop yield to biofortification. J. Biol. Chem. 2020, 295, 12002–12013. [Google Scholar] [CrossRef]
- Ejsmond, M.J.; Blackburn, N.; Fridolfsson, E.; Haecky, P.; Andersson, A.; Casini, M.; Belgrano, A.; Hylander, S. Modeling vitamin B1 transfer to consumers in the aquatic food web. Sci. Rep. 2019, 9, 10045. [Google Scholar] [CrossRef] [PubMed]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef]
- Fattal-Valevski, A. Thiamine (Vitamin B1). J. Evid. Based Integr. Med. 2011, 16, 12–20. [Google Scholar] [CrossRef]
- Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Dietary reference values for thiamin. EFSA J. 2016, 14, e04653. [Google Scholar] [CrossRef]
- Chawla, J.; Kvarnberg, D. Hydrosoluble Vitamins; Elsevier B.V.: Amsterdam, The Netherlands, 2014; Volume 120, pp. 891–914. [Google Scholar]
- O’Connor, A. An overview of the role of bread in the UK diet. Nutr. Bull. 2012, 37, 193–212. [Google Scholar] [CrossRef]
- Lockyer, S.; Spiro, A. The role of bread in the UK diet: An update. Nutr. Bull. 2020, 45, 133–164. [Google Scholar] [CrossRef]
- Bonku, R.; Yu, J.M. Health aspects of peanuts as an outcome of its chemical composition. Food Sci. Hum. Wellness 2020, 9, 21–30. [Google Scholar] [CrossRef]
- Stuetz, W.; Schlormann, W.; Glei, M. B-vitamins, carotenoids and alpha-/gamma-tocopherol in raw and roasted nuts. Food Chem. 2017, 221, 222–227. [Google Scholar] [CrossRef]
- Prinzo, Z.W. Thiamine Deficiency and Its Prevention and Control in Major Emergencies; Department of Nutrition for Health and Development, World Health Organisation: Geneva, Switzerland, 1999. [Google Scholar]
- Whitfield, K.C.; Bourassa, M.W.; Adamolekun, B.; Bergeron, G.; Bettendorff, L.; Brown, K.H.; Cox, L.; Fattal-Valevski, A.; Fischer, P.R.; Frank, E.L.; et al. Thiamine deficiency disorders: Diagnosis, prevalence, and a roadmap for global control programs. Ann. N. Y. Acad. Sci. 2018, 1430, 3–43. [Google Scholar] [CrossRef]
- Pacei, F.; Tesone, A.; Laudi, N.; Laudi, E.; Cretti, A.; Pnini, S.; Varesco, F.; Colombo, C. The Relevance of Thiamine Evaluation in a Practical Setting. Nutrients 2020, 12, 2810. [Google Scholar] [CrossRef] [PubMed]
- Panijpan, B.; Ratanaubolchai, K. Kinetics of thiamine-polyphenol interactions and mechanism of thiamine disulphide formation. Int. J. Vitam. Nutr. Res. 1980, 50, 247–253. [Google Scholar] [PubMed]
- Dhir, S.; Tarasenko, M.; Napoli, E.; Giulivi, C. Neurological, Psychiatric, and Biochemical Aspects of Thiamine Deficiency in Children and Adults. Front. Psychiatry 2019, 10, 207. [Google Scholar] [CrossRef] [PubMed]
- Hilker, D.M.; Somogyi, J.C. Antithiamins of plant origin: Their chemical nature and mode of action. Ann. N. Y. Acad. Sci. 1982, 378, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Frank, L.L. Thiamin in Clinical Practice. JPEN J. Parenter Enter. Nutr. 2015, 39, 503–520. [Google Scholar] [CrossRef]
- Vimokesant, S.; Kunjara, S.; Rungruangsak, K.; Nakornchai, S.; Panijpan, B. Beriberi caused by antithiamin factors in food and its prevention. Ann. N. Y. Acad. Sci. 1982, 378, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Fabre, B.; Geay, B.; Beaufils, P. Thiaminase activity in Equisetum arvense and its extracts. Plant Méd. Phytothér. 1993, 26, 190–197. [Google Scholar]
- Yang, P.F.; Pratt, D.E. Antithiamin Activity of Polyphenolic Antioxidants. J. Food Sci. 1984, 49, 489–492. [Google Scholar] [CrossRef]
- Sannino, D.; Angert, E.R. Genomic insights into the thiamin metabolism of Paenibacillus thiaminolyticus NRRL B-4156 and P. apiarius NRRL B-23460. Stand. Genom. Sci. 2017, 12, 59. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.S.; Kies, C. Niacin, thiamin, iron and protein status of humans as affected by the consumption of tea (Camellia sinensis) infusions. Plant Foods Hum. Nutr. 1991, 41, 337–353. [Google Scholar] [CrossRef] [PubMed]
- Nishimune, T.; Watanabe, Y.; Okazaki, H.; Akai, H. Thiamin is decomposed due to Anaphe spp. entomophagy in seasonal ataxia patients in Nigeria. J. Nutr. 2000, 130, 1625–1628. [Google Scholar] [CrossRef] [PubMed]
- Ringe, H.; Schuelke, M.; Weber, S.; Dorner, B.G.; Kirchner, S.; Dorner, M.B. Infant botulism: Is there an association with thiamine deficiency? Pediatrics 2014, 134, e1436–e1440. [Google Scholar] [CrossRef] [PubMed]
- Taungbodhitham, A.K. Thiamin Content and Activity of Antithiamin Factor in Vegetables of Southern Thailand. Food Chem. 1995, 52, 285–288. [Google Scholar] [CrossRef]
- Somogyi, J.C. On antithiamine factors of fern. J. Vitam. 1971, 17, 165–174. [Google Scholar] [CrossRef][Green Version]
- Murata, K.; Tanaka, R.; Yamaoka, M. Reaction mechanisms of thiamine with thermostable factors. J. Nutr. Sci. Vitam. 1974, 20, 351–362. [Google Scholar] [CrossRef][Green Version]
- Rungruangsak, K.; Tosukhowong, P.; Panijpan, B.; Vimokesant, S.L. Chemical interactions between thiamin and tannic acid. I. Kinetics, oxygen dependence and inhibition by ascorbic acid. Am. J. Clin. Nutr. 1977, 30, 1680–1685. [Google Scholar] [CrossRef] [PubMed]
- Wills, R.B.H.; McBrien, K.J. Antithiamin activity of tea fractions. Food Chem. 1980, 6, 111–114. [Google Scholar] [CrossRef]
- Somogyi, J.C.; Bonicke, R. Connection between chemical structure and antithiamine activity of various phenol derivatives. Bibl. Nutr. Dieta 1970, 15, 180. [Google Scholar] [PubMed]
- Somogyi, J.C.; Nageli, U. Antithiamine effect of coffee. Int. J. Vitam. Nutr. Res. 1976, 46, 149–153. [Google Scholar]
- Hilker, D.M. Antithiamine factors in blueberries. Int. Z. Vitam. 1968, 38, 387–391. [Google Scholar]
- Schaller, K.; Holler, H. Thiamine absorption in the rat. IV. Effects of caffeic acid (3,4-dihydroxycinnamic acid) upon absorption and active transport of thiamine. Int. J. Vitam. Nutr. Res. 1976, 46, 143–148. [Google Scholar] [PubMed]
- Beruter, J.; Somogyi, J.C. 3,4-Dihydroxycinnamic acid, an antithiamine factor of fern. Experientia 1967, 23, 996–997. [Google Scholar] [CrossRef]
- Horman, I.; Brambilla, E.; Stalder, R. Evidence against the reported antithiamine effect of caffeic and chlorogenic acids. Int. J. Vitam. Nutr. Res. 1981, 51, 385–390. [Google Scholar]
- Zhang, F.; Masania, J.; Anwar, A.; Xue, M.; Zehnder, D.; Kanji, H.; Rabbani, N.; Thornalley, P.J. The uremic toxin oxythiamine causes functional thiamine deficiency in end-stage renal disease by inhibiting transketolase activity. Kidney Int. 2016, 90, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Burns, A.; Gleadow, R.; Cliff, J.; Zacarias, A.; Cavagnaro, T. Cassava: The Drought, War and Famine Crop in a Changing World. Sustainability 2010, 2, 3572–3607. [Google Scholar] [CrossRef]
- Leichter, J.; Joslyn, M.A. Kinetics of thiamin cleavage by sulphite. Biochem. J. 1969, 113, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Vanier, N.L.; Paraginski, R.T.; Berrios, J.D.; Oliveira, L.D.; Elias, M.C. Thiamine content and technological quality properties of parboiled rice treated with sodium bisulfite: Benefits and food safety risk. J. Food Compos. Anal. 2015, 41, 98–103. [Google Scholar] [CrossRef]
- Ottaway, P.B. Stability of Vitamins during Food Processing and Storage; Woodhead Publishing Ltd.: Cambridge, UK, 2010; pp. 545–548, 553–556. [Google Scholar]
- Yagi, N.; Itokawa, Y. Cleavage of thiamine by chlorine in tap water. J. Nutr. Sci. Vitam. 1979, 25, 281–287. [Google Scholar] [CrossRef]
- Kimura, M.; Itokawa, Y.; Fujiwara, M. Cooking losses of thiamin in food and its nutritional significance. J. Nutr. Sci. Vitam. 1990, 36, S17–S24. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, B.K.; Arnold, R.G. Chemistry of thiamine degradation in food products and model systems: A review. J. Agric. Food Chem. 1973, 21, 54–60. [Google Scholar] [CrossRef]
- Kaplan Evlice, A.; Özkaya, H. Effects of wheat cultivar, cooking method, and bulgur type on nutritional quality characteristics of bulgur. J. Cereal Sci. 2020, 96, 103124. [Google Scholar] [CrossRef]
- Calinoiu, L.F.; Vodnar, D.C. Whole Grains and Phenolic Acids: A Review on Bioactivity, Functionality, Health Benefits and Bioavailability. Nutrients 2018, 10, 1615. [Google Scholar] [CrossRef] [PubMed]
- Oghbaei, M.; Prakash, J.; Yildiz, F. Effect of primary processing of cereals and legumes on its nutritional quality: A comprehensive review. Cogent. Food Agric. 2016, 2, 1136015. [Google Scholar] [CrossRef]
- Batifoulier, F.; Verny, M.A.; Chanliaud, E.; Remesy, C.; Demigne, C. Variability of B vitamin concentrations in wheat grain, milling fractions and bread products. Eur. J. Agron. 2006, 25, 163–169. [Google Scholar] [CrossRef]
- Létinois, U.; Moine, G.; Hohmann, H.P. 6. Vitamin B1(Thiamin). In Ullmann’s Encyclopedia of Industrial Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; pp. 1–22. [Google Scholar]
- Liu, K.L.; Zheng, J.B.; Chen, F.S. Relationships between degree of milling and loss of Vitamin B, minerals, and change in amino acid composition of brown rice. LWT Food Sci. Technol. 2017, 82, 429–436. [Google Scholar] [CrossRef]
- Tiozon, R.N.; Fernie, A.R.; Sreenivasulu, N. Meeting human dietary vitamin requirements in the staple rice via strategies of biofortification and post-harvest fortification. Trends Food Sci. Technol. 2021, 109, 65–82. [Google Scholar] [CrossRef]
- Suri, D.J.; Tanumihardjo, S.A. Effects of Different Processing Methods on the Micronutrient and Phytochemical Contents of Maize: From A to Z. Compr. Rev. Food Sci. Food Saf. 2016, 15, 912–926. [Google Scholar] [CrossRef]
- Gwirtz, J.A.; Garcia-Casal, M.N. Processing maize flour and corn meal food products. Ann. N. Y. Acad. Sci. 2014, 1312, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Voelker, A.L.; Miller, J.; Running, C.A.; Taylor, L.S.; Mauer, L.J. Chemical stability and reaction kinetics of two thiamine salts (thiamine mononitrate and thiamine chloride hydrochloride) in solution. Food Res. Int. 2018, 112, 443–456. [Google Scholar] [CrossRef]
- Rekha, P.N.; Singhal, S.; Pandit, A.B. A study on degradation kinetics of thiamine in red gram splits (Cajanus cajan L.). Food Chem. 2004, 85, 591–598. [Google Scholar] [CrossRef]
- European Food Safety Authority. Benfotiamine, thiamine monophosphate chloride and thiamine pyrophosphate chloride, as sources of vitamin B1 added for nutritional purposes to food supplements-Scientific Opinion of the Panel on Food Additives and Nutrient Sources added to Food (ANS). EFSA J. 2008, 6, 864. [Google Scholar] [CrossRef]
- Voelker, A.L.; Taylor, L.S.; Mauer, L.J. Chemical stability and reaction kinetics of thiamine mononitrate in the aqueous phase of bread dough. Food Res. Int. 2021, 140, 110084. [Google Scholar] [CrossRef]
- Dionísio, A.P.; Gomes, R.T.; Oetterer, M. Ionizing radiation effects on food vitamins: A review. Braz. Arch. Biol. Technol. 2009, 52, 1267–1278. [Google Scholar] [CrossRef]
- Godoy, H.T.; Amaya-Farfan, J.; Rodriguez-Amaya, D.B. Degradation of vitamins. In Chemical Changes During Processing and Storage of Foods; Rodriguez-Amaya, D.B., Amaya-Farfan, J., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 329–383. [Google Scholar]
- Bognár, A. Tables on Weight Yield of Food and Retention Factors of Food Constituents for the Calculation of Nutrient Composition of Cooked Foods (Dishes); Bundesforschungsanstalt für Ernährung: Karlsruhe, Germany, 2002. [Google Scholar]
- Öhrvik, V.; Carlsen, M.H.; Källman, A.; Martinsen, T.A. Improving Food Composition Data by Standardizing Calculation Methods; Nordisk Ministerråd: Copenhagen, Denmark, 2015; p. 56. [Google Scholar]
- USDA. USDA Table of Nutrient Retention Factors. Available online: https://www.ars.usda.gov/ARSUserFiles/80400525/Data/retn/retn06.pdf (accessed on 10 July 2021).
- Bell, S.; Becker, W.; Vásquez-Caicedo, A.; Hartmann, B.; Møller, A.; Butriss, J. Report on Nutrient Losses and Gains Factors Used in European Food Composition Databases; Federal Research Centre for Nutrition and Food: Karlsruhe, Germany, 2006. [Google Scholar]
- Lešková, E.; Kubíková, J.; Kováčiková, E.; Košická, M.; Porubská, J.; Holčíková, K. Vitamin losses: Retention during heat treatment and continual changes expressed by mathematical models. J. Food Compos. Anal. 2006, 19, 252–276. [Google Scholar] [CrossRef]
- Kumar, S.; Aalbersberg, B. Nutrient retention in foods after earth-oven cooking compared to other forms of domestic cooking-2. Vitamins. J. Food Compos. Anal. 2006, 19, 311–320. [Google Scholar] [CrossRef]
- Aktas-Akyildiz, E.; Koksel, H. Minimisation of vitamin losses in fortified cookies by response surface methodology and validation of the determination methods. Eur. Food Res. Technol. 2021, 247, 1345–1354. [Google Scholar] [CrossRef]
- Fillion, L.; Henry, C.J. Nutrient losses and gains during frying: A review. Int. J. Food Sci. Nutr. 1998, 49, 157–168. [Google Scholar] [CrossRef]
- Lombardi-Boccia, G.; Lanzi, S.; Aguzzi, A. Aspects of meat quality: Trace elements and B vitamins in raw and cooked meats. J. Food Compos. Anal. 2005, 18, 39–46. [Google Scholar] [CrossRef]
- Bognar, A. Comparative study of frying to other cooking techniques influence on the nutritive value. Grasas Aceites 1998, 49, 250–260. [Google Scholar] [CrossRef]
- Silveira, C.M.; Moreira, A.V.; Martino, H.S.; Gomide, R.S.; Pinheiro, S.S.; Della Lucia, C.M.; Pinheiro-Sant’ana, H.M. Effect of cooking methods on the stability of thiamin and folic acid in fortified rice. Int. J. Food Sci. Nutr. 2017, 68, 179–187. [Google Scholar] [CrossRef]
- Jaworska, G.; Bernas, E. The effect of preliminary processing and period of storage on the quality of frozen Boletus edulis (Bull: Fr.) mushrooms. Food Chem. 2009, 113, 936–943. [Google Scholar] [CrossRef]
- Liu, K.; Zheng, J.; Wang, X.; Chen, F. Effects of household cooking processes on mineral, vitamin B, and phytic acid contents and mineral bioaccessibility in rice. Food Chem. 2019, 280, 59–64. [Google Scholar] [CrossRef]
- Szymandera-Buszka, K.; Piechocka, J.; Zaremba, A.; Przeor, M.; Jedrusek-Golinska, A. Pumpkin, Cauliflower and Broccoli as New Carriers of Thiamine Compounds for Food Fortification. Foods 2021, 10, 578. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, M.; Açkurt, F.; Yildiz, M.; Biringen, G.; Gürcan, T.; Löker, M. Effect of roasting on some nutrients of hazelnuts (Corylus avellena L.). Food Chem. 2001, 73, 185–190. [Google Scholar] [CrossRef]
- Pinheiro-Sant’Ana, H.M.; Penteado, M.; Brandão, S.; Stringheta, P. Stability of B-vitamin in meats prepared by foodservice. 1. Thiamin. Foodserv. Res. Int. 1999, 11, 33–52. [Google Scholar]
- Williams, P.G. Vitamin retention in cook/chill and cook/hot-hold hospital food-services. J. Am. Diet Assoc. 1996, 96, 490–498. [Google Scholar] [CrossRef]
- Ryley, J.; Kajda, P. Vitamins in Thermal-Processing. Food Chem. 1994, 49, 119–129. [Google Scholar] [CrossRef]
- Hill, M.A. Vitamin Retention in Microwave Cooking and Cook-Chill Foods. Food Chem. 1994, 49, 131–136. [Google Scholar] [CrossRef]
- Severi, S.; Bedogni, G.; Manzieri, A.M.; Poli, M.; Battistini, N. Effects of cooking and storage methods on the micronutrient content of foods. Eur. J. Cancer Prev. 1997, 6, S21–S24. [Google Scholar] [CrossRef] [PubMed]
- Hubner, F.; Arendt, E.K. Germination of cereal grains as a way to improve the nutritional value: A review. Crit. Rev. Food Sci. Nutr. 2013, 53, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Freitag, S.; Verrall, S.R.; Pont, S.D.A.; McRae, D.; Sungurtas, J.A.; Palau, R.; Hawes, C.; Alexander, C.J.; Allwood, J.W.; Foito, A.; et al. Impact of Conventional and Integrated Management Systems on the Water-Soluble Vitamin Content in Potatoes, Field Beans, and Cereals. J. Agric. Food Chem. 2018, 66, 831–841. [Google Scholar] [CrossRef] [PubMed]
- Titcomb, T.J.; Tanumihardjo, S.A. Global Concerns with B Vitamin Statuses: Biofortification, Fortification, Hidden Hunger, Interactions, and Toxicity. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1968–1984. [Google Scholar] [CrossRef] [PubMed]
- FAO. Sorghum and millets in human nutrition. In FAO Food and Nutrition Series; FAO: Rome, Italy, 1995; pp. 52, 121–124. [Google Scholar]
- Malleshi, N.G.; Klopfenstein, C.E. Nutrient composition, amino acid and vitamin contents of malted sorghum, pearl millet, finger millet and their rootlets. Int. J. Food Sci. Technol. 1998, 49, 415–422. [Google Scholar] [CrossRef]
- Pinheiro, S.S.; Anunciacao, P.C.; Cardoso, L.M.; Della Lucia, C.M.; de Carvalho, C.W.P.; Queiroz, V.A.V.; Pinheiro Sant’Ana, H.M. Stability of B vitamins, vitamin E, xanthophylls and flavonoids during germination and maceration of sorghum (Sorghum bicolor L.). Food Chem. 2021, 345, 128775. [Google Scholar] [CrossRef]
- Prodanov, M.; Sierra, I.; Vidal-Valverde, C. Effect of germination on the thiamine, riboflavin and niacin contents in legumes. Eur. Food Res. Technol. 1997, 205, 48–52. [Google Scholar] [CrossRef]
- Frias, J.; Prodanov, M.; Sierra, I.; Vidal-Valverde, C. Effect of Light on Carbohydrates and Hydrosoluble Vitamins of Lentils during Soaking. J. Food Prot. 1995, 58, 692–695. [Google Scholar] [CrossRef] [PubMed]
- Roe, M.; Church, S.; Pinchen, H.; Finglas, P. Nutrient Analysis of Fruit and Vegetables; Analytical Report; Institute of Food Research: Norwich, UK, 2013; pp. 17–76. [Google Scholar]
- Garg, M.; Sharma, A.; Vats, S.; Tiwari, V.; Kumari, A.; Mishra, V.; Krishania, M. Vitamins in Cereals: A Critical Review of Content, Health Effects, Processing Losses, Bioaccessibility, Fortification, and Biofortification Strategies for Their Improvement. Front. Nutr. 2021, 8, 586815. [Google Scholar] [CrossRef]
- Maskova, E.R.; Fiedlerova, V.; Holasova, M. Vitamin and mineral retention in meat in various cooking methods. Czech J. Food Sci. 1994, 12, 407–416. [Google Scholar]
- USDA. USDA Food Composition Databases. Available online: https://fdc.nal.usda.gov/ (accessed on 12 June 2021).
- Roe, M.; Church, S.; Pinchen, H.; Finglas, P. Nutrient Analysis of Fish and Fish Products; Analytical Report; Institute of Food Research: Norwich, UK, 2013; pp. 14–69. [Google Scholar]
- Mattila, P.; Konko, K.; Eurola, M.; Pihlava, J.M.; Astola, J.; Vahteristo, L.; Hietaniemi, V.; Kumpulainen, J.; Valtonen, M.; Piironen, V. Contents of vitamins, mineral elements, and some phenolic compounds in cultivated mushrooms. J. Agric. Food Chem. 2001, 49, 2343–2348. [Google Scholar] [CrossRef]
- Sałata, A.; Lemieszek, M.; Parzymies, M. The Nutritional and Health Properties of an Oyster Mushroom (Pleurotus ostreatus (Jacq. Fr) P. Kumm.). Acta Sci. Pol. Hortorum Cultus 2018, 17, 185–197. [Google Scholar] [CrossRef]
- Bernaś, E.; Jaworska, G. Vitamins profile as an indicator of the quality of frozen Agaricus bisporus mushrooms. J. Food Compos. Anal. 2016, 49, 1–8. [Google Scholar] [CrossRef]
- Hashemi Gahruie, H.; Eskandari, M.H.; Mesbahi, G.; Hanifpour, M.A. Scientific and technical aspects of yogurt fortification: A review. Food Sci. Hum. Wellness 2015, 4, 1–8. [Google Scholar] [CrossRef]
- Roe, M.; Church, S.; Pinchen, H.; Finglas, P. Nutrient Analysis of Eggs; Analytical Report; Institute of Food Research: Norwich, UK, 2013; pp. 1–44. [Google Scholar]
- Awonorin, S.O.; Rotimi, D.K. Effects of oven temperature and time on the losses of some B vitamins in roasted beef and pork. Foodserv. Res. Int. 1991, 6, 89–105. [Google Scholar] [CrossRef]
- Kyritsi, A.; Tzia, C.; Karathanos, V.T. Vitamin fortified rice grain using spraying and soaking methods. LWT Food Sci. Technol. 2011, 44, 312–320. [Google Scholar] [CrossRef]
- Atungulu, G.G.; Pan, Z. Rice industrial processing worldwide and impact on macro- and micronutrient content, stability, and retention. Ann. N. Y. Acad. Sci. 2014, 1324, 15–28. [Google Scholar] [CrossRef]
- Rumm-Kreuter, D.; Demmel, I. Comparison of vitamin losses in vegetables due to various cooking methods. J. Nutr. Sci. Vitaminol. 1990, 36, S7–S15. [Google Scholar] [CrossRef]
- Díaz-Gómez, J.; Twyman, R.M.; Zhu, C.; Farré, G.; Serrano, J.C.; Portero-Otin, M.; Muñoz, P.; Sandmann, G.; Capell, T.; Christou, P. Biofortification of crops with nutrients: Factors affecting utilization and storage. Curr. Opin. Biotechnol. 2017, 44, 115–123. [Google Scholar] [CrossRef]
- Prodanov, M.; Sierra, I.; Vidal-Valverde, C. Influence of soaking and cooking on the thiamin, riboflavin and niacin contents of legumes. Food Chem. 2004, 84, 271–277. [Google Scholar] [CrossRef]
- Batifoulier, F.; Verny, M.A.; Chanliaud, E.; Remesy, C.; Demigne, C. Effect of different breadmaking methods on thiamine, riboflavin and pyridoxine contents of wheat bread. J. Cereal Sci. 2005, 42, 101–108. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Michalska, A.; Frias, J.; Piskula, M.K.; Vidal-Valverde, C.; Zielinski, H. Effect of flour extraction rate and baking on thiamine and riboflavin content and antioxidant capacity of traditional rye bread. J. Food Sci. 2009, 74, C49–C55. [Google Scholar] [CrossRef] [PubMed]
- Haddad, G.S.; Loewenstein, M. Effect of several heat treatments and frozen storage on thiamine, riboflavin, and ascorbic acid content of milk. J. Dairy Sci. 1983, 66, 1601–1606. [Google Scholar] [CrossRef]
- Graham, D.M. Alteration of nutritive value resulting from processing and fortification of milk and milk products. J. Dairy Sci. 1974, 57, 738–745. [Google Scholar] [CrossRef]
- Lima, H.; Vogel, K.; Wagner-Gillespie, M.; Wimer, C.; Dean, L.; Fogleman, A. Nutritional Comparison of Raw, Holder Pasteurized, and Shelf-stable Human Milk Products. J. Pediatr. Gastroenterol. Nutr. 2018, 67, 649–653. [Google Scholar] [CrossRef]
- Athar, N.; Hardacre, A.; Taylor, G.; Clark, S.; Harding, R.; McLaughlin, J. Vitamin retention in extruded food products. J. Food Compos. Anal. 2006, 19, 379–383. [Google Scholar] [CrossRef]
- Riaz, M.N.; Asif, M.; Ali, R. Stability of vitamins during extrusion. Crit. Rev. Food Sci. Nutr. 2009, 49, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Aylangan, A.; Ic, E.; Ozyardimci, B. Investigation of gamma irradiation and storage period effects on the nutritional and sensory quality of chickpeas, kidney beans and green lentils. Food Control 2017, 80, 428–434. [Google Scholar] [CrossRef]
- Fox, J.B.; Thayer, D.W.; Jenkins, R.K.; Phillips, J.G.; Ackerman, S.A.; Beecher, G.R.; Holden, J.M.; Morrow, F.D.; Quirbach, D.M. Effect of gamma irradiation on the B vitamins of pork chops and chicken breasts. Int. J. Radiat. Biol. 1989, 55, 689–703. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.B.; Lakritz, L.; Hampson, J.R.; Ward, K.; Thayer, D.W. Gamma Irradiation Effects on Thiamin and Riboflavin in Beef, Lamb, Pork, and Turkey. J. Food Sci. 1995, 60, 596–598. [Google Scholar] [CrossRef]
- Woodside, J. Nutritional aspects of irradiated food. Stewart Postharvest Rev. 2015, 11, 1–6. [Google Scholar] [CrossRef]
- Greenwood, D.A.; Kraybill, H.R.; Feaster, J.F.; Jackson, J.M. Vitamin Retention in Processed Meat. Ind. Eng. Chem. 1944, 36, 922–927. [Google Scholar] [CrossRef]
- Rickman, J.C.; Barrett, D.M.; Bruhn, C.M. Nutritional comparison of fresh, frozen and canned fruits and vegetables. Part 1. Vitamins C and B and phenolic compounds. J. Sci. Food Agric. 2007, 87, 930–944. [Google Scholar] [CrossRef]
- Martín-Belloso, O.; Llanos-Barriobero, E. Proximate composition, minerals and vitamins in selected canned vegetables. Eur. Food Res. Technol. 2001, 212, 182–187. [Google Scholar] [CrossRef]
- Marçal, S.; Sousa, A.S.; Taofiq, O.; Antunes, F.; Morais, A.M.; Freitas, A.C.; Barros, L.; Ferreira, I.C.; Pintado, M. Impact of postharvest preservation methods on nutritional value and bioactive properties of mushrooms. Trends Food Sci. Technol. 2021, 110, 418–431. [Google Scholar] [CrossRef]
- Coad, R.; Bui, L. Stability of Vitamins B1, B2, B6 and E in a Fortified Military Freeze-Dried Meal During Extended Storage. Foods 2020, 9, 39. [Google Scholar] [CrossRef] [PubMed]
- Ayhan, D.K.; Koksel, H. Investigation of the effect of different storage conditions on vitamin content of enriched pasta product. Qual. Assur. Saf. Crops 2019, 11, 701–712. [Google Scholar] [CrossRef]
- Walker, G.J. The nutritional value of processed foods. CSIRO Food Proc. 1979, 4. [Google Scholar] [CrossRef]
- Gan, R.Y.; Lui, W.Y.; Wu, K.; Chan, C.L.; Dai, S.H.; Sui, Z.Q.; Corke, H. Bioactive compounds and bioactivities of germinated edible seeds and sprouts: An updated review. Trends Food Sci. Technol. 2017, 59, 1–14. [Google Scholar] [CrossRef]
- Lemmens, E.; Moroni, A.V.; Pagand, J.; Heirbaut, P.; Ritala, A.; Karlen, Y.; Le, K.A.; Van den Broeck, H.C.; Brouns, F.; De Brier, N.; et al. Impact of Cereal Seed Sprouting on Its Nutritional and Technological Properties: A Critical Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 305–328. [Google Scholar] [CrossRef]
- Lonsdale, D. Thiamine tetrahydrofurfuryl disulfide: A little known therapeutic agent. Med. Sci. Monit. 2004, 10, RA199–RA203. [Google Scholar]
- Fujiwara, M.; Watanabe, H.; Matsui, K. “Allithiamine” a Newly Found Derivative of Vitamin B1. J. Biochem. 1954, 41, 29–39. [Google Scholar] [CrossRef]
- Fujiwara, M. Allithiamine and its properties. J. Nutr. Sci. Vitam. 1976, 22, S57–S62. [Google Scholar] [CrossRef]
- Matsukawa, T.; Kawasaki, H.; Iwatsu, T.; Yurugi, S. Syntheses of allithiamine and its homologues. J. Vitam. 1954, 1, 13–26. [Google Scholar] [CrossRef]
- Miah, M.A.K.; Haque, A.; Douglass, M.P.; Clarke, B. Parboiling of rice. Part II: Effect of hot soaking time on the degree of starch gelatinization. Int. J. Food Sci. Technol. 2002, 37, 539–545. [Google Scholar] [CrossRef]
- Ituen, E.; Ukpakha, A. Improved method of par-boiling paddy for better quality rice. World J. Appl. Sci. Technol. 2011, 3, 31–40. [Google Scholar]
- Oli, P.; Ward, R.; Adhikari, B.; Torley, P. Parboiled rice: Understanding from a materials science approach. J. Food Eng. 2014, 124, 173–183. [Google Scholar] [CrossRef]
- Hinton, J.J. Parboiling treatment of rice. Nature 1948, 162, 913–915. [Google Scholar] [CrossRef] [PubMed]
- Villota, S.M.A.; Tuates, A.M., Jr.; Capariño, O.A. Cooking Qualilites and Nutritional Contents of Parboiled Milled Rice. Asian J. Appl. Sci. 2016, 4, 1172–1178. [Google Scholar]
- Manful, J.; Swetman, A.; Coker, R.; Drunis, A. Changes in the thiamine and riboflavin contents of rice during artisanal parboiling in Ghana. Trop. Sci. 2007, 47, 211–217. [Google Scholar] [CrossRef]
- Padua, A.B.; Juliano, B.O. Effect of parboiling on thiamin, protein and fat of rice. J. Sci. Food Agric. 1974, 25, 697–701. [Google Scholar] [CrossRef]
- Cubadda, F.; Jackson, B.P.; Cottingham, K.L.; Van Horne, Y.O.; Kurzius-Spencer, M. Human exposure to dietary inorganic arsenic and other arsenic species: State of knowledge, gaps and uncertainties. Sci. Total Environ. 2017, 579, 1228–1239. [Google Scholar] [CrossRef]
- Davis, M.A.; Signes-Pastor, A.J.; Argos, M.; Slaughter, F.; Pendergrast, C.; Punshon, T.; Gossai, A.; Ahsan, H.; Karagas, M.R. Assessment of human dietary exposure to arsenic through rice. Sci. Total Environ. 2017, 586, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.X.; Williams, P.N.; Carey, A.M.; Zhu, Y.G.; Deacon, C.; Raab, A.; Feldmann, J.; Islam, R.M.; Meharg, A.A. Inorganic arsenic in rice bran and its products are an order of magnitude higher than in bulk grain. Environ. Sci. Technol. 2008, 42, 7542–7546. [Google Scholar] [CrossRef]
- Lombi, E.; Scheckel, K.G.; Pallon, J.; Carey, A.M.; Zhu, Y.G.; Meharg, A.A. Speciation and distribution of arsenic and localization of nutrients in rice grains. New Phytol. 2009, 184, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Meharg, A.A.; Lombi, E.; Williams, P.N.; Scheckel, K.G.; Feldmann, J.; Raab, A.; Zhu, Y.; Islam, R. Speciation and localization of arsenic in white and brown rice grains. Environ. Sci. Technol. 2008, 42, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Peng, B.; Tan, C.; Ma, L.; Rathinasabapathi, B. Recent advances in arsenic bioavailability, transport, and speciation in rice. Environ. Sci. Pollut. Res. Int. 2015, 22, 5742–5750. [Google Scholar] [CrossRef] [PubMed]
- USFDA, U.S.F.a.D. Arsenic in Rice and Rice Products Risk Assessment Report. Available online: http://www.fda.gov/Food/FoodScienceResearch/RiskSafetyAssessment/default.htm (accessed on 10 July 2021).
- Upadhyay, M.K.; Shukla, A.; Yadav, P.; Srivastava, S. A review of arsenic in crops, vegetables, animals and food products. Food Chem. 2019, 276, 608–618. [Google Scholar] [CrossRef]
- European Food Safety Authority; Arcella, D.; Cascio, C.; Gomez Ruiz, J.A. Chronic dietary exposure to inorganic arsenic. EFSA J. 2021, 19, 50. [Google Scholar] [CrossRef]
- EFSA. Dietary exposure to inorganic arsenic in the European population. EFSA J. 2014, 12, 3597. [Google Scholar] [CrossRef]
- Rasheed, H.; Kay, P.; Slack, R.; Gong, Y.Y. Arsenic species in wheat, raw and cooked rice: Exposure and associated health implications. Sci. Total Environ. 2018, 634, 366–373. [Google Scholar] [CrossRef]
- Lai, P.Y.; Cottingham, K.L.; Steinmaus, C.; Karagas, M.R.; Miller, M.D. Arsenic and Rice: Translating Research to Address Health Care Providers’ Needs. J. Pediatr. 2015, 167, 797–803. [Google Scholar] [CrossRef]
- Islam, S.; Rahman, M.M.; Rahman, M.A.; Naidu, R. Inorganic arsenic in rice and rice-based diets: Health risk assessment. Food Control 2017, 82, 196–202. [Google Scholar] [CrossRef]
- Mwale, T.; Rahman, M.M.; Mondal, D. Risk and Benefit of Different Cooking Methods on Essential Elements and Arsenic in Rice. Int. J. Environ. Res. Public Health 2018, 15, 1056. [Google Scholar] [CrossRef] [PubMed]
- Nachman, K.E.; Ginsberg, G.L.; Miller, M.D.; Murray, C.J.; Nigra, A.E.; Pendergrast, C.B. Mitigating dietary arsenic exposure: Current status in the United States and recommendations for an improved path forward. Sci. Total Environ. 2017, 581, 221–236. [Google Scholar] [CrossRef] [PubMed]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, International Agency for Research on Cancer, and World Health Organization. Arsenic, metals, fibres, and dusts. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100C, 41–93. [Google Scholar]
- EFSA. Scientific Opinion on Arsenic in Food. EFSA J. 2009, 7, 1351–1550. [Google Scholar] [CrossRef]
- Rahman, M.A.; Rahman, A.; Khan, M.Z.K.; Renzaho, A.M.N. Human health risks and socio-economic perspectives of arsenic exposure in Bangladesh: A scoping review. Ecotoxicol. Environ. Saf. 2018, 150, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Gray, P.J.; Conklin, S.D.; Todorov, T.I.; Kasko, S.M. Cooking rice in excess water reduces both arsenic and enriched vitamins in the cooked grain. Food Addit. Contam. Part A 2016, 33, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Kumarathilaka, P.; Seneweera, S.; Ok, Y.S.; Meharg, A.; Bundschuh, J. Arsenic in cooked rice foods: Assessing health risks and mitigation options. Environ. Int. 2019, 127, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Pedron, T.; Segura, F.R.; Paniz, F.P.; Souz, F.D.; dos Santos, M.C.; de Magalhaes, A.M.; Batista, B.L. Mitigation of arsenic in rice grains by polishing and washing: Evidencing the benefit and the cost. J. Cereal Sci. 2019, 87, 52–58. [Google Scholar] [CrossRef]
- Menon, M.; Dong, W.; Chen, X.; Hufton, J.; Rhodes, E.J. Improved rice cooking approach to maximise arsenic removal while preserving nutrient elements. Sci. Total Environ. 2021, 755, 143341. [Google Scholar] [CrossRef]
- Naito, S.; Matsumoto, E.; Shindoh, K.; Nishimura, T. Effects of polishing, cooking, and storing on total arsenic and arsenic species concentrations in rice cultivated in Japan. Food Chem. 2015, 168, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Atiaga, O.; Nunes, L.M.; Otero, X.L. Effect of cooking on arsenic concentration in rice. Environ. Sci. Pollut. Res. Int. 2020, 27, 10757–10765. [Google Scholar] [CrossRef] [PubMed]
- Raab, A.; Baskaran, C.; Feldmann, J.; Meharg, A.A. Cooking rice in a high water to rice ratio reduces inorganic arsenic content. J. Environ. Monit. 2009, 11, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Eggersdorfer, M.; Laudert, D.; Letinois, U.; McClymont, T.; Medlock, J.; Netscher, T.; Bonrath, W. One hundred years of vitamins-a success story of the natural sciences. Angew. Chem. Int. Ed. Engl. 2012, 51, 12960–12990. [Google Scholar] [CrossRef]
- Acevedo-Rocha, C.G.; Gronenberg, L.S.; Mack, M.; Commichau, F.M.; Genee, H.J. Microbial cell factories for the sustainable manufacturing of B vitamins. Curr. Opin. Biotechnol. 2019, 56, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, T.B.; Basset, G.J.; Borel, P.; Carrari, F.; DellaPenna, D.; Fraser, P.D.; Hellmann, H.; Osorio, S.; Rothan, C.; Valpuesta, V.; et al. Vitamin deficiencies in humans: Can plant science help? Plant Cell 2012, 24, 395–414. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L., 3rd; Keast, D.R.; Bailey, R.L.; Dwyer, J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J. Nutr. 2011, 141, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Liberato, S.C.; Pinheiro-Sant’Ana, H.M. Fortification of industrialized foods with vitamins. Rev. Nutr. 2006, 19, 215–231. [Google Scholar] [CrossRef]
- Berner, L.A.; Keast, D.R.; Bailey, R.L.; Dwyer, J.T. Fortified foods are major contributors to nutrient intakes in diets of US children and adolescents. J. Acad. Nutr. Diet 2014, 114, 1009–1022. [Google Scholar] [CrossRef]
- Whitfield, K.C.; Smith, T.J.; Rohner, F.; Wieringa, F.T.; Green, T.J. Thiamine fortification strategies in low- and middle-income settings: A review. Ann. N. Y. Acad. Sci. 2021, 1498, 29–45. [Google Scholar] [CrossRef]
- Allen, L.; Benoist, B.; Dary, O.; Hurrell, R. Guidelines on Food Fortification with Micronutrients; World Health Organization Food and Agriculture Organization United Nations: Geneva, Switzerland, 2006; pp. 67–71, 128. [Google Scholar]
- Newman, J.C.; Malek, A.M.; Hunt, K.J.; Marriott, B.P. Nutrients in the US Diet: Naturally Occurring or Enriched/Fortified Food and Beverage Sources, Plus Dietary Supplements: NHANES 2009–2012. J. Nutr. 2019, 149, 1404–1412. [Google Scholar] [CrossRef]
- EU Parliament. E. Regulation (EC) No 1925/2006 of the European Parliament and of the Council of 20 December 2006 on the addition of vitamins and minerals and of certain other substances to foods. OJ L 404. 2006, pp. 26–38. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32006R1925 (accessed on 10 July 2021).
- Gomes, F.; Bergeron, G.; Bourassa, M.W.; Fischer, P.R. Thiamine deficiency unrelated to alcohol consumption in high-income countries: A literature review. Ann. N. Y. Acad. Sci. 2021, 1498, 46–56. [Google Scholar] [CrossRef] [PubMed]
- GFD. Global Fortification Data Exchange. Available online: https://fortificationdata.org/ (accessed on 1 June 2021).
- FFI. Food Fortification Initiative. Available online: https://www.ffinetwork.org/country-profiles (accessed on 12 July 2021).
- De Pee, S. Proposing nutrients and nutrient levels for rice fortification. Ann. N. Y. Acad. Sci. 2014, 1324, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Roy, A. Whole grain rice fortification as a solution to micronutrient deficiency: Technologies and need for more viable alternatives. Food Chem. 2020, 326, 127049. [Google Scholar] [CrossRef]
- Steiger, G.; Muller-Fischer, N.; Cori, H.; Conde-Petit, B. Fortification of rice: Technologies and nutrients. Ann. N. Y. Acad. Sci. 2014, 1324, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Alavi, S.; Bugusu, B.; Cramer, G.; Dary, O.; Lee, T.-C.; Martin, L.; McEntire, J.; Wailes, E. Rice Fortification in Developing Countries: A Critical Review of the Technical and Economic Feasibility; Academy for Educational Development: Washington, DC, USA, 2008. [Google Scholar]
- Strobbe, S.; Van Der Straeten, D. Toward Eradication of B-Vitamin Deficiencies: Considerations for Crop Biofortification. Front. Plant Sci. 2018, 9, 443. [Google Scholar] [CrossRef] [PubMed]
- Minhas, A.P.; Tuli, R.; Puri, S. Pathway Editing Targets for Thiamine Biofortification in Rice Grains. Front. Plant Sci. 2018, 9, 975. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Thomas, N.; Ronald, P.C.; Goyer, A. Overexpression of thiamin biosynthesis genes in rice increases leaf and unpolished grain thiamin content but not resistance to Xanthomonas oryzae pv. oryzae. Front. Plant Sci. 2016, 7, 616. [Google Scholar] [CrossRef]
- Strobbe, S.; Verstraete, J.; Stove, C.; Van Der Straeten, D. Metabolic engineering of rice endosperm towards higher vitamin B1 accumulation. Plant Biotechnol. J. 2021, 19, 1253–1267. [Google Scholar] [CrossRef]
- Goyer, A. Thiamin biofortification of crops. Curr. Opin. Biotechnol. 2017, 44, 1–7. [Google Scholar] [CrossRef]
- Smithline, H.A.; Donnino, M.; Greenblatt, D.J. Pharmacokinetics of high-dose oral thiamine hydrochloride in healthy subjects. BMC Clin. Pharmacol. 2012, 12, 4. [Google Scholar] [CrossRef]
- Gangolf, M.; Czerniecki, J.; Radermecker, M.; Detry, O.; Nisolle, M.; Jouan, C.; Martin, D.; Chantraine, F.; Lakaye, B.; Wins, P.; et al. Thiamine status in humans and content of phosphorylated thiamine derivatives in biopsies and cultured cells. PLoS ONE 2010, 5, e13616. [Google Scholar] [CrossRef] [PubMed]
- Rindi, G.; Laforenza, U. Thiamine intestinal transport and related issues: Recent aspects. Proc. Soc. Exp. Biol. Med. 2000, 224, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Said, H.M.; Balamurugan, K.; Subramanian, V.S.; Marchant, J.S. Expression and functional contribution of hTHTR-2 in thiamin absorption in human intestine. Am. J. Physiol. Gastrointest. Liver. Physiol. 2004, 286, G491–G498. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, V.; Smith, S.B.; Prasad, P.D. SLC19: The folate/thiamine transporter family. Pflug. Arch. 2004, 447, 641–646. [Google Scholar] [CrossRef]
- Nabokina, S.M.; Said, H.M. A high-affinity and specific carrier-mediated mechanism for uptake of thiamine pyrophosphate by human colonic epithelial cells. Am. J. Physiol. Gastrointest. Liver. Physiol. 2012, 303, G389–G395. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.; Werneke, U. Wernicke’s encephalopathy-from basic science to clinical practice. Part 1: Understanding the role of thiamine. Adv. Ther. Psychopharmacol. 2020, 10, 2045125320978106. [Google Scholar] [CrossRef]
- Lu, J.; Frank, E.L. Rapid HPLC measurement of thiamine and its phosphate esters in whole blood. Clin. Chem. 2008, 54, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Labay, V.; Raz, T.; Baron, D.; Mandel, H.; Williams, H.; Barrett, T.; Szargel, R.; McDonald, L.; Shalata, A.; Nosaka, K.; et al. Mutations in SLC19A2 cause thiamine-responsive megaloblastic anaemia associated with diabetes mellitus and deafness. Nat. Genet. 1999, 22, 300–304. [Google Scholar] [CrossRef]
- Zhao, R.; Gao, F.; Goldman, I.D. Molecular cloning of human thiamin pyrophosphokinase. Biochim. Biophys. Acta 2001, 1517, 320–322. [Google Scholar] [CrossRef]
- Bettendorff, L. The compartmentation of phosphorylated thiamine derivatives in cultured neuroblastoma cells. Biochim. Biophys. Acta 1994, 1222, 7–14. [Google Scholar] [CrossRef]
- Eudy, J.D.; Spiegelstein, O.; Barber, R.C.; Wlodarczyk, B.J.; Talbot, J.; Finnell, R.H. Identification and characterization of the human and mouse SLC19A3 gene: A novel member of the reduced folate family of micronutrient transporter genes. Mol. Genet. Metab. 2000, 71, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Casteels, M.; Sniekers, M.; Fraccascia, P.; Mannaerts, G.P.; Van Veldhoven, P.P. The role of 2-hydroxyacyl-CoA lyase, a thiamin pyrophosphate-dependent enzyme, in the peroxisomal metabolism of 3-methyl-branched fatty acids and 2-hydroxy straight-chain fatty acids. Biochem. Soc. Trans. 2007, 35, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Ashokkumar, B.; Vaziri, N.D.; Said, H.M. Thiamin uptake by the human-derived renal epithelial (HEK-293) cells: Cellular and molecular mechanisms. Am. J. Physiol. Ren. Physiol. 2006, 291, F796–F805. [Google Scholar] [CrossRef]
- Patel, M.S.; Nemeria, N.S.; Furey, W.; Jordan, F. The pyruvate dehydrogenase complexes: Structure-based function and regulation. J. Biol. Chem. 2014, 289, 16615–16623. [Google Scholar] [CrossRef] [PubMed]
- Hutson, S.M.; Sweatt, A.J.; Lanoue, K.F. Branched-chain amino acid metabolism: Implications for establishing safe intakes. J. Nutr. 2005, 135, 1557S–1564S. [Google Scholar] [CrossRef]
- Sperringer, J.E.; Addington, A.; Hutson, S.M. Branched-Chain Amino Acids and Brain Metabolism. Neurochem. Res. 2017, 42, 1697–1709. [Google Scholar] [CrossRef]
- Schenk, G.; Duggleby, R.G.; Nixon, P.F. Properties and functions of the thiamin diphosphate dependent enzyme transketolase. Int. J. Biochem. Cell Biol. 1998, 30, 1297–1318. [Google Scholar] [CrossRef]
- Foulon, V.; Sniekers, M.; Huysmans, E.; Asselberghs, S.; Mahieu, V.; Mannaerts, G.P.; Van Veldhoven, P.P.; Casteels, M. Breakdown of 2-hydroxylated straight chain fatty acids via peroxisomal 2-hydroxyphytanoyl-CoA lyase: A revised pathway for the alpha-oxidation of straight chain fatty acids. J. Biol. Chem. 2005, 280, 9802–9812. [Google Scholar] [CrossRef]
- Lonsdale, D. Thiamin. Adv. Food. Nutr. Res. 2018, 83, 1–56. [Google Scholar] [CrossRef]
- Kelley, R.I.; Robinson, D.; Puffenberger, E.G.; Strauss, K.A.; Morton, D.H. Amish lethal microcephaly: A new metabolic disorder with severe congenital microcephaly and 2-ketoglutaric aciduria. Am. J. Med. Genet. 2002, 112, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Marce-Grau, A.; Marti-Sanchez, L.; Baide-Mairena, H.; Ortigoza-Escobar, J.D.; Perez-Duenas, B. Genetic defects of thiamine transport and metabolism: A review of clinical phenotypes, genetics, and functional studies. J. Inherit. Metab. Dis. 2019, 42, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Shible, A.A.; Ramadurai, D.; Gergen, D.; Reynolds, P.M. Dry Beriberi Due to Thiamine Deficiency Associated with Peripheral Neuropathy and Wernicke’s Encephalopathy Mimicking Guillain-Barre syndrome: A Case Report and Review of the Literature. Am. J. Case Rep. 2019, 20, 330–334. [Google Scholar] [CrossRef]
- Chisolm-Straker, M.; Cherkas, D. Altered and unstable: Wet beriberi, a clinical review. J. Emerg. Med. 2013, 45, 341–344. [Google Scholar] [CrossRef] [PubMed]
- DiNicolantonio, J.J.; Liu, J.; O’Keefe, J.H. Thiamine and Cardiovascular Disease: A Literature Review. Prog. Cardiovasc. Dis. 2018, 61, 27–32. [Google Scholar] [CrossRef]
- Greenspon, J.; Perrone, E.E.; Alaish, S.M. Shoshin beriberi mimicking central line sepsis in a child with short bowel syndrome. World J. Pediatr. 2010, 6, 366–368. [Google Scholar] [CrossRef]
- Dabar, G.; Harmouche, C.; Habr, B.; Riachi, M.; Jaber, B. Shoshin Beriberi in Critically-Ill patients: Case series. Nutr. J. 2015, 14, 51. [Google Scholar] [CrossRef]
- Fattal-Valevski, A.; Bloch-Mimouni, A.; Kivity, S.; Heyman, E.; Brezner, A.; Strausberg, R.; Inbar, D.; Kramer, U.; Goldberg-Stern, H. Epilepsy in children with infantile thiamine deficiency. Neurology 2009, 73, 828–833. [Google Scholar] [CrossRef]
- Nazir, M.; Lone, R.; Charoo, B.A. Infantile Thiamine Deficiency: New Insights into an Old Disease. Indian Pediatr. 2019, 56, 673–681. [Google Scholar] [CrossRef]
- Chandrakumar, A.; Bhardwaj, A.; Geert, W.; Jong, G.W. Review of thiamine deficiency disorders: Wernicke encephalopathy and Korsakoff psychosis. J. Basic Clin. Physiol. Pharmacol. 2018, 30, 153–162. [Google Scholar] [CrossRef]
- Butterworth, R.F. Thiamin deficiency and brain disorders. Nutr. Res. Rev. 2003, 16, 277–284. [Google Scholar] [CrossRef]
- Kopelman, M.D.; Thomson, A.D.; Guerrini, I.; Marshall, E.J. The Korsakoff syndrome: Clinical aspects, psychology and treatment. Alcohol Alcohol. 2009, 44, 148–154. [Google Scholar] [CrossRef]
- Arts, N.J.; Walvoort, S.J.; Kessels, R.P. Korsakoff’s syndrome: A critical review. Neuropsychiatr. Dis. Treat. 2017, 13, 2875–2890. [Google Scholar] [CrossRef] [PubMed]
- Kril, J.J.; Harper, C.G. Neuroanatomy and neuropathology associated with Korsakoff’s syndrome. Neuropsychol. Rev. 2012, 22, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Yates, A.A.; Schlicker, S.A.; Suitor, C.W. Dietary Reference Intakes: The new basis for recommendations for calcium and related nutrients, B vitamins, and choline. J. Am. Diet Assoc. 1998, 98, 699–706. [Google Scholar] [CrossRef]
- Armah, S.; Ferruzzi, M.G.; Gletsu-Miller, N. Feasibility of Mass-Spectrometry to Lower Cost and Blood Volume Requirements for Assessment of B Vitamins in Patients Undergoing Bariatric Surgery. Metabolites 2020, 10, 240. [Google Scholar] [CrossRef]
- Bishop, A.M.; Fernandez, C.; Whitehead, R.D., Jr.; Morales, A.P.; Barr, D.B.; Wilder, L.C.; Baker, S.E. Quantification of riboflavin in human urine using high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 1823–1826. [Google Scholar] [CrossRef]
- Diniz, M.; Dias, N.; Andrade, F.; Paulo, B.; Ferreira, A. Isotope dilution method for determination of vitamin B2 in human plasma using liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2019, 1113, 14–19. [Google Scholar] [CrossRef]
- Hampel, D.; York, E.R.; Allen, L.H. Ultra-performance liquid chromatography tandem mass-spectrometry (UPLC-MS/MS) for the rapid, simultaneous analysis of thiamin, riboflavin, flavin adenine dinucleotide, nicotinamide and pyridoxal in human milk. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2012, 903, 7–13. [Google Scholar] [CrossRef]
- Cheng, X.; Ma, D.; Fei, G.; Ma, Z.; Xiao, F.; Yu, Q.; Pan, X.; Zhou, F.; Zhao, L.; Zhong, C. A single-step method for simultaneous quantification of thiamine and its phosphate esters in whole blood sample by ultra-performance liquid chromatography-mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2018, 1095, 103–111. [Google Scholar] [CrossRef]
- Jeong Hyeon, M.; Shin Beom, S.; Shin, S. Liquid Chromatography-Tandem Mass Spectrometry Analysis of Riboflavin in Beagle Dog Plasma for Pharmacokinetic Studies. Mass Spectrom. Lett. 2020, 11, 10–14. [Google Scholar] [CrossRef]
- Kahoun, D.; Fojtíková, P.; Vácha, F.; Nováková, E.; Hypša, V. Development and validation of an LC-MS/MS method for determination of B vitamins and some its derivatives in whole blood. bioRxiv 2021. [Google Scholar] [CrossRef]
- Khaksari, M.; Mazzoleni, L.R.; Ruan, C.H.; Song, P.; Hershey, N.D.; Kennedy, R.T.; Burns, M.A.; Minerick, A.R. Detection and quantification of vitamins in microliter volumes of biological samples by LC-MS for clinical screening. Aiche J. 2018, 64, 3709–3718. [Google Scholar] [CrossRef]
- Meisser Redeuil, K.; Longet, K.; Benet, S.; Munari, C.; Campos-Gimenez, E. Simultaneous quantification of 21 water soluble vitamin circulating forms in human plasma by liquid chromatography-mass spectrometry. J. Chromatogr. A 2015, 1422, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.N.; Yin, S.A.; Yang, Z.Y.; Yang, X.G.; Shao, B.; Ren, Y.P.; Zhang, J. Application of UPLC-MS/MS Method for Analyzing B-vitamins in Human Milk. Biomed. Environ. Sci. 2015, 28, 738–750. [Google Scholar] [CrossRef] [PubMed]
- Roelofsen-de Beer, R.; Van Zelst, B.D.; Wardle, R.; Kooij, P.G.; de Rijke, Y.B. Simultaneous measurement of whole blood vitamin B1 and vitamin B6 using LC-ESI-MS/MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1063, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Verstraete, J.; Stove, C. Patient-Centric Assessment of Thiamine Status in Dried Blood Volumetric Absorptive Microsamples Using LC-MS/MS Analysis. Anal. Chem. 2021, 93, 2660–2668. [Google Scholar] [CrossRef]
- Zhang, Q.; Ford, L.A.; Goodman, K.D.; Freed, T.A.; Hauser, D.M.; Conner, J.K.; Vroom, K.E.; Toal, D.R. LC-MS/MS method for quantitation of seven biomarkers in human plasma for the assessment of insulin resistance and impaired glucose tolerance. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1038, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Gibson, R.A.; Green, T.J. Measuring thiamine status in dried blood spots. Clin. Chim. Acta 2020, 509, 52–59. [Google Scholar] [CrossRef]
- Jenčo, J.; Krčmová, L.K.; Sobotka, L.; Bláha, V.; Solich, P.; Švec, F. Development of novel liquid chromatography method for clinical monitoring of vitamin B1 metabolites and B6 status in the whole blood. Talanta 2020, 211, 120702. [Google Scholar] [CrossRef]
- Mathew, E.M.; Sakore, P.; Lewis, L.; Manokaran, K.; Rao, P.; Moorkoth, S. Development and validation of a dried blood spot test for thiamine deficiency among infants by HPLC-fluorimetry. Biomed. Chromatogr. 2019, 33, e4668. [Google Scholar] [CrossRef]
- Nguyen, V.L.; Darman, M.; Ireland, A.; Fitzpatrick, M. A high performance liquid chromatography fluorescence method for the analysis of both pyridoxal-5-phosphate and thiamine pyrophosphate in whole blood. Clin. Chim. Acta 2020, 506, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Petteys, B.J.; Frank, E.L. Rapid determination of vitamin B(2) (riboflavin) in plasma by HPLC. Clin. Chim. Acta 2011, 412, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Stuetz, W.; Carrara, V.I.; McGready, R.; Lee, S.J.; Biesalski, H.K.; Nosten, F.H. Thiamine diphosphate in whole blood, thiamine and thiamine monophosphate in breast-milk in a refugee population. PLoS ONE 2012, 7, e36280. [Google Scholar] [CrossRef]
- Heydari, R.; Elyasi, N.S. Ion-pair cloud-point extraction: A new method for the determination of water-soluble vitamins in plasma and urine. J. Sep. Sci. 2014, 37, 2724–2731. [Google Scholar] [CrossRef]
- Mandal, S.M.; Mandal, M.; Ghosh, A.K.; Dey, S. Rapid determination of vitamin B2 and B12 in human urine by isocratic liquid chromatography. Anal. Chim. Acta 2009, 640, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Asgharian Marzabad, M.; Jafari, B.; Norouzi, P. Determination of Riboflavin by Nanocomposite Modified Carbon Paste Electrode in Biological Fluids Using Fast Fourier Transform Square Wave Voltammetry. Int. J. Eng. 2020, 33, 1696–1702. [Google Scholar] [CrossRef]
- Prasad, B.B.; Singh, R.; Singh, K. Development of highly electrocatalytic and electroconducting imprinted film using Ni nanomer for ultra-trace detection of thiamine. Sens. Actuators B Chem. 2017, 246, 38–45. [Google Scholar] [CrossRef]
- Shankar, S.; John, S.A. Sensitive and highly selective determination of vitamin B1 in the presence of other vitamin B complexes using functionalized gold nanoparticles as fluorophore. Rsc. Adv. 2015, 5, 49920–49925. [Google Scholar] [CrossRef]
- Song, Z.; Hou, S. Determination of picomole amounts of thiamine through flow-injection analysis based on the suppression of luminol-KIO(4) chemiluminescence system. J. Pharm. Biomed. Anal. 2002, 28, 683–691. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, H.; Li, H.; Pan, S.; Ran, Y.; Hu, X. Construction of a novel turn-on-off fluorescence sensor used for highly selective detection of thiamine via its quenching effect on o-phen-Zn(2+) complex. Luminescence 2018, 33, 1128–1135. [Google Scholar] [CrossRef] [PubMed]
- Immundiagnostik AG. ID-Vit® Pantothenic acid. Available online: https://www.immundiagnostik.com/media/pages/testkits/kif004/1c6c7f961a-1633917660/kif004_2019-05-23_pantothensaeure.pdf (accessed on 10 July 2021).
- Immundiagnostik AG. ID-Vit® Niacin. Available online: https://www.immundiagnostik.com/media/pages/testkits/kif003/2d1c628e3b-1633917660/kif003_2019-05-23_niacin.pdf (accessed on 10 July 2021).
- RECIPE Chemicals+Instruments GmbH. VITAMIN B1, B2 AND B6 (COMBIKIT). Available online: https://recipe.de/products/combikit-vitamin-b1-b2-b6-whole-blood/ (accessed on 10 July 2021).
- Immundiagnostik AG. Vitamin B1 HPLC Kit. Available online: https://www.immundiagnostik.com/media/pages/testkits/kc2201/59011e2c72-1633658467/vitamin-b1_kc2201.pdf (accessed on 10 July 2021).
- RECIPE Chemicals+Instruments GmbH. VITAMIN B1. Available online: https://recipe.de/products/vitamin-b1-whole-blood/ (accessed on 10 July 2021).
- RECIPE Chemicals+Instruments GmbH. VITAMIN B2. Available online: https://recipe.de/products/vitamin-b2-whole-blood/ (accessed on 10 July 2021).
- MYBioSource. Thiamine Elisa Kit: Human Thiamine ELISA Kit. Available online: https://www.mybiosource.com/human-elisa-kits/thiamine/167383 (accessed on 10 July 2021).
- LSBio. Vitamin B2/Riboflavin (Competitive EIA) ELISA Kit-LS-F55485. Available online: https://www.lsbio.com/elisakits/vitamin-b2-riboflavin-competitive-eia-elisa-kit-ls-f55485/55485 (accessed on 10 July 2021).
- Antibodiesonline GmbH. Vitamin B2 (Riboflavin) ELISA Kit. Available online: https://www.antibodies-online.com/kit/1059863/Vitamin+B2+Riboflavin+ELISA+Kit/ (accessed on 10 July 2021).
- Amrein, K.; Oudemans-van Straaten, H.M.; Berger, M.M. Vitamin therapy in critically ill patients: Focus on thiamine, vitamin C, and vitamin D. Intensive Care Med. 2018, 44, 1940–1944. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.M.; Suter, P.M. Vitamin requirements of elderly people: An update. Am. J. Clin. Nutr. 1993, 58, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.; Guerrini, I.; Marshall, E.J. Incidence of Adverse Reactions to Parenteral Thiamine in the Treatment of Wernicke’s Encephalopathy, and Recommendations. Alcohol Alcohol. 2019, 54, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Claus, D.; Eggers, R.; Warecka, K.; Neundorfer, B. Thiamine deficiency and nervous system function disturbances. Eur. Arch. Psych. Neurol. Sci. 1985, 234, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Alaei Shahmiri, F.; Soares, M.J.; Zhao, Y.; Sherriff, J. High-dose thiamine supplementation improves glucose tolerance in hyperglycemic individuals: A randomized, double-blind cross-over trial. Eur. J. Nutr. 2013, 52, 1821–1824. [Google Scholar] [CrossRef]
- Gibson, G.E.; Hirsch, J.A.; Cirio, R.T.; Jordan, B.D.; Fonzetti, P.; Elder, J. Abnormal thiamine-dependent processes in Alzheimer’s Disease. Lessons from diabetes. Mol. Cell. Neurosci. 2013, 55, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Kv, L.N.; Nguyen, L.T. The role of thiamine in HIV infection. Int. J. Infect. Dis. 2013, 17, e221–e227. [Google Scholar] [CrossRef]
- Volvert, M.L.; Seyen, S.; Piette, M.; Evrard, B.; Gangolf, M.; Plumier, J.C.; Bettendorff, L. Benfotiamine, a synthetic S-acyl thiamine derivative, has different mechanisms of action and a different pharmacological profile than lipid-soluble thiamine disulfide derivatives. BMC Pharmacol. 2008, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- Loew, D. Pharmacokinetics of thiamine derivatives especially of benfotiamine. Int. J. Clin. Pharmacol. Ther. 1996, 34, 47–50. [Google Scholar]
- Nishikawa, T.; Edelstein, D.; Du, X.L.; Yamagishi, S.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.P.; et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Raj, V.; Ojha, S.; Howarth, F.C.; Belur, P.D.; Subramanya, S.B. Therapeutic potential of benfotiamine and its molecular targets. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 3261–3273. [Google Scholar] [CrossRef] [PubMed]
- Babaei-Jadidi, R.; Karachalias, N.; Ahmed, N.; Battah, S.; Thornalley, P.J. Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine. Diabetes 2003, 52, 2110–2120. [Google Scholar] [CrossRef]
- Stracke, H.; Lindemann, A.; Federlin, K. A benfotiamine-vitamin B combination in treatment of diabetic polyneuropathy. Exp. Clin. Endocrinol. Diabetes 1996, 104, 311–316. [Google Scholar] [CrossRef]
- Huang, W.C.; Huang, H.Y.; Hsu, Y.J.; Su, W.H.; Shen, S.Y.; Lee, M.C.; Lin, C.L.; Huang, C.C. The Effects of Thiamine Tetrahydrofurfuryl Disulfide on Physiological Adaption and Exercise Performance Improvement. Nutrients 2018, 10, 851. [Google Scholar] [CrossRef] [PubMed]
- Scientific Committee on Food. Opinion of the Scientific Committee on Food on the Tolerable Upper Intake Level of Vitamin B1; European Commission: Brussels, Belgium, 2001. [Google Scholar]
- Wrenn, K.D.; Murphy, F.; Slovis, C.M. A toxicity study of parenteral thiamine hydrochloride. Ann. Emerg. Med. 1989, 18, 867–870. [Google Scholar] [CrossRef]
- Sica, D.A. Loop diuretic therapy, thiamine balance, and heart failure. Congest. Heart Fail. 2007, 13, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Schumann, K. Interactions between drugs and vitamins at advanced age. Int. J. Vitam. Nutr. Res. 1999, 69, 173–178. [Google Scholar] [CrossRef]
- Vora, B.; Green, E.A.E.; Khuri, N.; Ballgren, F.; Sirota, M.; Giacomini, K.M. Drug-nutrient interactions: Discovering prescription drug inhibitors of the thiamine transporter ThTR-2 (SLC19A3). Am. J. Clin. Nutr. 2020, 111, 110–121. [Google Scholar] [CrossRef]
- Giacomini, M.M.; Hao, J.; Liang, X.; Chandrasekhar, J.; Twelves, J.; Whitney, J.A.; Lepist, E.I.; Ray, A.S. Interaction of 2,4-Diaminopyrimidine-Containing Drugs Including Fedratinib and Trimethoprim with Thiamine Transporters. Drug Metab. Dispos. 2017, 45, 76–85. [Google Scholar] [CrossRef]
- Hohmann, H.P.; Bretzel, W.; Hans, M.; Friedel, A.; Litta, G.; Lehmann, M.; Kurth, R.; Paust, J.; Haehnlein, W. Vitamins, 7. Vitamin B2 (Riboflavin). In Ullmann’s Encyclopedia of Industrial Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; pp. 1–12. [Google Scholar]
- Saedisomeolia, A.; Ashoori, M. Riboflavin in Human Health: A Review of Current Evidences. Adv. Food Nutr. Res. 2018, 83, 57–81. [Google Scholar] [CrossRef]
- Powers, H.J. Riboflavin (vitamin B-2) and health. Am. J. Clin. Nutr. 2003, 77, 1352–1360. [Google Scholar] [CrossRef] [PubMed]
- Mestdagh, F.; De Meulenaer, B.; De Clippeleer, J.; Devlieghere, F.; Huyghebaert, A. Protective influence of several packaging materials on light oxidation of milk. J. Dairy Sci. 2005, 88, 499–510. [Google Scholar] [CrossRef]
- Cardoso, D.R.; Libardi, S.H.; Skibsted, L.H. Riboflavin as a photosensitizer. Effects on human health and food quality. Food Funct. 2012, 3, 487–502. [Google Scholar] [CrossRef] [PubMed]
- Sheraz, M.A.; Kazi, S.H.; Ahmed, S.; Anwar, Z.; Ahmad, I. Photo, thermal and chemical degradation of riboflavin. Beilstein J. Org. Chem. 2014, 10, 1999–2012. [Google Scholar] [CrossRef] [PubMed]
- Choe, E.; Huang, R.M.; Min, D.B. Chemical reactions and stability of riboflavin in foods. J. Food Sci. 2005, 70, R28–R36. [Google Scholar] [CrossRef]
- Gaylord, A.M.; Warthesen, J.J.; Smith, D.E. Influence of milk fat, milk solids, and light intensity on the light stability of vitamin A and riboflavin in lowfat milk. J. Dairy Sci. 1986, 69, 2779–2784. [Google Scholar] [CrossRef]
- Semba, R.D. The discovery of the vitamins. Int. J. Vitam. Nutr. Res. 2012, 82, 310–315. [Google Scholar] [CrossRef]
- Northrop-Clewes, C.A.; Thurnham, D.I. The discovery and characterization of riboflavin. Ann. Nutr. Metab. 2012, 61, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Bacher, A. Biosynthesis of vitamin B2 and flavocoenzymes in plants. Adv. Bot. Res. 2011, 58, 93–152. [Google Scholar] [CrossRef]
- Fischer, M.; Bacher, A. Biosynthesis of vitamin B2: Structure and mechanism of riboflavin synthase. Arch. Biochem. Biophys. 2008, 474, 252–265. [Google Scholar] [CrossRef]
- Fischer, M.; Bacher, A. Biosynthesis of vitamin B2: A unique way to assemble a xylene ring. Chembiochem 2011, 12, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Bacher, A.; Eberhardt, S.; Fischer, M.; Kis, K.; Richter, G. Biosynthesis of vitamin b2 (riboflavin). Annu. Rev. Nutr. 2000, 20, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Angulo, V.A. Overlapping riboflavin supply pathways in bacteria. Crit. Rev. Microbiol. 2017, 43, 196–209. [Google Scholar] [CrossRef]
- Gutierrez-Preciado, A.; Torres, A.G.; Merino, E.; Bonomi, H.R.; Goldbaum, F.A.; Garcia-Angulo, V.A. Extensive Identification of Bacterial Riboflavin Transporters and Their Distribution across Bacterial Species. PLoS ONE 2015, 10, e0126124. [Google Scholar] [CrossRef] [PubMed]
- Zylberman, V.; Klinke, S.; Haase, I.; Bacher, A.; Fischer, M.; Goldbaum, F.A. Evolution of vitamin B2 biosynthesis: 6,7-dimethyl-8-ribityllumazine synthases of Brucella. J. Bacteriol. 2006, 188, 6135–6142. [Google Scholar] [CrossRef]
- Schwechheimer, S.K.; Park, E.Y.; Revuelta, J.L.; Becker, J.; Wittmann, C. Biotechnology of riboflavin. Appl. Microbiol. Biotechnol. 2016, 100, 2107–2119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-R.; Ge, Y.-Y.; Liu, P.-H.; Wu, D.-T.; Liu, H.-Y.; Li, H.-B.; Corke, H.; Gan, R.-Y. Biotechnological Strategies of Riboflavin Biosynthesis in Microbes. Engineering 2021. [Google Scholar] [CrossRef]
- Revuelta, J.L.; Ledesma-Amaro, R.; Lozano-Martinez, P.; Diaz-Fernandez, D.; Buey, R.M.; Jimenez, A. Bioproduction of riboflavin: A bright yellow history. J. Ind. Microbiol. Biotechnol. 2017, 44, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Auclair, O.; Han, Y.; Burgos, S.A. Consumption of Milk and Alternatives and Their Contribution to Nutrient Intakes among Canadian Adults: Evidence from the 2015 Canadian Community Health Survey-Nutrition. Nutrients 2019, 11, 1948. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Aparicio-Ugarriza, R.; Olza, J.; Aranceta-Bartrina, J.; Gil, A.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; Gonzalez-Gross, M. Dietary Intake and Food Sources of Niacin, Riboflavin, Thiamin and Vitamin B (6) in a Representative Sample of the Spanish Population. The Anthropometry, Intake, and Energy Balance in Spain (ANIBES) Study dagger. Nutrients 2018, 10, 846. [Google Scholar] [CrossRef] [PubMed]
- Gorska-Warsewicz, H.; Rejman, K.; Laskowski, W.; Czeczotko, M. Milk and Dairy Products and Their Nutritional Contribution to the Average Polish Diet. Nutrients 2019, 11, 1771. [Google Scholar] [CrossRef]
- Efsa Panel on Dietetic Products; Nutrition and Allergies; Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; et al. Dietary Reference Values for riboflavin. EFSA J. 2017, 15, e04919. [Google Scholar] [CrossRef] [PubMed]
- Revuelta, J.L.; Ledesma-Amaro, R.; Jiménez, A. Industrial production of vitamin B2 by microbial fermentation. In Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; pp. 15–40. [Google Scholar]
- Mosegaard, S.; Dipace, G.; Bross, P.; Carlsen, J.; Gregersen, N.; Olsen, R.K.J. Riboflavin Deficiency-Implications for General Human Health and Inborn Errors of Metabolism. Int. J. Mol. Sci. 2020, 21, 3847. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Fulgoni Iii, V.L. Nutritional impact of adding a serving of mushrooms to USDA Food Patterns-a dietary modeling analysis. Food Nutr. Res. 2021, 65. [Google Scholar] [CrossRef] [PubMed]
- Škrovánková, S.; Sikorová, P. Vitamin B2 (riboflavin) content in cereal products. Acta Univ. Agric. Silvic. Mendel. Brun. 2010. [Google Scholar] [CrossRef]
- Vidal-Valverde, C.; Prodanov, M.; Sierra, I. Natural fermentation of lentils. Z. Lebensm. Unters. Forsch. 1997, 205, 464–469. [Google Scholar] [CrossRef]
- Melse-Boonstra, A. Bioavailability of Micronutrients From Nutrient-Dense Whole Foods: Zooming in on Dairy, Vegetables, and Fruits. Front. Nutr. 2020, 7, 101. [Google Scholar] [CrossRef] [PubMed]
- Kanno, C.; Kanehara, N.; Shirafuji, K.; Tanji, R.; Imai, T. Binding form of vitamin B2 in bovine milk: Its concentration, distribution and binding linkage. J. Nutr. Sci. Vitam. 1991, 37, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Thielecke, F.; Lecerf, J.M.; Nugent, A.P. Processing in the food chain: Do cereals have to be processed to add value to the human diet? Nutr. Res. Rev. 2021, 34, 159–173. [Google Scholar] [CrossRef]
- Pinheiro-Sant’Ana, H.M.; Stringheta, P.C.P.; Penteado, M.V.; Brandão, S.C. Stability of B-vitamins in meats prepared by foodservice. 2.Riboflavin. Foodserv. Res. Int. 1999, 11, 53–67. [Google Scholar] [CrossRef]
- Guneser, O.; Karagul Yuceer, Y. Effect of ultraviolet light on water- and fat-soluble vitamins in cow and goat milk. J. Dairy Sci. 2012, 95, 6230–6241. [Google Scholar] [CrossRef]
- Asadullah; Khair-un-nisa; Tarar, O.M.; Ali, S.A.; Jamil, K.; Begum, A. Study to evaluate the impact of heat treatment on water soluble vitamins in milk. J. Pak. Med. Assoc. 2010, 60, 909–912. [Google Scholar]
- Golbach, J.L.; Ricke, S.C.; O’Bryan, C.A.; Crandall, P.G. Riboflavin in nutrition, food processing, and analysis-A Review. J. Food Res. 2014, 3, 23. [Google Scholar] [CrossRef]
- Sharabi, S.; Okun, Z.; Shpigelman, A. Changes in the shelf life stability of riboflavin, vitamin C and antioxidant properties of milk after (ultra) high pressure homogenization: Direct and indirect effects. Innov. Food Sci. Emerg. Technol. 2018, 47, 161–169. [Google Scholar] [CrossRef]
- Allen, C.; Parks, O.W. Photodegradation of riboflavin in milks exposed to fluorescent light. J. Dairy Sci. 1979, 62, 1377–1379. [Google Scholar] [CrossRef]
- Dror, D.K.; Allen, L.H. Overview of Nutrients in Human Milk. Adv. Nutr. 2018, 9, 278S–294S. [Google Scholar] [CrossRef] [PubMed]
- Bates, C.J.; Liu, D.S.; Fuller, N.J.; Lucas, A. Susceptibility of riboflavin and vitamin A in breast milk to photodegradation and its implications for the use of banked breast milk in infant feeding. Acta Paediatr. Scand. 1985, 74, 40–44. [Google Scholar] [CrossRef]
- Lima, H.K.; Vogel, K.; Hampel, D.; Wagner-Gillespie, M.; Fogleman, A.D. The Associations Between Light Exposure During Pumping and Holder Pasteurization and the Macronutrient and Vitamin Concentrations in Human Milk. J. Hum. Lact. 2020, 36, 254–263. [Google Scholar] [CrossRef]
- Rico, D.; Penas, E.; Garcia, M.D.C.; Martinez-Villaluenga, C.; Rai, D.K.; Birsan, R.I.; Frias, J.; Martin-Diana, A.B. Sprouted Barley Flour as a Nutritious and Functional Ingredient. Foods 2020, 9, 296. [Google Scholar] [CrossRef]
- Tishler, M.; Pfister, K., 3rd; Babson, R.D.; Ladenburg, K.; Fleming, A.J. The reaction between o-aminoazo compounds and barbituric acid; a new synthesis of riboflavin. J. Am. Chem. Soc. 1947, 69, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Tischler, M.; Wellman, J.W.; Ladenburg, K. The preparation of riboflavin; the synthesis of alloxazines and isoalloxazines. J. Am. Chem. Soc. 1945, 67, 2165–2168. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hu, W.; Wang, Z.; Chen, T. Production of riboflavin and related cofactors by biotechnological processes. Microb. Cell Fact. 2020, 19, 31. [Google Scholar] [CrossRef] [PubMed]
- Revuelta, J.L.; Buey, R.M.; Ledesma-Amaro, R.; Vandamme, E.J. Microbial biotechnology for the synthesis of (pro)vitamins, biopigments and antioxidants: Challenges and opportunities. Microb. Biotechnol. 2016, 9, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Perkins, J.B.; Sloma, A.; Hermann, T.; Theriault, K.; Zachgo, E.; Erdenberger, T.; Hannett, N.; Chatterjee, N.P.; Williams, V.; Rufo, G.A.; et al. Genetic engineering of Bacillus subtilis for the commercial production of riboflavin. J. Ind. Microbiol. Biotechnol. 1999, 22, 8–18. [Google Scholar] [CrossRef]
- Aguiar, T.Q.; Silva, R.; Domingues, L. Ashbya gossypii beyond industrial riboflavin production: A historical perspective and emerging biotechnological applications. Biotechnol. Adv. 2015, 33, 1774–1786. [Google Scholar] [CrossRef] [PubMed]
- Man, Z.W.; Rao, Z.M.; Cheng, Y.P.; Yang, T.W.; Zhang, X.; Xu, M.J.; Xu, Z.H. Enhanced riboflavin production by recombinant Bacillus subtilis RF1 through the optimization of agitation speed. World J. Microbiol. Biotechnol. 2014, 30, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Stahmann, K.P.; Revuelta, J.L.; Seulberger, H. Three biotechnical processes using Ashbya gossypii, Candida famata, or Bacillus subtilis compete with chemical riboflavin production. Appl. Microbiol. Biotechnol. 2000, 53, 509–516. [Google Scholar] [CrossRef]
- Shi, T.; Wang, Y.; Wang, Z.; Wang, G.; Liu, D.; Fu, J.; Chen, T.; Zhao, X. Deregulation of purine pathway in Bacillus subtilis and its use in riboflavin biosynthesis. Microb. Cell Fact. 2014, 13, 101. [Google Scholar] [CrossRef]
- Abbas, C.A.; Sibirny, A.A. Genetic control of biosynthesis and transport of riboflavin and flavin nucleotides and construction of robust biotechnological producers. Microbiol. Mol. Biol. Rev. 2011, 75, 321–360. [Google Scholar] [CrossRef]
- Wang, G.; Shi, T.; Chen, T.; Wang, X.; Wang, Y.; Liu, D.; Guo, J.; Fu, J.; Feng, L.; Wang, Z.; et al. Integrated whole-genome and transcriptome sequence analysis reveals the genetic characteristics of a riboflavin-overproducing Bacillus subtilis. Metab. Eng. 2018, 48, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Averianova, L.A.; Balabanova, L.A.; Son, O.M.; Podvolotskaya, A.B.; Tekutyeva, L.A. Production of Vitamin B2 (Riboflavin) by Microorganisms: An Overview. Front. Bioeng. Biotechnol. 2020, 8, 570828. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Park, E.Y. Riboflavin production by Ashbya gossypii. Biotechnol. Lett. 2012, 34, 611–618. [Google Scholar] [CrossRef]
- EU Commision. E. Commission Directive 2006/125/EC of 5 December 2006 on Processed Cereal-Based Foods and Baby Foods for Infants and Young Children. Available online: https://eur-lex.europa.eu/eli/dir/2006/125/oj (accessed on 11 June 2021).
- Levit, R.; Savoy de Giori, G.; de Moreno de LeBlanc, A.; LeBlanc, J.G. Recent update on lactic acid bacteria producing riboflavin and folates: Application for food fortification and treatment of intestinal inflammation. J. Appl. Microbiol. 2021, 130, 1412–1424. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, V.; Russo, P.; Duenas, M.T.; Lopez, P.; Spano, G. Lactic acid bacteria producing B-group vitamins: A great potential for functional cereals products. Appl. Microbiol. Biotechnol. 2012, 96, 1383–1394. [Google Scholar] [CrossRef]
- Thakur, K.; Tomar, S.K.; De, S. Lactic acid bacteria as a cell factory for riboflavin production. Microb. Biotechnol. 2016, 9, 441–451. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Laino, J.E.; del Valle, M.J.; Vannini, V.; Van Sinderen, D.; Taranto, M.P.; de Valdez, G.F.; de Giori, G.S.; Sesma, F. B-group vitamin production by lactic acid bacteria-current knowledge and potential applications. J. Appl. Microbiol. 2011, 111, 1297–1309. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Milani, C.; de Giori, G.S.; Sesma, F.; Van Sinderen, D.; Ventura, M. Bacteria as vitamin suppliers to their host: A gut microbiota perspective. Curr. Opin. Biotechnol. 2013, 24, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Solopova, A.; Bottacini, F.; Venturi Degli Esposti, E.; Amaretti, A.; Raimondi, S.; Rossi, M.; Van Sinderen, D. Riboflavin Biosynthesis and Overproduction by a Derivative of the Human Gut Commensal Bifidobacterium longum subsp. infantis ATCC 15697. Front. Microbiol. 2020, 11, 573335. [Google Scholar] [CrossRef]
- Burgess, C.M.; Smid, E.J.; Rutten, G.; van Sinderen, D. A general method for selection of riboflavin-overproducing food grade micro-organisms. Microb. Cell Fact. 2006, 5, 24. [Google Scholar] [CrossRef]
- del Valle, M.J.; Laiño, J.E.; de Giori, G.S.; LeBlanc, J. Riboflavin producing lactic acid bacteria as a biotechnological strategy to obtain bio-enriched soymilk. Food Res. Int. 2014, 62, 1015–1019. [Google Scholar] [CrossRef]
- Daniel, H.; Binninger, E.; Rehner, G. Hydrolysis of FMN and FAD by alkaline phosphatase of the intestinal brush-border membrane. Int. J. Vitam. Nutr. Res. 1983, 53, 109–114. [Google Scholar]
- LeBlanc, J.G.; Burgess, C.; Sesma, F.; de Giori, G.S.; van Sinderen, D. Lactococcus lactis is capable of improving the riboflavin status in deficient rats. Br. J. Nutr. 2005, 94, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Kasper, H. Vitamin absorption in the colon. Am. J. Proctol 1970, 21, 341–345. [Google Scholar]
- Iinuma, S. Synthesis of riboflavin by intestinal bacteria. J. Vitam. 1955, 1, 6–13. [Google Scholar] [CrossRef]
- Yonezawa, A.; Inui, K. Novel riboflavin transporter family RFVT/SLC52: Identification, nomenclature, functional characterization and genetic diseases of RFVT/SLC52. Mol. Asp. Med. 2013, 34, 693–701. [Google Scholar] [CrossRef]
- Yonezawa, A.; Masuda, S.; Katsura, T.; Inui, K. Identification and functional characterization of a novel human and rat riboflavin transporter, RFT1. Am. J. Physiol. Cell Physiol. 2008, 295, C632–C641. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Yonezawa, A.; Yoshimatsu, H.; Masuda, S.; Katsura, T.; Inui, K. Identification and comparative functional characterization of a new human riboflavin transporter hRFT3 expressed in the brain. J. Nutr. 2010, 140, 1220–1226. [Google Scholar] [CrossRef]
- Yamamoto, S.; Inoue, K.; Ohta, K.Y.; Fukatsu, R.; Maeda, J.Y.; Yoshida, Y.; Yuasa, H. Identification and functional characterization of rat riboflavin transporter 2. J. Biochem. 2009, 145, 437–443. [Google Scholar] [CrossRef]
- Jaeger, B.; Bosch, A.M. Clinical presentation and outcome of riboflavin transporter deficiency: Mini review after five years of experience. J. Inherit. Metab. Dis. 2016, 39, 559–564. [Google Scholar] [CrossRef]
- Barile, M.; Giancaspero, T.A.; Leone, P.; Galluccio, M.; Indiveri, C. Riboflavin transport and metabolism in humans. J. Inherit. Metab. Dis. 2016, 39, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Hustad, S.; McKinley, M.C.; McNulty, H.; Schneede, J.; Strain, J.J.; Scott, J.M.; Ueland, P.M. Riboflavin, flavin mononucleotide, and flavin adenine dinucleotide in human plasma and erythrocytes at baseline and after low-dose riboflavin supplementation. Clin. Chem. 2002, 48, 1571–1577. [Google Scholar] [CrossRef] [PubMed]
- Frago, S.; Martinez-Julvez, M.; Serrano, A.; Medina, M. Structural analysis of FAD synthetase from Corynebacterium ammoniagenes. BMC Microbiol. 2008, 8, 160. [Google Scholar] [CrossRef] [PubMed]
- Herguedas, B.; Martinez-Julvez, M.; Frago, S.; Medina, M.; Hermoso, J.A. Oligomeric state in the crystal structure of modular FAD synthetase provides insights into its sequential catalysis in prokaryotes. J. Mol. Biol. 2010, 400, 218–230. [Google Scholar] [CrossRef]
- Barile, M.; Giancaspero, T.A.; Brizio, C.; Panebianco, C.; Indiveri, C.; Galluccio, M.; Vergani, L.; Eberini, I.; Gianazza, E. Biosynthesis of flavin cofactors in man: Implications in health and disease. Curr. Pharm. Des. 2013, 19, 2649–2675. [Google Scholar] [CrossRef]
- Serrano, A.; Ferreira, P.; Martinez-Julvez, M.; Medina, M. The prokaryotic FAD synthetase family: A potential drug target. Curr. Pharm. Des. 2013, 19, 2637–2648. [Google Scholar] [CrossRef]
- Chastain, J.L.; McCormick, D.B. Flavin catabolites: Identification and quantitation in human urine. Am. J. Clin. Nutr. 1987, 46, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Lienhart, W.D.; Gudipati, V.; Macheroux, P. The human flavoproteome. Arch. Biochem. Biophys. 2013, 535, 150–162. [Google Scholar] [CrossRef]
- Macheroux, P.; Kappes, B.; Ealick, S.E. Flavogenomics–A genomic and structural view of flavin-dependent proteins. FEBS J. 2011, 278, 2625–2634. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.K.; Anderson, K.E. Variegate Porphyria. In GeneReviews®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Mirzaa, G., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Musayev, F.N.; Di Salvo, M.L.; Saavedra, M.A.; Contestabile, R.; Ghatge, M.S.; Haynes, A.; Schirch, V.; Safo, M.K. Molecular basis of reduced pyridoxine 5′-phosphate oxidase catalytic activity in neonatal epileptic encephalopathy disorder. J. Biol. Chem. 2009, 284, 30949–30956. [Google Scholar] [CrossRef]
- Manoj, N.; Ealick, S.E. Unusual space-group pseudosymmetry in crystals of human phosphopantothenoylcysteine decarboxylase. Acta Cryst. D Biol. Cryst. 2003, 59, 1762–1766. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, I.; Carecchio, M.; Tiranti, V. Inborn errors of coenzyme A metabolism and neurodegeneration. J. Inherit. Metab. Dis. 2019, 42, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Heeringa, S.F.; Chernin, G.; Chaki, M.; Zhou, W.; Sloan, A.J.; Ji, Z.; Xie, L.X.; Salviati, L.; Hurd, T.W.; Vega-Warner, V.; et al. COQ6 mutations in human patients produce nephrotic syndrome with sensorineural deafness. J. Clin. Investig. 2011, 121, 2013–2024. [Google Scholar] [CrossRef]
- Acosta, M.J.; Vazquez Fonseca, L.; Desbats, M.A.; Cerqua, C.; Zordan, R.; Trevisson, E.; Salviati, L. Coenzyme Q biosynthesis in health and disease. Biochim. Biophys. Acta 2016, 1857, 1079–1085. [Google Scholar] [CrossRef]
- Cao, Q.; Li, G.M.; Xu, H.; Shen, Q.; Sun, L.; Fang, X.Y.; Liu, H.M.; Guo, W.; Zhai, Y.H.; Wu, B.B. Coenzyme Q(10) treatment for one child with COQ6 gene mutation induced nephrotic syndrome and literature review. Zhonghua Er Ke Za Zhi 2017, 55, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Afink, G.; Kulik, W.; Overmars, H.; de Randamie, J.; Veenboer, T.; van Cruchten, A.; Craen, M.; Ris-Stalpers, C. Molecular characterization of iodotyrosine dehalogenase deficiency in patients with hypothyroidism. J. Clin. Endocrinol. Metab. 2008, 93, 4894–4901. [Google Scholar] [CrossRef]
- Friedman, J.E.; Watson, J.A., Jr.; Lam, D.W.; Rokita, S.E. Iodotyrosine deiodinase is the first mammalian member of the NADH oxidase/flavin reductase superfamily. J. Biol. Chem. 2006, 281, 2812–2819. [Google Scholar] [CrossRef]
- Moreno, J.C.; Klootwijk, W.; Van Toor, H.; Pinto, G.; D’Alessandro, M.; Leger, A.; Goudie, D.; Polak, M.; Gruters, A.; Visser, T.J. Mutations in the iodotyrosine deiodinase gene and hypothyroidism. N. Engl. J. Med. 2008, 358, 1811–1818. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, M.M.; Kiely, M.; Harrington, K.E.; Robson, P.J.; Strain, J.J.; Flynn, A. The North/South Ireland Food Consumption Survey: Vitamin intakes in 18–64-year-old adults. Public Health Nutr. 2001, 4, 1069–1079. [Google Scholar] [CrossRef] [PubMed]
- Thakur, K.; Tomar, S.K.; Singh, A.K.; Mandal, S.; Arora, S. Riboflavin and health: A review of recent human research. Crit. Rev. Food Sci. Nutr. 2017, 57, 3650–3660. [Google Scholar] [CrossRef]
- Hoppel, C.L.; Tandler, B. Riboflavin and mouse hepatic cell structure and function. Mitochondrial oxidative metabolism in severe deficiency states. J. Nutr. 1975, 105, 562–570. [Google Scholar] [CrossRef]
- Mushtaq, S.; Su, H.; Hill, M.H.; Powers, H.J. Erythrocyte pyridoxamine phosphate oxidase activity: A potential biomarker of riboflavin status? Am. J. Clin. Nutr. 2009, 90, 1151–1159. [Google Scholar] [CrossRef]
- Grunert, S.C. Clinical and genetical heterogeneity of late-onset multiple acyl-coenzyme A dehydrogenase deficiency. Orphanet J. Rare Dis. 2014, 9, 117. [Google Scholar] [CrossRef]
- Balasubramaniam, S.; Christodoulou, J.; Rahman, S. Disorders of riboflavin metabolism. J. Inherit. Metab. Dis. 2019, 42, 608–619. [Google Scholar] [CrossRef]
- O’Callaghan, B.; Bosch, A.M.; Houlden, H. An update on the genetics, clinical presentation, and pathomechanisms of human riboflavin transporter deficiency. J. Inherit. Metab. Dis. 2019, 42, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Hellebrekers, D.; Sallevelt, S.; Theunissen, T.E.J.; Hendrickx, A.T.M.; Gottschalk, R.W.; Hoeijmakers, J.G.J.; Habets, D.D.; Bierau, J.; Schoonderwoerd, K.G.; Smeets, H.J.M. Novel SLC25A32 mutation in a patient with a severe neuromuscular phenotype. Eur. J. Hum. Genet. 2017, 25, 886–888. [Google Scholar] [CrossRef] [PubMed]
- Schiff, M.; Veauville-Merllie, A.; Su, C.H.; Tzagoloff, A.; Rak, M.; Ogier de Baulny, H.; Boutron, A.; Smedts-Walters, H.; Romero, N.B.; Rigal, O.; et al. SLC25A32 Mutations and Riboflavin-Responsive Exercise Intolerance. N. Engl. J. Med. 2016, 374, 795–797. [Google Scholar] [CrossRef]
- Thompson, D.F.; Saluja, H.S. Prophylaxis of migraine headaches with riboflavin: A systematic review. J. Clin. Pharmacol. Ther. 2017, 42, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Namazi, N.; Heshmati, J.; Tarighat-Esfanjani, A. Supplementation with Riboflavin (Vitamin B2) for Migraine Prophylaxis in Adults and Children: A Review. Int. J. Vitam. Nutr. Res. 2015, 85, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.K.; Dwivedi, A.; Pal, M.K.; Rastogi, N.; Gupta, P.; Ali, S.; Prabhu, M.B.; Kushwaha, H.N.; Ray, R.S.; Singh, S.K.; et al. Attenuated neuroprotective effect of riboflavin under UV-B irradiation via miR-203/c-Jun signaling pathway in vivo and in vitro. J. Biomed. Sci. 2014, 21, 39. [Google Scholar] [CrossRef] [PubMed]
- Barbre, A.B.; Hoane, M.R. Magnesium and riboflavin combination therapy following cortical contusion injury in the rat. Brain Res. Bull. 2006, 69, 639–646. [Google Scholar] [CrossRef]
- Seekamp, A.; Hultquist, D.E.; Till, G.O. Protection by vitamin B2 against oxidant-mediated acute lung injury. Inflammation 1999, 23, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Mack, C.P.; Hultquist, D.E.; Shlafer, M. Myocardial flavin reductase and riboflavin: A potential role in decreasing reoxygenation injury. Biochem. Biophys. Res. Commun. 1995, 212, 35–40. [Google Scholar] [CrossRef]
- Suwannasom, N.; Kao, I.; Pruss, A.; Georgieva, R.; Baumler, H. Riboflavin: The Health Benefits of a Forgotten Natural Vitamin. Int. J. Mol. Sci. 2020, 21, 950. [Google Scholar] [CrossRef] [PubMed]
- George, B.O.; Ojegbemi, O. Oxidative stress and the effect of riboflavin supplementation in individuals with uncomplicated malaria infection. Afr. J. Biotechnol. 2009, 8, 849–853. [Google Scholar]
- Akompong, T.; Ghori, N.; Haldar, K. In vitro activity of riboflavin against the human malaria parasite Plasmodium falciparum. Antimicrob. Agents Chemother. 2000, 44, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Araki, S.; Suzuki, M.; Fujimoto, M.; Kimura, M. Enhancement of resistance to bacterial infection in mice by vitamin B2. J. Vet. Med. Sci. 1995, 57, 599–602. [Google Scholar] [CrossRef] [PubMed]
- Mazur-Bialy, A.I.; Buchala, B.; Plytycz, B. Riboflavin deprivation inhibits macrophage viability and activity-a study on the RAW 264.7 cell line. Br. J. Nutr. 2013, 110, 509–514. [Google Scholar] [CrossRef]
- Bertollo, C.M.; Oliveira, A.C.; Rocha, L.T.; Costa, K.A.; Nascimento, E.B., Jr.; Coelho, M.M. Characterization of the antinociceptive and anti-inflammatory activities of riboflavin in different experimental models. Eur. J. Pharmacol. 2006, 547, 184–191. [Google Scholar] [CrossRef]
- Buehler, B.A. Vitamin B2: Riboflavin. J. Evid. Based Integr. Med. 2011, 16, 88–90. [Google Scholar] [CrossRef]
- Mazzotta, C.; Caragiuli, S.; Caporossi, A. Riboflavin and the Cornea and Implications for Cataracts. In Handbook of Nutrition, Diet and the Eye; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 123–130. [Google Scholar]
- Chocano-Bedoya, P.O.; Manson, J.E.; Hankinson, S.E.; Willett, W.C.; Johnson, S.R.; Chasan-Taber, L.; Ronnenberg, A.G.; Bigelow, C.; Bertone-Johnson, E.R. Dietary B vitamin intake and incident premenstrual syndrome. Am. J. Clin. Nutr. 2011, 93, 1080–1086. [Google Scholar] [CrossRef]
- Alam, M.M.; Iqbal, S.; Naseem, I. Ameliorative effect of riboflavin on hyperglycemia, oxidative stress and DNA damage in type-2 diabetic mice: Mechanistic and therapeutic strategies. Arch. Biochem. Biophys. 2015, 584, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Schoenen, J.; Lenaerts, M.; Bastings, E. High-dose riboflavin as a prophylactic treatment of migraine: Results of an open pilot study. Cephalalgia 1994, 14, 328–329. [Google Scholar] [CrossRef] [PubMed]
- MacLennan, S.C.; Wade, F.M.; Forrest, K.M.; Ratanayake, P.D.; Fagan, E.; Antony, J. High-dose riboflavin for migraine prophylaxis in children: A double-blind, randomized, placebo-controlled trial. J. Child Neurol. 2008, 23, 1300–1304. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.T.; Rivlin, R.S. Drugs that promote renal excretion of riboflavin. Drug Nutr. Interact. 1987, 5, 143–151. [Google Scholar]
- Pinto, J.; Huang, Y.P.; McConnell, R.J.; Rivlin, R.S. Increased urinary riboflavin excretion resulting from boric acid ingestion. J. Lab. Clin. Med. 1978, 92, 126–134. [Google Scholar]
- Ogura, R.; Ueta, H.; Hino, Y.; Hidaka, T.; Sugiyama, M. Riboflavin deficiency caused by treatment with adriamycin. J. Nutr. Sci. Vitam. 1991, 37, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.T.; Delman, B.N.; Dutta, P.; Nisselbaum, J. Adriamycin-induced increase in serum aldosterone levels: Effects in riboflavin-sufficient and riboflavin-deficient rats. Endocrinology 1990, 127, 1495–1501. [Google Scholar] [CrossRef]
- Pinto, J.; Wolinsky, M.; Rivlin, R.S. Chlorpromazine antagonism of thyroxine-induced flavin formation. Biochem. Pharmacol. 1979, 28, 597–600. [Google Scholar] [CrossRef]
- Rivlin, R.S.; Langdon, R.G. Effects of thyroxine upon biosynthesis of flavin mononucleotide and flavin adenine dinucleotide. Endocrinology 1969, 84, 584–588. [Google Scholar] [CrossRef]
- Pinto, J.; Huang, Y.P.; Rivlin, R.S. Inhibition of riboflavin metabolism in rat tissues by chlorpromazine, imipramine, and amitriptyline. J. Clin. Investig. 1981, 67, 1500–1506. [Google Scholar] [CrossRef]
- Rivlin, R.S.; Menendez, C.; Langdon, R.G. Biochemical similarities between hypothyroidism and riboflavin deficiency. Endocrinology 1968, 83, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Pelliccione, N.; Pinto, J.; Huang, Y.P.; Rivlin, R.S. Accelerated development of riboflavin deficiency by treatment with chlorpromazine. Biochem. Pharmacol. 1983, 32, 2949–2953. [Google Scholar] [CrossRef]
- Lee, S.S.; McCormick, D.B. Thyroid hormone regulation of flavocoenzyme biosynthesis. Arch. Biochem. Biophys. 1985, 237, 197–201. [Google Scholar] [CrossRef]
- Pinto, J.; Huang, Y.P.; Pelliccione, N.; Rivlin, R.S. Cardiac sensitivity to the inhibitory effects of chlorpromazine, imipramine and amitriptyline upon formation of flavins. Biochem. Pharmacol. 1982, 31, 3495–3499. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- Ross, A.C.; Caballero, B.; Cousins, R.J.; Tucker, K.L.; Ziegler, T.R. Modern Nutrition in Health and Disease, 11th ed.; Wolters Kluwer Health Adis: Philadelphia, PA, USA, 2012; pp. 1–1616. [Google Scholar]
- Erdman, J.W., Jr.; MacDonald, I.A.; Zeisel, S.H.; Penberthy WT, K.J. Present Knowledge in Nutrition, 10th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 293–306. [Google Scholar]
- Berry Ottaway, P. Stability of vitamins during food processing and storage. In Chemical Deterioration and Physical Instability of Food and Beverages; Skibsted, L.H., Risbo, J., Andersen, M.L., Eds.; Woodhead Publishing: Cambridge, UK, 2010; pp. 548, 553–554. [Google Scholar]
- World Health Organization. Pellagra and Its Prevention and Control in Major Emergencies; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Bhalla, T.C. Vitamin B3, Niacin. In Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants; Vandamme, E.J., Revuelta, J.L., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016. [Google Scholar]
- Allen, L.; Benoist, B.; Dary, O.; Hurrell, R. WHO/FAO Guidelines on Food Fortification with Micronutrients; World Health Organization: Geneva, Switzerland, 2006; pp. 73–76, 127. [Google Scholar]
- Gazzaniga, F.; Stebbins, R.; Chang, S.Z.; McPeek, M.A.; Brenner, C. Microbial NAD metabolism: Lessons from comparative genomics. Microbiol. Mol. Biol. Rev. 2009, 73, 529–541. [Google Scholar] [CrossRef]
- Li, Y.F.; Bao, W.G. Why do some yeast species require niacin for growth? Different modes of NAD synthesis. FEMS Yeast Res. 2007, 7, 657–664. [Google Scholar] [CrossRef]
- Kurnasov, O.; Goral, V.; Colabroy, K.; Gerdes, S.; Anantha, S.; Osterman, A.; Begley, T.P. NAD biosynthesis: Identification of the tryptophan to quinolinate pathway in bacteria. Chem. Biol. 2003, 10, 1195–1204. [Google Scholar] [CrossRef]
- Noctor, G.; Hager, J.; Li, S. Biosynthesis of NAD and Its Manipulation in Plants. In Advances in Botanical Research; Rébeillé, F., Douce, R., Eds.; Academic Press: Cambridge, MA, USA, 2011; Volume 58, pp. 153–201. [Google Scholar]
- Magnusdottir, S.; Ravcheev, D.; de Crecy-Lagard, V.; Thiele, I. Systematic genome assessment of B-vitamin biosynthesis suggests co-operation among gut microbes. Front. Genet. 2015, 6, 148. [Google Scholar] [CrossRef]
- Kirkland, J.B.; Meyer-Ficca, M.L. Chapter Three-Niacin. Adv. Food. Nutr. Res. 2018, 83, 83–149. [Google Scholar] [CrossRef]
- Bauer, J.E. Nutritional uniqueness of cats. Vet. Q 1998, 20, S78–S79. [Google Scholar] [CrossRef] [PubMed]
- Reeds, P.J. Dispensable and indispensable amino acids for humans. J. Nutr. 2000, 130, 1835S–1840S. [Google Scholar] [CrossRef]
- Shibata, K. Organ Co-Relationship in Tryptophan Metabolism and Factors That Govern the Biosynthesis of Nicotinamide from Tryptophan. J. Nutr. Sci. Vitam. 2018, 64, 90–98. [Google Scholar] [CrossRef]
- Gasperi, V.; Sibilano, M.; Savini, I.; Catani, M.V. Niacin in the Central Nervous System: An Update of Biological Aspects and Clinical Applications. Int. J. Mol. Sci. 2019, 20, 428. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.F. Tryptophan depletion and HIV infection: A metabolic link to pathogenesis. Lancet Infect. Dis. 2003, 3, 644–652. [Google Scholar] [CrossRef]
- Fukuwatari, T.; Shibata, K. Nutritional aspect of tryptophan metabolism. Int. J. Tryptophan Res. 2013, 6, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Meir, Z.; Osherov, N. Vitamin Biosynthesis as an Antifungal Target. J. Fungi 2018, 4, 72. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Dietary Reference Values for Nutrients Summary report. EFSA Support. Publ. 2017, 14, e15121. [Google Scholar] [CrossRef]
- Food and Drug Administration. Converting Units of Measure for Folate, Niacin, and Vitamins A, D, and E on the Nutrition and Supplement Facts Labels: Guidance for Industry. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-converting-units-measure-folate-niacin-and-vitamins-d-and-e-nutrition-and (accessed on 10 July 2021).
- Fukuwatari, T.; Ohta, M.; Kimtjra, N.; Sasaki, R.; Shibata, K. Conversion ratio of tryptophan to niacin in Japanese women fed a purified diet conforming to the Japanese Dietary Reference Intakes. J. Nutr. Sci. Vitam. 2004, 50, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F.; McClung, J.P. (Eds.) Niacin. In The Vitamins; Academic Press: Cambridge, MA, USA, 2017; pp. 331–350. [Google Scholar]
- Lanska, D.J. The discovery of niacin, biotin, and pantothenic acid. Ann. Nutr. Metab. 2012, 61, 246–253. [Google Scholar] [CrossRef]
- Henderson, L.M.; Koski, R.E.; D’Angeli, F. The role of riboflavin and vitamin B6 in tryptophan metabolism. J. Biol. Chem. 1955, 215, 369–376. [Google Scholar] [CrossRef]
- Shibata, K.; Mushiage, M.; Kondo, T.; Hayakawa, T.; Tsuge, H. Effects of vitamin B6 deficiency on the conversion ratio of tryptophan to niacin. Biosci. Biotechnol. Biochem. 1995, 59, 2060–2063. [Google Scholar] [CrossRef] [PubMed]
- Shibata, K.; Kobayashi, R.; Fukuwatari, T. Vitamin B1 deficiency inhibits the increased conversion of tryptophan to nicotinamide in severe food-restricted rats. Biosci. Biotechnol. Biochem. 2015, 79, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Fukuwatari, T.; Shibata, K. Effect of nicotinamide administration on the tryptophan-nicotinamide pathway in humans. Int. J. Vitam. Nutr. Res. 2007, 77, 255–262. [Google Scholar] [CrossRef]
- Lule, V.K.; Garg, S.; Gosewade, S.C.; Tomar, S.K.; Khedkar, C.D. Niacin. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 63–72. [Google Scholar]
- Wall, J.S.; Carpenter, K.J. Variation in Availability of Niacin in Grain Products. Food Technol. 1988, 42, 198. [Google Scholar]
- Blum, R. Vitamins, 8. Vitamin B3 (Niacin). In Ullmann’s Encyclopedia of Industrial Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; pp. 1–9. [Google Scholar]
- EFSA. Scientific opinion on dietary reference values for niacin. EFSA J. 2014, 12, 3759. [Google Scholar] [CrossRef]
- Chawla, J.; Kvarnberg, D. Chapter 59—Hydrosoluble vitamins. In Handbook of Clinical Neurology; Biller, J., Ferro, J.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 120, pp. 891–914. [Google Scholar]
- Raman, J.; Jang, K.Y.; Oh, Y.L.; Oh, M.; Im, J.H.; Lakshmanan, H.; Sabaratnam, V. Cultivation and Nutritional Value of Prominent Pleurotus spp.: An Overview. Mycobiology 2020, 49, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K. Nutraceutical Potential and Processing Aspects of Oyster Mushrooms (Pleurotus Species). Curr. Nutr. Food Sci. 2020, 16, 3–14. [Google Scholar] [CrossRef]
- Çatak, J.; Yaman, M. Determination of Nicotinic Acid and Nicotinamide Forms of Vitamin B3 (Niacin) in Fruits and Vegetables by HPLC Using Postcolumn Derivatization System. Pak. J. Nutr. 2019, 18, 563–570. [Google Scholar] [CrossRef][Green Version]
- Prousky, J.; Millman, C.G.; Kirkland, J.B. Pharmacologic Use of Niacin. J. Evid. Based Integr. Med. 2011, 16, 91–101. [Google Scholar] [CrossRef]
- Angelino, D.; Tassotti, M.; Brighenti, F.; Del Rio, D.; Mena, P. Niacin, alkaloids and (poly)phenolic compounds in the most widespread Italian capsule-brewed coffees. Sci. Rep. 2018, 8, 17874. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.; Yagar, E.F.; Eggers, R.; Hofmann, T. Quantitative investigation of trigonelline, nicotinic acid, and nicotinamide in foods, urine, and plasma by means of LC-MS/MS and stable isotope dilution analysis. J. Agric. Food Chem. 2008, 56, 11114–11121. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A. Variability of the Niacin Content in Coffee. Nature 1962, 194, 1096. [Google Scholar] [CrossRef]
- Stadler, R.H.; Varga, N.; Hau, J.; Vera, F.A.; Welti, D.H. Alkylpyridiniums. 1. Formation in model systems via thermal degradation of trigonelline. J. Agric. Food Chem. 2002, 50, 1192–1199. [Google Scholar] [CrossRef]
- Kremer, J.I.; Gompel, K.; Bakuradze, T.; Eisenbrand, G.; Richling, E. Urinary Excretion of Niacin Metabolites in Humans After Coffee Consumption. Mol. Nutr. Food Res. 2018, 62, e1700735. [Google Scholar] [CrossRef]
- Ghafoorunissa; Rao, B.S. Effect of leucine on enzymes of the tryptophan-niacin metabolic pathway in rat liver and kidney. Biochem. J. 1973, 134, 425–430. [Google Scholar] [CrossRef]
- Badawy, A.A.; Lake, S.L.; Dougherty, D.M. Mechanisms of the pellagragenic effect of leucine: Stimulation of hepatic tryptophan oxidation by administration of branched-chain amino acids to healthy human volunteers and the role of plasma free tryptophan and total kynurenines. Int. J. Tryptophan Res. 2014, 7, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.A. Kynurenine Pathway of Tryptophan Metabolism: Regulatory and Functional Aspects. Int. J. Tryptophan Res. 2017, 10, 1178646917691938. [Google Scholar] [CrossRef]
- Katz, S.H.; Hediger, M.L.; Valleroy, L.A. Traditional maize processing techniques in the new world. Science 1974, 184, 765–773. [Google Scholar] [CrossRef]
- Bender, D.A. Effects of a dietary excess of leucine on the metabolism of tryptophan in the rat: A mechanism for the pellagragenic action of leucine. Br. J. Nutr. 1983, 50, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Salter, M.; Bender, D.A.; Pogson, C.I. Leucine and tryptophan metabolism in rats. Biochem. J. 1985, 225, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Bates, C.J. Niacin and Pellagra. In Encyclopedia of Human Nutrition; Caballero, B., Ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 182–188. [Google Scholar]
- Cook, N.E.; Carpenter, K.J. Leucine excess and niacin status in rats. J. Nutr. 1987, 117, 519–526. [Google Scholar] [CrossRef]
- Manson, J.A.; Carpenter, K.J. The effect of a high level of dietary leucine on the niacin status of dogs. J. Nutr. 1978, 108, 1889–1898. [Google Scholar] [CrossRef] [PubMed]
- Hegedus, M.; Pedersen, B.; Eggum, B.O. The influence of milling on the nutritive value of flour from cereal grains. 7. Vitamins and tryptophan. Plant Foods Hum. Nutr. 1985, 35, 175–180. [Google Scholar] [CrossRef]
- Chamlagain, B.; Rautio, S.; Edelmann, M.; Ollilainen, V.; Piironen, V. Niacin contents of cereal-milling products in food-composition databases need to be updated. J. Food Compos. Anal. 2020, 91, 103518. [Google Scholar] [CrossRef]
- Çatak, J. Determination of niacin profiles in some animal and plant based foods by high performance liquid chromatography: Association with healthy nutrition. J. Anim. Sci. Technol. 2019, 61, 138–146. [Google Scholar] [CrossRef]
- Saleh, A.S.M.; Wang, P.; Wang, N.; Yang, L.; Xiao, Z. Brown Rice Versus White Rice: Nutritional Quality, Potential Health Benefits, Development of Food Products, and Preservation Technologies. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1070–1096. [Google Scholar] [CrossRef]
- Adebo, O.A. African Sorghum-Based Fermented Foods: Past, Current and Future Prospects. Nutrients 2020, 12, 1111. [Google Scholar] [CrossRef] [PubMed]
- Wyness, L. The role of red meat in the diet: Nutrition and health benefits. Proc. Nutr. Soc. 2016, 75, 227–232. [Google Scholar] [CrossRef]
- Feeney, M.J.; Dwyer, J.; Hasler-Lewis, C.M.; Milner, J.A.; Noakes, M.; Rowe, S.; Wach, M.; Beelman, R.B.; Caldwell, J.; Cantorna, M.T.; et al. Mushrooms and Health Summit proceedings. J. Nutr. 2014, 144, 1128S–1136S. [Google Scholar] [CrossRef]
- Muehlhoff, E.; Bennett, A.; McMahon, D. Milk and Dairy Products in Human Nutrition; FAO: Rome, Italy, 2013. [Google Scholar]
- Biesalksi, H.K.; Back, E.I. VITAMINS|Niacin, Nutritional Significance. In Encyclopedia of Dairy Sciences; Roginski, H., Ed.; Elsevier: Amsterdam, The Netherlands, 2002; pp. 2703–2707. [Google Scholar]
- Satya, S.; Kaushik, G.; Naik, S.N. Processing of food legumes: A boon to human nutrition. Med. J. Nutr. Metab. 2010, 3, 183–195. [Google Scholar] [CrossRef]
- Sobral, M.M.C.; Cunha, S.C.; Faria, M.A.; Ferreira, I.M. Domestic Cooking of Muscle Foods: Impact on Composition of Nutrients and Contaminants. Compr. Rev. Food Sci. Food Saf. 2018, 17, 309–333. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro-Sant’Ana, H.M.; Penteado, M.V.C.; Stringheta, P.C.; Chaves, J.B.P. Stability of B-Vitamins in Meats Prepared by Foodservice. 3. Nicotinic Acid. Foodserv. Res. Int. 1999, 11, 69–82. [Google Scholar] [CrossRef]
- Meyer, B.H.; Hinman, W.F.; Halliday, E.G. Retention of some vitamins of the B-complex in beef during cooking. Food Res. 1947, 12, 203–211. [Google Scholar] [CrossRef]
- Kilcast, D. Effect of Irradiation on Vitamins. Food Chem. 1994, 49, 157–164. [Google Scholar] [CrossRef]
- Yaman, M.; Catak, J.; Ugur, H.; Gurbuz, M.; Belli, I.; Tanyildiz, S.N.; Yildirim, H.; Cengiz, S.; Yavuz, B.B.; Kismiroglu, C.; et al. The bioaccessibility of water-soluble vitamins: A review. Trends Food Sci. Technol. 2021, 109, 552–563. [Google Scholar] [CrossRef]
- Akça, S.N.; Sargın, H.S.; Mızrak, Ö.F.; Yaman, M. Determination and assessment of the bioaccessibility of vitamins B1, B2, and B3 in commercially available cereal-based baby foods. Microchem. J. 2019, 150, 104192. [Google Scholar] [CrossRef]
- Gregory, J.F., 3rd. Accounting for differences in the bioactivity and bioavailability of vitamers. Food Nutr. Res. 2012, 56. [Google Scholar] [CrossRef]
- Zaupa, M.; Scazzina, F.; Dall’Asta, M.; Calani, L.; Del Rio, D.; Bianchi, M.A.; Melegari, C.; De Albertis, P.; Tribuzio, G.; Pellegrini, N.; et al. In vitro bioaccessibility of phenolics and vitamins from durum wheat aleurone fractions. J. Agric. Food Chem. 2014, 62, 1543–1549. [Google Scholar] [CrossRef]
- Carter, E.G.; Carpenter, K.J. The bioavailability for humans of bound niacin from wheat bran. Am. J. Clin. Nutr. 1982, 36, 855–861. [Google Scholar] [CrossRef]
- Harper, A.E.; Punekar, B.D.; Elvehjem, C.A. Effect of alkali treatment on the availability of niacin and amino acids in maize. J. Nutr. 1958, 66, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, K.J. The relationship of pellagra to corn and the low availability of niacin in cereals. Experientia Suppl. 1983, 44, 197–222. [Google Scholar] [CrossRef] [PubMed]
- Kodicek, E.; Braude, R.; Kon, S.K.; Mitchell, K.G. The availability to pigs of nicotinic acid in tortilla baked from maize treated with lime-water. Br. J. Nutr. 1959, 13, 363–384. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kodicek, E.; Braude, R.; Kon, S.K.; Mitchell, K.G. The effect of alkaline hydrolysis of maize on the availability of its nicotinic acid to the pig. Br. J. Nutr. 1956, 10, 51–67. [Google Scholar] [CrossRef] [PubMed]
- Wacher, C. Nixtamalization, a Mesoamerican technology to process maize at small-scale with great potential for improving the nutritional quality of maize based foods. In Proceedings of the Food-Based Approaches for a Healthy Nutrition, Ouagadougou, Burkina Faso, 1 January 2003; pp. 735–743. [Google Scholar]
- Escalante-Aburto, A.; Mariscal-Moreno, R.M.; Santiago-Ramos, D.; Ponce-García, N. An Update of Different Nixtamalization Technologies, and Its Effects on Chemical Composition and Nutritional Value of Corn Tortillas. Food Rev. Int. 2020, 36, 456–498. [Google Scholar] [CrossRef]
- Salazar, R.; Arambula-Villa, G.; Luna-Barcenas, G.; Figueroa-Cardenas, J.D.; Azuara, E.; Vazquez-Landaverde, P.A. Effect of added calcium hydroxide during corn nixtamalization on acrylamide content in tortilla chips. LWT Food Sci. Technol. 2014, 56, 87–92. [Google Scholar] [CrossRef]
- Maureen, N.; Kaaya, A.N.; Kauffman, J.; Narrod, C.; Atukwase, A. Enhancing Nutritional Benefits and Reducing Mycotoxin Contamination of Maize through Nixtamalization. J. Biol. Sci. 2020, 20, 153–162. [Google Scholar] [CrossRef]
- Sefa-Dedeh, S.; Cornelius, B.; Sakyi-Dawson, E.; Afoakwa, E.O. Effect of nixtamalization on the chemical and functional properties of maize. Food Chem. 2004, 86, 317–324. [Google Scholar] [CrossRef]
- Kamau, E.H.; Nkhata, S.G.; Ayua, E.O. Extrusion and nixtamalization conditions influence the magnitude of change in the nutrients and bioactive components of cereals and legumes. Food Sci. Nutr. 2020, 8, 1753–1765. [Google Scholar] [CrossRef] [PubMed]
- de la Parra, C.; Saldivar, S.O.; Liu, R.H. Effect of processing on the phytochemical profiles and antioxidant activity of corn for production of masa, tortillas, and tortilla chips. J. Agric. Food Chem. 2007, 55, 4177–4183. [Google Scholar] [CrossRef] [PubMed]
- Schaarschmidt, S.; Fauhl-Hassek, C. Mycotoxins during the Processes of Nixtamalization and Tortilla Production. Toxins 2019, 11, 227. [Google Scholar] [CrossRef] [PubMed]
- FAO. Maize in Human Nutrition; Food and Agriculture Organization of the United Nations: Rome, Italy, 1992. [Google Scholar]
- Bressani, R.; Paz y Paz, R.; Scrimshaw, N.S. Corn Nutrient Losses, Chemical Changes in Corn during Preparation of Tortillas. J. Agric. Food Chem. 1958, 6, 770–774. [Google Scholar] [CrossRef]
- Carter, E.G.; Carpenter, K.J. The available niacin values of foods for rats and their relation to analytical values. J. Nutr. 1982, 112, 2091–2103. [Google Scholar] [CrossRef]
- Dunn, M.L.; Jain, V.; Klein, B.P. Stability of key micronutrients added to fortified maize flours and corn meal. Ann. N. Y. Acad. Sci. 2014, 1312, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Laguna, J.; Carpenter, K.J. Raw versus processed corn in niacin-deficient diets. J. Nutr. 1951, 45, 21–28. [Google Scholar] [CrossRef]
- Braham, J.E.; Villarreal, A.; Bressani, R. Effect of lime treatment of corn on the availability of niacin for cats. J. Nutr. 1962, 76, 183–186. [Google Scholar] [CrossRef]
- Kodicek, E.; Ashby, D.R.; Muller, M.; Carpenter, K.J. The conversion of bound nicotinic acid to free nicotinamide on roasting sweet corn. Proc. Nutr. Soc. 1974, 33, 105A–106A. [Google Scholar]
- Buckel, L.; Kremer, J.I.; Stegmüller, S.; Richling, E. Fast, Sensitive and Robust Determination of Nicotinic Acid (Vitamin B3) Contents in Coffee Beverages Depending on the Degree of Roasting and Brewing Technique. Proceedings 2019, 11, 13. [Google Scholar] [CrossRef]
- Taguchi, H.; Sakaguchi, M.; Shimabayashi, Y. Trigonelline Content in Coffee Beans and the Thermal-Conversion of Trigonelline into Nicotinic-Acid during the Roasting of Coffee Beans. Agr. Biol. Chem. 1985, 49, 3467–3471. [Google Scholar] [CrossRef]
- Bressani, R.; Navarrete, D.A. Niacin Content of Coffee in Central America. J. Food Sci. 1959, 24, 344–351. [Google Scholar] [CrossRef]
- Teply, L.J.; Prier, R.F. Nutrients in Coffee-Nutritional Evaluation of Coffee Including Niacin Bioassay. J. Agric. Food Chem. 1957, 5, 375–377. [Google Scholar] [CrossRef]
- Caprioli, G.; Cortese, M.; Maggi, F.; Minnetti, C.; Odello, L.; Sagratini, G.; Vittori, S. Quantification of caffeine, trigonelline and nicotinic acid in espresso coffee: The influence of espresso machines and coffee cultivars. Int. J. Food Sci. Nutr. 2014, 65, 465–469. [Google Scholar] [CrossRef]
- Chaturvedi, A.; Geervani, P. Bioavailability of niacin from processed groundnuts. J. Nutr. Sci. Vitam. 1986, 32, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Nurit, E.; Lyan, B.; Pujos-Guillot, E.; Branlard, G.; Piquet, A. Change in B and E vitamin and lutein, β-sitosterol contents in industrial milling fractions and during toasted bread production. J. Cereal Sci. 2016, 69, 290–296. [Google Scholar] [CrossRef]
- Asiedu, M.; Lied, E.; Nilsen, R.; Sandnes, K. Effect of processing (sprouting and/or fermentation) on sorghum and maize: II. Vitamins and amino acid composition. Biological utilization of maize protein. Food Chem. 1993, 48, 201–204. [Google Scholar] [CrossRef]
- Žilić, S.; Basić, Z.; Hadži-Tašković Šukalović, V.; Maksimović, V.; Janković, M.; Filipović, M. Can the sprouting process applied to wheat improve the contents of vitamins and phenolic compounds and antioxidant capacity of the flour? Int. J. Food Sci. Technol. 2014, 49, 1040–1047. [Google Scholar] [CrossRef]
- Lay, M.M.G.; Fields, M.L. Nutritive-Value of Germinated Corn and Corn Fermented after Germination. J. Food Sci. 1981, 46, 1069–1073. [Google Scholar] [CrossRef]
- Mihhalevski, A.; Nisamedtinov, I.; Halvin, K.; Oseka, A.; Paalme, T. Stability of B-complex vitamins and dietary fiber during rye sourdough bread production. J. Cereal Sci. 2013, 57, 30–38. [Google Scholar] [CrossRef]
- Mani, I. Microbial Production of Vitamins; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Kumar, S.; Babu, B.V. Process Intensification of Nicotinic Acid Production via Enzymatic Conversion using Reactive Extraction. Chem. Biochem. Eng. Q. 2009, 23, 367–376. [Google Scholar] [CrossRef]
- Chuck, R. Green Sustainable Chemistry in the Production of Nicotinates. In Sustainable Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2009; pp. 541–550. [Google Scholar]
- Chuck, R. A catalytic green process for the production of niacin. Chimia 2000, 54, 508–513. [Google Scholar]
- Eschenmoser, W. 100 years of progress with LONZA. Chimia 1997, 51, 259–269. [Google Scholar]
- Chuck, R. Technology development in nicotinate production. Appl. Catal. A-Gen. 2005, 280, 75–82. [Google Scholar] [CrossRef]
- Gong, J.S.; Zhang, Q.; Gu, B.C.; Dong, T.T.; Li, H.; Li, H.; Lu, Z.M.; Shi, J.S.; Xu, Z.H. Efficient biocatalytic synthesis of nicotinic acid by recombinant nitrilase via high density culture. Bioresour. Technol. 2018, 260, 427–431. [Google Scholar] [CrossRef]
- Shaw, N.M.; Robins, K.T.; Kiener, A. Lonza: 20 years of biotransformations. Adv. Synth. Catal. 2003, 345, 425–435. [Google Scholar] [CrossRef]
- de Carvalho, C.C. Whole cell biocatalysts: Essential workers from Nature to the industry. Microb. Biotechnol. 2017, 10, 250–263. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, Z.; Cui, W.; Zhou, Z. Establishment of Bioprocess for Synthesis of Nicotinamide by Recombinant Escherichia coli Expressing High-Molecular-Mass Nitrile Hydratase. Appl. Biochem. Biotechnol. 2017, 182, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Raj, J.; Bhalla, T.C. Bench scale conversion of 3-cyanopyidine to nicotinamide using resting cells of Rhodococcus rhodochrous PA-34. Indian J. Microbiol. 2007, 47, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.D.; Cai, X.; Liu, Z.Q.; Zheng, Y.G. Nitrilase: A promising biocatalyst in industrial applications for green chemistry. Crit. Rev. Biotechnol. 2021, 41, 72–93. [Google Scholar] [CrossRef] [PubMed]
- Mathew, C.D.; Nagasawa, T.; Kobayashi, M.; Yamada, H. Nitrilase-Catalyzed Production of Nicotinic Acid from 3-Cyanopyridine in Rhodococcus rhodochrous J1. Appl. Environ. Microbiol. 1988, 54, 1030–1032. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Bhalla, T.C. Nitrile hydratases (NHases): At the interface of academia and industry. Biotechnol. Adv. 2010, 28, 725–741. [Google Scholar] [CrossRef]
- Nagasawa, T.; Mathew, C.D.; Mauger, J.; Yamada, H. Nitrile Hydratase-Catalyzed Production of Nicotinamide from 3-Cyanopyridine in Rhodococcus rhodochrous J1. Appl. Environ. Microbiol. 1988, 54, 1766–1769. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, T.C.; Kumar, V.; Kumar, V.; Thakur, N.; Savitri. Nitrile Metabolizing Enzymes in Biocatalysis and Biotransformation. Appl. Biochem. Biotechnol. 2018, 185, 925–946. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.S.; Lu, Z.M.; Li, H.; Shi, J.S.; Zhou, Z.M.; Xu, Z.H. Nitrilases in nitrile biocatalysis: Recent progress and forthcoming research. Microb. Cell Fact. 2012, 11, 142. [Google Scholar] [CrossRef]
- Gong, J.S.; Shi, J.S.; Lu, Z.M.; Li, H.; Zhou, Z.M.; Xu, Z.H. Nitrile-converting enzymes as a tool to improve biocatalysis in organic synthesis: Recent insights and promises. Crit. Rev. Biotechnol. 2017, 37, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Xia, Y.; Zhou, Z. Recent Advances and Promises in Nitrile Hydratase: From Mechanism to Industrial Applications. Front. Bioeng. Biotechnol. 2020, 8, 352. [Google Scholar] [CrossRef]
- Busch, H.; Hagedoorn, P.L.; Hanefeld, U. Rhodococcus as a Versatile Biocatalyst in Organic Synthesis. Int. J. Mol. Sci. 2019, 20, 4787. [Google Scholar] [CrossRef]
- Berner, L.A.; Clydesdale, F.M.; Douglass, J.S. Fortification contributed greatly to vitamin and mineral intakes in the United States, 1989–1991. J. Nutr. 2001, 131, 2177–2183. [Google Scholar] [CrossRef]
- Muthayya, S.; Hall, J.; Bagriansky, J.; Sugimoto, J.; Gundry, D.; Matthias, D.; Prigge, S.; Hindle, P.; Moench-Pfanner, R.; Maberly, G. Rice fortification: An emerging opportunity to contribute to the elimination of vitamin and mineral deficiency worldwide. Food Nutr. Bull. 2012, 33, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Ficca, M.; Kirkland, J.B. Niacin. Adv. Nutr. 2016, 7, 556–558. [Google Scholar] [CrossRef]
- De Dios Figueroa Cardenas, J.; Godinez, M.G.; Mendez, N.L.; Guzman, A.L.; Acosta, L.M. Nutritional quality of nixtamal tortillas fortified with vitamins and soy proteins. Int. J. Food Sci. Nutr. 2003, 54, 189–200. [Google Scholar] [CrossRef]
- Shewry, P.R.; Hawkesford, M.J.; Piironen, V.; Lampi, A.M.; Gebruers, K.; Boros, D.; Andersson, A.A.; Aman, P.; Rakszegi, M.; Bedo, Z.; et al. Natural variation in grain composition of wheat and related cereals. J. Agric. Food Chem. 2013, 61, 8295–8303. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Van Schaik, F.; Ravel, C.; Charmet, G.; Rakszegi, M.; Bedo, Z.; Ward, J.L. Genotype and environment effects on the contents of vitamins B1, B2, B3, and B6 in wheat grain. J. Agric. Food Chem. 2011, 59, 10564–10571. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.R.; Jung, E.S.; Lee, S.; Lim, S.H.; Ha, S.H.; Lee, C.H. Combined mass spectrometry-based metabolite profiling of different pigmented rice (Oryza sativa L.) seeds and correlation with antioxidant activities. Molecules 2014, 19, 15673–15686. [Google Scholar] [CrossRef] [PubMed]
- Gerdes, S.; Lerma-Ortiz, C.; Frelin, O.; Seaver, S.M.; Henry, C.S.; de Crecy-Lagard, V.; Hanson, A.D. Plant B vitamin pathways and their compartmentation: A guide for the perplexed. J. Exp. Bot. 2012, 63, 5379–5395. [Google Scholar] [CrossRef] [PubMed]
- Nuss, E.T.; Tanumihardjo, S.A. Quality protein maize for Africa: Closing the protein inadequacy gap in vulnerable populations. Adv. Nutr. 2011, 2, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, B.M.; Vasal, S.K.; Kassahun, B.; Singh, N.N. Quality protein maize. Curr. Sci. 2001, 81, 1308–1319. [Google Scholar]
- Prasanna, B.M.; Palacios-Rojas, N.; Hossain, F.; Muthusamy, V.; Menkir, A.; Dhliwayo, T.; Ndhlela, T.; San Vicente, F.; Nair, S.K.; Vivek, B.S.; et al. Molecular Breeding for Nutritionally Enriched Maize: Status and Prospects. Front. Genet. 2019, 10, 1392. [Google Scholar] [CrossRef] [PubMed]
- Goredema-Matongera, N.; Ndhlela, T.; Magorokosho, C.; Kamutando, C.N.; Van Biljon, A.; Labuschagne, M. Multinutrient Biofortification of Maize (Zea mays L.) in Africa: Current Status, Opportunities and Limitations. Nutrients 2021, 13, 1039. [Google Scholar] [CrossRef] [PubMed]
- Maqbool, M.A.; Issa, A.B.; Khokhar, E.S. Quality protein maize (QPM): Importance, genetics, timeline of different events, breeding strategies and varietal adoption. Plant Breed. 2021, 140, 375–399. [Google Scholar] [CrossRef]
- Bhat, J.S.; Patil, B.S.; Hariprasanna, K.; Hossain, F.; Muthusamy, V.; Mukri, G.; Mallikarjuna, M.G.; Zunjare, R.; Singh, S.P.; Sankar, S.M.; et al. Genetic Enhancement of Micronutrient Content in Cereals. SABRAO J. Breed. Genet. 2018, 50, 373–429. [Google Scholar]
- Coates, P.M.; Betz, J.M.; Blackman, M.R.; Cragg, G.M.; Levine, M.; Moss, J.; White, J.D. Encyclopedia of Dietary Supplements; Informa Healthcare: Boca Raton, FL, USA, 2010. [Google Scholar]
- Bechgaard, H.; Jespersen, S. GI absorption of niacin in humans. J. Pharm. Sci. 1977, 66, 871–872. [Google Scholar] [CrossRef] [PubMed]
- Lan, S.J.; Henderson, L.M. Uptake of nicotinic acid and nicotinamide by rat erythrocytes. J. Biol. Chem. 1968, 243, 3388–3394. [Google Scholar] [CrossRef]
- Revollo, J.R.; Grimm, A.A.; Imai, S. The NAD biosynthesis pathway mediated by nicotinamide phosphoribosyltransferase regulates Sir2 activity in mammalian cells. J. Biol. Chem. 2004, 279, 50754–50763. [Google Scholar] [CrossRef] [PubMed]
- Van der Veer, E.; Ho, C.; O’Neil, C.; Barbosa, N.; Scott, R.; Cregan, S.P.; Pickering, J.G. Extension of human cell lifespan by nicotinamide phosphoribosyltransferase. J. Biol. Chem. 2007, 282, 10841–10845. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, K.M.; Yoshino, J.; Brace, C.S.; Abrassart, D.; Kobayashi, Y.; Marcheva, B.; Hong, H.K.; Chong, J.L.; Buhr, E.D.; Lee, C.; et al. Circadian clock feedback cycle through NAMPT-mediated NAD+ biosynthesis. Science 2009, 324, 651–654. [Google Scholar] [CrossRef] [PubMed]
- Savitz, J. The kynurenine pathway: A finger in every pie. Mol. Psychiatry 2020, 25, 131–147. [Google Scholar] [CrossRef]
- Li, R.; Yu, K.; Wang, Q.; Wang, L.; Mao, J.; Qian, J. Pellagra Secondary to Medication and Alcoholism: A Case Report and Review of the Literature. Nutr. Clin. Pract. 2016, 31, 785–789. [Google Scholar] [CrossRef]
- Yang, H.; Yang, T.; Baur, J.A.; Perez, E.; Matsui, T.; Carmona, J.J.; Lamming, D.W.; Souza-Pinto, N.C.; Bohr, V.A.; Rosenzweig, A.; et al. Nutrient-sensitive mitochondrial NAD+ levels dictate cell survival. Cell 2007, 130, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Agledal, L.; Niere, M.; Ziegler, M. The phosphate makes a difference: Cellular functions of NADP. Redox Rep. 2010, 15, 2–10. [Google Scholar] [CrossRef]
- Piepho, R.W. The pharmacokinetics and pharmacodynamics of agents proven to raise high-density lipoprotein cholesterol. Am. J. Cardiol. 2000, 86, 35L–40L. [Google Scholar] [CrossRef]
- Pieper, J.A. Overview of niacin formulations: Differences in pharmacokinetics, efficacy, and safety. Am. J. Health Syst. Pharm. 2003, 60, S9–S14. [Google Scholar] [CrossRef] [PubMed]
- Wilkin, J.K.; Wilkin, O.; Kapp, R.; Donachie, R.; Chernosky, M.E.; Buckner, J. Aspirin blocks nicotinic acid-induced flushing. Clin. Pharmacol. Ther. 1982, 31, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Lenglet, A.; Liabeuf, S.; Bodeau, S.; Louvet, L.; Mary, A.; Boullier, A.; Lemaire-Hurtel, A.S.; Jonet, A.; Sonnet, P.; Kamel, S.; et al. N-methyl-2-pyridone-5-carboxamide (2PY)-Major Metabolite of Nicotinamide: An Update on an Old Uremic Toxin. Toxins 2016, 8, 339. [Google Scholar] [CrossRef]
- Breen, L.T.; Smyth, L.M.; Yamboliev, I.A.; Mutafova-Yambolieva, V.N. beta-NAD is a novel nucleotide released on stimulation of nerve terminals in human urinary bladder detrusor muscle. Am. J. Physiol. Ren. Physiol. 2006, 290, F486–F495. [Google Scholar] [CrossRef] [PubMed]
- Mutafova-Yambolieva, V.N. Neuronal and extraneuronal release of ATP and NAD(+) in smooth muscle. IUBMB Life 2012, 64, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Gruenbacher, G.; Gander, H.; Rahm, A.; Dobler, G.; Drasche, A.; Troppmair, J.; Nussbaumer, W.; Thurnher, M. The Human G Protein-Coupled ATP Receptor P2Y11 Is Associated With IL-10 Driven Macrophage Differentiation. Front. Immunol. 2019, 10, 1870. [Google Scholar] [CrossRef]
- Durnin, L.; Dai, Y.; Aiba, I.; Shuttleworth, C.W.; Yamboliev, I.A.; Mutafova-Yambolieva, V.N. Release, neuronal effects and removal of extracellular beta-nicotinamide adenine dinucleotide (beta-NAD(+)) in the rat brain. Eur. J. Neurosci. 2012, 35, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Durnin, L.; Kurahashi, M.; Sanders, K.M.; Mutafova-Yambolieva, V.N. Extracellular metabolism of the enteric inhibitory neurotransmitter beta-nicotinamide adenine dinucleotide (beta-NAD) in the murine colon. J. Physiol. 2020, 598, 4509–4521. [Google Scholar] [CrossRef] [PubMed]
- Umapathy, N.S.; Zemskov, E.A.; Gonzales, J.; Gorshkov, B.A.; Sridhar, S.; Chakraborty, T.; Lucas, R.; Verin, A.D. Extracellular beta-nicotinamide adenine dinucleotide (beta-NAD) promotes the endothelial cell barrier integrity via PKA- and EPAC1/Rac1-dependent actin cytoskeleton rearrangement. J. Cell Physiol. 2010, 223, 215–223. [Google Scholar] [CrossRef]
- Hiller, S.D.; Heldmann, S.; Richter, K.; Jurastow, I.; Kullmar, M.; Hecker, A.; Wilker, S.; Fuchs-Moll, G.; Manzini, I.; Schmalzing, G.; et al. beta-Nicotinamide Adenine Dinucleotide (beta-NAD) Inhibits ATP-Dependent IL-1beta Release from Human Monocytic Cells. Int. J. Mol. Sci. 2018, 19, 1126. [Google Scholar] [CrossRef]
- Nikiforov, A.; Kulikova, V.; Ziegler, M. The human NAD metabolome: Functions, metabolism and compartmentalization. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 284–297. [Google Scholar] [CrossRef]
- Pollak, N.; Dolle, C.; Ziegler, M. The power to reduce: Pyridine nucleotides—Small molecules with a multitude of functions. Biochem. J. 2007, 402, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Edenberg, H.J. The genetics of alcohol metabolism: Role of alcohol dehydrogenase and aldehyde dehydrogenase variants. Alcohol Res. Health 2007, 30, 5–13. [Google Scholar] [PubMed]
- Nakahata, Y.; Sahar, S.; Astarita, G.; Kaluzova, M.; Sassone-Corsi, P. Circadian control of the NAD+ salvage pathway by CLOCK-SIRT1. Science 2009, 324, 654–657. [Google Scholar] [CrossRef]
- Frederick, D.W.; Davis, J.G.; Davila, A., Jr.; Agarwal, B.; Michan, S.; Puchowicz, M.A.; Nakamaru-Ogiso, E.; Baur, J.A. Increasing NAD synthesis in muscle via nicotinamide phosphoribosyltransferase is not sufficient to promote oxidative metabolism. J. Biol. Chem. 2015, 290, 1546–1558. [Google Scholar] [CrossRef]
- Canto, C.; Menzies, K.J.; Auwerx, J. NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab. 2015, 22, 31–53. [Google Scholar] [CrossRef]
- Guse, A.H.; Lee, H.C. NAADP: A universal Ca2+ trigger. Sci. Signal. 2008, 1, re10. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, E.; Lo Buono, N.; Horenstein, A.L.; Funaro, A.; Malavasi, F. The ADP-ribosyl cyclases—The current evolutionary state of the ARCs. Front. Biosci. 2014, 19, 986–1002. [Google Scholar] [CrossRef] [PubMed]
- Partida-Sanchez, S.; Cockayne, D.A.; Monard, S.; Jacobson, E.L.; Oppenheimer, N.; Garvy, B.; Kusser, K.; Goodrich, S.; Howard, M.; Harmsen, A.; et al. Cyclic ADP-ribose production by CD38 regulates intracellular calcium release, extracellular calcium influx and chemotaxis in neutrophils and is required for bacterial clearance in vivo. Nat. Med. 2001, 7, 1209–1216. [Google Scholar] [CrossRef]
- Chong, A.; Malavasi, F.; Israel, S.; Khor, C.C.; Yap, V.B.; Monakhov, M.; Chew, S.H.; Lai, P.S.; Ebstein, R.P. ADP ribosyl-cyclases (CD38/CD157), social skills and friendship. Psychoneuroendocrinology 2017, 78, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.L. PARPs. Curr. Biol. 2017, 27, R1256–R1258. [Google Scholar] [CrossRef] [PubMed]
- Perina, D.; Mikoc, A.; Ahel, J.; Cetkovic, H.; Zaja, R.; Ahel, I. Distribution of protein poly (ADP-ribosyl) ation systems across all domains of life. DNA Repair 2014, 23, 4–16. [Google Scholar] [CrossRef]
- Aravind, L.; Zhang, D.; de Souza, R.F.; Anand, S.; Iyer, L.M. The natural history of ADP-ribosyltransferases and the ADP-ribosylation system. Curr. Top. Microbiol. Immunol. 2015, 384, 3–32. [Google Scholar] [CrossRef] [PubMed]
- Trucco, C.; Rolli, V.; Oliver, F.J.; Flatter, E.; Masson, M.; Dantzer, F.; Niedergang, C.; Dutrillaux, B.; Menissier-de Murcia, J.; de Murcia, G. A dual approach in the study of poly (ADP-ribose) polymerase: In vitro random mutagenesis and generation of deficient mice. Mol. Cell. Biochem. 1999, 193, 53–60. [Google Scholar] [CrossRef]
- Shall, S.; de Murcia, G. Poly (ADP-ribose) polymerase-1: What have we learned from the deficient mouse model? Mutat. Res. 2000, 460, 1–15. [Google Scholar] [CrossRef]
- Herceg, Z.; Wang, Z.Q. Functions of poly (ADP-ribose) polymerase (PARP) in DNA repair, genomic integrity and cell death. Mutat. Res. 2001, 477, 97–110. [Google Scholar] [CrossRef]
- Berger, N.A.; Besson, V.C.; Boulares, A.H.; Burkle, A.; Chiarugi, A.; Clark, R.S.; Curtin, N.J.; Cuzzocrea, S.; Dawson, T.M.; Dawson, V.L.; et al. Opportunities for the repurposing of PARP inhibitors for the therapy of non-oncological diseases. Br. J. Pharmacol. 2018, 175, 192–222. [Google Scholar] [CrossRef]
- Buisson, R.; Dion-Cote, A.M.; Coulombe, Y.; Launay, H.; Cai, H.; Stasiak, A.Z.; Stasiak, A.; Xia, B.; Masson, J.Y. Cooperation of breast cancer proteins PALB2 and piccolo BRCA2 in stimulating homologous recombination. Nat. Struct. Mol. Biol. 2010, 17, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Bochum, S.; Berger, S.; Martens, U.M. Olaparib. Recent Results Cancer Res. 2018, 211, 217–233. [Google Scholar] [CrossRef]
- Lord, C.J.; Ashworth, A. PARP inhibitors: Synthetic lethality in the clinic. Science 2017, 355, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, L.; Mikoc, A.; Ahel, I. ADP-ribosylation: New facets of an ancient modification. FEBS J. 2017, 284, 2932–2946. [Google Scholar] [CrossRef]
- Moraes, D.S.; Moreira, D.C.; Andrade, J.M.O.; Santos, S.H.S. Sirtuins, brain and cognition: A review of resveratrol effects. IBRO Rep. 2020, 9, 46–51. [Google Scholar] [CrossRef]
- Tanner, K.G.; Landry, J.; Sternglanz, R.; Denu, J.M. Silent information regulator 2 family of NAD- dependent histone/protein deacetylases generates a unique product, 1-O-acetyl-ADP-ribose. Proc. Natl. Acad. Sci. USA 2000, 97, 14178–14182. [Google Scholar] [CrossRef]
- Asher, G.; Gatfield, D.; Stratmann, M.; Reinke, H.; Dibner, C.; Kreppel, F.; Mostoslavsky, R.; Alt, F.W.; Schibler, U. SIRT1 regulates circadian clock gene expression through PER2 deacetylation. Cell 2008, 134, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Nakahata, Y.; Kaluzova, M.; Grimaldi, B.; Sahar, S.; Hirayama, J.; Chen, D.; Guarente, L.P.; Sassone-Corsi, P. The NAD+-dependent deacetylase SIRT1 modulates CLOCK-mediated chromatin remodeling and circadian control. Cell 2008, 134, 329–340. [Google Scholar] [CrossRef]
- Imai, S.I.; Guarente, L. It takes two to tango: NAD(+) and sirtuins in aging/longevity control. NPJ Aging Mech. Dis. 2016, 2, 16017. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liu, J.; Chen, D.; Yan, L.; Zheng, W. Sirtuin Inhibition: Strategies, Inhibitors, and Therapeutic Potential. Trends Pharmacol. Sci. 2017, 38, 459–472. [Google Scholar] [CrossRef]
- Van de Ven, R.A.H.; Santos, D.; Haigis, M.C. Mitochondrial Sirtuins and Molecular Mechanisms of Aging. Trends Mol. Med. 2017, 23, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Bayele, H.K. Sirtuins transduce STACs signals through steroid hormone receptors. Sci. Rep. 2020, 10, 5338. [Google Scholar] [CrossRef] [PubMed]
- Gaal, Z.; Csernoch, L. Impact of Sirtuin Enzymes on the Altered Metabolic Phenotype of Malignantly Transformed Cells. Front. Oncol. 2020, 10, 45. [Google Scholar] [CrossRef]
- Carpenter, K.J. Pellagra; Hutchinson Ross: Stroudsburg, PA, USA, 1981. [Google Scholar]
- Prabhu, D.; Dawe, R.S.; Mponda, K. Pellagra a review exploring causes and mechanisms, including isoniazid-induced pellagra. Photodermatol. Photoimmunol. Photomed. 2021, 37, 99–104. [Google Scholar] [CrossRef]
- Williams, A.C.; Hill, L.J. The 4 D’s of Pellagra and Progress. Int. J. Tryptophan Res. 2020, 13, 1178646920910159. [Google Scholar] [CrossRef]
- Ramirez-Cabral, N.Y.Z.; Kumar, L.; Shabani, F. Global alterations in areas of suitability for maize production from climate change and using a mechanistic species distribution model (CLIMEX). Sci. Rep. 2017, 7, 5910. [Google Scholar] [CrossRef]
- Schmid, M.A.; Salomeyesudas, B.; Satheesh, P.; Hanley, J.; Kuhnlein, H.V. Intervention with traditional food as a major source of energy, protein, iron, vitamin C and vitamin A for rural Dalit mothers and young children in Andhra Pradesh, South India. Asia Pac. J. Clin. Nutr. 2007, 16, 84–93. [Google Scholar]
- Malfait, P.; Moren, A.; Dillon, J.C.; Brodel, A.; Begkoyian, G.; Etchegorry, M.G.; Malenga, G.; Hakewill, P. An outbreak of pellagra related to changes in dietary niacin among Mozambican refugees in Malawi. Int. J. Epidemiol. 1993, 22, 504–511. [Google Scholar] [CrossRef]
- Altschul, R.; Hoffer, A.; Stephen, J.D. Influence of nicotinic acid on serum cholesterol in man. Arch. Biochem. Biophys. 1955, 54, 558–559. [Google Scholar] [CrossRef]
- Carlson, L.A. Nicotinic acid: The broad-spectrum lipid drug. A 50th anniversary review. J. Intern. Med. 2005, 258, 94–114. [Google Scholar] [CrossRef] [PubMed]
- Wise, A.; Foord, S.M.; Fraser, N.J.; Barnes, A.A.; Elshourbagy, N.; Eilert, M.; Ignar, D.M.; Murdock, P.R.; Steplewski, K.; Green, A.; et al. Molecular identification of high and low affinity receptors for nicotinic acid. J. Biol. Chem. 2003, 278, 9869–9874. [Google Scholar] [CrossRef]
- Soga, T.; Kamohara, M.; Takasaki, J.; Matsumoto, S.; Saito, T.; Ohishi, T.; Hiyama, H.; Matsuo, A.; Matsushime, H.; Furuichi, K. Molecular identification of nicotinic acid receptor. Biochem. Biophys. Res. Commun. 2003, 303, 364–369. [Google Scholar] [CrossRef]
- Taggart, A.K.; Kero, J.; Gan, X.; Cai, T.Q.; Cheng, K.; Ippolito, M.; Ren, N.; Kaplan, R.; Wu, K.; Wu, T.J.; et al. (D)-beta-Hydroxybutyrate inhibits adipocyte lipolysis via the nicotinic acid receptor PUMA-G. J. Biol. Chem. 2005, 280, 26649–26652. [Google Scholar] [CrossRef]
- Gille, A.; Bodor, E.T.; Ahmed, K.; Offermanns, S. Nicotinic acid: Pharmacological effects and mechanisms of action. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 79–106. [Google Scholar] [CrossRef]
- Carlson, L.A.; Oro, L.; Ostman, J. Effect of a single dose of nicotinic acid on plasma lipids in patients with hyperlipoproteinemia. Acta Med. Scand. 1968, 183, 457–465. [Google Scholar] [CrossRef]
- Ganji, S.H.; Tavintharan, S.; Zhu, D.; Xing, Y.; Kamanna, V.S.; Kashyap, M.L. Niacin noncompetitively inhibits DGAT2 but not DGAT1 activity in HepG2 cells. J. Lipid Res. 2004, 45, 1835–1845. [Google Scholar] [CrossRef]
- Jin, F.Y.; Kamanna, V.S.; Kashyap, M.L. Niacin accelerates intracellular ApoB degradation by inhibiting triacylglycerol synthesis in human hepatoblastoma (HepG2) cells. Arter. Thromb. Vasc. Biol. 1999, 19, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Svedmyr, N.; Harthon, L.; Lundholm, L. The relationship between the plasma concentration of free nicotinic acid and some of its pharmacologic effects in man. Clin. Pharmacol. Ther. 1969, 10, 559–570. [Google Scholar] [CrossRef]
- Barter, P.J.; Brewer, H.B., Jr.; Chapman, M.J.; Hennekens, C.H.; Rader, D.J.; Tall, A.R. Cholesteryl ester transfer protein: A novel target for raising HDL and inhibiting atherosclerosis. Arter. Thromb. Vasc. Biol. 2003, 23, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, M.; Wright, S.D.; Cai, T.Q. Critical role of cholesterol ester transfer protein in nicotinic acid-mediated HDL elevation in mice. Biochem. Biophys. Res. Commun. 2007, 355, 1075–1080. [Google Scholar] [CrossRef]
- Le Goff, W.; Guerin, M.; Chapman, M.J. Pharmacological modulation of cholesteryl ester transfer protein, a new therapeutic target in atherogenic dyslipidemia. Pharmacol. Ther. 2004, 101, 17–38. [Google Scholar] [CrossRef]
- Mousa, S.S.; Block, R.C.; Mousa, S.A. High Density Lipoprotein (HDL) Modulation Targets. Drugs Future 2010, 35, 33–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Olsson, A.G. Nicotinic Acid and Derivatives. In Principles and Treatment of Lipoprotein Disorders; Handbook of Experimental, Pharmacology; Schettler, G., Habenicht, A.J.R., Eds.; Springer: Berlin/Heidelberg, Germany, 1994; pp. 349–400. [Google Scholar]
- Jin, F.Y.; Kamanna, V.S.; Kashyap, M.L. Niacin decreases removal of high-density lipoprotein apolipoprotein A-I but not cholesterol ester by Hep G2 cells. Implication for reverse cholesterol transport. Arter. Thromb. Vasc. Biol. 1997, 17, 2020–2028. [Google Scholar] [CrossRef]
- Meyers, C.D.; Kashyap, M.L. Pharmacologic elevation of high-density lipoproteins: Recent insights on mechanism of action and atherosclerosis protection. Curr. Opin. Cardiol. 2004, 19, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.H.; Zhao, S.P. Niacin promotes cholesterol efflux through stimulation of the PPARgamma-LXRalpha-ABCA1 pathway in 3T3-L1 adipocytes. Pharmacology 2009, 84, 282–287. [Google Scholar] [CrossRef]
- Knowles, H.J.; te Poele, R.H.; Workman, P.; Harris, A.L. Niacin induces PPARgamma expression and transcriptional activation in macrophages via HM74 and HM74a-mediated induction of prostaglandin synthesis pathways. Biochem. Pharmacol. 2006, 71, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Oram, J.F.; Lawn, R.M.; Garvin, M.R.; Wade, D.P. ABCA1 is the cAMP-inducible apolipoprotein receptor that mediates cholesterol secretion from macrophages. J. Biol. Chem. 2000, 275, 34508–34511. [Google Scholar] [CrossRef] [PubMed]
- Yvan-Charvet, L.; Wang, N.; Tall, A.R. Role of HDL, ABCA1, and ABCG1 transporters in cholesterol efflux and immune responses. Arter. Thromb. Vasc. Biol. 2010, 30, 139–143. [Google Scholar] [CrossRef]
- Takahashi, Y.; Miyata, M.; Zheng, P.; Imazato, T.; Horwitz, A.; Smith, J.D. Identification of cAMP analogue inducible genes in RAW264 macrophages. Biochim. Biophys. Acta 2000, 1492, 385–394. [Google Scholar] [CrossRef]
- Zhao, S.P.; Yang, J.; Li, J.; Dong, S.Z.; Wu, Z.H. Effect of niacin on LXRalpha and PPARgamma expression and HDL-induced cholesterol efflux in adipocytes of hypercholesterolemic rabbits. Int. J. Cardiol. 2008, 124, 172–178. [Google Scholar] [CrossRef]
- Johnson, S.; Imai, S.I. NAD(+) biosynthesis, aging, and disease. F1000Research 2018, 7, 132. [Google Scholar] [CrossRef]
- Imai, S. From heterochromatin islands to the NAD World: A hierarchical view of aging through the functions of mammalian Sirt1 and systemic NAD biosynthesis. Biochim. Biophys. Acta 2009, 1790, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Imai, S. Dissecting systemic control of metabolism and aging in the NAD World: The importance of SIRT1 and NAMPT-mediated NAD biosynthesis. FEBS Lett. 2011, 585, 1657–1662. [Google Scholar] [CrossRef]
- Rehan, L.; Laszki-Szczachor, K.; Sobieszczanska, M.; Polak-Jonkisz, D. SIRT1 and NAD as regulators of ageing. Life Sci. 2014, 105, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Poljsak, B. NAMPT-Mediated NAD Biosynthesis as the Internal Timing Mechanism: In NAD+ World, Time Is Running in Its Own Way. Rejuvenation Res. 2018, 21, 210–224. [Google Scholar] [CrossRef]
- Massudi, H.; Grant, R.; Braidy, N.; Guest, J.; Farnsworth, B.; Guillemin, G.J. Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PLoS ONE 2012, 7, e42357. [Google Scholar] [CrossRef]
- Schultz, M.B.; Sinclair, D.A. Why NAD(+) Declines during Aging: It’s Destroyed. Cell Metab. 2016, 23, 965–966. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Pereira, J.; Tarrago, M.G.; Chini, C.C.S.; Nin, V.; Escande, C.; Warner, G.M.; Puranik, A.S.; Schoon, R.A.; Reid, J.M.; Galina, A.; et al. CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metab. 2016, 23, 1127–1139. [Google Scholar] [CrossRef] [PubMed]
- Mao, K.; Zhang, G. The role of PARP1 in neurodegenerative diseases and aging. FEBS J. 2021. [Google Scholar] [CrossRef]
- Covarrubias, A.J.; Perrone, R.; Grozio, A.; Verdin, E. NAD(+) metabolism and its roles in cellular processes during ageing. Nat. Rev. Mol. Cell Biol. 2021, 22, 119–141. [Google Scholar] [CrossRef]
- Bai, P.; Canto, C.; Oudart, H.; Brunyanszki, A.; Cen, Y.; Thomas, C.; Yamamoto, H.; Huber, A.; Kiss, B.; Houtkooper, R.H.; et al. PARP-1 inhibition increases mitochondrial metabolism through SIRT1 activation. Cell Metab. 2011, 13, 461–468. [Google Scholar] [CrossRef]
- Belenky, P.; Racette, F.G.; Bogan, K.L.; McClure, J.M.; Smith, J.S.; Brenner, C. Nicotinamide riboside promotes Sir2 silencing and extends lifespan via Nrk and Urh1/Pnp1/Meu1 pathways to NAD+. Cell 2007, 129, 473–484. [Google Scholar] [CrossRef]
- Canto, C.; Houtkooper, R.H.; Pirinen, E.; Youn, D.Y.; Oosterveer, M.H.; Cen, Y.; Fernandez-Marcos, P.J.; Yamamoto, H.; Andreux, P.A.; Cettour-Rose, P.; et al. The NAD(+) precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell Metab. 2012, 15, 838–847. [Google Scholar] [CrossRef]
- Wang, X.; Li, H.; Ding, S. The effects of NAD+ on apoptotic neuronal death and mitochondrial biogenesis and function after glutamate excitotoxicity. Int. J. Mol. Sci. 2014, 15, 20449–20468. [Google Scholar] [CrossRef] [PubMed]
- Yaku, K.; Okabe, K.; Hikosaka, K.; Nakagawa, T. NAD Metabolism in Cancer Therapeutics. Front. Oncol. 2018, 8, 622. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.E.; Sharif, T.; Martell, E.; Dai, C.; Kim, Y.; Lee, P.W.; Gujar, S.A. NAD(+) salvage pathway in cancer metabolism and therapy. Pharmacol. Res. 2016, 114, 274–283. [Google Scholar] [CrossRef]
- Wakade, C.; Chong, R.; Bradley, E.; Thomas, B.; Morgan, J. Upregulation of GPR109A in Parkinson’s disease. PLoS ONE 2014, 9, e109818. [Google Scholar] [CrossRef] [PubMed]
- Wakade, C.; Chong, R.; Bradley, E.; Morgan, J.C. Low-dose niacin supplementation modulates GPR109A, niacin index and ameliorates Parkinson’s disease symptoms without side effects. Clin. Case Rep. 2015, 3, 635–637. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Li, X.; Gao, H.; Feng, Z.; Li, X.; Zhao, L.; Jia, X.; Zhang, H.; Liu, J. High doses of nicotinamide prevent oxidative mitochondrial dysfunction in a cellular model and improve motor deficit in a Drosophila model of Parkinson’s disease. J. Neurosci. Res. 2008, 86, 2083–2090. [Google Scholar] [CrossRef] [PubMed]
- Nimmagadda, V.K.; Makar, T.K.; Chandrasekaran, K.; Sagi, A.R.; Ray, J.; Russell, J.W.; Bever, C.T., Jr. SIRT1 and NAD+ precursors: Therapeutic targets in multiple sclerosis a review. J. Neuroimmunol. 2017, 304, 29–34. [Google Scholar] [CrossRef]
- Kim, S.Y.; Cohen, B.M.; Chen, X.; Lukas, S.E.; Shinn, A.K.; Yuksel, A.C.; Li, T.; Du, F.; Ongur, D. Redox Dysregulation in Schizophrenia Revealed by in vivo NAD+/NADH Measurement. Schizophr. Bull. 2017, 43, 197–204. [Google Scholar] [CrossRef]
- Benavente, C.A.; Schnell, S.A.; Jacobson, E.L. Effects of niacin restriction on sirtuin and PARP responses to photodamage in human skin. PLoS ONE 2012, 7, e42276. [Google Scholar] [CrossRef] [PubMed]
- Gensler, H.L.; Williams, T.; Huang, A.C.; Jacobson, E.L. Oral niacin prevents photocarcinogenesis and photoimmunosuppression in mice. Nutr. Cancer 1999, 34, 36–41. [Google Scholar] [CrossRef]
- Gehring, W. Nicotinic acid/niacinamide and the skin. J. Cosmet. Dermatol. 2004, 3, 88–93. [Google Scholar] [CrossRef]
- Levine, D.; Even-Chen, Z.; Lipets, I.; Pritulo, O.A.; Svyatenko, T.V.; Andrashko, Y.; Lebwohl, M.; Gottlieb, A. Pilot, multicenter, double-blind, randomized placebo-controlled bilateral comparative study of a combination of calcipotriene and nicotinamide for the treatment of psoriasis. J. Am. Acad. Dermatol. 2010, 63, 775–781. [Google Scholar] [CrossRef]
- Park, S.M.; Li, T.; Wu, S.; Li, W.Q.; Weinstock, M.; Qureshi, A.A.; Cho, E. Niacin intake and risk of skin cancer in US women and men. Int. J. Cancer 2017, 140, 2023–2031. [Google Scholar] [CrossRef]
- Oberwittler, H.; Baccara-Dinet, M. Clinical evidence for use of acetyl salicylic acid in control of flushing related to nicotinic acid treatment. Int. J. Clin. Pract. 2006, 60, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Benyo, Z.; Gille, A.; Bennett, C.L.; Clausen, B.E.; Offermanns, S. Nicotinic acid-induced flushing is mediated by activation of epidermal langerhans cells. Mol. Pharmacol. 2006, 70, 1844–1849. [Google Scholar] [CrossRef]
- Hanson, J.; Gille, A.; Zwykiel, S.; Lukasova, M.; Clausen, B.E.; Ahmed, K.; Tunaru, S.; Wirth, A.; Offermanns, S. Nicotinic acid- and monomethyl fumarate-induced flushing involves GPR109A expressed by keratinocytes and COX-2-dependent prostanoid formation in mice. J. Clin. Investig. 2010, 120, 2910–2919. [Google Scholar] [CrossRef]
- Hay, D.L.; Poyner, D.R. Calcitonin gene-related peptide, adrenomedullin and flushing. Maturitas 2009, 64, 104–108. [Google Scholar] [CrossRef]
- Wierzbicki, A.S. Niacin: The only vitamin that reduces cardiovascular events. Int. J. Clin. Pr. 2011, 65, 379–385. [Google Scholar] [CrossRef] [PubMed]
- McKenney, J.M.; Proctor, J.D.; Harris, S.; Chinchili, V.M. A comparison of the efficacy and toxic effects of sustained-vs immediate-release niacin in hypercholesterolemic patients. JAMA 1994, 271, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Dalton, T.A.; Berry, R.S. Hepatotoxicity associated with sustained-release niacin. Am. J. Med. 1992, 93, 102–104. [Google Scholar] [CrossRef]
- Lawrence, S.P. Transient focal hepatic defects related to sustained-release niacin. J. Clin. Gastroenterol. 1993, 16, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Wu, T.J.; Wu, K.K.; Sturino, C.; Metters, K.; Gottesdiener, K.; Wright, S.D.; Wang, Z.; O’Neill, G.; Lai, E.; et al. Antagonism of the prostaglandin D2 receptor 1 suppresses nicotinic acid-induced vasodilation in mice and humans. Proc. Natl. Acad. Sci. USA 2006, 103, 6682–6687. [Google Scholar] [CrossRef]
- Parsons, W.B., Jr. Activation of peptic ulcer by nicotinic acid. Report of five cases. JAMA 1960, 173, 1466–1470. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, D.K.; Kahn, S.E.; Schwartz, M.W.; Koerker, D.J.; Palmer, J.P. Effect of nicotinic acid-induced insulin resistance on pancreatic B cell function in normal and streptozocin-treated baboons. J. Clin. Investig. 1991, 87, 1395–1401. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garg, A.; Grundy, S.M. Nicotinic acid as therapy for dyslipidemia in non-insulin-dependent diabetes mellitus. JAMA 1990, 264, 723–726. [Google Scholar] [CrossRef]
- Canner, P.L.; Furberg, C.D.; Terrin, M.L.; McGovern, M.E. Benefits of niacin by glycemic status in patients with healed myocardial infarction (from the Coronary Drug Project). Am. J. Cardiol. 2005, 95, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Elam, M.B.; Hunninghake, D.B.; Davis, K.B.; Garg, R.; Johnson, C.; Egan, D.; Kostis, J.B.; Sheps, D.S.; Brinton, E.A. Effect of niacin on lipid and lipoprotein levels and glycemic control in patients with diabetes and peripheral arterial disease: The ADMIT study: A randomized trial. Arterial Disease Multiple Intervention Trial. JAMA 2000, 284, 1263–1270. [Google Scholar] [CrossRef]
- Grundy, S.M.; Vega, G.L.; McGovern, M.E.; Tulloch, B.R.; Kendall, D.M.; Fitz-Patrick, D.; Ganda, O.P.; Rosenson, R.S.; Buse, J.B.; Robertson, D.D.; et al. Efficacy, safety, and tolerability of once-daily niacin for the treatment of dyslipidemia associated with type 2 diabetes: Results of the assessment of diabetes control and evaluation of the efficacy of niaspan trial. Arch. Intern. Med. 2002, 162, 1568–1576. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.S.; Song, S.B. Possible Adverse Effects of High-Dose Nicotinamide: Mechanisms and Safety Assessment. Biomolecules 2020, 10, 687. [Google Scholar] [CrossRef]
- Williams, R.J.; Bradway, E.M. The further fractination of yeast nutrilites and their relationship to vitamin B and Wildiers’ "bios". J. Am. Chem. Soc. 1931, 53, 783–789. [Google Scholar] [CrossRef]
- Williams, R.J.; Lyman, C.M.; Goodyear, G.H.; Truesdail, J.H.; Holaday, D. “Pantothenic Acid,” a Growth Determinant of Universal Biological Occurrence. J. Am. Chem. Soc. 1933, 55, 2912–2927. [Google Scholar] [CrossRef]
- Müller, M.A.; Medlock, J.; Prágai, Z.; Warnke, I.; Litta, G.; Kleefeldt, A.; Kaiser, K.; De Potzolli, B. Vitamins, 9. Vitamin B5. In Ullmann’s Encyclopedia of Industrial Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 1–16. [Google Scholar]
- Gonzalez-Lopez, J.; Aliaga, L.; Gonzalez-Martinez, A.; Martinez-Toledo, M.V. Pantothenic Acid. In Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; pp. 67–101. [Google Scholar]
- Schnellbaecher, A.; Binder, D.; Bellmaine, S.; Zimmer, A. Vitamins in cell culture media: Stability and stabilization strategies. Biotechnol. Bioeng. 2019, 116, 1537–1555. [Google Scholar] [CrossRef]
- Webb, M.E.; Smith, A.G.; Abell, C. Biosynthesis of pantothenate. Nat. Prod. Rep. 2004, 21, 695–721. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, R.; Jackowski, S. Biosynthesis of Pantothenic Acid and Coenzyme A. EcoSal Plus 2007, 2. [Google Scholar] [CrossRef]
- Martinez, D.L.; Tsuchiya, Y.; Gout, I. Coenzyme A biosynthetic machinery in mammalian cells. Biochem. Soc. Trans. 2014, 42, 1112–1117. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, R.; Zhang, Y.M.; Rock, C.O.; Jackowski, S. Coenzyme A: Back in action. Prog. Lipid Res. 2005, 44, 125–153. [Google Scholar] [CrossRef] [PubMed]
- Ottenhof, H.H.; Ashurst, J.L.; Whitney, H.M.; Saldanha, S.A.; Schmitzberger, F.; Gweon, H.S.; Blundell, T.L.; Abell, C.; Smith, A.G. Organisation of the pantothenate (vitamin B5) biosynthesis pathway in higher plants. Plant J. 2004, 37, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Chakauya, E.; Coxon, K.M.; Whitney, H.M.; Ashurst, J.L.; Abell, C.; Smith, A.G. Pantothenate biosynthesis in higher plants: Advances and challenges. Physiol. Plant 2006, 126, 319–329. [Google Scholar] [CrossRef]
- Webb, M.E.; Smith, A.G. Pantothenate Biosynthesis in Higher Plants. In Advances in Botanical Research; Rébeillé, F., Douce, R., Eds.; Academic Press: Cambridge, MA, USA, 2011; Volume 58, pp. 203–255. [Google Scholar]
- Webb, M.E.; Marquet, A.; Mendel, R.R.; Rebeille, F.; Smith, A.G. Elucidating biosynthetic pathways for vitamins and cofactors. Nat. Prod. Rep. 2007, 24, 988–1008. [Google Scholar] [CrossRef]
- White, W.H.; Gunyuzlu, P.L.; Toyn, J.H. Saccharomyces cerevisiae is capable of de Novo pantothenic acid biosynthesis involving a novel pathway of beta-alanine production from spermine. J. Biol. Chem. 2001, 276, 10794–10800. [Google Scholar] [CrossRef] [PubMed]
- Spry, C.; Kirk, K.; Saliba, K.J. Coenzyme A biosynthesis: An antimicrobial drug target. FEMS Microbiol. Rev. 2008, 32, 56–106. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.M.; Song, W.O. Comparative nutrition of pantothenic acid. J. Nutr. Biochem. 1996, 7, 312–321. [Google Scholar] [CrossRef]
- Roje, S. Vitamin B biosynthesis in plants. Phytochemistry 2007, 68, 1904–1921. [Google Scholar] [CrossRef]
- Coxon, K.M.; Chakauya, E.; Ottenhof, H.H.; Whitney, H.M.; Blundell, T.L.; Abell, C.; Smith, A.G. Pantothenate biosynthesis in higher plants. Biochem. Soc. Trans. 2005, 33, 743–746. [Google Scholar] [CrossRef]
- Miller, J.W.; Rucker, R.B. Pantothenic Acid. In Present Knowledge in Nutrition; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 375–390. [Google Scholar]
- Walsh, J.H.; Wyse, B.W.; Hansen, R.G. Pantothenic acid content of 75 processed and cooked foods. J. Am. Diet. Assoc. 1981, 78, 140–144. [Google Scholar] [CrossRef]
- Scientific Committee on Food. Tolerable Upper Intake Levels for Vitamins and Minerals; EFSA: Parma, Italy, 2006. [Google Scholar]
- Kelly, G.S. Pantothenic acid. Monograph. Altern. Med. Rev. 2011, 16, 263–274. [Google Scholar] [PubMed]
- Willerton, E.; Cromwell, H. Microbiologic Assay of Natural Pantothenic Acid in Yeast and Liver. Influence of Clarase Digestion. Ind. Eng. Chem. Anal. Ed. 2002, 14, 603–604. [Google Scholar] [CrossRef]
- Ball, G.F.M. Pantothenic acid. In Bioavailability and Analysis of Vitamins in Foods; Springer US: Boston, MA, USA, 1998; pp. 409–422. [Google Scholar]
- Hall, A.P.; Moore, J.G.; Morgan, A.F. B Vitamin content of avocados: Studies reveal California-grown avocados are in superior group of foods as source of pantothenic acid and vitamin B. Calif. Agric. 1956, 10, 13–14. [Google Scholar]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on Dietary Reference Values for pantothenic acid. EFSA J. 2014, 12, 3581. [Google Scholar] [CrossRef][Green Version]
- Ciulu, M.; Floris, I.; Nurchi, V.M.; Panzanelli, A.; Pilo, M.I.; Spano, N.; Sanna, G. HPLC determination of pantothenic acid in royal jelly. Anal. Methods 2013, 5, 6682–6685. [Google Scholar] [CrossRef]
- Kunugi, H.; Mohammed Ali, A. Royal Jelly and Its Components Promote Healthy Aging and Longevity: From Animal Models to Humans. Int. J. Mol. Sci. 2019, 20, 4662. [Google Scholar] [CrossRef] [PubMed]
- Pearson, P.B.; Burgin, C.J. The Pantothenic Acid Content of Royal Jelly. Exp. Biol. Med. 1941, 48, 415–417. [Google Scholar] [CrossRef]
- Uebanso, T.; Shimohata, T.; Mawatari, K.; Takahashi, A. Functional Roles of B-Vitamins in the Gut and Gut Microbiome. Mol. Nutr. Food Res. 2020, 64, e2000426. [Google Scholar] [CrossRef]
- Bates, C.J. Pantothenic Acid. In Encyclopedia of Human Nutrition; Caballero, B., Ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 1–5. [Google Scholar]
- Said, H.M.; Mohammed, Z.M. Intestinal absorption of water-soluble vitamins: An update. Curr. Opin. Gastroenterol. 2006, 22, 140–146. [Google Scholar] [CrossRef]
- Said, H.M.; Ortiz, A.; McCloud, E.; Dyer, D.; Moyer, M.P.; Rubin, S. Biotin uptake by human colonic epithelial NCM460 cells: A carrier-mediated process shared with pantothenic acid. Am. J. Physiol. 1998, 275, C1365–C1371. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, A.; Lambrecht, N.; Subramanya, S.B.; Kapadia, R.; Said, H.M. Conditional knockout of the Slc5a6 gene in mouse intestine impairs biotin absorption. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G64–G71. [Google Scholar] [CrossRef]
- Said, H.M. Intestinal absorption of water-soluble vitamins in health and disease. Biochem J. 2011, 437, 357–372. [Google Scholar] [CrossRef]
- MacDonald, R.; Reitmeier, C. (Eds.) Food Processing. In Understanding Food Systems; Academic Press: Cambridge, MA, USA, 2017; pp. 179–225. [Google Scholar]
- Schroeder, H.A. Losses of vitamins and trace minerals resulting from processing and preservation of foods. Am. J. Clin. Nutr. 1971, 24, 562–573. [Google Scholar] [CrossRef]
- Bodwell, C.; Anderson, B. Nutritional Composition and Value of Meat and Meat Products; Academic Press: Cambridge, MA, USA, 1986; pp. 321–369. [Google Scholar]
- Engler, P.P.; Bowers, J.A. B-vitamin retention in meat during storage and preparation. A review. J. Am. Diet. Assoc. 1976, 69, 253–257. [Google Scholar] [CrossRef]
- Meyer, B.H.; Mysinger, M.A.; Wodarski, L.A. Pantothenic acid and vitamin B6 in beef. J. Am. Diet Assoc. 1969, 54, 122–125. [Google Scholar] [CrossRef]
- Cheng, T.S.; Eitenmiller, R.R. Effects of Processing and Storage on the Pantothenic-Acid Content of Spinach and Broccoli. J. Food Process. Preserv. 1988, 12, 115–123. [Google Scholar] [CrossRef]
- Hoppner, K.; Lampi, B. Pantothenic-Acid and Biotin Retention in Cooked Legumes. J. Food Sci. 1993, 58, 1084–1085. [Google Scholar] [CrossRef]
- Khalil, A.H.; Mansour, E.H. The Effect of Cooking, Autoclaving and Germination on the Nutritional Quality of Faba Beans. Food Chem. 1995, 54, 177–182. [Google Scholar] [CrossRef]
- Kilgore, S.M.; Sistrunk, W.A. Effects of Soaking Treatments and Cooking Upon Selected B-Vitamins and the Quality of Blackeyed Peas. J. Food Sci. 1981, 46, 909–911. [Google Scholar] [CrossRef]
- Rolls, B.A.; Porter, J.W.G. Some effects of processing and storage on the nutritive value of milk and milk products. Proc. Nutr Soc. 1973, 32, 9–15. [Google Scholar] [CrossRef]
- King, J.C.; Blumberg, J.; Ingwersen, L.; Jenab, M.; Tucker, K.L. Tree nuts and peanuts as components of a healthy diet. J. Nutr. 2008, 138, 1736S–1740S. [Google Scholar] [CrossRef]
- Arya, S.S.; Salve, A.R.; Chauhan, S. Peanuts as functional food: A review. J. Food Sci. Technol. 2016, 53, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Sathe, S.K.; Monaghan, E.K.; Kshirsagar, H.H.; Venkatachalam, M. Chemical composition of edible nut seeds and its implications in human health. In Tree Nuts: Composition, Phytochemicals, and Health Effects; CRC Press: Boca Raton, FL, USA, 2009; pp. 11–35. [Google Scholar] [CrossRef]
- Dreher, M.L.; Davenport, A.J. Hass avocado composition and potential health effects. Crit. Rev. Food Sci. Nutr. 2013, 53, 738–750. [Google Scholar] [CrossRef]
- Chen, C.Y.; Lapsley, K.; Blumberg, J. A nutrition and health perspective on almonds. J. Sci. Food Agric. 2006, 86, 2245–2250. [Google Scholar] [CrossRef]
- Yada, S.; Lapsley, K.; Huang, G.W. A review of composition studies of cultivated almonds: Macronutrients and micronutrients. J. Food Compos. Anal. 2011, 24, 469–480. [Google Scholar] [CrossRef]
- Barreca, D.; Nabavi, S.M.; Sureda, A.; Rasekhian, M.; Raciti, R.; Silva, A.S.; Annunziata, G.; Arnone, A.; Tenore, G.C.; Suntar, I.; et al. Almonds (Prunus Dulcis Mill. D. A. Webb): A Source of Nutrients and Health-Promoting Compounds. Nutrients 2020, 12, 672. [Google Scholar] [CrossRef]
- Roncero, J.M.; Alvarez-Orti, M.; Pardo-Gimenez, A.; Rabadan, A.; Pardo, J.E. Review about Non-Lipid Components and Minor Fat-Soluble Bioactive Compounds of Almond Kernel. Foods 2020, 9, 1646. [Google Scholar] [CrossRef]
- Ahmad, R.S.; Imran, A.; Hussain, M.B. Nutritional Composition of Meat; IntechOpen Limited: London, UK, 2018; Volume 61. [Google Scholar]
- Li, C. The role of beef in human nutrition and health. In Ensuring Safety and Quality in the Production of Beef; Burleigh Dodds Series in Agricultural Science: Cambridge, UK, 2017; Volume 2, pp. 329–338. [Google Scholar]
- Probst, Y. Nutrient Compostion of Chicken Meat; Rural Industries Research and Development Corporation: Kingston, Australia, 2009. [Google Scholar]
- Van Heerden, S.M.; Schönfeldt, H.C.; Smith, M.F.; Jansen van Rensburg, D.M. Nutrient Content of South African Chickens. J. Food Compos. Anal. 2002, 15, 47–64. [Google Scholar] [CrossRef]
- Dunn, K.R.; Goddard, V.R. Effect of heat upon the nutritive values of peanuts; riboflavin and pantothenic acid content. Food Res. 1948, 13, 512–517. [Google Scholar] [CrossRef]
- Muhamad, N.; Yusoff, M.M.; Gimbun, J. Thermal degradation kinetics of nicotinic acid, pantothenic acid and catechin derived from Averrhoa bilimbi fruits. RSC Adv. 2015, 5, 74132–74137. [Google Scholar] [CrossRef]
- Ford, J.E.; Hurrell, R.F.; Finot, P.A. Storage of milk powders under adverse conditions. 2. Influence on the content of water-soluble vitamins. Br. J. Nutr. 1983, 49, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Gutzeit, D.; Klaubert, B.; Rychlik, M.; Winterhalter, P.; Jerz, G. Effects of processing and of storage on the stability of pantothenic acid in sea buckthorn products (Hippophae rhamnoides L. ssp. rhamnoides) assessed by stable isotope dilution assay. J. Agric. Food Chem. 2007, 55, 3978–3984. [Google Scholar] [CrossRef]
- Pearson, A.; West, R.; Luecke, R. The vitamin and amino acid content of drip obtained upon defrosting frozen pork. Food Res. 1959, 24, 515–519. [Google Scholar] [CrossRef]
- Pearson, A.M.; Burnside, J.E.; Edwards, H.M.; Glasscock, R.S.; Cunha, T.J.; Novak, A.F. Vitamin losses in drip obtained upon defrosting frozen meat. Food Res. 1951, 16, 85–87. [Google Scholar] [CrossRef]
- Ledesma-Amaro, R.; Santos, M.A.; Jiménez, A.; Revuelta, J.L. Microbial production of vitamins. In Microbial Production of Food Ingredients, Enzymes and Nutraceuticals; McNeil, B., Archer, D., Giavasis, I., Harvey, L., Eds.; Woodhead Publishing: Cambridge, UK, 2013; pp. 571–594. [Google Scholar]
- Shimizu, S. Vitamins and Related Compounds: Microbial Production. In Biotechnology; Wiley-VCH: Weinheim, Germany, 2001; pp. 318–340. [Google Scholar]
- Shimizu, S.; Kataoka, M.; Honda, K.; Sakamoto, K. Lactone-ring-cleaving enzymes of microorganisms: Their diversity and applications. J. Biotechnol. 2001, 92, 187–194. [Google Scholar] [CrossRef]
- Li, H.; Lu, X.; Chen, K.; Yang, J.; Zhang, A.; Wang, X.; Ouyang, P. β-alanine production using whole-cell biocatalysts in recombinant Escherichia coli. Mol. Catal. 2018, 449, 93–98. [Google Scholar] [CrossRef]
- Shen, Y.; Zhao, L.; Li, Y.; Zhang, L.; Shi, G. Synthesis of beta-alanine from L-aspartate using L-aspartate-alpha-decarboxylase from Corynebacterium glutamicum. Biotechnol. Lett. 2014, 36, 1681–1686. [Google Scholar] [CrossRef]
- Wang, L.; Mao, Y.; Wang, Z.; Ma, H.; Chen, T. Advances in biotechnological production of beta-alanine. World J. Microbiol. Biotechnol. 2021, 37, 79. [Google Scholar] [CrossRef]
- Laudert, D.; Hohmann, H.P. Application of Enzymes and Microbes for the Industrial Production of Vitamins and Vitamin-like Compounds. In Comprehensive Biotechnology; Moo-Young, M., Ed.; Academic Press: Cambridge, MA, USA, 2011; pp. 583–602. [Google Scholar]
- Bonrath, W.; Netscher, T.; Eggersdorfer, M.; Adam, G. Vitamins, 1. In troduction. In Ullmann’s Encyclopedia of Industrial Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 1–5. [Google Scholar]
- Zu Berstenhorst, S.M.; Hohmann, H.P.; Stahmann, K.P. Vitamins and Vitamin-like Compounds: Microbial Production. In Encyclopedia of Microbiology; Schaechter, M., Ed.; Academic Press: Cambridge, MA, USA, 2009; pp. 549–561. [Google Scholar]
- Wang, Y.; Liu, L.; Jin, Z.; Zhang, D. Microbial Cell Factories for Green Production of Vitamins. Front. Bioeng. Biotechnol. 2021, 9, 661562. [Google Scholar] [CrossRef]
- Dusch, N.; Puhler, A.; Kalinowski, J. Expression of the Corynebacterium glutamicum panD gene encoding L-aspartate-alpha-decarboxylase leads to pantothenate overproduction in Escherichia coli. Appl. Environ. Microbiol. 1999, 65, 1530–1539. [Google Scholar] [CrossRef]
- Huser, A.T.; Chassagnole, C.; Lindley, N.D.; Merkamm, M.; Guyonvarch, A.; Elisakova, V.; Patek, M.; Kalinowski, J.; Brune, I.; Puhler, A.; et al. Rational design of a Corynebacterium glutamicum pantothenate production strain and its characterization by metabolic flux analysis and genome-wide transcriptional profiling. Appl. Environ. Microbiol. 2005, 71, 3255–3268. [Google Scholar] [CrossRef]
- Tigu, F.; Zhang, J.; Liu, G.; Cai, Z.; Li, Y. A highly active pantothenate synthetase from Corynebacterium glutamicum enables the production of D-pantothenic acid with high productivity. Appl. Microbiol. Biotechnol. 2018, 102, 6039–6046. [Google Scholar] [CrossRef]
- Chassagnole, C.; Diano, A.; Letisse, F.; Lindley, N.D. Metabolic network analysis during fed-batch cultivation of Corynebacterium glutamicum for pantothenic acid production: First quantitative data and analysis of by-product formation. J. Biotechnol. 2003, 104, 261–272. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, X.M.; Wang, W.; Liu, Z.Q.; Zheng, Y.G. Metabolic engineering of Escherichia coli for d-pantothenic acid production. Food Chem. 2019, 294, 267–275. [Google Scholar] [CrossRef]
- Zou, S.P.; Wang, Z.J.; Zhao, K.; Zhang, B.; Niu, K.; Liu, Z.Q.; Zheng, Y.G. High-level production of d-pantothenic acid from glucose by fed-batch cultivation of Escherichia coli. Biotechnol. Appl. Biochem. 2020, 68, 1227–1235. [Google Scholar] [CrossRef]
- Zou, S.P.; Zhao, K.; Wang, Z.J.; Zhang, B.; Liu, Z.Q.; Zheng, Y.G. Overproduction of D-pantothenic acid via fermentation conditions optimization and isoleucine feeding from recombinant Escherichia coli W3110. 3 Biotech 2021, 11, 295. [Google Scholar] [CrossRef]
- Woollard, D.C.; Indyk, H.E.; Christiansen, S.K. The analysis of pantothenic acid in milk and infant formulas by HPLC. Food Chem. 2000, 69, 201–208. [Google Scholar] [CrossRef]
- Romera, J.M.; Ramirez, M.; Gil, A. Determination of pantothenic acid in infant milk formulas by high performance liquid chromatography. J. Dairy Sci. 1996, 79, 523–526. [Google Scholar] [CrossRef]
- Mittermayr, R.; Kalman, A.; Trisconi, M.J.; Heudi, O. Determination of vitamin B5 in a range of fortified food products by reversed-phase liquid chromatography-mass spectrometry with electrospray ionisation. J. Chromatogr. A 2004, 1032, 1–6. [Google Scholar] [CrossRef]
- Andrieux, P.; Fontannaz, P.; Kilinc, T.; Gimenez, E.C. Pantothenic acid (vitamin B5) in fortified foods: Comparison of a novel ultra-performance liquid chromatography-tandem mass spectrometry method and a microbiological assay (AOAC Official Method 992.07). J. AOAC Int. 2012, 95, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Ren, Y.; Huang, B.; Liao, W.; Cai, Z.; Tie, X. Simultaneous determination of four water-soluble vitamins in fortified infant foods by ultra-performance liquid chromatography coupled with triple quadrupole mass spectrometry. J. Chromatogr. Sci. 2008, 46, 225–232. [Google Scholar] [CrossRef]
- Chakauya, E.; Coxon, K.M.; Wei, M.; Macdonald, M.V.; Barsby, T.; Abell, C.; Smith, A.G. Towards engineering increased pantothenate (vitamin B(5)) levels in plants. Plant Mol. Biol. 2008, 68, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Rucker, R.B. Pantothenic Acid; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Czumaj, A.; Szrok-Jurga, S.; Hebanowska, A.; Turyn, J.; Swierczynski, J.; Sledzinski, T.; Stelmanska, E. The Pathophysiological Role of CoA. Int. J. Mol. Sci. 2020, 21, 9057. [Google Scholar] [CrossRef] [PubMed]
- Mindrebo, J.T.; Patel, A.; Misson, L.E.; Kim, W.E.; Davis, T.D.; Ni, Q.Z.; La Clair, J.J.; Burkart, M.D. 1.04-Structural Basis of Acyl-Carrier Protein Interactions in Fatty Acid and Polyketide Biosynthesis. In Comprehensive Natural Products III; Hung-Wen, L., Begley, T.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Naquet, P.; Kerr, E.W.; Vickers, S.D.; Leonardi, R. Regulation of coenzyme A levels by degradation: The “Ins and Outs”. Prog. Lipid Res. 2020, 78, 101028. [Google Scholar] [CrossRef]
- Goding, J.W.; Grobben, B.; Slegers, H. Physiological and pathophysiological functions of the ecto-nucleotide pyrophosphatase/phosphodiesterase family. Biochim. Biophys. Acta 2003, 1638, 1–19. [Google Scholar] [CrossRef]
- Shibata, K.; Gross, C.J.; Henderson, L.M. Hydrolysis and absorption of pantothenate and its coenzymes in the rat small intestine. J. Nutr. 1983, 113, 2107–2115. [Google Scholar] [CrossRef] [PubMed]
- Bartucci, R.; Salvati, A.; Olinga, P.; Boersma, Y.L. Vanin 1: Its Physiological Function and Role in Diseases. Int. J. Mol. Sci. 2019, 20, 3891. [Google Scholar] [CrossRef]
- Turner, J.B.; Hughes, D.E. The absorption of some B-group vitamins by surviving rat intestine preparations. Q. J. Exp. Physiol. Cogn. Med. Sci. 1962, 47, 107–133. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.D.; Wang, H.; Huang, W.; Fei, Y.J.; Leibach, F.H.; Devoe, L.D.; Ganapathy, V. Molecular and functional characterization of the intestinal Na+-dependent multivitamin transporter. Arch. Biochem. Biophys. 1999, 366, 95–106. [Google Scholar] [CrossRef]
- Ono, S.; Kameda, K.; Abiko, Y. Metabolism of panthethine in the rat. J. Nutr. Sci. Vitam. 1974, 20, 203–213. [Google Scholar] [CrossRef]
- Wittwer, C.T.; Gahl, W.A.; Butler, J.D.; Zatz, M.; Thoene, J.G. Metabolism of pantethine in cystinosis. J. Clin. Investig. 1985, 76, 1665–1672. [Google Scholar] [CrossRef]
- Karnitz, L.M.; Gross, C.J.; Henderson, L.M. Transport and metabolism of pantothenic acid by rat kidney. Biochim. Biophys. Acta 1984, 769, 486–492. [Google Scholar] [CrossRef]
- Eissenstat, B.R.; Wyse, B.W.; Hansen, R.G. Pantothenic acid status of adolescents. Am. J. Clin. Nutr. 1986, 44, 931–937. [Google Scholar] [CrossRef]
- Annous, K.F.; Song, W.O. Pantothenic acid uptake and metabolism by red blood cells of rats. J. Nutr. 1995, 125, 2586–2593. [Google Scholar] [CrossRef]
- Vadlapudi, A.D.; Vadlapatla, R.K.; Mitra, A.K. Sodium dependent multivitamin transporter (SMVT): A potential target for drug delivery. Curr. Drug Targets 2012, 13, 994–1003. [Google Scholar] [CrossRef]
- Grassl, S.M. Human placental brush-border membrane Na(+)-pantothenate cotransport. J. Biol. Chem. 1992, 267, 22902–22906. [Google Scholar] [CrossRef]
- Uchida, Y.; Ito, K.; Ohtsuki, S.; Kubo, Y.; Suzuki, T.; Terasaki, T. Major involvement of Na(+) -dependent multivitamin transporter (SLC5A6/SMVT) in uptake of biotin and pantothenic acid by human brain capillary endothelial cells. J. Neurochem. 2015, 134, 97–112. [Google Scholar] [CrossRef]
- Boger, W.P.; Bayne, G.M.; Gylfe, J.; Wright, L.D. Renal clearance of pantothenic acid in man; inhibition by probenecid (benemid). Proc. Soc. Exp. Biol. Med. 1953, 82, 604–608. [Google Scholar] [CrossRef]
- Spector, H.; Hamilton, T.S.; Mitchell, H.H. The effect of pantothenic acid dosage and environmental temperature and humidity upon the dermal and renal excretion of pantothenic acid. J. Biol. Chem. 1945, 161, 145–152. [Google Scholar] [CrossRef]
- Tsuji, T.; Fukuwatari, T.; Sasaki, S.; Shibata, K. Urinary excretion of vitamin B1, B2, B6, niacin, pantothenic acid, folate, and vitamin C correlates with dietary intakes of free-living elderly, female Japanese. Nutr. Res. 2010, 30, 171–178. [Google Scholar] [CrossRef]
- Tsuji, T.; Fukuwatari, T.; Sasaki, S.; Shibata, K. Twenty-four-hour urinary water-soluble vitamin levels correlate with their intakes in free-living Japanese university students. Eur. J. Clin. Nutr. 2010, 64, 800–807. [Google Scholar] [CrossRef]
- Hodges, R.E.; Ohlson, M.A.; Bean, W.B. Pantothenic acid deficiency in man. J. Clin. Investig. 1958, 37, 1642–1657. [Google Scholar] [CrossRef]
- Hodges, R.E.; Bean, W.B.; Ohlson, M.A.; Bleiler, R. Human pantothenic acid deficiency produced by omega-methyl pantothenic acid. J. Clin. Investig. 1959, 38, 1421–1425. [Google Scholar] [CrossRef]
- Drell, W.; Dunn, M.S. Production of pantothenic acid deficiency syndrome in mice with-methylpantothenic acid. Arch. Biochem. Biophys. 1951, 33, 110–119. [Google Scholar] [CrossRef]
- Pudelkewicz, C.; Roderuck, C. Pantothenic acid deficiency in the young guinea pig. J. Nutr. 1960, 70, 348–352. [Google Scholar] [CrossRef]
- Bean, W.B.; Hodges, R.E.; Daum, K. Pantothenic acid deficiency induced in human subjects. J. Clin. Investig. 1955, 34, 1073–1084. [Google Scholar] [CrossRef]
- Nelson, M.M.; Evans, H.M. Pantothenic acid deficiency and reproduction in the rat. J. Nutr. 1946, 31, 497–507. [Google Scholar] [CrossRef]
- Chen, M.-C.; Song, Y.; Song, W.O. Fetal growth retardation and death in pantothenic acid-deficient rats is due to imparired placental function. J. Nutr. Biochem. 1996, 7, 451–456. [Google Scholar] [CrossRef]
- Olson, R.E.; Kaplan, N.O. The effect of pantothenic acid deficiency upon the coenzyme A content and pyruvate utilization of rat and duck tissues. J. Biol. Chem. 1948, 175, 515–529. [Google Scholar] [CrossRef]
- Schaefer, A.E.; McKibbin, J.M.; Elvehjem, C.A. Pantothenic acid deficiency studies in the dog. J. Biol. Chem. 1942, 143, 321–330. [Google Scholar] [CrossRef]
- Guehring, R.R.; Hurley, L.S.; Morgan, A.F. Cholesterol metabolism in pantothenic acid deficiency. J. Biol. Chem. 1952, 197, 485–493. [Google Scholar] [CrossRef]
- Fry, P.C.; Fox, H.M.; Tao, H.G. Metabolic response to a pantothenic acid deficient diet in humans. J. Nutr. Sci. Vitam. 1976, 22, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Patassini, S.; Begley, P.; Church, S.; Waldvogel, H.J.; Faull, R.L.M.; Unwin, R.D.; Cooper, G.J.S. Cerebral deficiency of vitamin B5 (d-pantothenic acid; pantothenate) as a potentially-reversible cause of neurodegeneration and dementia in sporadic Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2020, 527, 676–681. [Google Scholar] [CrossRef]
- Johnson, M.A.; Kuo, Y.M.; Westaway, S.K.; Parker, S.M.; Ching, K.H.; Gitschier, J.; Hayflick, S.J. Mitochondrial localization of human PANK2 and hypotheses of secondary iron accumulation in pantothenate kinase-associated neurodegeneration. Ann. N. Y. Acad. Sci. 2004, 1012, 282–298. [Google Scholar] [CrossRef]
- Kurian, M.A.; McNeill, A.; Lin, J.P.; Maher, E.R. Childhood disorders of neurodegeneration with brain iron accumulation (NBIA). Dev. Med. Child Neurol. 2011, 53, 394–404. [Google Scholar] [CrossRef]
- Pratini, N.R.; Sweeters, N.; Vichinsky, E.; Neufeld, J.A. Treatment of classic pantothenate kinase-associated neurodegeneration with deferiprone and intrathecal baclofen. Am. J. Phys. Med. Rehabil. 2013, 92, 728–733. [Google Scholar] [CrossRef]
- Gregory, A.; Hayflick, S.J. Pantothenate Kinase-Associated Neurodegeneration. In GeneReviews®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Mirzaa, G., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Hatano, M. Pantothenic acid deficiency in rats. J. Vitam. 1962, 8, 143–159. [Google Scholar] [CrossRef]
- Seronde, J., Jr. The Pathogenesis of Duodenal Ulcer Disease in the Pantothenate-Deficient Rat. Yale J. Biol. Med. 1963, 36, 141–156. [Google Scholar] [PubMed]
- Jones, J.H.; Foster, C.; Dorfman, F.; Hunter, G.L.; Quinby, M.E.; Alexander, D.L. Effects on the Albino Mouse of Feeding Diets Very Deficient in Each of Several Vitamin B Factors. J. Nutr. 1945, 29, 127–136. [Google Scholar] [CrossRef]
- Berg, B.N. Duodenitis and duodenal ulcers produced in rats by pantothenic acid deficiency. Br. J. Exp. Pathol. 1959, 40, 371–374. [Google Scholar] [PubMed]
- Osborn, M.O.; Weaver, C.; Anderson, J. Cholesterol in blood and tissues of adult pantothenic acid-deficient rats. J. Nutr. 1958, 64, 313–319. [Google Scholar] [CrossRef]
- Groody, T.C.; Groody, M.E. Feather Depigmentation and Pantothenic Acid Deficiency in Chicks. Science 1942, 95, 655–656. [Google Scholar] [CrossRef] [PubMed]
- Gries, C.L.; Scott, M.L. The pathology of thiamin, riboflavin, pantothenic acid and niacin deficiencies in the chick. J. Nutr. 1972, 102, 1269–1285. [Google Scholar] [CrossRef]
- Jukes, T.H. Pantothenic acid and the filtrate (chick anti-dermatitis) factor. J. Am. Chem. Soc. 1939, 61, 975–976. [Google Scholar] [CrossRef]
- Wintrobe, M.M.; Follis, R.H., Jr.; Alcayaga, R.; Paulson, M.; Humphreys, S. Pantothenic acid deficiency in swine with particular reference to the effects on growth and on the alimentary tract. Bul. Johns Hopkins Hosp. 1943, 73, 313–341. [Google Scholar]
- Follis, R.H.; Wintrobe, M.M. A Comparison of the Effects of Pyridoxine and Pantothenic Acid Deficiencies on the Nervous Tissues of Swine. J. Exp. Med. 1945, 81, 539–552. [Google Scholar] [CrossRef]
- Nelson, R.A. Intestinal transport, coenzyme A, and colitis in pantothenic acid deficiency. Am. J. Clin. Nutr. 1968, 21, 495–501. [Google Scholar] [CrossRef]
- Silber, R.H. Studies of Pantothenic Acid Deficiency in Dogs: Three Figures. J. Nutr. 1944, 27, 425–433. [Google Scholar] [CrossRef]
- Wittwer, C.T.; Graves, C.P.; Peterson, M.A.; Jorgensen, E.; Wilson, D.E.; Thoene, J.G.; Wyse, B.W.; Windham, C.T.; Hansen, R.G. Pantethine lipomodulation: Evidence for cysteamine mediation in vitro and in vivo. Atherosclerosis 1987, 68, 41–49. [Google Scholar] [CrossRef]
- Evans, M.; Rumberger, J.A.; Azumano, I.; Napolitano, J.J.; Citrolo, D.; Kamiya, T. Pantethine, a derivative of vitamin B5, favorably alters total, LDL and non-HDL cholesterol in low to moderate cardiovascular risk subjects eligible for statin therapy: A triple-blinded placebo and diet-controlled investigation. Vasc. Health Risk Manag. 2014, 10, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Gaddi, A.; Descovich, G.C.; Noseda, G.; Fragiacomo, C.; Colombo, L.; Craveri, A.; Montanari, G.; Sirtori, C.R. Controlled evaluation of pantethine, a natural hypolipidemic compound, in patients with different forms of hyperlipoproteinemia. Atherosclerosis 1984, 50, 73–83. [Google Scholar] [CrossRef]
- Arsenio, L.; Caronna, S.; Lateana, M.; Magnati, G.; Strata, A.; Zammarchi, G. Hyperlipidemia, diabetes and atherosclerosis: Efficacy of treatment with pantethine. Acta Biomed. Ateneo Parm. 1984, 55, 25–42. [Google Scholar]
- Donati, C.; Bertieri, R.S.; Barbi, G. Pantethine, diabetes mellitus and atherosclerosis. Clinical study of 1045 patients. Clin. Ter. 1989, 128, 411–422. [Google Scholar]
- Coronel, F.; Tornero, F.; Torrente, J.; Naranjo, P.; De Oleo, P.; Macia, M.; Barrientos, A. Treatment of hyperlipemia in diabetic patients on dialysis with a physiological substance. Am. J. Nephrol. 1991, 11, 32–36. [Google Scholar] [CrossRef]
- Orloff, S.; Butler, J.D.; Towne, D.; Mukherjee, A.B.; Schulman, J.D. Pantetheinase activity and cysteamine content in cystinotic and normal fibroblasts and leukocytes. Pediatr. Res. 1981, 15, 1063–1067. [Google Scholar] [CrossRef]
- Capodice, J.L. Feasibility, Tolerability, Safety and Efficacy of a Pantothenic Acid Based Dietary Supplement in Subjects with Mild to Moderate Facial Acne Blemishes. J. Cosmet. Dermatol. Sci. Appl. 2012, 2, 132–135. [Google Scholar] [CrossRef]
- Yang, M.; Moclair, B.; Hatcher, V.; Kaminetsky, J.; Mekas, M.; Chapas, A.; Capodice, J. A randomized, double-blind, placebo-controlled study of a novel pantothenic Acid-based dietary supplement in subjects with mild to moderate facial acne. Dermatol. Ther. 2014, 4, 93–101. [Google Scholar] [CrossRef]
- Proksch, E.; de Bony, R.; Trapp, S.; Boudon, S. Topical use of dexpanthenol: A 70th anniversary article. J. Dermatol. Treat. 2017, 28, 766–773. [Google Scholar] [CrossRef]
- Wollina, U.; Kubicki, J. Dexpanthenol supports healing of superficial wounds and injuries. Kosm. Med. 2006, 27, 240–249. [Google Scholar]
- Proksch, E.; Nissen, H.P. Dexpanthenol enhances skin barrier repair and reduces inflammation after sodium lauryl sulphate-induced irritation. J. Dermatol. Treat. 2002, 13, 173–178. [Google Scholar] [CrossRef]
- Stettler, H.; Kurka, P.; Lunau, N.; Manger, C.; Bohling, A.; Bielfeldt, S.; Wilhelm, K.P.; Dahnhardt-Pfeiffer, S.; Dahnhardt, D.; Brill, F.H.; et al. A new topical panthenol-containing emollient: Results from two randomized controlled studies assessing its skin moisturization and barrier restoration potential, and the effect on skin microflora. J. Dermatol. Treat. 2017, 28, 173–180. [Google Scholar] [CrossRef]
- Heise, R.; Schmitt, L.; Huth, L.; Krings, L.; Kluwig, D.; Katsoulari, K.V.; Steiner, T.; Holzle, F.; Baron, J.M.; Huth, S. Accelerated wound healing with a dexpanthenol-containing ointment after fractional ablative CO2 laser resurfacing of photo-damaged skin in a randomized prospective clinical trial. Cutan. Ocul. Toxicol. 2019, 38, 274–278. [Google Scholar] [CrossRef]
- Wananukul, S.; Limpongsanuruk, W.; Singalavanija, S.; Wisuthsarewong, W. Comparison of dexpanthenol and zinc oxide ointment with ointment base in the treatment of irritant diaper dermatitis from diarrhea: A multicenter study. J. Med. Assoc. Thai 2006, 89, 1654–1658. [Google Scholar]
- Olsavszky, R.; Nanu, E.A.; Macura-Biegun, A.; Kurka, P.; Trapp, S. Skin barrier restoration upon topical use of two 5% dexpanthenol water-in-oil formulations on freshly tattooed skin: Results from a single-blind prospective study. Wounds Int. 2019, 10, 33–39. [Google Scholar]
- Udompataikul, M.; Limpa-o-vart, D. Comparative trial of 5% dexpanthenol in water-in-oil formulation with 1% hydrocortisone ointment in the treatment of childhood atopic dermatitis: A pilot study. J. Drugs Dermatol. 2012, 11, 366–374. [Google Scholar]
- Shanazi, M.; Farshbaf Khalili, A.; Kamalifard, M.; Asghari Jafarabadi, M.; Masoudin, K.; Esmaeli, F. Comparison of the Effects of Lanolin, Peppermint, and Dexpanthenol Creams on Treatment of Traumatic Nipples in Breastfeeding Mothers. J. Caring Sci. 2015, 4, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Hamdi, I.M. Effect of D-Panthenol on Corneal Epithelial Healing after Surface Laser Ablation. J. Ophthalmol. 2018, 2018, 6537413. [Google Scholar] [CrossRef]
- Gobbels, M.; Gross, D. Clinical study of the effectiveness of a dexpanthenol containing artificial tears solution (Siccaprotect) in treatment of dry eyes. Klin. Monbl. Augenheilkd. 1996, 209, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Jagade, M.V.; Langade, D.G.; Pophale, R.R.; Prabhu, A. Oxymetazoline plus dexpanthenol in nasal congestion. Indian J. Otolaryngol. Head Neck Surg. 2008, 60, 393–397. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kehrl, W.; Sonnemann, U.; Dethlefsen, U. Advance in therapy of acute rhinitis—Comparison of efficacy and safety of xylometazoline in combination xylometazoline-dexpanthenol in patients with acute rhinitis. Laryngo-Rhino-Otologie 2003, 82, 266–271. [Google Scholar] [CrossRef]
- Kehrl, W.; Sonnemann, U. Improving wound healing after nose surgery by combined administration of xylometazoline and dexpanthenol. Laryngo-Rhino-Otologie 2000, 79, 151–154. [Google Scholar] [CrossRef]
- Unna, K.; Greslin, J.G. Studies on the toxicity and pharmacology of pantothenic acid. J. Pharmacol. Exp. Ther. 1941, 76, 85–90. [Google Scholar]
- Flodin, N.W. Pharmacology of Micronutrients; A.R. Liss: New York, NY, USA, 1988; 340p. [Google Scholar]
- Debourdeau, P.M.; Djezzar, S.; Estival, J.L.; Zammit, C.M.; Richard, R.C.; Castot, A.C. Life-threatening eosinophilic pleuropericardial effusion related to vitamins B5 and H. Ann. Pharmacother. 2001, 35, 424–426. [Google Scholar] [CrossRef]
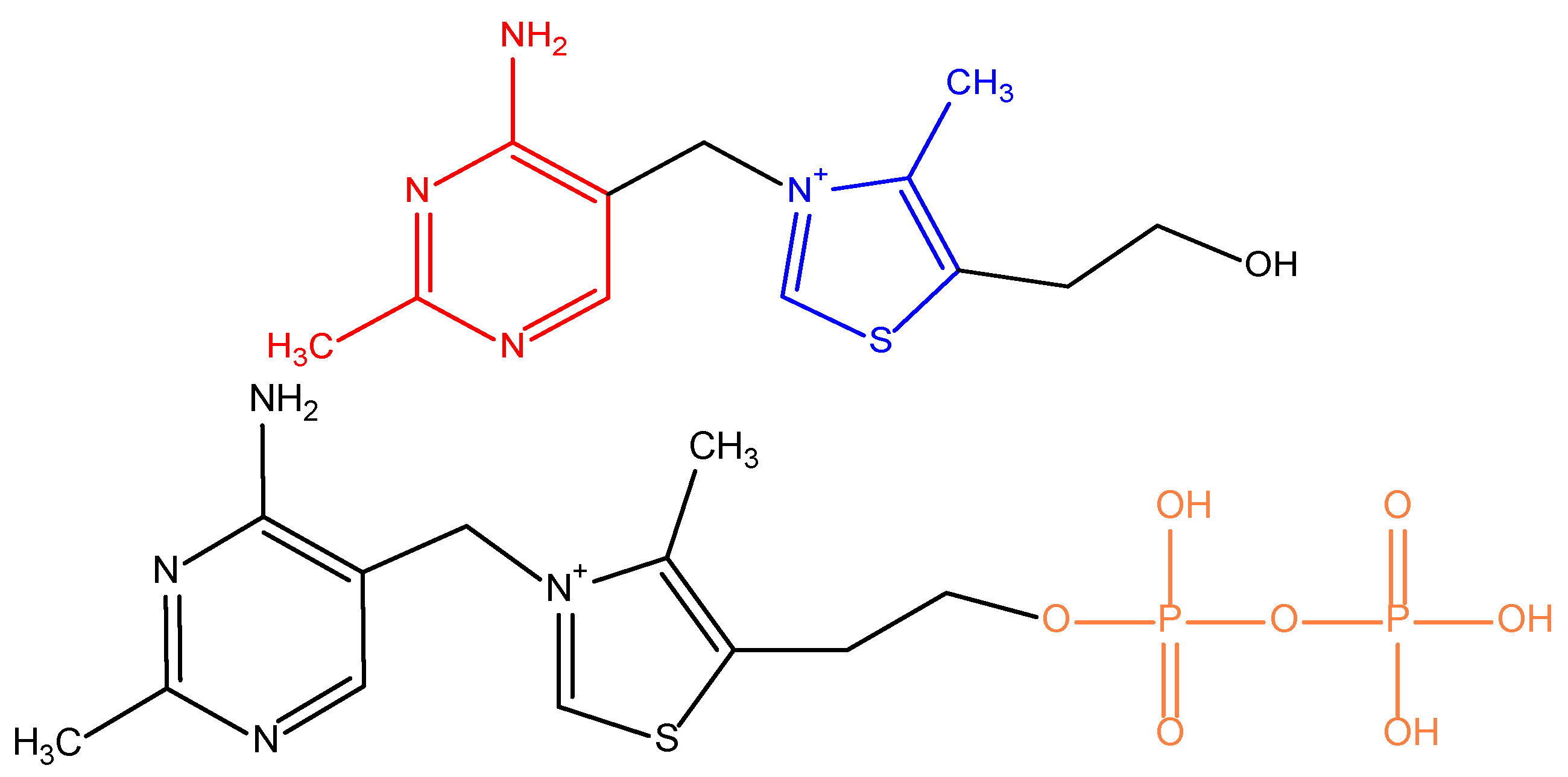
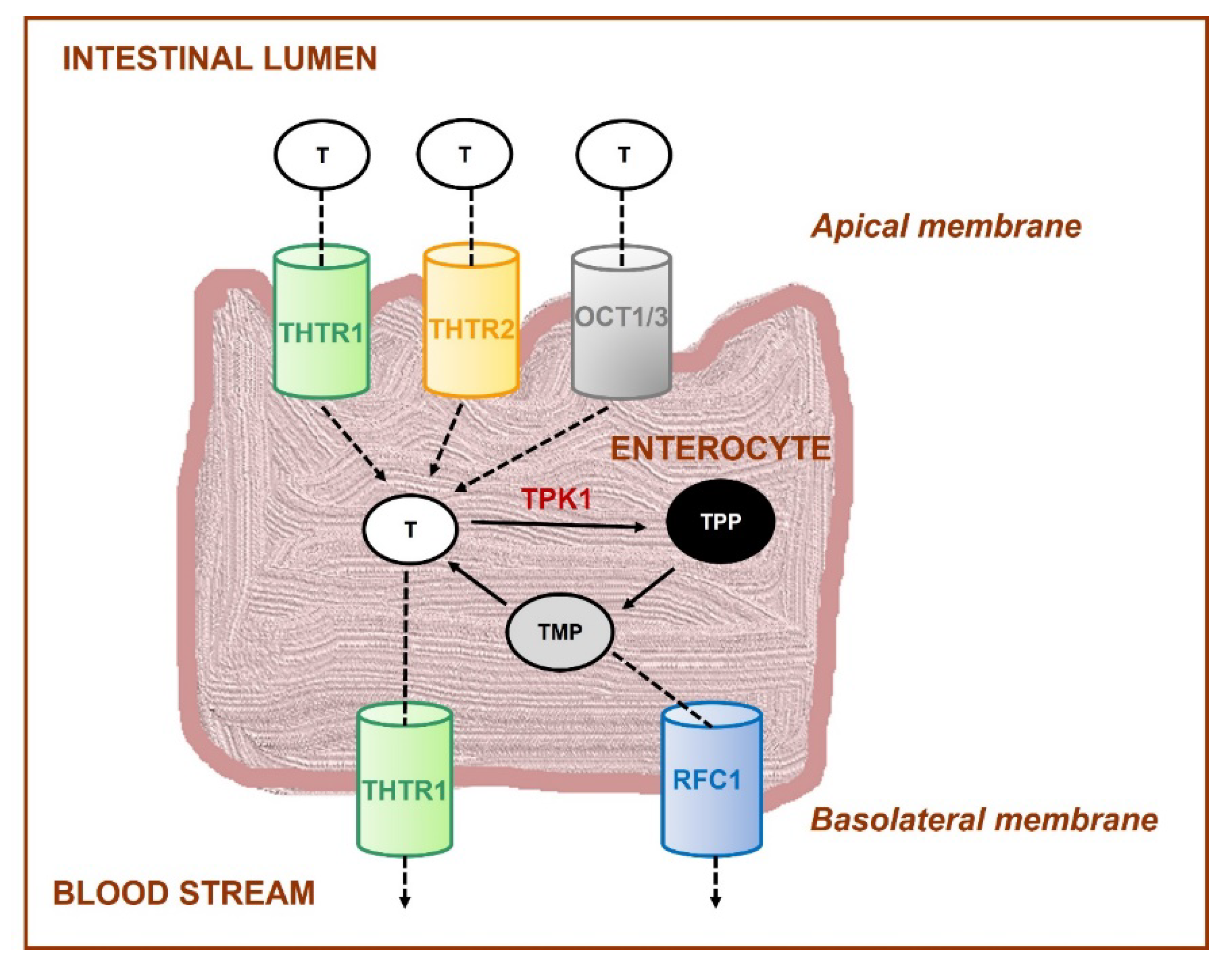

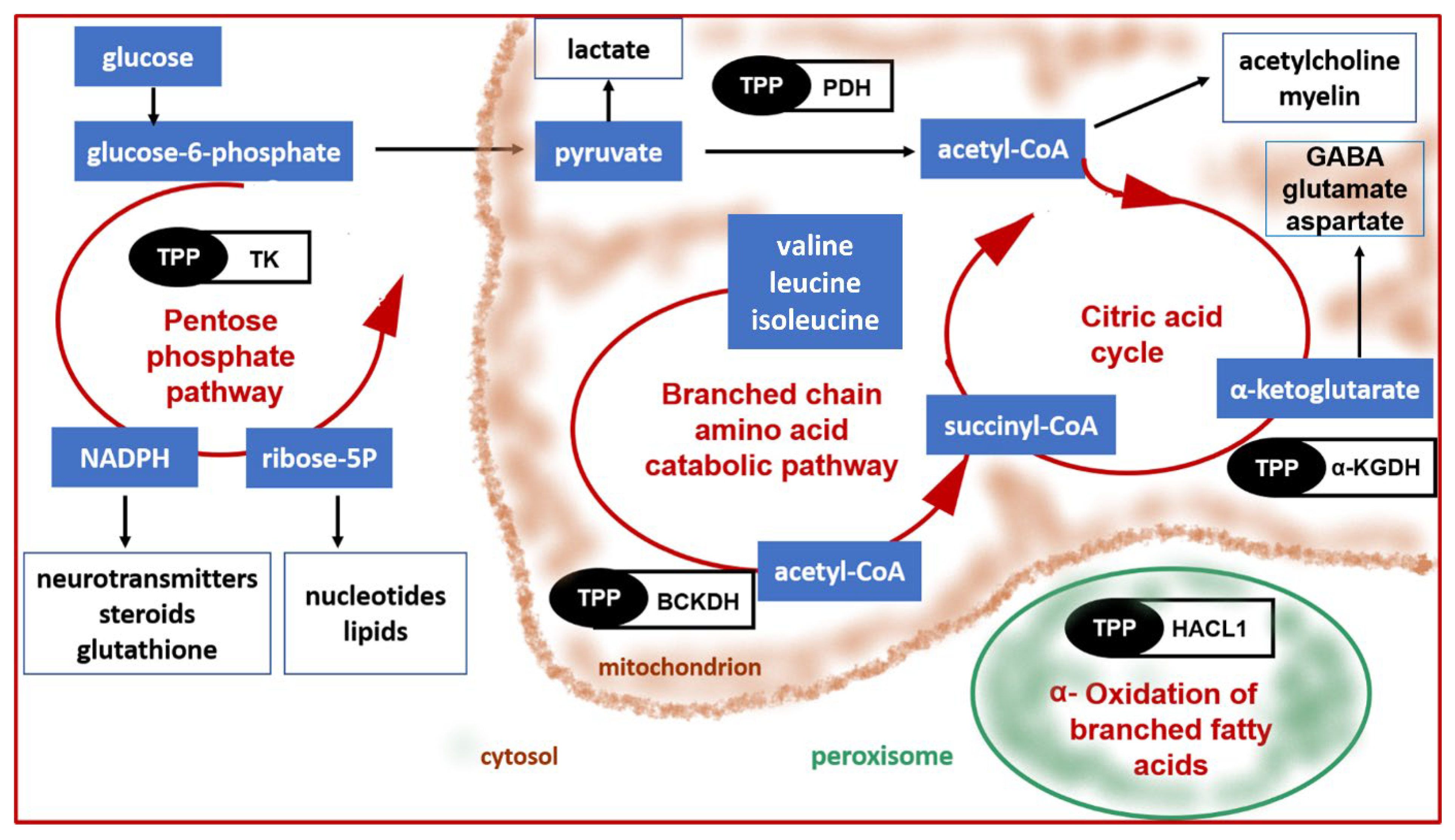
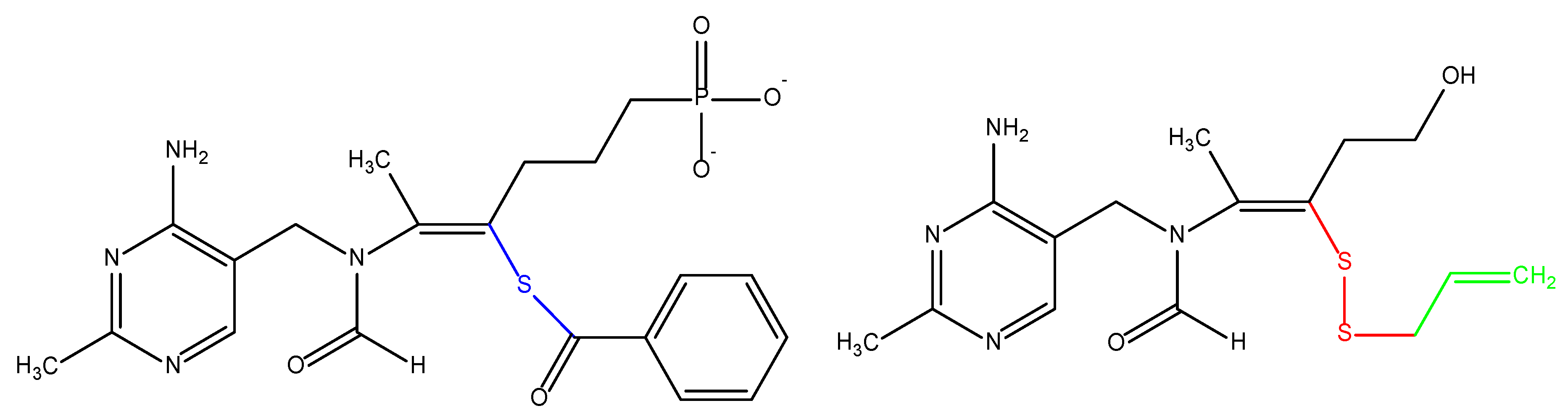
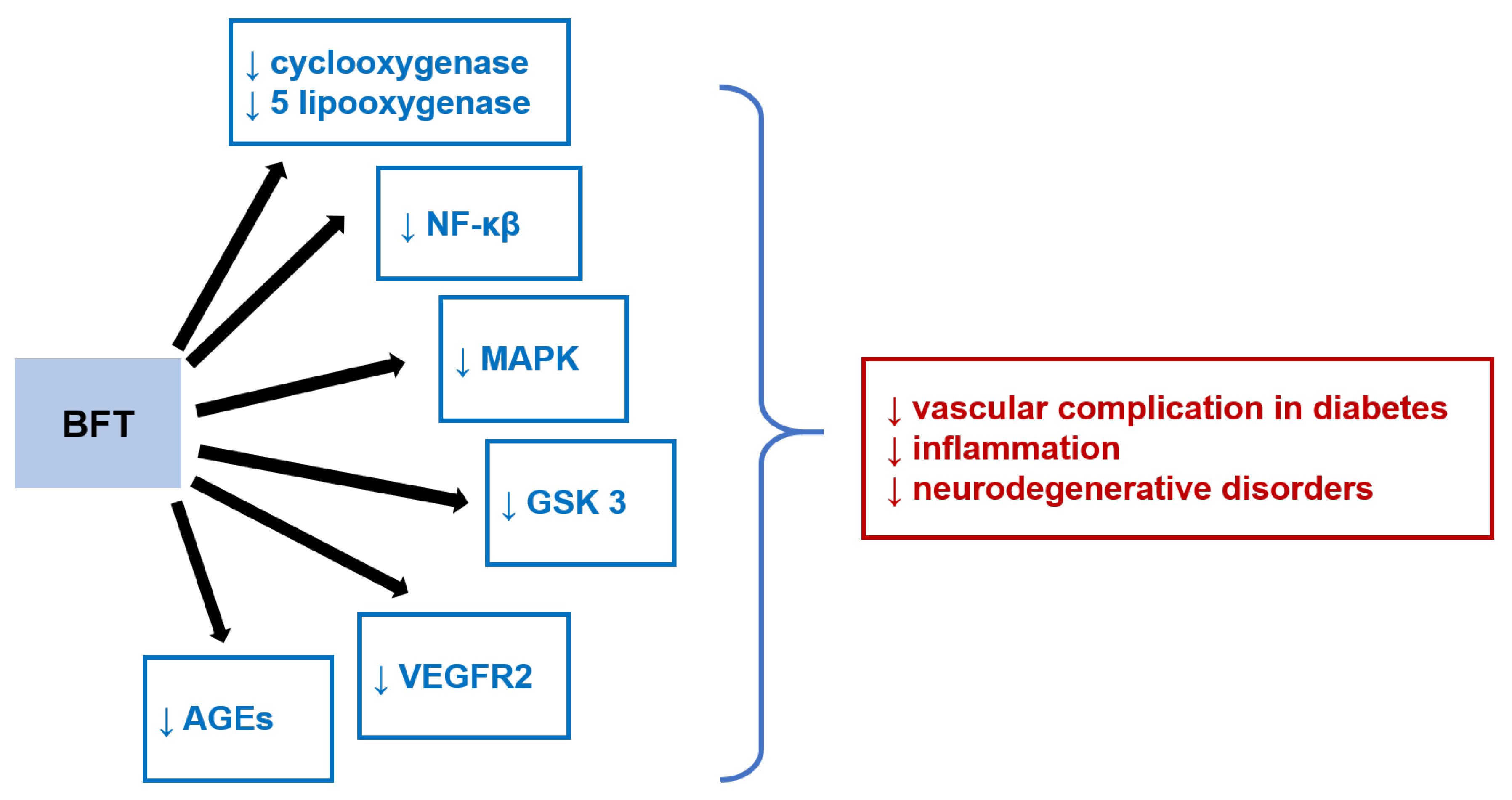
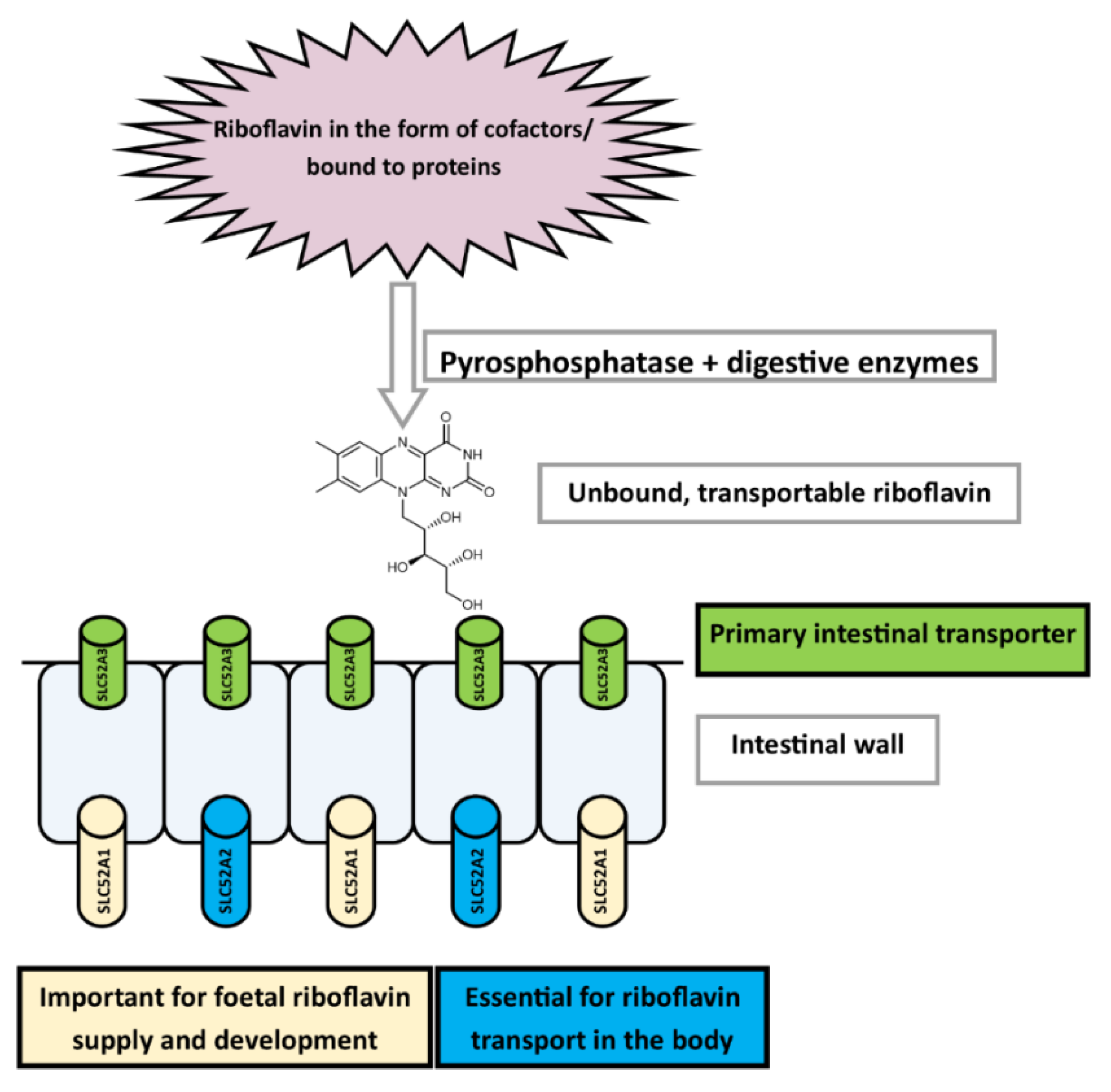
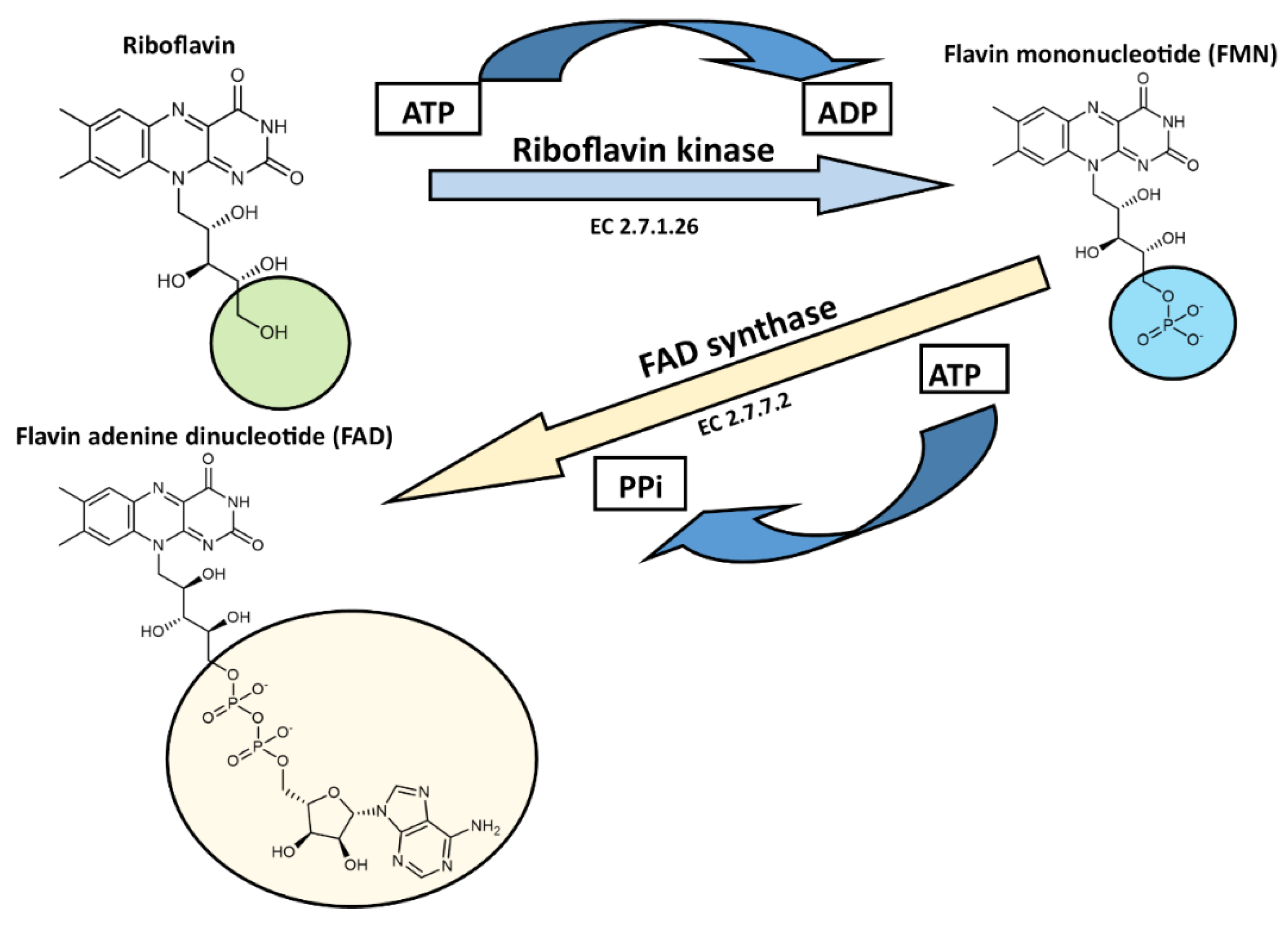
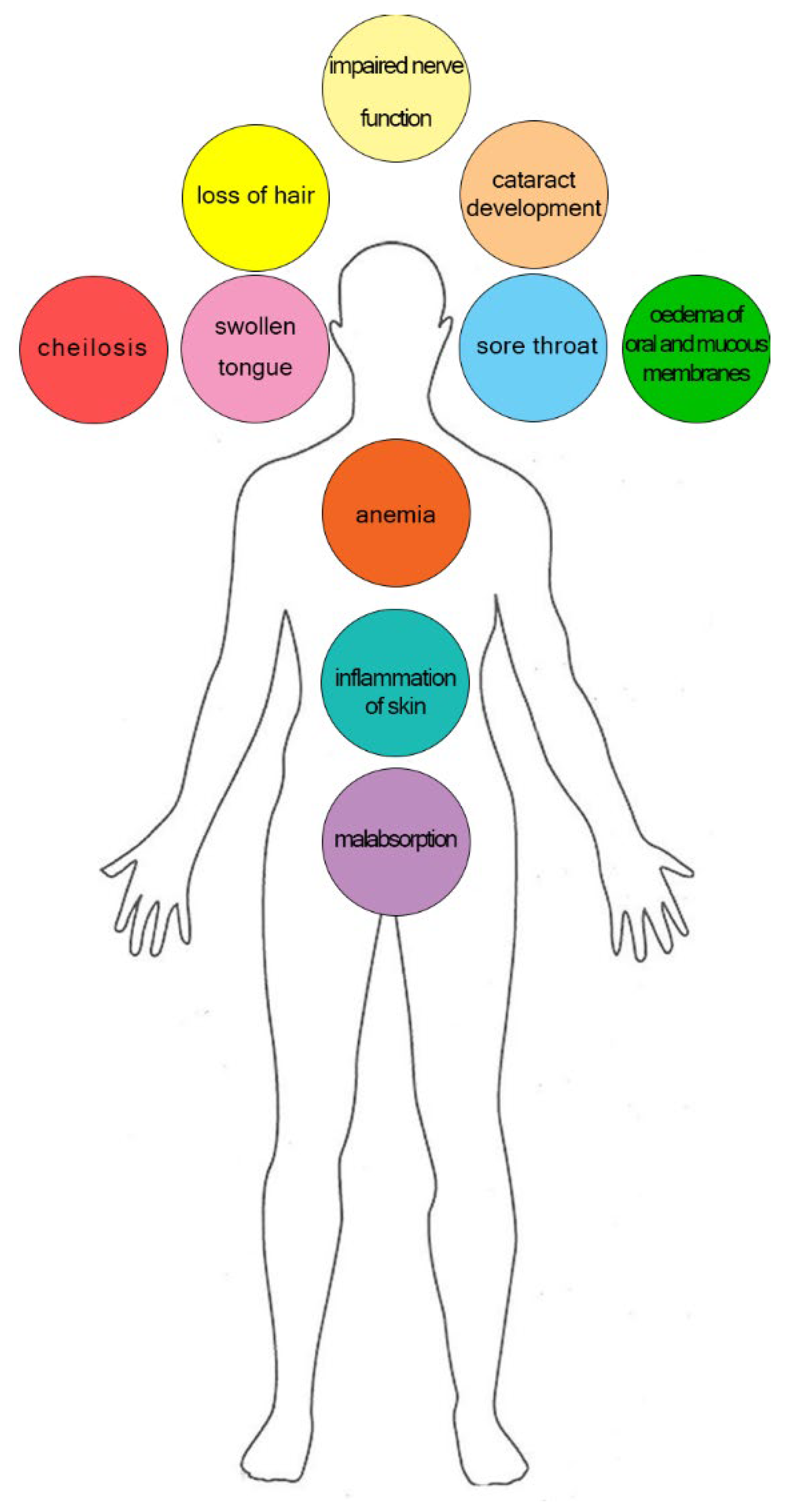
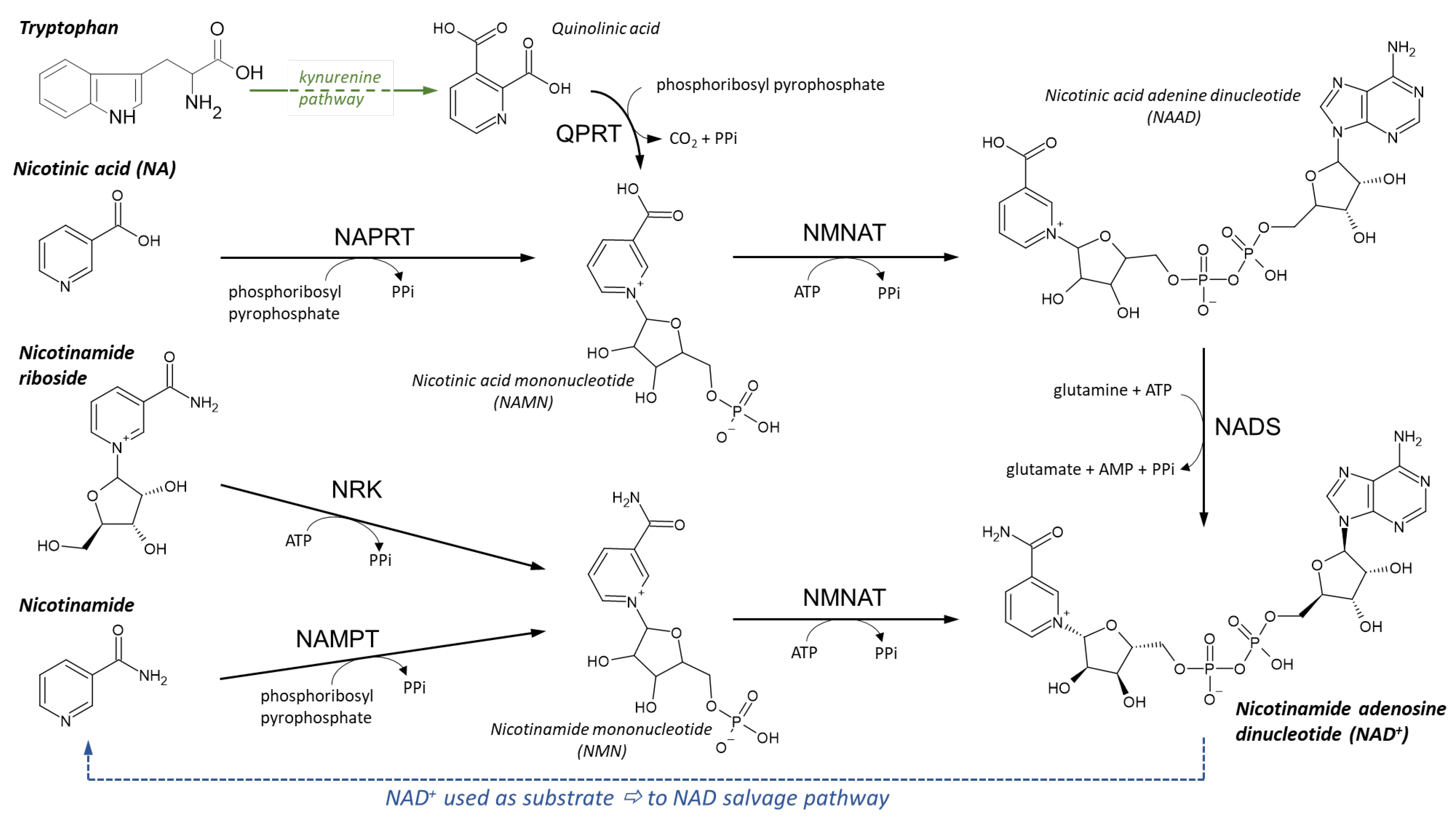
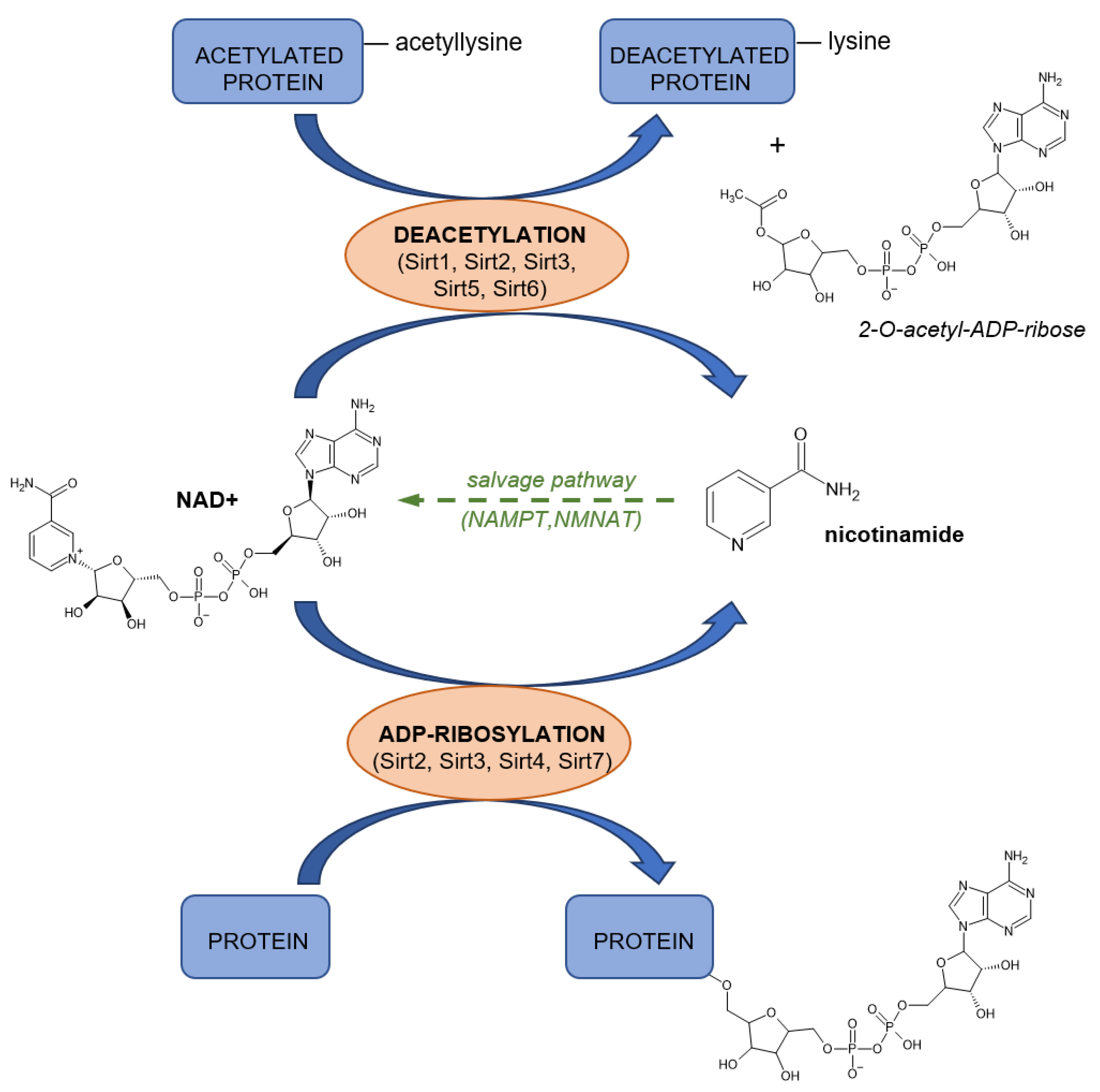
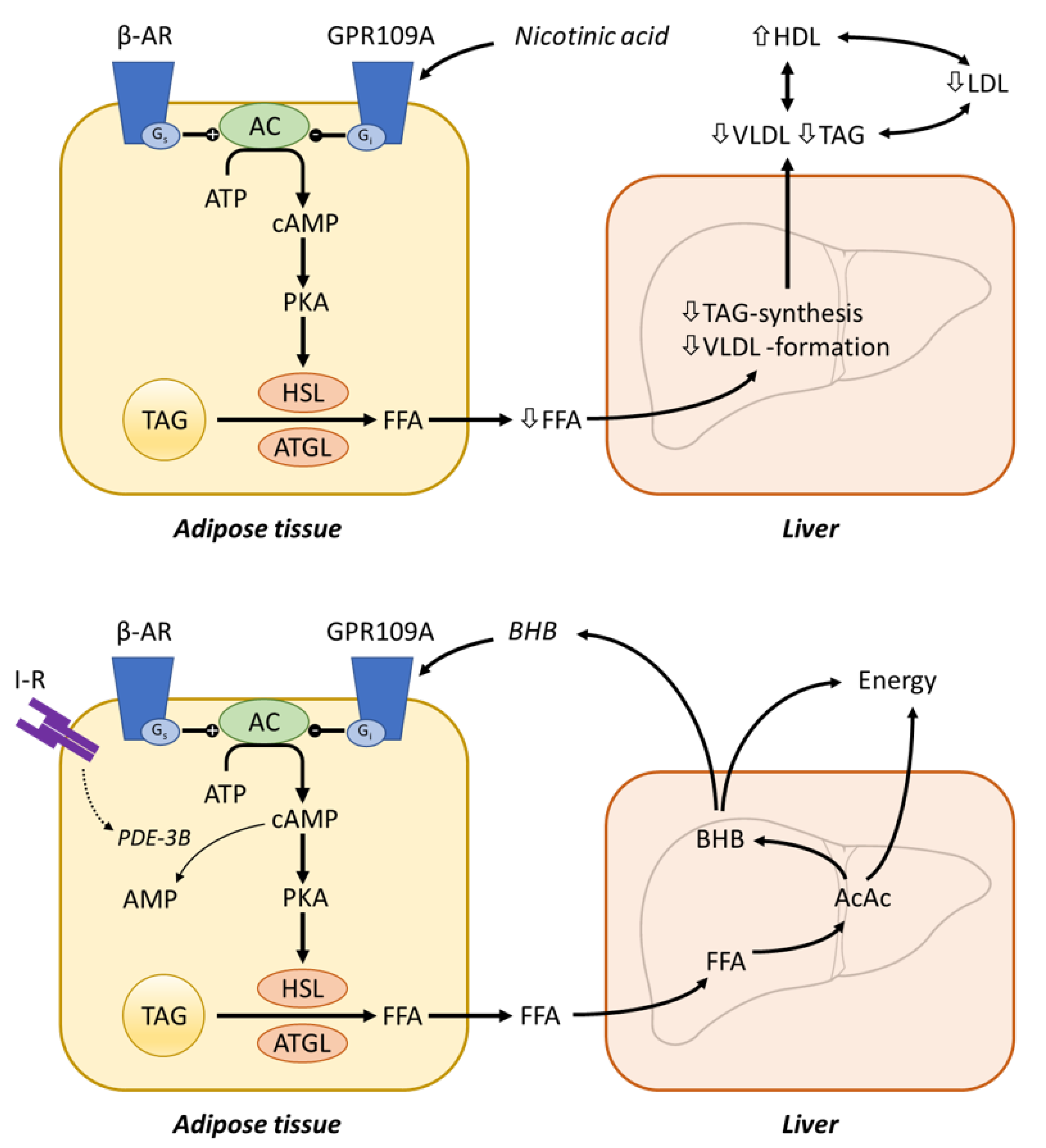
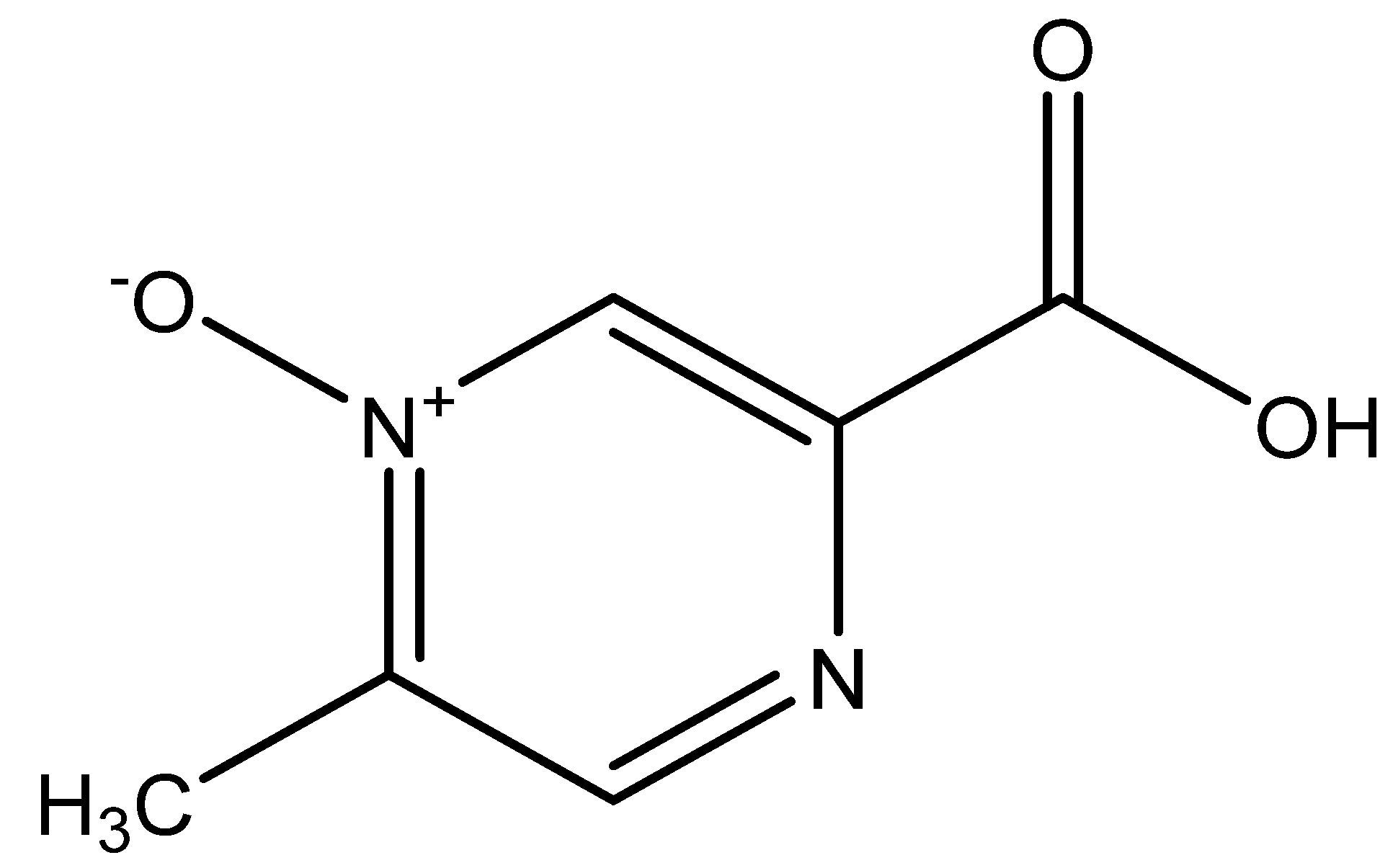
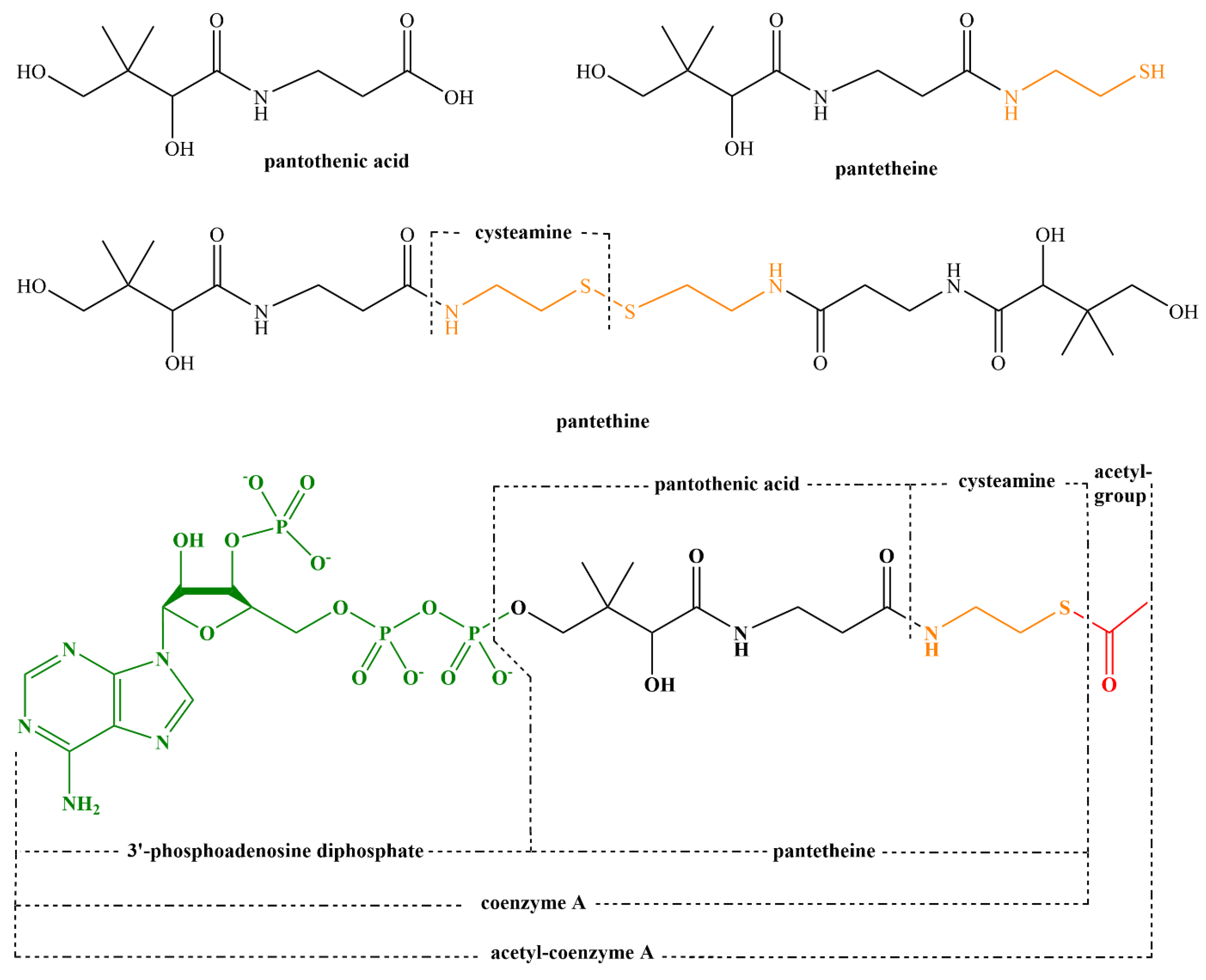
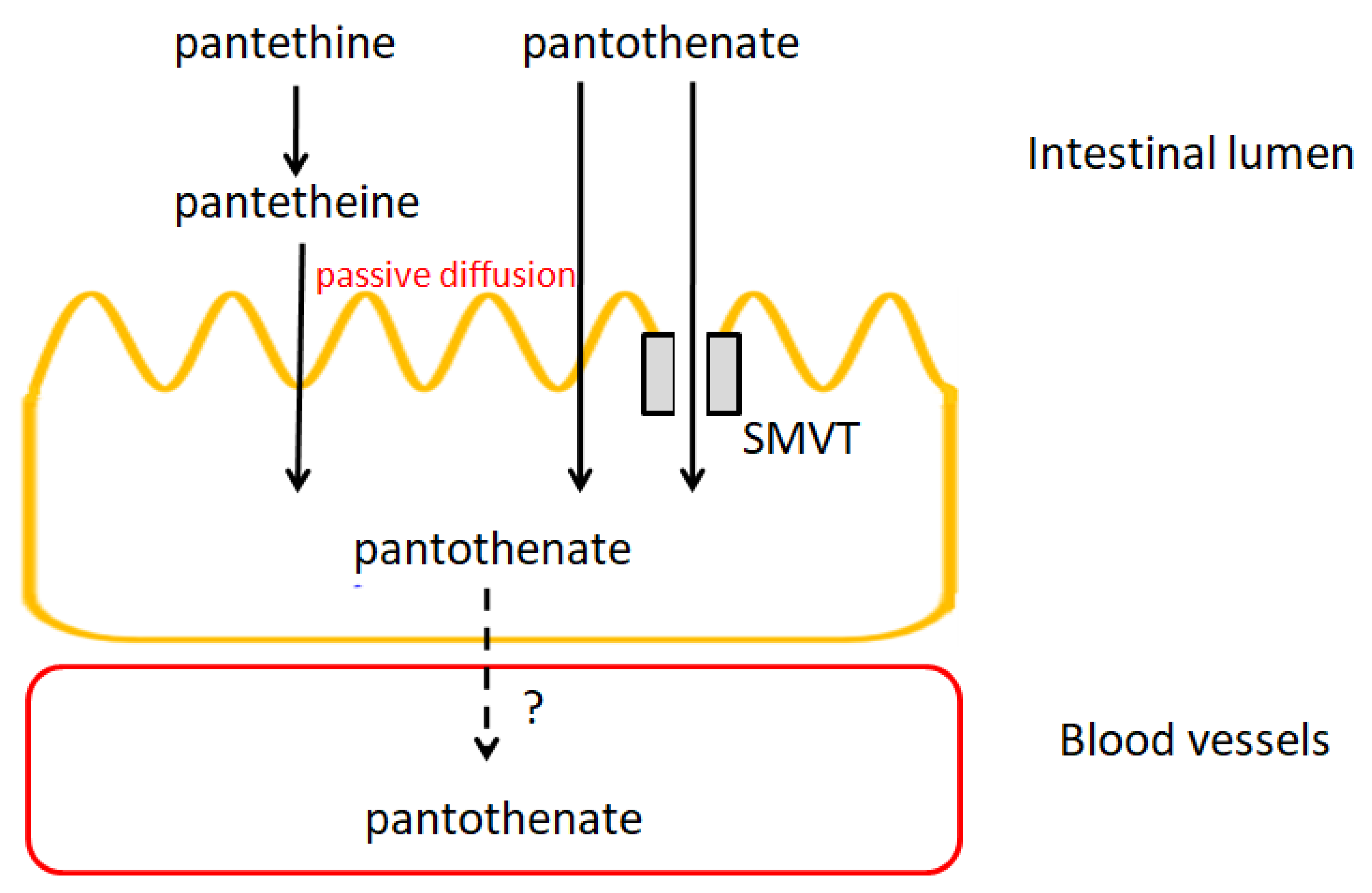
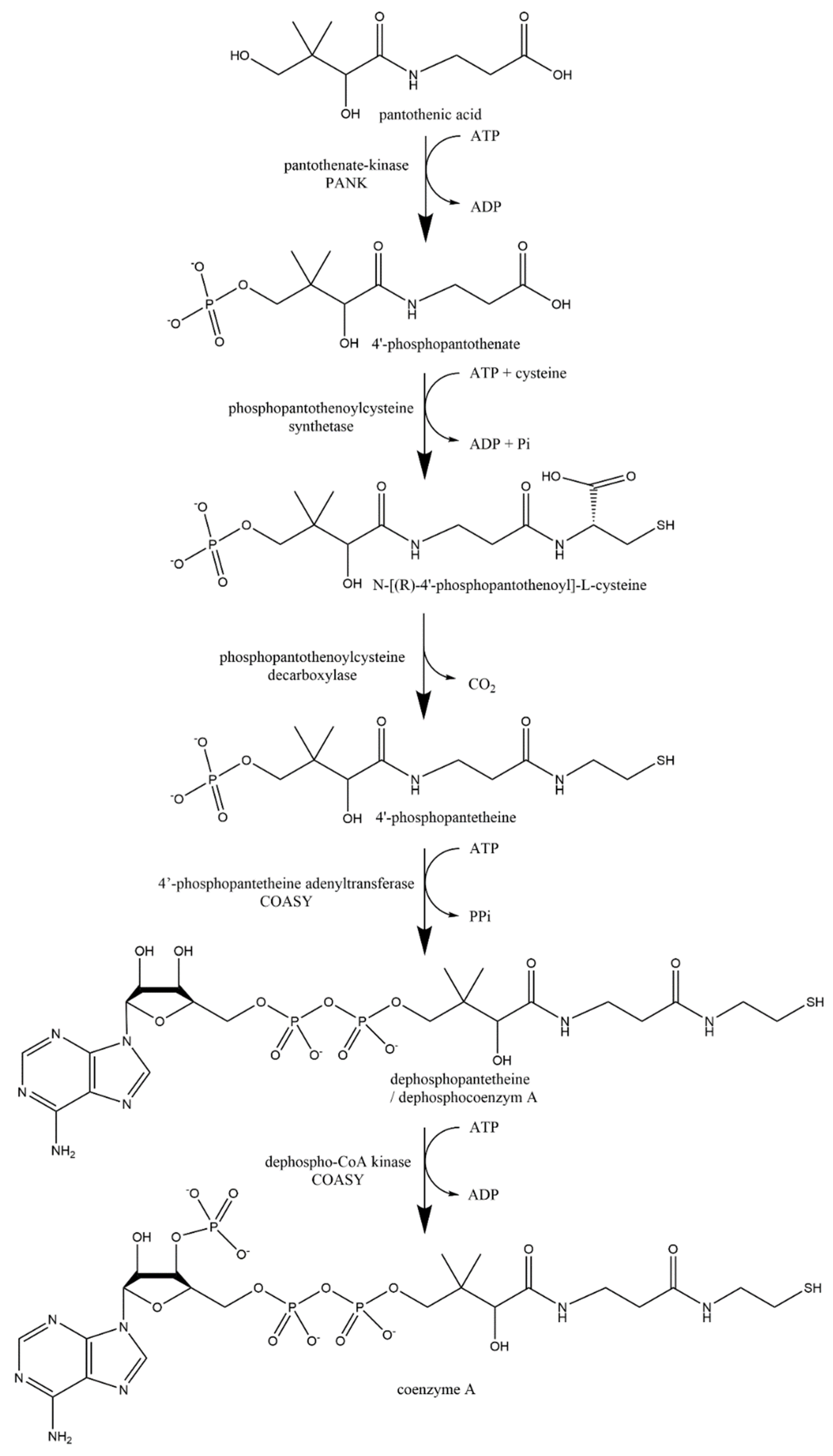
| Food | Thiamine Content (μg/100 g) | References |
|---|---|---|
| Oats | 520–763 | [11,90] |
| Wheat | 276–525 | [58,90,91] |
| Rice, brown | 300–413 | [11,22,59,92] |
| Rice, white | 50–80 | [11,22,59,92] |
| Maize | 246–385 | [11,90,92] |
| Rye | 316–350 | [11,90] |
| Barley | 191–399 | [11,56,91] |
| Millet | 358–421 | [11,93,94] |
| Sorghum | 277–380 | [93,94,95] |
| Soybean | 874–1300 | [11,20,59] |
| Lentil | 433–873 | [11,96,97] |
| Peanut | 600 | [20] |
| Macadamia nut | 365–1195 | [11,21] |
| Pistachio nut | 654–870 | [11,20,21] |
| Hazelnut | 317–643 | [11,21] |
| Walnut | 227–340 | [20,21] |
| Almond | 192–210 | [11,20,21] |
| Garlic | 200 | [11] |
| Potato | 80–170 | [11,59,92] |
| Carrot | 66–130 | [11,98] |
| Cabbage | 61–230 | [11,98] |
| Tomato | 37–50 | [11,98] |
| Broccoli | 71–150 | [11,98] |
| Cauliflower | 60 | [98] |
| Spinach | 78–90 | [11,98] |
| Orange | 87 | [11] |
| Avocado | 67 | [11] |
| Strawberry | 20–24 | [11,98] |
| Apple | 17–40 | [11,98] |
| White bread | 100 | [99] |
| Brown bread | 210 | [99] |
| Pork | 600–950 | [59,78,100] |
| Beef | 50–160 | [78,100] |
| Chicken breast | 40–170 | [78,100] |
| Liver, beef | 189 | [101] |
| Liver, pork | 283 | [101] |
| Tuna | 130 | [102] |
| Sardines | 10 | [102] |
| Baker’s yeasts | 1880 | [101] |
| Oyster mushroom | 50–150 | [103,104] |
| Button mushroom | 70–94 | [103,105] |
| Milk | 30–70 | [11,59,92,106] |
| Yogurt | 50–60 | [106] |
| Cheese, cheddar | 29 | [101] |
| Eggs | 40–80 | [92,107] |
| Deficiency | Gene Mutation | Disease | Symptoms |
|---|---|---|---|
| THTR1 | SLC19A2 | Thiamine-responsive megaloblastic anemia (TRMA, also known as the Rogger syndrome) | Megaloblastic anemia, diabetes mellitus, hearing loss |
| THTR2 | SLC19A3 | Biotin-thiamine responsive encephalopathy | Episodic encephalopathy, gait ataxia, seizures, bulbar dysfunction |
| Mitochondrial TPP transporter | SLC25A19 | Amish lethal microcephaly | Severe congenital microencephaly, death within the first year, episodic encephalopathy |
| TPK1 | TPK1 gene | Thiamine metabolism dysfunction syndrome 5 | Episodic encephalopathy, ataxia, dystonia and spasticity, loss of ability to walk |
| BCKDH | DBT gene | Maple syrup urine disease (MSUD) | Impaired mental development, seizures, lethargy, progressive neurodegeneration, maple syrup odor in the cerumen and the urine |
| Technique | Sensitivity nmol/L | Analytes | Matrix | Advantages | Disadvantages | Refs. | Publication Year |
|---|---|---|---|---|---|---|---|
| LC-MS | LOQ-LLOQ 0.15–246.32 (B1–B5) | B1, B1-TMP, B1-TPP, B2, B2-FAD, B3-NAM, B5 | human milk serum whole blood dry blood (VAMS) plasma tears urine |
|
| [227,228,229,230,231,232,233,234,235,236,237,238,239] | 2011–2020 |
| HPLC-FLD | LOQ-LLOQ 0.5–23.51 (B1-TPP) | B1, B1-TMP, B1-TPP, B2, B2-FAD, B2-FMN | whole blood dry blood spot plasma h uman milk |
|
| [240,241,242,243,244,245] | 2011–2020 |
| HPLC-PDA | LOD 212.56–4.09 × 103 (B2–B3-NAM) | B1, B2, B3-NAM | plasma urine |
|
| [246,247] | 2009–2014 |
| Sensors/nanodots/ CL/FLD/ECD | LOD 6.8 × 10−6–0.25 × 103 B1 | B1, B2 | urine serum plasma |
|
| [248,249,250,251,252] | 2002–2020 |
| Microbiological test kits | LLOQ 64.98–83.93 (B3–B5) | B3, B5, | serum |
|
| [253,254] | 2021 |
| HPLC-FLD kits | LOD 1.18–12.71 (B1-TPP–B2-FAD) | B1, B1-TPP, B1-TMP, B2, B2-FAD, B2-FMN | plasma whole blood |
|
| [255,256,257,258] | 2021 |
| ELISA kits | LOD 0.93 × 10−3–6.93 (B1–B2) | B1, B2 | serum plasma cell culture supernatant tissue breast milk sperm urine |
|
| [259,260,261] | 2021 |
| Individuals | Condition, Age | Dose (mg/kg) |
|---|---|---|
| Adults male | 1.2 | |
| Adults female | 1.1 | |
| pregnancy | 1.4 | |
| lactation | 1.4 | |
| Children | 0–6 month | 0.2 |
| 7–12 month | 0.3 | |
| 1–3 years | 0.5 | |
| 4–8 years | 0.6 | |
| 9–13 years | 0.9 | |
| Adolescent male | 14–18 years | 1.2 |
| Adolescent female | 14–18 years | 1.0 |
| Food | Riboflavin Content (μg/100 g) | References |
|---|---|---|
| Oat | 139 | [11] |
| Wheat | 57–265 | [56,58,309] |
| Rice, brown | 40–140 | [11,61] |
| Rice, white | 20–60 | [11,61] |
| Maize | 80–201 | [11,92] |
| Rye | 200–251 | [11,56] |
| Barley | 100–114 | [11,56] |
| Millet | 210–290 | [11,93] |
| Sorghum | 50–150 | [93,94,95] |
| Soybean | 870 | [11,20] |
| Lentil | 61–211 | [11,113,310] |
| Peanut | 100 | [20] |
| Macadamia nut | 162–367 | [11,21] |
| Pistachio nut | 160–447 | [11,20,21] |
| Hazelnut | 113–370 | [11,21] |
| Walnut | 150–395 | [20,21] |
| Almond | 1138–1432 | [11,20,21] |
| Garlic | 110 | [11] |
| Potato | 32–36 | [11,92,111] |
| Carrot | 10–58 | [11,98] |
| Cabbage | 20–40 | [11,98] |
| Tomato | 20–10月 | [11,98] |
| Broccoli | 117–120 | [11,98] |
| Spinach | 180–189 | [11,98] |
| Cauliflower | 90 | [98] |
| Orange | 40 | [11] |
| Avocado | 130 | [11] |
| Strawberry | 20–22 | [11,98] |
| Apple | 26–40 | [11,98] |
| White bread | 110 | [99] |
| Brown bread | 160–322 | [99,309] |
| Pork | 100–309 | [78,92,100] |
| Beef | 90–170 | [78,92,100] |
| Chicken breast | 30–120 | [78,92,100] |
| Liver, beef | 2760 | [101] |
| Liver, pork | 3000 | [101] |
| Tuna | 70 | [102] |
| Sardines | 340 | [102] |
| Oyster mushroom | 200–210 | [103,104] |
| Button mushroom | 384–390 | [103,105] |
| Baker´s yeast | 1113 | [101] |
| Milk | 169–180 | [11,92,311,312] |
| Yogurt | 160–270 | [106,311] |
| Cheese, cheddar | 441 | [101] |
| Eggs | 457–500 | [92,107] |
| Transporter | Localization | Mechanism |
|---|---|---|
| SLC52A1 | Placenta, small intestine | Na+, Cl–, pH-independent |
| SLC52A2 | Ubiquitous, higher in brain and salivary gland | Na+, Cl–, pH-independent |
| SLC52A3 | Testis, small intestine | Na+, Cl– independent, pH sensitive with about ½ half of the activity at neutral pH and 1/3 at pH 8.5 when compared to pH 5.5 |
| Vitamer | Median Plasma Concentration (nmol/L) | Median Erythrocyte Concentration (nmol/L) |
|---|---|---|
| Riboflavin | 10.5 | negligible |
| FMN | 6.6 | 44 |
| FAD | 74 | 469 |
| Food | Niacin Content (μg/100 g) | References |
|---|---|---|
| Oat | 961–2370 | [11,90,339,466] |
| Wheat | 4957–5700 | [56,90,92,93,339,466] |
| Rice, brown | 3500–5433 | [11,61,92,93,109,339,467] |
| Rice, white | 1300–2400 | [11,61,92,109,466,467] |
| Maize | 1900–3630 | [11,92,93,339] |
| Rye | 1700–4270 | [11,56,90,339,466] |
| Barley | 4523–5200 | [11,56,339,466] |
| Millet | 4500–4720 | [11,93,339] |
| Sorghum | 2920–4880 | [93,94,339,468] |
| Soybean | 1623 | [11,20] |
| Lentil | 1930–2605 | [11,96] |
| Peanut | 12100 | [20] |
| Macadamia nut | 2473 | [11] |
| Pistachio nut | 1300 | [11,20] |
| Hazelnut | 1800 | [11] |
| Walnut | 570 | [20] |
| Almond | 3618 | [11,20] |
| Garlic | 700 | [11] |
| Potato | 1035–1573 | [11,92,111] |
| Carrot | 837–983 | [11,448] |
| Cabbage | 234–323 | [11,448] |
| Tomato | 400–683 | [11,448] |
| Broccoli | 639–814 | [11,448] |
| Cauliflower | 600 | [98] |
| Spinach | 724–1000 | [11,448] |
| Orange | 249–282 | [11,448] |
| Avocado | 1738 | [11] |
| Strawberry | 291–600 | [11,448] |
| Apple | 91–126 | [11,448] |
| White bread | 1600 | [99] |
| Brown bread | 3800 | [99] |
| Pork | 5600–5900 | [78,92,100,469] |
| Beef | 4600–6500 | [78,92,100,466,469] |
| Chicken breast | 6801–9181 | [78,92,100,466] |
| Liver, pork | 13,200 | [101] |
| Liver, beef | 15,300 | [101] |
| Tuna | 21,900 | [102] |
| Sardines | 10,100 | [102] |
| Oyster mushroom | 4952–5870 | [103,104,308,470] |
| Button mushroom | 2800–3300 | [103,470] |
| Baker’s yeasts | 12,300 | [101] |
| Milk | 89–130 | [11,92,471] |
| Yogurt | 90–200 | [106,472] |
| Cheese, cheddar | 52 | [101] |
| Eggs | 50–75 | [92,107] |
| NAD+/NADH | catabolism | NAD+ → NADH | Glycolysis | glucose → pyruvate |
| Anaerobic glucose oxidation | Lactate ↔ pyruvate | |||
| B-oxidation of fatty acids | palmitoyl CoA → acetyl CoA | |||
| Amino acid catabolism | amino acid → acetyl/palmitoyl CoA | |||
| The Krebs cycle | acetyl CoA/pyruvate → energy | |||
| anabolism | NADH → NAD+ | Glyconeogenesis | lactate/pyruvate ↔ glycogen | |
| Fat synthesis | glucose → TAG | |||
| Steroid synthesis | cholesterol → various steroids | |||
| NAD+ → NADH | Cholesterol synthesis | acetyl CoA → cholesterol | ||
| NADPH/NADP+ | anabolism | NADPH → NADP+ | Fatty acid synthesis | acetyl CoA → palmitate |
| Cholesterol synthesis | acetyl CoA → cholesterol | |||
| Bile acid synthesis | cholesterol → cholate, chenodeoxycholate | |||
| Steroid synthesis | cholesterol → various steroids |
| Food | Pantothenic Acid Content (μg/100 g) | References |
|---|---|---|
| Oat | 800–1350 | [339,671] |
| Wheat | 950–1200 | [339,671] |
| Rice, brown | 660–1860 | [61,99,109,339,467] |
| Rice, white | 250–1080 | [61,99,109,467,671] |
| Maize | 420–650 | [339,671,705] |
| Rye | 1340–1460 | [339,512] |
| Barley | 280 | [339] |
| Millet | 850 | [339] |
| Sorghum | 1550–1630 | [468] |
| Soybean | 793–1431 | [101,710] |
| Lentil | 1030–1430 | [710] |
| Peanut | 1412–1767 | [714,715] |
| Macadamia nut | 800 | [716] |
| Pistachio nut | 470–500 | [716,717] |
| Hazelnut | 900 | [716] |
| Walnut | 470–600 | [716,717] |
| Almond | 300–471 | [716,718,719,720,721] |
| Garlic | 596 | [101] |
| Potato | 350–440 | [98,671] |
| Carrot | 270 | [98] |
| Cabbage | 210 | [98] |
| Tomato | 290–320 | [98,671] |
| Broccoli | 610–1300 | [98,671] |
| Cauliflower | 1010–1040 | [98,671] |
| Spinach | 280 | [98] |
| Orange | 240–370 | [671,699] |
| Avocado | 1390–1460 | [101,717] |
| Strawberry | 300–370 | [98,671,699] |
| Apple | 61–100 | [98,101,671] |
| Pear | 70 | [699] |
| White bread | 300–460 | [99,699] |
| Brown bread | 630–760 | [99,699,705] |
| Pork | 500–700 | [101,671,722] |
| Beef | 500–750 | [101,671,722,723] |
| Chicken breast | 870–1500 | [101,724,725] |
| Liver, beef | 7170–7900 | [101,671] |
| Liver, pork | 6650–6800 | [101,671] |
| Tuna | 230–500 | [102,705] |
| Sardines | 690–1090 | [102,705] |
| Oyster mushroom | 1300 | [470] |
| Button mushroom | 1360 | [470] |
| Baker’s yeasts | 4900 | [101] |
| Milk | 320–580 | [106,471,671,699] |
| Yogurt | 450–500 | [106,699] |
| Cheese, cheddar | 413–500 | [101,699] |
| Eggs | 1350–1600 | [107,671] |
| Symptoms | Sources | |
|---|---|---|
| Rodents (rats, mice, guinea pigs) | Growth: retardation, decrease in weight | [778,779,783,792,793,794,795,796] |
| Skin and mucosa: ruffing and discoloration of the fur, thinning of hair, alopecia, dryness of the skin with scaly desquamation, nasal discharge, watering of the eyes | ||
| Digestive track: diarrhea, duodenal changes (Lieberkühn crypts—enlargement, hyperplasia, increase in space between crypts, atrophy; villi diminution, epithelial changes to cuboid or flat, leading to ulcerations, perforation and chronic lesions), salivation | ||
| Nervous system: muscle weakness of the hind legs, convulsions, coma | ||
| Glands: adrenal lesions | ||
| Birds (ducklings and chicks) | Growth: retardation, decrease in weight | [783,797,798,799] |
| Skin: scaly dermatitis, skin lesions, scabs around beak and eyes, feather depigmentation, dermal edema | ||
| Nervous system: severe ataxia, tendency to fall and inability to rise and laying panting | ||
| Glands: lymphoid cell necrosis in the bursa of Fabricius and the thymus, and a lymphocytic paucity in the spleen | ||
| Pigs | Growth: failure to gain in weight, loss of appetite | [800,801,802] |
| Skin: loss of hair, roughness of the coat | ||
| Digestive track: diarrhea, severe colonic lesions | ||
| Nervous system: ataxia, lesions in sensory neurons, sudden lifting one of the limbs from the ground, unusual walk, inability to walk or stand | ||
| Respiratory system: cough and nasal secretion | ||
| Dogs | Growth: retardation Nervous system: sudden weakness, coma, rapid respiratory and heart rate, convulsions, spasticity of the hind legs | [784,803] |
| Digestive track: decreased appetite, gastrointestinal symptoms, gastritis or enteritis | ||
| Glands: fatty liver, mottled thymusis | ||
| Blood: blood level of glucose and chlorides were lower and non-protein nitrogen was elevated | ||
| Urinary system: hemorrhagic kidney degeneration | ||
| Humans | Nervous system: headache, irritability, restlessness, quarrelsomeness, excessive fatigue, numbness, paresthesia, muscle cramps, faulty coordination associated with tremor and peculiar gait | [776] |
| Digestive track: abdominal rumbling, diarrhea, epigastric burning, regurgitation | ||
| Glands: loss of eosinophilic response to adrenocorticotropic hormone, increased sensitivity to insulin |
| Vitamin | Transport | Blood Storage | Excretion | Toxicity/Adverse Effects |
|---|---|---|---|---|
| Thiamine | transporter mediated + passive diffusion | erythrocytes—as TPP | urinary | none/minor |
| Riboflavin | transporter mediated | erythrocytes—as FAD | urinary | none/minor |
| Niacin | transporter mediated + passive diffusion | erythrocytes | urinary | None/minor with exception of high doses of nicotinic acid but not of nicotinamide which can cause flushing, headache, lactic acidosis or hepatotoxicity |
| Pantothenic acid | transporter mediated + passive diffusion | erythrocytes | Urinary | none/minor |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hrubša, M.; Siatka, T.; Nejmanová, I.; Vopršalová, M.; Kujovská Krčmová, L.; Matoušová, K.; Javorská, L.; Macáková, K.; Mercolini, L.; Remião, F.; et al. Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5. Nutrients 2022, 14, 484. https://doi.org/10.3390/nu14030484
Hrubša M, Siatka T, Nejmanová I, Vopršalová M, Kujovská Krčmová L, Matoušová K, Javorská L, Macáková K, Mercolini L, Remião F, et al. Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5. Nutrients. 2022; 14(3):484. https://doi.org/10.3390/nu14030484
Chicago/Turabian StyleHrubša, Marcel, Tomáš Siatka, Iveta Nejmanová, Marie Vopršalová, Lenka Kujovská Krčmová, Kateřina Matoušová, Lenka Javorská, Kateřina Macáková, Laura Mercolini, Fernando Remião, and et al. 2022. "Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5" Nutrients 14, no. 3: 484. https://doi.org/10.3390/nu14030484
APA StyleHrubša, M., Siatka, T., Nejmanová, I., Vopršalová, M., Kujovská Krčmová, L., Matoušová, K., Javorská, L., Macáková, K., Mercolini, L., Remião, F., Máťuš, M., Mladěnka, P., & on behalf of the OEMONOM. (2022). Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5. Nutrients, 14(3), 484. https://doi.org/10.3390/nu14030484










