Association of Dietary Live Microbe Intake with Cardiovascular Disease in US Adults: A Cross-Sectional Study of NHANES 2007–2018
Abstract
1. Introduction
2. Methods
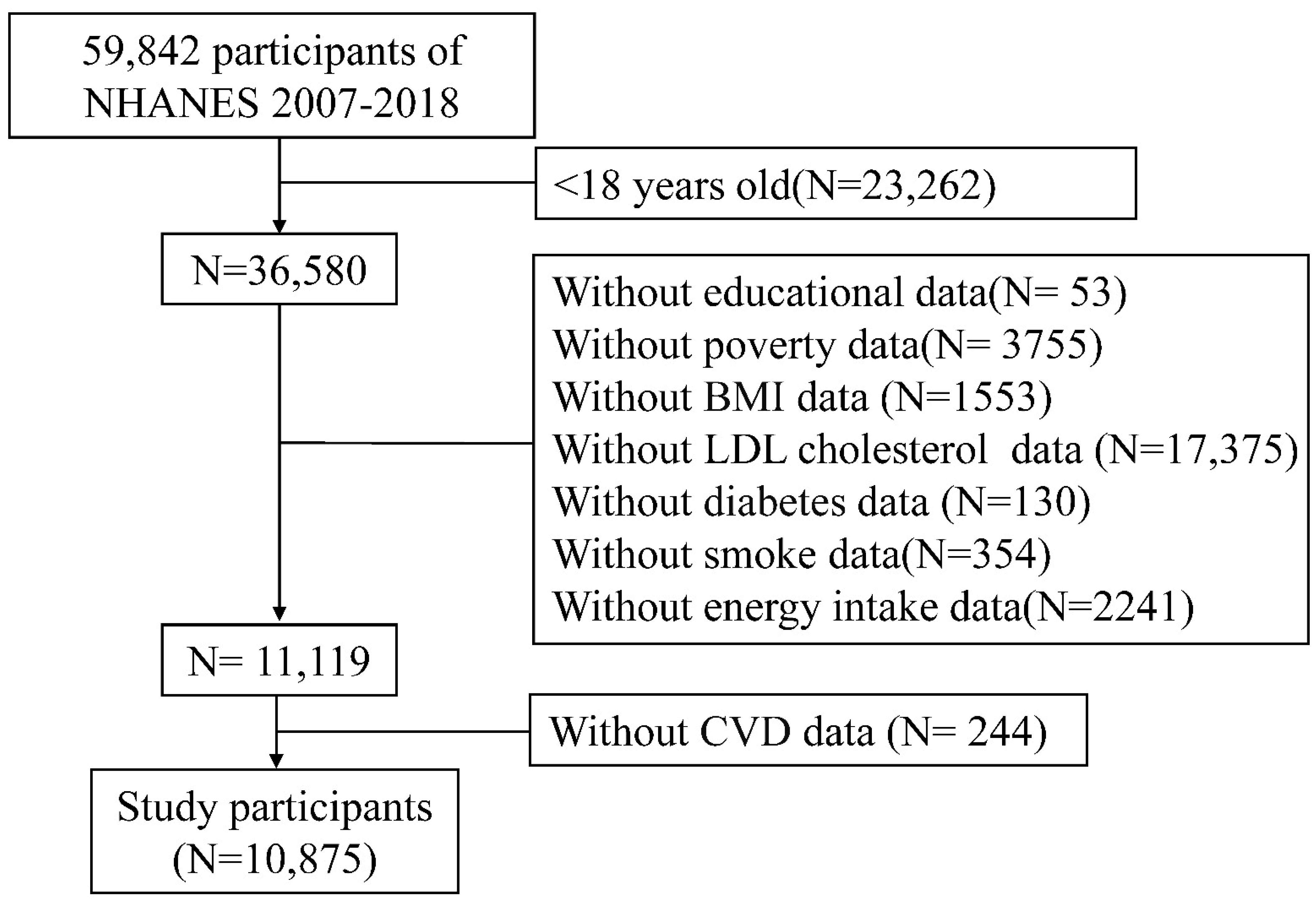
2.1. Data Source and Participants
2.2. Dietary Intakes and Live Microbial Category
2.3. Outcome Definitions
2.4. Covariates
2.5. Statistical Analyses
3. Results
3.1. Characteristics of the Included Population
3.2. Subjects in Different Dietary Live Microbe Groups
3.3. Association between Different Dietary Live Microbe Groups and CVD
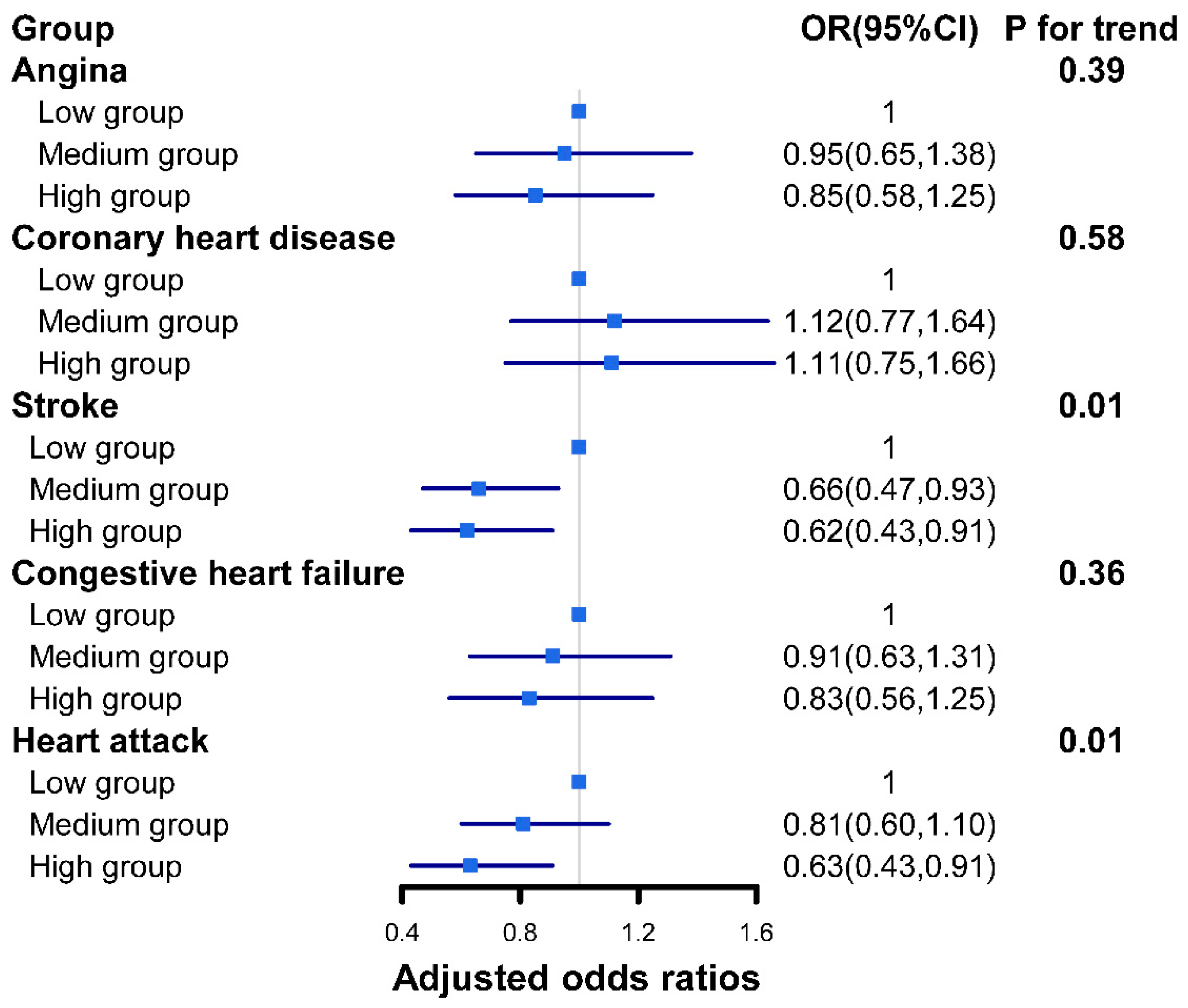
3.4. Association between Different Dietary Live Microbe Groups and Subtypes of CVD
3.5. Subgroup Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rook, G.A. 99th Dahlem conference on infection, inflammation and chronic inflammatory disorders: Darwinian medicine and the ‘hygiene’ or ‘old friends’ hypothesis. Clin. Exp. Immunol. 2010, 160, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Wastyk, H.C.; Fragiadakis, G.K.; Perelman, D.; Dahan, D.; Merrill, B.D.; Yu, F.B.; Topf, M.; Gonzalez, C.G.; Treuren, W.V.; Han, S.; et al. Gut-microbiota-targeted diets modulate human immune status. Cell 2021, 184, 4137–4153. [Google Scholar] [CrossRef] [PubMed]
- Roselli, M.; Natella, F.; Zinno, P.; Guantario, B.; Canali, R.; Schifano, E.; De Angelis, M.; Nikoloudaki, O.; Gobbetti, M.; Perozzi, G.; et al. Colonization Ability and Impact on Human Gut Microbiota of Foodborne Microbes From Traditional or Probiotic-Added Fermented Foods: A Systematic Review. Front. Nutr. 2021, 8, 689084. [Google Scholar] [CrossRef]
- Marco, M.L.; Hill, C.; Hutkins, R.; Slavin, J.; Tancredi, D.J.; Merenstein, D.; Sanders, E.M. Should There Be a Recommended Daily Intake of Microbes? J. Nutr. 2020, 150, 3061–3067. [Google Scholar] [CrossRef] [PubMed]
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Alonso, A.; Beaton, A.Z.; Bittencourt, M.S.; Boehme, A.K.; Buxton, A.E.; Carson, A.P.; Commodore-Mensah, Y.; et al. Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association. Circulation 2022, 145, e153–e639. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P.; Leong, D.; McKee, M.; Anand, S.S.; Schwalm, J.D.; Teo, K.; Mente, A.; Yusuf, S. Reducing the Global Burden of Cardiovascular Disease, Part 1: The Epidemiology and Risk Factors. Circ. Res. 2017, 121, 677–694. [Google Scholar] [CrossRef]
- Sonestedt, E.; Wirfalt, E.; Wallstrom, P.; Gullberg, B.; Orho-Melander, M.; Hedblad, B. Dairy products and its association with incidence of cardiovascular disease: The Malmo diet and cancer cohort. Eur. J. Epidemiol. 2011, 26, 609–618. [Google Scholar] [CrossRef]
- Praagman, J.; Franco, O.H.; Ikram, M.A.; Soedamah-Muthu, S.S.; Engberink, M.F.; van Rooij, F.J.; Hofman, A.; Geleijnse, J.M. Dairy products and the risk of stroke and coronary heart disease: The Rotterdam Study. Eur. J. Nutr. 2015, 54, 981–990. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Rangarajan, S.; Sheridan, P.; Mohan, V.; Gupta, R.; Lear, S.; Wentzel-Viljoen, E.; Avezum, A. Association of dairy intake with cardiovascular disease and mortality in 21 countries from five continents (PURE): A prospective cohort study. Lancet 2018, 392, 2288–2297. [Google Scholar] [CrossRef]
- Munoz Alferez, M.J.; Munoz-Garcia, A.; Moreno-Fernandez, J.; Lopez-Aliaga, I.; Diaz-Castro, J. Fermented goat milk consumption improves cardiovascular health during anemia recovery. J. Sci. Food. Agric. 2019, 99, 473–481. [Google Scholar] [CrossRef]
- Nozue, M.; Shimazu, T.; Charvat, H.; Mori, N.; Mutoh, M.; Sawada, N.; Iwasaki, M.; Yamaji, T.; Inoue, M.; Kokubo, Y.; et al. Fermented soy products intake and risk of cardiovascular disease and total cancer incidence: The Japan Public Health Center-based Prospective study. Eur. J. Clin. Nutr. 2021, 75, 954–968. [Google Scholar] [CrossRef]
- Jeddi, M.Z.; Yunesian, M.; Gorji, M.E.; Noori, N.; Pourmand, M.R.; Khaniki, G.R. Microbial evaluation of fresh, minimally-processed vegetables and bagged sprouts from chain supermarkets. J. Health Popu. l Nutr. 2014, 32, 391–399. [Google Scholar]
- Valentin-Bon, I.; Jacobson, A.; Monday, S.R.; Feng, P.C. Microbiological quality of bagged cut spinach and lettuce mixes. Appl. Environ. Microbiol. 2008, 74, 1240–1242. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Hutkins, R.; Hill, C.; Fulgoni, V.L.; Cifelli, C.J.; Gahche, J.; Slavin, J.L.; Merenstein, D.; Tancredi, D.J.; Sanders, M.E.; et al. A Classification System for Defining and Estimating Dietary Intake of Live Microbes in US Adults and Children. J. Nutr. 2022, 152, 1729–1736. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhu, Z.; Bundy, J.D.; Dorans, K.S.; Chen, J.; Hamm, L.L. Trends in Cardiovascular Risk Factors in US Adults by Race and Ethnicity and Socioeconomic Status, 1999–2018. JAMA 2021, 326, 1286–1298. [Google Scholar] [CrossRef]
- Lee, P.H.; Chan, C.W. Energy intake, energy required and mortality in an older population. Public Health Nutr. 2016, 19, 3178–3184. [Google Scholar] [CrossRef]
- Sirtori, C.R.; Fumagalli, R. LDL-cholesterol lowering or HDL-cholesterol raising for cardiovascular prevention. A lesson from cholesterol turnover studies and others. Atherosclerosis 2006, 186, 1–11. [Google Scholar] [CrossRef]
- McQueen, M.J.; Hawken, S.; Wang, X.; Ounpuu, S.; Sniderman, A.; Probstfield, J.; Steyn, K.; Sanderson, J.E.; Hasani, M.; Volkova, E.; et al. Lipids, lipoproteins, and apolipoproteins as risk markers of myocardial infarction in 52 countries (the INTERHEART study): A case-control study. Lancet 2008, 372, 224–233. [Google Scholar] [CrossRef]
- Ambrose, J.A.; Barua, R.S. The pathophysiology of cigarette smoking and cardiovascular disease: An update. J. Am. Coll. Cardiol. 2004, 43, 1731–1737. [Google Scholar] [CrossRef]
- Colafella, K.M.M.; Denton, K.M. Sex-specific differences in hypertension and associated cardiovascular disease. Nat. Rev. Nephrol. 2018, 14, 185–201. [Google Scholar] [CrossRef]
- Riar, S.S.; Fitchett, D.; FitzGerald, J.; Dehghani, P. Diabetes Mellitus and Cardiovascular Disease: An Evidence-Based Review of Provincial Formulary Coverage. Can. J. Cardiol. 2018, 34, 1362–1364. [Google Scholar] [CrossRef]
- Zhang, Y.; Moran, A.E. Trends in the Prevalence, Awareness, Treatment, and Control of Hypertension Among Young Adults in the United States, 1999 to 2014. Hypertension 2017, 70, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Menke, A.; Casagrande, S.; Geiss, L.; Cowie, C.C. Prevalence of and Trends in Diabetes Among Adults in the United States, 1988–2012. JAMA 2015, 314, 1021–1029. [Google Scholar] [CrossRef]
- Taylor, B.C.; Lejzerowicz, F.; Poirel, M.; Shaffer, J.P.; Jiang, L.; Aksenov, A.; Litwin, N.; Humphrey, G.; Martino, C.; Miller-Montgomery, S.; et al. Consumption of Fermented Foods Is Associated with Systematic Differences in the Gut Microbiome and Metabolome. Msystems 2020, 5, e00901–e00919. [Google Scholar] [CrossRef] [PubMed]
- Oniszczuk, A.; Oniszczuk, T.; Gancarz, M.; Szymanska, J. Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases. Molecules 2021, 26, 1172. [Google Scholar] [CrossRef] [PubMed]
- Gadelha, C.; Bezerra, A.N. Effects of probiotics on the lipid profile: Systematic review. J. Vasc. Bras. 2019, 18, e20180124. [Google Scholar] [CrossRef] [PubMed]
- Niamah, A.K.; Sahi, A.A.; Al-Sharifi, A.S.N. Effect of Feeding Soy Milk Fermented by Probiotic Bacteria on Some Blood Criteria and Weight of Experimental Animals. Probiotics Antimicrob. Proteins 2017, 9, 284–291. [Google Scholar] [CrossRef]
- Yadav, R.; Khan, S.H.; Mada, S.B.; Meena, S.; Kapila, R.; Kapila, S. Consumption of Probiotic Lactobacillus fermentum MTCC: 5898-Fermented Milk Attenuates Dyslipidemia, Oxidative Stress, and Inflammation in Male Rats Fed on Cholesterol-Enriched Diet. Probiotics Antimicrob. Proteins 2019, 11, 509–518. [Google Scholar] [CrossRef]
- Tenorio-Jimenez, C.; Martinez-Ramirez, M.J.; Tercero-Lozano, M.; Arraiza-Irigoyen, C.; Del Castillo-Codes, I.; Olza, J.; Plaza-Diaz, J.; Fontana, L.; Migueles, H.J.; Olivares, M.; et al. Evaluation of the effect of Lactobacillus reuteri V3401 on biomarkers of inflammation, cardiovascular risk and liver steatosis in obese adults with metabolic syndrome: A randomized clinical trial (PROSIR). BMC Complement Altern. Med. 2018, 18, 306. [Google Scholar] [CrossRef]
- Olas, B. Probiotics, Prebiotics and Synbiotics-A Promising Strategy in Prevention and Treatment of Cardiovascular Diseases? Int. J. Mol. Sci. 2020, 21, 9737. [Google Scholar] [CrossRef]
- Redman, M.G.; Ward, E.J.; Phillips, R.S. The efficacy and safety of probiotics in people with cancer: A systematic review. Ann. Oncol. 2014, 25, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
| Variable | CVD | p-Value | |
|---|---|---|---|
| No | Yes | ||
| No. of participants | 9616 | 1269 | |
| Age [years] | 47.05 (0.30) | 65.39 (0.52) | <0.0001 |
| Energy intake [kcal] | 4189.88 (23.69) | 3823.38 (64.73) | <0.0001 |
| Serum ldl-cholesterol [mg/dL] | 99.72 (1.43) | 116.08 (0.58) | <0.0001 |
| Gender (%) | <0.001 | ||
| Female | 5164 (53.34) | 555 (46.54) | |
| Male | 4442 (46.66) | 714(53.46) | |
| Race/ethnicity (%) | <0.0001 | ||
| Mexican American | 1445 (7.95) | 107 (4.58) | |
| Non-Hispanic Black | 1886 (10.02) | 268 (11.38) | |
| Non-Hispanic White | 4191 (68.83) | 722 (75.40) | |
| Other Hispanic | 979 (5.62) | 103 (3.25) | |
| Other Racer | 1105 (7.58) | 69 (5.40) | |
| FIR (%) | 0.03 | ||
| <1.2 | 2590 (18.78) | 383 (22.05) | |
| ≥1.2 | 7016 (81.22) | 7016 (81.22) | |
| Educational status (%) | <0.0001 | ||
| >high school | 5460 (63.91) | 547 (49.37) | |
| high school | 2122 (22.54) | 333 (27.34) | |
| <high school | 2024 (13.55) | 389 (23.29) | |
| Vigorous/moderate recreational activities (%) | <0.0001 | ||
| Yes | 4870 (55.66) | 422 (39.86) | |
| No | 4736 (44.34) | 847 (60.14) | |
| Smoke (%) | |||
| Now | 1803 (18.14) | 266 (20.89) | <0.0001 |
| Former | 2261 (25.13) | 485 (40.73) | |
| Never | 5542 (56.73) | 518 (38.38) | |
| Body Mass Index (%) | <0.0001 | ||
| Normal | 2827 (30.41) | 273 (22.95)) | |
| Overweight | 3159 (32.99) | 400 (29.17) | |
| Obesity | 3620 (36.60) | 596 (47.88) | |
| Hypertension (%) | <0.0001 | ||
| Yes | 3734 (35.97) | 1008 (75.74) | |
| No | 5872 (64.03) | 261 (24.26) | |
| Diabetes mellitus (%) | <0.0001 | ||
| Yes | 1752 (14.14) | 579 (40.47) | |
| Borderline | 1681 (17.38) | 237 (18.72) | |
| No | 6173 (68.48) | 453 (40.81) | |
| Dietary livemicrobe group (%) | 0.01 | ||
| Low | 3380 (31.67) | 515 (36.71) | |
| Medium | 3910 (38.54) | 503 (39.15) | |
| High | 2316 (29.78) | 251(24.14) | |
| Variable | Low Dietary Live Microbe Group | Medium Dietary Live Microbe Group | High Dietary Live Microbe Group | p-Value |
|---|---|---|---|---|
| No. of participants | 3805 | 4413 | 2567 | |
| Age [years] | 46.71 (0.45) | 50.43 (0.44) | 48.99 (0.46) | <0.0001 |
| Energy intake [kcal] | 4003.75 (43.68) | 4167.64 (37.62) | 4303.42 (47.05) | <0.0001 |
| Serum ldl-cholesterol [mg/dL] | 114.53 (0.95) | 113.84 (0.82) | 115.35 (1.11) | 0.51 |
| Gender (%) | <0.001 | |||
| Female | 1906 (47.97) | 2362 (54.35) | 1451 (55.69) | |
| Male | 1989 (52.03) | 2051 (45.65) | 1116 (44.31) | |
| Race/ethnicity (%) | <0.0001 | |||
| Mexican American | 469 (6.86) | 786 (9.87) | 297 (5.52) | |
| Non-Hispanic Black | 1063 (15.07) | 771 (9.43) | 320 (5.68) | |
| Non-Hispanic White | 1569 (64.55) | 1935 (67.40) | 1409 (77.59) | |
| Other Hispanic | 366 (5.48) | 454 (5.82) | 262 (4.73) | |
| Other Racer | 428 (8.03) | 467 (7.49) | 279 (6.48) | |
| FIR (%) | <0.0001 | |||
| <1.2 | 1315 (27.00) | 1120 (16.80) | 538 (13.42) | |
| ≥1.2 | 2580 (73.00) | 3293 (83.20) | 2029 (86.58) | |
| Educational status (%) | <0.0001 | |||
| >high school | 1867 (53.44) | 2418 (62.94) | 1722 (71.94) | |
| high school | 995 (27.22) | 979 (22.08)) | 481 (19.58) | |
| <high school | 1033 (19.34) | 1016 (14.99) | 364 (8.48) | |
| Vigorous/moderate recreational activities (%) | <0.0001 | |||
| Yes | 1657 (45.49) | 2196 (55.88) | 1439 (61.37) | |
| No | 2238 (54.51) | 2217 (44.12) | 1128 (38.63) | |
| Smoke (%) | <0.0001 | |||
| Now | 985 (26.77) | 707 (14.92) | 377 (13.79) | |
| Former | 896 (23.57) | 1176 (28.22) | 674 (27.89) | |
| Never | 2014 (49.66) | 2530 (56.85) | 1516 (58.32) | |
| Body Mass Index (%) | <0.0001 | |||
| Normal | 1043 (26.23) | 1268 (30.38) | 789 (32.59) | |
| Overweight | 1214 (30.95) | 1489 (32.87) | 856 (34.13) | |
| Obesity | 1638 (42.82) | 1656 (36.75) | 922 (33.28) | |
| Hypertension (%) | <0.001 | |||
| Yes | 1765 (41.53) | 1951 (41.02) | 1026 (36.26) | |
| No | 2130 (58.47) | 2462 (58.98) | 1541 (63.74) | |
| Diabetes mellitus (%) | <0.0001 | |||
| Yes | 845 (16.92) | 1019 (18.67) | 467 (13.77) | |
| Borderline | 706 (18.17) | 792 (17.89) | 420 (16.28) | |
| No | 2344 (64.90) | 2602 (63.44) | 1680 (69.96) | |
| CVD (%) | 0.01 | |||
| Yes | 515 (10.98) | 503 (9.75) | 251 (7.94) | |
| No | 3380 (89.02) | 3910 (90.25) | 2316 (92.06) |
| Outcomes | Model | Low Dietary Live Microbe Group OR (95%) | Medium Dietary Live Microbe Group OR (95%) | High Dietary Live Microbe Group OR (95%) | p for Trend |
|---|---|---|---|---|---|
| CVD | Crude | 1.00 (Reference) | 0.88(0.72,1.06) | 0.70(0.55,0.88) * | 0.003 |
| Model1 | 1.00 (Reference) | 0.67(0.53,0.84) * | 0.61(0.48,0.79) * | <0.001 | |
| Model2 | 1.00 (Reference) | 0.77(0.61,0.97) * | 0.76(0.59,0.99) * | 0.039 | |
| Model3 | 1.00 (Reference) | 0.78(0.62,0.99) * | 0.83(0.64,1.08) | 0.148 |
| Subgroup Variable | Total Number = 6372, Stroke = 450 | Total Number = 6372, Heart Attack = 438 | ||||
|---|---|---|---|---|---|---|
| OR High vs. Low (95% CI) | p-Value | p-Interaction | OR High vs. Low (95% CI) | p-Value | p-Interaction | |
| Gender | 0.03 | 0.93 | ||||
| Female | 1.07 (0.65,1.78) | 0.41 | 0.63 (0.41,0.97) | 0.04 | ||
| Male | 0.42 (0.25,0.73) | <0.01 | 0.74 (0.43,1.29) | 0.29 | ||
| Age | 0.05 | 0.81 | ||||
| <60 | 0.42 (0.18,0.99) | 0.05 | 0.95 (0.44, 2.04) | 0.89 | ||
| ≥60 | 0.77 (0.54,1.12) | 0.17 | 0.59 (0.37,0.93) | 0.02 | ||
| Race/ethnicity | 0.93 | 0.59 | ||||
| Mexican American | 0.32 (0.10, 1.06) | 0.06 | 0.86 (0.21, 3.60) | 0.83 | ||
| Non-Hispanic Black | 0.75 (0.31,1.81) | 0.51 | 0.85 (0.30, 2.45) | 0.76 | ||
| Non-Hispanic White | 0.59 (0.37,0.94) | 0.03 | 0.65 (0.44,0.98) | 0.04 | ||
| Other Hispanic | 1.21 (0.30, 4.92) | 0.78 | 0.72 (0.15, 3.53) | 0.68 | ||
| Other Racer | 0.70 (0.27, 1.83) | 0.46 | 0.54 (0.10, 2.96) | 0.47 | ||
| FIR | 0.83 | 0.33 | ||||
| <1.2 | 0.57 (0.21,1.52) | 0.25 | 0.62 (0.41,0.92) | 0.02 | ||
| ≥1.2 | 0.63 (0.41,0.96) | 0.03 | 0.82 (0.35, 1.89) | 0.63 | ||
| Year cycle | 0.62 | 0.89 | ||||
| 2007–2012 | 0.60 (0.41,0.88) | 0.01 | 0.72 (0.42,1.21) | 0.20 | ||
| 2013–2018 | 0.65 (0.33,1.28) | 0.19 | 0.56 (0.32, 0.98) | 0.04 | ||
| Vigorous/moderate recreational activities | 0.36 | 0.20 | ||||
| Yes | 0.80 (0.37, 1.72) | 0.56 | 0.77 (0.42,1.41) | 0.39 | ||
| No | 0.53 (0.32,0.88) | 0.02 | 0.52 (0.32, 0.85) | 0.01 | ||
| Body Mass Index | 0.51 | 0.83 | ||||
| Normal | 0.65 (0.29,1.48) | 0.30 | 0.78 (0.37, 1.62) | 0.50 | ||
| Overweight | 0.83 (0.45,1.55) | 0.56 | 0.62 (0.32, 1.17) | 0.14 | ||
| Obesity | 0.52 (0.28,0.96) | 0.04 | 0.64 (0.37,1.10) | 0.10 | ||
| Educational status | 0.87 | 0.89 | ||||
| >high school | 0.59 (0.33,1.04) | 0.07 | 0.67 (0.42,1.07) | 0.09 | ||
| high school | 0.72 (0.34,1.52) | 0.38 | 0.62 (0.28, 1.35) | 0.22 | ||
| <high school | 0.67 (0.30,1.49) | 0.32 | 0.79 (0.37, 1.67) | 0.53 | ||
| Smoke | 0.73 | 0.21 | ||||
| Now | 0.55 (0.21, 1.47) | 0.23 | 0.98 (0.40, 2.40) | 0.96 | ||
| Former | 0.82 (0.46,1.46) | 0.50 | 0.40 (0.20, 0.79) | 0.01 | ||
| Never | 0.53 (0.30,0.93) | 0.03 | 0.74 (0.41,1.34) | 0.31 | ||
| Hypertension | 0.15 | 0.94 | ||||
| Yes | 0.75 (0.51,1.10) | 0.14 | 0.69 (0.43,1.13) | 0.14 | ||
| No | 0.44 (0.20, 0.97) | 0.04 | 0.67 (0.31, 1.44) | 0.30 | ||
| Diabetes mellitus | 0.30 | 0.14 | ||||
| Yes | 0.51 (0.32,0.84) | 0.01 | 0.40 (0.21,0.77) | 0.01 | ||
| Borderline | 0.45 (0.16, 1.30) | 0.14 | 1.17 (0.53, 2.58) | 0.70 | ||
| No | 0.72 (0.39,1.33) | 0.30 | 0.68 (0.35, 1.33) | 0.26 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, L.; Wang, Q. Association of Dietary Live Microbe Intake with Cardiovascular Disease in US Adults: A Cross-Sectional Study of NHANES 2007–2018. Nutrients 2022, 14, 4908. https://doi.org/10.3390/nu14224908
Han L, Wang Q. Association of Dietary Live Microbe Intake with Cardiovascular Disease in US Adults: A Cross-Sectional Study of NHANES 2007–2018. Nutrients. 2022; 14(22):4908. https://doi.org/10.3390/nu14224908
Chicago/Turabian StyleHan, Lu, and Qi Wang. 2022. "Association of Dietary Live Microbe Intake with Cardiovascular Disease in US Adults: A Cross-Sectional Study of NHANES 2007–2018" Nutrients 14, no. 22: 4908. https://doi.org/10.3390/nu14224908
APA StyleHan, L., & Wang, Q. (2022). Association of Dietary Live Microbe Intake with Cardiovascular Disease in US Adults: A Cross-Sectional Study of NHANES 2007–2018. Nutrients, 14(22), 4908. https://doi.org/10.3390/nu14224908





