Effect of a Multi-Strain Probiotic on the Incidence and Severity of Necrotizing Enterocolitis and Feeding Intolerances in Preterm Neonates
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Setting
2.3. Study Participants
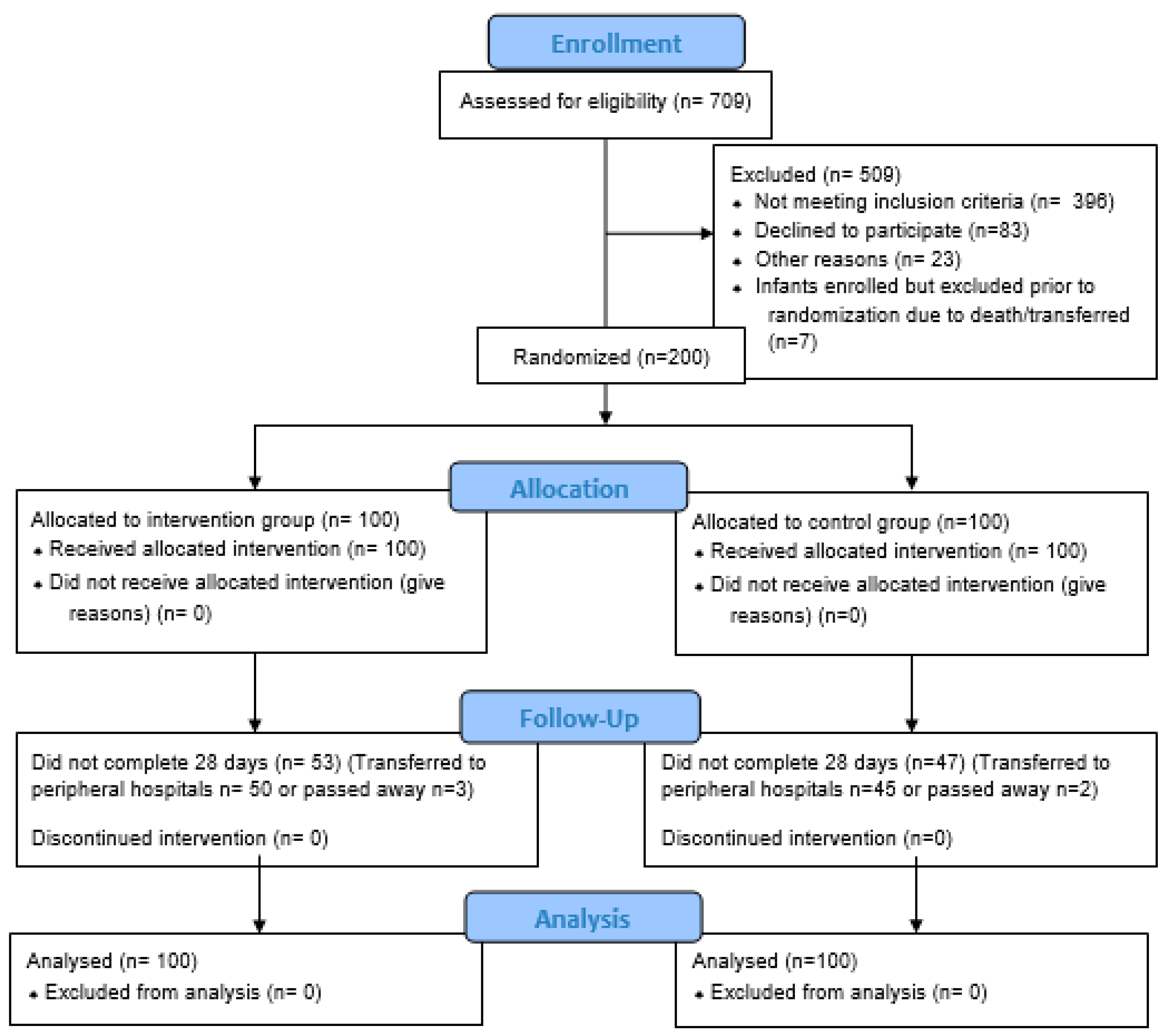
2.4. Randomization
2.5. Procedures
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rees, C.M.; Eaton, S.; Pierro, A. National prospective surveillance study of necrotizing enterocolitis in neonatal intensive care units. J. Pediatr. Surg. 2010, 45, 1391. [Google Scholar] [CrossRef]
- Battersby, C.; Santhalingam, T.; Costeloe, K.; Modi, N. Incidence of neonatal necrotising enterocolitis in high-income countries: A systematic review. Arch. Dis. Child. Fetal Neonatal Ed. 2018, 103, F182. [Google Scholar] [CrossRef]
- Han, S.M.; Hong, C.R.; Knell, J.; Edwards, E.M.; Morrow, K.A.; Soll, R.F.; Modi, B.P.; Horbar, J.D.; Jaksic, T. Trends in incidence and outcomes of necrotizing enterocolitis over the last 12 years: A multicenter cohort analysis. J. Pediatr. Surg. 2020, 55, 998. [Google Scholar] [CrossRef] [PubMed]
- Kafetzis, D.; Skevaki, C.; Costalos, C. Neonatal necrotizing enterocolitis: An overview. Curr. Opin. Infect. Dis. 2003, 16, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Denning, N.; Prince, J. Neonatal intestinal dysbiosis in necrotizing enterocolitis. Mol. Med. 2018, 24, 4. [Google Scholar] [CrossRef]
- Wandro, S.; Osborne, S.; Enriquez, C.; Bixby, C.; Arrieta, A.; Whiteson, K. The microbiome and metabolome of preterm infant stool are personalized and not driven by health outcomes, including Necrotizing Enterocolitis and late-onset sepsis. mSphere 2018, 3, e00104-18. [Google Scholar] [CrossRef] [PubMed]
- AlFaleh, K.; Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 2014, 2–85. [Google Scholar] [CrossRef]
- Aceti, A.; Gori, D.; Barone, G.; Callegari, M.L.; Di Mauro, A.; Fantini, M.P.; Indrio, F.; Maggio, L.; Meneghin, F.; Morelli, L.; et al. Probiotics for prevention of necrotizing enterocolitis in preterm infants: Systematic review and meta-analysis. Ital. J. Pediatr. 2015, 41, 89. [Google Scholar] [CrossRef]
- Caplan, M. Probiotic and prebiotic supplementation for the prevention of neonatal necrotizing enterocoliti. J. Perinatol. 2009, 29, S2–S6. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Luedtke, S.; Yang, J.; Wild, H. Probiotics and Necrotizing Enterocolitis: Finding the Missing Pieces of the Probiotic Puzzle. J. Pediatr. Pharmacol. Ther. 2012, 17, 308–328. [Google Scholar] [CrossRef]
- Walsh, M.; Kliegman, R. Necrotizing enterocolitis: Treatment based on staging criteria. Pediatr. Clin. N. Am. 1986, 33, 179–201. [Google Scholar] [CrossRef]
- Gregory, K.E.; DeForge, C.E.; Natale, K.M.; Phillips, M.; Van Marter, L.J. Necrotizing enterocolitis in the premature infant: Neonatal nursing assessment, disease pathogenesis, and clinical presentation. Adv. Neonatal Care 2011, 11, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Gareau, M.; Sherman, P.; Walker, W. Probiotics and the gut microbiota in intestinal health and disease. Nat. Rev. Gastroenterol. Ad. Hetapol. 2010, 7, 503–514. [Google Scholar] [CrossRef]
- Desfrere, L.; de Oliveira, I.; Goffinet, F.; El Ayoubi, M.; Firtion, G.; Bavoux, F.; Françoise, B.; Pierre-Henri, J.; Moriette, G. Increased incidence of necrotizing enterocolitis in premature infants born to HIV-positive mothers. AIDS 2005, 19, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.; Marrs, E.C.L.; Nelson, A.; Lanyon, C.; Perry, J.D.; Embleton, N.; Cummings, S.P.; Berrington, J. Development of the Preterm Gut Microbiome in Twins at Risk of Necrotising Enterocolitis and Sepsis. PLoS ONE 2013, 8, e73465. [Google Scholar]
- Deshpande, G.; Rao, S.; Patole, S. Probiotics for prevention of necrotising enterocolitis in preterm neonates with very low birthweight: A systematic review of randomised controlled trials. Lancet 2007, 369, 1614–1620. [Google Scholar] [CrossRef]
- Claud, E.; Walker, W. Hypothesis: Inappropriate colonization of the premature intestine can cause neonatal necrotizing enterocolitis. FASEB J. 2001, 15, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Elgin, T.; Kern, S.; McElroy, S. Develpment of the Neontal Intetinal Microbiome and its Association with Necrotizing Enterocolitis. Clin. Ther. 2016, 38, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Riezzo, G.; Cavallo, L.; Di Mauro, A.; Francavilla, R. Physiological basis of food intolerance in VLBW. J. Matern. Neonatal Med. 2011, 24, 64–66. [Google Scholar] [CrossRef]
- Fanaro, S. Feeding intolerance in the preterm infant. Early Hum. Dev. 2013, 89, S13–S20. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, G.; Jape, G.; Rao, S.; Patole, S. Benefits of probiotics in preterm neonates in low-income and mediumincome countries: A systematic review of randomised controlled trials. BMJ Open 2017, 7, e017638. [Google Scholar] [CrossRef]
- Olse, R.; Greisen, G.; Schroder, M.; Brok, J. Prophylactic probiotics for preterm infants: A systematic review and meta-analysis of observational studies. Neonatology 2016, 109, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Caplan, M.; Frost, B. Myth: Necrotizing enterocolitis: Probiotics will end the disease, and surgical intervention improves the outcome. Semin. Fetal Neonatal Med. 2011, 16, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Athalye-Jape, G.; Deshpande, G.; Rao, S.; Patole, S. Benefits of probiotics on enteral nutrition in preterm neonates: A systematic review. Am. J. Clin. Nutr. 2014, 100, 1508–1519. [Google Scholar] [CrossRef] [PubMed]
- Stoll, B.J.; Hansen, N.; Fanaroff, A.A.; Wright, L.L.; Carlo, W.A.; Ehrenkranz, R.A.; Lemons, J.A.; Donovan, E.F.; Stark, A.R.; Tyson, J.E.; et al. Late-onset sepsis in very low birth weight neonates: The experience of the NICHD Neonatal Research Network. Pediatrics 2002, 110, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.; Wilson, M. Feeding Intolerance, A Concept Analysis. Adv. Neonatal Care 2011, 11, 149–154. [Google Scholar] [CrossRef]
- Xin-Tian, H.; Jun, T.; De-Zhi, M. Effect of oral administration of probiotics on intestinal colonization with drug-resistant bacteria in preterm infants. CJCP 2014, 16, 606–609. [Google Scholar]
- Murphy, K.; Ross, R.P.; Ryan, A.; Dempsey, E.M.; Stanton, C. Protiotics, prebiotics and synbiotics for the prevention of necrotizing enterocolitis. Front. Nutr. 2021, 8, 1–10. [Google Scholar] [CrossRef]
- Alsaied, A.; Islam, N.; Thalib, L. Global incidence of Necrotizing Enterocolitis: A systemati review and Meta-analysis. BMC Pediatr. 2020, 20, 344. [Google Scholar] [CrossRef] [PubMed]
- Chokoe, M.J.; Wright, C.A.; Bezuidenhout, J.; Moore, S.W.; Smith, J. Necrotizing Enterocolitis in HIV-Exposed and Nonexposed Infants: Clinical Presentation and Histopathological Features. Pediatr. Dev. Pathol. 2014, 15, 293–297. [Google Scholar] [CrossRef] [PubMed]
- van Niekerk, E.; Nel, D.; Blaauw, R.; Gert, F.K. Probiotics Reduce Necrotizing Enterocolitis Severity in HIV-exposed Premature Infants. J. Trop. Pediatr. 2015, 61, 155–164. [Google Scholar] [CrossRef]
- Bi, L.; Yan, B.; Yang, Q.L.M.; Cui, H. Which is the best probiotic treatment strategy to prevent the necrotizing enterocolitis in premature infants. Medicine 2019, 98, e17521. [Google Scholar] [CrossRef]
- Sawh, S.C.; Desphande, S.; Jansen, S.; Reynaert, C.J.; Jones, P.M. Prevention of necrotizing enterocolitis with probiotics: A systematic review and meta-analysis. PeerJ 2016, 4, e2429. [Google Scholar] [CrossRef] [PubMed]
- Robertson, C.; Savva, G.M.; Clapuci, R.; Jones, J.; Maimouni, H.; Brown, E.; Minocha, A.; Hall, L.J.; Clarke, P. Incidence of necrotising enterocolitis before and after introducing routine prophylactic Lactobacillus and Bifidobacterium probiotics. Arch. Dis. Child. Fetal Neonatal Ed. 2020, 105, F380–F386. [Google Scholar] [CrossRef] [PubMed]
- Hoyos, A. Reduced Incidence of Necrotizing Enterocoloitis Associated With Enteral Administration of Lactobacillus acidophils and Bifidobacterium infantis in an Intenstive Care Unit. Int. J. Infect. Dis. 1999, 3, 197–202. [Google Scholar] [CrossRef]
- Totsu, S.; Yamasaki, C.; Terahara, M.; Uchiyama, A.; Kusuda, S.; Probiotics Study Group in Japan. Bifidobacterium and enteral feeding in preterm infants: Cluster-randomized tria. Pediatr. Int. 2014, 56, 714–719. [Google Scholar] [CrossRef]
| Probiotic Group (n = 100) | Placebo Group (n = 100) | |
|---|---|---|
| Gender | ||
| Male (n, %) | 47 (47) | 37 (37) |
| Birth weight | ||
| 750–1000 g (n, %) | 30 (30) | 32 (32) |
| 1001–1500 g (n, %) | 70 (70) | 68 (68) |
| Gestational Age | ||
| 26–28 weeks (n, %) | 34 (34) | 30 (30) |
| 29–32 weeks (n, %) | 60 (60) | 62 (62) |
| 33–36 weeks (n, %) | 6 (6) | 8 (8) |
| Apgar score (10 min) | ||
| <4 (n, %) | 0 (0) | 1 (1) |
| 4–7 (n, %) | 10 (10) | 9 (9) |
| >7 (n, %) | 89 (89) | 89 (89) |
| No Apgar (born before arrival (n, %) | 1 (1) | 1 (1) |
| HIV | ||
| Exposed (n, %) | 22 (22) | 26 (26) |
| Birth number | ||
| Single neonate (n, %) | 79 (79) | 86 (86) |
| Twin neonates (n, %) | 21 (21) | 14 (14) |
| Reason for premature delivery | ||
| SPPROM (n, %) | 16 (16) | 20 (20) |
| FD (n, %) | 57 (57) | 43 (43) |
| EOPET (n, %) | 2 (2) | 4 (4) |
| Placenta abruption (n, %) | 2 (2) | 7 (7) |
| IUGR (n, %) | 1 (1) | 6 (6) |
| SPTL (n, %) | 18 (18) | 18 (18) |
| HELLP (n, %) | 2 (2) | 1 (1) |
| Placenta praevia (n, %) | 2 (2) | 1 (1) |
| Empiric antibiotic use for presumed sepsis at birth | ||
| Neonates classified as at septic risk at birth and received empiric antibiotics (n, %) | 57 (57) | 55 (55) |
| Days (mean days ± SD) | 3.8 ± 2.1, (range 1–12) | 3.8 ± 2.0, (range 1–10) |
| Probiotic Group (n = 100) n (%) | Placebo Group (n = 100) n (%) | |
|---|---|---|
| NEC | 0 (0%) | 5 (5%) |
| NEC Bell’s diagnosed Stage 1A | 0 | 2 (2%) |
| NEC Bell’s diagnosed Stage 1B | 0 | 1 (1%) |
| NEC Bell’s diagnosed Stage 2A | 0 | 1 (1%) |
| NEC Bell’s diagnosed Stage 3B | 0 | 1 (1%) |
| Neonate passed away due to NEC | 0 | 1 (1%) |
| Bell’s I | Bell’s II | Bell’s III | |
|---|---|---|---|
| Treatment group | |||
| Probiotic | 0 | 0 | 0 |
| Placebo | 3 | 1 | 1 |
| HIV-exposure | |||
| HIV-exposed | 0 | 0 | 0 |
| HIV-unexposed | 3 | 1 | 1 |
| Gestational age | |||
| 26–28 weeks | 1 | 0 | 1 |
| 29–32 weeks | 2 | 1 | 0 |
| 33–36 weeks | 0 | 0 | 0 |
| Birth weight (grams) | |||
| 750–1000 g | 2 | 0 | 1 |
| 1001–1500 g | 1 | 1 | 0 |
| Probiotic Group | Placebo Group | |
|---|---|---|
| (n = 100) | (n = 100) | |
| Day first feed received (days), (mean days ± SD) | 3.1 ± 1.1 | 3.00 ± 1.0 |
| Day full feeds reached (days), (mean days ± SD) | 8.7 ± 2.0 | 9.7; ± 4.3 |
| Duration of TPN (days) (mean days ± SD) | 0.1 ± 1.0 | 0.5 ± 2.2 |
| Probiotic Group (n = 100) n (%) | Placebo Group (n = 100) n (%) | |
|---|---|---|
| Feeding tolerance | 86 (86%) | 65 (65%) |
| Feeding intolerance diagnosed (number of neonates) | 14 (14%) | 35 (35%) |
| Emesis observed (number of neonates) | 49 (49%) | 69 (69%) |
| Abdominal distension observed (number of neonates) | 24 (24%) | 48 (48%) |
| Neonates NPO for 1 or more days due to emesis (number of neonates) | 8 (8%) | 20 (20%) |
| Neonate received TPN (number of neonates) | 2 (2%) | 5 (5%) |
| Duration NPO (days) (mean ± SD) | 0.2 ± 0.4 | 0.4 ± 0.7 |
| Crude Odds Ratio | Adjusted Odds Ratio * | |
| Development of feeding intolerance | 0.19 (0.09 to 0.43); p < 0.001 | 0.11 (0.04 to 0.30); p < 0.001 |
| Crude Rate Ratio | Adjusted Rate Ratio * | |
| Number of days with emesis | 0.41 (0.28 to 0.60); p < 0.001 | 0.39 (0.26 to 0.56); p < 0.001 |
| Number of emesis observed | 0.40 (0.26 to 0.43); p < 0.001 | 0.35 (0.23 to 0.54); p < 0.001 |
| Days with abdominal distention | 0.20 (0.11 to 0.35); p < 0.001 | 0.20 (0.12 to 0.35); p < 0.001 |
| Number of days NPO | 0.35 (0.15 to 0.84); p = 0.019 | 0.27 (0.10 to 0.71); p = 0.008 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sowden, M.; van Weissenbruch, M.M.; Bulabula, A.N.H.; van Wyk, L.; Twisk, J.; van Niekerk, E. Effect of a Multi-Strain Probiotic on the Incidence and Severity of Necrotizing Enterocolitis and Feeding Intolerances in Preterm Neonates. Nutrients 2022, 14, 3305. https://doi.org/10.3390/nu14163305
Sowden M, van Weissenbruch MM, Bulabula ANH, van Wyk L, Twisk J, van Niekerk E. Effect of a Multi-Strain Probiotic on the Incidence and Severity of Necrotizing Enterocolitis and Feeding Intolerances in Preterm Neonates. Nutrients. 2022; 14(16):3305. https://doi.org/10.3390/nu14163305
Chicago/Turabian StyleSowden, Marwyn, Mirjam Maria van Weissenbruch, Andre Nyandwe Hamama Bulabula, Lizelle van Wyk, Jos Twisk, and Evette van Niekerk. 2022. "Effect of a Multi-Strain Probiotic on the Incidence and Severity of Necrotizing Enterocolitis and Feeding Intolerances in Preterm Neonates" Nutrients 14, no. 16: 3305. https://doi.org/10.3390/nu14163305
APA StyleSowden, M., van Weissenbruch, M. M., Bulabula, A. N. H., van Wyk, L., Twisk, J., & van Niekerk, E. (2022). Effect of a Multi-Strain Probiotic on the Incidence and Severity of Necrotizing Enterocolitis and Feeding Intolerances in Preterm Neonates. Nutrients, 14(16), 3305. https://doi.org/10.3390/nu14163305






