Effect of Dietary Fiber (Oat Bran) Supplement in Heart Rate Lowering in Patients with Hypertension: A Randomized DASH-Diet-Controlled Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
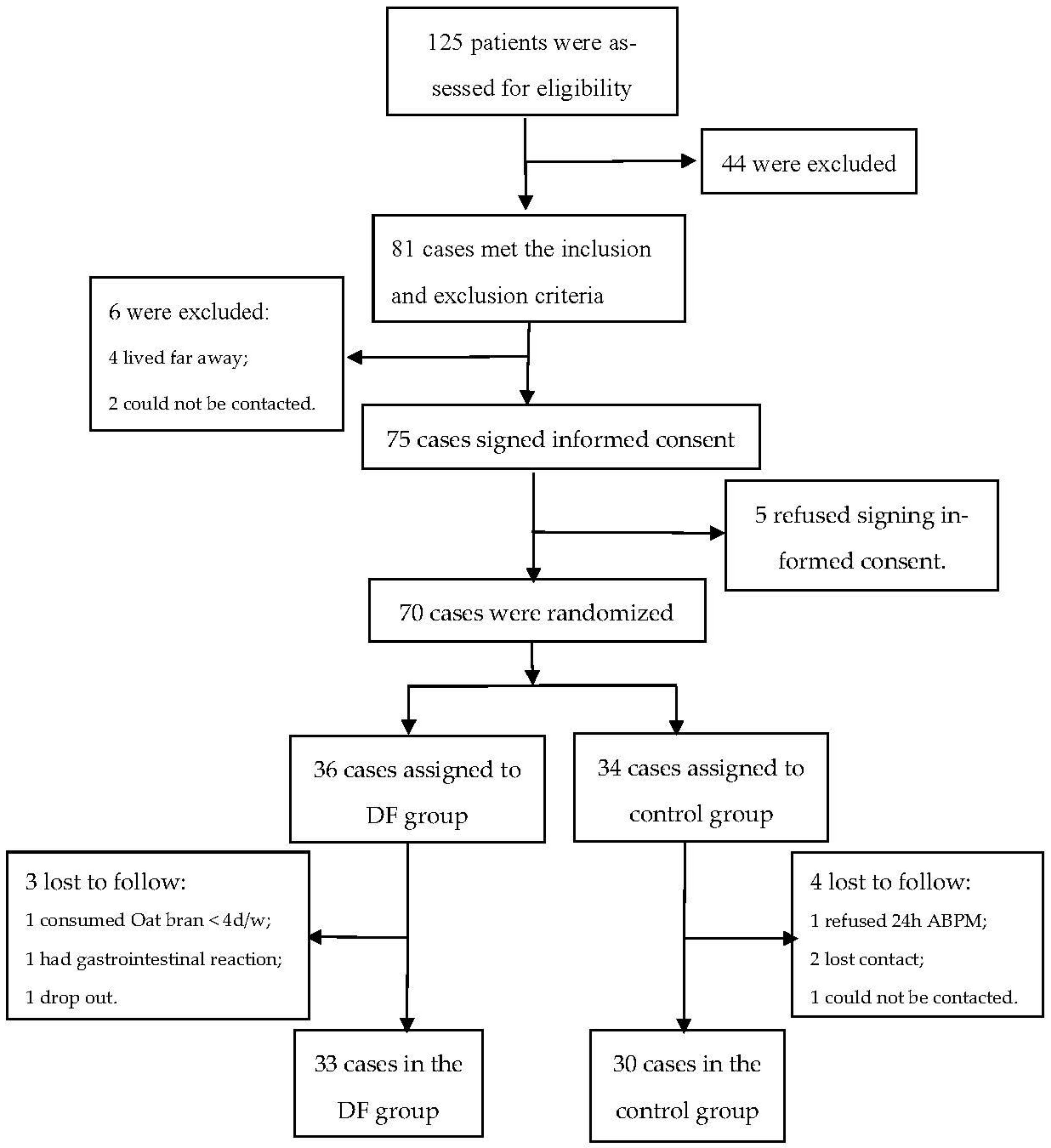
2.1. Study Design and Population
2.2. Subjects
2.3. Sample Size Calculation
2.4. Intervention
2.4.1. Control Group
2.4.2. Intervention Group
2.5. Follow-Up
2.6. Outcomes
2.6.1. Primary Outcomes
2.6.2. Secondary Outcomes
Compliance to Oat Consumption
Side Effect of Dietary Fiber
Change of Medication
2.7. Measurement
2.7.1. Clinical Data and Anthropometric Measurements
2.7.2. Diet and Oat Bran Consumption Record
2.7.3. The 24 h Heart Rate
2.8. Statistical Analysis
- (1)
- Demographic, clinical data, and nutrients: First of all, the Kolmogorov–Smirov test and the Levene’s test were applied to test whether the data of different variables were normally distributed and of equal variance, respectively. The continuous variables being normally distributed were expressed as mean (M) ± standard deviation (SD), and comparisons between groups were made using the independent samples t-test if the variables were normally distributed and of equal variance; otherwise, the data of variables were expressed as median (P25, P75), and their comparisons between groups were made using the Mann–Whitney U test. For categorical variables, the data were described as the frequency (percentage), and comparisons between the two groups was conducted using the Pearson chi-squared test or Fisher’s exact test.
- (2)
- Comparisons of HR between the two groups were conducted at baseline and 3 months: if it was normally distributed, the comparison between two groups was carried out using the independent samples t-test; otherwise, the Mann–Whitney U test was used. For the changes of medication, Pearson’s chi-squared test or Fisher’s exact test was used for comparison. With respect to demographic data, clinical data, and nutrient data, analysis of covariance was applied to correct the relative biases if the comparison of variables between two groups was p < 0.1.
- (3)
- The paired t-test was used to compare the changes of HR within a group before and after intervention.
- (4)
- Intention-to-treat of HR was performed to ensure the reliability of results. A p-value of <0.05 was considered statistically significant.
3. Results
3.1. Baseline Characteristics
3.2. Dietary Fiber Intake
3.3. Effect of Dietary Fiber (Oat Bran) Supplementation on HR in Patients with Hypertension
3.3.1. 24h aveHR, 24hminHR, and 24h maxHR
3.3.2. D-aveHR, D-minHR, and D-maxHR
3.3.3. N-aveHR, N-minHR, and N-maxHR
3.4. Change of Medication
3.5. Side Effect of Dietary Fiber
4. Discussion
4.1. General Data
4.2. Compliance to Dietary Fiber Intake
4.3. Dietary Fiber on Heart Rate
4.3.1. Dietary Fiber on 24 h Heart Rates
4.3.2. Dietary Fiber on Daytime and Nighttime Heart Rates
4.4. Dietary Fiber on Medication Change
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ferrer, E. 2016-European Society of Hypertension Guidelines for the management of high blood pressure in children and adolescents. An. Pediat. 2016, 85, 167–169. [Google Scholar]
- Wang, Z.; Chen, Z.; Zhang, L.; Wang, X.; Hao, G.; Zhang, Z.; Shao, L.; Tian, Y.; Dong, Y.; Zheng, C. Status of Hypertension in China: Results from the China Hypertension Survey, 2012–2015. Circulation 2018, 137, 2344–2356. [Google Scholar] [CrossRef]
- Kazmi, I.; Al-Maliki, W.H.; Ali, H.; Al-Abbasi, F.A. Biochemical interaction of salt sensitivity: A key player for the development of essential hypertension. Mol. Cell. Biochem. 2020, 476, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Kouvas, N.; Tsioufis, C.; Vogiatzakis, N.; Sanidas, E.; Konstantinidis, D.; Kintis, K.; Dimitriadis, K.; Kakosaiou, Z.; Tsioufis, P.; Kouremeti, M. Heart Rate and Blood Pressure: “Connecting the Dots” in Epidemiology and Pathophysiology. Angiology 2017, 69, 660–665. [Google Scholar] [CrossRef]
- Sun, N.L.; Huo, Y.; Huang, J. The current status of heart rate in Chinese hypertensive patients. Chin. J. Hypertens. 2015, 23, 934–939. [Google Scholar]
- Salles, G.F.; Cardoso, C.R.L.; Fonseca, L.L. Prognostic Significance of Baseline Heart Rate and Its Interaction with Beta-Blocker Use in Resistant Hypertension: A Cohort Study. Am. J. Hypertens. 2012, 26, 218–226. [Google Scholar] [CrossRef] [Green Version]
- Saxena, A.; Minton, D.; Lee, D.C.; Sui, X.; Fayad, R.; Lavie, C.J.; Blair, S.N. Protective Role of Resting Heart Rate on All-Cause and Cardiovascular Disease Mortality. Mayo Clin. Proc. 2013, 88, 1420–1426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Management, M.C.G.O. Multidisciplinary expert consensus on heart rate management in Hypertensive Patients in China (2021). Zhonghua Xinxueguanbing Zazhi 2021, 24, 8. [Google Scholar]
- Williams, B.; Mancia, G.; Spiering, W.; Rosei, E.A.; Azizi, M.; Burnier, M.; Clement, D.; Coca, A.; Simone, G.D.; Dominiczak, A. 2018 ESC/ESH Guidelines for the management of arterial hypertension. The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). J. Hypertens. 2018, 36, 1953–2041. [Google Scholar] [CrossRef] [Green Version]
- Zhao, M.X.; Zhao, Q.; Zheng, M.; Liu, T.; Wu, S. Effect of resting heart rate on the risk of all-cause death in Chinese patients with hypertension: Analysis of the Kailuan follow-up study. BMJ Open 2020, 10, e32699. [Google Scholar] [CrossRef] [Green Version]
- Tomlinson, B.; Dalal, J.J.; Huang, J.; Low, L.P.; Park, C.G.; Rahman, A.R.; Reyes, E.B.; Soenarta, A.A.; Heagerty, A.; Follath, F. The role of ββ-blockers in the management of hypertension: An Asian perspective. Curr. Med. Res. Opin. 2011, 27, 1021–1033. [Google Scholar] [CrossRef] [PubMed]
- Wahlstrom, M.; Karlsson, M.R.; Medin, J.; Frykman, V. Effects of yoga in patients with paroxysmal atrial fibrillation-a randomized controlled study. Eur. J. Cardiovasc. Nurs. 2017, 16, 57–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zou, Y.; Zhao, X.; Hou, Y.Y.; Liu, T.; Wu, Q.; Huang, Y.H.; Wang, X.H. Meta-Analysis of Effects of Voluntary Slow Breathing Exercises for Control of Heart Rate and Blood Pressure in Patients with Cardiovascular Diseases. Am. J. Cardiol. 2017, 120, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.; Veronesi, M.; Fogacci, F. Dietary Intervention to Improve Blood Pressure Control: Beyond Salt Restriction. High Blood Press Cardiovasc. Prev. 2021, 28, 547–553. [Google Scholar] [CrossRef]
- Hadi, A.; Askarpour, M.; Miraghajani, M.; Symonds, M.E.; Sheikhi, A.; Ghaedi, E. Effects of strawberry supplementation on cardiovascular risk factors: A comprehensive systematic review and meta-analysis of randomized controlled trials. Food Funct. 2019, 10, 6987–6998. [Google Scholar] [CrossRef]
- Ma, A.; Ega, B.; Nr, C.; Ahd, E.; Hm, F.; Mes, G.; Mmhg, I. Policosanol supplementation significantly improves blood pressure among adults: A systematic review and meta-analysis of randomized controlled trials. Complement. Ther. Med. 2019, 45, 89–97. [Google Scholar]
- Foundation, H.A.S. The DASH Diet to Lower High Blood Pressure. Available online: https://medlineplus.gov/ency/patientinstructions/000770.htm (accessed on 9 July 2022).
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Makki, K.; Deehan, E.C.; Walter, J.; Bckhed, F. The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Muniz, F.J. Dietary fiber and cardiovascular health. In Nutricion Hospitalaria: Organo Oficial de la Sociedad Espanola de Nutricion Parenteral y Enteral; Un Encuentro en la Real Academia Nacional de Farmacia: Madrid, Spain, 2012; Volume 27, pp. 40–54. [Google Scholar]
- Ho, H.V.T.; Sievenpiper, J.L.; Zurbau, A.; Blanco Mejia, S.; Jovanovski, E.; Au-Yeung, F.; Jenkins, A.L.; Vuksan, V. The effect of oat β-glucan on LDL-cholesterol, non-HDL-cholesterol and apoB for CVD risk reduction: A systematic review and meta-analysis of randomised-controlled trials. Brit. J. Nutr. 2016, 116, 1369–1382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whelton, S.P.; Hyre, A.D.; Pedersen, B.; Yi, Y.; Whelton, P.K.; He, J. Effect of dietary fiber intake on blood pressure: A meta-analysis of randomized, controlled clinical trials. J. Hypertens. 2005, 23, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Cui, L.; Qi, J.; Ojo, O.; Wang, X. The Effect of Dietary Fiber (Oat bran) Supplement on Blood Pressure in Patients with Essential Hypertension: A randomized controlled trial. Nutr. Metab. Cardiovas. 2021, 31, 2458–2470. [Google Scholar] [CrossRef]
- Catapano, G.; Pedone, C.; Nunziata, E.; Zizzo, A.; Incalzi, R.A. Nutrient intake and serum cytokine pattern in elderly people with heart failure. Eur. J. Heart Fail. 2014, 10, 428–434. [Google Scholar] [CrossRef]
- Liu, L.S.; Wu, Z.S.; Wang, J.G.; Wang, W.; Bao, Y.J.; Cai, J.; Chen, L.Y.; Chen, W.W.; Chu, S.L.; Feng, Y.Q.; et al. 2018 Chinese Guidelines for Prevention and Treatment of Hypertension—A report of the Revision Committee of Chinese Guidelines for Prevention and Treatment of Hypertension. J. Geriatr. Cardiol. 2019, 16, 182–241. [Google Scholar]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Wright, J.T. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 2017, 19, 213–221. [Google Scholar]
- Strilchuk, L.; Cincione, R.I.; Fogacci, F.; Cicero, A. Dietary interventions in blood pressure lowering: Current evidence in 2020. Kardiol. Pol. 2020, 78, 659–666. [Google Scholar] [CrossRef]
- Wang, F.; Luo, X.; Zhang, J.; Chen, F. Visual Analogue Scale in the diagnosis of Influenza. Chin. Gen. Pract. 2019, 22, 2472–2475. [Google Scholar]
- Drossman, D.A. The functional gastrointestinal disorders and the Rome III process. Gastroenterology 2006, 130, 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Sharman, J.E.; Andre, L.G.; Coombes, J.S. Exercise and Cardiovascular Risk in Patients with Hypertension. Am. J. Hypertens. 2015, 28, 147–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oparil, S.; Acelajado, M.C.; Bakris, G.L.; Berlowitz, D.R.; Cífková, R.; Dominiczak, A.F.; Grassi, G.; Jordan, J.; Poulter, N.R.; Rodgers, A. Hypertension. Nat. Rev. Dis. Primers 2018, 4, 18014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrova, M.M.; Moskaleva, P.V.; Shnayder, N.A.; Nasyrova, R.F. Comorbidity of arterial hypertension and tension-type headache. Kardiologiia 2020, 60, 132–140. [Google Scholar] [CrossRef]
- Measurement, C.W.G.O. Guidelines for blood pressure measurement in China. Chin. J. Hypertens. 2011, 19, 1101–1115. [Google Scholar]
- Yang, Y. 24h ambulatory blood pressure monitoring and its clinical application. Chin. J. Integr. Med. Cardio-/Cereb. Ascular Dis. 2008, 6, 246–247. [Google Scholar]
- Palatini, P.; Rosei, E.A.; Casiglia, E.; Chalmers, J.; Ferrari, R.; Grassi, G.; Inoue, T.; Jelakovic, B.; Jensen, M.T.; Julius, S.; et al. Management of the hypertensive patient with elevated heart rate: Statement of the Second Consensus Conference endorsed by the European Society of Hypertension. J. Hypertens. 2016, 34, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, N.; Kasuga, C.; Tanaka, A.; Kamachi, K.; Ai, M.; Urayama, K.Y.; Tanaka, A. Association between dietary fibre: Carbohydrate intake ratio and insulin resistance in Japanese adults without type 2 diabetes. Br. J. Nutr. 2018, 119, 620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.; Je, Y. Dietary fibre intake and mortality from cardiovascular disease and all cancers: A meta-analysis of prospective cohort studies. Arch. Cardiovasc. Dis. 2016, 109, 39–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rong, Y.M.; Cheng, N.; He, C.L. The relationship between body mass index, waist circumference and hypertension. Chin. J. Hypertens. 2018, 4, 379–383. [Google Scholar]
- Wu, J.; Li, T.; Song, X.; Sun, W.; Zhang, Y.; Liu, Y.; Li, L.; Yu, Y.; Liu, Y.; Qi, C. Prevalence and distribution of hypertension and related risk factors in Jilin Province, China 2015: A cross-sectional study. BMJ Open 2018, 8, e20126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, F.J.; Xie, M.K.; Yan, M.X.; Yang, Q.F. Status of self-management in young and middle-aged patients with chronic heart failure and its influencing factors. Nurs. J. Chin. PLA 2021, 6, 5–8. [Google Scholar]
- Liu, Y.; Ju, Y.; Cui, L.; Liu, T.; Hou, Y.; Wu, Q.; Ojo, O.; Du, X.; Wang, X. Association between Dietary Fiber Intake and Incidence of Depression and Anxiety in Patients with Essential Hypertension. Nutrients 2021, 13, 4159. [Google Scholar] [CrossRef]
- Qi, J.D.; Xue, Y.; Du, X.J.; Cui, L.L.; Jiang, T.B.; Ying, T.T.; Wang, X.H. Dietary fiber intake levels and its relationship with blood pressure in patients with essential hypertension in Suzhou. Mod. Prev. Med. 2021, 48, 636–639. [Google Scholar]
- Okin, P.M.; Kjeldsen, S.E.; Julius, S.; Hille, D.A.; Dahl, F.B.; Devereux, R.B. Effect of Changing Heart Rate during Treatment of Hypertension on Incidence of Heart Failure. Am. J. Cardiol. 2012, 109, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.F. Investigation on the Relationship between the Average Heart Rate within 24-Hour after Admission and Cardiovascular Events in Patients with Acute Myocardial Infarction; Zhejiang Chinese Medicine University: Hangzhou, China, 2016. [Google Scholar]
- Kolloch, R.; Legler, U.F.; Champion, A.; Cooper-Dehoff, R.M.; Handberg, E.; Zhou, Q.; Pepine, C.J. Impact of resting heart rate on outcomes in hypertensive patients with coronary artery disease: Findings from the INternational VErapamil-SR/trandolapril STudy (INVEST). Eur. Heart J. 2008, 29, 1327–1334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lonn, E.M.; Rambihar, S.; Gao, P.; Custodis, F.F.; Sliwa, K.; Teo, K.K.; Yusuf, S.; Bohm, M. Heart rate is associated with increased risk of major cardiovascular events, cardiovascular and all-cause death in patients with stable chronic cardiovascular disease: An analysis of ONTARGET/TRANSCEND. Clin. Res. Cardiol. 2014, 103, 149–159. [Google Scholar] [CrossRef]
- Onyszkiewicz, M.; Gawry-Kopczyńska, M.; Konopelski, P.; Aleksandrowicz, M.; Ufnal, M. Butyric acid, a gut bacteria metabolite, lowers arterial blood pressure via colon-vagus nerve signaling and GPR41/43 receptors. Pflügers Arch. Eur. J. Physiol. 2019, 471, 1441–1453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomasz, H.; Marek, K.; Maksymilian, O.; Piotr, P.; Agnieszka, S.; Jadwiga, T.; Marcin, U. Colonic indole, gut bacteria metabolite of tryptophan, increases portal blood pressure in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R646–R655. [Google Scholar]
- Natarajan, N.; Hori, D.; Flavahan, S.; Steppan, J.; Flavahan, N.A.; Berkowitz, D.E.; Pluznick, J.L. Microbial short chain fatty acid metabolites lower blood pressure via endothelial G-protein coupled receptor 41. Physiol. Genom. 2016, 48, 826–834. [Google Scholar] [CrossRef]
- Miyamoto, J.; Kasubuchi, M.; Nakajima, A.; Irie, J.; Itoh, H.; Kimura, I. The role of short-chain fatty acid on blood pressure regulation. Curr. Opin. Nephrol. Hypertens. 2016, 25, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Schilderink, R.; Verseijden, C.; Seppen, J.; Muncan, V.; Van, D.; Lambers, T.T.; Tol, E.V.; Jonge, W.D. The SCFA butyrate stimulates the epithelial production of retinoic acid via inhibition of epithelial HDAC. AJP Gastrointest. Liver Physiol. 2016, 310, G1138–G1146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, L.H. Evaluation of Heart Rate in Patients with Heart Failure Bu Short-Term Ambulatory Electrocardiography. Master’s Thesis, Dali University, Dali, China, 2021. [Google Scholar]
- Bishehsari, F.; Voigt, R.M.; Keshavarzian, A. Circadian rhythms and the gut microbiota: From the metabolic syndrome to cancer. Nat. Rev. Endocrinol. 2020, 16, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Hayter, E.A.; Wehrens, S.; Van Dongen, H.; Stangherlin, A.; Gaddameedhi, S.; Crooks, E.; Barron, N.J.; Venetucci, L.A.; O’Neill, J.S.; Brown, T.M.; et al. Distinct circadian mechanisms govern cardiac rhythms and susceptibility to arrhythmia. Nat. Commun. 2021, 12, 2472. [Google Scholar] [CrossRef]
| Characteristic | DF Group (n = 33) | Control Group (n = 30) | t/χ2/z | p | |
|---|---|---|---|---|---|
| x ± s/n(%)/ M(P25, P75) | x ± s/n(%)/ M(P25, P75) | ||||
| Ages (years) | 40.09 ± 9.32 | 40.80 ± 10.94 | 0.736 a | 0.465 | |
| Sex | Male | 26 (78.8) | 16 (53.3) | 4.582 b | 0.032 |
| Female | 7 (21.2) | 14 (46.7) | |||
| Marital status | Unmarried | 11 (33.3) | 5 (16.7) | 2.304 b | 0.129 |
| Married | 22 (66.7) | 25 (83.3) | |||
| Education | Primary school | 1 (3.0) | 0 (0.0) | 1.443 b | 0.695 |
| Junior middle school | 4 (12.1) | 5 (16.7) | |||
| High school/SSS | 5 (15.2) | 6 (20.0) | |||
| College or higher | 23 (66.7) | 19 (63.3) | |||
| Professional situation | On the job | 31 (94.0) | 25 (83.3) | 1.838 b | 0.399 |
| Retired | 1 (3.0) | 3 (10.0) | |||
| Job-waiting | 1 (3.0) | 2 (6.7) | |||
| Medical payment | Self-funded | 3 (9.0) | 1 (3.3) | 3.938 c | 0.140 |
| NRCMI | 3 (9.0) | 0(0.0) | |||
| Medical insurance | 27 (81.8) | 29 (96.7) | |||
| Exercise | Yes | 11 (33.3) | 12 (40.0) | 0.301 b | 0.583 |
| No | 22 (66.7) | 18 (60.0) | |||
| DOS(hours/day) | 6.48 (6.00, 7.63) | 6.90 (6.00, 7.50) | −0.561 d | 0.743 | |
| Sleep quality | Poor | 5 (15.2) | 0(0.0) | 5.491 b | 0.064 |
| Fair | 17 (51.5) | 21 (70.0) | |||
| Good | 11 (33.3) | 9 (30.0) | |||
| Smoke | Yes | 8 (24.2) | 7 (23.3) | 0.007 b | 0.933 |
| No | 25 (75.8) | 23 (76.7) | |||
| Alcohol intake | Yes | 9 (27.3) | 9 (30.0) | 0.057 b | 0.811 |
| No | 24 (72.7) | 21 (70.0) | |||
| BMI (kg/m2) | 24.93 ± 3.12 | 25.38 ± 4.83 | 0.436 a | 0.664 | |
| WHR | 0.92 ± 0.18 | 0.96 ± 0.37 | 0.561 d | 0.743 | |
| Constipation | Yes | 2 (6.1) | 3 (10.0) | 0.334 c | 0.563 |
| No | 31 (93.9) | 27 (90.0) | |||
| DOH (years) | 1.98 ± 3.70 | 1.67 ± 3.17 | −0.182 a | 0.856 | |
| Anti-hypertensive drugs | Yes | 6 (18.2) | 10 (33.3) | 1.904 c | 0.168 |
| No | 27 (81.8) | 20 (66.7) | |||
| Comorbidity | Yes | 2 (6.1) | 5 (16.7) | 1.790 c | 0.181 |
| No | 31 (93.9) | 25 (83.3) | |||
| HR-lowing drugs | Yes | 1 | 3 | 1.284 c | 0.257 |
| No | 32 | 27 | |||
| SBP (mmHg) | 131.30 ± 4.80 | 137.13 ± 8.09 | 3.514 a | 0.001 * | |
| DBP (mmHg) | 84.58 ± 4.72 | 85.40 ± 5.26 | 0.655 a | 0.515 | |
| Complication | Yes | 0 (0.0) | 1 (3.3) | 1.118 c | 0.290 |
| No | 33 (100.0) | 29 (96.7) | |||
| Characteristic | DF Group (n = 33) | Control Group (n = 30) | t | p |
|---|---|---|---|---|
| x ± s/n(%) | x ± s/n(%) | |||
| Total calories (kcal/day) | 1660.55 ± 386.66 | 1607.41 ± 381.21 | −0.543 | 0.589 |
| Carbohydrate (g/day) | 210.66 ± 66.34 | 202.17 ± 62.18 | −0.517 | 0.607 |
| Protein (g/day) | 79.17 ± 22.97 | 77.38 ± 21.74 | −0.315 | 0.754 |
| Fat (g/day) | 57.66 ± 22.83 | 54.66 ± 21.49 | −0.530 | 0.598 |
| Cholesterol (mg/day) | 534.67 ± 290.31 | 510.48 ± 291.59 | −0.327 | 0.745 |
| Calcium (mg/day) | 563.45 ± 172.85 | 577.57 ± 261.02 | 0.254 | 0.800 |
| Potassium (mg/day) | 1971.68 ± 467.21 | 1993.56 ± 663.66 | 0.153 | 0.879 |
| Sodium (mg/day) | 1282.50 ± 800.98 | 1391.69 ± 878.15 | 0.514 | 0.609 |
| Vitamin D (μg/day) | 3.10 ± 3.74 | 4.20 ± 5.20 | 0.962 | 0.340 |
| Folic acid (μg/day) | 118.32 ± 61.78 | 124.0 ± 96.51 | 0.284 | 0.777 |
| Vitamin B6 (μ/day) | 0.36 ± 0.37 | 0.24 ± 0.18 | −1.567 | 0.122 |
| Magnesium (mg/day) | 296.65 ± 77.49 | 310.45 ± 115.51 | 0.558 | 0.579 |
| Zinc (mg/day) | 10.18 ± 2.59 | 11.47 ± 4.51 | 1.402 | 0.166 |
| Selenium (μg/day) | 52.47 ± 17.47 | 55.51 ± 27.11 | 0.215 | 0.830 |
| DF (g/d) | DF Group (n = 33) | Control Group (n = 30) | t | p |
|---|---|---|---|---|
| x ± s/n(%) | x ± s/n(%) | |||
| Baseline | 11.07 ± 3.73 | 10.66 ± 4.27 | −0.388 | 0.700 |
| 3 month | 21.45 ± 3.86 | 10.40 ± 3.76 | −10.75 | <0.001 * |
| HR | Time | DF Group (n = 33) ± s | Control Group (n = 30) ± s | t/F | p |
|---|---|---|---|---|---|
| 24h aveHR | Baseline | 75.15 ± 9.69 | 73.73 ± 8.21 | −0.624 | 0.535 |
| 3 month | 71.03 ± 9.91 | 73.00 ± 7.62 | 0.734 | 0.466 | |
| t | 2.401 | 0.629 | |||
| p | 0.022 * | 0.534 | |||
| 3 m, adjusted | 70.74 ± 1.59 | 72.70 ± 1.80 | 0.924 | 0.341 | |
| MD | 4.12 ± 9.86 | 0.73 ± 6.39 | −1.600 | 0.115 | |
| 24hminHR | Baseline | 54.15 ± 6.51 | 55.33 ± 7.55 | 0.667 | 0.507 |
| 3 month | 53.88 ± 7.65 | 54.63 ± 5.39 | 0.336 | 0.738 | |
| t | 0.284 | 0.639 | |||
| p | 0.778 | 0.528 | |||
| 3 month, adjusted | 53.81 ± 1.20 | 54.47 ± 1.31 | 0.197 | 0.684 | |
| MD | 0.27 ± 5.51 | 0.700 ± 6.00 | 0.295 | 0.769 | |
| 24h maxHR | Baseline | 103.03 ± 18.43 | 99.67 ± 13.74 | −0.822 | 0.414 |
| 3 month | 93.24 ± 15.26 | 98.17 ± 16.99 | 1.099 | 0.276 | |
| t | 2.966 | 0.638 | |||
| p | 0.006 * | 0.529 | |||
| 3 month, adjusted | 92.96 ± 2.93 | 96.80 ± 3.27 | 1.062 | 0.308 | |
| MD | 9.79 ± 18.96 | 1.50 ± 12.88 | −2.009 | 0.049 * | |
| D-aveHR | Baseline | 78.30 ± 10.53 | 76.43 ± 8.30 | −0.777 | 0.440 |
| 3 month | 73.30 ± 10.13 | 74.97 ± 8.08 | 0.637 | 0.527 | |
| t | 2.956 | 1.239 | |||
| p | 0.006 * | 0.225 | |||
| 3 month, adjusted | 73.17 ± 1.66 | 74.54 ± 1.93 | 0.447 | 0.507 | |
| MD | 5.00 ± 9.71 | 1.46 ± 6.48 | −1.681 | 0.098 | |
| D-minHR | Baseline | 60.61 ± 9.05 | 60.17 ± 8.01 | −0.203 | 0.840 |
| 3 month | 56.97 ± 8.01 | 57.97 ± 7.37 | 0.490 | 0.626 | |
| t | 2.048 | 1.760 | |||
| p | 0.049 * | 0.089 | |||
| 3 month, adjusted | 56.97 ± 1.45 | 57.82 ± 1.49 | 0.159 | 0.692 | |
| MD | 3.64 ± 10.20 | 2.14 ± 6.95 | −0.649 | 0.518 | |
| D-maxHR | Baseline | 101.76 ± 20.73 | 98.83 ± 12.31 | −0.672 | 0.504 |
| 3 month | 92.79 ± 15.26 | 98.17 ± 16.39 | 1.213 | 0.230 | |
| t | 2.278 | 0.264 | |||
| p | 0.030 * | 0.794 | |||
| 3 month, adjusted | 92.60 ± 2.96 | 96.72 ± 3.31 | 1.169 | 0.285 | |
| MD | 8.97 ± 22.63 | 0.67 ± 13.86 | −1.735 | 0.088 | |
| N-aveHR | Baseline | 61.64 ± 7.45 | 63.53 ± 8.90 | 0.920 | 0.361 |
| 3 month | 62.91 ± 9.48 | 63.20 ± 6.14 | 0.219 | 0.828 | |
| t | −0.868 | 0.241 | |||
| p | 0.392 | −0.668 | |||
| 3 month, adjusted | 62.87 ± 1.37 | 63.11 ± 1.54 | 0.032 | 0.859 | |
| MD | −1.27 ± 8.43 | 0.33 ± 7.57 | 0.793 | 0.431 | |
| N-minHR | Baseline | 53.94 ± 6.71 | 56. 83 ± 9.20 | 1.436 | 0.156 |
| 3 month | 54.88 ± 7.96 | 54.80 ± 6.05 | −0.024 | 0.981 | |
| t | −0.866 | 1.698 | |||
| p | 0.392 | 0.100 | |||
| 3 month, adjusted | 54.54 ± 1.25 | 55.06 ± 1.40 | 0.072 | 0.790 | |
| MD | −0.94 ± 6.23 | 2.03 ± 6.56 | 1.844 | 0.070 | |
| N-maxHR | Baseline | 73.58 ± 12.20 | 74.17 ± 11.91 | 0.194 | 0.847 |
| 3 month | 74.97 ± 14.88 | 73.97 ± 10.14 | −0.257 | 0.798 | |
| t | −0.493 | 0.097 | |||
| p | 0.625 | 0.923 | |||
| 3 month, adjusted | 75.08 ± 2.26 | 73.83 ± 2.52 | 0.108 | 0.744 | |
| MD | −1.39 ± 16.23 | 0.200 ± 11.26 | 0.449 | 0.655 |
| HR | Time | DF Group (n = 36) ± s | Control Group (n = 34) ± s | t | p |
|---|---|---|---|---|---|
| 24h aveHR | Baseline | 74.97 ± 9.37 | 74.18 ± 7.99 | −0.381 | 0.704 |
| 3 month | 71.19 ± 9.58 | 73.53 ± 7.49 | 0.1.31 | 0.262 | |
| t | 2.386 | 0.629 | |||
| p | 0.023 * | 0.533 | |||
| MD | 3.77 ± 9.50 | 0.65 ± 5.99 | −1.638 | 0.106 | |
| 24hminHR | Baseline | 54.36 ± 6.47 | 55.47 ± 7.12 | 0.683 | 0.497 |
| 3 month | 54.11 ± 7.53 | 54.85 ± 5.14 | 0.479 | 0.634 | |
| t | 0.285 | 0.640 | |||
| p | 0.778 | 0.527 | |||
| MD | 0.25 ± 5.27 | 0.62 ± 5.63 | 0.282 | 0.779 | |
| 24h maxHR | Baseline | 102.64 ± 17.67 | 99.91 ± 13.06 | −0.731 | 0.467 |
| 3 month | 93.67 ± 14.67 | 98.59 ± 16.36 | 1.326 | 0.189 | |
| t | 2.936 | 0.639 | |||
| p | 0.006 * | 0.527 | |||
| MD | 8.97 ± 18.34 | 1.32 ± 16.08 | −2.048 | 0.044 * | |
| D-aveHR | Baseline | 78.08 ± 10.16 | 76.85 ± 8.08 | −0.559 | 0.578 |
| 3 month | 73.50 ± 9.77 | 75.56 ± 7.97 | 0.963 | 0.339 | |
| t | 2.929 | 1.237 | |||
| p | 0.006 * | 0.225 | |||
| MD | 4.58 ± 9.38 | 1.29 ± 6.10 | −1.717 | 0.089 | |
| D-minHR | Baseline | 60.61 ± 8.79 | 60.26 ± 7.63 | −0.176 | 0.861 |
| 3 month | 57.28 ± 7.89 | 58.32 ± 7.09 | 0.582 | 0.562 | |
| t | 2.039 | 1.753 | |||
| p | 0.049 * | 0.089 | |||
| MD | 3.33 ± 9.81 | 1.82 ± 6.45 | −0.697 | 0.488 | |
| D-maxHR | Baseline | 101.47 ± 20.73 | 99.18 ± 12.12 | −0.580 | 0.564 |
| 3 month | 93.25 ± 14.68 | 98.59 ± 16.36 | 1.438 | 0.155 | |
| t | 2.265 | 0.264 | |||
| p | 0.030 * | 0.793 | |||
| MD | 8.22 ± 21.78 | 0.59 ± 12.99 | −1.768 | 0.082 | |
| N-aveHR | Baseline | 61.75 ± 7.38 | 64.15 ± 8.95 | 1.226 | 0.225 |
| 3 month | 62.92 ± 9.26 | 63.85 ± 6.63 | 0.484 | 0.630 | |
| t | −0.868 | 0.242 | |||
| p | 0.391 | 0.810 | |||
| MD | −1.16 ± 8.07 | 0.29 ± 7.09 | 0.803 | 0.425 | |
| N-minHR | Baseline | 54.17± 6.66 | 58.09 ± 11.61 | 1.745 | 0.086 |
| 3 month | 55.03 ± 7.80 | 56.29 ± 9.90 | 0.596 | 0.553 | |
| t | −0.866 | 1.639 | |||
| p | 0.392 | 0.100 | |||
| MD | −0.86 ± 5.97 | 1.79 ± 6.18 | 1828 | 0.072 | |
| N-maxHR | Baseline | 73.44 ± 11.94 | 73.74 ± 11.69 | 0.103 | 0.918 |
| 3 month | 74.72 ± 14.47 | 73.56 ± 10.12 | −0.388 | 0.700 | |
| t | −0.494 | 0.098 | |||
| p | 0.624 | 0.923 | |||
| MD | −1.28 ± 15.52 | −0.38 ± 10.02 | 0.456 | 0.650 |
| Changes of Medication Dosage | DF Group (n = 33) | Control Group (n = 30) | χ2 | p |
|---|---|---|---|---|
| Reduced | 4 (12.1) | 2 (6.7) | 5.926 | 0.115 |
| No change | 3 (9.1) | 7 (23.3) | ||
| Increased | 3 (9.1) | 5 (16.7) | ||
| Not taking medicine | 23 (69.7) | 16 (53.3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ju, Y.; Zhang, C.; Zhang, Z.; Zhu, H.; Liu, Y.; Liu, T.; Ojo, O.; Qiu, J.; Wang, X. Effect of Dietary Fiber (Oat Bran) Supplement in Heart Rate Lowering in Patients with Hypertension: A Randomized DASH-Diet-Controlled Clinical Trial. Nutrients 2022, 14, 3148. https://doi.org/10.3390/nu14153148
Ju Y, Zhang C, Zhang Z, Zhu H, Liu Y, Liu T, Ojo O, Qiu J, Wang X. Effect of Dietary Fiber (Oat Bran) Supplement in Heart Rate Lowering in Patients with Hypertension: A Randomized DASH-Diet-Controlled Clinical Trial. Nutrients. 2022; 14(15):3148. https://doi.org/10.3390/nu14153148
Chicago/Turabian StyleJu, Yang, Chenglin Zhang, Zhirong Zhang, Hongying Zhu, Yuanyuan Liu, Ting Liu, Omorogieva Ojo, Jingbo Qiu, and Xiaohua Wang. 2022. "Effect of Dietary Fiber (Oat Bran) Supplement in Heart Rate Lowering in Patients with Hypertension: A Randomized DASH-Diet-Controlled Clinical Trial" Nutrients 14, no. 15: 3148. https://doi.org/10.3390/nu14153148
APA StyleJu, Y., Zhang, C., Zhang, Z., Zhu, H., Liu, Y., Liu, T., Ojo, O., Qiu, J., & Wang, X. (2022). Effect of Dietary Fiber (Oat Bran) Supplement in Heart Rate Lowering in Patients with Hypertension: A Randomized DASH-Diet-Controlled Clinical Trial. Nutrients, 14(15), 3148. https://doi.org/10.3390/nu14153148








