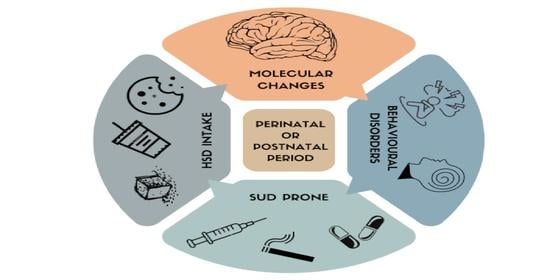
A High-Sugar Diet Consumption, Metabolism and Health Impacts with a Focus on the Development of Substance Use Disorder: A Narrative Review
Abstract
1. Introduction
1.1. Daily Limits for Carbohydrate Intake
1.2. Added Sugar Consumption
1.3. Carbohydrate ADME Processes
2. Hormonal and Neuronal Regulation of SUGAR Intake
2.1. Pancreatic Hormones Control Blood Glucose Homeostasis
2.2. Sweetness Perception and Regulation
2.3. Energy-Balanced Peptide Hormones
2.4. Stress-Induced Bingeing “Comfort Food”
2.5. Changes in the Reward Brain System Following HSD
2.6. Behavioural Consequences of HSD
3. Maternal HSD Overabundance as Offspring Disease Factor
4. HSD as a Risk Factor to Develop SUD and Food Addiction
5. HSD Evokes Behavioural Predictors of Drug of Abuse
6. Conclusions and Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Asif, M.; Akram, M.; Profile, S.; Saeed, T.; Naveed, A.; Asif, H.M.; Khan, M.I.; Akhtar, N.; Ur Rehman, R.; Shah, S.M.A.; et al. Carbohydrates. Int. Res. J. Biochem. Bioinform. 2011, 1, 1–5. [Google Scholar]
- Lee, C.K. The Chemistry and Biochemistry of the Sweetness of Sugars. Adv. Carbohydr. Chem. Biochem. 1987, 45, 199–351. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Stephen, A.M. Carbohydrate Terminology and Classification. Eur. J. Clin. Nutr. 2007, 61, S5–S18. [Google Scholar] [CrossRef]
- Hannou, S.A.; Haslam, D.E.; McKeown, N.M.; Herman, M.A. Fructose Metabolism and Metabolic Disease. J. Clin. Investig. 2018, 128, 545. [Google Scholar] [CrossRef] [PubMed]
- Rippe, J.M.; Angelopoulos, T.J. Relationship between Added Sugars Consumption and Chronic Disease Risk Factors: Current Understanding. Nutrients 2016, 8, 697. [Google Scholar] [CrossRef]
- Slavin, J.; Carlson, J. Carbohydrates. Adv. Nutr. 2014, 5, 760. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services; U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th Ed.; December 2015. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 13 June 2021).
- Dietary Guidelines Advisory Committee. Scientif ic Report of the 2020 Dietary Guidelines Advisory Committee: Advisory Report to the Secretary of Agriculture and the Secretary of Health Human Services; U.S. Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2020. [Google Scholar]
- Toop, C.R.; Gentili, S. Fructose Beverage Consumption Induces a Metabolic Syndrome Phenotype in the Rat: A Systematic Review and Meta-Analysis. Nutrients 2016, 8, 577. [Google Scholar] [CrossRef]
- National Research Council (US) Subcommittee on Laboratory Animal Nutrition. Nutrient Requirements of Laboratory Animals, 4th ed.; National Academies Press: Washington, DC, USA, 1995. [Google Scholar] [CrossRef]
- Ricci, M. Laboratory Animal Control Diets: Very Important, Often Neglected. Lab Anim. 2015, 44, 240–241. [Google Scholar] [CrossRef]
- Bruce, H.M.; Parkes, A.S. Feeding and Breeding of Laboratory Animals: IX. A Complete Cubed Diet for Mice and Rats. J. Hyg. 1949, 47, 202. [Google Scholar] [CrossRef]
- Tuck, C.J.; de Palma, G.; Takami, K.; Brant, B.; Caminero, A.; Reed, D.E.; Muir, J.G.; Gibson, P.R.; Winterborn, A.; Verdu, E.F.; et al. Nutritional Profile of Rodent Diets Impacts Experimental Reproducibility in Microbiome Preclinical Research. Sci. Rep. 2020, 10, 17784. [Google Scholar] [CrossRef]
- Welsh, J.A.; Sharma, A.J.; Grellinger, L.; Vos, M.B. Consumption of Added Sugars Is Decreasing in the United States. Am. J. Clin. Nutr. 2011, 94, 726–734. [Google Scholar] [CrossRef] [PubMed]
- McGuire, S.; Ervin, R.B.; Kit, B.K.; Carroll, M.D.; Ogden, C.L. Consumption of Added Sugar among U.S. Children and Adolescents, 2005–2008. NCHS Data Brief No 87. Hyattsville, MD: National Center for Health Statistics. Adv. Nutr. 2012, 3, 534. [Google Scholar] [CrossRef]
- Herrick, K.A.; Fryar, C.D.; Hamner, H.C.; Park, S.; Ogden, C.L. Added Sugars Intake among US Infants and Toddlers. J. Acad. Nutr. Diet. 2020, 120, 23. [Google Scholar] [CrossRef] [PubMed]
- Bowman, S.A.; Clemens, J.C.; Friday, J.E.; Moshfegh, A.J. 2020. Food Patterns Equivalent Intakes from Food: Mean Amounts Consumed per Individual, What We Eat in America, NHANES 2017–2018, Tables 1–4. Worldwide Web Site: Food Surveys Research Group. Available online: https://www.ars.usda.gov/nea/bhnrc/fsrg/fped (accessed on 10 April 2022).
- Rosinger, A.; Herrick, K.; Gahche, J.; Park, S. Sugar-Sweetened Beverage Consumption Among U.S. Youth, 2011–2014. NCHS Data Brief. 2017, 271, 1–8. [Google Scholar]
- Lundeen, E.A.; Park, S.; Pan, L.; Blanck, H.M. Daily Intake of Sugar-Sweetened Beverages among US Adults in 9 States, by State and Sociodemographic and Behavioral Characteristics, 2016. Prev. Chronic Dis. 2018, 15, E154. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids. J. Am. Diet Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025. 9th Edition. December 2020. Available online: DietaryGuidelines.gov (accessed on 10 April 2022).
- Yang, Q.; Zhang, Z.; Gregg, E.W.; Flanders, W.D.; Merritt, R.; Hu, F.B. Added Sugar Intake and Cardiovascular Diseases Mortality Among US Adults. JAMA Int. Med. 2014, 174, 516–524. [Google Scholar] [CrossRef]
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global Nutrition Transition and the Pandemic of Obesity in Developing Countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef]
- Bandy, L.K.; Scarborough, P.; Harrington, R.A.; Rayner, M.; Jebb, S.A. Reductions in Sugar Sales from Soft Drinks in the UK from 2015 to 2018. BMC Med. 2020, 18, 20. [Google Scholar] [CrossRef]
- Stephan, B.C.M.; Wells, J.C.K.; Brayne, C.; Albanese, E.; Siervo, M. Increased Fructose Intake as a Risk Factor for Dementia. J. Gerontol. Ser. A 2010, 65A, 809–814. [Google Scholar] [CrossRef]
- Malik, V.S.; Hu, F.B. Fructose and Cardiometabolic Health: What the Evidence from Sugar-Sweetened Beverages Tells Us. J. Am. Coll. Cardiol. 2015, 66, 1615. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Bremer, A.A.; Medici, V.; Nakajima, K.; Ito, Y.; Nakano, T.; Chen, G.; Fong, T.H.; Lee, V.; Menorca, R.I.; et al. Consumption of Fructose and High Fructose Corn Syrup Increase Postprandial Triglycerides, LDL-Cholesterol, and Apolipoprotein-B in Young Men and Women. J. Clin. Endocrinol. Metab. 2011, 96, E1596–E1605. [Google Scholar] [CrossRef]
- Vos, M.B.; Kimmons, J.E.; Gillespie, C.; Welsh, J.; Blank, H.M. Dietary Fructose Consumption Among US Children and Adults: The Third National Health and Nutrition Examination Survey. Medscape J. Med. 2008, 10, 160. [Google Scholar] [PubMed]
- Elliott, S.S.; Keim, N.L.; Stern, J.S.; Teff, K.; Havel, P.J. Fructose, Weight Gain, and the Insulin Resistance Syndrome. Am. J. Clin. Nutr. 2002, 76, 911–922. [Google Scholar] [CrossRef]
- Bray, G.A. Energy and Fructose from Beverages Sweetened With Sugar or High-Fructose Corn Syrup Pose a Health Risk for Some People. Adv. Nutr. 2013, 4, 220. [Google Scholar] [CrossRef]
- Bray, G.A.; Nielsen, S.J.; Popkin, B.M. Consumption of High-Fructose Corn Syrup in Beverages May Play a Role in the Epidemic of Obesity. Am. J. Clin. Nutr. 2004, 79, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L. Sugar Consumption, Metabolic Disease and Obesity: The State of the Controversy. Crit. Rev. Clin. Lab. Sci. 2016, 53, 52–67. [Google Scholar] [CrossRef]
- Macdonald, I.A. A Review of Recent Evidence Relating to Sugars, Insulin Resistance and Diabetes. Eur. J. Nutr. 2016, 55, 17. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Sánchez-Lozada, L.G.; Andrews, P.; Lanaspa, M.A. Perspective: A Historical and Scientific Perspective of Sugar and Its Relation with Obesity and Diabetes. Adv. Nutr. 2017, 8, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Simoes, E.J. Sugar-Sweetened Beverage, Obesity, and Type 2 Diabetes in Children and Adolescents: Policies, Taxation, and Programs. Curr. Diab. Rep. 2018, 18, 1–10. [Google Scholar] [CrossRef]
- Schetz, M.; de Jong, A.; Deane, A.M.; Druml, W.; Hemelaar, P.; Pelosi, P.; Pickkers, P.; Reintam-Blaser, A.; Roberts, J.; Sakr, Y.; et al. Obesity in the Critically Ill: A Narrative Review. Intensive Care Med. 2019, 45, 757–769. [Google Scholar] [CrossRef] [PubMed]
- Faruque, S.; Tong, J.; Lacmanovic, V.; Agbonghae, C.; Minaya, D.M.; Czaja, K. The Dose Makes the Poison: Sugar and Obesity in the United States—A Review. Pol. J. Food Nutr. Sci. 2019, 69, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Haththotuwa, R.N.; Wijeyaratne, C.N.; Senarath, U. Worldwide Epidemic of Obesity; Elsevier: Oxford, UK, 2013; Volume 1, pp. 3–11. [Google Scholar] [CrossRef]
- Iadecola, C. Sugar and Alzheimer’s Disease: A Bittersweet. Nat. Neurosci. 2015, 18, 477. [Google Scholar] [CrossRef]
- Johnson, R.J.; Gomez-Pinilla, F.; Nagel, M.; Nakagawa, T.; Rodriguez-Iturbe, B.; Sanchez-Lozada, L.G.; Tolan, D.R.; Lanaspa, M.A. Cerebral Fructose Metabolism as a Potential Mechanism Driving Alzheimer’s Disease. Front. Aging Neurosci. 2020, 12, 560865. [Google Scholar] [CrossRef]
- Mohamed, H.E.; Asker, M.E.; Shaheen, M.A.; Eissa, R.G.; Younis, N.N. Alleviation of Fructose-Induced Alzheimer’s Disease in Rats by Pioglitazone and Decaffeinated Green Coffee Bean Extract. J. Food Biochem. 2021, 45, e13715. [Google Scholar] [CrossRef]
- Marques, A.; Dutheil, F.; Durand, E.; Rieu, I.; Mulliez, A.; Fantini, M.L.; Boirie, Y.; Durif, F. Glucose Dysregulation in Parkinson’s Disease: Too Much Glucose or Not Enough Insulin? Parkinsonism Relat. Disord. 2018, 55, 122–127. [Google Scholar] [CrossRef]
- Palavra, N.C.; Lubomski, M.; Flood, V.M.; Davis, R.L.; Sue, C.M. Increased Added Sugar Consumption Is Common in Parkinson’s Disease. Front. Nutr. 2021, 8, 628845. [Google Scholar] [CrossRef]
- Zhang, S.; Lachance, B.B.; Mattson, M.P.; Jia, X. Glucose Metabolic Crosstalk and Regulation in Brain Function and Diseases. Prog. Neurobiol. 2021, 204, 102089. [Google Scholar] [CrossRef]
- Mergenthaler, P.; Lindauer, U.; Dienel, G.A.; Meisel, A. Sugar for the Brain: The Role of Glucose in Physiological and Pathological Brain Function. Trends Neurosci. 2013, 36, 587. [Google Scholar] [CrossRef]
- Tang, B.L. Glucose, Glycolysis, and Neurodegenerative Diseases. J. Cell. Physiol. 2020, 235, 7653–7662. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, M.S.; Iossa, S.; Cigliano, L. Sweet but Bitter: Focus on Fructose Impact on Brain Function in Rodent Models. Nutrients 2021, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Molteni, R.; Barnard, R.J.; Ying, Z.; Roberts, C.K.; Gomez-Pinilla, F. A High-Fat, Refined Sugar Diet Reduces Hippocampal Brain-Derived Neurotrophic Factor, Neuronal Plasticity, and Learning. Neuroscience 2002, 112, 803–814. [Google Scholar] [CrossRef]
- Beilharz, J.E.; Maniam, J.; Morris, M.J. Diet-Induced Cognitive Deficits: The Role of Fat and Sugar, Potential Mechanisms and Nutritional Interventions. Nutrients 2015, 7, 6719–6738. [Google Scholar] [CrossRef]
- Jacques, A.; Chaaya, N.; Beecher, K.; Ali, S.A.; Belmer, A.; Bartlett, S. The Impact of Sugar Consumption on Stress Driven, Emotional and Addictive Behaviors. Neurosci. Biobehav. Rev. 2019, 103, 178–199. [Google Scholar] [CrossRef] [PubMed]
- Bagnol, D.; Al-Shamma, H.A.; Behan, D.; Whelan, K.; Grottick, A.J. Diet-Induced Models of Obesity (DIO) in Rodents. Curr. Protoc. Neurosci. 2012, 59, 9–38. [Google Scholar] [CrossRef] [PubMed]
- Rosini, T.C.; Ramos da Silva, A.S.; de Moraes, C. Diet-Induced Obesity: Rodent Model for the Study of Obesity-Related Disorders. Rev. Assoc. Méd. Bras. 2012, 58, 383–387. [Google Scholar] [CrossRef]
- Dashty, M. A Quick Look at Biochemistry: Carbohydrate Metabolism. Clin. Biochem. 2013, 46, 1339–1352. [Google Scholar] [CrossRef]
- Nakrani, M.N.; Wineland, R.H.; Anjum, F. Physiology, Glucose Metabolism; StatPearls Publishing LLC: Tampa, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK560599/ (accessed on 10 April 2022).
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry, 5th ed.; Freeman, W.H., Ed.; Madison Avenue: New York, NY, USA, 2002; Chapter 17; pp. 497–514. ISBN 9788131248850. [Google Scholar]
- Deng, D.; Yan, N. GLUT, SGLT, and SWEET: Structural and Mechanistic Investigations of the Glucose Transporters. Protein Sci. 2016, 25, 546–558. [Google Scholar] [CrossRef]
- Navale, A.M.; Paranjape, A.N. Glucose Transporters: Physiological and Pathological Roles. Biophys. Rev. 2016, 8, 5. [Google Scholar] [CrossRef]
- Scheepers, A.; Joost, H.G.; Schurmann, A. The Glucose Transporter Families SGLT and GLUT: Molecular Basis of Normal and Aberrant Function. JPEN J. Parenter. Enter. Nutr. 2004, 28, 364–371. [Google Scholar] [CrossRef]
- Koepsell, H. Glucose Transporters in Brain in Health and Disease. Pflug. Arch. Eur. J. Physiol. 2020, 472, 1299–1343. [Google Scholar] [CrossRef]
- Benarroch, E.E. Brain Glucose Transporters. Neurology 2014, 82, 1374–1379. [Google Scholar] [CrossRef] [PubMed]
- McMillin, J.M. Blood Glucose. In Clinical Methods: The History, Physical, and Laboratory Examinations, 3rd ed.; Walker, H.K., Hall, W.D., Hurst, J.W., Eds.; Butterworths: Boston, MA, USA, 1990; Chapter 141. [Google Scholar]
- Georga, E.I.; Fotiadis, D.I.; Tigas, S.K. Pathophysiology and Management of Type 1 Diabetes. In Personalized Predictive Modeling in Type 1 Diabetes; Academic Press: Cambridge, MA, USA, 2018; pp. 15–35. [Google Scholar] [CrossRef]
- Douard, V.; Ferraris, R.P. Regulation of the Fructose Transporter GLUT5 in Health and Disease. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E227. [Google Scholar] [CrossRef] [PubMed]
- Merino, B.; Fernández-Díaz, C.M.; Cózar-Castellano, I.; Perdomo, G. Intestinal Fructose and Glucose Metabolism in Health and Disease. Nutrients 2020, 12, 94. [Google Scholar] [CrossRef] [PubMed]
- Regnault, T.R.; Gentili, S.; Sarr, O.; Toop, C.R.; Sloboda, D.M. Fructose, Pregnancy and Later Life Impacts. Clin. Exp. Pharmacol. Physiol. 2013, 40, 824–837. [Google Scholar] [CrossRef]
- Chadt, A.; Al-Hasani, H. Glucose Transporters in Adipose Tissue, Liver, and Skeletal Muscle in Metabolic Health and Disease. Pflug. Arch. 2020, 472, 1273. [Google Scholar] [CrossRef]
- Hassel, B.; Elsais, A.; Frøland, A.S.; Taubøll, E.; Gjerstad, L.; Quan, Y.; Dingledine, R.; Rise, F. Uptake and Metabolism of Fructose by Rat Neocortical Cells in Vivo and by Isolated Nerve Terminals in Vitro. J. Neurochem. 2015, 133, 572–581. [Google Scholar] [CrossRef]
- Chiba, Y.; Murakami, R.; Matsumoto, K.; Wakamatsu, K.; Nonaka, W.; Uemura, N.; Yanase, K.; Kamada, M.; Ueno, M. Glucose, Fructose, and Urate Transporters in the Choroid Plexus Epithelium. Int. J. Mol. Sci. 2020, 21, 7230. [Google Scholar] [CrossRef]
- Glimcher, L.H.; Lee, A.H. From Sugar to Fat: How the Transcription Factor XBP1 Regulates Hepatic Lipogenesis. Ann. N. Y. Acad. Sci. 2009, 1173, E2. [Google Scholar] [CrossRef]
- Schwarz, J.M.; Clearfield, M.; Mulligan, K. Conversion of Sugar to Fat: Is Hepatic de Novo Lipogenesis Leading to Metabolic Syndrome and Associated Chronic Diseases? J. Am. Osteopath. Assoc. 2017, 117, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.S.; Miller, J.B. Sugars and Blood Glucose Control. Am. J. Clin. Nutr. 1995, 62, 212S–221S. [Google Scholar] [CrossRef] [PubMed]
- Röder, P.v.; Wu, B.; Liu, Y.; Han, W. Pancreatic Regulation of Glucose Homeostasis. Exp. Mol. Med. 2016, 48, e219. [Google Scholar] [CrossRef]
- Jerry Kaneko, J.; Harvey, J.J.; Bruss, M.L. Clinical Biochemistry of Domestic Animals; Academic Press: Cambridge, MA, USA, 2008. [Google Scholar] [CrossRef]
- Nadkarni, P.; Chepurny, O.G.; Holz, G.G. Regulation of Glucose Homeostasis by GLP-1. Prog. Mol. Biol. Transl. Sci. 2014, 121, 23–65. [Google Scholar] [CrossRef]
- Melmed, S.; Koenig, R.; Rosen, C.; Auchus, R.; Goldfine, A. Shlomo Melmed Ronald Koenig Clifford Rosen Richard Auchus Allison Goldfine 37. Complications of Diabetes Mellitus. In Williams Textbook of Endocrinology; Elsevier Health Sciences: Amsterdam, The Netherlands, 2019; pp. 1438–1520. [Google Scholar]
- Lee, A.A.; Owyang, C. Sugars, Sweet Taste Receptors, and Brain Responses. Nutrients 2017, 9, 653. [Google Scholar] [CrossRef]
- Chaudhari, N.; Roper, S.D. The Cell Biology of Taste. J. Cell Biol. 2010, 190, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Depoortere, I. Taste Receptors of the Gut: Emerging Roles in Health and Disease. Gut 2014, 63, 179–190. [Google Scholar] [CrossRef]
- Furness, J.B.; Rivera, L.R.; Cho, H.J.; Bravo, D.M.; Callaghan, B. The Gut as a Sensory Organ. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 729–740. [Google Scholar] [CrossRef]
- Fernstrom, J.D.; Munger, S.D.; Sclafani, A.; de Araujo, I.E.; Roberts, A.; Molinary, S. Mechanisms for Sweetness. J. Nutr. 2012, 142, 1134S. [Google Scholar] [CrossRef]
- Scott, T.R.; Small, D.M. The Role of the Parabrachial Nucleus in Taste Processing and Feeding. Ann. N. Y. Acad. Sci. 2009, 1170, 372–377. [Google Scholar] [CrossRef]
- Abdalla, M.M.I. Ghrelin—Physiological Functions and Regulation. Eur. Endocrinol. 2015, 11, 90. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Chen, Y.; Heiman, M.; DiMarchi, R. Leptin: Structure, Function and Biology. Vitam. Horm. 2005, 71, 345–372. [Google Scholar] [CrossRef]
- Cummings, D.E.; Foster, K.E. Ghrelin-Leptin Tango in Body-Weight Regulation. Gastroenterology 2003, 124, 1532–1535. [Google Scholar] [CrossRef]
- Klok, M.D.; Jakobsdottir, S.; Drent, M.L. The Role of Leptin and Ghrelin in the Regulation of Food Intake and Body Weight in Humans: A Review. Obes. Rev. 2007, 8, 21–34. [Google Scholar] [CrossRef] [PubMed]
- García, A.S.E.; Moreno, A.G.M.; Reyes Castillo, Z. Papel de La Grelina y la Leptina en el Comportamiento Alimentario: Evidencias Genéticas y Moleculares. Endocrinol. Diabetes Nutr. 2021, 68, 654–663. [Google Scholar] [CrossRef]
- Nogueiras, R.; Tschöp, M.H.; Zigman, J.M. CNS Regulation of Energy Metabolism: Ghrelin versus Leptin. Ann. N. Y. Acad. Sci. 2008, 1126, 14. [Google Scholar] [CrossRef]
- Dallman, M.F.; Pecoraro, N.; Akana, S.F.; la Fleur, S.E.; Gomez, F.; Houshyar, H.; Bell, M.E.; Bhatnagar, S.; Laugero, K.D.; Manalo, S. Chronic Stress and Obesity: A New View of “Comfort Food”. Proc. Natl. Acad. Sci. USA 2003, 100, 11696. [Google Scholar] [CrossRef]
- Smith, S.M.; Vale, W.W. The Role of the Hypothalamic-Pituitary-Adrenal Axis in Neuroendocrine Responses to Stress. Dialogues Clin. Neurosci. 2006, 8, 383. [Google Scholar] [CrossRef]
- Miller, W.L. The Hypothalamic-Pituitary-Adrenal Axis: A Brief History. Horm. Res. Paediatr. 2018, 89, 212–223. [Google Scholar] [CrossRef]
- Hiller-Sturmhöfel, S.; Bartke, A. The Endocrine System: An Overview. Alcohol Health Res. World 1998, 22, 153. [Google Scholar]
- Tsigos, C.; Kyrou, I.; Kassi, E.; Chrousos, G.P.; Feingold, K.R.; Anawalt, B.; Boyce, A.; Chrousos, G.; de Herder, W.W.; Dhatariya, K.; et al. (Eds.) Stress: Endocrine Physiology and Pathophysiology. [Updated 2020 Oct 17]; Endotext [Internet], com, Inc.: South Dartmouth, MA, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK278995/#strss-endo-pathophys.STRESS_ENDOCRINE_PA (accessed on 10 April 2022).
- Kinsella, M.T.; Monk, C. Impact of Maternal Stress, Depression & Anxiety on Fetal Neurobehavioral Development. Clin. Obstet. Gynecol. 2009, 52, 425. [Google Scholar] [CrossRef] [PubMed]
- Ulrich-Lai, Y.M.; Fulton, S.; Wilson, M.; Petrovich, G.; Rinaman, L. Stress Exposure, Food Intake, and Emotional State. Stress 2015, 18, 381. [Google Scholar] [CrossRef] [PubMed]
- Wardle, J.; Steptoe, A.; Oliver, G.; Lipsey, Z. Stress, Dietary Restraint and Food Intake. J. Psychosom. Res. 2000, 48, 195–202. [Google Scholar] [CrossRef]
- Dallman, M.F.; la Fleur, S.E.; Pecoraro, N.C.; Gomez, F.; Houshyar, H.; Akana, S.F. Minireview: Glucocorticoids—Food Intake, Abdominal Obesity, and Wealthy Nations in 2004. Endocrinology 2004, 145, 2633–2638. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Christiansen, A.M.; Ostrander, M.M.; Jones, A.A.; Jones, K.R.; Choi, D.C.; Krause, E.G.; Evanson, N.K.; Furay, A.R.; Davis, J.F.; et al. Pleasurable Behaviors Reduce Stress via Brain Reward Pathways. Proc. Natl. Acad. Sci. USA 2010, 107, 20529–20534. [Google Scholar] [CrossRef] [PubMed]
- Nagy, M.R.; Gill, A.; Adams, T.; Gerras, J.; Mazin, L.; Leung, C.; Hasson, R.E. Stress-Induced Suppression of Food Intake in Overweight and Obese Adolescents. Psychosom. Med. 2019, 81, 814–820. [Google Scholar] [CrossRef]
- Herhaus, B.; Ullmann, E.; Chrousos, G.; Petrowski, K. High/Low Cortisol Reactivity and Food Intake in People with Obesity and Healthy Weight. Transl. Psychiatry 2020, 10, 40. [Google Scholar] [CrossRef]
- Inam, Q.U.A.; Ikram, H.; Shireen, E.; Haleem, D.J. Effects of Sugar Rich Diet on Brain Serotonin, Hyperphagia and Anxiety in Animal Model of Both Genders. Pak. J. Pharm. Sci. 2016, 29, 757–763. [Google Scholar]
- Egan, A.E.; Seemiller, L.R.; Packard, A.E.B.; Solomon, M.B.; Ulrich-Lai, Y.M. Palatable Food Reduces Anxiety-like Behaviors and HPA Axis Responses to Stress in Female Rats in an Estrous-Cycle Specific Manner. Horm. Behav. 2019, 115, 104557. [Google Scholar] [CrossRef]
- Packard, A.E.B.; Di, S.; Egan, A.E.; Fourman, S.M.; Tasker, J.G.; Ulrich-Lai, Y.M. Sucrose-Induced Plasticity in the Basolateral Amygdala in a ‘Comfort’ Feeding Paradigm. Brain Struct. Funct. 2017, 222, 4035. [Google Scholar] [CrossRef]
- Dess, N.K. Divergent Responses to Saccharin vs. Sucrose Availability after Stress in Rats. Physiol. Behav. 1992, 52, 115–125. [Google Scholar] [CrossRef]
- Gordon, E.L.; Ariel-Donges, A.H.; Bauman, V.; Merlo, L.J. What Is the Evidence for “Food Addiction?” A Systematic Review. Nutrients 2018, 10, 477. [Google Scholar] [CrossRef] [PubMed]
- Hoebel, B.G.; Avena, N.M.; Bocarsly, M.E.; Rada, P. A Behavioral and Circuit Model Based on Sugar Addiction in Rats. J. Addict. Med. 2009, 3, 33. [Google Scholar] [CrossRef] [PubMed]
- Singh, M. Mood, Food and Obesity. Front. Psychol. 2014, 5, 925. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Wang, H.L.; Li, X.; Ng, T.H.; Morales, M. Mesocorticolimbic Glutamatergic Pathway. J. Neurosci. 2011, 31, 8476–8490. [Google Scholar] [CrossRef]
- Volkow, N.D.; Michaelides, M.; Baler, R. The Neuroscience of Drug Reward and Addiction. Physiol. Rev. 2019, 99, 2115. [Google Scholar] [CrossRef]
- Arias-Carrión, O.; Stamelou, M.; Murillo-Rodríguez, E.; Menéndez-Gonzlez, M.; Pöppel, E. Dopaminergic Reward System: A Short Integrative Review. Int. Arch. Med. 2010, 3, 24. [Google Scholar] [CrossRef]
- Blaess, S.; Stott, S.R.W.; Ang, S.-L. The Generation of Midbrain Dopaminergic Neurons. In Patterning and Cell Type Specification in the Developing CNS and PNS; Academic Press: Cambridge, MA, USA, 2020; pp. 369–398. [Google Scholar] [CrossRef]
- Van Opstal, A.M.; Kaal, I.; van den Berg-Huysmans, A.A.; Hoeksma, M.; Blonk, C.; Pijl, H.; Rombouts, S.A.R.B.; van der Grond, J. Dietary Sugars and Non-Caloric Sweeteners Elicit Different Homeostatic and Hedonic Responses in the Brain. Nutrition 2019, 60, 80–86. [Google Scholar] [CrossRef]
- Van Opstal, A.M.; Hafkemeijer, A.; van den Berg-Huysmans, A.A.; Hoeksma, M.; Blonk, C.; Pijl, H.; Rombouts, S.A.R.B.; van der Grond, J. Brain Activity and Connectivity Changes in Response to Glucose Ingestion. Nutr. Neurosci. 2020, 23, 110–117. [Google Scholar] [CrossRef]
- Van Opstal, A.M.; Hafkemeijer, A.; van den Berg-Huysmans, A.A.; Hoeksma, M.; Mulder, T.P.J.; Pijl, H.; Rombouts, S.A.R.B.; van der Grond, J. Brain Activity and Connectivity Changes in Response to Nutritive Natural Sugars, Non-Nutritive Natural Sugar Replacements and Artificial Sweeteners. Nutr. Neurosci. 2021, 24, 395–405. [Google Scholar] [CrossRef]
- Shestov, A.A.; Emir, U.E.; Kumar, A.; Henry, P.G.; Seaquist, E.R.; Öz, G. Simultaneous Measurement of Glucose Transport and Utilization in the Human Brain. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E1040–E1049. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.; White, D.J.; Scholey, A. Resting State FMRI Reveals Differential Effects of Glucose Administration on Central Appetite Signalling in Young and Old Adults. J. Psychopharmacol. 2020, 34, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Benton, D. The Plausibility of Sugar Addiction and Its Role in Obesity and Eating Disorders. Clin. Nutr. 2010, 29, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Ventura, T.; Santander, J.; Torres, R.; Contreras, A.M. Neurobiologic Basis of Craving for Carbohydrates. Nutrition 2014, 30, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Goran, M.I.; Plows, J.F.; Ventura, E.E. Effects of Consuming Sugars and Alternative Sweeteners during Pregnancy on Maternal and Child Health: Evidence for a Secondhand Sugar Effect. Proc. Nutr. Soc. 2019, 78, 262–271. [Google Scholar] [CrossRef]
- Wiss, D.A.; Avena, N.; Rada, P. Sugar Addiction: From Evolution to Revolution. Front. Psychiatry 2018, 9, 545. [Google Scholar] [CrossRef]
- Avena, N.M.; Rada, P.; Hoebel, B.G. Evidence for Sugar Addiction: Behavioral and Neurochemical Effects of Intermittent, Excessive Sugar Intake. Neurosci. Biobehav. Rev. 2008, 32, 20. [Google Scholar] [CrossRef]
- Avena, N.M.; Long, K.A.; Hoebel, B.G. Sugar-Dependent Rats Show Enhanced Responding for Sugar after Abstinence: Evidence of a Sugar Deprivation Effect. Physiol. Behav. 2005, 84, 359–362. [Google Scholar] [CrossRef]
- Spangler, R.; Wittkowski, K.M.; Goddard, N.L.; Avena, N.M.; Hoebel, B.G.; Leibowitz, S.F. Opiate-like Effects of Sugar on Gene Expression in Reward Areas of the Rat Brain. Brain Res. Mol. Brain Res. 2004, 124, 134–142. [Google Scholar] [CrossRef]
- Avena, N.M.; Bocarsly, M.E.; Rada, P.; Kim, A.; Hoebel, B.G. After Daily Bingeing on a Sucrose Solution, Food Deprivation Induces Anxiety and Accumbens Dopamine/Acetylcholine Imbalance. Physiol. Behav. 2008, 94, 309–315. [Google Scholar] [CrossRef]
- Avena, N.M.; Rada, P.; Moise, N.; Hoebel, B.G. Sucrose Sham Feeding on a Binge Schedule Releases Accumbens Dopamine Repeatedly and Eliminates the Acetylcholine Satiety Response. Neuroscience 2006, 139, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Rada, P.; Avena, N.M.; Hoebel, B.G. Daily Bingeing on Sugar Repeatedly Releases Dopamine in the Accumbens Shell. Neuroscience 2005, 134, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Shariff, M.; Quik, M.; Holgate, J.; Morgan, M.; Patkar, O.L.; Tam, V.; Belmer, A.; Bartlett, S.E. Neuronal Nicotinic Acetylcholine Receptor Modulators Reduce Sugar Intake. PLoS ONE 2016, 11, e0150270. [Google Scholar] [CrossRef] [PubMed]
- Stuber, G.D.; Sparta, D.R.; Stamatakis, A.M.; van Leeuwen, W.A.; Hardjoprajitno, J.E.; Cho, S.; Tye, K.M.; Kempadoo, K.A.; Zhang, F.; Deisseroth, K.; et al. Excitatory Transmission from the Amygdala to Nucleus Accumbens Facilitates Reward Seeking. Nature 2011, 475, 377–382. [Google Scholar] [CrossRef]
- Hajnal, A.; Smith, G.P.; Norgren, R. Oral Sucrose Stimulation Increases Accumbens Dopamine in the Rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, R31–R37. [Google Scholar] [CrossRef]
- Colantuoni, C.; Schwenker, J.; McCarthy, J.; Rada, P.; Ladenheim, B.; Cadet, J.L.; Schwartz, G.J.; Moran, T.H.; Hoebel, B.G. Excessive Sugar Intake Alters Binding to Dopamine and Mu-Opioid Receptors in the Brain. Neuroreport 2001, 12, 3549–3552. [Google Scholar] [CrossRef]
- Gawliński, D.; Gawlińska, K.; Frankowska, M.; Filip, M. Maternal Diet Influences the Reinstatement of Cocaine-Seeking Behavior and the Expression of Melanocortin-4 Receptors in Female Offspring of Rats. Nutrients 2020, 12, 1462. [Google Scholar] [CrossRef]
- Gawliński, D.; Gawlińska, K.; Frankowska, M.; Filip, M. Maternal High-Sugar Diet Changes Offspring Vulnerability to Reinstatement of Cocaine-Seeking Behavior: Role of Melanocortin-4 Receptors. FASEB J. 2020, 34, 9192–9206. [Google Scholar] [CrossRef]
- Archer, Z.A.; Rayner, D.v.; Duncan, J.S.; Bell, L.M.; Mercer, J.G. Introduction of a High-Energy Diet Acutely up-Regulates Hypothalamic Cocaine and Amphetamine-Regulated Transcript, Mc4R and Brown Adipose Tissue Uncoupling Protein-1 Gene Expression in Male Sprague-Dawley Rats. J. Neuroendocrinol. 2005, 17, 10–17. [Google Scholar] [CrossRef]
- Klenowski, P.M.; Shariff, M.R.; Belmer, A.; Fogarty, M.J.; Mu, E.W.H.; Bellingham, M.C.; Bartlett, S.E. Prolonged Consumption of Sucrose in a Binge-Like Manner, Alters the Morphology of Medium Spiny Neurons in the Nucleus Accumbens Shell. Front. Behav. Neurosci. 2016, 10, 54. [Google Scholar] [CrossRef]
- Scheggi, S.; Secci, M.E.; Marchese, G.; de Montis, M.G.; Gambarana, C. Influence of Palatability on Motivation to Operate for Caloric and Non-Caloric Food in Non Food-Deprived and Food-Deprived Rats. Neuroscience 2013, 236, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Tukey, D.S.; Ferreira, J.M.; Antoine, S.O.; D’Amour, J.A.; Ninan, I.; de Vaca, S.C.; Incontro, S.; Wincott, C.; Horwitz, J.K.; Hartner, D.T.; et al. Sucrose Ingestion Induces Rapid AMPA Receptor Trafficking. J. Neurosci. 2013, 33, 6123–6132. [Google Scholar] [CrossRef] [PubMed]
- Carelli, R.M.; West, E.A. When a Good Taste Turns Bad: Neural Mechanisms Underlying the Emergence of Negative Affect and Associated Natural Reward Devaluation by Cocaine. Neuropharmacology 2014, 76, 360–369. [Google Scholar] [CrossRef]
- Beecher, K.; Wang, J.; Jacques, A.; Chaaya, N.; Chehrehasa, F.; Belmer, A.; Bartlett, S.E. Sucrose Consumption Alters Serotonin/Glutamate Co-Localisation Within the Prefrontal Cortex and Hippocampus of Mice. Front. Mol. Neurosci. 2021, 14, 133. [Google Scholar] [CrossRef]
- Chong, C.P.; Shahar, S.; Haron, H.; Din, N.C. Habitual Sugar Intake and Cognitive Impairment among Multi-Ethnic Malaysian Older Adults. Clin. Interv. Aging 2019, 14, 1331. [Google Scholar] [CrossRef] [PubMed]
- Stollery, B.; Christian, L. Glucose and Memory: The Influence of Drink, Expectancy, and Beliefs. Psychopharmacology 2013, 228, 685–697. [Google Scholar] [CrossRef] [PubMed]
- Del-Ponte, B.; Anselmi, L.; Assunção, M.C.F.; Tovo-Rodrigues, L.; Munhoz, T.N.; Matijasevich, A.; Rohde, L.A.; Santos, I.S. Sugar Consumption and Attention-Deficit/Hyperactivity Disorder (ADHD): A Birth Cohort Study. J. Affect. Disord. 2019, 243, 290–296. [Google Scholar] [CrossRef]
- Beecher, K.; Alvarez Cooper, I.; Wang, J.; Walters, S.B.; Chehrehasa, F.; Bartlett, S.E.; Belmer, A. Long-Term Overconsumption of Sugar Starting at Adolescence Produces Persistent Hyperactivity and Neurocognitive Deficits in Adulthood. Front. Neurosci. 2021, 15, 670430. [Google Scholar] [CrossRef]
- Wong, A.; Dogra, V.R.; Reichelt, A.C. High-Sucrose Diets in Male Rats Disrupt Aspects of Decision Making Tasks, Motivation and Spatial Memory, but Not Impulsivity Measured by Operant Delay-Discounting. Behav. Brain Res. 2017, 327, 144–154. [Google Scholar] [CrossRef]
- Colantuoni, C.; Rada, P.; McCarthy, J.; Patten, C.; Avena, N.M.; Chadeayne, A.; Hoebel, B.G. Evidence That Intermittent, Excessive Sugar Intake Causes Endogenous Opioid Dependence. Obes. Res. 2002, 10, 478–488. [Google Scholar] [CrossRef]
- Parylak, S.L.; Cottone, P.; Sabino, V.; Rice, K.C.; Zorrilla, E.P. Effects of CB1 and CRF1 Receptor Antagonists on Binge-like Eating in Rats with Limited Access to a Sweet Fat Diet: Lack of Withdrawal-like Responses. Physiol. Behav. 2012, 107, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Harrell, C.S.; Burgado, J.; Kelly, S.D.; Johnson, Z.P.; Neigh, G.N. High-Fructose Diet during Periadolescent Development Increases Depressive-like Behavior and Remodels the Hypothalamic Transcriptome in Male Rats. Psychoneuroendocrinology 2015, 62, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Eudave, D.M.; BeLow, M.N.; Flandreau, E.I. Effects of High Fat or High Sucrose Diet on Behavioral-Response to Social Defeat Stress in Mice. Neurobiol. Stress 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Shou, J.; Abera, S.; Ziff, E.B. Sucrose Withdrawal Induces Depression and Anxiety-like Behavior by Kir2.1 Upregulation in the Nucleus Accumbens. Neuropharmacology 2018, 130, 10–17. [Google Scholar] [CrossRef]
- Santos, C.J.; Ferreira, A.V.M.; Oliveira, A.L.; Oliveira, M.C.; Gomes, J.S.; Aguiar, D.C. Carbohydrate-Enriched Diet Predispose to Anxiety and Depression-like Behavior after Stress in Mice. Nutr. Neurosci. 2018, 21, 33–39. [Google Scholar] [CrossRef]
- Gueye, A.B.; Vendruscolo, L.F.; de Avila, C.; le Moine, C.; Darnaudéry, M.; Cador, M. Unlimited Sucrose Consumption during Adolescence Generates a Depressive-like Phenotype in Adulthood. Neuropsychopharmacology 2018, 43, 2627–2635. [Google Scholar] [CrossRef]
- Choi, C.S.; Kim, P.; Park, J.H.; Gonzales, E.L.T.; Kim, K.C.; Cho, K.S.; Ko, M.J.; Yang, S.M.; Seung, H.; Han, S.H.; et al. High Sucrose Consumption during Pregnancy Induced ADHD-like Behavioral Phenotypes in Mice Offspring. J. Nutr. Biochem. 2015, 26, 1520–1526. [Google Scholar] [CrossRef]
- Barker, D.J.P. The Origins of the Developmental Origins Theory. J. Intern. Med. 2007, 261, 412–417. [Google Scholar] [CrossRef]
- Wadhwa, P.D.; Buss, C.; Entringer, S.; Swanson, J.M. Developmental Origins of Health and Disease: Brief History of the Approach and Current Focus on Epigenetic Mechanisms. Semin. Reprod. Med. 2009, 27, 358–368. [Google Scholar] [CrossRef]
- Heindel, J.J.; Vandenberg, L.N. Developmental Origins of Health and Disease: A Paradigm for Understanding Disease Cause and Prevention. Curr. Opin. Pediatr. 2015, 27, 248–253. [Google Scholar] [CrossRef]
- Netting, M.J.; Middleton, P.F.; Makrides, M. Does Maternal Diet during Pregnancy and Lactation Affect Outcomes in Offspring? A Systematic Review of Food-Based Approaches. Nutrition 2014, 30, 1225–1241. [Google Scholar] [CrossRef] [PubMed]
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in Utero and Early-Life Conditions on Adult Health and Disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Montalvo-Martínez, L.; Maldonado-Ruiz, R.; Cárdenas-Tueme, M.; Reséndez-Pérez, D.; Camacho, A. Maternal Overnutrition Programs Central Inflammation and Addiction-Like Behavior in Offspring. Biomed Res. Int. 2018, 2018, 8061389. [Google Scholar] [CrossRef] [PubMed]
- Kominiarek, M.A.; Rajan, P. Nutrition Recommendations in Pregnancy and Lactation. Med. Clin. N. Am. 2016, 100, 1199–1215. [Google Scholar] [CrossRef] [PubMed]
- ACOG Practice Bulletin No 156: Obesity in Pregnancy. Obstet. Gynecol. 2015, 126, e112–e126. [CrossRef] [PubMed]
- Torloni, M.R.; Betran, A.P.; Horta, B.L.; Nakamura, M.U.; Atallah, A.N.; Moron, A.F.; Valente, O. Prepregnancy BMI and the Risk of Gestational Diabetes: A Systematic Review of the Literature with Meta-Analysis. Obes. Rev. 2009, 10, 194–203. [Google Scholar] [CrossRef]
- Metzger, B.E.; Lowe, L.P.; Dyer, A.R.; Trimble, E.R.; Sheridan, B.; Hod, M.; Chen, R.; Yogev, Y.; Coustan, D.R.; Catalano, P.M.; et al. Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study: Associations with Neonatal Anthropometrics. Diabetes 2009, 58, 453–459. [Google Scholar] [CrossRef]
- Stuebe, A.M.; Rich-Edwards, J.W. The Reset Hypothesis: Lactation and Maternal Metabolism. Am. J. Perinatol. 2009, 26, 81–88. [Google Scholar] [CrossRef]
- Kjos, S.L.; Henry, O.A.; Montoro, M.; Buchanan, T.A.; Mestman, J.H. Insulin-Requiring Diabetes in Pregnancy: A Randomized Trial of Active Induction of Labor and Expectant Management. Am. J. Obstet. Gynecol. 1993, 169, 611–615. [Google Scholar] [CrossRef]
- Diniz, J.M.M.; da Costa, T.H.M. Independent of Body Adiposity, Breast-Feeding Has a Protective Effect on Glucose Metabolism in Young Adult Women. Br. J. Nutr. 2004, 92, 905–912. [Google Scholar] [CrossRef]
- Blackmore, H.L.; Ozanne, S.E. Maternal Diet-Induced Obesity and Offspring Cardiovascular Health. J. Dev. Orig. Health Dis. 2013, 4, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.B.; MacKenzie, K.C.; Gahagan, S. The Effect of Maternal Obesity on the Offspring. Clin. Obstet. Gynecol. 2014, 57, 508. [Google Scholar] [CrossRef]
- Francis, E.C.; Dabelea, D.; Shankar, K.; Perng, W. Maternal Diet Quality during Pregnancy Is Associated with Biomarkers of Metabolic Risk among Male Offspring. Diabetologia 2021, 64, 2478–2490. [Google Scholar] [CrossRef] [PubMed]
- Giordano, F.; Carbone, P.; Nori, F.; Mantovani, A.; Taruscio, D.; Figà-Talamanca, I. Maternal Diet and the Risk of Hypospadias and Cryptorchidism in the Offspring. Paediatr. Perinat. Epidemiol. 2008, 22, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Radzicka-Mularczyk, S.A.; Pietryga, M.; Brazert, J. How Mother’s Obesity May Affect the Pregnancy and Offspring. Ginekol. Polska 2020, 91, 769–772. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, K.M.; Reynolds, R.M.; Prescott, S.L.; Nyirenda, M.; Jaddoe, V.W.V.; Eriksson, J.G.; Broekman, B.F.P. Influence of Maternal Obesity on the Long-Term Health of Offspring. Lancet Diabetes Endocrinol. 2017, 5, 53–64. [Google Scholar] [CrossRef]
- Sharma, N.; Okere, I.C.; Barrows, B.R.; Lei, B.; Duda, M.K.; Yuan, C.L.; Previs, S.F.; Sharov, V.G.; Azimzadeh, A.M.; Ernsberger, P.; et al. High-Sugar Diets Increase Cardiac Dysfunction and Mortality in Hypertension Compared to Low-Carbohydrate or High-Starch Diets. J. Hypertens. 2008, 26, 1402–1410. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Iyer, A.; Nazer, R.; Alam, A.; Diwan, V.; Kauter, K.; Sernia, C.; Campbell, F.; Ward, L.; et al. High-Carbohydrate High-Fat Diet–Induced Metabolic Syndrome and Cardiovascular Remodeling in Rats. J. Cardiovasc. Pharmacol. 2011, 57, 51–64. [Google Scholar] [CrossRef]
- Toop, C.R.; Muhlhausler, B.S.; O’Dea, K.; Gentili, S. Impact of Perinatal Exposure to Sucrose or High Fructose Corn Syrup (HFCS-55) on Adiposity and Hepatic Lipid Composition in Rat Offspring. J. Physiol. 2017, 595, 4379–4398. [Google Scholar] [CrossRef]
- Tobar-Bernal, F.A.; Zamudio, S.R.; Quevedo-Corona, L. The High-Fructose Intake of Dams during Pregnancy and Lactation Exerts Sex-Specific Effects on Adult Rat Offspring Metabolism. J. Dev. Orig. Health Dis. 2021, 12, 411–419. [Google Scholar] [CrossRef]
- Ornellas, F.; Carapeto, P.V.; Aguila, M.B.; Mandarim-de-Lacerda, C.A. Sex-Linked Changes and High Cardiovascular Risk Markers in the Mature Progeny of Father, Mother, or Both Father and Mother Consuming a High-Fructose Diet. Nutrition 2020, 71, 110612. [Google Scholar] [CrossRef] [PubMed]
- Trujillo-Villarreal, L.A.; Romero-Díaz, V.J.; Marino-Martínez, I.A.; Fuentes-Mera, L.; Ponce-Camacho, M.A.; Devenyi, G.A.; Mallar Chakravarty, M.; Camacho-Morales, A.; Garza-Villarreal, E.E. Maternal Cafeteria Diet Exposure Primes Depression-like Behavior in the Offspring Evoking Lower Brain Volume Related to Changes in Synaptic Terminals and Gliosis. Transl. Psychiatry 2021, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Ribaroff, G.A.; Wastnedge, E.; Drake, A.J.; Sharpe, R.M.; Chambers, T.J.G. Animal Models of Maternal High Fat Diet Exposure and Effects on Metabolism in Offspring: A Meta-Regression Analysis. Obes. Rev. 2017, 18, 673–686. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, H.; Zeng, M.; Tang, X.; Zhu, X.; Guo, Y.; Qi, L.; Xie, Y.; Zhang, M.; Chen, D. Maternal High Sugar and Fat Diet Benefits Offspring Brain Function via Targeting on the Gut-Brain Axis. Aging 2021, 13, 10240–10274. [Google Scholar] [CrossRef] [PubMed]
- Andreas, E.; Reid, M.; Zhang, W.; Moley, K.H. The Effect of Maternal High-Fat/High-Sugar Diet on Offspring Oocytes and Early Embryo Development. Mol. Hum. Reprod. 2019, 25, 717–728. [Google Scholar] [CrossRef]
- Gawlińska, K.; Gawliński, D.; Filip, M.; Przegaliński, E. Relationship of Maternal High-Fat Diet during Pregnancy and Lactation to Offspring Health. Nutr. Rev. 2021, 79, 709–725. [Google Scholar] [CrossRef] [PubMed]
- Menting, M.D.; Mintjens, S.; van de Beek, C.; Frick, C.J.; Ozanne, S.E.; Limpens, J.; Roseboom, T.J.; Hooijmans, C.R.; van Deutekom, A.W.; Painter, R.C. Maternal Obesity in Pregnancy Impacts Offspring Cardiometabolic Health: Systematic Review and Meta-Analysis of Animal Studies. Obes. Rev. 2019, 20, 675–685. [Google Scholar] [CrossRef]
- Menting, M.D.; van de Beek, C.; Mintjens, S.; Wever, K.E.; Korosi, A.; Ozanne, S.E.; Limpens, J.; Roseboom, T.J.; Hooijmans, C.; Painter, R.C. The Link between Maternal Obesity and Offspring Neurobehavior: A Systematic Review of Animal Experiments. Neurosci. Biobehav. Rev. 2019, 98, 107–121. [Google Scholar] [CrossRef]
- Mizera, J.; Kazek, G.; Niedzielska-Andres, E.; Pomierny-Chamiolo, L. Maternal High-Sugar Diet Results in NMDA Receptors Abnormalities and Cognitive Impairment in Rat Offspring. FASEB J. 2021, 35, e21547. [Google Scholar] [CrossRef]
- Bayol, S.A.; Farrington, S.J.; Stickland, N.C. A Maternal “junk Food” Diet in Pregnancy and Lactation Promotes an Exacerbated Taste for “Junk Food” and a Greater Propensity for Obesity in Rat Offspring. Br. J. Nutr. 2007, 98, 843–851. [Google Scholar] [CrossRef]
- Volkow, N.D.; Poznyak, V.; Saxena, S.; Gerra, G. Drug Use Disorders: Impact of a Public Health Rather than a Criminal Justice Approach. World Psychiatry 2017, 16, 213. [Google Scholar] [CrossRef] [PubMed]
- The United Nations Office on Drugs and Crime. World Drug Report 2021; United Nations Publication: Vienna, Austria, 2021; Sales No. E.21.XI.8; ISBN 9789211483611. [Google Scholar]
- Castilla-Ortega, E.; Serrano, A.; Blanco, E.; Araos, P.; Suárez, J.; Pavón, F.J.; de Fonseca, F.R.; Santín, L.J. A Place for the Hippocampus in the Cocaine Addiction Circuit: Potential Roles for Adult Hippocampal Neurogenesis. Neurosci. Biobehav. Rev. 2016, 66, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Venton, B.J.; Seipel, A.T.; Phillips, P.E.M.; Wetsel, W.C.; Gitler, D.; Greengard, P.; Augustine, G.J.; Wightman, R.M. Cocaine Increases Dopamine Release by Mobilization of a Synapsin-Dependent Reserve Pool. J. Neurosci. 2006, 26, 3206–3209. [Google Scholar] [CrossRef]
- Egilmez, Y.; Jung, M.E.; Lane, J.D.; Emmett-Oglesby, M.W. Dopamine Release during Cocaine Self-Administration in Rats: Effect of SCH23390. Brain Res. 1995, 701, 142–150. [Google Scholar] [CrossRef]
- Ahmed, S.H.; Guillem, K.; Vandaele, Y. Sugar Addiction: Pushing the Drug-Sugar Analogy to the Limit. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 434–439. [Google Scholar] [CrossRef]
- Onaolapo, A.Y.; Onaolapo, O.J.; Olowe, O.A. An Overview of Addiction to Sugar. In Dietary Sugar, Salt and Fat in Human Health; Academic Press: Cambridge, MA, USA, 2020; pp. 195–216. [Google Scholar] [CrossRef]
- Hartmann, H.; Pauli, L.K.; Janssen, L.K.; Huhn, S.; Ceglarek, U.; Horstmann, A. Preliminary Evidence for an Association between Intake of High-Fat High-Sugar Diet, Variations in Peripheral Dopamine Precursor Availability and Dopamine-Dependent Cognition in Humans. J. Neuroendocrinol. 2020, 32, e12917. [Google Scholar] [CrossRef]
- Lenoir, M.; Serre, F.; Cantin, L.; Ahmed, S.H. Intense Sweetness Surpasses Cocaine Reward. PLoS ONE 2007, 2, e698. [Google Scholar] [CrossRef]
- Lopes, F.M.; Boos, F.Z.; Zanini, A.M.; Czermainski, F.R. Neuropsychological and Behavioral Aspects of Drug Use and Abuse: Theory, Research, and Intervention. Drugs Hum. Behav. 2021, 1, 151–170. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H.; Wilson, W.L. Sugar Addiction: Is It Real? A Narrative Review. Br. J. Sports Med. 2018, 52, 910–913. [Google Scholar] [CrossRef]
- Rorabaugh, J.M.; Stratford, J.M.; Zahniser, N.R. A Relationship between Reduced Nucleus Accumbens Shell and Enhanced Lateral Hypothalamic Orexin Neuronal Activation in Long-Term Fructose Bingeing Behavior. PLoS ONE 2014, 9, e95019. [Google Scholar] [CrossRef]
- Peleg-Raibstein, D.; Sarker, G.; Litwan, K.; Krämer, S.D.; Ametamey, S.M.; Schibli, R.; Wolfrum, C. Enhanced Sensitivity to Drugs of Abuse and Palatable Foods Following Maternal Overnutrition. Transl. Psychiatry 2016, 6, e911. [Google Scholar] [CrossRef] [PubMed]
- Duart-Castells, L.; Cantacorps, L.; López-Arnau, R.; Montagud-Romero, S.; Puster, B.; Mera, P.; Serra, D.; Camarasa, J.; Pubill, D.; Valverde, O.; et al. Effects of High-Fat Diet and Maternal Binge-Like Alcohol Consumption and Their Influence on Cocaine Response in Female Mice Offspring. Int. J. Neuropsychopharmacol. 2021, 24, 77. [Google Scholar] [CrossRef] [PubMed]
- Wijenayake, S.; Rahman, M.F.; Lum, C.M.W.; de Vega, W.C.; Sasaki, A.; McGowan, P.O. Maternal High-Fat Diet Induces Sex-Specific Changes to Glucocorticoid and Inflammatory Signaling in Response to Corticosterone and Lipopolysaccharide Challenge in Adult Rat Offspring. J. Neuroinflamm. 2020, 17, 116. [Google Scholar] [CrossRef] [PubMed]
- Bocarsly, M.E.; Barson, J.R.; Hauca, J.M.; Hoebel, B.G.; Leibowitz, S.F.; Avena, N.M. Effects of Perinatal Exposure to Palatable Diets on Body Weight and Sensitivity to Drugs of Abuse in Rats. Physiol. Behav. 2012, 107, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Collins, G.T.; Chen, Y.; Tschumi, C.; Rush, E.L.; Mensah, A.; Koek, W.; France, C.P. Effects of Consuming a Diet High in Fat and/or Sugar on the Locomotor Effects of Acute and Repeated Cocaine in Male and Female C57BL/6J Mice. Exp. Clin. Psychopharmacol. 2015, 23, 228–237. [Google Scholar] [CrossRef]
- Avena, N.M.; Hoebel, B.G. Amphetamine-Sensitized Rats Show Sugar-Induced Hyperactivity (Cross-Sensitization) and Sugar Hyperphagia. Pharmacol. Biochem. Behav. 2003, 74, 635–639. [Google Scholar] [CrossRef]
- Avena, N.M.; Hoebel, B.G. A Diet Promoting Sugar Dependency Causes Behavioral Cross-Sensitization to a Low Dose of Amphetamine. Neuroscience 2003, 122, 17–20. [Google Scholar] [CrossRef]
- Moore, C.F.; Leonard, M.Z.; Micovic, N.M.; Miczek, K.A.; Sabino, V.; Cottone, P. Reward Sensitivity Deficits in a Rat Model of Compulsive Eating Behavior. Neuropsychopharmacology 2019, 45, 589–596. [Google Scholar] [CrossRef]
- Rorabaugh, J.M.; Stratford, J.M.; Zahniser, N.R. Differences in Bingeing Behavior and Cocaine Reward Following Intermittent Access to Sucrose, Glucose or Fructose Solutions. Neuroscience 2015, 301, 213–220. [Google Scholar] [CrossRef]
- Vitale, M.A.; Chen, D.; Kanarek, R.B. Chronic Access to a Sucrose Solution Enhances the Development of Conditioned Place Preferences for Fentanyl and Amphetamine in Male Long-Evans Rats. Pharmacol. Biochem. Behav. 2003, 74, 529–539. [Google Scholar] [CrossRef]
- Bruggeman, E.C.; Li, C.; Ross, A.P.; Doherty, J.M.; Williams, B.F.; Frantz, K.J.; Parent, M.B. A High Fructose Diet Does Not Affect Amphetamine Self-Administration or Spatial Water Maze Learning and Memory in Female Rats. Pharmacol. Biochem. Behav. 2011, 99, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Orsini, C.A.; Ginton, G.; Shimp, K.G.; Avena, N.M.; Gold, M.S.; Setlow, B. Food Consumption and Weight Gain after Cessation of Chronic Amphetamine Administration. Appetite 2014, 78, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Foltin, R.W. Consumption of Palatable Food Decreases the Anorectic Effects of Serotonergic, but Not Dopaminergic Drugs in Baboons. Physiol. Behav. 2011, 103, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Loebens, M.; Barros, H.M.T. Diet Influences Cocaine Withdrawal Behaviors in the Forced Swimming Test. Pharmacol. Biochem. Behav. 2003, 74, 259–267. [Google Scholar] [CrossRef]
- Ströhle, A.; Gensichen, J.; Domschke, K. The Diagnosis and Treatment of Anxiety Disorders. Deutsches Ärzteblatt Int. 2018, 115, 611. [Google Scholar] [CrossRef] [PubMed]
- Bandelow, B.; Baldwin, D.; Abelli, M.; Bolea-Alamanac, B.; Bourin, M.; Chamberlain, S.R.; Cinosi, E.; Davies, S.; Domschke, K.; Fineberg, N.; et al. Biological Markers for Anxiety Disorders, OCD and PTSD: A Consensus Statement. Part II: Neurochemistry, Neurophysiology and Neurocognition. World J. Biol. Psychiatry 2017, 18, 162–214. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.P.; Book, S.W. Anxiety and Substance Use Disorders: A Review. Psychiatr. Times 2008, 25, 19. [Google Scholar] [PubMed]
- Stathopoulou, G.; Gold, A.K.; Hoyt, D.L.; Milligan, M.; Hearon, B.A.; Otto, M.W. Does Anxiety Sensitivity Predict Addiction Severity in Opioid Use Disorder? Addict. Behav. 2021, 112, 106644. [Google Scholar] [CrossRef]
- Horigian, V.E.; Schmidt, R.D.; Feaster, D.J. Loneliness, Mental Health, and Substance Use among US Young Adults during COVID-19. J. Psychoact. Drugs 2021, 53, 1–9. [Google Scholar] [CrossRef]
- Vorspan, F.; Mehtelli, W.; Dupuy, G.; Bloch, V.; Lépine, J.P. Anxiety and Substance Use Disorders: Co-Occurrence and Clinical Issues. Curr. Psychiatry Rep. 2015, 17, 1–7. [Google Scholar] [CrossRef]
- Goodwin, R.D.; Stein, D.J. Anxiety Disorders and Drug Dependence: Evidence on Sequence and Specificity among Adults. Psychiatry Clin. Neurosci. 2013, 67, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Zvolensky, M.J.; Schmidt, N.B. Anxiety and Substance Use Disorders: Introduction to the Special Series. J. Anxiety Disord. 2004, 18, 1–6. [Google Scholar] [CrossRef]
- Smith, J.P.; Randall, C.L. Anxiety and Alcohol Use Disorders: Comorbidity and Treatment Considerations. Alcohol Res. Curr. Rev. 2012, 34, 414. [Google Scholar]
- Johannessen, E.L.; Andersson, H.W.; Bjørngaard, J.H.; Pape, K. Anxiety and Depression Symptoms and Alcohol Use among Adolescents—A Cross Sectional Study of Norwegian Secondary School Students. BMC Public Health 2017, 17, 494. [Google Scholar] [CrossRef] [PubMed]
- Grasing, M.J.; Xu, H.; Idowu, J.Y.; Grasing, K. Changes Depression- and Anxiety- like Behaviors Following Selective Breeding for Cocaine Reinforcement. Psychiatry Res. 2021, 295, 113637. [Google Scholar] [CrossRef] [PubMed]
- Markou, A.; Kosten, T.R.; Koob, G.F. Neurobiological Similarities in Depression and Drug Dependence: A Self-Medication Hypothesis. Neuropsychopharmacology 1998, 18, 135–174. [Google Scholar] [CrossRef]
- DiGirolamo, G.J.; Gonzalez, G.; Smelson, D.; Guevremont, N.; Andre, M.I.; Patnaik, P.O.; Zaniewski, Z.R. Increased Depression and Anxiety Symptoms Are Associated with More Breakdowns in Cognitive Control to Cocaine Cues in Veterans with Cocaine Use Disorder. J. Dual Diagn. 2017, 13, 298–304. [Google Scholar] [CrossRef]
- Ayran, G.; Köse, S.; Küçükoğlu, S.; Özdemir, A.A. The Effect of Anxiety on Nicotine Dependence among University Students during the COVID-19 Pandemic. Perspect. Psychiatr. Care 2022, 58, 114–123. [Google Scholar] [CrossRef]
- Benowitz, N.L. Nicotine Addiction. N. Engl. J. Med. 2010, 362, 2295. [Google Scholar] [CrossRef]
- Kobiella, A.; Ulshöfer, D.E.; Vollmert, C.; Bühler, M.; Esslinger, C.; Smolka, M.N. Nicotine Increases Neural Response to Unpleasant Stimuli and Anxiety in Non-Smokers. Addict. Biol. 2011, 16, 285–295. [Google Scholar] [CrossRef]
- Conner, K.R.; Pinquart, M.; Holbrook, A.P. Meta-Analysis of Depression and Substance Use and Impairment among Cocaine Users. Drug Alcohol Depend. 2008, 98, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Poling, J.; Kosten, T.R.; Sofuoglu, M. Treatment Outcome Predictors for Cocaine Dependence. Am. J. Drug Alcohol Abuse 2007, 33, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Rounsaville, B.J. Treatment of Cocaine Dependence and Depression. Biol. Psychiatry 2004, 56, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Leventhal, A.M.; Mooney, M.E.; DeLaune, K.A.; Schmitz, J.M. Using Addiction Severity Profiles to Differentiate Cocaine-Dependent Patients with and without Comorbid Major Depression. Am. J. Addict. Am. Acad. Psychiatr. Alcohol. Addict. 2006, 15, 362. [Google Scholar] [CrossRef] [PubMed]
- Ford, J.D.; Gelernter, J.; DeVoe, J.S.; Zhang, W.; Weiss, R.D.; Brady, K.; Farrer, L.; Kranzler, H.R. Association of Psychiatric and Substance Use Disorder Comorbidity with Cocaine Dependence Severity and Treatment Utilization in Cocaine-Dependent Individuals. Drug Alcohol Depend. 2009, 99, 193–203. [Google Scholar] [CrossRef]
- Leventhal, A.M.; Brightman, M.; Ameringer, K.J.; Greenberg, J.; Mickens, L.; Ray, L.A.; Sun, P.; Sussman, S. Anhedonia Associated with Stimulant Use and Dependence in a Population-Based Sample of American Adults. Exp. Clin. Psychopharmacol. 2010, 18, 562–569. [Google Scholar] [CrossRef]
- Wardle, M.C.; Vincent, J.N.; Suchting, R.; Green, C.E.; Lane, S.D.; Schmitz, J.M. Anhedonia Is Associated with Poorer Outcomes in Contingency Management for Cocaine Use Disorder. J. Subst. Abuse Treat. 2017, 72, 32–39. [Google Scholar] [CrossRef]
- Lee, R.S.C.; Hoppenbrouwers, S.; Franken, I. A Systematic Meta-Review of Impulsivity and Compulsivity in Addictive Behaviors. Neuropsychol. Rev. 2019, 29, 14–26. [Google Scholar] [CrossRef]
- Dawe, S.; Loxton, N.J. The Role of Impulsivity in the Development of Substance Use and Eating Disorders. Neurosci. Biobehav. Rev. 2004, 28, 343–351. [Google Scholar] [CrossRef]
- Whiteside, S.P.; Lynam, D.R. The Five Factor Model and Impulsivity: Using a Structural Model of Personality to Understand Impulsivity. Personal. Individ. Differ. 2001, 30, 669–689. [Google Scholar] [CrossRef]
- Homberg, J.R.; Karel, P.; Verheij, M.M.M. Individual Differences in Cocaine Addiction: Maladaptive Behavioural Traits. Addict. Biol. 2014, 19, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Cohen, A.; Davids, M.; Rabindranath, A. Attention-Deficit/Hyperactivity Disorder in Relation to Addictive Behaviors: A Moderated-Mediation Analysis of Personality-Risk Factors and Sex. Front. Psychiatry 2015, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Dalley, J.W.; Robbins, T.W. Fractionating Impulsivity: Neuropsychiatric Implications. Nat. Rev. Neurosci. 2017, 18, 158–171. [Google Scholar] [CrossRef] [PubMed]
- Stamates, A.L.; Linden-Carmichael, A.N.; Preonas, P.D.; Lau-Barraco, C. Testing Daily Associations between Impulsivity, Affect, and Alcohol Outcomes: A Pilot Study. Addict. Res. Theory 2019, 27, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Lees, B.; Mewton, L.; Stapinski, L.A.; Squeglia, L.M.; Rae, C.D.; Teesson, M. Neurobiological and Cognitive Profile of Young Binge Drinkers: A Systematic Review and Meta-Analysis. Neuropsychol. Rev. 2019, 29, 357–385. [Google Scholar] [CrossRef]
- Bozkurt, M.; Evren, C.; Umut, G.; Evren, B. Relationship of Attention-Deficit/Hyperactivity Disorder Symptom Severity with Severity of Alcohol-Related Problems in a Sample of Inpatients with Alcohol Use Disorder. Neuropsychiatr. Dis. Treat. 2016, 12, 1661–1667. [Google Scholar] [CrossRef]
- Andersson, H.W.; Lauvsnes, A.D.F.; Nordfjærn, T. Emerging Adults in Inpatient Substance Use Treatment: A Prospective Cohort Study of Patient Characteristics and Treatment Outcomes. Eur. Addict. Res. 2021, 27, 206–215. [Google Scholar] [CrossRef]
- Franques, P.; Auriacombe, M.; Piquemal, E.; Verger, M.; Brisseau-Gimenez, S.; Grabot, D.; Tignol, J. Sensation Seeking as a Common Factor in Opioid Dependent Subjects and High Risk Sport Practicing Subjects. A Cross Sectional Study. Drug Alcohol Depend. 2003, 69, 121–126. [Google Scholar] [CrossRef]
- Kreek, M.J.; Nielsen, D.A.; Butelman, E.R.; LaForge, K.S. Genetic Influences on Impulsivity, Risk Taking, Stress Responsivity and Vulnerability to Drug Abuse and Addiction. Nat. Neurosci. 2005, 8, 1450–1457. [Google Scholar] [CrossRef]
- Zuckerman, M.; Eysenck, S.B.; Eysenck, H.J. Sensation Seeking in England and America: Cross-Cultural, Age, and Sex Comparisons. J. Consult. Clin. Psychol. 1978, 46, 139–149. [Google Scholar] [CrossRef]
- Zuckerman, M.; Ball, S.; Black, J. Influences of Sensation Seeking, Gender, Risk Appraisal, and Situational Motivation on Smoking. Addict. Behav. 1990, 15, 209–220. [Google Scholar] [CrossRef]
- Pelloux, Y.; Costentin, J.; Duterte-Boucher, D. Differential Effects of Novelty Exposure on Place Preference Conditioning to Amphetamine and Its Oral Consumption. Psychopharmacology 2004, 171, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Wingo, T.; Nesil, T.; Choi, J.S.; Li, M.D. Novelty Seeking and Drug Addiction in Humans and Animals: From Behavior to Molecules. J. Neuroimmune Pharmacol. 2015, 11, 456–470. [Google Scholar] [CrossRef]
- Belin, D.; Berson, N.; Balado, E.; Piazza, P.V.; Deroche-Gamonet, V. High-Novelty-Preference Rats Are Predisposed to Compulsive Cocaine Self-Administration. Neuropsychopharmacology 2011, 36, 569. [Google Scholar] [CrossRef] [PubMed]
- Yetnikoff, L.; Arvanitogiannis, A. Differential Sensitivity to the Acute and Sensitizing Behavioral Effects of Methylphenidate as a Function of Strain in Adolescent and Young Adult Rats. Behav. Brain Funct. 2013, 9, 38. [Google Scholar] [CrossRef]
- Zakharova, E.; Leoni, G.; Kichko, I.; Izenwasser, S. Differential Effects of Methamphetamine and Cocaine on Conditioned Place Preference and Locomotor Activity in Adult and Adolescent Male Rats. Behav. Brain Res. 2009, 198, 45. [Google Scholar] [CrossRef] [PubMed]
- Mofatteh, M. Risk Factors Associated with Stress, Anxiety, and Depression among University Undergraduate Students. AIMS Public Health 2021, 8, 36. [Google Scholar] [CrossRef]
- de Souza, L.; Barros, W.d.M.; de Souza, R.M.; Delanogare, E.; Machado, A.E.; Braga, S.P.; Rosa, G.K.; Nardi, G.M.; Rafacho, A.; Speretta, G.F.F.; et al. Impact of Different Fructose Concentrations on Metabolic and Behavioral Parameters of Male and Female Mice. Physiol. Behav. 2021, 228, 113187. [Google Scholar] [CrossRef]
- Carneiro-Nascimento, S.; Opacka-Juffry, J.; Costabile, A.; Boyle, C.N.; Herde, A.M.; Ametamey, S.M.; Sigrist, H.; Pryce, C.R.; Patterson, M. Chronic Social Stress in Mice Alters Energy Status Including Higher Glucose Need but Lower Brain Utilization. Psychoneuroendocrinology 2020, 119, 104747. [Google Scholar] [CrossRef]
- Iemolo, A.; Valenza, M.; Tozier, L.; Knapp, C.M.; Kornetsky, C.; Steardo, L.; Sabino, V.; Cottone, P. Withdrawal from Chronic, Intermittent Access to a Highly Palatable Food Induces Depressive-like Behavior in Compulsive Eating Rats. Behav. Pharmacol. 2012, 23, 593–602. [Google Scholar] [CrossRef]
- Aucoin, M.; Lachance, L.; Naidoo, U.; Remy, D.; Shekdar, T.; Sayar, N.; Cardozo, V.; Rawana, T.; Chan, I.; Cooley, K. Diet and Anxiety: A Scoping Review. Nutrients 2021, 13, 4418. [Google Scholar] [CrossRef] [PubMed]
- Kanter, J.W.; Rusch, L.C.; Brondino, M.J. Depression Self-Stigma: A New Measure and Preliminary Findings. J. Nerv. Ment. Dis. 2008, 196, 663–670. [Google Scholar] [CrossRef] [PubMed]
- McCarron, R.M.; Vanderlip, E.R.; Rado, J. Depression. Ann. Intern. Med. 2016, 165, ITC49–ITC63. [Google Scholar] [CrossRef]
- Kloster, A.; Hyer, M.M.; Dyer, S.; Salome-Sanchez, C.; Neigh, G.N. High Fructose Diet Induces Sex-Specific Modifications in Synaptic Respiration and Affective-like Behaviors in Rats. Neuroscience 2021, 454, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Reis, D.J.; Ilardi, S.S.; Namekata, M.S.; Wing, E.K.; Fowler, C.H. The Depressogenic Potential of Added Dietary Sugars. Med. Hypotheses 2020, 134, 109421. [Google Scholar] [CrossRef]
- Xu, Y.; Zeng, L.; Zou, K.; Shan, S.; Wang, X.; Xiong, J.; Zhao, L.; Zhang, L.; Cheng, G. Role of Dietary Factors in the Prevention and Treatment for Depression: An Umbrella Review of Meta-Analyses of Prospective Studies. Transl. Psychiatry 2021, 11, 478. [Google Scholar] [CrossRef]
- Huang, Q.; Liu, H.; Suzuki, K.; Ma, S.; Liu, C. Linking What We Eat to Our Mood: A Review of Diet, Dietary Antioxidants, and Depression. Antioxidants 2019, 8, 376. [Google Scholar] [CrossRef]
- Pérez-Ara, M.Á.; Gili, M.; Visser, M.; Penninx, B.W.J.H.; Brouwer, I.A.; Watkins, E.; Owens, M.; García-Toro, M.; Hegerl, U.; Kohls, E.; et al. Associations of Non-Alcoholic Beverages with Major Depressive Disorder History and Depressive Symptoms Clusters in a Sample of Overweight Adults. Nutrients 2020, 12, 3202. [Google Scholar] [CrossRef]
- Grant, J.E.; Valle, S.; Chamberlain, S.R. Nutrition in Skin Picking Disorder and Trichotillomania. Front. Psychiatry 2021, 12, 761321. [Google Scholar] [CrossRef]
- Steele, C.C.; Steele, T.J.; Gwinner, M.; Rosenkranz, S.K.; Kirkpatrick, K. The Relationship between Dietary Fat Intake, Impulsive Choice, and Metabolic Health. Appetite 2021, 165, 105292. [Google Scholar] [CrossRef]
- Steele, C.C.; Pirkle, J.R.A.; Davis, I.R.; Kirkpatrick, K. Dietary Effects on the Determinants of Food Choice: Impulsive Choice, Discrimination, Incentive Motivation, Preference, and Liking in Male Rats. Appetite 2019, 136, 160–172. [Google Scholar] [CrossRef] [PubMed]
| Types (DP) | Subgroup | Major Components | Natural Sources |
|---|---|---|---|
| Sugars (DP1, and DP2) | monosaccharides | glucose, galactose, fructose | fruit, vegetables, honey, seeds |
| disaccharides | sucrose, maltose, lactose | table sugar (sugar cane or sugar beet), grains, dairy products | |
| Oligosaccharides (DP3–9) | maltooligosaccharides | maltodextrins | corn starch |
| Polysaccharides (DP ≥ 10) | starch | amylose, amylopectin | cereals, vegetables, processed flour products |
| non-starch | cellulose, hemicellulose, pectins, glycogen | vegetables, fruits, seeds, meat |
| Localization | SGLTs | GLUTs |
|---|---|---|
| Intestine | SGLT1, SGLT3, SGLT4 *, SGLT6 | GLUT2 *, GLUT5 *, GLUT7 *, GLUT12 |
| Kidney | SGLT1, SGLT2 *, SGLT3, SGLT4 *, SGLT5 *, SGLT6 | GLUT1, GLUT2 *, GLUT3, GLUT5 *, GLUT9, GLUT10, GLUT11 *, GLUT13 |
| Liver | SGLT2 *, SGLT3, SGLT4 * | GLUT1, GLUT2 *, GLUT3, GLUT5 *, GLUT7, GLUT8 *, GLUT9 |
| Brain | SGLT1, SGLT2, SGLT3, SGLT4 *, SGLT6 | GLUT1, GLUT2 *, GLUT3, GLUT4, GLUT5 *, GLUT6, GLUT8 *, GLUT13 |
| Prenatal Exposure | Animals | Drug of Abuse | Consequences Enhanced by HSD | Ref | ||
|---|---|---|---|---|---|---|
| HSD Model | Period | Behavioural | Molecular/Neurochemical | |||
| 70% carbohydrates (44% sucrose) | Pregnancy and lactation | Female WR offspring | Cocaine hydrochloride | ↑ CUE- and cocaine reinstatement relapse in cocaine SA ↑ cocaine-seeking behaviour | ↑ MC4R expression in the NAc, and dorsal striatum | [130] |
| Male WR offspring | ↓ MC4R expression in the PFC, NAc, and dorsal striatum ↑ MC4R expression in the amygdala | [131] | ||||
| 10% sucrose or 16% HFCS solution | Male and female SD offspring | Amphetamine | ↑ female alcohol intake during alcohol training ↑ male hyperactivity in LAM | - | [199] | |
| Postnatal Exposure | Animals | Drug of Abuse | Consequences Enhanced by HSD | Ref | ||
|---|---|---|---|---|---|---|
| HSD Model | Period (Days) | Behavioural | Molecular/Neurochemical | |||
| 10% wt/vol sucrose | 84 | Male and female C57BL/6J mice | Cocaine | ↑ female postcocaine tests locomotor activity, ↑ female sensitization to cocaine in LAM | - | [200] |
| 0.2% saccharin (0.3 mL/trial) | 15 | Male WR | ↓ sensitization to cocaine in discrete-trials choice procedures | - | [192] | |
| 70% from corn starch | 38 | Male WR | ↓ immobility, head shake, and locomotion after drug treatment before FST ↓ immobility, climbing, swim, dive, and locomotion in cocaine withdrawal FST | - | [209] | |
| 8% fructose or glucose or sucrose | 9 | Male SD | ↑ cocaine-CPP after sucrose and fructose, ↓ cocaine-CPP after glucose bingeing | - | [204] | |
| 10% wt/v sucrose | 21 | Female SD | Amphetamine | ↑ activity and sensitivity to low dose of drug in LAM | - | [202] |
| 10% sucrose | 5 | Male SD | ↑ activity in LAM | - | [201] | |
| 32% (w/v) sucrose solution | 21 | Male LE | ↑ conditioning scores in CPP | - | [205] | |
| 50% kcal from sucrose | >35 | Male WR | ↑ locomotor activity in LAM, ↓ sensitivity to drug in CPP, and ICSS | ↓ NAc DA ↑ VTA DAT mRNA ↓ extracellular DA | [203] | |
| 60% fructose | 47, 88, and 144 | Female SD | ↑↓SA, and spatial memory testing in water maze test | - | [206] | |
| 64.3% sugar in palatable food | 30 | Male LE | ↑ chow intake after SA | - | [207] | |
| 75% sugar in Skittles candy | 28 | Male baboons (Papio cynocephalus anubis) | ↓ sensitivity to candy reward during pellet meal sessions | - | [208] | |
| 32% (w/v) sucrose solution | 21 | Male LE | Fentanyl | ↑ conditioning scores ↑ time spent in the previously drug-paired side in CPP | - | [205] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Witek, K.; Wydra, K.; Filip, M. A High-Sugar Diet Consumption, Metabolism and Health Impacts with a Focus on the Development of Substance Use Disorder: A Narrative Review. Nutrients 2022, 14, 2940. https://doi.org/10.3390/nu14142940
Witek K, Wydra K, Filip M. A High-Sugar Diet Consumption, Metabolism and Health Impacts with a Focus on the Development of Substance Use Disorder: A Narrative Review. Nutrients. 2022; 14(14):2940. https://doi.org/10.3390/nu14142940
Chicago/Turabian StyleWitek, Kacper, Karolina Wydra, and Małgorzata Filip. 2022. "A High-Sugar Diet Consumption, Metabolism and Health Impacts with a Focus on the Development of Substance Use Disorder: A Narrative Review" Nutrients 14, no. 14: 2940. https://doi.org/10.3390/nu14142940
APA StyleWitek, K., Wydra, K., & Filip, M. (2022). A High-Sugar Diet Consumption, Metabolism and Health Impacts with a Focus on the Development of Substance Use Disorder: A Narrative Review. Nutrients, 14(14), 2940. https://doi.org/10.3390/nu14142940