Use of Dietary Supplements and Perceived Knowledge among Adults Living with Fibromyalgia in Norway: A Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population and Recruitment
2.3. Survey
2.4. Variables
2.4.1. Outcome Variables
2.4.2. Covariates
2.5. Statistical Analysis
3. Results
3.1. Sample Characteristics
3.2. Prevalence of DS
3.3. Lifestyle Factor and Health Conditions
3.4. Knowledge of DS
3.5. Predictors of Spending Money on DS
3.6. Predictors of Being in the Group of Users
3.7. Summary of the Main Findings
4. Discussion
Strengths and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bellato, E.; Marini, E.; Costoldi, F.; Barbasetti, N.; Mattei, L.; Bonasia, D.E.; Blonna, D. Fibromyalgia syndrome: Etiology, pathogenesis, diagnosis, and treatment. Pain Res. Treat. 2012, 2012, 426130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clauw, D.J. Fibromyalgia and related conditions. In Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 2015; Volume 90, pp. 680–692. [Google Scholar] [CrossRef] [Green Version]
- Gracely, R.H.; Schweinhardt, P. Key mechanisms mediating fibromyalgia. Clin. Exp. Rheumatol. 2015, 33 (Suppl. 88), s3–s6. [Google Scholar] [PubMed]
- Littlejohn, G.; Guymer, E. Key milestones contributing to the understanding of the mechanisms underlying fibromylagia. Biomedicines 2020, 8, 223. [Google Scholar] [CrossRef]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.-A.; Goldenberg, D.L.; Häuser, W.; Katz, R.L.; Mease, P.J.; Russel, A.S.; Russel, I.J.; Walitt, B. Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin. Arthritis Rheum. 2016, 46, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Heidari, F.; Afshari, M.; Moosazadeh, M. Prevalence of fibromyalgia in general population and patients, a systematic review and meta-analysis. Rheumatol. Int. 2017, 37, 1527–1539. [Google Scholar] [CrossRef] [PubMed]
- Fitzcharles, M.-A.; Rampakakis, E.; Ste-Marie, P.A.; Sampalis, J.S.; Shir, Y. The Association of Socioeconomic Status and Symptom Severity in Persons with Fibromyalgia. J. Rheumatol. 2014, 41, 1398–1404. [Google Scholar] [CrossRef]
- Okifuji, A.; Gao, J.; Bokat, C.; Hare, B.D. Management of fibromyalgia syndrome in 2016. Pain Manag. 2016, 6, 383–400. [Google Scholar] [CrossRef] [Green Version]
- Walitt, B.; Fitzcharles, M.-A.; Hasset, A.L.; Katz, R.S.; Häuser, W.; Wolfe, F. The longitudinal outcome of fibromyalgia: A study of 1555 patients. J. Rheumatol. 2011, 38, 2238–2246. [Google Scholar] [CrossRef]
- Lind, B.K.; Lafferty, W.E.; Tyree, P.T.; Diehr, P.K.; Grembowski, D.E. Use of complementary and alternative medicine providers by fibromyaligia patients under insurance coverage. Arthritis Rheum. 2007, 57, 71–76. [Google Scholar] [CrossRef] [Green Version]
- Silva, A.R.; Bernardo, A.; Costa, J.; Cardoso, A.; Santos, P.; de Mesquita, M.F.; Patto, J.V.; Moreira, P.; Silva, M.L.; Padrão, P. Dietary interventions in fibromyalgia: A systematic review. Ann. Med. 2019, 51 (Suppl. 1), 2–14. [Google Scholar] [CrossRef] [Green Version]
- Lowry, E.; Marley, J.; McVeigh, J.G.; McSorley, E.; Allsopp, P.; Kerr, D. Dietary Interventions in the Management of Fibromyalgia: A Systematic Review and Best-Evidence Synthesis. Nutrients 2020, 12, 2664. [Google Scholar] [CrossRef]
- Pagliai, G.; Giangrandi, I.; Dinu, M.; Sofi, F.; Colombini, B. Nutritional Interventions in the Management of Fibromyalgia Syndrome. Nutrients 2020, 12, 2525. [Google Scholar] [CrossRef]
- Merriam-Webster. Dictionary. Dietary Supplements. Available online: https://www.merriam-webster.com/dictionary/dietary%20supplement (accessed on 25 August 2021).
- Skaer, T.L. Fibromyalgia: Disease synopsis, medication cost effectiveness and economic burden. Pharmacoeconomics 2014, 32, 457–466. [Google Scholar] [CrossRef]
- Panus, S.R. Complementary and Alternative Remedies for Rheumatic Disorders. Available online: https://www.uptodate.com/contents/complementary-and-alternative-remedies-for-rheumatic-disorders (accessed on 12 August 2021).
- Arranz, L.I.; Canela, M.-Á.; Rafecas, M. Dietary aspects in fibromyalgia patients: Results of a surevey on food awareness, allergies, and nutritional supplementation. Rheumatol. Int. 2012, 32, 2615–2621. [Google Scholar] [CrossRef]
- Feinberg, T.; Lilly, C.; Innes, K. Nonvitamin, Nonmineral Dietary Supplement Use among Adults with Fibromyalgia: United States, 2007–2012. Evid.-Based Complementary Altern. Med. 2017, 2017, 6751856. [Google Scholar] [CrossRef] [Green Version]
- European Food Safety Authority. Food supplements. Available online: https://www.efsa.europa.eu/en/topics/topic/food-supplements (accessed on 18 July 2021).
- Wooltorton, E. Too much of a good thing? Toxic effects of vitamin and mineral supplements. CMAJ 2003, 169, 47–48. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/pmc164945/ (accessed on 14 August 2021). [PubMed]
- Karbownik, M.S.; Horne, R.; Paul, E.; Kowalczky, E.; Szemraj, J. Determinants of Knowledge About Dietary Supplements Among Polish Internet Users: Nationwide Cross-sectional Study. J. Med. Internet Res. 2021, 23, e25228. [Google Scholar] [CrossRef] [PubMed]
- Totland, T.H.; Melnæs, B.K.; Lundberg-Hallén, N.; Helland-Kigen, K.M.; Lund-Blix, N.A.; Myhre, J.B.; Johansen, A.M.W.; Løken, E.B.; Andersen, L.F. Norkost 3: The Third National Diet Survey Conducted in Norway among Men and Women Aged 18–70 Years. The Norwegian Directorate of Health, 2010/2011. Available online: http://ghdx.healthdata.org/record/norway-norkost-national-food-consumption-survey-2010-2011 (accessed on 21 August 2021).
- Norway’s National Research Center in Complementary and Alternative Medicine (NAFKAM). Use of Alternative Treatment in Norway 2018. Available online: https://nafkam.no/sites/default/files/2019-07/NAFKAM-2018%20rapport%20finale%20250619_0.pdf (accessed on 23 August 2021).
- Norwegian Institute of Public Health. Norwegian Mother, Father and Child Cohort Study (MoBa). Available online: https://www.fhi.no/en/studies/moba/ (accessed on 15 August 2021).
- Kofoed, C.L.F.; Christensen, J.; Dragsted, L.O.; Tjønneland, A.; Roswall, N. Determinants of dietary supplement use—healthy individuals use dietary supplements. Br. J. Nutr. 2015, 113, 1993–2000. [Google Scholar] [CrossRef] [Green Version]
- Streiner, D.L.; Norman, G.R.; Cairney, J. Health Measurement Scales: A Practical Guide to Their Development and Use, 5th ed.; Oxford University Press: Oxford, UK, 2015; p. 416. [Google Scholar]
- Pallant, J. SPSS Survival Manual. In A Step by Step Guide to Data Analysis Using IBM SPSS, 7th ed.; Taylor & Francis: Oxford, Australia, 2020; p. 378. [Google Scholar]
- Yunus, M.B. The Role of Gender in Fibromyalgia Syndrome. Curr. Rheumatol. Rep. 2001, 3, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, A.; Blatman, J.; El-Dash, N.; Franco, J.C. Consumer Usage and Reasons for Using Dietary Supplements: Report of a Series of Surveys. J. Am. Coll. Nutr. 2014, 33, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Kantor, E.D.; Rehm, C.D.; Du, M.; White, E.; Giovannucci, E.L. Trends in Dietary Supplements Use Among US Adults From 1999–2012. JAMA 2016, 316, 1464–1474. [Google Scholar] [CrossRef] [PubMed]
- Arnesen, E. Use of dietary supplements and medicines in a group of people over 50 years of age. Nor. Tidsskr. Ernæring 2017, 1, 6–12. Available online: http://urn.nb.no/URN:NBN:no-66536 (accessed on 19 June 2021).
- Waaseth, M.; Larsen, H.N.; Thoresen, M.B.S.; Skeie, G. Use of dietary supplements among middle-aged Norwegian women. Nor. Tidsskr. Ernæring 2017, 1, 8–16. Available online: https://hdl.handle.net/10037/16831.1 (accessed on 25 June 2021).
- Wall, G.C.; Krypel, L.L.; Miller, M.J.; Rees, D.M. A pilot study of complementary and alternative medicine use in patients with fibromyalgia syndrome. Pharm. Pract. 2007, 5, 185–190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Council for Responsible Nutrition. 2019 CRN Consumer Survey on Dietary Supplements. Available online: https://www.crnusa.org/2019survey/Topline-Infographic#more (accessed on 9 May 2021).
- Karras, S.; Rapti, E.; Matsoukas, S.; Kotsa, K. Vitamin D in Fibromyalgia: A Causative or Confounding Biological Interplay? Nutrients 2016, 8, 343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pajor, E.M.; Eggers, S.M.; Kurfs, K.C.; Oenema, A.; de Vries, H. Why do Dutch people use dietary supplements? Exploring the role of socio-cognitive and psychosocial determinants. Appetite 2017, 114, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Frey, A.; Hoffman, I.; Heuer, T. Characterisation of vitamin and mineral supplement users differentiated according to their motives for using supplements: Results of the German National Nutrition Monitoring (NEMONIT). Public Health Nutr. 2017, 20, 2173–2182. [Google Scholar] [CrossRef] [Green Version]
- Dickinson, A.; MacKay, D.; Wong, A. Consumer attitudes about the role of multivitamins and other dietary supplements: Report of a survey. Nutr. J. 2015, 14, 66. [Google Scholar] [CrossRef] [Green Version]
- Rock, C.L. Multivitamin-multimineral supplements: Who uses them? Am. J. Clin. Nutr. 2007, 85, 277S–279S. [Google Scholar] [CrossRef] [Green Version]
- Bailey, R.L.; Gahche, J.J.; Miller, P.E.; Thomas, P.R.; Dwyer, J.T. Why US adults use dietary supplements. JAMA Intern. Med. 2013, 173, 355–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruger, J.; Dunning, D. Unskilled and unaware of It: Difficulties in Recognizing One’s own Incompetence Lead to Inflated Self-Assessments. J. Personal. Soc. Psychol. 1999, 77, 1121–1134. [Google Scholar] [CrossRef]
- Sirico, F.; Miressi, S.; Costaldo, C.; Spera, R.; Montagnani, S.; Di Meglio, F.; Nurzynska, D. Habits and beliefs related to food supplements: Results of a survey among Italian students of different education fields and levels. PLoS ONE 2018, 13, e0191424. [Google Scholar] [CrossRef] [PubMed]
- Le, C.; Finbråten, H.S.; Pettersen, K.S.; Guttersrud, Ø. Health Literacy in the Norwegian population. Oslo: The Norwegian Directorate of Health. 2021. Available online: https://www.helsedirektoratet.no/rapporter/befolkningens-helsekompetanse (accessed on 10 September 2021).
- Creswell, J.W.; Creswell, J.D. Research Design: Qualitative, Quantitative, and Mixed Methods Approaches; SAGE Publications, Inc.: Thousands Oaks, CA, USA, 2018; p. 304. [Google Scholar]
- Bowling, A. Research Methods in Health; Open University Press: New York, NY, USA, 2014; p. 568. [Google Scholar]
- Weir, P.T.; Harlan, G.A.; Nkoy, F.L.; Jones, S.S.; Hegmann, K.T.; Gren, L.H.; Lyon, J.L. The incidence of fibromyalgia and its associated comorbidities: A population-based retrospective cohort study based on International Classification of Diseases, 9th revision codes. J. Clin. Rheumatol. 2006, 12, 124–128. [Google Scholar] [CrossRef]
- Wigers, S.H. Fibromyalgia—An update. Tidsskr. Nor. Lægeforen 2002, 122, 1300–1304. Available online: https://tidsskriftet.no/2002/05/tema-funksjonelle-lidelser/fibromyalgi-en-oppdatering (accessed on 14 August 2021).
- Ihlebæk, C.; Brage, S.; Natvig, B.; Bruusgaard, D. Prevalence of musculoskeletal disorders in Norway. Tidsskr. Nor. Lægeforen 2010, 130, 2365–2368. Available online: https://tidsskriftet.no/2010/12/oversiktsartikkel/forekomst-av-muskel-og-skjelettlidelser-i-norge (accessed on 25 July 2021). [CrossRef] [Green Version]
- Gilbert, G.E.; Prion, S. Making Sense of Methods and Measurement: Lawshe’s Content Validity Index. Clin. Simul. Nurs. 2016, 12, 530–531. [Google Scholar] [CrossRef]
- Bornstein, M.H.; Jager, J.; Putnick, D.L. Sampling in Development Science: Situations, shortcomings, solutions, and standards. Dev. Rev. 2013, 33, 357–370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cuschieri, S. The STROBE guidelines. Saudi J. Anasthesia 2019, 13 (Suppl. 1), S31–S34. [Google Scholar] [CrossRef]
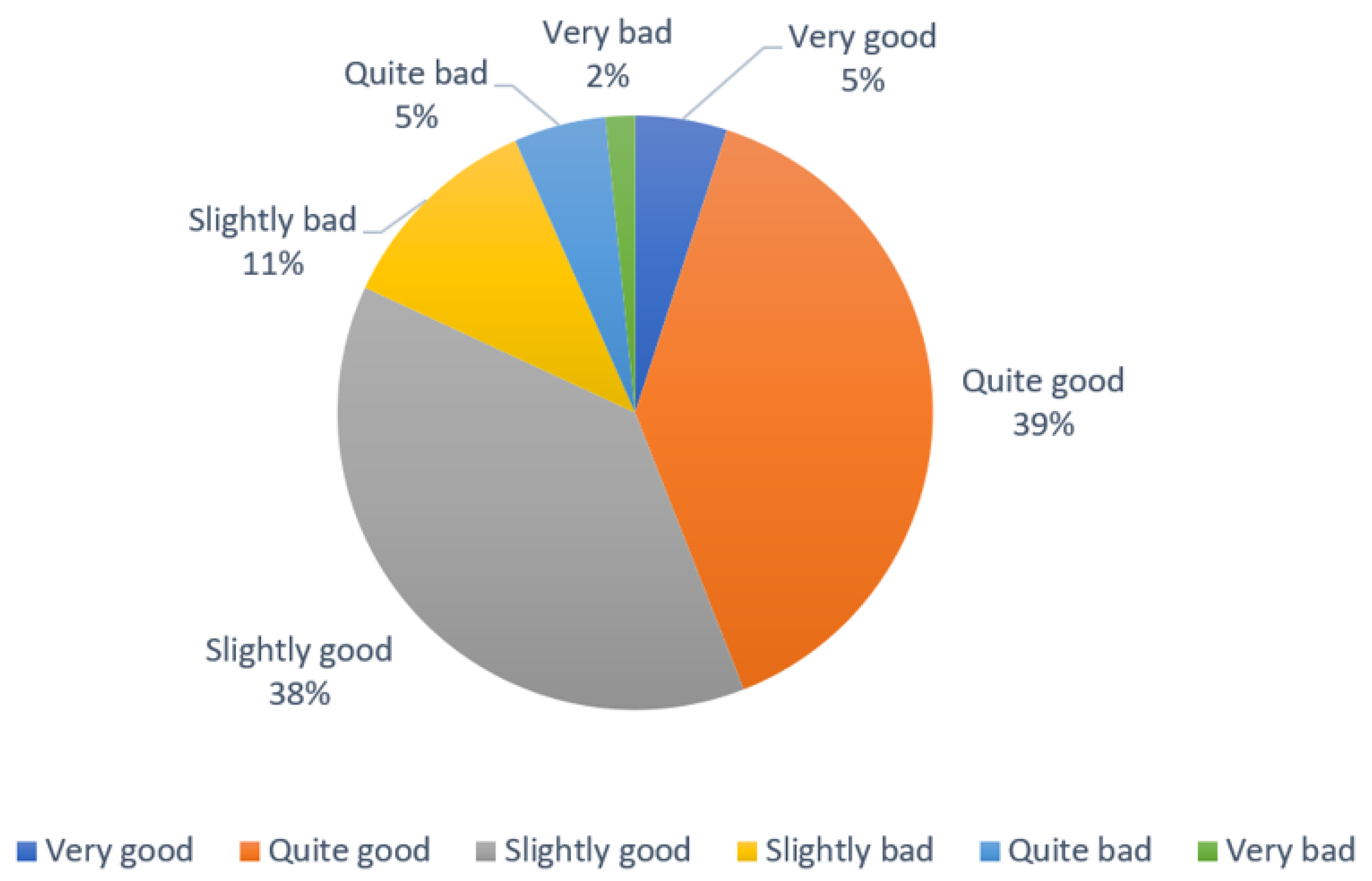
| Reg (n) | Whole Sample (n = 504) | Range (Min/Max) | Users of DS (n = 430) | Non-Users of DS (n = 74) | p | |
|---|---|---|---|---|---|---|
| Gender, women (n) (%) | 504 | 476 (94.4) | 407 (94.7) | 69 (93.2) | 0.63 | |
| Age in years, mean (SD) | 504 | 52.7 (10.1) | 21–81 | 52.8 (10.1) | 51.8 (10) | 0.43 |
| Marital status, living with a partner (n) (%) | 504 | 382 (75.8) | 324 (75.3) | 58 (78.4) | 0.57 | |
| Education, high (n) (%) | 504 | 253 (50.2) | 232 (54) | 21 (28.4) | 0.01 * | |
| Employment, active (n) (%) | 504 | 171 (33.9) | 145 (33.7) | 26 (35.1) | 0.81 | |
| Health professional background (n) (%) | 504 | 132 (26.2) | 117 (27.2) | 15 (20.3) | 0.21 | |
| Number of diagnoses, mean (SD) | 504 | 2.2 (1.3) | 1–7 | 2.3 (1.3) | 2.1 (1.3) | 0.24 |
| Comorbidity, two or more diagnoses (n) (%) | 504 | 311 (61.7) | 271 (63) | 40 (54.1) | 0.14 | |
| Number of medications, high (n) (%) | 412 | 230 (45.6) | 201 (56.9) | 29 (49.2) | 0.27 | |
| BMI, kg/m2, mean (SD) | 499 | 28.5 (5.4) | 16–46.1 | 28.4 (5.4) | 29.2 (5.4) | 0.25 |
| Lifestyle factors, health index, mean (SD) | 486 | 3.4 (0.9) | 0–5 | 3.4 (0.9) | 3.3 (0.9) | 0.38 |
| Self-reported health in general, mean (SD) | 504 | 3.2 (1.1) | 1–6 | 3.2 (1.1) | 3.1 (1.1) | 0.36 |
| Self-reported health today, mean (SD) | 504 | 50.3 (17.6) | 0–90 | 50.3 (17.7) | 50 (17.3) | 0.88 |
| Self-reported knowledge of DS, mean (SD) | 504 | 4.2 (1) | 1–6 | 4.3 (0.9) | 3.6 (1.3) | 0.01 * |
| Knowledge about health claims, mean (SD) | 504 | 26.9 (6.2) | 10–48 | 26.7 (6.3) | 28.5 (5.7) | 0.02 * |
| Sources of information, reliable (n) (%) | 504 | 402 (79.8) | 351 (81.6) | 51 (31.1) | 0.01 * |
| Types of DS | Group * | Not Used | Last Year | Monthly | Weekly |
|---|---|---|---|---|---|
| Fish oil | Group 1 | 273 (63.5) | 28 (6.5) | 12 (2.8) | 117 (27.2) |
| Omega-3 fatty acids | Group 1 | 165 (38.4) | 40 (9.3) | 11 (2.6) | 214 (49.9) |
| Multivitamin/-mineral | Group 2 | 227 (52.8) | 43 (10.0) | 10 (2.3) | 150 (34.9) |
| Vitamin A | Group 3 | 398 (92.6) | 9 (2.1) | 2 (0.5) | 21 (4.9) |
| Vitamin D | Group 3 | 102 (23.7) | 44 (10.2) | 10 (2.3) | 274 (63.7) |
| Vitamin E | Group 3 | 389 (90.5) | 17 (4.0) | 5 (1.2) | 19 (4.5) |
| Vitamin C | Group 3 | 206 (47.9) | 61 (14.2) | 34 (7.9) | 129 (30) |
| Vitamin K | Group 3 | 377 (87.7) | 17 (4.0) | 1 (0.2) | 35 (8.2) |
| Vitamin B12 | Group 3 | 220 (51.2) | 63 (14.7) | 14 (3.3) | 133 (31) |
| Folic acid/folate | Group 3 | 346 (80.5) | 30 (7.0) | 3 (0.7) | 51 (11.9) |
| Iodine | Group 3 | 385 (89.5) | 15 (3.5) | 3 (0.7) | 27 (6.3) |
| Calcium | Group 3 | 298 (69.3) | 31 (7.2) | 8 (1.9) | 93 (21.7) |
| Iron | Group 3 | 337 (78.4) | 39 (9.1) | 11 (2.6) | 43 (9.9) |
| Zink | Group 3 | 355 (82.6) | 22 (5.1) | 4 (0.9) | 49 (11.1) |
| Magnesium | Group 3 | 151 (35.1) | 43 (10.0) | 16 (3.7) | 220 (51.1) |
| Selenium | Group 3 | 386 (89.8) | 11 (2.6) | 1 (0.2) | 32 (7.4) |
| Chromium | Group 3 | 380 (88.4) | 19 (4.4) | 4 (0.9) | 27 (6.3) |
| Probiotics/prebiotics | Group 4 | 327 (76.0) | 49 (11.4) | 7 (1.6) | 47 (10.9) |
| Omega 6 fatty acids | Group 4 | 370 (86.0) | 17 (4.0) | 2 (0.5) | 41 (9.5) |
| CLA | Group 4 | 419 (97.4) | 6 (1.4) | 0 (0.0) | 5 (1.2) |
| Q10 | Group 4 | 406 (94.4) | 12 (2.8) | 4 (0.9) | 8 (1.8) |
| Garlic | Group 4 | 308 (71.6) | 26 (6.0) | 34 (7.9) | 62 (14.4) |
| Ginseng | Group 4 | 405 (94.2) | 12 (2.8) | 5 (1.2) | 8 (1.9) |
| Valerian | Group 4 | 419 (97.4) | 10 (2.3) | 0 (0.0) | 1 (0.2) |
| Red sunflower/echinacea | Group 4 | 406 (94.4) | 15 (3.5) | 7 (1.6) | 2 (0.4) |
| Rhodiola rosea | Group 4 | 415 (97.6) | 6 (1.4) | 2 (0.5) | 2 (0.4) |
| St. John’s wort | Group 4 | 421 (97.9) | 5 (1.2) | 3 (0.7) | 1 (0.2) |
| Collagen Plus | Group 5 | 384 (89.3) | 18 (4.2) | 2 (0.5) | 26 (6.0) |
| Medox | Group 5 | 417 (97.0) | 6 (1.4) | 2 (0.5) | 5 (1.1) |
| VitaPro | Group 5 | 411 (95.6) | 9 (2.1) | 0 (0.0) | 10 (2.3) |
| Glucosamine | Group 5 | 409 (95.1) | 14 (3.3) | 0 (0.0) | 7 (1.6) |
| Lifestyle Factors | Registered | Never/Less often than 1 Day | 1–4 Days | 5–7 Days |
|---|---|---|---|---|
| Smoking status | n = 501 | 439 (87.1) | 5 (1.0) | 57 (11.3) |
| Snuff habits | n = 501 | 474 (94.0) | 5 (1.0) | 22 (4.4) |
| Alcohol use | n = 500 | 296 (58.7) | 199 (39.5) | 5 (1.0) |
| Physical activity | n = 499 | 45 (8.9) | 332 (65.9) | 122 (24.2) |
| Fruits/vegetables | n = 498 | 55 (10.9) | 251 (49.8) | 192 (38.2) |
| Health Claims | Correct Answers | |
|---|---|---|
| 1. All adults need to take DS | 245 (48.7) | No |
| 2. DS must refer to the effect before distribution in the market | 28 (5.6) | No |
| 3. DS must undergo tests to ensure that they are safe before they can be sold on the market | 18 (3.6) | No |
| 4. Taking multivitamin/-mineral products prevents diseases in healthy adults | 232 (46.1) | No |
| 5. Taking vitamin C supplements regularly prevents colds | 196 (38.9) | No |
| 6. Taking an omega-3 supplement or fish oil regularly prevents cardiovascular disease | 67 (13.3) | No |
| 7. Taking antioxidant supplements regularly prevents various cancers | 262 (52) | No |
| 8. For the elderly, taking vitamin D supplements regularly will reduce the risk of bone fractures | 102 (20.3) | No |
| 9. The use of DS may influence the effect of medicines | 388 (77) | Yes |
| Independent Variables | Unadjusted Analyses | ||||
|---|---|---|---|---|---|
| n | R2 | B (95% CI) | β | Sig. | |
| Gender (0 = women, 1 = men) | 504 | −0.002 | −255.48 (−1836.5–1325.6) | −0.015 | 0.751 |
| Age in years | 504 | 0.001 | 21.95 (−13.2–57.1) | 0.059 | 0.221 |
| Marital status (no partner = 0, partner = 1) | 504 | 0.007 | −847.71 (−1669.3–−26.1) | −0.098 | 0.043 * |
| Education (low = 0, high = 1) | 504 | 0.016 | 939.27 (231.1–1647.5) | 0.125 | 0.009 * |
| Employment (not active = 0, active = 1) | 504 | −0.002 | 84.87 (−667.7–837.4) | 0.011 | 0.825 |
| Health professional background (no = 0, yes = 1) | 504 | −0.002 | −88.08 (−887.5–711.3) | −0.010 | 0.829 |
| Number of diagnoses | 504 | −0.001 | 102.1 (−170.8–377) | 0.036 | 0.460 |
| Number of medications (low = 0, high = 1) | 412 | 0.002 | 524.92 (−257.8–1307.7) | 0.07 | 0.19 |
| BMI, kg/m2 | 499 | 0.002 | −44.23 (−110.1–21.6) | −0.064 | 0.187 |
| Lifestyle factors, health index | 486 | −0.001 | −143.75 (−570.7–283.2) | −0.033 | 0.508 |
| Self-reported health in general | 504 | −0.002 | 36.89 (−286.7–360.5) | 0.011 | 0.823 |
| Self-reported health today | 504 | −0.002 | −4.71 (−24.9–15.4) | −0.022 | 0.646 |
| Self-reported knowledge of DS | 504 | 0.010 | 465.40 (6936–861.4) | 0.111 | 0.021 * |
| Knowledge about health claims | 504 | 0.048 | −132.97 (−188.2–−77.8) | −0.223 | 0.001 * |
| Sources of information, reliable | 504 | −0.001 | −390.52 (−1308.5–527.5) | −0.04 | 0.404 |
| Omega-3 fatty acids or fish oil users of DS (profile 1) | 332 | 0.010 | 1010.65 (161.7–1859.6) | 0.112 | 0.020 * |
| Multivitamin or -mineral users of DS (profile 2) | 171 | 0.003 | 552.22 (−158.5–1263) | 0.074 | 0.127 |
| Single-vitamin or -mineral users of DS (profile 3) | 373 | 0.001 | 1132.46 (−640.8–2905.7) | 0.061 | 0.210 |
| Non-vitamin or -mineral users of DS (profile 4) | 96 | 0.008 | 873.66 (73.5–1673.8) | 0.104 | 0.032 * |
| Users of the DS marketed for muscles/joints (profile 5) | 88 | 0.036 | 1908.21 (993.3–2823.1) | 0.194 | 0.001 * |
| Independent Variables | Adjusted Analysis R2 = 0.087 | |||
|---|---|---|---|---|
| n | B (95% KI) | β | Sig. | |
| Gender (0 = women, 1 = men) | 504 | −815.65 (−2352.2–721) | −0.049 | 0.297 |
| Age in years | 504 | 11.81 (−22.7–46.3) | 0.032 | 0.501 |
| Marital status (no partner = 0, partner = 1) | 504 | −768.69 (−1567.2–29.8) | −0.090 | 0.059 # |
| Education (low = 0, high = 1) | 504 | 819.01 (115.8–1522.3) | 0.110 | 0.023 * |
| Self-reported knowledge of DS | 504 | 263.82 (−128–655.7) | 0.063 | 0.186 |
| Knowledge of health claims | 504 | −114.01 (−169.5–−58.5) | −0.193 | 0.001 * |
| Omega-3 fatty acids or fish oil users of DS (profile 1) | 332 | 552.62 (−271.8–1379.1) | 0.062 | 0.189 |
| Non-vitamin or -mineral users of DS (profile 4) | 96 | 283.41 (−516.6–1083.4) | 0.034 | 0.487 |
| Users of the DS marketed for muscles or joints (profile 5) | 88 | 1224.99 (294–2156) | 0.125 | 0.010 * |
| Independent Variables | Unadjusted Analyses | Adjusted Analysis | ||||
|---|---|---|---|---|---|---|
| B | OR (95% CI) | Sig. | B | OR (95% CI) | Sig. | |
| Gender (0 = women, 1 = men) | −0.25 | 0.78 (0.29–2.12) | 0.63 | −0.21 | 0.81 (0.27–2.41) | 0.71 |
| Age in years | 0.01 | 1.01 (0.99–1.04) | 0.43 | 0.01 | 1.01 (0.98–1.03) | 0.60 |
| Marital status (non-partner = 0, partner = 1) | −0.71 | 0.84 (0.47–1.53) | 0.57 | |||
| Education (low = 0, high = 1) | 1.08 | 2.96 (1.72–5.07) | 0.001 * | 0.88 | 2.41 (1.36–4.27) | 0.003 * |
| Employment (not active = 0, active = 1) | −0.06 | 0.94 (0.56–1.58) | 0.81 | |||
| Health professional background (no = 0, yes = 1) | 0.39 | 1.47 (0.80–2.69) | 0.21 | |||
| Number of diagnoses | 0.12 | 1.13 (0.92–1.38) | 0.28 | |||
| Number of medications (low = 0, high = 1) | 0.31 | 1.37 (0.79–2.38) | 0.27 | |||
| BMI, kg/m2 | −0.03 | 0.97 (0.93–1.02) | 0.25 | |||
| Lifestyle factors, health index | 0.13 | 1.14 (0.85–1.53) | 0.38 | |||
| Self-reported health in general | 0.11 | 1.12 (0.89–1.40) | 0.33 | |||
| Self-reported health today | 0.01 | 1.01 (0.99–1.02) | 0.87 | |||
| Self-reported knowledge of DS | 0.70 | 2.02 (1.59–2.56) | 0.001 * | 0.59 | 1.81 (1.40–2.32) | 0.001 * |
| Knowledge about health claims | −0.05 | 0.95 (0.92–0.99 | 0.02 * | −0.07 | 0.94 (0.90–0.98) | 0.004 * |
| Sources of information, reliable | 0.70 | 2.01 (1.16–3.47) | 0.01 * | 0.52 | 1.69 (0.92–3.11) | 0.09 # |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kvæl, L.A.H.; Løchting, I.; Molin, M. Use of Dietary Supplements and Perceived Knowledge among Adults Living with Fibromyalgia in Norway: A Cross-Sectional Study. Nutrients 2022, 14, 5. https://doi.org/10.3390/nu14010005
Kvæl LAH, Løchting I, Molin M. Use of Dietary Supplements and Perceived Knowledge among Adults Living with Fibromyalgia in Norway: A Cross-Sectional Study. Nutrients. 2022; 14(1):5. https://doi.org/10.3390/nu14010005
Chicago/Turabian StyleKvæl, Linda Aimée Hartford, Ida Løchting, and Marianne Molin. 2022. "Use of Dietary Supplements and Perceived Knowledge among Adults Living with Fibromyalgia in Norway: A Cross-Sectional Study" Nutrients 14, no. 1: 5. https://doi.org/10.3390/nu14010005
APA StyleKvæl, L. A. H., Løchting, I., & Molin, M. (2022). Use of Dietary Supplements and Perceived Knowledge among Adults Living with Fibromyalgia in Norway: A Cross-Sectional Study. Nutrients, 14(1), 5. https://doi.org/10.3390/nu14010005






