Nuts: Natural Pleiotropic Nutraceuticals
Abstract
1. Introduction
2. Data Sources and Selection of Studies on Nuts and Human Health
3. Historical Aspects
4. Nutrient Content
5. Nut Consumption and Health Outcomes
5.1. CVD Incidence and Mortality
5.2. Hypertension Incidence and Mortality
5.3. Diabetes Incidence and Mortality
5.4. Cancer Incidence and Mortality
5.5. Brain Health
5.6. All-Cause Mortality
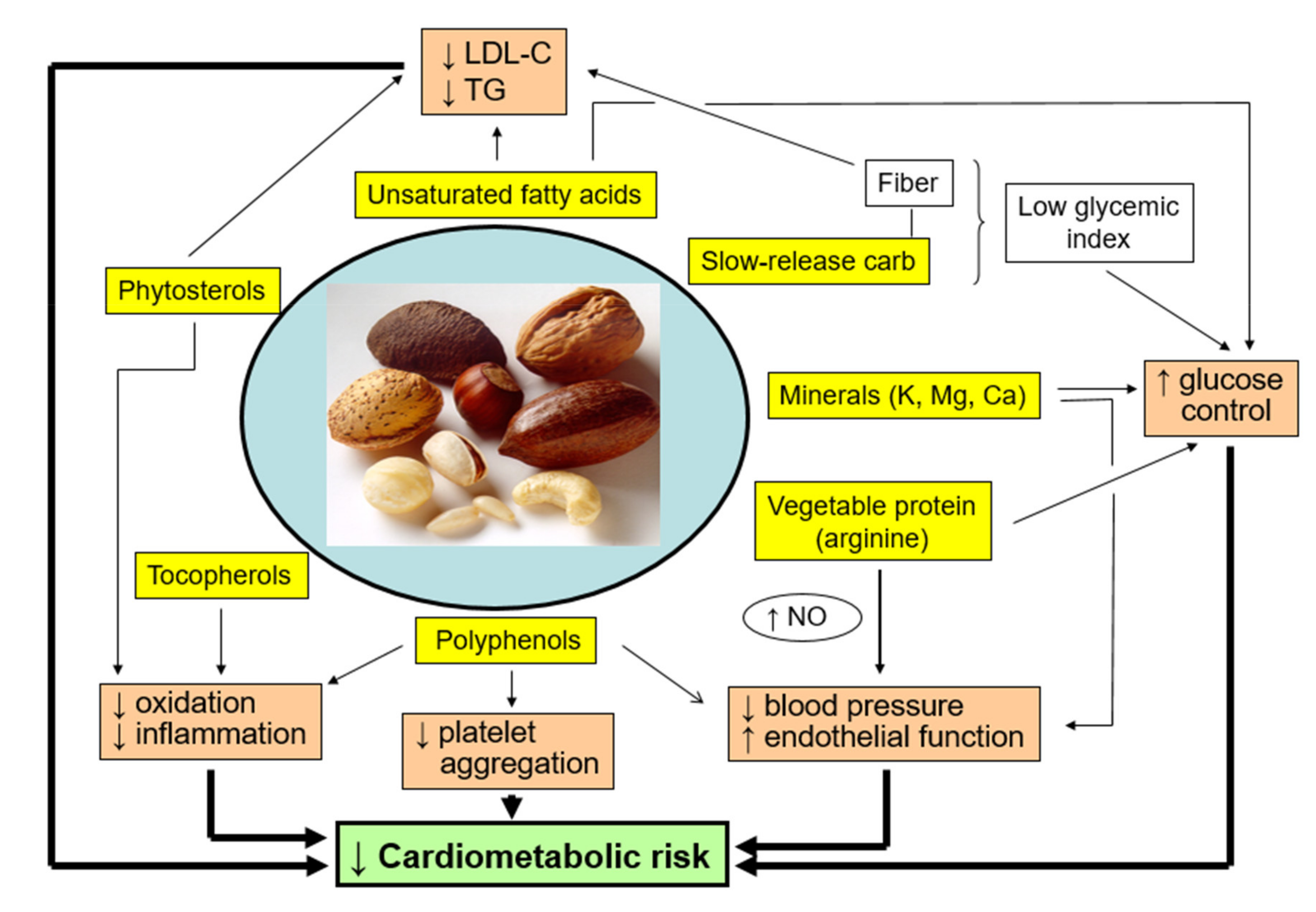
5.7. Intermediate Markers: Adiposity, Lipids, Blood Pressure, Glycemic Control, Endothelial Function, and Inflammation
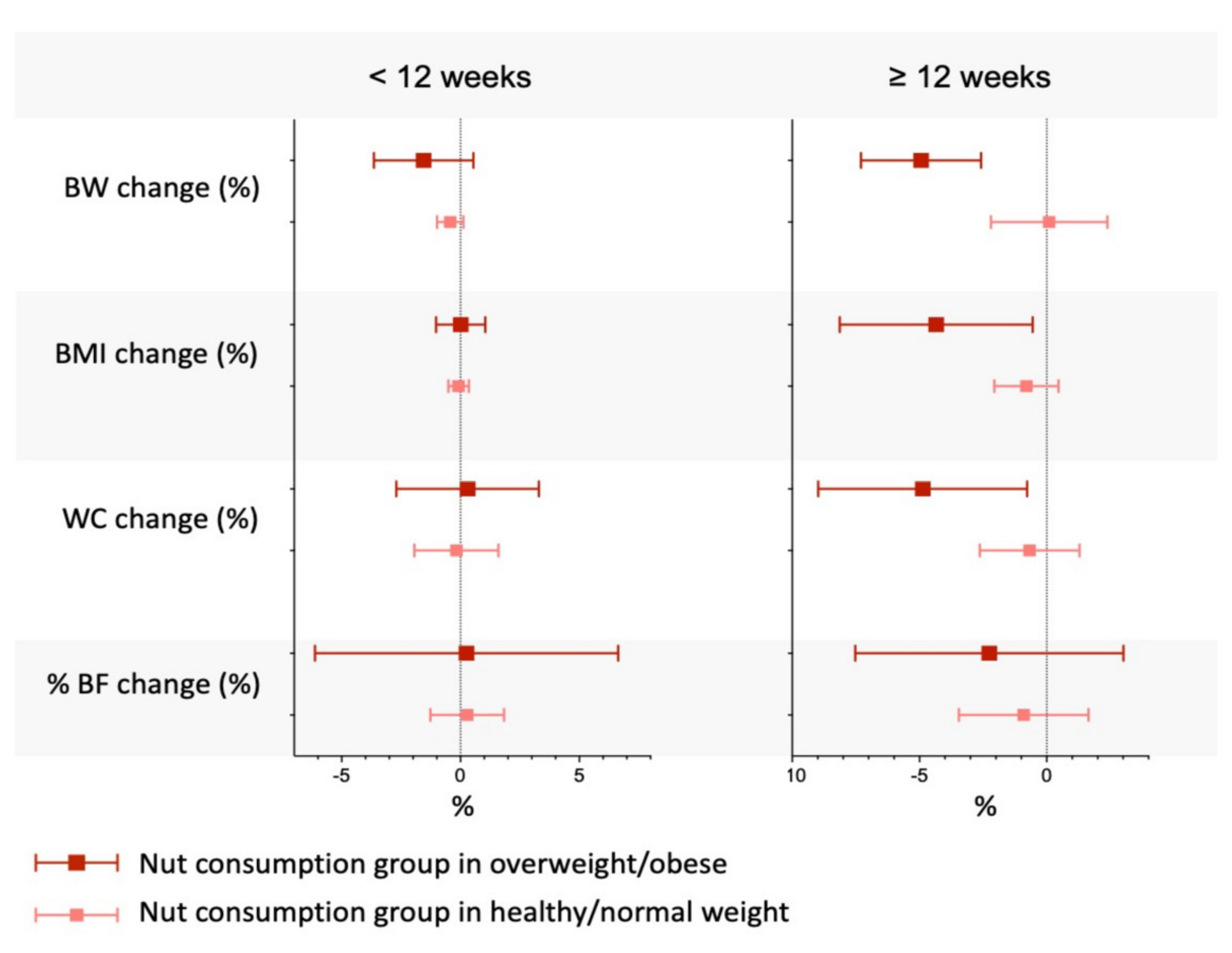
5.7.1. Adiposity
5.7.2. Blood Lipids
5.7.3. Blood Pressure
5.7.4. Glycemic Control
5.7.5. Endothelial Function
5.7.6. Inflammation
5.8. Other Health Outcomes
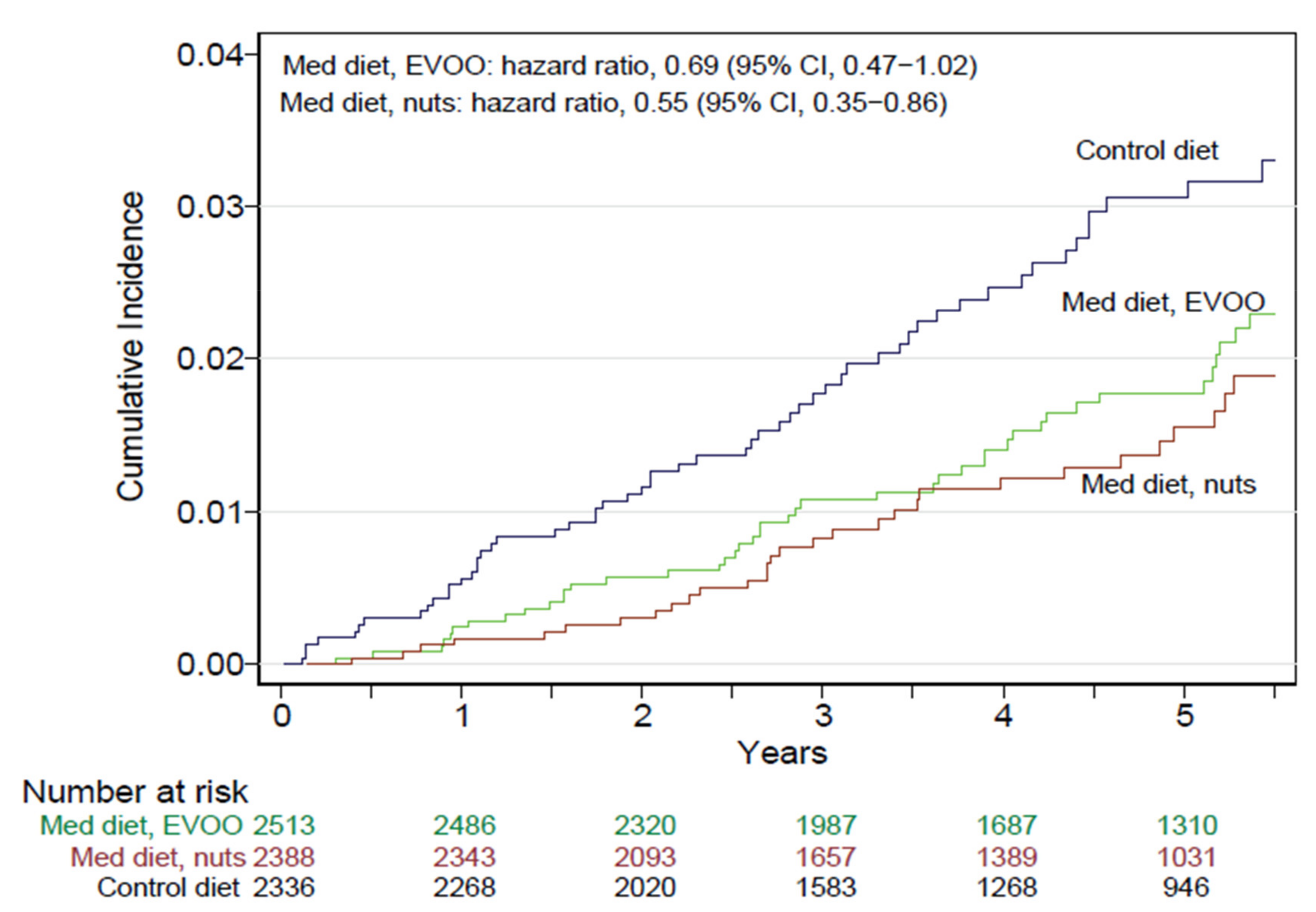
5.9. Health Effects of Nuts in the Predimed Trial
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALA—α-Linolenic Acid |
| BP—Blood Pressure |
| BMI—Body Mass Index |
| CHD—Coronary Heart Disease |
| CI—Confidence Interval |
| CRP—C-Reactive Protein |
| CV—Cardiovascular |
| CVD—Cardiovascular Disease |
| DBP—Diastolic Blood Pressure |
| FMD—Flow-Mediated Dilation |
| HOMA-IR—Homeostasis Model Assessment of Insulin Resistance |
| HPFS—Health Professionals Follow-Up Study |
| HR—Hazard Ratio |
| ICAM—Intercellular Adhesion Molecule |
| IL—Interleukin |
| MetS—Metabolic syndrome |
| MUFA—Monounsaturated Fatty Acids |
| NAFLD—Non-Alcoholic Fatty Liver Disease |
| NHS—Nurses’ Health Study |
| NO—Nitric Oxide |
| OR—Odds ratio |
| PREDIMED—PREvención con DIeta MEDiterránea |
| PUFA—Polyunsaturated Fatty Acids |
| PURE—Prospective Urban and Rural Epidemiology |
| RCT—Randomized Controlled Trial |
| RR—Relative Risk |
| SBP—Systolic Blood Pressure |
| SFA—Saturated Fatty Acids |
| T2D—Type-2 Diabetes Mellitus |
| TNF-α—Tumor-necrosis Factor-α |
| VCAM—Vascular Cell Adhesion Molecule |
| WAHA—Walnuts and Healthy Aging |
| WC—Waist Circumference |
| WMD—Weighted Mean Difference |
References
- Ros, E. Health benefits of nut consumption. Nutrients 2010, 2, 652–683. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.E.; Sabaté, J.; Beeson, W.L.; Strahan, T.M. A possible protective effect of nut consumption on risk of coronary heart disease. The Adventist Health Study. Arch. Intern. Med. 1992, 152, 1416–1424. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Fraser, G.E.; Burke, K.; Knutsen, S.F.; Bennett, H.; Lindsted, K.D. Effects of walnuts on serum lipid levels and blood pressure in normal men. N. Engl. J. Med. 1993, 328, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Eaton, S.B.; Konner, M. Paleolithic nutrition. A consideration of its nature and current implications. N. Engl. J. Med. 1985, 312, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J. Nut consumption, vegetarian diets, ischemic heart disease risk, and all-cause mortality: Evidence from epidemiologic studies. Am. J. Clin. Nutr. 1999, 70, 500S–503S. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.L. Qualified Health Claims: Letter of Enforcement Discretion—Nuts and Coronary Heart Disease (Docket No 02P-0505); Food and Drug Administration, Ed.; Office of Nutritional Products, Labeling and Dietary Supplements: Washington, DC, USA, 2003. Available online: http://wayback.archive-it.org/7993/20171114183724/https://www.fda.gov/Food/IngredientsPackagingLabeling/LabelingNutrition/ucm072926.htm (accessed on 25 July 2021).
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; Van Horn, L.; Greenlund, K.; Daniels, S.; Nichol, G.; Tomaselli, G.F.; et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: The American Heart Association’s strategic Impact Goal through 2020 and beyond. Circulation 2010, 121, 586–613. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 207. [Google Scholar] [CrossRef]
- Del Gobbo, L.C.; Falk, M.C.; Feldman, R.; Lewis, K.; Mozaffarian, D. Effects of tree nuts on blood lipids, apolipoproteins, and blood pressure: Systematic review, meta-analysis, and dose-response of 61 controlled intervention trials. Am. J. Clin. Nutr. 2015, 102, 1347–1356. [Google Scholar] [CrossRef]
- Jackson, C.L.; Hu, F.B. Long-term associations of nut consumption with body weight and obesity. Am. J. Clin. Nutr. 2014, 100, 408S–411S. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Casas-Agustench, P.; Salas-Huetos, A. Cultural and historical aspects of Mediterranean nuts with emphasis on their attributed healthy and nutritional properties. Nutr. Metab. Cardiovasc. Dis. 2011, 21, S1–S6. [Google Scholar] [CrossRef]
- Arya, S.S.; Salve, A.R.; Chauhan, S. Peanuts as functional food: A review. J. Food Sci. Technol. 2016, 53, 31–41. [Google Scholar] [CrossRef]
- Jenab, M.; Sabate, J.; Slimani, N.; Ferrari, P.; Mazuir, M.; Casagrande, C.; Deharveng, G.; Tjonneland, A.; Olsen, A.; Overvad, K.; et al. Consumption and portion sizes of tree nuts, peanuts and seeds in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohorts from 10 European countries. Br. J. Nutr. 2006, 96, S12–S23. [Google Scholar] [CrossRef]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef]
- ARS. FoodData Central; US Department of Agriculture, Agricultural Research Service, Ed.; US Government: Washington, DC, USA, 2019. Available online: http://www.fdc.nal.usda.gov (accessed on 25 July 2021).
- Kornsteiner-Krenn, M.; Wagner, K.H.; Elmadfa, I. Phytosterol content and fatty acid pattern of ten different nut types. Int. J. Vitam. Nutr. Res. 2013, 83, 263–270. [Google Scholar] [CrossRef]
- Moncada, S.; Higgs, A. The L-arginine-nitric oxide pathway. N. Engl. J. Med. 1993, 329, 2002–2012. [Google Scholar] [CrossRef] [PubMed]
- Neveu, V.; Pérez-Jiménez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. In Database; Phenol-Explorer, Ed.; University of Alberta: Alberta, Canada, 2010. [Google Scholar] [CrossRef]
- Ros, E.; Hu, F.B. Consumption of plant seeds and cardiovascular health: Epidemiological and clinical trial evidence. Circulation 2013, 128, 553–565. [Google Scholar] [CrossRef]
- Moreau, R.A.; Nystrom, L.; Whitaker, B.D.; Winkler-Moser, J.K.; Baer, D.J.; Gebauer, S.K.; Hicks, K.B. Phytosterols and their derivatives: Structural diversity, distribution, metabolism, analysis, and health-promoting uses. Prog. Lipid. Res. 2018, 70, 35–61. [Google Scholar] [CrossRef]
- Del Gobbo, L.C.; Falk, M.C.; Feldman, R.; Lewis, K.; Mozaffarian, D. Are Phytosterols Responsible for the Low-Density Lipoprotein-Lowering Effects of Tree Nuts?: A Systematic Review and Meta-Analysis. J. Am. Coll. Cardiol. 2015, 65, 2765–2767. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Karppanen, H.; Karppanen, P.; Mervaala, E. Why and how to implement sodium, potassium, calcium, and magnesium changes in food items and diets? J. Hum. Hypertens. 2005, 19, S10–S19. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gomez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- WHO. Cardiovascular Diseases (CVDs). Available online: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 29 July 2021).
- Becerra-Tomás, N.; Paz-Graniel, I.; Kendall, C.W.C.; Kahleova, H.; Rahelic, D.; Sievenpiper, J.L.; Salas-Salvado, J. Nut consumption and incidence of cardiovascular diseases and cardiovascular disease mortality: A meta-analysis of prospective cohort studies. Nutr. Rev. 2019, 77, 691–709. [Google Scholar] [CrossRef]
- Liu, G.; Guasch-Ferré, M.; Hu, Y.; Li, Y.; Hu, F.B.; Rimm, E.B.; Manson, J.E.; Rexrode, K.M.; Sun, Q. Nut Consumption in Relation to Cardiovascular Disease Incidence and Mortality Among Patients With Diabetes Mellitus. Circ. Res. 2019, 124, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Amba, V.; Murphy, G.; Etemadi, A.; Wang, S.; Abnet, C.C.; Hashemian, M. Nut and Peanut Butter Consumption and Mortality in the National Institutes of Health-AARP Diet and Health Study. Nutrients 2019, 11, 1508. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.J.; Dehghan, M.; Mente, A.; Bangdiwala, S.I.; Ahmed, S.H.; Alhabib, K.F.; Altuntas, Y.; Basiak-Rasala, A.; Dagenais, G.R.; Diaz, R.; et al. Association of nut intake with risk factors, cardiovascular disease, and mortality in 16 countries from 5 continents: Analysis from the Prospective Urban and Rural Epidemiology (PURE) study. Am. J. Clin. Nutr. 2020, 112, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Imran, T.F.; Kim, E.; Buring, J.E.; Lee, I.M.; Gaziano, J.M.; Djousse, L. Nut consumption, risk of cardiovascular mortality, and potential mediating mechanisms: The Women’s Health Study. J. Clin. Lipidol. 2021, 15, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Mohammadifard, N.; Ghaderian, N.; Hassannejad, R.; Sajjadi, F.; Sadeghi, M.; Roohafza, H.; Salas-Salvado, J.; Sarrafzadegan, N. Longitudinal Association of Nut Consumption and the Risk of Cardiovascular Events: A Prospective Cohort Study in the Eastern Mediterranean Region. Front. Nutr. 2020, 7, 610467. [Google Scholar] [CrossRef]
- Yamakawa, M.; Wada, K.; Koda, S.; Uji, T.; Nakashima, Y.; Onuma, S.; Oba, S.; Nagata, C. Associations of total nut and peanut intakes with all-cause and cause-specific mortality in a Japanese community: The Takayama study. Br. J. Nutr. 2021, 1–8. [Google Scholar] [CrossRef]
- Liu, X.; Guasch-Ferré, M.; Drouin-Chartier, J.P.; Tobias, D.K.; Bhupathiraju, S.N.; Rexrode, K.M.; Willett, W.C.; Sun, Q.; Li, Y. Changes in Nut Consumption and Subsequent Cardiovascular Disease Risk Among US Men and Women: 3 Large Prospective Cohort Studies. J. Am. Heart Assoc. 2020, 9, e013877. [Google Scholar] [CrossRef]
- Meier, T.; Grafe, K.; Senn, F.; Sur, P.; Stangl, G.I.; Dawczynski, C.; Marz, W.; Kleber, M.E.; Lorkowski, S. Cardiovascular mortality attributable to dietary risk factors in 51 countries in the WHO European Region from 1990 to 2016: A systematic analysis of the Global Burden of Disease Study. Eur. J. Epidemiol. 2019, 34, 37–55. [Google Scholar] [CrossRef] [PubMed]
- Sisa, I.; Abeya-Gilardon, E.; Fisberg, R.M.; Jackson, M.D.; Mangialavori, G.L.; Sichieri, R.; Cudhea, F.; Bannuru, R.R.; Ruthazer, R.; Mozaffarian, D.; et al. Impact of diet on CVD and diabetes mortality in Latin America and the Caribbean: A comparative risk assessment analysis. Public Health Nutr. 2021, 24, 2577–2591. [Google Scholar] [CrossRef]
- Kim, Y.; Keogh, J.; Clifton, P.M. Nuts and Cardio-Metabolic Disease: A Review of Meta-Analyses. Nutrients 2018, 10, 1935. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knuppel, S.; Iqbal, K.; Andriolo, V.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food Groups and Risk of Hypertension: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Adv. Nutr. 2017, 8, 793–803. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G.; Lampousi, A.M.; Knuppel, S.; Iqbal, K.; Schwedhelm, C.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2017, 32, 363–375. [Google Scholar] [CrossRef]
- Becerra-Tomás, N.; Paz-Graniel, I.; Hernández-Alonso, P.; Jenkins, D.J.A.; Kendall, C.W.C.; Sievenpiper, J.L.; Salas-Salvadó, J. Nut consumption and type 2 diabetes risk: A systematic review and meta-analysis of observational studies. Am. J. Clin. Nutr. 2021, 113, 960–971. [Google Scholar] [CrossRef]
- Pan, A.; Sun, Q.; Manson, J.E.; Willett, W.C.; Hu, F.B. Walnut consumption is associated with lower risk of type 2 diabetes in women. J. Nutr. 2013, 143, 512–518. [Google Scholar] [CrossRef]
- Arab, L.; Dhaliwal, S.K.; Martin, C.J.; Larios, A.D.; Jackson, N.J.; Elashoff, D. Association between walnut consumption and diabetes risk in NHANES. Diabetes Metab. Res. Rev. 2018, 34, e3031. [Google Scholar] [CrossRef] [PubMed]
- Ros, E.; Izquierdo-Pulido, M.; Sala-Vila, A. Beneficial effects of walnut consumption on human health: Role of micronutrients. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 498–504. [Google Scholar] [CrossRef] [PubMed]
- WHO. Cancer Tomorrow. Available online: https://gco.iarc.fr/tomorrow/en (accessed on 29 July 2021).
- Islami, F.; Goding Sauer, A.; Miller, K.D.; Siegel, R.L.; Fedewa, S.A.; Jacobs, E.J.; McCullough, M.L.; Patel, A.V.; Ma, J.; Soerjomataram, I.; et al. Proportion and number of cancer cases and deaths attributable to potentially modifiable risk factors in the United States. CA Cancer J. Clin. 2018, 68, 31–54. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1063. [Google Scholar] [CrossRef] [PubMed]
- Naghshi, S.; Sadeghian, M.; Nasiri, M.; Mobarak, S.; Asadi, M.; Sadeghi, O. Association of Total Nut, Tree Nut, Peanut, and Peanut Butter Consumption with Cancer Incidence and Mortality: A Comprehensive Systematic Review and Dose-Response Meta-Analysis of Observational Studies. Adv. Nutr. 2021, 12, 793–808. [Google Scholar] [CrossRef]
- NIPHE. Dutch Food Composition Database. Available online: https://www.rivm.nl/en/dutch-food-composition-database (accessed on 29 July 2021).
- Berkey, C.S.; Tamimi, R.M.; Willett, W.C.; Rosner, B.; Hickey, M.; Toriola, A.T.; Frazier, A.L.; Colditz, G.A. Adolescent alcohol, nuts, and fiber: Combined effects on benign breast disease risk in young women. NPJ Breast Cancer 2020, 6, 61. [Google Scholar] [CrossRef]
- Zhang, D.; Dai, C.; Zhou, L.; Li, Y.; Liu, K.; Deng, Y.J.; Li, N.; Zheng, Y.; Hao, Q.; Yang, S.; et al. Meta-analysis of the association between nut consumption and the risks of cancer incidence and cancer-specific mortality. Aging 2020, 12, 10772–10794. [Google Scholar] [CrossRef]
- Arvanitakis, Z.; Shah, R.C.; Bennett, D.A. Diagnosis and Management of Dementia: Review. JAMA 2019, 322, 1589–1599. [Google Scholar] [CrossRef] [PubMed]
- Kivipelto, M.; Mangialasche, F.; Ngandu, T. Lifestyle interventions to prevent cognitive impairment, dementia and Alzheimer disease. Nat. Rev. Neurol. 2018, 14, 653–666. [Google Scholar] [CrossRef]
- Scarmeas, N.; Anastasiou, C.A.; Yannakoulia, M. Nutrition and prevention of cognitive impairment. Lancet Neurol. 2018, 17, 1006–1015. [Google Scholar] [CrossRef]
- Mecocci, P.; Boccardi, V.; Cecchetti, R.; Bastiani, P.; Scamosci, M.; Ruggiero, C.; Baroni, M. A Long Journey into Aging, Brain Aging, and Alzheimer’s Disease Following the Oxidative Stress Tracks. J. Alzheimers Dis. 2018, 62, 1319–1335. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.Y.; Snyder, P.J.; Wu, W.C.; Zhang, M.; Echeverria, A.; Alber, J. Pathophysiologic relationship between Alzheimer’s disease, cerebrovascular disease, and cardiovascular risk: A review and synthesis. Alzheimers Dement. 2017, 7, 69–87. [Google Scholar] [CrossRef]
- Theodore, L.E.; Kellow, N.J.; McNeil, E.A.; Close, E.O.; Coad, E.G.; Cardoso, B.R. Nut Consumption for Cognitive Performance: A Systematic Review. Adv. Nutr. 2021, 12, 777–792. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A.; Chauhan, V. Beneficial Effects of Walnuts on Cognition and Brain Health. Nutrients 2020, 12, 550. [Google Scholar] [CrossRef]
- Jiang, Y.W.; Sheng, L.T.; Feng, L.; Pan, A.; Koh, W.P. Consumption of dietary nuts in midlife and risk of cognitive impairment in late-life: The Singapore Chinese Health Study. Age Ageing 2021, 50, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Leonard, B.E. Inflammation and depression: A causal or coincidental link to the pathophysiology? Acta Neuropsychiatr. 2018, 30, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Vaccarino, V.; Badimón, L.; Bremner, J.D.; Cenko, E.; Cubedo, J.; Dorobantu, M.; Duncker, D.J.; Koller, A.; Manfrini, O.; Milicic, D.; et al. Depression and coronary heart disease: 2018 position paper of the ESC working group on coronary pathophysiology and microcirculation. Eur. Heart J. 2020, 41, 1687–1696. [Google Scholar] [CrossRef]
- Su, Q.; Yu, B.; He, H.; Zhang, Q.; Meng, G.; Wu, H.; Du, H.; Liu, L.; Shi, H.; Xia, Y.; et al. Nut Consumption Is Associated with Depressive Symptoms among Chinese Adults. Depress. Anxiety 2016, 33, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Arab, L.; Guo, R.; Elashoff, D. Lower Depression Scores among Walnut Consumers in NHANES. Nutrients 2019, 11, 275. [Google Scholar] [CrossRef]
- Elstgeest, L.E.M.; Visser, M.; Penninx, B.; Colpo, M.; Bandinelli, S.; Brouwer, I.A. Bidirectional associations between food groups and depressive symptoms: Longitudinal findings from the Invecchiare in Chianti (InCHIANTI) study. Br. J. Nutr. 2019, 121, 439–450. [Google Scholar] [CrossRef]
- Chen, G.C.; Zhang, R.; Martínez-González, M.A.; Zhang, Z.L.; Bonaccio, M.; van Dam, R.M.; Qin, L.Q. Nut consumption in relation to all-cause and cause-specific mortality: A meta-analysis 18 prospective studies. Food Funct. 2017, 8, 3893–3905. [Google Scholar] [CrossRef] [PubMed]
- Jo, G.; Oh, H.; Singh, G.M.; Park, D.; Shin, M.J. Impact of dietary risk factors on cardiometabolic and cancer mortality burden among Korean adults: Results from nationally representative repeated cross-sectional surveys 1998–2016. Nutr. Res. Pract. 2020, 14, 384–400. [Google Scholar] [CrossRef]
- Fernández-Rodríguez, R.; Mesas, A.E.; Garrido-Miguel, M.; Martínez-Ortega, I.A.; Jiménez-López, E.; Martínez-Vizcaino, V. The Relationship of Tree Nuts and Peanuts with Adiposity Parameters: A Systematic Review and Network Meta-Analysis. Nutrients 2021, 13, 2251. [Google Scholar] [CrossRef] [PubMed]
- Guarneiri, L.L.; Cooper, J.A. Intake of Nuts or Nut Products Does Not Lead to Weight Gain, Independent of Dietary Substitution Instructions: A Systematic Review and Meta-Analysis of Randomized Trials. Adv. Nutr. 2021, 12, 384–401. [Google Scholar] [CrossRef]
- Liu, X.; Li, Y.; Guasch-Ferré, M.; Willett, W.C.; Drouin-Chartier, J.P.; Bhupathiraju, S.N.; Tobias, D.K. Changes in nut consumption influence long-term weight change in US men and women. BMJ Nutr. Prev. Health 2019, 2, 90–99. [Google Scholar] [CrossRef]
- Sabaté, J.; Oda, K.; Ros, E. Nut consumption and blood lipid levels: A pooled analysis of 25 intervention trials. Arch. Intern. Med. 2010, 170, 821–827. [Google Scholar] [CrossRef]
- Cofán, M.; Ros, E. Use of Plant Sterol and Stanol Fortified Foods in Clinical Practice. Curr. Med. Chem. 2019, 26, 6691–6703. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G.; Iqbal, K.; Schwedhelm, C.; Boeing, H. Food groups and intermediate disease markers: A systematic review and network meta-analysis of randomized trials. Am. J. Clin. Nutr. 2018, 108, 576–586. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Li, J.; Hu, F.B.; Salas-Salvadó, J.; Tobias, D.K. Effects of walnut consumption on blood lipids and other cardiovascular risk factors: An updated meta-analysis and systematic review of controlled trials. Am. J. Clin. Nutr. 2018, 108, 174–187. [Google Scholar] [CrossRef]
- Asbaghi, O.; Moodi, V.; Hadi, A.; Eslampour, E.; Shirinbakhshmasoleh, M.; Ghaedi, E.; Miraghajani, M. The effect of almond intake on lipid profile: A systematic review and meta-analysis of randomized controlled trials. Food Funct. 2021, 12, 1882–1896. [Google Scholar] [CrossRef] [PubMed]
- Ghanavati, M.; Rahmani, J.; Clark, C.C.T.; Hosseinabadi, S.M.; Rahimlou, M. Pistachios and cardiometabolic risk factors: A systematic review and meta-analysis of randomized controlled clinical trials. Complement. Ther. Med. 2020, 52, 102513. [Google Scholar] [CrossRef] [PubMed]
- Perna, S.; Giacosa, A.; Bonitta, G.; Bologna, C.; Isu, A.; Guido, D.; Rondanelli, M. Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis. Nutrients 2016, 8, 747. [Google Scholar] [CrossRef] [PubMed]
- Jalali, M.; Karamizadeh, M.; Ferns, G.A.; Zare, M.; Moosavian, S.P.; Akbarzadeh, M. The effects of cashew nut intake on lipid profile and blood pressure: A systematic review and meta-analysis of randomized controlled trials. Complement. Ther. Med. 2020, 50, 102387. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Hui, S.; Wang, B.; Kaliannan, K.; Guo, X.; Liang, L. Comparative effects of different types of tree nut consumption on blood lipids: A network meta-analysis of clinical trials. Am. J. Clin. Nutr. 2020, 111, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Mohammadifard, N.; Salehi-Abargouei, A.; Salas-Salvado, J.; Guasch-Ferre, M.; Humphries, K.; Sarrafzadegan, N. The effect of tree nut, peanut, and soy nut consumption on blood pressure: A systematic review and meta-analysis of randomized controlled clinical trials. Am. J. Clin. Nutr. 2015, 101, 966–982. [Google Scholar] [CrossRef]
- Eslampour, E.; Asbaghi, O.; Hadi, A.; Abedi, S.; Ghaedi, E.; Lazaridi, A.V.; Miraghajani, M. The effect of almond intake on blood pressure: A systematic review and meta-analysis of randomized controlled trials. Complement. Ther. Med. 2020, 50, 102399. [Google Scholar] [CrossRef]
- Asbaghi, O.; Hadi, A.; Campbell, M.S.; Venkatakrishnan, K.; Ghaedi, E. Effects of pistachios on anthropometric indices, inflammatory markers, endothelial function and blood pressure in adults: A systematic review and meta-analysis of randomised controlled trials. Br. J. Nutr. 2021, 126, 718–729. [Google Scholar] [CrossRef]
- Domènech, M.; Serra-Mir, M.; Roth, I.; Freitas-Simoes, T.; Valls-Pedret, C.; Cofán, M.; López, A.; Sala-Vila, A.; Calvo, C.; Rajaram, S.; et al. Effect of a Walnut Diet on Office and 24-Hour Ambulatory Blood Pressure in Elderly Individuals. Hypertension 2019, 73, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Kendall, C.W.; Josse, A.R.; Esfahani, A.; Jenkins, D.J. Nuts, metabolic syndrome and diabetes. Br. J. Nutr. 2010, 104, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Dreher, M.L. A Comprehensive Review of Almond Clinical Trials on Weight Measures, Metabolic Health Biomarkers and Outcomes, and the Gut Microbiota. Nutrients 2021, 13, 1968. [Google Scholar] [CrossRef] [PubMed]
- Tindall, A.M.; Johnston, E.A.; Kris-Etherton, P.M.; Petersen, K.S. The effect of nuts on markers of glycemic control: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2019, 109, 297–314. [Google Scholar] [CrossRef]
- Neale, E.P.; Guan, V.; Tapsell, L.C.; Probst, Y.C. Effect of walnut consumption on markers of blood glucose control: A systematic review and meta-analysis. Br. J. Nutr. 2020, 124, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Matsuzawa, Y.; Kwon, T.G.; Lennon, R.J.; Lerman, L.O.; Lerman, A. Prognostic Value of Flow-Mediated Vasodilation in Brachial Artery and Fingertip Artery for Cardiovascular Events: A Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2015, 4, e002270. [Google Scholar] [CrossRef] [PubMed]
- Deanfield, J.E.; Halcox, J.P.; Rabelink, T.J. Endothelial function and dysfunction: Testing and clinical relevance. Circulation 2007, 115, 1285–1295. [Google Scholar] [CrossRef]
- Neale, E.P.; Tapsell, L.C.; Guan, V.; Batterham, M.J. The effect of nut consumption on markers of inflammation and endothelial function: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2017, 7, e016863. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Huang, W.; Peng, C.; Zhang, J.; Wong, C.; Kim, J.H.; Yeoh, E.K.; Su, X. Effect of nut consumption on vascular endothelial function: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. 2018, 37, 831–839. [Google Scholar] [CrossRef]
- Dikariyanto, V.; Smith, L.; Francis, L.; Robertson, M.; Kusaslan, E.; O’Callaghan-Latham, M.; Palanche, C.; D’Annibale, M.; Christodoulou, D.; Basty, N.; et al. Snacking on whole almonds for 6 weeks improves endothelial function and lowers LDL cholesterol but does not affect liver fat and other cardiometabolic risk factors in healthy adults: The ATTIS study, a randomized controlled trial. Am. J. Clin. Nutr. 2020, 111, 1178–1189. [Google Scholar] [CrossRef]
- Morgillo, S.; Hill, A.M.; Coates, A.M. The Effects of Nut Consumption on Vascular Function. Nutrients 2019, 11, 116. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Crea, F. Clinical implications of inflammation for cardiovascular primary prevention. Eur. Heart J. 2010, 31, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Inflammation in Atherosclerosis-No Longer a Theory. Clin. Chem. 2021, 67, 131–142. [Google Scholar] [CrossRef]
- Xiao, Y.; Xia, J.; Ke, Y.; Cheng, J.; Yuan, J.; Wu, S.; Lv, Z.; Huang, S.; Kim, J.H.; Wong, S.Y.; et al. Effects of nut consumption on selected inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Nutrition 2018, 54, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Cofan, M.; Rajaram, S.; Sala-Vila, A.; Valls-Pedret, C.; Serra-Mir, M.; Roth, I.; Freitas-Simoes, T.M.; Bitok, E.; Sabate, J.; Ros, E. Effects of 2-Year Walnut-Supplemented Diet on Inflammatory Biomarkers. J. Am. Coll. Cardiol. 2020, 76, 2282–2284. [Google Scholar] [CrossRef]
- Tsai, C.J.; Leitzmann, M.F.; Hu, F.B.; Willett, W.C.; Giovannucci, E.L. Frequent nut consumption and decreased risk of cholecystectomy in women. Am. J. Clin. Nutr. 2004, 80, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.J.; Leitzmann, M.F.; Hu, F.B.; Willett, W.C.; Giovannucci, E.L. A prospective cohort study of nut consumption and the risk of gallstone disease in men. Am. J. Epidemiol. 2004, 160, 961–968. [Google Scholar] [CrossRef]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr.; et al. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Perez-Martinez, P.; Mikhailidis, D.P.; Athyros, V.G.; Bullo, M.; Couture, P.; Covas, M.I.; de Koning, L.; Delgado-Lista, J.; Diaz-Lopez, A.; Drevon, C.A.; et al. Lifestyle recommendations for the prevention and management of metabolic syndrome: An international panel recommendation. Nutr. Rev. 2017, 75, 307–326. [Google Scholar] [CrossRef]
- Blanco Mejia, S.; Kendall, C.W.; Viguiliouk, E.; Augustin, L.S.; Ha, V.; Cozma, A.I.; Mirrahimi, A.; Maroleanu, A.; Chiavaroli, L.; Leiter, L.A.; et al. Effect of tree nuts on metabolic syndrome criteria: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2014, 4, e004660. [Google Scholar] [CrossRef]
- Al Abdrabalnabi, A.; Rajaram, S.; Bitok, E.; Oda, K.; Beeson, W.L.; Kaur, A.; Cofán, M.; Serra-Mir, M.; Roth, I.; Ros, E.; et al. Effects of Supplementing the Usual Diet with a Daily Dose of Walnuts for Two Years on Metabolic Syndrome and Its Components in an Elderly Cohort. Nutrients 2020, 12, 451. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.; Anstee, Q.M.; Marietti, M.; Hardy, T.; Henry, L.; Eslam, M.; George, J.; Bugianesi, E. Global burden of NAFLD and NASH: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 11–20. [Google Scholar] [CrossRef]
- Plaz Torres, M.C.; Bodini, G.; Furnari, M.; Marabotto, E.; Zentilin, P.; Giannini, E.G. Nuts and Non-Alcoholic Fatty Liver Disease: Are Nuts Safe for Patients with Fatty Liver Disease? Nutrients 2020, 12, 3363. [Google Scholar] [CrossRef] [PubMed]
- Arias-Fernández, L.; Machado-Fragua, M.D.; Graciani, A.; Guallar-Castillón, P.; Banegas, J.R.; Rodríguez-Artalejo, F.; Lana, A.; López-García, E. Prospective Association Between Nut Consumption and Physical Function in Older Men and Women. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 74, 1091–1097. [Google Scholar] [CrossRef]
- Freitas-Simoes, T.M.; Wagner, M.; Samieri, C.; Sala-Vila, A.; Grodstein, F. Consumption of Nuts at Midlife and Healthy Aging in Women. J. Aging Res. 2020, 2020, 5651737. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Garach, A.; Garcia-Fontana, B.; Munoz-Torres, M. Nutrients and Dietary Patterns Related to Osteoporosis. Nutrients 2020, 12, 1986. [Google Scholar] [CrossRef] [PubMed]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196. [Google Scholar] [CrossRef]
- Griel, A.E.; Kris-Etherton, P.M.; Hilpert, K.F.; Zhao, G.; West, S.G.; Corwin, R.L. An increase in dietary n-3 fatty acids decreases a marker of bone resorption in humans. Nutr. J. 2007, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Baskaran, S.; Parekh, N.; Cho, C.L.; Henkel, R.; Vij, S.; Arafa, M.; Panner Selvam, M.K.; Shah, R. Male infertility. Lancet 2021, 397, 319–333. [Google Scholar] [CrossRef]
- Salas-Huetos, A.; James, E.R.; Aston, K.I.; Jenkins, T.G.; Carrell, D.T. Diet and sperm quality: Nutrients, foods and dietary patterns. Reprod. Biol. 2019, 19, 219–224. [Google Scholar] [CrossRef]
- Robbins, W.A.; Xun, L.; FitzGerald, L.Z.; Esguerra, S.; Henning, S.M.; Carpenter, C.L. Walnuts improve semen quality in men consuming a Western-style diet: Randomized control dietary intervention trial. Biol. Reprod. 2012, 87, 101. [Google Scholar] [CrossRef] [PubMed]
- Salas-Huetos, A.; Moraleda, R.; Giardina, S.; Antón, E.; Blanco, J.; Salas-Salvadó, J.; Bulló, M. Effect of nut consumption on semen quality and functionality in healthy men consuming a Western-style diet: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.S.; Walter, E.E. Erectile Dysfunction: An Umbrella Review of Meta-Analyses of Risk-Factors, Treatment, and Prevalence Outcomes. J. Sex. Med. 2019, 16, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Ostfeld, R.J.; Allen, K.E.; Aspry, K.; Brandt, E.J.; Spitz, A.; Liberman, J.; Belardo, D.; O’Keefe, J.H.; Aggarwal, M.; Miller, M.; et al. Vasculogenic Erectile Dysfunction: The Impact of Diet and Lifestyle. Am. J. Med. 2021, 134, 310–316. [Google Scholar] [CrossRef]
- Salas-Huetos, A.; Muralidharan, J.; Galie, S.; Salas-Salvado, J.; Bullo, M. Effect of Nut Consumption on Erectile and Sexual Function in Healthy Males: A Secondary Outcome Analysis of the FERTINUTS Randomized Controlled Trial. Nutrients 2019, 11, 1372. [Google Scholar] [CrossRef]
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef]
- Lamuel-Raventos, R.M.; Onge, M.S. Prebiotic nut compounds and human microbiota. Crit. Rev. Food Sci. Nutr. 2017, 57, 3154–3163. [Google Scholar] [CrossRef] [PubMed]
- Creedon, A.C.; Hung, E.S.; Berry, S.E.; Whelan, K. Nuts and their Effect on Gut Microbiota, Gut Function and Symptoms in Adults: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2020, 12, 2347. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Canela, M.; Estruch, R.; Corella, D.; Salas-Salvado, J.; Martinez-Gonzalez, M.A. Association of Mediterranean diet with peripheral artery disease: The PREDIMED randomized trial. JAMA 2014, 311, 415–417. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Bulló, M.; Estruch, R.; Ros, E.; Covas, M.I.; Ibarrola-Jurado, N.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; et al. Prevention of diabetes with Mediterranean diets: A subgroup analysis of a randomized trial. Ann. Intern. Med. 2014, 160, 1–10. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Toledo, E.; Arós, F.; Fiol, M.; Corella, D.; Salas-Salvadó, J.; Ros, E.; Covas, M.I.; Fernández-Crehuet, J.; Lapetra, J.; et al. Extravirgin olive oil consumption reduces risk of atrial fibrillation: The PREDIMED (Prevencion con Dieta Mediterranea) trial. Circulation 2014, 130, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, A.; Martínez-González, M.A.; Alonso-Gómez, A.; Rekondo, J.; Salas-Salvadó, J.; Corella, D.; Ros, E.; Fitó, M.; Estruch, R.; Lapetra, J.; et al. Mediterranean diet and risk of heart failure: Results from the PREDIMED randomized controlled trial. Eur. J. Heart Fail. 2017, 19, 1179–1185. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Bulló, M.; Martínez-González, M.A.; Ros, E.; Corella, D.; Estruch, R.; Fitó, M.; Arós, F.; Warnberg, J.; Fiol, M.; et al. Frequency of nut consumption and mortality risk in the PREDIMED nutrition intervention trial. BMC Med. 2013, 11, 164. [Google Scholar] [CrossRef]
- Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Fitó, M.; Chiva-Blanch, G.; Fiol, M.; Gómez-Gracia, E.; Arós, F.; Lapetra, J.; et al. Effect of a high-fat Mediterranean diet on bodyweight and waist circumference: A prespecified secondary outcomes analysis of the PREDIMED randomised controlled trial. Lancet Diabetes Endocrinol. 2019, 7, e6–e17. [Google Scholar] [CrossRef]
- Domènech, M.; Roman, P.; Lapetra, J.; García de la Corte, F.J.; Sala-Vila, A.; de la Torre, R.; Corella, D.; Salas-Salvadó, J.; Ruiz-Gutiérrez, V.; Lamuela-Raventós, R.M.; et al. Mediterranean diet reduces 24-hour ambulatory blood pressure, blood glucose, and lipids: One-year randomized, clinical trial. Hypertension 2014, 64, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Sala-Vila, A.; Romero-Mamani, E.S.; Gilabert, R.; Núñez, I.; de la Torre, R.; Corella, D.; Ruiz-Gutiérrez, V.; López-Sabater, M.C.; Pintó, X.; Rekondo, J.; et al. Changes in ultrasound-assessed carotid intima-media thickness and plaque with a Mediterranean diet: A substudy of the PREDIMED trial. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; de la Torre, R.; Martínez-González, M.A.; Martínez-Lapiscina, E.H.; Fitó, M.; Pérez-Heras, A.; Salas-Salvadó, J.; et al. Mediterranean Diet and Age-Related Cognitive Decline: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 1094–1103. [Google Scholar] [CrossRef]
- Ros, E. Eat Nuts, Live Longer. J. Am. Coll. Cardiol. 2017, 70, 2533–2535. [Google Scholar] [CrossRef] [PubMed]
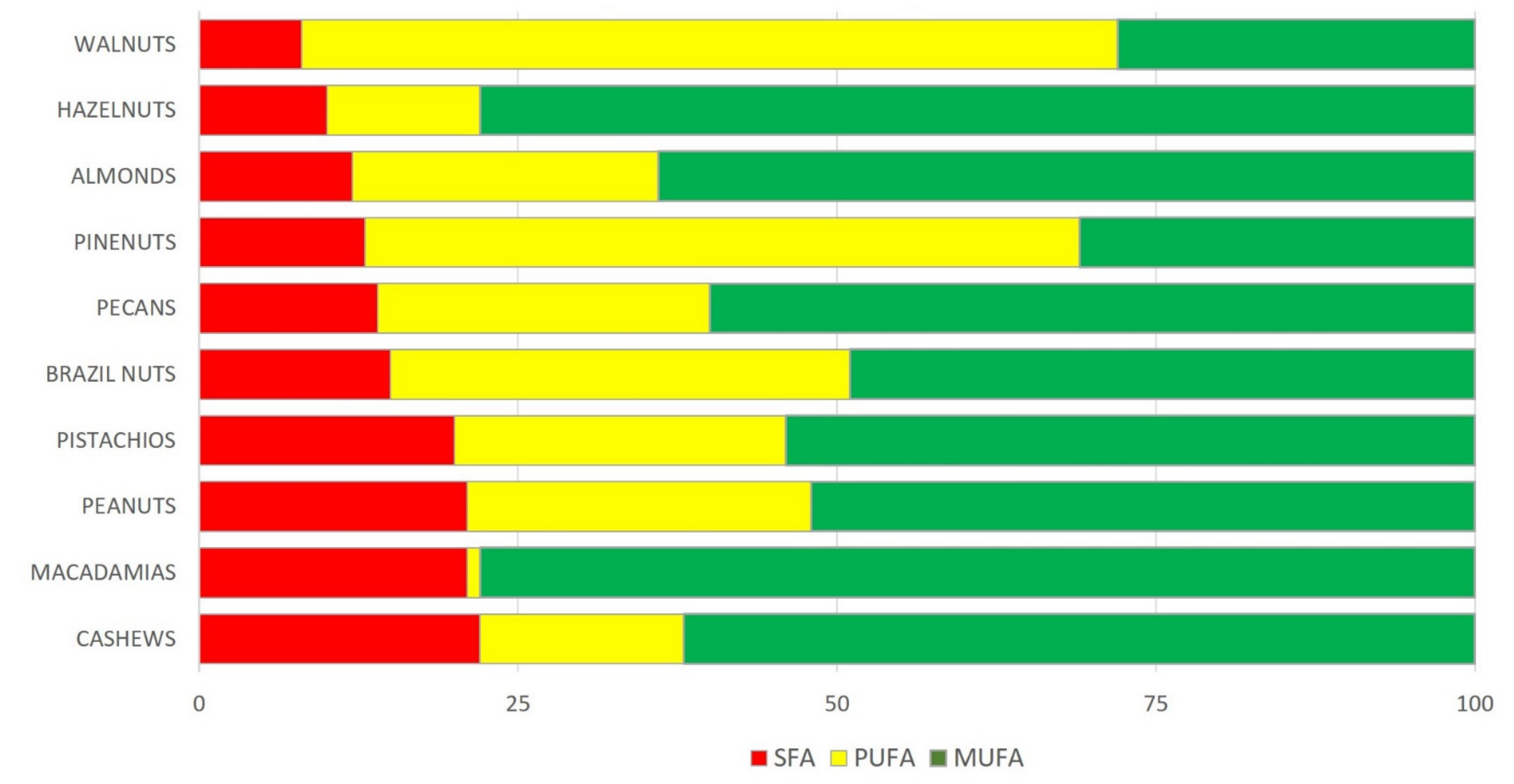

| Nuts |
Energy (kJ) |
Protein (g) |
Fiber (g) |
Fat (g) |
SFA (g) |
MUFA (g) |
PUFA (g) |
LA (g) |
ALA (g) |
Phytosterols (g) |
|---|---|---|---|---|---|---|---|---|---|---|
| Almonds | 2409 | 21.1 | 12.5 | 49.9 | 3.9 | 31.5 | 12.2 | 12.2 | 0.00 | 162 |
| Brazil nuts (dried) | 2743 | 14.3 | 7.5 | 66.4 | 15.1 | 24.5 | 24.4 | 20.5 | 0.05 | 72 |
| Cashews | 2401 | 15.3 | 3 | 46.4 | 9.2 | 27.3 | 7.8 | 7.7 | 0.15 | 120 |
| Hazelnuts | 2669 | 15.0 | 9.7 | 60.8 | 4.5 | 45.7 | 7.9 | 7.8 | 0.09 | 115 |
| Macadamias | 2995 | 7.9 | 8.0 | 76 | 11.9 | 58.9 | 1.4 | 1.3 | 0.21 | 119 |
| Peanuts | 2372 | 26 | 8.5 | 49.2 | 6.2 | 24.4 | 15.6 | 15.6 | 0.00 | 126 |
| Pecans | 2891 | 9.2 | 9.6 | 72.0 | 6.2 | 40.8 | 21.6 | 20.6 | 1.00 | 113 |
| Pine nuts (dried) | 2816 | 13.7 | 3.7 | 68.4 | 4.9 | 18.8 | 34.1 | 33.2 | 0.16 | 120 |
| Pistachios | 2430 | 20.6 | 10.0 | 47 | 5.4 | 25.0 | 14.0 | 13.2 | 0.25 | 272 |
| Walnuts | 2738 | 15.2 | 6.7 | 65.2 | 6.1 | 8.9 | 47.2 | 38.1 | 9.08 | 143 |
| Nuts |
Folate (µg) |
Calcium (mg) |
Magnesium (mg) |
Sodium (mg) |
Potassium (mg) |
Polyphenols (mg) |
|---|---|---|---|---|---|---|
| Almonds | 44 | 269 | 270 | 1 | 733 | 287 |
| Brazil nuts | 22 | 160 | 376 | 3 | 659 | 244 |
| Cashews | 69 | 45 | 260 | 16 | 565 | 233 |
| Hazelnuts | 113 | 114 | 163 | 0 | 680 | 671 |
| Macadamias | 10 | 70 | 118 | 4 | 363 | 126 |
| Peanuts | 240 | 92 | 168 | 18 | 705 | 406 |
| Pecans | 22 | 70 | 121 | 0 | 410 | 1284 |
| Pine nuts | 34 | 16 | 251 | 2 | 597 | 58 |
| Pistachios | 49 | 104 | 106 | 6 | 977 | 1420 |
| Walnuts | 98 | 98 | 158 | 2 | 441 | 1579 |
| Disease/Factor | Association | Level of Evidence |
|---|---|---|
| Epidemiologic studies | ||
| Cardiovascular disease | Reduction | ++ |
| Coronary heart disease | Reduction | ++ |
| Stroke | No change//reduction | +/− |
| Heart failure | No change/reduction | +/− |
| Atrial fibrillation | Reduction | + |
| Hypertension | Reduction | + |
| Diabetes | No change/reduction | +/− |
| Cognitive dysfunction | Improvement | + |
| Depression | No change/reduction | +/− |
| Cancer | Reduction | ++ |
| Obesity | No change/reduction | ++ |
| All-cause mortality | Reduction | ++ |
| Randomized clinical trials | ||
| Blood lipid profile | ||
| Total cholesterol | Reduction * | ++ |
| LDL-cholesterol | Reduction * | ++ |
| HDL-cholesterol | No change | ++ |
| Triglycerides | Reduction * | ++ |
| Insulin sensitivity | No change/increase * | + |
| Diabetes control | Improvement | + |
| Blood pressure | No change/reduction * | +/− |
| Inflammation | No change/reduction * | + |
| Vascular reactivity | Improvement | + |
| Body weight | No change/slight reduction * | ++ |
| Waist circumference | No change/slight reduction * | ++ |
| Metabolic syndrome | Improvement or reversion ** | + |
| Type-2 diabetes incidence | No change ** | + |
| CVD incidence | Reduction ** | + |
| Stroke incidence | Reduction ** | + |
| PAD incidence | Reduction ** | + |
| Cognitive function | Improvement * | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ros, E.; Singh, A.; O’Keefe, J.H. Nuts: Natural Pleiotropic Nutraceuticals. Nutrients 2021, 13, 3269. https://doi.org/10.3390/nu13093269
Ros E, Singh A, O’Keefe JH. Nuts: Natural Pleiotropic Nutraceuticals. Nutrients. 2021; 13(9):3269. https://doi.org/10.3390/nu13093269
Chicago/Turabian StyleRos, Emilio, Annapoorna Singh, and James H. O’Keefe. 2021. "Nuts: Natural Pleiotropic Nutraceuticals" Nutrients 13, no. 9: 3269. https://doi.org/10.3390/nu13093269
APA StyleRos, E., Singh, A., & O’Keefe, J. H. (2021). Nuts: Natural Pleiotropic Nutraceuticals. Nutrients, 13(9), 3269. https://doi.org/10.3390/nu13093269







