Night Vision and Carotenoids (NVC): A Randomized Placebo Controlled Clinical Trial on Effects of Carotenoid Supplementation on Night Vision in Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Acquisition
2.2. Instrumentation
3. Statistical Analysis
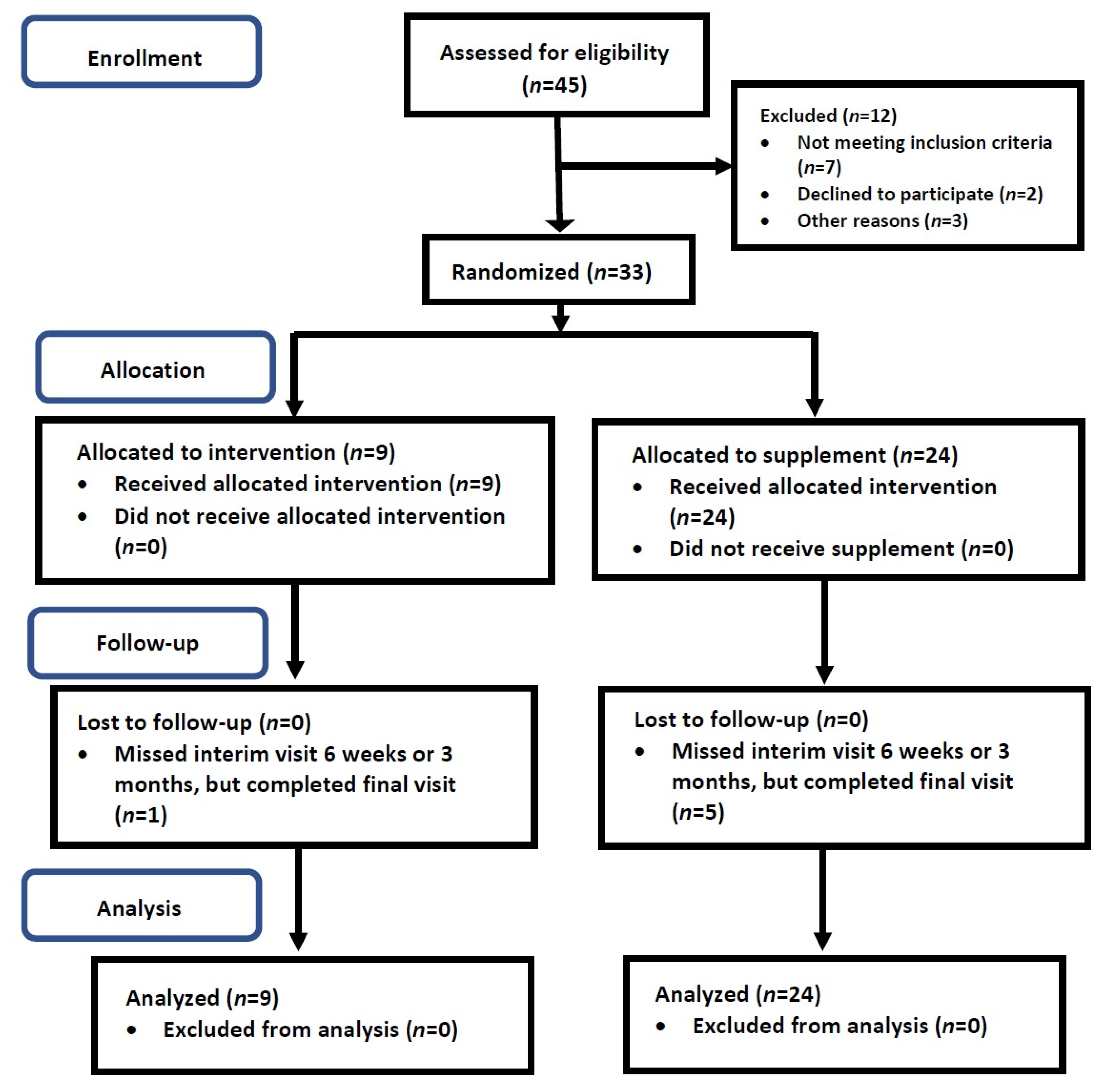
4. Results
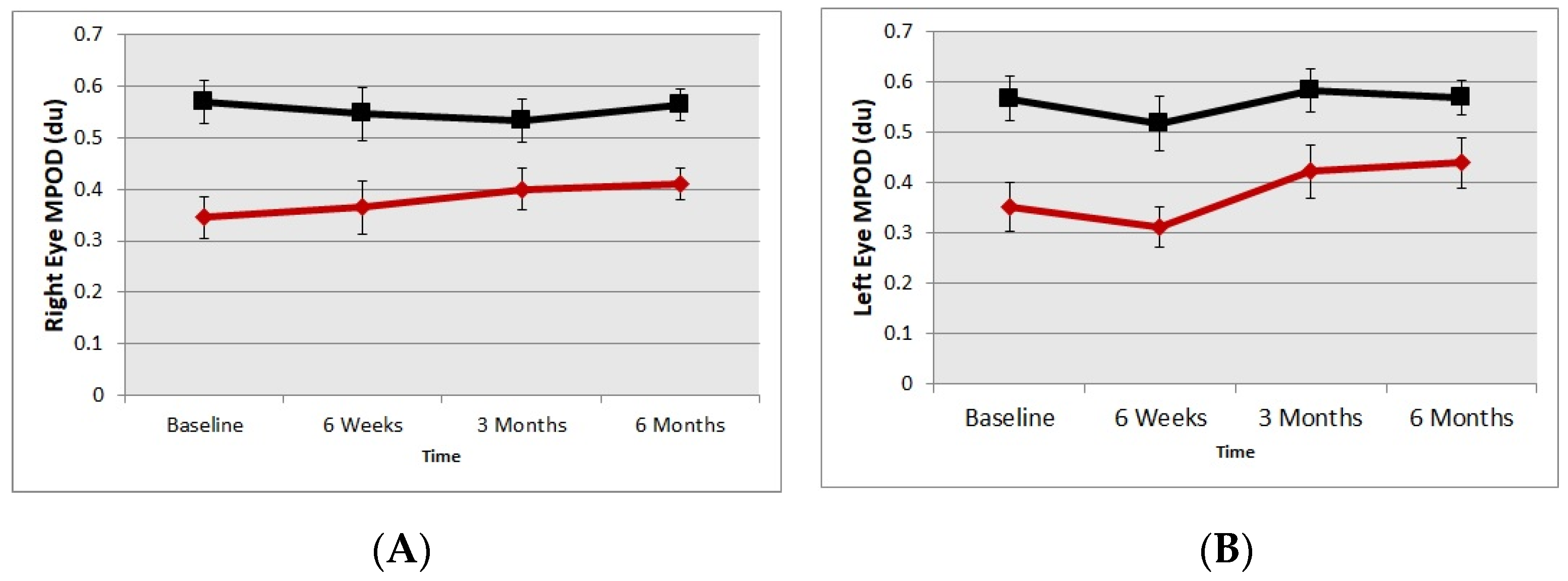
4.1. Macular Pigment Optical Density
4.2. Glare & Contrast Improvement
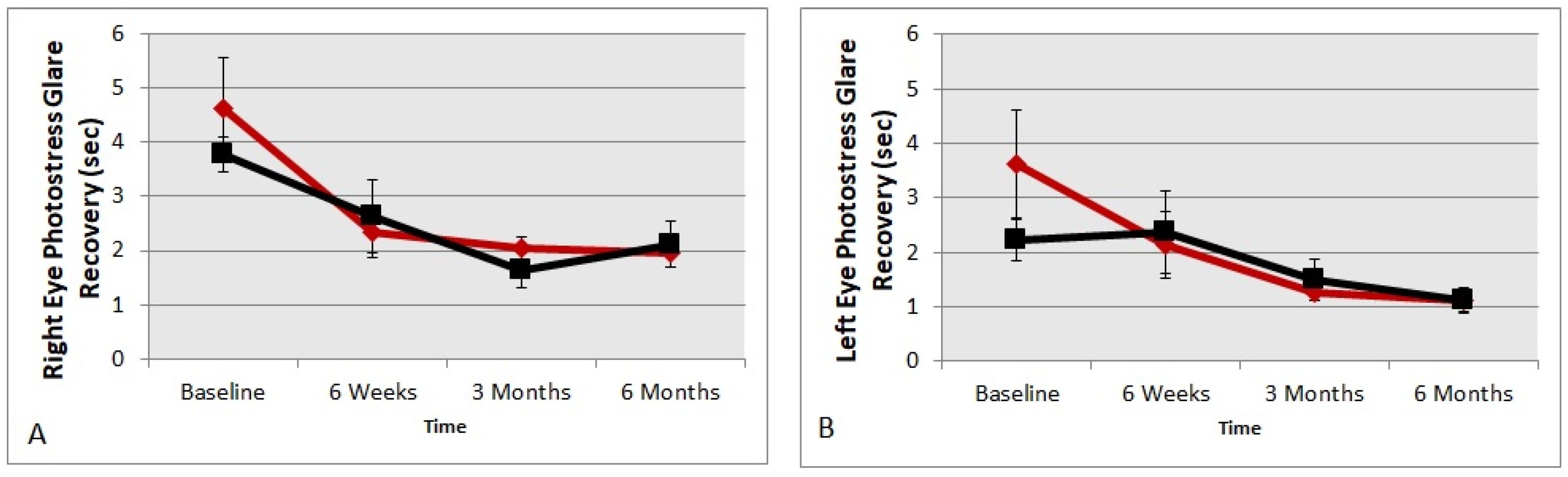
4.3. Glare Recovery Improvement
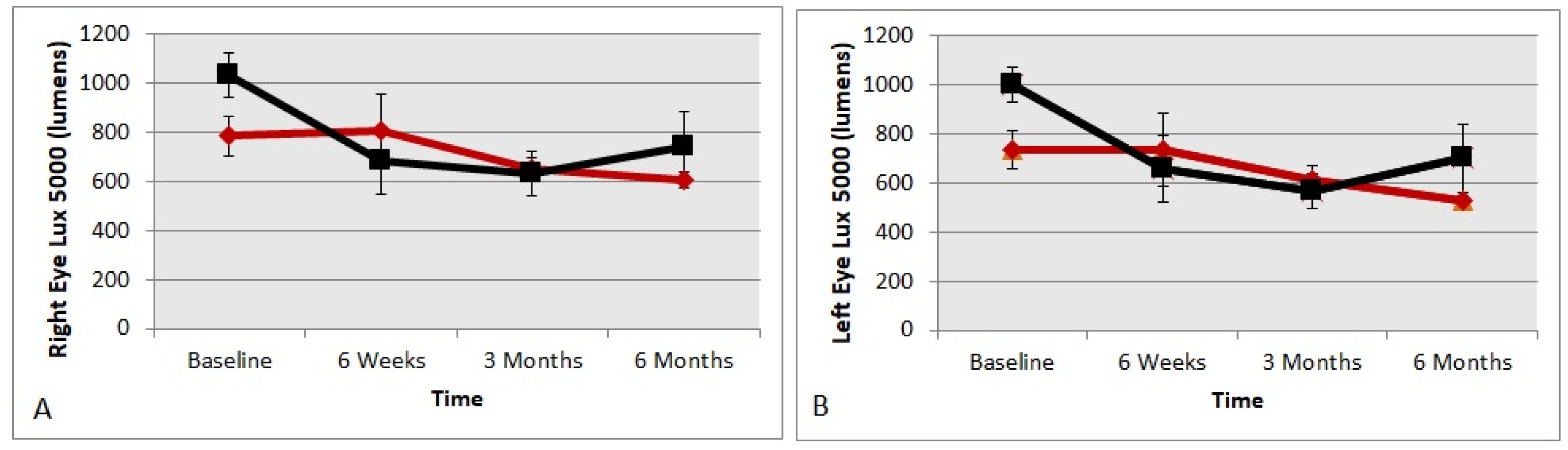
4.4. Preferred Luminance in Lumens
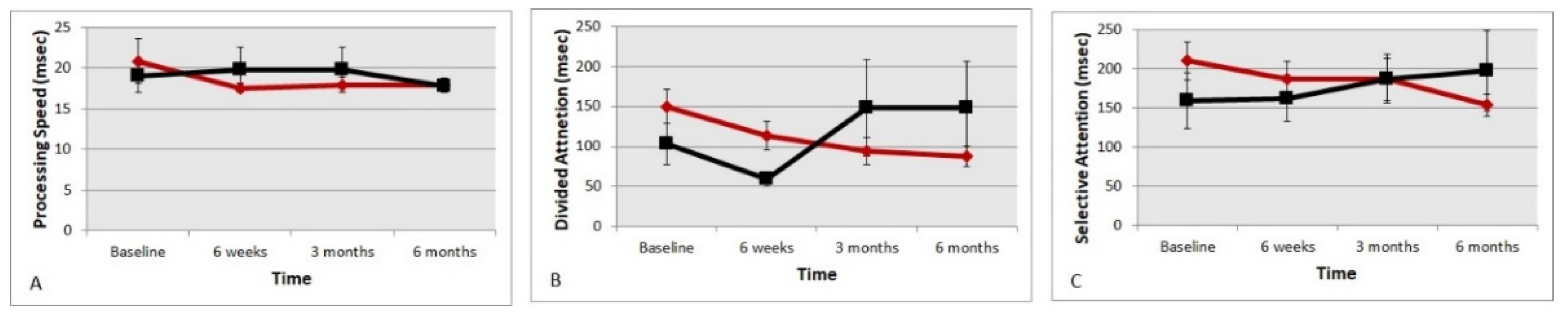
4.5. Useful Field of (UFOV) Reaction Times
4.6. Low Contrast Snellen Visual Acuity Letters
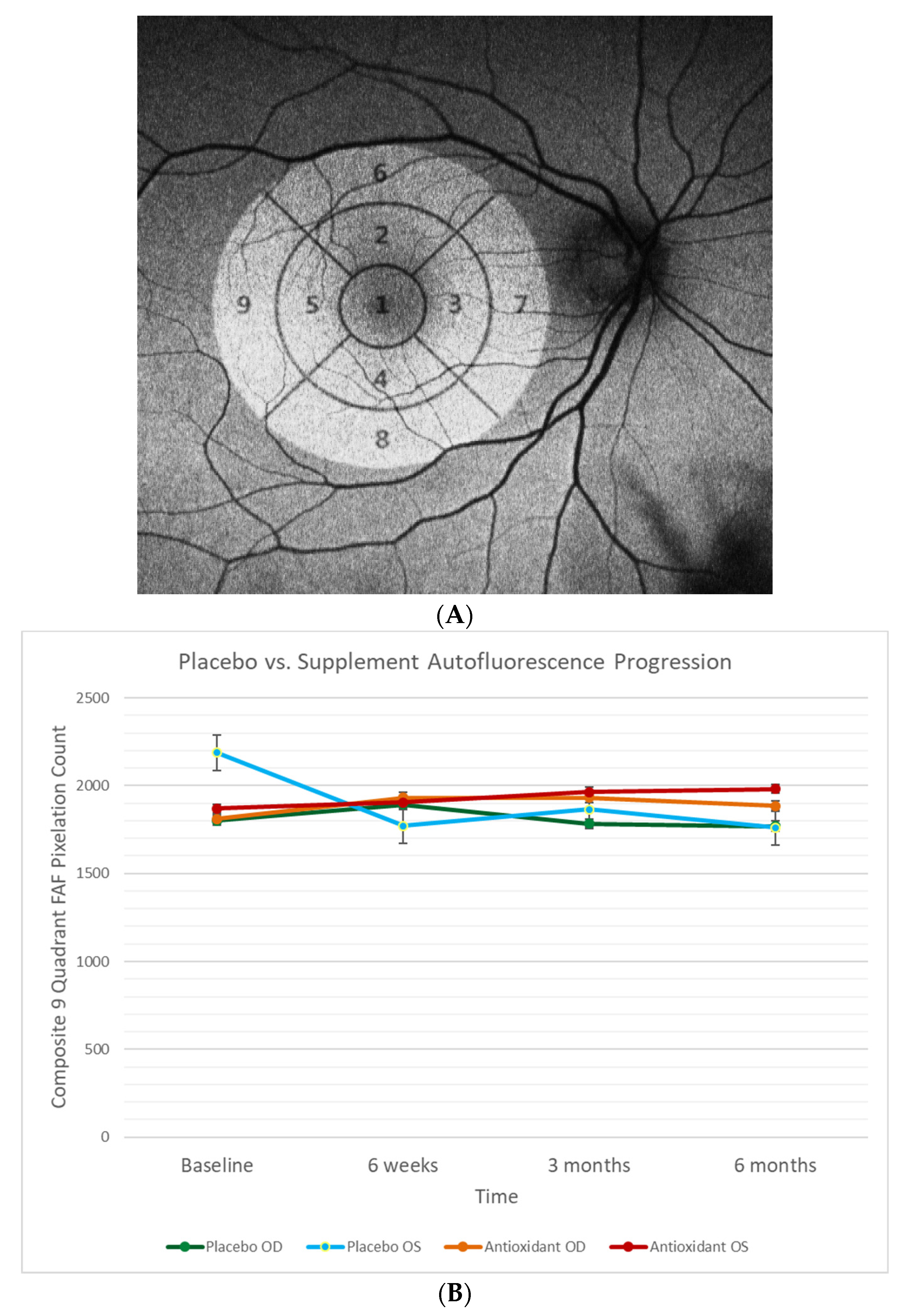
4.7. Retinal Fundus Auto-Fluorescence
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Active Ingredients | Amount per Serving |
|---|---|
| Zeaxanthin | 14 mg |
| Lutein | 7 mg |
| Vitamin A | 2500 IU |
| Vitamin C | 60 mg |
| Vitamin D | 1000 IU |
| Vitamin E | 60 IU |
| Vitamin B6 | 2 mg |
| Folic Acid | 400 mcg |
| Vitamin B12 | 6 mcg |
| Zinc | 15 mg |
| Selenium | 70 mcg |
| Manganese | 2 mg |
| Fish Oil | 150 mg |
| Coenzyme Q10 | 15 mg |
| Bilberry | 15 mg |
| Berry Anthocyanin Extract | 15 mg |
| Alpha Lipoic Acid | 10 mg |
| Mixed Tocopherols | 6 mg |
References
- Andysz, A.; Merecz, D. Visual abilities of older drivers—Review of driving simulator studies. Med. Pr. 2012, 63, 677–687. [Google Scholar]
- Prevalence of Impairments of Visual Function among European Drivers—Vision Impact Institute. Available online: https://www.visionimpactinstitute.org/research/prevalence-of-impairments-of-visual-function-among-european-drivers (accessed on 16 March 2021).
- Van Rijn, L.J.; Nischler, C.; Michael, R.; Heine, C.; Coeckelbergh, T.; Wilhelm, H.; Grabner, G.; Barraquer, R.I.; Van Den Berg, T.J.T.P. Prevalence of impairment of visual function in European drivers. Acta Ophthalmol. 2011, 89, 124–131. [Google Scholar] [CrossRef]
- Hessburg, P.C. Autonomous Vehicles, Drivers Fitness to Function and Naturalistic Driving Methods. In Proceedings of the 9th World Congress on Health and Modern Mobility, Detroit, MI, USA, 7–8 December 2002; Available online: www.henryford.com/TheEyeAndTheAuto (accessed on 16 March 2021).
- Wood, J.M.; Black, A.A.; Mallon, K.; Kwan, A.S.; Owsley, C. Effects of Age-Related Macular Degeneration on Driving Performance. Investig. Opthalmol. Vis. Sci. 2018, 59, 273. [Google Scholar] [CrossRef]
- NHTSA Report. Available online: https://one.nhtsa.gov/nhtsa/whatis/planning/2020Report/2020report.html (accessed on 27 March 2021).
- Chew, E.Y.; Clemons, T.E.; Sangiovanni, J.P.; Danis, R.P.; Ferris, F.L.; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; Bressler, S.B.; et al. Secondary Analyses of the Effects of Lutein/Zeaxanthin on Age-Related Macular Degeneration Progression. JAMA Ophthalmol. 2014, 132, 142. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, M.E.; Carroll, Y.; Corridan, B.; Olmedilla, B.; Granado, F.; Blanco, I.; Berg, H.V.D.; Hininger, I.; Rousell, A.M.; Chopra, M.; et al. A European carotenoid database to assess carotenoid intakes and its use in a five-country comparative study. Br. J. Nutr. 2001, 85, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.; Park, D.W.; Epstein, R.; Wrobel, J.S.; Thomas, C. Macular Re-pigmentation Enhances Driving Vision in Elderly Adult Males with Macular Degeneration. J. Clin. Exp. Ophthalmol. 2012, 3, 217. [Google Scholar] [CrossRef]
- Yao, Y.; Qiu, Q.-H.; Wu, X.-W.; Cai, Z.-Y.; Xu, S.; Liang, X.-Q. Lutein supplementation improves visual performance in Chinese drivers: 1-year randomized, double-blind, placebo-controlled study. Nutrition 2013, 29, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Arunkumar, R.; Calvo, C.M.; Conrady, C.D.; Bernstein, P.S. What do we know about the macular pigment in AMD: The past, the present, and the future. Eye 2018, 32, 992–1004. [Google Scholar] [CrossRef]
- Hammond, B.R.; Fletcher, L.M.; Elliott, J.G. Glare Disability, Photostress Recovery, and Chromatic Contrast: Relation to Macular Pigment and Serum Lutein and Zeaxanthin. Investig. Opthalmol. Vis. Sci. 2013, 54, 476. [Google Scholar] [CrossRef]
- Owsley, C.; Wood, J.M.; McGwin, G., Jr. A roadmap for interpreting the literature on vision and driving. Surv. Ophthalmol. 2015, 60, 250–262. [Google Scholar] [CrossRef]
- Stringham, J.M.; Garcia, P.V.; Smith, P.A.; McLin, L.N.; Foutch, B.K. Macular Pigment and Visual Performance in Glare: Benefits for Photostress Recovery, Disability Glare, and Visual Discomfort. Investig. Opthalmol. Vis. Sci. 2011, 52, 7406. [Google Scholar] [CrossRef]
- Oliver, W.; Renzi-Hammond, L.M.; Thorne, S.A.; Clementz, B.; Miller, L.S.; Hammond, B.R. Neural Activation During Visual Attention Differs in Individuals with High versus Low Macular Pigment Density. Mol. Nutr. Food Res. 2019, 63, 1801052. [Google Scholar] [CrossRef]
- Kaido, M.; Matsutani, T.; Negishi, K.; Dogru, M.; Tsubota, K. Aged Drivers May Experience Decreased Visual Function While Driving. Asia-Pac. J. Ophthalmol. 2013, 2, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Green, R.C.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between Serum and Brain Carotenoids,α-Tocopherol, and Retinol Concentrations and Cognitive Performance in the Oldest Old from the Georgia Centenarian Study. J. Aging Res. 2013, 2013, 951786. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Schalch, W.; Johnson, E.J. Macular pigment carotenoids in the retina and occipital cortex are related in humans. Nutr. Neurosci. 2016, 19, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Feeney, J.; Finucane, C.; Savva, G.M.; Cronin, H.; Beatty, S.; Nolan, J.M.; Kenny, R.A. Low macular pigment optical density is associated with lower cognitive performance in a large, population-based sample of older adults. Neurobiol. Aging 2013, 34, 2449–2456. [Google Scholar] [CrossRef]
- Hoffmann, K.G.; Richer, S.P.; Wrobel, J.S.; Chen, E.; Podella, C.J. A Prospective Study of Neuro-Cognitive Enhancement with Carotenoids in Elderly Adult Males with Early Age Related Macular Degeneration. Ophthalmol. Res. Int. J. 2015, 4, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S.; Delori, F.C.; Richer, S.; Van Kuijk, F.J.M.; Wenzel, A.J. The value of measurement of macular carotenoid pigment optical densities and distributions in age-related macular degeneration and other retinal disorders. Vis. Res. 2010, 50, 716–728. [Google Scholar] [CrossRef]
- Davey, P.G.; Henderson, T.; Lem, D.W.; Weis, R.; Amonoo-Monney, S.; Evans, D.W. Visual Function and Macular Carotenoid Changes in Eyes with Retinal Drusen—An Open Label Randomized Controlled Trial to Compare a Micronized Lipid-Based Carotenoid Liquid Supplementation and AREDS-2 Formula. Nutrients 2020, 12, 3271. [Google Scholar] [CrossRef] [PubMed]
- Lem, D.W.; Davey, P.G.; Gierhart, D.L.; Rosen, R.B. A Systematic Review of Carotenoids in the Management of Age-Related Macular Degeneration. Antioxidants 2021, 10, 1255. [Google Scholar] [CrossRef]
- Lem, D.W.; Gierhart, D.L.; Davey, P.G. Carotenoids in the Management of Glaucoma: A Systematic Review of the Evidence. Nutrients 2021, 13, 1949. [Google Scholar] [CrossRef] [PubMed]
- Lem, D.W.; Gierhart, D.L.; Davey, P.G. A Systematic Review of Carotenoids in the Management of Diabetic Retinopathy. Nutrients 2021, 13, 2441. [Google Scholar] [CrossRef]
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The Veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Richer, S.P.; Stiles, W.; Graham-Hoffman, K.; Levin, M.; Ruskin, D.; Wrobel, J.; Park, D.-W.; Thomas, C. Randomized, double-blind, placebo-controlled study of zeaxanthin and visual function in patients with atrophic age-related macular degeneration. Optom. J. Am. Optom. Assoc. 2011, 82, 667–680.e666. [Google Scholar] [CrossRef] [PubMed]
- Owsley, C.; McGwin, G.; Scilley, K.; Kallies, K. Development of a Questionnaire to Assess Vision Problems under Low Luminance in Age-Related Maculopathy. Investig. Opthalmol. Vis. Sci. 2006, 47, 528. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.; Robert, D.; Novil, S.; Dervishi, A.; Nassiri, S.; Davey, P.G. Predicting night vision difficulties during an eye examination. (Night Vision & Carotenoids RCT; Hines VA IRB 1052607-1, Baseline Data). Investig. Ophthalmol. Vis. Sci. 2019, 60, 1828. [Google Scholar]
- Jackson, G.R.; Scott, I.U.; Kim, I.K.; Quillen, D.A.; Iannaccone, A.; Edwards, J.G. Diagnostic Sensitivity and Specificity of Dark Adaptometry for Detection of Age-Related Macular Degeneration. Investig. Opthalmol. Vis. Sci. 2014, 55, 1427. [Google Scholar] [CrossRef]
- Intelligent Refractor RT-6100. Available online: https://www.nidek-intl.com/product/ophthaloptom/refraction/ref_optometry/rt-6100.html (accessed on 16 March 2021).
- Smart System Standard. Available online: https://premierop.com/smart-system-standard/ (accessed on 16 March 2021).
- Davey, P.-G.; Alvarez, S.; Lee, J. Macular pigment optical density: Repeatability, intereye correlation, and effect of ocular dominance. Clin. Ophthalmol. 2016, 10, 1671–1678. [Google Scholar] [CrossRef]
- Van Der Veen, R.L.P.; Berendschot, T.T.J.M.; Hendrikse, F.; Carden, D.; Makridaki, M.; Murray, I.J. A new desktop instrument for measuring macular pigment optical density based on a novel technique for setting flicker thresholds. Ophthalmic Physiol. Opt. 2009, 29, 127–137. [Google Scholar] [CrossRef]
- Davey, P.G.; Rosen, R.B.; Gierhart, D.L. Macular Pigment Reflectometry: Developing Clinical Protocols, Comparison with Heterochromatic Flicker Photometry and Individual Carotenoid Levels. Nutrients 2021, 13, 2553. [Google Scholar] [CrossRef]
- Sanabria, J.C.; Bass, J.; Spors, F.; Gierhart, D.L.; Davey, P.G. Measurement of Carotenoids in Perifovea using the Macular Pigment Reflectometer. J. Vis. Exp. 2020, 155, e60429. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Teussink, M.M.; Sloan, K.R.; Surya, J.; Raman, R. Evaluation of Macular Pigment Optical Density in Healthy Eyes Based on Dual-Wavelength Autofluorescence Imaging in South Indian Population. Transl. Vis. Sci. Technol. 2020, 9, 40. [Google Scholar] [CrossRef]
- Davey, P.G.; Lievens, C.; Amonoo-Monney, S. Differences in macular pigment optical density across four ethnicities: A comparative study. Ther. Adv. Ophthalmol. 2020, 12, 251584142092416. [Google Scholar] [CrossRef] [PubMed]
- Central Vision Analyzer. Available online: https://sinclairtechnologiesllc.com/project/real-world-functional-vision-analysis/cva/ (accessed on 16 March 2021).
- Wittich, W.; St Amour, L.; Jarry, J.; Seiple, W. Test-retest Variability of a Standardized Low Vision Lighting Assessment. Optom. Vis. Sci. 2018, 95, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Newsome, D.A.; Negreiro, M. Reproducible measurement of macular light flash recovery time using a novel device can indicate the presence and worsening of macular diseases. Curr. Eye Res. 2009, 34, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Loughman, J.; Hewitt, C.; Judge, C.; Martin, L.; Moulds, C.; Davison, P.A. Clinical applicability of the Macular Degeneration Detection Device (MDD-2): A novel photostress recovery measurement device. Clin. Exp. Optom. 2013, 96, 272–277. [Google Scholar] [CrossRef][Green Version]
- Brain Training That Works. Available online: https://www.brainhq.com/?v4=true&fr=y (accessed on 21 March 2021).
- Holz, F.G.; Schmitz-Valckenberg, S.; Spaide, R.F.; Bird, A.C. Atlas of Fundus Autofluorescence Imaging, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Xu, Q.; Li, Y.; Cheng, Y.; Qu, Y. Assessment of the effect of age on macular layer thickness in a healthy Chinese cohort using spectral-domain optical coherence tomography. BMC Ophthalmol. 2018, 18, 169. [Google Scholar] [CrossRef]
- Chous, A.P.; Richer, S.P.; Gerson, J.D.; Kowluru, R.A. The Diabetes Visual Function Supplement Study (DiVFuSS). Br. J. Ophthalmol. 2016, 100, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Loughman, J.; Loskutova, E.; Butler, J.S.; Siah, W.F.; O’Brien, C. Macular pigment response to lutein, zeaxanthin and meso-zeaxanthin supplementation in open angle glaucoma: A randomized controlled trial. Ophthalmol. Sci. 2021, 1, 100039. [Google Scholar] [CrossRef]
- Siah, W.F.; O’Brien, C.; Loughman, J.J. Macular pigment is associated with glare-affected visual function and central visual field loss in glaucoma. Br. J. Ophthalmol. 2018, 102, 929–935. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Davey, P.G.; Roman, B.O.; Evans, D.W. Efficacy of Commercially Available Nutritional Supplements: Analysis of Serum Uptake, Macular Pigment Optical Density and Visual Functional Response. Nutrients 2020, 12, 1321. [Google Scholar] [CrossRef] [PubMed]
- Dukic Willstrand, T.; Broberg, T.; Selander, H. Driving Characteristics of Older Drivers and Their Relationship to the Useful Field of View Test. Gerontology 2017, 63, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Rush, E.; Amoah, I.; Diep, T.; Jalili-Moghaddam, S. Determinants and Suitability of Carotenoid Reflection Score as a Measure of Carotenoid Status. Nutrients 2020, 12, 113. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.R.; Sandberg, K.A.; Foutch, B.K. Macular pigment optical density and visual quality of life. J. Optom. 2021, 14, 92–99. [Google Scholar] [CrossRef]
| Placebo Group | Treatment Group | p-Value | |
|---|---|---|---|
| Mean (Standard Deviation) | |||
| Age | 65.7 (8.0) | 61.6 (11.63) | 0.027 |
| Body Mass Index | 26.3 (3.42) | 31.30 (6.54) | 0.06 |
| Percent Body Fat | 28.2 (4.56) | 33.5 (6.81) | 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richer, S.; Novil, S.; Gullett, T.; Dervishi, A.; Nassiri, S.; Duong, C.; Davis, R.; Davey, P.G. Night Vision and Carotenoids (NVC): A Randomized Placebo Controlled Clinical Trial on Effects of Carotenoid Supplementation on Night Vision in Older Adults. Nutrients 2021, 13, 3191. https://doi.org/10.3390/nu13093191
Richer S, Novil S, Gullett T, Dervishi A, Nassiri S, Duong C, Davis R, Davey PG. Night Vision and Carotenoids (NVC): A Randomized Placebo Controlled Clinical Trial on Effects of Carotenoid Supplementation on Night Vision in Older Adults. Nutrients. 2021; 13(9):3191. https://doi.org/10.3390/nu13093191
Chicago/Turabian StyleRicher, Stuart, Steven Novil, Taylor Gullett, Avni Dervishi, Sherwin Nassiri, Co Duong, Robert Davis, and Pinakin Gunvant Davey. 2021. "Night Vision and Carotenoids (NVC): A Randomized Placebo Controlled Clinical Trial on Effects of Carotenoid Supplementation on Night Vision in Older Adults" Nutrients 13, no. 9: 3191. https://doi.org/10.3390/nu13093191
APA StyleRicher, S., Novil, S., Gullett, T., Dervishi, A., Nassiri, S., Duong, C., Davis, R., & Davey, P. G. (2021). Night Vision and Carotenoids (NVC): A Randomized Placebo Controlled Clinical Trial on Effects of Carotenoid Supplementation on Night Vision in Older Adults. Nutrients, 13(9), 3191. https://doi.org/10.3390/nu13093191







