Changes in Gut Microbiota Induced by Doxycycline Influence in Vascular Function and Development of Hypertension in DOCA-Salt Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Groups
2.2. Blood Pressure Measurements
2.3. Cardiac and Renal Weight Indices
2.4. Plasma Determinations
2.5. Vascular Reactivity Studies
2.6. Vascular NADPH Oxidase Activity and Measurement of Ex Vivo Vascular Reactive Oxygen Species (ROS)
2.7. Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) Analysis
2.8. 16S rDNA V4-V5 Region Sequencing
2.9. Bioinformatics Analysis
2.10. Reagents
2.11. Statistical Analysis
3. Results
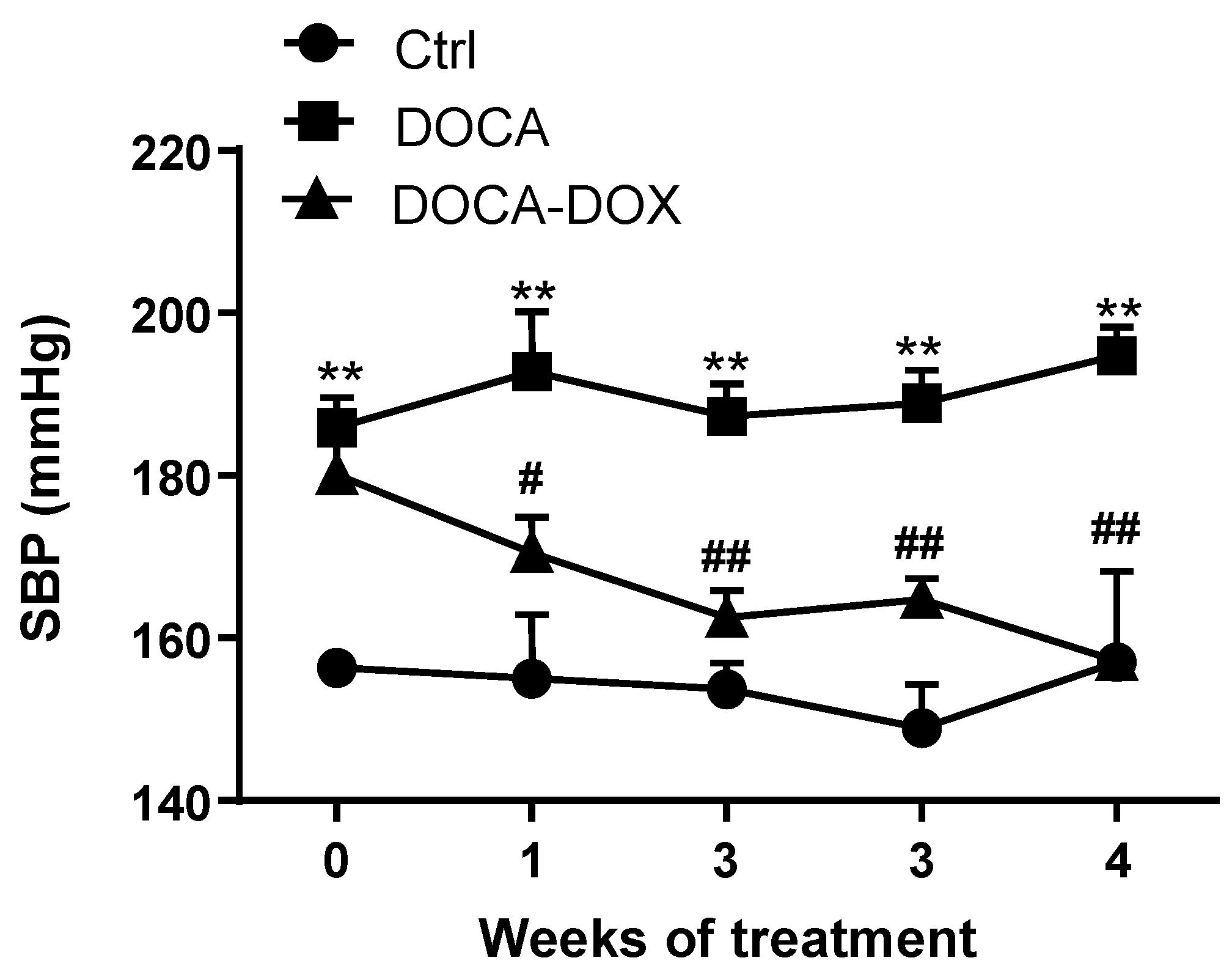
3.1. Doxycycline Treatment Improved Morphologic Variables and Blood Pressure
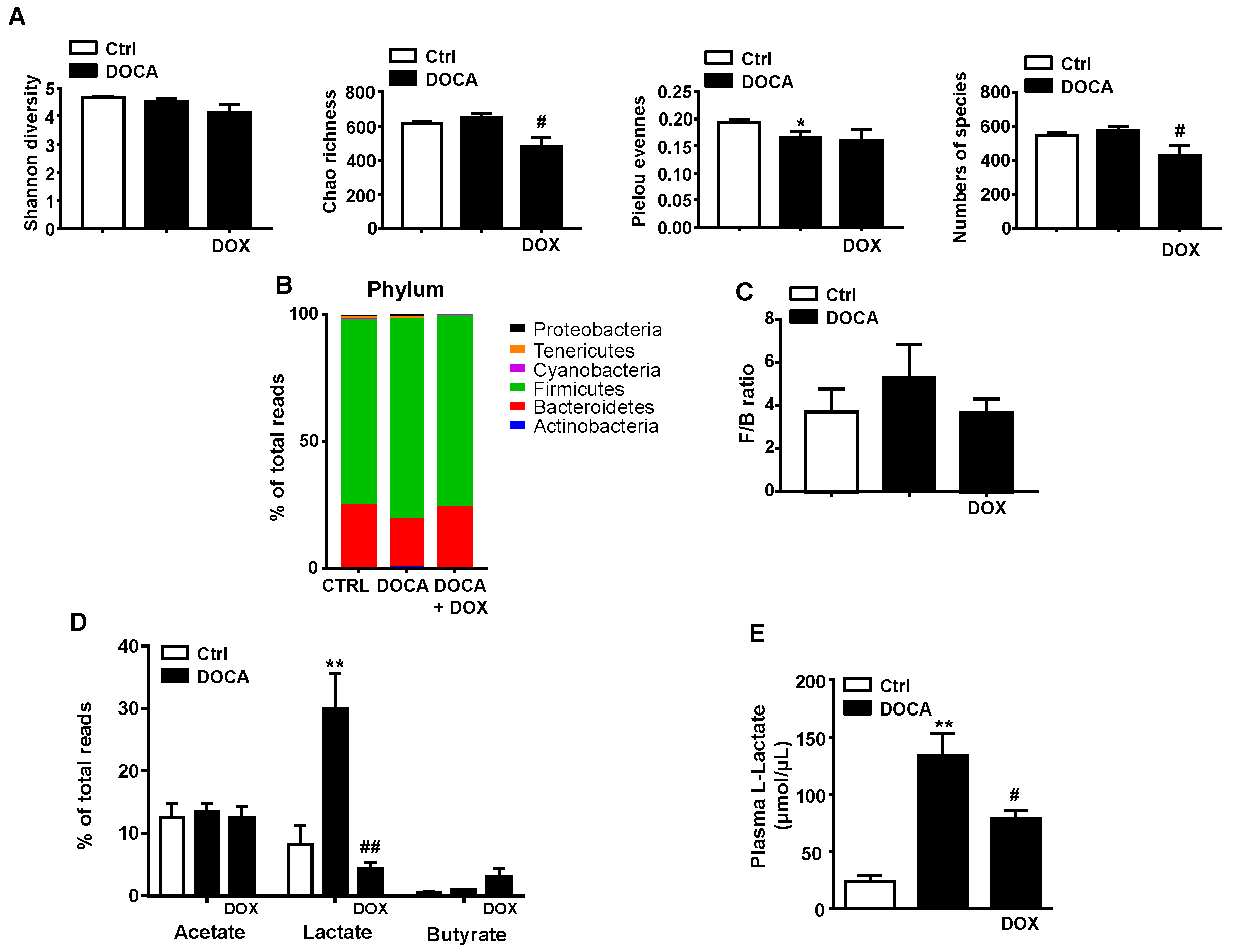
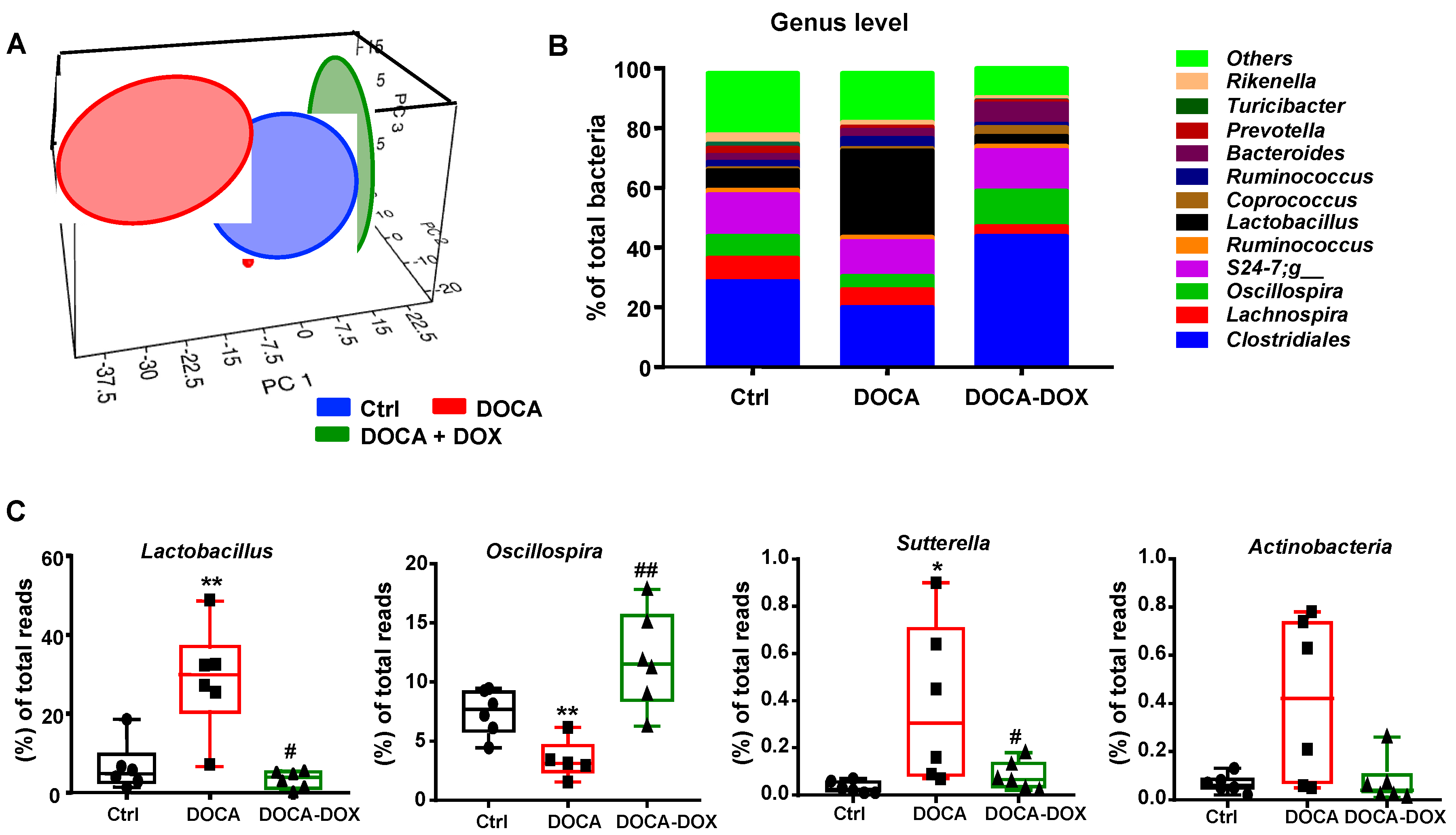
3.2. Doxycycline Treatment Induced Changes in Gut Microbiota in DOCA-Salt Treated Rats
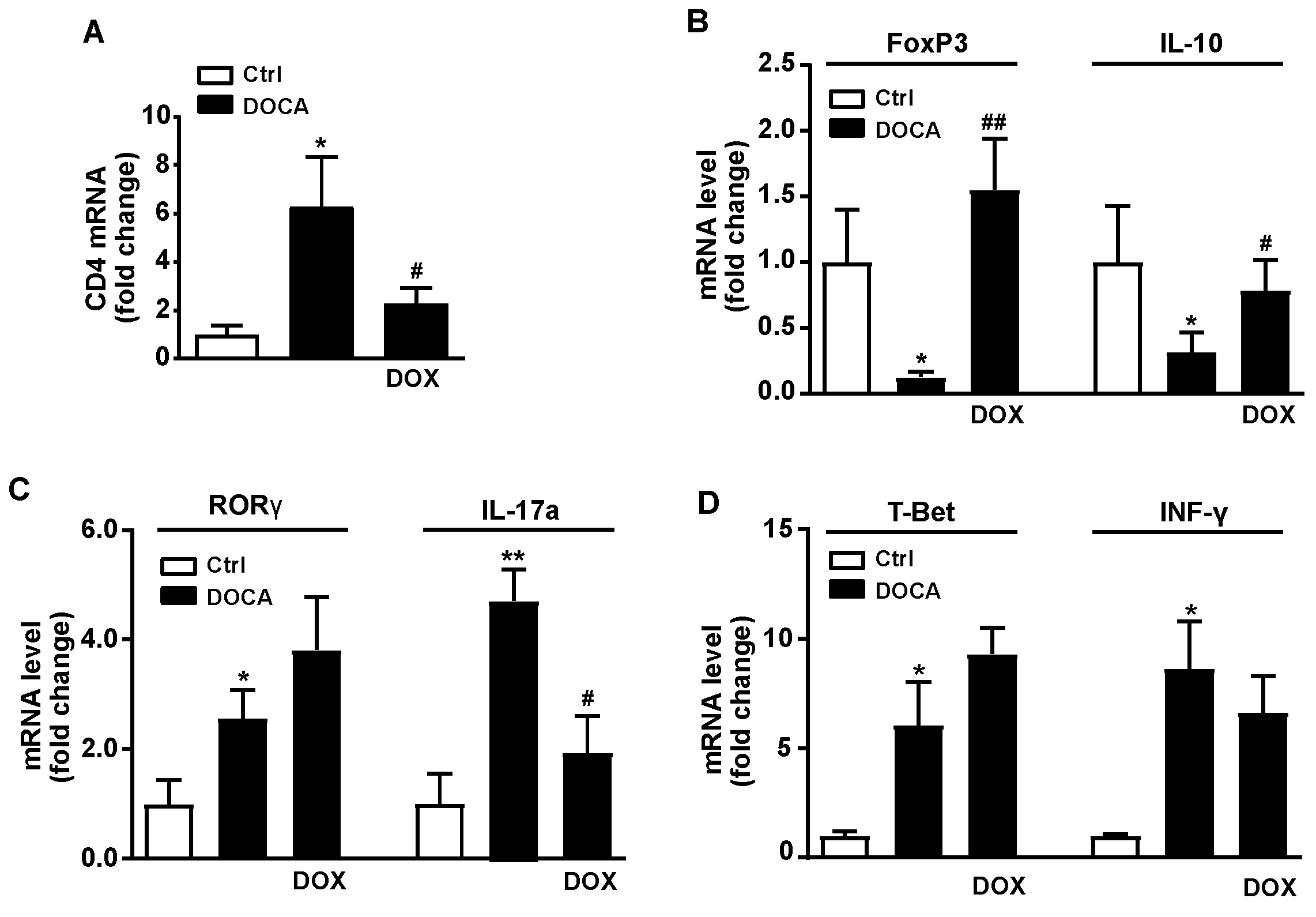
3.3. Doxycycline Treatment Improved Intestinal Integrity, Colonic Inflammation and Reduced Endotoxemia and Plasma Noradrenaline in DOCA-Salt Rats
3.4. Doxycycline Treatment Improved Endothelial Function and Reduced Vascular Oxidative Stress and Inflammation in DOCA-Salt Rats
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| Ang II | angiotensin II |
| DOCA | deoxycorticosterone acetate |
| DOX | doxycycline |
| Ctrl | control |
| DAPI | 4,6-diamidino-2-phenylindoledichlorohydrate |
| DHE | dihydroethidium |
| F/B | Firmicutes/Bacteroidetes |
| FoxP3 | forkhead box P3 |
| LDA | linear discriminant analysis |
| L-NAME | NG-nitro-L-arginine methyl ester |
| LPS | lipopolysaccharide |
| MUC | mucin |
| NIH | National Institutes of Health |
| NO | nitric oxide |
| NOX | NADPH oxidase |
| OTU | operational taxonomic unit |
| PCA | principal component analysis |
| RORγ | retinoid-related orphan receptor-γ |
| ROS | reactive oxygen species |
| Rpm | revolutions per minute |
| RT | real-time |
| SBP | Systolic blood pressure |
| SCFAs | short chain fatty acids |
| SHR | spontaneously hypertensive rat |
| SNP | sodium nitroprusside |
| Th | T-helper |
| TLRs | Toll-like receptors |
| Tregs | regulatory T cell |
| ZO-1 | zonula occludens-1 |
References
- North, B.J.; Sinclair, D.A. The intersection between aging and cardiovascular disease. Circ. Res. 2012, 110, 1097–1108. [Google Scholar] [CrossRef]
- Cutler, J.A.; Sorlie, P.D.; Wolz, M.; Thom, T.; Fields, L.E.; Roccella, E.J. Trends in hypertension prevalence, awareness, treatment, and control rates in United States adults between 1988–1994 and 1999–2004. Hypertension 2008, 52, 818–827. [Google Scholar] [CrossRef] [Green Version]
- Yang, T.; Santisteban, M.M.; Rodriguez, V.; Li, E.; Ahmari, N.; Carvajal, J.M.; Zadeh, M.; Gong, M.; Qi, Y.; Zubcevic, J.; et al. Gut dysbiosis is linked to hypertension. Hypertension 2015, 65, 1331–1340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santisteban, M.M.; Qi, Y.; Zubcevic, J.; Kim, S.; Yang, T.; Shenoy, V.; Cole-Jeffrey, C.T.; Lobaton, G.O.; Stewart, D.C.; Rubiano, A.; et al. Hypertension-Linked Pathophysiological Alterations in the Gut. Circ. Res. 2017, 120, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Mell, B.; Jala, V.R.; Mathew, A.V.; Byun, J.; Waghulde, H.; Zhang, Y.; Haribabu, B.; Vijay-Kumar, M.; Pennathur, S.; Joe, B. Evidence for a link between gut microbiota and hypertension in the Dahl rat. Physiol. Genom. 2015, 47, 187–197. [Google Scholar] [CrossRef] [Green Version]
- Adnan, S.; Nelson, J.W.; Ajami, N.J.; Venna, V.R.; Petrosino, J.F.; Bryan, R.M., Jr.; Durgan, D.J. Alterations in the gut microbiota can elicit hypertension in rats. Physiol. Genom. 2017, 49, 96–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marques, F.Z.; Nelson, E.; Chu, P.Y.; Horlock, D.; Fiedler, A.; Ziemann, M.; Tan, J.K.; Kuruppu, S.; Rajapakse, N.W.; El-Osta, A.; et al. High-Fiber Diet and Acetate Supplementation Change the Gut Microbiota and Prevent the Development of Hypertension and Heart Failure in Hypertensive Mice. Circulation 2017, 135, 964–977. [Google Scholar] [CrossRef]
- Karbach, S.H.; Schönfelder, T.; Brandão, I.; Wilms, E.; Hörmann, N.; Jäckel, S.; Schüler, R.; Finger, S.; Knorr, M.; Lagrange, J.; et al. Gut microbiota promote angiotensin II-induced arterial hypertension and vascular dysfunction. J. Am. Heart Assoc. 2016, 5, e003698. [Google Scholar] [CrossRef] [Green Version]
- Honour, J.W.; Borriello, S.P.; Ganten, U.; Honour, P. Antibiotics attenuate experimental hypertension in rats. J. Endocrinol. 1985, 105, 347–350. [Google Scholar] [CrossRef]
- Honour, J.W. Historical perspective: Gut dysbiosis and hypertension. Physiol. Genom. 2015, 47, 443–446. [Google Scholar] [CrossRef]
- Galla, S.; Chakraborty, S.; Cheng, X.; Yeo, J.; Mell, B.; Zhang, H.; Mathew, A.V.; Vijay-Kumar, M.; Joe, B. Disparate effects of antibiotics on hypertension. Physiol. Genom. 2018, 50, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Antonio, R.C.; Ceron, C.S.; Rizzi, E.; Coelho, E.B.; Tanus-Santos, J.E.; Gerlach, R.F. Antioxidant effect of doxycycline decreases MMP activity and blood pressure in SHR. Mol. Cell. Biochem. 2014, 386, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.M.; Rizzi, E.; Ceron, C.S.; Guimaraes, D.A.; Rodrigues, G.J.; Bendhack, L.M.; Gerlach, R.F.; Tanus-Santos, J.E. Doxycycline ameliorates 2K-1C hypertension-induced vascular dysfunction in rats by attenuating oxidative stress and improving nitric oxide bioavailability. Nitric Oxide 2012, 26, 162–168. [Google Scholar] [CrossRef]
- Nascimento, R.A.; Mendes, G.; Possomato-Vieira, J.S.; Goncalves-Rizzi, V.H.; Kushima, H.; Delella, F.K.; Dias-Junior, C.A. Metalloproteinase inhibition protects against reductions in circulating adrenomedullin during lead-induced acute hypertension. Basic Clin. Pharmacol. Toxicol. 2015, 116, 508–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nascimento, R.A.; Possomato-Vieira, J.S.; Gonçalves-Rizzi, V.H.; Bonacio, G.F.; Rizzi, E.; Dias-Junior, C.A. Hypertension, augmented activity of matrix metalloproteinases-2 and -9 and angiogenic imbalance in hypertensive pregnancy are attenuated by doxycycline. Eur. J. Pharmacol. 2018, 840, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.M.; Tanus-Santos, J.E.; Gerlach, R.F. Matrix metalloproteinases: Targets for doxycycline to prevent the vascular alterations of hypertension. Pharmacol. Res. 2011, 64, 567–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guimaraes, D.A.; Rizzi, E.; Ceron, C.S.; Oliveira, A.M.; Oliveira, D.M.; Castro, M.M.; Tirapelli, C.R.; Gerlach, R.F.; Tanus-Santos, J.E. Doxycycline dose-dependently inhibits MMP-2-mediated vascular changes in 2K1C hypertension. Basic Clin. Pharmacol. Toxicol. 2011, 108, 318–325. [Google Scholar] [CrossRef]
- Basting, T.; Lazartigues, E. DOCA-Salt Hypertension: An Update. Curr. Hypertens. Rep. 2017, 19, 32. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Jiménez, R.; Sánchez, M.; Zarzuelo, M.J.; Galindo, P.; Quintela, A.M.; López-Sepúlveda, R.; Romero, M.; Tamargo, J.; Vargas, F.; et al. Epicatechin lowers blood pressure, restores endothelial function, and decreases oxidative stress and endothelin-1 and NADPH oxidase activity in DOCA-salt hypertension. Free Radic. Biol. Med. 2012, 52, 70–79. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Toral, M.; Romero, M.; Jiménez, R.; Galindo, P.; Sánchez, M.; Zarzuelo, M.J.; Olivares, M.; Gálvez, J.; Duarte, J. Antihypertensive effects of probiotics Lactobacillus strains in spontaneously hypertensive rats. Mol. Nutr. Food Res. 2015, 59, 2326–2336. [Google Scholar] [CrossRef]
- Toral, M.; Robles-Vera, I.; de la Visitación, N.; Romero, M.; Sánchez, M.; Gómez-Guzmán, M.; Rodriguez-Nogales, A.; Yang, T.; Jiménez, R.; Algieri, F.; et al. Role of the immune system in vascular function and blood pressure control induced by faecal microbiota transplantation in rats. Acta Physiol. 2019, 227, e13285. [Google Scholar] [CrossRef] [PubMed]
- Robles-Vera, I.; Toral, M.; de la Visitación, N.; Sánchez, M.; Romero, M.; Olivares, M.; Jiménez, R.; Duarte, J. The Probiotic Lactobacillus fermentum Prevents Dysbiosis and Vascular Oxidative Stress in Rats with Hypertension Induced by Chronic Nitric Oxide Blockade. Mol. Nutr. Food Res. 2018, 62, e1800298. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Subramanian, S.; Faith, J.J.; Gevers, D.; Gordon, J.I.; Knight, R.; Mills, D.A.; Caporaso, J.G. Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nat. Methods 2013, 10, 57–59. [Google Scholar] [CrossRef]
- Angelakis, E.; Million, M.; Kankoe, S.; Lagier, J.C.; Armougom, F.; Giorgi, R.; Raoult, D. Abnormal weight gain and gut microbiota modifications are side effects of long-term doxycycline and hydroxychloroquine treatment. Antimicrob. Agents Chemother. 2014, 58, 3342–3347. [Google Scholar] [CrossRef] [Green Version]
- Becker, E.; Schmidt, T.S.B.; Bengs, S.; Poveda, L.; Opitz, L.; Atrott, K.; Stanzel, C.; Biedermann, L.; Rehman, A.; Jonas, D.; et al. Effects of oral antibiotics and isotretinoin on the murine gut microbiota. Int. J. Antimicrob. Agents 2017, 50, 342–351. [Google Scholar] [CrossRef] [Green Version]
- Boynton, F.D.D.; Ericsson, A.C.; Uchihashi, M.; Dunbar, M.L.; Wilkinson, J.E. Doxycycline induces dysbiosis in female C57BL/6NCrl mice. BMC Res. Notes 2017, 10, 644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wall, R.; Cryan, J.F.; Ross, R.P.; Fitzgerald, G.F.; Dinan, T.G.; Stanton, C. Bacterial neuroactive compounds produced by psychobiotics. Adv. Exp. Med. Biol. 2014, 817, 221–239. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.; Corrales, R.M.; Villarreal, A.L.; Farley, W.J.; Li, D.-Q.; Stern, M.E.; Pflugfelder, S.C. Corticosteroid and doxycycline suppress MMP-9 and inflammatory cytokine expression, MAPK activation in the corneal epithelium in experimental dry eye. Exp. Eye Res. 2006, 83, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Cazalis, J.; Bodet, C.; Gagnon, G.; Greiner, D. Doxycycline reduces lipopolysaccharide-induced inflammatory mediator secretion in macrophage and ex vivo human blood models. J. Peridontol. 2008, 79, 1762–1768. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, K.; Martin, T.; Andreas, S.T.; Kim, Y.J. Chemically modified tetracyclines selectively inhibit IL-6 expression in osteoblasts by decreasing mRNA stability. Transplant 2008, 17, 223–229. [Google Scholar] [CrossRef]
- Toral, M.; Gómez-Guzmán, M.; Jiménez, R.; Romero, M.; Sánchez, M.; Utrilla, M.P.; Garrido-Mesa, N.; Rodríguez-Cabezas, M.E.; Olivares, M.; Gálvez, J.; et al. The probiotic Lactobacillus coryniformis CECT5711 reduces the vascular pro-oxidant and pro-inflammatory status in obese mice. Clin. Sci. 2014, 127, 33–45. [Google Scholar] [CrossRef]
- Kim, F.; Pham, M.; Luttrell, I.; Bannerman, D.D.; Tupper, J.; Thaler, J.; Hawn, T.R.; Raines, E.W.; Schwartz, M.W. Toll-like receptor-4 mediates vascular inflammation and insulin resistance in diet-induced obesity. Circ. Res. 2007, 100, 1589–1596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maloney, E.; Sweet, I.R.; Hockenbery, D.M.; Pham, M.; Rizzo, N.O.; Tateya, S.; Handa, P.; Schwartz, M.W.; Kim, F. Activation of NF-kappaB by palmitate in endothelial cells: A key role for NADPH oxidase-derived superoxide in response to TLR4 activation. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1370–1975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, C.F.; Liu, J.T.; Wang, Y.; Xu, A.; Vanhoutte, P.M. Toll-like receptor 4 mutation protects obese mice against endothelial dysfunction by decreasing NADPH oxidase isoforms 1 and 4. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 777–784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Funder, J.W.; Carey, R.M.; Fardella, C.; Gomez-Sanchez, C.E.; Mantero, F.; Stowasser, M.; Young, W.F., Jr.; Montori, V.M.; Endocrine Society. Case detection, diagnosis, and treatment of patients with primary aldosteronism: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2008, 93, 3266–3281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, P.; Diez-Freire, C.; Jun, J.Y.; Qi, Y.; Katovich, M.J.; Li, Q.; Sriramula, S.; Francis, J.; Sumners, C.; Raizada, M.K. Brain microglial cytokines in neurogenic hypertension. Hypertension 2010, 56, 297–303. [Google Scholar] [CrossRef] [Green Version]
- Qi, Y.F.; Aranda, J.M.; Rodriguez, V.; Raizada, M.K.; Pepine, C.J. Impact of antibiotcs on arterial blood pressure in a patient with resistan hypertension—A case report. Int. J. Cardiol. 2015, 201, 157–158. [Google Scholar] [CrossRef] [Green Version]
- Sharma, R.K.; Yang, T.; Oliveira, A.C.; Lobaton, G.O.; Aquino, V.; Kim, S.; Richards, E.M.; Pepine, C.J.; Sumners, C.; Raizada, M.K. Microglial Cells Impact Gut Microbiota and Gut Pathology in Angiotensin II-Induced Hypertension. Circ. Res. 2019, 124, 727–736. [Google Scholar] [CrossRef]
- Toral, M.; Robles-Vera, I.; de la Visitación, N.; Romero, M.; Yang, T.; Sánchez, M.; Gómez-Guzmán, M.; Jiménez, R.; Raizada, M.K.; Duarte, J. Critical Role of the Interaction Gut Microbiota-Sympathetic Nervous System in the Regulation of Blood Pressure. Front. Physiol. 2019, 10, 231. [Google Scholar] [CrossRef] [Green Version]
- Robles-Vera, I.; de la Visitación, N.; Toral, M.; Sánchez, M.; Romero, M.; Gómez-Guzmán, M.; Yang, T.; Izquierdo-García, J.L.; Guerra-Hernández, E.; Ruiz-Cabello, J.; et al. Probiotic Bifidobacterium breve prevents DOCA-salt hypertension. FASEB J. 2020, 34, 13626–13640. [Google Scholar] [CrossRef]
- Robles-Vera, I.; de la Visitación, N.; Sánchez, M.; Gómez-Guzmán, M.; Jiménez, R.; Moleón, J.; González-Correa, C.; Romero, M.; Yang, T.; Raizada, M.K.; et al. Mycophenolate Improves Brain-Gut Axis Inducing Remodeling of Gut Microbiota in DOCA-Salt Hypertensive Rats. Antioxidants 2020, 9, 1199. [Google Scholar] [CrossRef]
- Jansson, P.A.; Larsson, A.; Lönnroth, P.N. Relationship between blood pressure, metabolic variables and blood flow in obese subjects with or without non-insulin-dependent diabetes mellitus. Eur. J. Clin. Investig. 1998, 28, 813–818. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Bower, J.K.; Selvin, E.; Subash Shantha, G.P.; Hoogeveen, R.C.; Ballantyne, C.M.; Young, J.H. Plasma lactate and incident hypertension in the atherosclerosis risk in communities study. Am. J. Hypertens. 2015, 28, 216–224. [Google Scholar] [CrossRef] [Green Version]
- Sapadin, A.N.; Fleischmajer, R. Tetracyclines: Nonantibiotic properties and their clinical implications. J. Am. Acad. Dermatol. 2006, 54, 258–265. [Google Scholar] [CrossRef]
- Garrido-Mesa, J.; Algieri, F.; Rodriguez-Nogales, A.; Utrilla, M.P.; Rodriguez-Cabezas, M.E.; Zarzuelo, A.; Ocete, M.A.; Garrido-Mesa, N.; Galvez, J. A new therapeutic association to manage relapsing experimental colitis: Doxycycline plus Saccharomyces boulardii. Pharmacol. Res. 2015, 97, 48–63. [Google Scholar] [CrossRef]
- Garrido-Mesa, J.; Algieri, F.; Rodríguez-Nogales, A.; Vezza, T.; Utrilla, M.P.; Garcia, F.; Chueca, N.; Rodríguez-Cabezas, M.E.; Garrido-Mesa, N.; Gálvez, J. Immunomodulatory tetracyclines ameliorate DNBS-colitis: Impact on microRNA expression and microbiota composition. Biochem. Pharmacol. 2018, 155, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Hummitzsch, L.; Zitta, K.; Berndt, R.; Kott, M.; Schildhauer, C.; Parczany, K.; Steinfath, M.; Albrecht, M. Doxycycline protects human intestinal cells from hypoxia/reoxygenation injury: Implications from an in-vitro hypoxia model. Exp. Cell Res. 2017, 353, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Grylls, A.; Seidler, K.; Neil, J. Link between microbiota and hypertension: Focus on LPS/TLR4 pathway in endothelial dysfunction and vascular inflammation, and therapeutic implication of probiotics. Biomed. Pharmacother. 2021, 137, 111334. [Google Scholar] [CrossRef] [PubMed]
- Suganya, K.; Son, T.; Kim, K.W.; Koo, B.S. Impact of gut microbiota: How it could play roles beyond the digestive system on development of cardiovascular and renal diseases. Microb. Pathog. 2021, 152, 104583. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, P.; La Rosa, M.; Barbera, C.; Arcoleo, F.; Di Bella, G.; Milano, S.; Cillari, E. Doxycycline reduces mortality to lethal endotoxemia by reducing nitric oxide synthesis via an interleukin-10-independent mechanism. J. Infect. Dis. 1998, 177, 489–492. [Google Scholar] [CrossRef] [Green Version]
- Bode, C.; Diedrich, B.; Muenster, S.; Hentschel, V.; Weisheit, C.; Rommelsheim, K.; Hoeft, A.; Meyer, R.; Boehm, O.; Knuefermann, P.; et al. Antibiotics regulate the immune response in both presence and absence of lipopolysaccharide through modulation of Toll-like receptors, cytokine production and phagocytosis in vitro. Int. Immunopharmacol. 2014, 18, 27–34. [Google Scholar] [CrossRef] [Green Version]
- Bomfim, G.F.; Dos Santos, R.A.; Oliveira, M.A.; Giachini, F.R.; Akamine, E.; Tostes, R.; Fortes, Z.B.; Webb, R.C.; Carvalho, M.H.C. Toll-like receptor 4 contributes to blood pressure regulation and vascular contraction in spontaneously hypertensive rats. Clin. Sci. 2012, 122, 535–543. [Google Scholar] [CrossRef] [Green Version]
- Nunes, K.P.; de Oliveira, A.A.; Lima, V.V.; Webb, R.C. Toll-like receptor 4 and blood pressure: Lessons from animal studies. Front. Physiol. 2019, 10, 655. [Google Scholar] [CrossRef] [PubMed]
- Amador, C.A.; Barrientos, V.; Pena, J.; Herrada, A.A.; Gonzalez, M.; Valdes, S.; Carrasco, L.; Alzamora, R.; Figueroa, F.; Kalergis, A.M.; et al. Spironolactone decreases DOCA-salt-induced organ damage by blocking the activation of T helper 17 and the downregulation of regulatory T lymphocytes. Hypertension 2014, 63, 797–803. [Google Scholar] [CrossRef] [Green Version]
- Du, Y.N.; Tang, X.F.; Xu, L.; Gao, P.J.; Han, W.Q. Th17/Treg Imbalance in Hypertension. Cardiovasc. Pharm. Open Access 2018, 7, 3. [Google Scholar] [CrossRef]
- Nguyen, H.; Chiasson, V.L.; Chatterjee, P.; Kopriva, S.E.; Young, K.J.; Mitchell, B.M. Interleukin-17 causes Rho-kinase-mediated endothelial dysfunction and hypertension. Cardiovasc. Res. 2013, 97, 696–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gunnett, C.A.; Heistad, D.D.; Berg, D.J.; Faraci, F.M. IL-10 deficiency increases superoxide and endothelial dysfunction during inflammation. Am. J. Physiol. Heart Circ. Physiol. 2000, 279, H1555–H1562. [Google Scholar] [CrossRef] [PubMed]
- Kasal, D.A.; Barhoumi, T.; Li, M.W.; Yamamoto, N.; Zdanovich, E.; Rehman, A.; Neves, M.F.; Laurant, P.; Paradis, P.; Schiffrin, E.L. T regulatory lymphocytes prevent aldosterone-induced vascular injury. Hypertension 2012, 59, 324–330. [Google Scholar] [CrossRef] [Green Version]
- Kassan, M.; Galan, M.; Partyka, M.; Trebak, M.; Matrougui, K. Interleukin-10 released by CD4(+)CD25(+) natural regulatory T cells improves microvascular endothelial function through inhibition of NADPH oxidase activity in hypertensive mice. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2534–2542. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robles-Vera, I.; de la Visitación, N.; Toral, M.; Sánchez, M.; Romero, M.; Gómez-Guzmán, M.; Vargas, F.; Duarte, J.; Jiménez, R. Changes in Gut Microbiota Induced by Doxycycline Influence in Vascular Function and Development of Hypertension in DOCA-Salt Rats. Nutrients 2021, 13, 2971. https://doi.org/10.3390/nu13092971
Robles-Vera I, de la Visitación N, Toral M, Sánchez M, Romero M, Gómez-Guzmán M, Vargas F, Duarte J, Jiménez R. Changes in Gut Microbiota Induced by Doxycycline Influence in Vascular Function and Development of Hypertension in DOCA-Salt Rats. Nutrients. 2021; 13(9):2971. https://doi.org/10.3390/nu13092971
Chicago/Turabian StyleRobles-Vera, Iñaki, Néstor de la Visitación, Marta Toral, Manuel Sánchez, Miguel Romero, Manuel Gómez-Guzmán, Félix Vargas, Juan Duarte, and Rosario Jiménez. 2021. "Changes in Gut Microbiota Induced by Doxycycline Influence in Vascular Function and Development of Hypertension in DOCA-Salt Rats" Nutrients 13, no. 9: 2971. https://doi.org/10.3390/nu13092971
APA StyleRobles-Vera, I., de la Visitación, N., Toral, M., Sánchez, M., Romero, M., Gómez-Guzmán, M., Vargas, F., Duarte, J., & Jiménez, R. (2021). Changes in Gut Microbiota Induced by Doxycycline Influence in Vascular Function and Development of Hypertension in DOCA-Salt Rats. Nutrients, 13(9), 2971. https://doi.org/10.3390/nu13092971









