Phytosterols, Cholesterol Control, and Cardiovascular Disease
Abstract
:1. Introduction
2. Phytosterols’ Chemistry
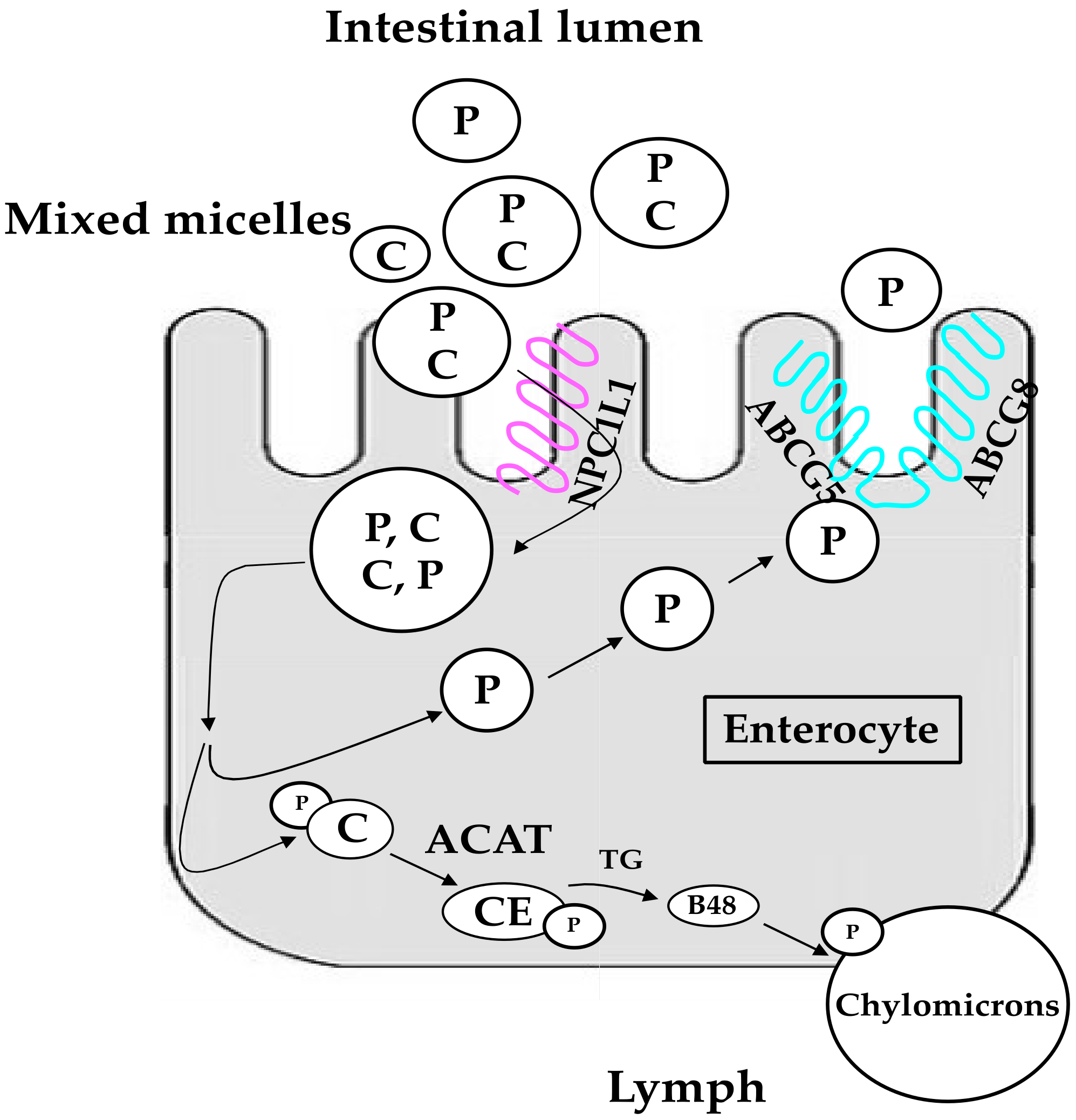
3. Human Metabolism and Metabolic Effects of Phytosterols
4. Effects of Phytosterols on Low Density Lipoprotein (LDL) Cholesterol: Characteristics and Clinical Relevance
5. Variables Affecting the Cholesterol-Lowering Effect of Phytosterols
6. Regulatory Framework
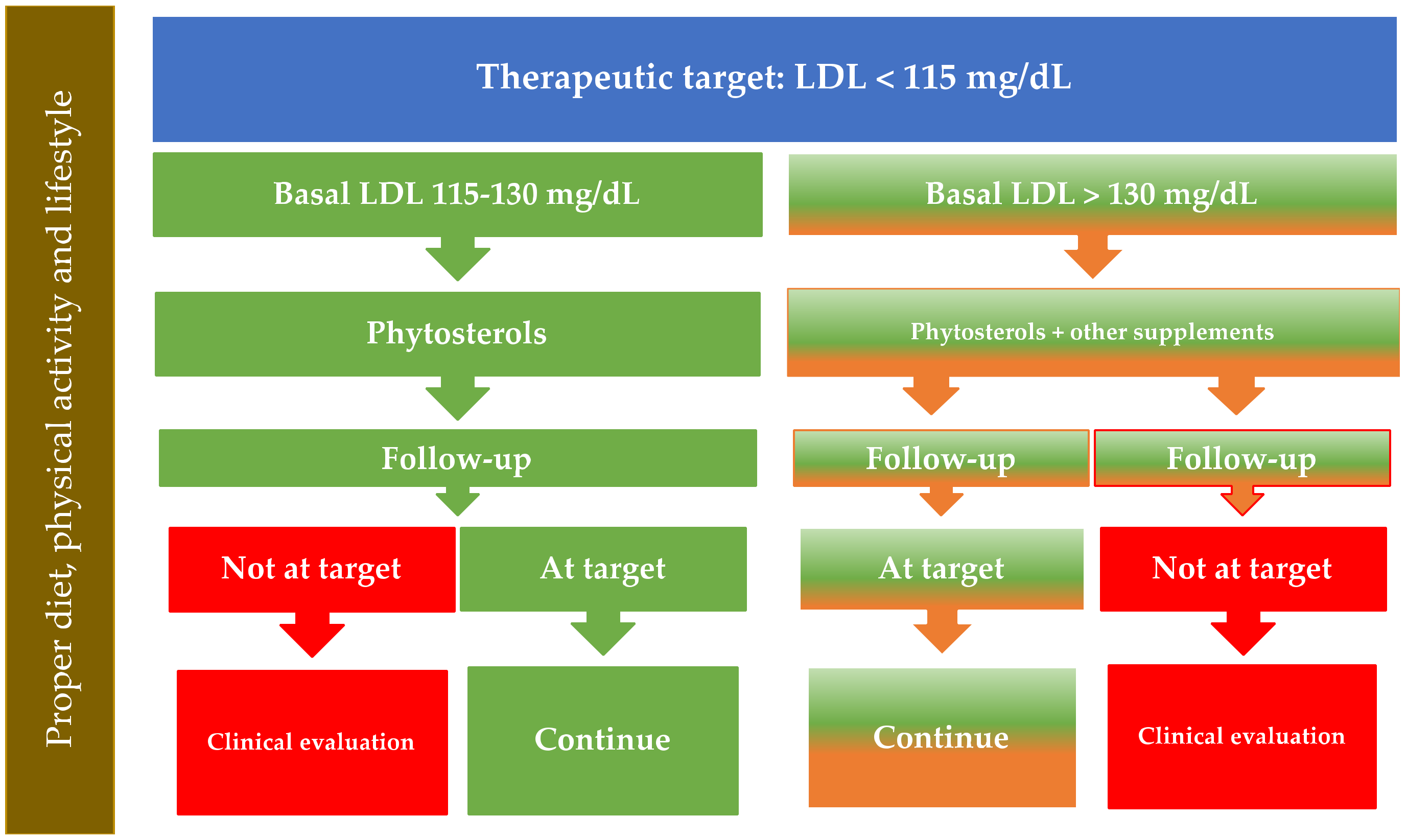
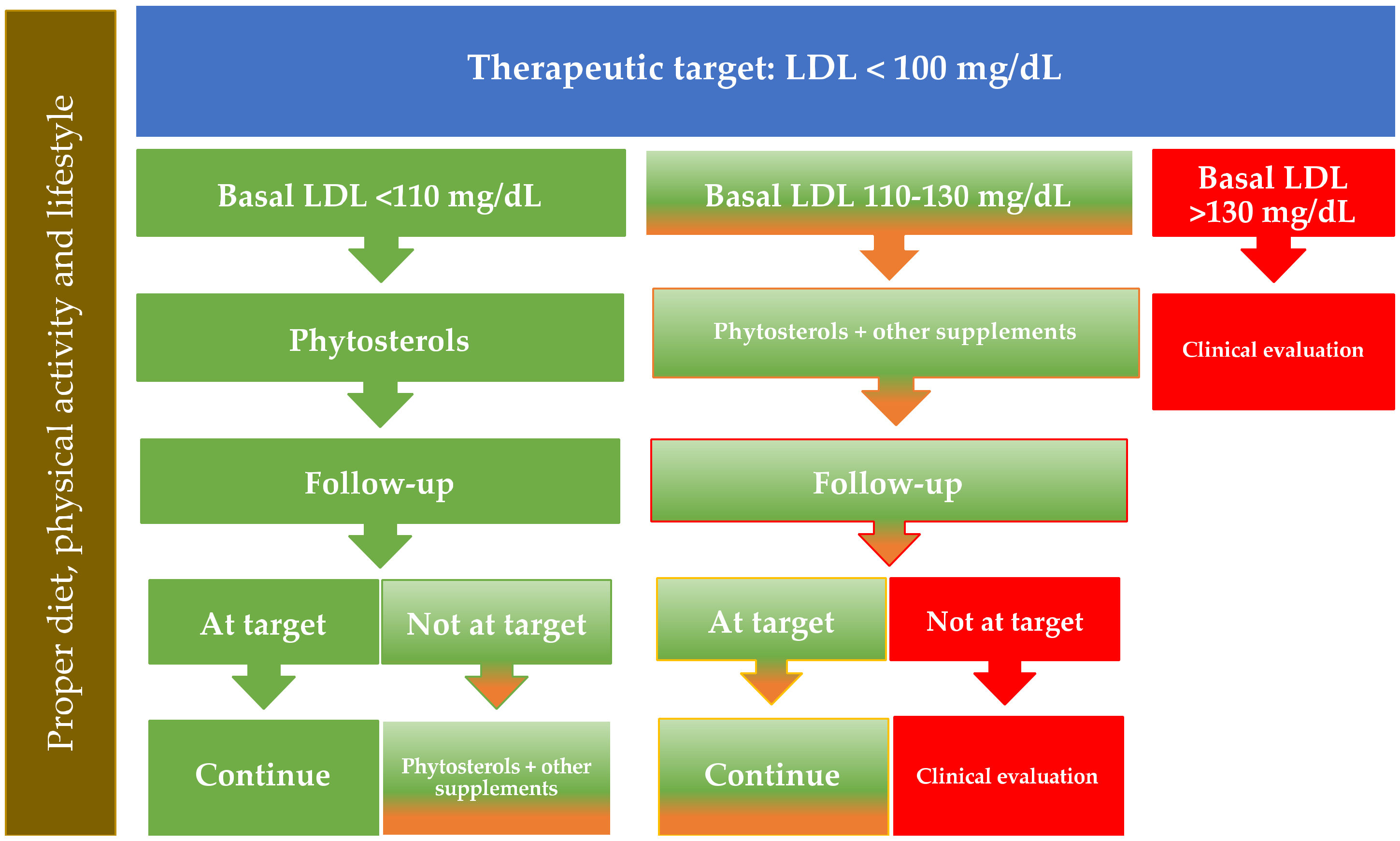
7. How to Identify Optimal Candidates for the Use of Phytosterols to Reduce LDL Cholesterol Levels
8. Side Effects of Phytosterols Use
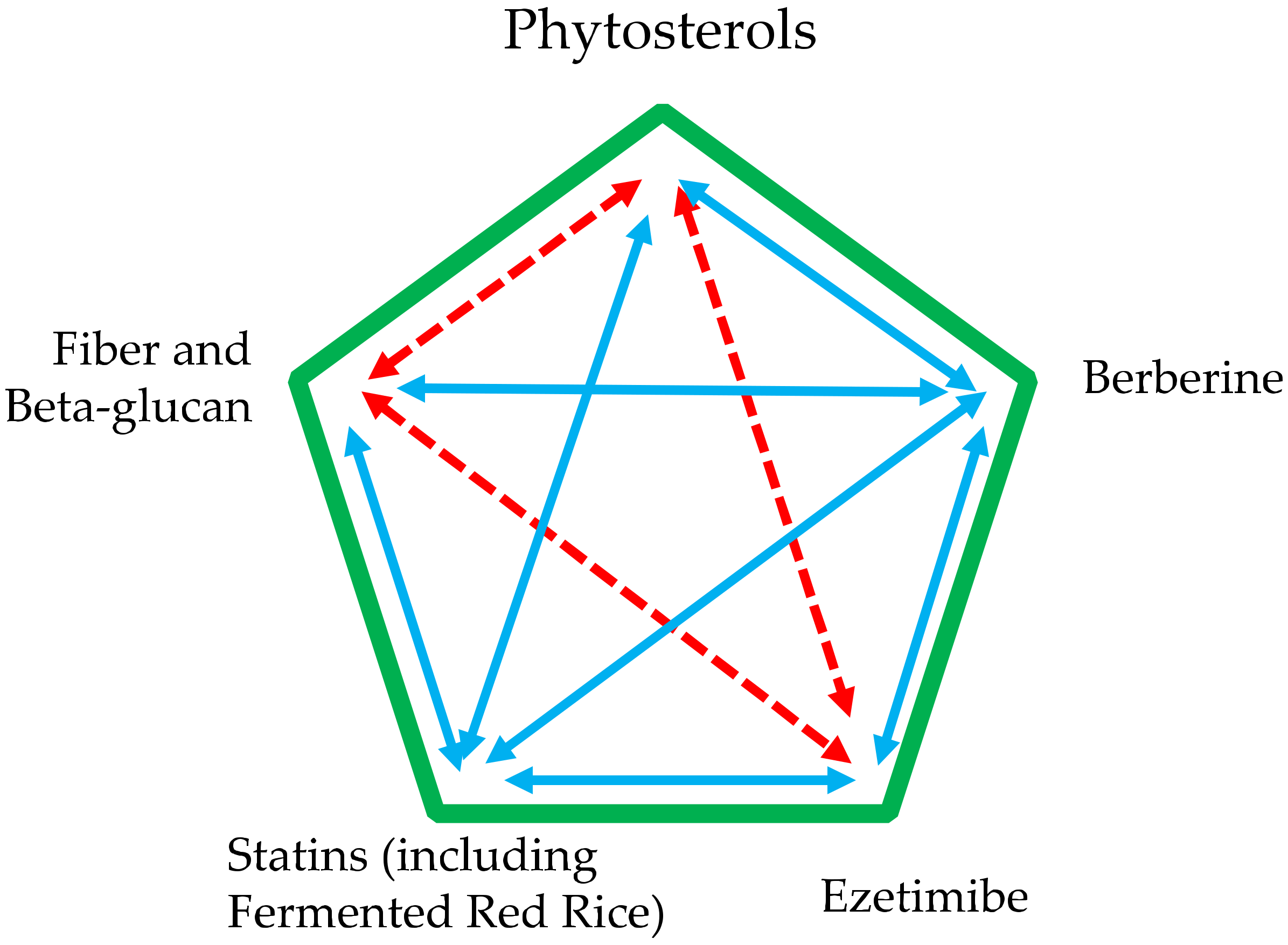
9. Use of Phytosterols in Addition to Other Supplements and Drugs
10. Conclusions and Practical Suggestions
Author Contributions
Funding
Conflicts of Interest
References
- Baumgartner, S.; Bruckert, E.; Gallo, A.; Plat, J. The position of functional foods and supplements with a serum LDL-C lowering effect in the spectrum ranging from universal to care-related CVD risk management. Atherosclerosis 2020, 311, 116–123. [Google Scholar] [CrossRef]
- Poli, A.; Barbagallo, C.M.; Cicero, A.F.G.; Corsini, A.; Manzato, E.; Trimarco, B.; Bernini, F.; Visioli, F.; Bianchi, A.; Canzone, G.; et al. Nutraceuticals and functional foods for the control of plasma cholesterol levels. An intersociety position paper. Pharm. Res. 2018, 134, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Fogacci, F.; Stoian, A.P.; Vrablik, M.; Al Rasadi, K.; Banach, M.; Toth, P.P.; Rizzo, M. Nutraceuticals in the Management of Dyslipidemia: Which, When, and for Whom? Could Nutraceuticals Help Low-Risk Individuals with Non-optimal Lipid Levels? Curr. Atheroscler. Rep. 2021, 23, 57. [Google Scholar] [CrossRef] [PubMed]
- Gylling, H.; Plat, J.; Turley, S.; Ginsberg, H.N.; Ellegard, L.; Jessup, W.; Jones, P.J.; Lutjohann, D.; Maerz, W.; Masana, L.; et al. Plant sterols and plant stanols in the management of dyslipidaemia and prevention of cardiovascular disease. Atherosclerosis 2014, 232, 346–360. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products Nutrition and Allergies. Scientific Opinion on the substantiation of a health claim related to 3 g/day plant sterols/stanols and lowering blood LDL-cholesterol and reduced risk of (coronary) heart disease pursuant to Article 19 of Regulation (EC) No 1924/2006. EFSA J. 2012, 10, 2693. [Google Scholar] [CrossRef] [Green Version]
- EFSA Panel on Dietetic Products Nutrition and Allergies. European Commission and a similar request from France in relation to the authorisation procedure for health claims on plant sterols/stanols and lowering/reducing blood LDL-cholesterol pursuant to Article 14 of Regulation (EC) No 1924/2006. EFSA J. 2009, 1175, 1–9. [Google Scholar]
- Moreau, R.A.; Nystrom, L.; Whitaker, B.D.; Winkler-Moser, J.K.; Baer, D.J.; Gebauer, S.K.; Hicks, K.B. Phytosterols and their derivatives: Structural diversity, distribution, metabolism, analysis, and health-promoting uses. Prog. Lipid Res. 2018, 70, 35–61. [Google Scholar] [CrossRef]
- Gylling, H.; Simonen, P. Phytosterols, Phytostanols, and Lipoprotein Metabolism. Nutrients 2015, 7, 7965–7977. [Google Scholar] [CrossRef] [Green Version]
- Feng, S.; Belwal, T.; Li, L.; Limwachiranon, J.; Liu, X.; Luo, Z. Phytosterols and their derivatives: Potential health-promoting uses against lipid metabolism and associated diseases, mechanism, and safety issues. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1243–1267. [Google Scholar] [CrossRef] [PubMed]
- Vezza, T.; Canet, F.; de Maranon, A.M.; Banuls, C.; Rocha, M.; Victor, V.M. Phytosterols: Nutritional Health Players in the Management of Obesity and Its Related Disorders. Antioxidants 2020, 9, 1266. [Google Scholar] [CrossRef]
- Ostlund, R.E., Jr.; McGill, J.B.; Zeng, C.M.; Covey, D.F.; Stearns, J.; Stenson, W.F.; Spilburg, C.A. Gastrointestinal absorption and plasma kinetics of soy Delta(5)-phytosterols and phytostanols in humans. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E911–E916. [Google Scholar] [CrossRef]
- Jimenez-Escrig, A.; Santos-Hidalgo, A.B.; Saura-Calixto, F. Common sources and estimated intake of plant sterols in the Spanish diet. J. Agric. Food Chem. 2006, 54, 3462–3471. [Google Scholar] [CrossRef] [Green Version]
- Esche, R.; Muller, L.; Engel, K.H. Online LC-GC-based analysis of minor lipids in various tree nuts and peanuts. J. Agric. Food Chem. 2013, 61, 11636–11644. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Huang, W.; Hu, Y.; Zhang, L.; Shao, Y.; Wang, M.; Zhang, F.; Zhao, Z.; Mei, X.; Li, T.; et al. Phytosterol Profiles of Common Foods and Estimated Natural Intake of Different Structures and Forms in China. J. Agric. Food Chem. 2018, 66, 2669–2676. [Google Scholar] [CrossRef]
- Ras, R.T.; van der Schouw, Y.T.; Trautwein, E.A.; Sioen, I.; Dalmeijer, G.W.; Zock, P.L.; Beulens, J.W. Intake of phytosterols from natural sources and risk of cardiovascular disease in the European Prospective Investigation into Cancer and Nutrition-the Netherlands (EPIC-NL) population. Eur. J. Prev. Cardiol. 2015, 22, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Racette, S.B.; Spearie, C.A.; Phillips, K.M.; Lin, X.; Ma, L.; Ostlund, R.E., Jr. Phytosterol-deficient and high-phytosterol diets developed for controlled feeding studies. J. Am. Diet. Assoc. 2009, 109, 2043–2051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berge, K.E.; Tian, H.; Graf, G.A.; Yu, L.; Grishin, N.V.; Schultz, J.; Kwiterovich, P.; Shan, B.; Barnes, R.; Hobbs, H.H. Accumulation of dietary cholesterol in sitosterolemia caused by mutations in adjacent ABC transporters. Science 2000, 290, 1771–1775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ostlund, R.E., Jr. Phytosterols and cholesterol metabolism. Curr. Opin. Lipidol. 2004, 15, 37–41. [Google Scholar] [CrossRef]
- Huff, M.W.; Pollex, R.L.; Hegele, R.A. NPC1L1: Evolution from pharmacological target to physiological sterol transporter. Arter. Thromb. Vasc. Biol. 2006, 26, 2433–2438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bosner, M.S.; Lange, L.G.; Stenson, W.F.; Ostlund, R.E., Jr. Percent cholesterol absorption in normal women and men quantified with dual stable isotopic tracers and negative ion mass spectrometry. J. Lipid Res. 1999, 40, 302–308. [Google Scholar] [CrossRef]
- Klingberg, S.; Ellegard, L.; Johansson, I.; Hallmans, G.; Weinehall, L.; Andersson, H.; Winkvist, A. Inverse relation between dietary intake of naturally occurring plant sterols and serum cholesterol in northern Sweden. Am. J. Clin. Nutr. 2008, 87, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Katan, M.B.; Grundy, S.M.; Jones, P.; Law, M.; Miettinen, T.; Paoletti, R.; Stresa Workshop, P. Efficacy and safety of plant stanols and sterols in the management of blood cholesterol levels. Mayo Clin. Proc. 2003, 78, 965–978. [Google Scholar] [CrossRef] [Green Version]
- Ras, R.T.; Geleijnse, J.M.; Trautwein, E.A. LDL-cholesterol-lowering effect of plant sterols and stanols across different dose ranges: A meta-analysis of randomised controlled studies. Br. J. Nutr. 2014, 112, 214–219. [Google Scholar] [CrossRef] [Green Version]
- Ying, J.; Zhang, Y.; Yu, K. Phytosterol compositions of enriched products influence their cholesterol-lowering efficacy: A meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2019, 73, 1579–1593. [Google Scholar] [CrossRef] [PubMed]
- Abumweis, S.S.; Barake, R.; Jones, P.J. Plant sterols/stanols as cholesterol lowering agents: A meta-analysis of randomized controlled trials. Food Nutr. Res. 2008, 52. [Google Scholar] [CrossRef]
- Moruisi, K.G.; Oosthuizen, W.; Opperman, A.M. Phytosterols/stanols lower cholesterol concentrations in familial hypercholesterolemic subjects: A systematic review with meta-analysis. J. Am. Coll. Nutr. 2006, 25, 41–48. [Google Scholar] [CrossRef]
- Demonty, I.; Ras, R.T.; van der Knaap, H.C.; Meijer, L.; Zock, P.L.; Geleijnse, J.M.; Trautwein, E.A. The effect of plant sterols on serum triglyceride concentrations is dependent on baseline concentrations: A pooled analysis of 12 randomised controlled trials. Eur. J. Nutr. 2013, 52, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Demonty, I.; Ras, R.T.; van der Knaap, H.C.; Duchateau, G.S.; Meijer, L.; Zock, P.L.; Geleijnse, J.M.; Trautwein, E.A. Continuous dose-response relationship of the LDL-cholesterol-lowering effect of phytosterol intake. J. Nutr. 2009, 139, 271–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocha, V.Z.; Ras, R.T.; Gagliardi, A.C.; Mangili, L.C.; Trautwein, E.A.; Santos, R.D. Effects of phytosterols on markers of inflammation: A systematic review and meta-analysis. Atherosclerosis 2016, 248, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Scarmozzino, F.; Poli, A.; Visioli, F. Microbiota and cardiovascular disease risk: A scoping review. Pharmacol. Res. 2020, 159, 104952. [Google Scholar] [CrossRef]
- Cuevas-Tena, M.; Bermudez, J.D.; Silvestre, R.L.A.; Alegria, A.; Lagarda, M.J. Impact of colonic fermentation on sterols after the intake of a plant sterol-enriched beverage: A randomized, double-blind crossover trial. Clin. Nutr. 2019, 38, 1549–1560. [Google Scholar] [CrossRef] [PubMed]
- Gibney, E.R.; Milenkovic, D.; Combet, E.; Ruskovska, T.; Greyling, A.; Gonzalez-Sarrias, A.; de Roos, B.; Tomas-Barberan, F.; Morand, C.; Rodriguez-Mateos, A. Factors influencing the cardiometabolic response to (poly)phenols and phytosterols: A review of the COST Action POSITIVe activities. Eur. J. Nutr. 2019, 58, 37–47. [Google Scholar] [CrossRef] [Green Version]
- Gylling, H.; Strandberg, T.E.; Kovanen, P.T.; Simonen, P. Lowering Low-Density Lipoprotein Cholesterol Concentration with Plant Stanol Esters to Reduce the Risk of Atherosclerotic Cardiovascular Disease Events at a Population Level: A Critical Discussion. Nutrients 2020, 12, 2346. [Google Scholar] [CrossRef]
- Myocardial Infarction Genetics Consortium, I.; Stitziel, N.O.; Won, H.H.; Morrison, A.C.; Peloso, G.M.; Do, R.; Lange, L.A.; Fontanillas, P.; Gupta, N.; Duga, S.; et al. Inactivating mutations in NPC1L1 and protection from coronary heart disease. N. Engl. J. Med. 2014, 371, 2072–2082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matthan, N.R.; Pencina, M.; LaRocque, J.M.; Jacques, P.F.; D’Agostino, R.B.; Schaefer, E.J.; Lichtenstein, A.H. Alterations in cholesterol absorption/synthesis markers characterize Framingham offspring study participants with CHD. J. Lipid. Res. 2009, 50, 1927–1935. [Google Scholar] [CrossRef] [Green Version]
- Silbernagel, G.; Chapman, M.J.; Genser, B.; Kleber, M.E.; Fauler, G.; Scharnagl, H.; Grammer, T.B.; Boehm, B.O.; Makela, K.M.; Kahonen, M.; et al. High intestinal cholesterol absorption is associated with cardiovascular disease and risk alleles in ABCG8 and ABO: Evidence from the LURIC and YFS cohorts and from a meta-analysis. J. Am. Coll. Cardiol. 2013, 62, 291–299. [Google Scholar] [CrossRef]
- Rogacev, K.S.; Pinsdorf, T.; Weingartner, O.; Gerhart, M.K.; Welzel, E.; van Bentum, K.; Popp, J.; Menzner, A.; Fliser, D.; Lutjohann, D.; et al. Cholesterol synthesis, cholesterol absorption, and mortality in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2012, 7, 943–948. [Google Scholar] [CrossRef] [Green Version]
- Emrich, I.E.; Heine, G.H.; Schulze, P.C.; Rogacev, K.S.; Fliser, D.; Wagenpfeil, S.; Bohm, M.; Lutjohann, D.; Weingartner, O. Markers of cholesterol synthesis to cholesterol absorption across the spectrum of non-dialysis CKD: An observational study. Pharm. Res. Perspect. 2021, 9, e00801. [Google Scholar] [CrossRef]
- Sato, K.; Nakano, K.; Katsuki, S.; Matoba, T.; Osada, K.; Sawamura, T.; Sunagawa, K.; Egashira, K. Dietary cholesterol oxidation products accelerate plaque destabilization and rupture associated with monocyte infiltration/activation via the MCP-1-CCR2 pathway in mouse brachiocephalic arteries: Therapeutic effects of ezetimibe. J. Atheroscler. Thromb. 2012, 19, 986–998. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Xu, M.; Fang, Y.J.; Lu, M.S.; Pan, Z.Z.; Huang, W.Q.; Chen, Y.M.; Zhang, C.X. Association between phytosterol intake and colorectal cancer risk: A case-control study. Br. J. Nutr. 2017, 117, 839–850. [Google Scholar] [CrossRef] [Green Version]
- Rideout, T.C.; Harding, S.V.; Mackay, D.; Abumweis, S.S.; Jones, P.J. High basal fractional cholesterol synthesis is associated with nonresponse of plasma LDL cholesterol to plant sterol therapy. Am. J. Clin. Nutr. 2010, 92, 41–46. [Google Scholar] [CrossRef] [Green Version]
- MacKay, D.S.; Eck, P.K.; Gebauer, S.K.; Baer, D.J.; Jones, P.J. CYP7A1-rs3808607 and APOE isoform associate with LDL cholesterol lowering after plant sterol consumption in a randomized clinical trial. Am. J. Clin. Nutr. 2015, 102, 951–957. [Google Scholar] [CrossRef] [Green Version]
- Trautwein, E.A.; Vermeer, M.A.; Hiemstra, H.; Ras, R.T. LDL-Cholesterol Lowering of Plant Sterols and Stanols-Which Factors Influence Their Efficacy? Nutrients 2018, 10, 1262. [Google Scholar] [CrossRef] [Green Version]
- Racette, S.B.; Lin, X.; Lefevre, M.; Spearie, C.A.; Most, M.M.; Ma, L.; Ostlund, R.E., Jr. Dose effects of dietary phytosterols on cholesterol metabolism: A controlled feeding study. Am. J. Clin. Nutr. 2010, 91, 32–38. [Google Scholar] [CrossRef] [Green Version]
- Amir Shaghaghi, M.; Abumweis, S.S.; Jones, P.J. Cholesterol-lowering efficacy of plant sterols/stanols provided in capsule and tablet formats: Results of a systematic review and meta-analysis. J. Acad. Nutr. Diet. 2013, 113, 1494–1503. [Google Scholar] [CrossRef]
- Cusack, L.K.; Fernandez, M.L.; Volek, J.S. The food matrix and sterol characteristics affect the plasma cholesterol lowering of phytosterol/phytostanol. Adv. Nutr. 2013, 4, 633–643. [Google Scholar] [CrossRef] [Green Version]
- Plat, J.; van Onselen, E.N.; van Heugten, M.M.; Mensink, R.P. Effects on serum lipids, lipoproteins and fat soluble antioxidant concentrations of consumption frequency of margarines and shortenings enriched with plant stanol esters. Eur. J. Clin. Nutr. 2000, 54, 671–677. [Google Scholar] [CrossRef] [Green Version]
- Doornbos, A.M.; Meynen, E.M.; Duchateau, G.S.; van der Knaap, H.C.; Trautwein, E.A. Intake occasion affects the serum cholesterol lowering of a plant sterol-enriched single-dose yoghurt drink in mildly hypercholesterolaemic subjects. Eur. J. Clin. Nutr. 2006, 60, 325–333. [Google Scholar] [CrossRef]
- Lenssen, K.G.M.; Bast, A.; de Boer, A. Should botanical health claims be substantiated with evidence on traditional use? Reviewing the stakeholders’ arguments. PharmaNutrition 2020, 14, 100232. [Google Scholar] [CrossRef]
- Van der Waal, M.B.; Veldhuizen, C.K.; van der Waal, R.X.; Claassen, E.; van der Burgwal, L.H.M. A critical appreciation of intangible resources in PharmaNutrition. PharmaNutrition 2020, 13, 100208. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corra, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts) Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef] [PubMed]
- Stock, J. Focus on lifestyle: EAS Consensus Panel Position Statement on Phytosterol-added Foods. Atherosclerosis 2014, 234, 142–145. [Google Scholar] [CrossRef]
- Assmann, G.; Cullen, P.; Erbey, J.; Ramey, D.R.; Kannenberg, F.; Schulte, H. Plasma sitosterol elevations are associated with an increased incidence of coronary events in men: Results of a nested case-control analysis of the Prospective Cardiovascular Munster (PROCAM) study. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 13–21. [Google Scholar] [CrossRef]
- Willems, J.I.; Blommaert, M.A.; Trautwein, E.A. Results from a post-launch monitoring survey on consumer purchases of foods with added phytosterols in five European countries. Food Chem. Toxicol. 2013, 62, 48–53. [Google Scholar] [CrossRef]
- Ras, R.T.; Trautwein, E.A. Consumer purchase behaviour of foods with added phytosterols in six European countries: Data from a post-launch monitoring survey. Food Chem. Toxicol. 2017, 110, 42–48. [Google Scholar] [CrossRef]
- Bays, H.E.; Neff, D.; Tomassini, J.E.; Tershakovec, A.M. Ezetimibe: Cholesterol lowering and beyond. Expert Rev. Cardiovasc. Ther. 2008, 6, 447–470. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS); Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Dusemund, B.; Filipic, M.; Frutos, M.J.; Galtier, P.; Gott, D.; et al. Scientific opinion on the safety of monacolins in red yeast rice. EFSA J. 2018, 16, e05368. [Google Scholar] [CrossRef] [Green Version]
- Erguson, J.J.; Stojanovski, E.; MacDonald-Wicks, L.; Garg, M.L. High molecular weight oat beta-glucan enhances lipid-lowering effects of phytosterols. A randomised controlled trial. Clin. Nutr. 2020, 39, 80–89. [Google Scholar] [CrossRef]
- Han, S.; Jiao, J.; Xu, J.; Zimmermann, D.; Actis-Goretta, L.; Guan, L.; Zhao, Y.; Qin, L. Effects of plant stanol or sterol-enriched diets on lipid profiles in patients treated with statins: Systematic review and meta-analysis. Sci. Rep. 2016, 6, 31337. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.E.; Moore, R.A. Statins in hypercholesterolaemia: A dose-specific meta-analysis of lipid changes in randomised, double blind trials. BMC Fam. Pract. 2003, 4, 18. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Jia, X.; Ghanam, K.; Beaurepaire, C.; Zidichouski, J.; Miller, L. Berberine and plant stanols synergistically inhibit cholesterol absorption in hamsters. Atherosclerosis 2010, 209, 111–117. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Colletti, A.; Bajraktari, G.; Descamps, O.; Djuric, D.M.; Ezhov, M.; Fras, Z.; Katsiki, N.; Langlois, M.; Latkovskis, G.; et al. Lipid-lowering nutraceuticals in clinical practice: Position paper from an International Lipid Expert Panel. Nutr. Rev. 2017, 75, 731–767. [Google Scholar] [CrossRef] [PubMed]
- Cofan, M.; Ros, E. Use of Plant Sterol and Stanol Fortified Foods in Clinical Practice. Curr. Med. Chem. 2019, 26, 6691–6703. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, F.; Poli, A. Phytosterols and cardiovascular health. Pharm. Res. 2010, 61, 193–199. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poli, A.; Marangoni, F.; Corsini, A.; Manzato, E.; Marrocco, W.; Martini, D.; Medea, G.; Visioli, F. Phytosterols, Cholesterol Control, and Cardiovascular Disease. Nutrients 2021, 13, 2810. https://doi.org/10.3390/nu13082810
Poli A, Marangoni F, Corsini A, Manzato E, Marrocco W, Martini D, Medea G, Visioli F. Phytosterols, Cholesterol Control, and Cardiovascular Disease. Nutrients. 2021; 13(8):2810. https://doi.org/10.3390/nu13082810
Chicago/Turabian StylePoli, Andrea, Franca Marangoni, Alberto Corsini, Enzo Manzato, Walter Marrocco, Daniela Martini, Gerardo Medea, and Francesco Visioli. 2021. "Phytosterols, Cholesterol Control, and Cardiovascular Disease" Nutrients 13, no. 8: 2810. https://doi.org/10.3390/nu13082810
APA StylePoli, A., Marangoni, F., Corsini, A., Manzato, E., Marrocco, W., Martini, D., Medea, G., & Visioli, F. (2021). Phytosterols, Cholesterol Control, and Cardiovascular Disease. Nutrients, 13(8), 2810. https://doi.org/10.3390/nu13082810







