Sex Differences in Supplement Intake in Recreational Endurance Runners—Results from the NURMI Study (Step 2)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Ethical Approval
2.2. Participants and Experimental Approach
2.3. Data Clearance
2.4. Measures
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rossi, K.A. Nutritional aspects of the female athlete. Clin. Sports Med. 2017, 36, 627–653. [Google Scholar] [CrossRef]
- Hull, M.V.; Jagim, A.R.; Oliver, J.M.; Greenwood, M.; Busteed, D.R.; Jones, M.T. Gender differences and access to a sports dietitian influence dietary habits of collegiate athletes. J. Int. Soc. Sports Nutr. 2016, 13, 38. [Google Scholar] [CrossRef] [Green Version]
- Devries, M.C. Sex-based differences in endurance exercise muscle metabolism: Impact on exercise and nutritional strategies to optimize health and performance in women. Exp. Physiol. 2016, 101, 243–249. [Google Scholar] [CrossRef]
- Tiller, N.B.; Elliott-Sale, K.J.; Knechtle, B.; Wilson, P.B.; Roberts, J.D.; Millet, G.Y. Do sex differences in physiology confer a female advantage in ultra-endurance sport? Sports Med. 2021, 51, 895–915. [Google Scholar] [CrossRef]
- Cheuvront, S.N.; Carter, R.; Deruisseau, K.C.; Moffatt, R.J. Running performance differences between men and women: An update. Sports Med. 2005, 35, 1017–1024. [Google Scholar] [CrossRef]
- Burke, L.M.; Jeukendrup, A.E.; Jones, A.M.; Mooses, M. Contemporary nutrition strategies to optimize performance in distance runners and race walkers. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 117–129. [Google Scholar] [CrossRef] [Green Version]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and athletic performance. J. Acad. Nutr. Diet. 2016, 116, 501–528. [Google Scholar] [CrossRef] [PubMed]
- Burrows, M.; Bird, S. The physiology of the highly trained female endurance runner. Sports Med. 2000, 30, 281–300. [Google Scholar] [CrossRef] [PubMed]
- Deldicque, L.; Francaux, M. Recommendations for healthy nutrition in female endurance runners: An update. Front. Nutr. 2015, 2, 17. [Google Scholar] [CrossRef]
- Devries, M.C.; Hamadeh, M.J.; Phillips, S.M.; Tarnopolsky, M.A. Menstrual cycle phase and sex influence muscle glycogen utilization and glucose turnover during moderate-intensity endurance exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, R1120–R1128. [Google Scholar] [CrossRef] [Green Version]
- Tarnopolsky, M.A.; Rennie, C.D.; Robertshaw, H.A.; Fedak-Tarnopolsky, S.N.; Devries, M.C.; Hamadeh, M.J. Influence of endurance exercise training and sex on intramyocellular lipid and mitochondrial ultrastructure, substrate use, and mitochondrial enzyme activity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, R1271–R1278. [Google Scholar] [CrossRef] [Green Version]
- Fortunato, R.S.; Ferreira, A.C.; Hecht, F.; Dupuy, C.; Carvalho, D.P. Sexual dimorphism and thyroid dysfunction: A matter of oxidative stress? J. Endocrinol. 2014, 221, R31–R40. [Google Scholar] [CrossRef]
- Duhig, T.J.; McKeag, D. Thyroid disorders in athletes. Curr. Sports Med. Rep. 2009, 8, 16–19. [Google Scholar] [CrossRef] [Green Version]
- Dunn, D.; Turner, C. Hypothyroidism in women. Nurs. Womens Health 2016, 20, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Maas, A.H.; Appelman, Y.E. Gender differences in coronary heart disease. Neth. Heart J. 2010, 18, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Wasfy, M.M.; Hutter, A.M.; Weiner, R.B. Sudden cardiac death in athletes. Methodist Debakey Cardiovasc. J. 2016, 12, 76–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leynaert, B.; Sunyer, J.; Garcia-Esteban, R.; Svanes, C.; Jarvis, D.; Cerveri, I.; Dratva, J.; Gislason, T.; Heinrich, J.; Janson, C.; et al. Gender differences in prevalence, diagnosis and incidence of allergic and non-allergic asthma: A population-based cohort. Thorax 2012, 67, 625–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Wall, E.E. Long-distance running: Running for a long life? Neth. Heart J. 2014, 22, 89–90. [Google Scholar] [CrossRef] [Green Version]
- Zeiger, J.S.; Zeiger, R.S. Mental toughness latent profiles in endurance athletes. PLoS ONE 2018, 13, e0193071. [Google Scholar] [CrossRef]
- Dandoy, C.; Gereige, R.S. Performance-enhancing drugs. Pediatr. Rev. 2012, 33, 265–272. [Google Scholar] [CrossRef]
- Yusko, D.A.; Buckman, J.F.; White, H.R.; Pandina, R.J. Alcohol, tobacco, illicit drugs, and performance enhancers: A comparison of use by college student athletes and nonathletes. J. Am. Coll. Health 2008, 57, 281–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wirnitzer, K.; Boldt, P.; Lechleitner, C.; Wirnitzer, G.; Leitzmann, C.; Rosemann, T.; Knechtle, B. Health status of female and male vegetarian and vegan endurance runners compared to omnivores-results from the NURMI study (step 2). Nutrients 2018, 11, 29. [Google Scholar] [CrossRef] [Green Version]
- Regitz-Zagrosek, V. Sex and gender differences in health. Science & Society Series on Sex and Science. EMBO Rep. 2012, 13, 596–603. [Google Scholar] [CrossRef] [Green Version]
- Bredella, M.A. Sex differences in body composition. Adv. Exp. Med. Biol. 2017, 1043, 9–27. [Google Scholar] [CrossRef] [PubMed]
- Boldt, P.; Knechtle, B.; Nikolaidis, P.; Lechleitner, C.; Wirnitzer, G.; Leitzmann, C.; Wirnitzer, K. Sex differences in the health status of endurance runners: Results from the NURMI Study (Step 2). J. Strength Cond. Res. 2019, 33, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Heydenreich, J.; Kayser, B.; Schutz, Y.; Melzer, K. Total energy expenditure, energy intake, and body composition in endurance athletes across the training season: A systematic review. Sports Med. Open 2017, 3, 8. [Google Scholar] [CrossRef] [Green Version]
- Wardle, J.; Haase, A.M.; Steptoe, A.; Nillapun, M.; Jonwutiwes, K.; Bellisle, F. Gender differences in food choice: The contribution of health beliefs and dieting. Ann. Behav. Med. 2004, 27, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Rauma, A.L.; Mykkänen, H. Antioxidant status in vegetarians versus omnivores. Nutrition 2000, 16, 111–119. [Google Scholar] [CrossRef]
- Nieman, D. Vegetarian dietary practices and endurance performance. Am. J. Clin. Nutr. 1988, 48, 754–761. [Google Scholar] [CrossRef]
- Kårlund, A.; Gómez-Gallego, C.; Turpeinen, A.M.; Palo-Oja, O.M.; El-Nezami, H.; Kolehmainen, M. Protein supplements and their relation with nutrition, microbiota composition and health: Is more protein always better for sportspeople? Nutrients 2019, 11, 829. [Google Scholar] [CrossRef] [Green Version]
- Wirnitzer, K.C.; Kornexl, E. Energy and macronutrient intake of a female vegan cyclist during an 8-day mountain bike stage race. Bayl. Univ. Med. Cent. 2014, 27, 42–45. [Google Scholar] [CrossRef]
- Tiller, N.B.; Roberts, J.D.; Beasley, L.; Chapman, S.; Pinto, J.M.; Smith, L.; Wiffin, M.; Russell, M.; Sparks, S.A.; Duckworth, L.; et al. International Society of Sports Nutrition Position Stand: Nutritional considerations for single-stage ultra-marathon training and racing. J. Int. Soc. Sports Nutr. 2019, 16, 50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Black, K.; Slater, J.; Brown, R.C.; Cooke, R. Low energy availability, plasma lipids, and hormonal profiles of recreational athletes. J. Strength Cond. Res. 2018, 32, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, P.T.; Veniamakis, E.; Rosemann, T.; Knechtle, B. Nutrition in ultra-endurance: State of the art. Nutrients 2018, 10, 1995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wirnitzer, K.C. Vegan diet in sports and exercise–health benefits and advantages to athletes and physically active people: A narrative review. Int. J. Sports Exerc. Med. 2020, 6, 165. [Google Scholar] [CrossRef]
- Schröder, S.; Fischer, A.; Vock, C.; Böhme, M.; Schmelzer, C.; Döpner, M.; Hülsmann, O.; Döring, F. Nutrition concepts for elite distance runners based on macronutrient and energy expenditure. J. Athl. Train. 2008, 43, 489–504. [Google Scholar] [CrossRef]
- Heaney, S.; O’Connor, H.; Michael, S.; Gifford, J.; Naughton, G. Nutrition knowledge in athletes: A systematic review. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 248–261. [Google Scholar] [CrossRef] [Green Version]
- Vitale, K.; Getzin, A. Nutrition and supplement update for the endurance athlete: Review and recommendations. Nutrients 2019, 11, 1289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenner, S.L.; Buckley, G.L.; Belski, R.; Devlin, B.L.; Forsyth, A.K. Dietary intakes of professional and semi-professional team sport athletes do not meet sport nutrition recommendations-a systematic literature review. Nutrients 2019, 11, 1160. [Google Scholar] [CrossRef] [Green Version]
- Lentjes, M.A.H. The balance between food and dietary supplements in the general population. Proc. Nutr. Soc. 2019, 78, 97–109. [Google Scholar] [CrossRef] [Green Version]
- Schwingshackl, L.; Boeing, H.; Stelmach-Mardas, M.; Gottschald, M.; Dietrich, S.; Hoffmann, G.; Chaimani, A. Dietary sup-plements and risk of cause-specific death, cardiovascular disease, and cancer: A systematic review and meta-analysis of pri-mary prevention trials. Adv. Nutr. 2017, 8, 27–39. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Di-etary supplements and the high-performance athlete. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 104–125. [Google Scholar] [CrossRef] [Green Version]
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN exercise & sports nutrition review update: Research & recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 38. [Google Scholar] [CrossRef] [Green Version]
- Carlsohn, A.; Cassel, M.; Linné, K.; Mayer, F. How much is too much? a case report of nutritional supplement use of a high-performance athlete. Br. J. Nutr. 2011, 105, 1724–1728. [Google Scholar] [CrossRef] [PubMed]
- Parnell, J.A.; Wagner-Jones, K.; Madden, R.F.; Erdman, K.A. Dietary restrictions in endurance runners to mitigate exercise-induced gastrointestinal symptoms. J. Int. Soc. Sports Nutr. 2020, 17, 32. [Google Scholar] [CrossRef] [PubMed]
- Grimble, G.K. Adverse gastrointestinal effects of arginine and related amino acids. J. Nutr. 2007, 137, 1693–1701. [Google Scholar] [CrossRef] [Green Version]
- Giannopoulou, I.; Noutsos, K.; Apostolidis, N.; Bayios, I.; Nassis, G.P. Performance level affects the dietary supplement intake of both individual and team sports athletes. J. Sports Sci. Med. 2013, 12, 190–196. [Google Scholar] [PubMed]
- Nieper, A. Nutritional supplement practices in UK junior national track and field athletes. Br. J. Sports Med. 2005, 39, 645–649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nebl, J.; Schuchardt, J.P.; Wasserfurth, P.; Haufe, S.; Eigendorf, J.; Tegtbur, U.; Hahn, A. Characterization, dietary habits and nutritional intake of omnivorous, lacto-ovo vegetarian and vegan runners–a pilot study. BMC Nutr. 2019, 5, 51. [Google Scholar] [CrossRef] [Green Version]
- Wirnitzer, K.; Seyfart, T.; Leitzmann, C.; Keller, M.; Wirnitzer, G.; Lechleitner, C.; Rüst, C.A.; Rosemann, T.; Knechtle, B. Prevalence in running events and running performance of endurance runners following a vegetarian or vegan diet compared to non-vegetarian endurance runners: The NURMI study. SpringerPlus 2016, 5, 458. [Google Scholar] [CrossRef] [Green Version]
- Boldt, P.; Knechtle, B.; Nikolaidis, P.; Lechleitner, C.; Wirnitzer, G.; Leitzmann, C.; Rosemann, T.; Wirnitzer, K. Quality of life of female and male vegetarian and vegan endurance runners compared to omnivores-results from the NURMI study (step 2). J. Int. Soc. Sports Nutr. 2018, 15, 33. [Google Scholar] [CrossRef] [Green Version]
- Nutrition and Running High Mileage. Available online: https://www.nurmi-study.com/en (accessed on 11 March 2021).
- World Health Organization (WHO). Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 11 May 2018).
- World Health Organization (WHO). Available online: http://www.who.int/gho/ncd/risk_factors/bmi_text/en/ (accessed on 11 May 2018).
- Elmadfa, I.; Leitzmann, C. Ernährung des Menschen (Human Nutrition), 6th ed.; UTB GmbH: Berlin, Germany, 2019; p. 618. [Google Scholar]
- König, D. Kohlenhydrate im Sport (Carbohydrates in sport). In Lehrbuch der Sporternährung (Textbook of Sports Nutrition), 1st ed.; Lamprecht, M., Ed.; ÖGSE–österreichische Gesellschaft für Sporternährung: Graz, Austria, 2007; p. 448. [Google Scholar]
- Craig, W.J.; Mangels, A.R.; American Dietetic Association. Position of the American Dietetic Association: Vegetarian diets. J. Am. Diet. Assoc. 2009, 109, 1266–1282. [Google Scholar] [CrossRef] [PubMed]
- Melina, V.; Craig, W.; Levin, S. Position of the Academy of Nutrition and Dietetics: Vegetarian diets. J. Acad. Nutr. Diet. 2016, 116, 1970–1980. [Google Scholar] [CrossRef]
- Scheidt-Nave, C.; Kamtsiuris, P.; Gößwald, A.; Hölling, H.; Lange, M.; Busch, M.A.; Dahm, S.; Dölle, R.; Ellert, U.; Fuchs, J.; et al. German health interview and examination survey for adults (DEGS)-design, ob-jectives and implementation of the first data collection wave. BMC Public Health 2012, 12, 730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haftenberger, M.; Heuer, T.; Heidemann, C.; Kube, F.; Krems, C.; Mensink, G.B. Relative validation of a food frequency questionnaire for national health and nutrition monitoring. Nutr. J. 2010, 9, 36. [Google Scholar] [CrossRef] [Green Version]
- Gahagan, J.; Gray, K.; Whynacht, A. Sex and gender matter in health research: Addressing health inequities in health research reporting. Int. J. Equity Health 2015, 14, 12. [Google Scholar] [CrossRef] [Green Version]
- Knapik, J.J.; Steelman, R.A.; Hoedebecke, S.S.; Austin, K.G.; Farina, E.K.; Lieberman, H.R. Prevalence of dietary supplement use by athletes: Systematic review and meta-analysis. Sports Med. 2016, 46, 103–123. [Google Scholar] [CrossRef] [Green Version]
- Wiens, K.; Erdman, K.A.; Stadnyk, M.; Parnell, J.A. Dietary supplement usage, motivation, and education in young, Canadian athletes. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, N.; Lee, J.; Jung, S.S.; Kang, S.K.; Yoon, J.D. Dietary supplementation of high-performance Korean and Japanese judoists. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 119–127. [Google Scholar] [CrossRef]
- Barrack, M.T.; Muster, M.; Nguyen, J.; Rafferty, A.; Lisagor, T. An investigation of habitual dietary supplement use among 557 NCAA division I athletes. J. Am. Coll. Nutr. 2020, 39, 619–627. [Google Scholar] [CrossRef]
- Aguilar-Navarro, M.; Baltazar-Martins, G.; Brito de Souza, D.; Muñoz-Guerra, J.; del Mar Plata, M.; del Coso, J. Gender differences in prevalence and patterns of dietary supplement use in elite athletes. Res. Q. Exerc. Sports 2020, 18, 1–10. [Google Scholar] [CrossRef]
- Sundgot-Borgen, J.; Berglund, B.; Torstveit, M.K. Nutritional supplements in Norwegian elite athletes--impact of international ranking and advisors. Scand. J. Med. Sci. Sports 2003, 13, 138–144. [Google Scholar] [CrossRef]
- Karimian, J.; Esfahani, P.S. Supplement consumption in body builder athletes. J. Res. Med. Sci. 2011, 16, 1347–1353. [Google Scholar]
- Baranauskas, M.; Jablonskienė, V.; Abaravičius, J.A.; Stukas, R. Actual nutrition and dietary supplementation in Lithuanian elite athletes. Medicina 2020, 56, 247. [Google Scholar] [CrossRef] [PubMed]
- Lun, V.; Erdman, K.A.; Fung, T.S.; Reimer, R.A. Dietary supplementation practices in Canadian high-performance athletes. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 31–37. [Google Scholar] [CrossRef]
- Froiland, K.; Koszewski, W.; Hingst, J.; Kopecky, L. Nutritional supplement use among college athletes and their sources of information. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 104–120. [Google Scholar] [CrossRef]
- Wardenaar, F.; Brinkmans, N.; Ceelen, I.; van Rooij, B.; Mensink, M.; Witkamp, R.; de Vries, J. Micronutrient intakes in 553 Dutch elite and sub-elite athletes: Prevalence of low and high intakes in users and non-users of nutritional supplements. Nutrients 2017, 9, 142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heikkinen, A.; Alaranta, A.; Helenius, I.; Vasankari, T. Dietary supplementation habits and perceptions of supplement use among elite Finnish athletes. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 271–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maughan, R.J.; Depiesse, F.; Geyer, H.; International Association of Athletics Federations. The use of dietary supplements by athletes. J. Sports Sci. 2007, 25, S103–S113. [Google Scholar] [CrossRef] [PubMed]
- Tabata, S.H.; Yamasawa, F.; Torii, S.; Manabe, T.; Kamada, H.; Namba, A.; Kato, J.; Kaneko, H.; Tahara, K.; Tsukahara, Y.; et al. Use of nutritional supplements by elite Japanese track and field athletes. J. Int. Soc. Sports Nutr. 2020, 17, 38. [Google Scholar] [CrossRef]
- Martínez-Sanz, J.M.; Fernández Nuñez, A.; Sospedra, I.; Martínez-Rodríguez, A.; Domínguez, R.; González-Jurado, J.A.; Sánchez-Oliver, A.J. Nutrition-related adverse outcomes in endurance sports competitions: A review of incidence and practical recommendations. Int. J. Environ. Res. Public Health. 2020, 17, 4082. [Google Scholar] [CrossRef] [PubMed]
- Devrim-Lanpir, A.; Hill, L.; Knechtle, B. Efficacy of popular diets applied by endurance athletes on sports performance: Beneficial or detrimental? A narrative review. Nutrients 2021, 13, 491. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.B. Nutrition behaviors, perceptions, and beliefs of recent marathon finishers. Phys. Sportsmed. 2016, 44, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Ruby, M.B. Vegetarianism. A blossoming field of study. Appetite 2012, 58, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Rogerson, D. Vegan diets: Practical advice for athletes and exercisers. J. Int. Soc. Sports Nutr. 2017, 14, 36. [Google Scholar] [CrossRef] [Green Version]
- Nebl, J.; Schuchardt, J.P.; Ströhle, A.; Wasserfurth, P.; Haufe, S.; Eigendorf, J.; Tegtbur, U.; Hahn, A. Micronutrient Status of recreational runners with vegetarian or non-vegetarian dietary patterns. Nutrients 2019, 11, 1146. [Google Scholar] [CrossRef] [Green Version]
- Schüpbach, R.; Wegmüller, R.; Berguerand, C.; Bui, M.; Herter-Aeberli, I. Micronutrient status and intake in omnivores, vegetarians and vegans in Switzerland. Eur. J. Nutr. 2017, 56, 283–293. [Google Scholar] [CrossRef]
- McDougall, C.; McDougall, J. Plant-based diets are not nutritionally deficient. Perm. J. 2013, 17, 93. [Google Scholar] [CrossRef] [Green Version]
- Petróczi, A.; Naughton, D.P.; Pearce, G.; Bailey, R.; Bloodworth, A.; McNamee, M. Nutritional supplement use by elite young UK athletes: Fallacies of advice regarding efficacy. J. Int. Soc. Sports Nutr. 2008, 5, 22. [Google Scholar] [CrossRef] [Green Version]
- Pedrinelli, A.; Ejnisman, L.; Fagotti, L.; Dvorak, J.; Tscholl, P.M. Medications and nutritional supplements in athletes during the 2000, 2004, 2008, and 2012 FIFA Futsal World Cups. BioMed Res. Int. 2015, 2015, 870308. [Google Scholar] [CrossRef] [Green Version]
- Baltazar-Martins, G.; Brito de Souza, D.; Aguilar-Navarro, M.; Muñoz-Guerra, J.; del Mar Plata, M.; del Coso, J. Prevalence and patterns of dietary supplement use in elite Spanish athletes. J. Int. Soc. Sports Nutr. 2019, 16, 30. [Google Scholar] [CrossRef] [Green Version]
- Garthe, I.; Maughan, R.J. Athletes and supplements: Prevalence and perspectives. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 126–138. [Google Scholar] [CrossRef] [Green Version]
- Sobal, J.; Marquart, L.F. Vitamin/mineral supplement use among athletes: A review of the literature. Int. J. Sport Nutr. 1994, 4, 320–334. [Google Scholar] [CrossRef]
- Wilson, P.B.; Rhodes, G.S.; Ingraham, S.J. Self-report versus direct measurement for assessment of fluid intake during a 70.3-mile triathlon. Int. J. Sports Physiol. Perform. 2015, 10, 600–604. [Google Scholar] [CrossRef]
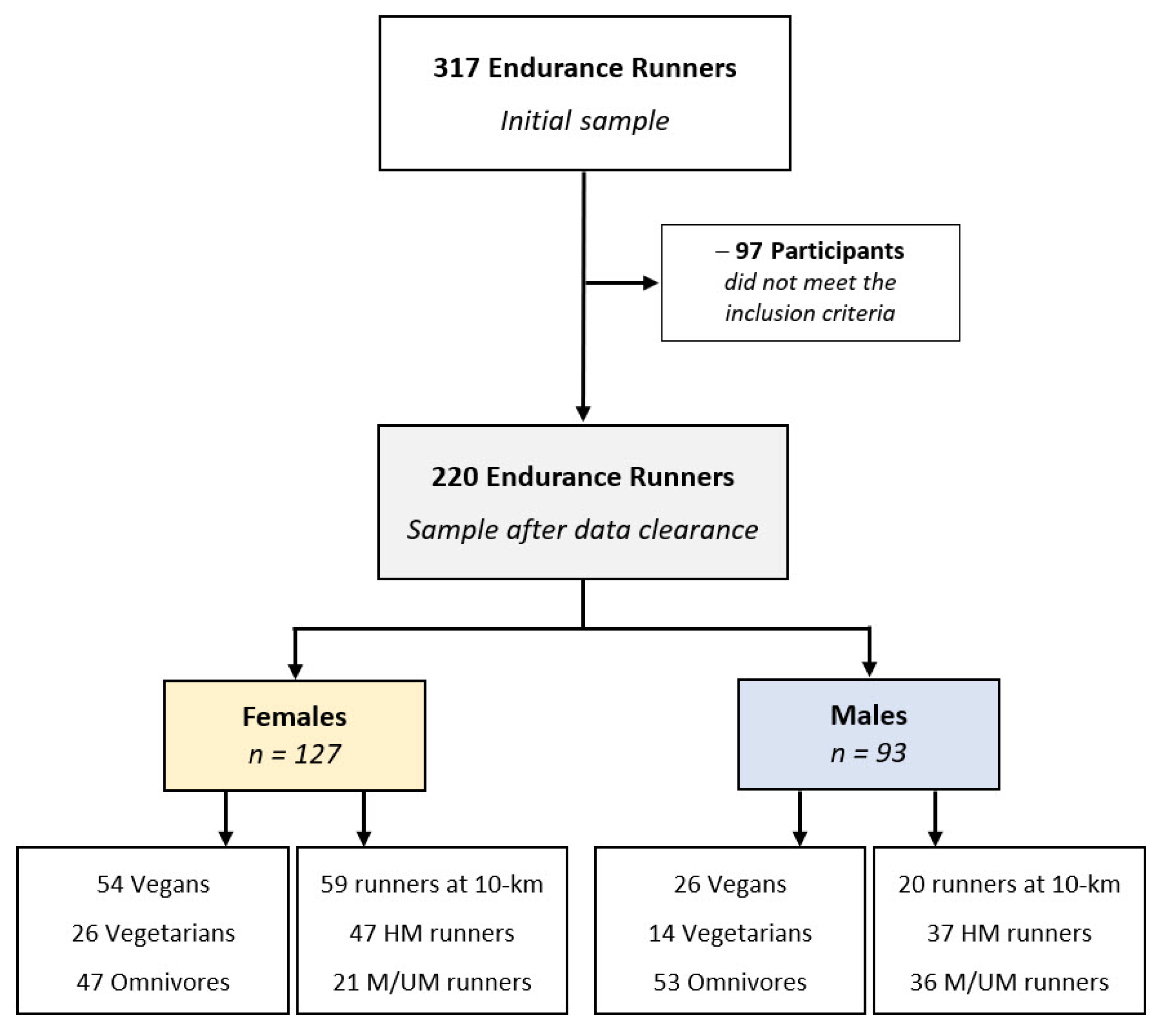
| Total | Females | Males | Statistics | ||
|---|---|---|---|---|---|
| n = 220 | n = 127 | n = 93 | |||
| Age (years) | 38.5 (30, 48) | 37.0 (30, 46) | 42.0 (33, 50) | F(1, 218) = 4.57; p = 0.034 | |
| Body Weight (kg) | 65.0 (59, 73) | 59.8 (54, 65) | 73.5 (68, 80) | F(1, 218) = 180.39; p < 0.001 | |
| Height (m) | 1.70 (1.70, 1.80) | 1.70 (1.60, 1.70) | 1.80 (1.80, 1.80) | F(1, 218) = 201.03; p < 0.001 | |
| BMI (kg/m2) | <18.5 | 13 (6%) | 11 (9%) | 2 (2%) | χ2(2) = 7.86; p = 0.020 |
| 18.5–25 | 181 (82%) | 106 (83%) | 75 (81%) | ||
| >25 | 26 (12%) | 10 (8%) | 16 (17%) | ||
| Academic Qualification | No Qualification | 1 (<1%) | 1 (<1%) | 0 (0%) | χ2(4) = 1.96; p = 0.742 |
| Upper Secondary | 72 (33%) | 38 (30%) | 34 (37%) | ||
| Education/Technical | |||||
| A Levels or Equivalent | 52 (24%) | 30 (24%) | 22 (24%) | ||
| University/Higher Degree | 73 (33%) | 44 (35%) | 29 (31%) | ||
| No Answer | 22 (10%) | 14 (11%) | 8 (9%) | ||
| Marital Status | Divorced/Separated | 11 (5%) | 8 (6%) | 3 (3%) | χ2(2) = 5.55; p = 0.062 |
| Married/Living with Partner | 149 (68%) | 78 (61%) | 71 (76%) | ||
| Single | 60 (27%) | 41 (32%) | 19 (20%) | ||
| Country of Residence | Austria | 39 (18%) | 14 (11%) | 25 (27%) | χ2(3) = 10.41; p = 0.015 |
| Germany | 161 (73%) | 100 (79%) | 61 (66%) | ||
| Switzerland | 11 (5%) | 6 (5%) | 5 (5%) | ||
| Other Countries | 9 (4%) | 7 (6%) | 2 (2%) | ||
| Race Distance | 10-km | 79 (36%) | 59 (46%) | 20 (22%) | χ2(2) = 19.60; p < 0.001 |
| HM | 84 (38%) | 47 (37%) | 37 (40%) | ||
| M/UM | 57 (26%) | 21 (17%) | 36 (39%) | ||
| Diet Type | Omnivorous | 100 (45%) | 47 (37%) | 53 (57%) | χ2(2) = 8.71; p = 0.013 |
| Vegetarian | 40 (18%) | 26 (20%) | 14 (15%) | ||
| Vegan | 80 (36%) | 54 (43%) | 26 (28%) | ||
| Supplement Intake | CHO/Protein Intake | Mineral Intake | Vitamin Intake | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | Females | Males | Females | Males | Females | Males | Females | Males | |
| Total participants | 113 (51%) | 69 (54%) | 44 (47%) | 24 (19%) | 19 (20%) | 46 (36%) | 26 (28%) | 58 (46%) | 35 (38%) |
| (♀ = 127; ♂ = 93) | |||||||||
| Dietary subgroups | |||||||||
| Omnivorous | 41 (41%) | 20 (43%) | 21 (40%) | 9 (19%) | 11 (21%) | 17 (36%) | 19 (36%) | 14 (30%) | 17 (32%) |
| (♀ = 47; ♂ = 53) | |||||||||
| Vegetarian | 14 (35%) | 9 (35%) | 5 (36%) | 4 (15%) | 3 (21%) | 7 (27%) | 2 (14%) | 8 (31%) | 2 (14%) |
| (♀ = 26; ♂ = 14) | |||||||||
| Vegan | 58 (72%) | 40 (74%) | 18 (69%) | 11 (20%) | 5 (19%) | 22 (41%) | 5 (19%) | 36 (67%) | 16 (62%) |
| (♀ = 54; ♂ = 26) | |||||||||
| Distance subgroups | |||||||||
| 10-km | 42 (53%) | 34 (58%) | 8 (40%) | 12 (20%) | 3 (15%) | 24 (41%) | 5 (25%) | 32 (54%) | 6 (30%) |
| (♀ = 59; ♂ = 20) | |||||||||
| HM | 41 (49%) | 23 (49%) | 18 (49%) | 6 (13%) | 9 (24%) | 13 (28%) | 10 (27%) | 18 (38%) | 14 (38%) |
| (♀ = 47; ♂ = 37) | |||||||||
| M/UM | 30 (53%) | 12 (57%) | 18 (50%) | 6 (29%) | 7 (19%) | 9 (43%) | 11 (31%) | 8 (38%) | 15 (42%) |
| (♀ = 21; ♂ = 36) | |||||||||
| BMI subgroups | |||||||||
| <18.5 | 8 (62%) | 7 (64%) | 1 (50%) | 3 (27%) | 1 (50%) | 3 (27%) | 1 (50%) | 7 (64%) | 1 (50%) |
| (♀ = 11; ♂ = 2) | |||||||||
| 18.5–25 | 97 (54%) | 58 (55%) | 39 (52%) | 20 (19%) | 14 (19%) | 41 (39%) | 23 (31%) | 46 (43%) | 33 (44%) |
| (♀ = 106; ♂ = 75) | |||||||||
| >25 | 8 (31%) | 4 (40%) | 4 (25%) | 1 (10%) | 4 (25%) | 2 (20%) | 2 (12%) | 5 (50%) | 1 (6%) |
| (♀ = 10; ♂ = 16) | |||||||||
| Daily Intake | 5–6 Days per Week | 3–4 Days per Week | 1–2 Days per Week | <1 Days per Week | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Females | Males | Females | Males | Females | Males | Females | Males | Females | Males | |
| Total participants | 41 (59%) | 25 (57%) | 4 (6%) | 6 (14%) | 9 (13%) | 4 (9%) | 10 (14%) | 7 (16%) | 5 (7%) | 2 (5%) |
| (♀ = 69; ♂ = 44) | ||||||||||
| Dietary subgroups | ||||||||||
| Omnivorous. | 14 (70%) | 11 (52%) | - | 3 (14%) | 2 (10%) | 2 (10%) | 2 (10%) | 5 (24%) | 2 (10%) | - |
| (♀ = 20; ♂ = 21) | ||||||||||
| Vegetarian | 4 (44%) | 1 (20%) | 1 (11%) | 1 (20%) | 2 (22%) | 1 (20%) | 1 (11%) | 1 (20%) | 1 (11%) | 1 (20%) |
| (♀ = 9; ♂ = 5) | ||||||||||
| Vegan | 23 (57%) | 13 (72%) | 3 (8%) | 2 (11%) | 5 (12%) | 1 (6%) | 7 (18%) | 1 (6%) | 2 (5%) | 1 (6%) |
| (♀ = 40; ♂ = 18) | ||||||||||
| Distance subgroups | ||||||||||
| 10-km | 23 (68%) | 4 (50%) | 2 (6%) | - | 4 (12%) | 2 (25%) | 2 (6%) | 1 (12%) | 3 (9%) | 1 (12%) |
| (♀ = 34; ♂ = 8) | ||||||||||
| HM | 10 (43%) | 12 (67%) | 1 (4%) | 1 (6%) | 3 (13%) | 1 (6%) | 7 (30%) | 3 (17%) | 2 (9%) | 1 (6%) |
| (♀ = 23; ♂ = 18) | ||||||||||
| M/UM | 8 (67%) | 9 (50%) | 1 (8%) | 5 (28%) | 2 (17%) | 1 (6%) | 1 (8%) | 3 (17%) | - | - |
| (♀ = 12; ♂ = 18) | ||||||||||
| BMI subgroups | ||||||||||
| <18.5 | 5 (71%) | 1 (100%) | - | - | - | - | 2 (29%) | - | - | - |
| (♀ = 7; ♂ = 1) | ||||||||||
| 18.5–25 | 32 (55%) | 22 (56%) | 4 (7%) | 6 (15%) | 9 (16%) | 4 (10%) | 8 (14%) | 5 (13%) | 5 (9%) | 2 (5%) |
| (♀ = 58; ♂ = 39) | ||||||||||
| >25 | 4 (100%) | 2 (50%) | - | - | - | - | - | 2 (50%) | - | - |
| (♀ = 4; ♂ = 4) | ||||||||||
| Males | Females | ||||||
|---|---|---|---|---|---|---|---|
| CHO/Protein | Minerals | Vitamins | CHO/Protein | Minerals | Vitamins | ||
| 10-km | Omnivorous | 0.30 [0.15–0.52] | 0.43 [0.25–0.64] | 0.35 [0.18–0.56] | 0.17 [0.04–0.48] | 0.25 [0.08–0.55] | 0.17 [0.04–0.48] |
| Vegetarian | 0.11 [0.02–0.50] | 0.11 [0.02–0.50] | 0.33 [0.11–0.67] | 0.00 [0.00–1.00] | 0.33 [0.04–0.85] | 0.33 [0.04–0.85] | |
| Vegan | 0.15 [0.06–0.33] | 0.48 [0.30–0.66] | 0.78 [0.59–0.90] | 0.20 [0.03–0.69] | 0.20 [0.03–0.69] | 0.60 [0.20–0.90] | |
| Total | 0.20 [0.12–0.32] | 0.41 [0.29–0.54] | 0.54 [0.42–0.66] | 0.15 [0.05–0.38] | 0.25 [0.11–0.48] | 0.30 [0.14–0.53] | |
| HM | Omnivorous | 0.00 [0.00–1.00] | 0.21 [0.07–0.49] | 0.21 [0.07–0.49] | 0.35 [0.18–0.56] | 0.35 [0.18–0.56] | 0.39 [0.22–0.60] |
| Vegetarian | 0.14 [0.04–0.43] | 0.29 [0.11–0.56] | 0.29 [0.11–0.56] | 0.00 [0.00–1.00] | 0.00 [0.00–1.00] | 0.00 [0.00–1.00] | |
| Vegan | 0.21 [0.08–0.45] | 0.32 [0.15–0.55] | 0.58 [0.36–0.77] | 0.11 [0.02–0.50] | 0.22 [0.06–0.58] | 0.56 [0.25–0.82] | |
| Total | 0.13 [0.06–0.26] | 0.28 [0.17–0.42] | 0.38 [0.26–0.53] | 0.24 [0.13–0.41] | 0.27 [0.15–0.43] | 0.38 [0.24–0.54] | |
| M/UM | Omnivorous | 0.20 [0.05–0.54] | 0.40 [0.16–0.70] | 0.30 [0.10–0.62] | 0.06 [0.01–0.31] | 0.44 [0.24–0.67] | 0.33 [0.16–0.57] |
| Vegetarian | 0.33 [0.04–0.85] | 0.67 [0.15–0.96] | 0.33 [0.04–0.85] | 0.50 [0.17–0.83] | 0.17 [0.02–0.63] | 0.17 [0.02–0.63] | |
| Vegan | 0.38 [0.13–0.72] | 0.38 [0.31–0.72] | 0.50 [0.20–0.80] | 0.25 [0.08–0.55] | 0.17 [0.04–0.48] | 0.67 [0.38–0.87] | |
| Total | 0.29 [0.13–0.51] | 0.43 [0.24–0.64] | 0.38 [0.20–0.60] | 0.19 [0.10–0.36] | 0.31 [0.18–0.47] | 0.42 [0.27–0.58] | |
| Total | Omnivorous | 0.19 [0.10–0.33] | 0.36 [0.24–0.51] | 0.30 [0.19–0.44] | 0.21 [0.12–0.34] | 0.36 [0.24–0.49] | 0.32 [0.21–0.46] |
| Vegetarian | 0.15 [0.06–0.35] | 0.27 [0.13–0.47] | 0.31 [0.16–0.51] | 0.21 [0.07–0.49] | 0.14 [0.04–0.43] | 0.14 [0.04–0.43] | |
| Vegan | 0.20 [0.12–0.33] | 0.41 [0.29–0.54] | 0.67 [0.53–0.78] | 0.19 [0.08–0.39] | 0.19 [0.08–0.39] | 0.62 [0.42–0.78] | |
| Total | 0.19 [0.13–0.27] | 0.36 [0.28–0.45] | 0.46 [0.37–0.54] | 0.20 [0.13–0.30] | 0.28 [0.20–0.38] | 0.38 [0.28–0.48] | |
| CHO/Protein | Minerals | Vitamins | |||||||
|---|---|---|---|---|---|---|---|---|---|
| χ² | Df | p | χ² | Df | p | χ² | Df | p | |
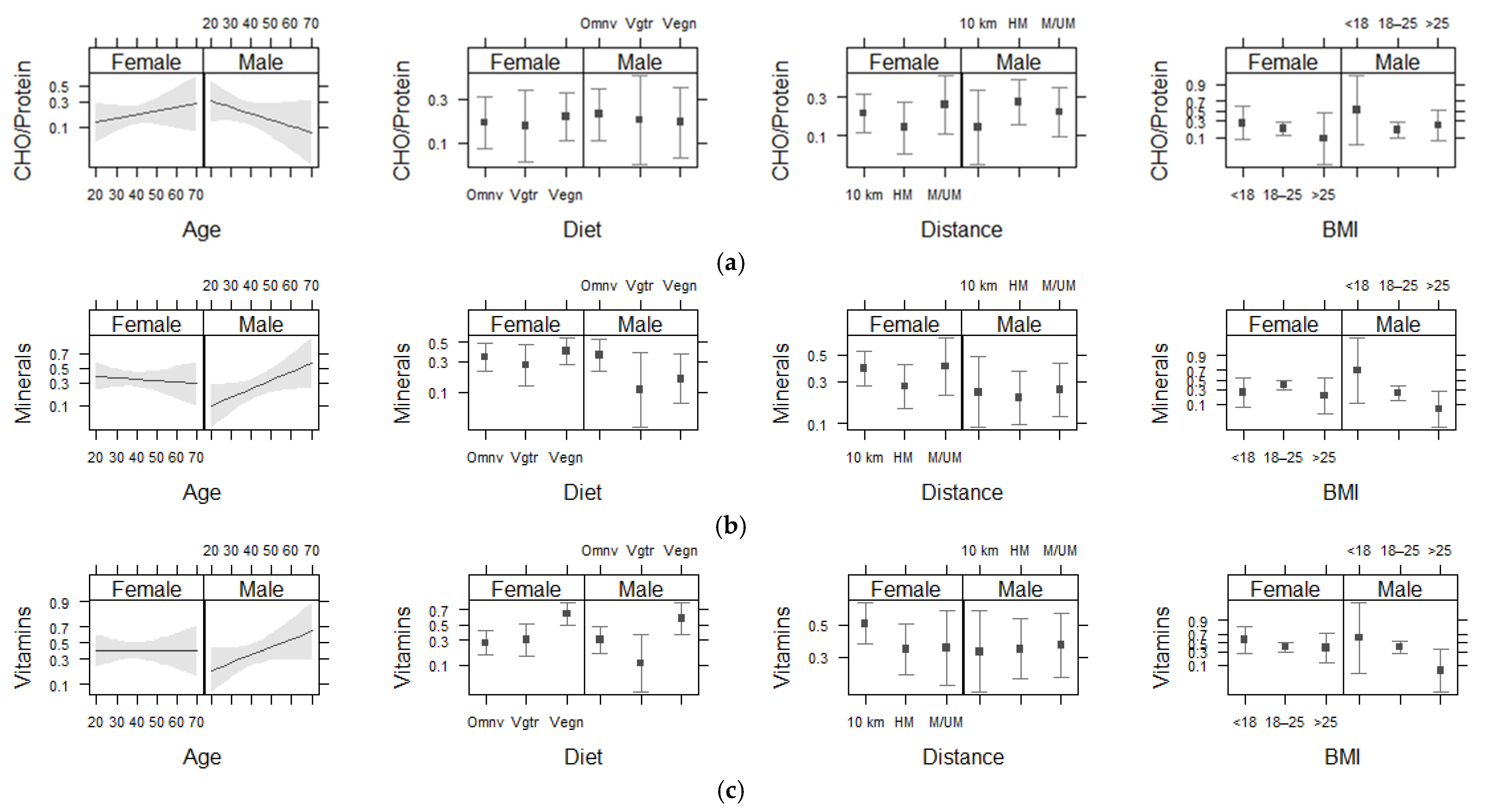
| Sex | 0.02 | 1 | 0.875 | 1.92 | 1 | 0.166 | 0.14 | 1 | 0.713 |
| Age | 0.00 | 1 | 0.997 | 1.48 | 1 | 0.224 | 1.37 | 1 | 0.242 |
| BMI | 1.13 | 2 | 0.567 | 5.28 | 2 | 0.071 | 5.11 | 2 | 0.078 |
| Diet | 0.04 | 2 | 0.979 | 2.50 | 2 | 0.287 | 25.33 | 2 | > 0.001 † |
| Distance | 0.46 | 2 | 0.794 | 1.84 | 2 | 0.398 | 1.03 | 2 | 0.598 |
| Sex * Age | 2.86 | 1 | 0.091 | 3.16 | 1 | 0.076 | 2.08 | 1 | 0.149 |
| Sex * BMI | 1.26 | 2 | 0.533 | 2.58 | 2 | 0.275 | 3.80 | 2 | 0.149 |
| Sex * Diet | 0.96 | 2 | 0.619 | 2.68 | 2 | 0.262 | 1.28 | 2 | 0.526 |
| Sex * Distance | 2.64 | 2 | 0.268 | 0.82 | 2 | 0.664 | 1.70 | 2 | 0.426 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wirnitzer, K.; Motevalli, M.; Tanous, D.R.; Gregori, M.; Wirnitzer, G.; Leitzmann, C.; Rosemann, T.; Knechtle, B. Sex Differences in Supplement Intake in Recreational Endurance Runners—Results from the NURMI Study (Step 2). Nutrients 2021, 13, 2776. https://doi.org/10.3390/nu13082776
Wirnitzer K, Motevalli M, Tanous DR, Gregori M, Wirnitzer G, Leitzmann C, Rosemann T, Knechtle B. Sex Differences in Supplement Intake in Recreational Endurance Runners—Results from the NURMI Study (Step 2). Nutrients. 2021; 13(8):2776. https://doi.org/10.3390/nu13082776
Chicago/Turabian StyleWirnitzer, Katharina, Mohamad Motevalli, Derrick R. Tanous, Martina Gregori, Gerold Wirnitzer, Claus Leitzmann, Thomas Rosemann, and Beat Knechtle. 2021. "Sex Differences in Supplement Intake in Recreational Endurance Runners—Results from the NURMI Study (Step 2)" Nutrients 13, no. 8: 2776. https://doi.org/10.3390/nu13082776
APA StyleWirnitzer, K., Motevalli, M., Tanous, D. R., Gregori, M., Wirnitzer, G., Leitzmann, C., Rosemann, T., & Knechtle, B. (2021). Sex Differences in Supplement Intake in Recreational Endurance Runners—Results from the NURMI Study (Step 2). Nutrients, 13(8), 2776. https://doi.org/10.3390/nu13082776









