Differences in the Concentration of Vitamin D Metabolites in Plasma Due to the Low-Carbohydrate-High-Fat Diet and the Eastern European Diet—A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
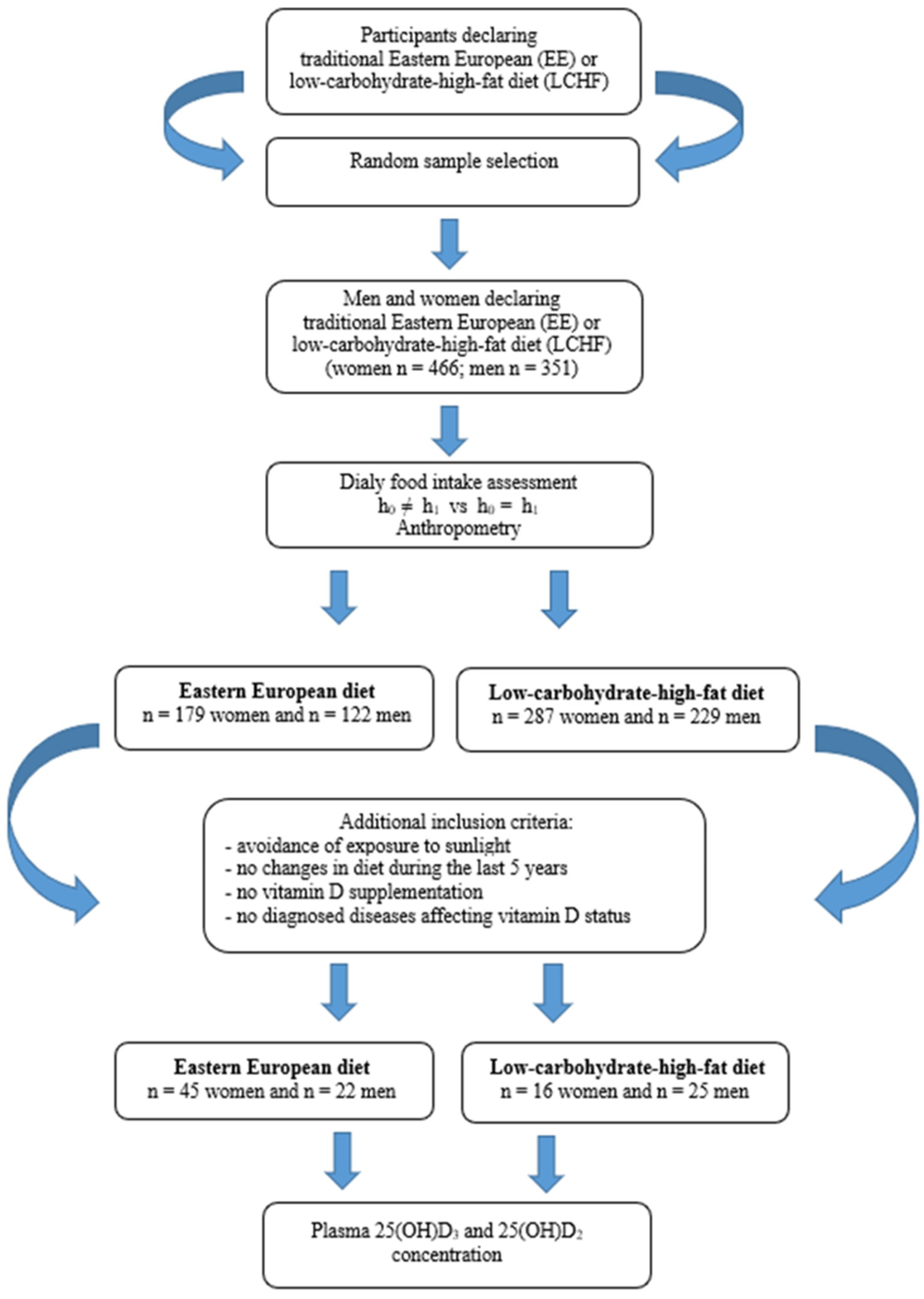
2.1. Study Design and Data Collection
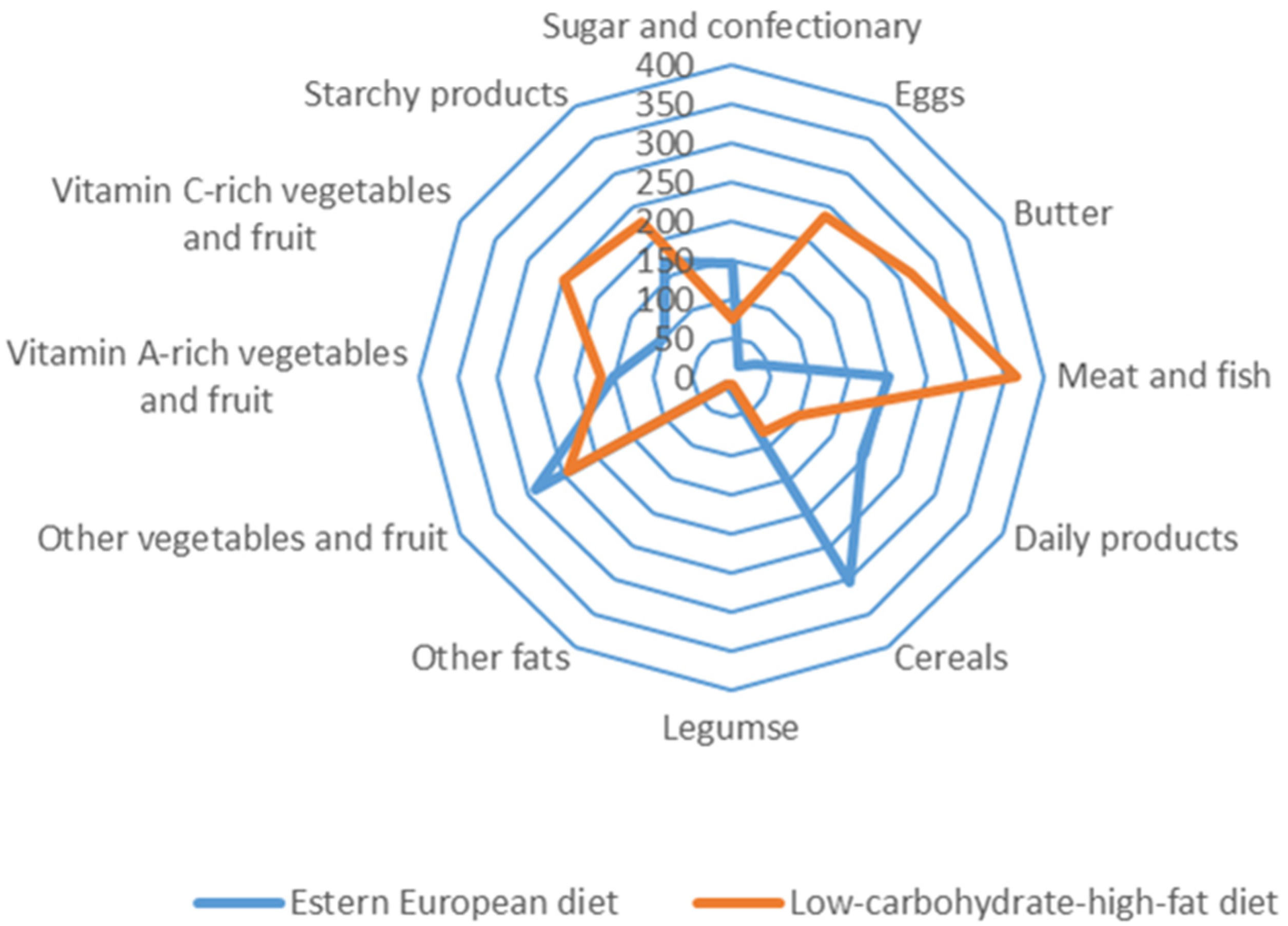
2.2. Assessment of Nutrition and Anthropometric Measures
2.3. Determination of Vitamin D Metabolites
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lamberg-Allardt, C. Vitamin D in Foods and as Supplements. Prog. Biophys. Mol. Biol. 2006, 92, 33–38. [Google Scholar] [CrossRef]
- Cardwell, G.; Bornman, J.F.; James, A.P.; Black, L.J. A Review of Mushrooms as a Potential Source of Dietary Vitamin D. Nutrients 2018, 10, 1498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalajian, T.A.; Aldoukhi, A.; Veronikis, A.J.; Persons, K.; Holick, M.F. Ultraviolet B Light Emitting Diodes (LEDs) Are More Efficient and Effective in Producing Vitamin D 3 in Human Skin Compared to Natural Sunlight. Sci. Rep. 2017, 7, 11489. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.; Christakos, S. New aspects of vitamin D metabolism and action—Addressing the skin as source and target. Nat. Rev. Endocrinol. 2020, 16, 234. [Google Scholar] [CrossRef] [PubMed]
- Saponaro, F.; Saba, A.; Zucchi, R. An Update on Vitamin D Metabolism. Int. J. Mol. Sci. 2020, 21, 6573. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Vitamin D Metabolism, Mechanism of Action, and Clinical Applications. Chem. Biol. 2014, 21, 319–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zmijewski, M. Vitamin D and Human Health. Int. J. Mol. Sci. 2019, 20, 145. [Google Scholar] [CrossRef] [Green Version]
- Spiro, A.; Buttriss, J.L. Vitamin D: An Overview of Vitamin D Status and Intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef] [Green Version]
- Touvier, M.; Deschasaux, M.; Montourcy, M.; Sutton, A.; Charnaux, N.; Kesse-Guyot, E.; Assmann, K.E.; Fezeu, L.; Latino-Martel, P.; Druesne-Pecollo, N.; et al. Determinants of Vitamin D Status in Caucasian Adults: Influence of Sun Exposure, Dietary Intake, Sociodemographic, Lifestyle, Anthropometric, and Genetic Factors. J. Investig. Dermatol. 2015, 135, 378–388. [Google Scholar] [CrossRef] [Green Version]
- Meoli, M.; Muggli, F.; Lava, S.A.G.; Bianchetti, M.G.; Agostoni, C.; Kocher, C.; Bührer, T.W.; Ciliberti, L.; Simonetti, G.D.; Milani, G.P. Vitamin D Status in Adolescents during COVID-19 Pandemic: A Cross-Sectional Comparative Study. Nutrients 2021, 13, 1467. [Google Scholar] [CrossRef]
- Milani, G.P.; Simonetti, G.D.; Edefonti, V.; Lava, S.A.G.; Agostoni, C.; Curti, M.; Stettbacher, A.; Bianchetti, M.G.; Muggli, F. Seasonal Variability of the Vitamin D Effect on Physical Fitness in Adolescents. Sci. Rep. 2021, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Dymarska, E.; Grochowalska, A.; Krauss, H. The influence of nutrition on immune system. Immunomodulation by fatty acids, vitamins, minerals and antioxidants. J. Med. Sci. 2013, 82, 222–231. [Google Scholar]
- Seamans, K.M.; Cashman, K.D. Existing and Potentially Novel Functional Markers of Vitamin D Status: A Systematic Review. Am. J. Clin. Nutr. 2009, 89, 1997S–2008S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, K.S.; Assar, S.; Harnpanich, D.; Bouillon, R.; Lambrechts, D.; Prentice, A.; Schoenmakers, I. 25(OH)D2 Half-Life Is Shorter Than 25(OH)D3 Half-Life and Is Influenced by DBP Concentration and Genotype. J. Clin. Endocrinol. Metab. 2014, 99, 3373–3381. [Google Scholar] [CrossRef] [Green Version]
- Bartoszewicz, Z.; Kondracka, A.; Jaźwiec, R.; Popow, M.; Dadlez, M.; Bednarczuk, T. Can We Accurately Measure the Concentration of Clinically Relevant Vitamin D Metabolites in the Circulation? The Problems and Their Consequences. Endokrynol. Pol. 2013, 64, 238–245. [Google Scholar]
- Płudowski, P.; Ducki, C.; Konstantynowicz, J.; Jaworski, M. Vitamin D Status in Poland. Pol. Arch. Med. Wewn. 2016, 126, 530–539. [Google Scholar] [CrossRef] [Green Version]
- Karczmarewicz, E.; Czekuć-Kryśkiewicz, E.; Płudowski, P. Effect of Vitamin D Status on Pharmacological Treatment Efficiency. Dermato-Endocrinology 2013, 5, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Yoo, K.; Cho, J.; Ly, S. Vitamin D Intake and Serum 25-Hydroxyvitamin D Levels in Korean Adults: Analysis of the 2009 Korea National Health and Nutrition Examination Survey (KNHANES IV-3) Using a Newly Established Vitamin D Database. Nutrients 2016, 8, 610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, C.L.; Patterson, K.Y.; Roseland, J.M.; Wise, S.A.; Merkel, J.M.; Pehrsson, P.R.; Yetley, E.A. Including Food 25-Hydroxyvitamin D in Intake Estimates May Reduce the Discrepancy between Dietary and Serum Measures of Vitamin D Status. J. Nutr. 2014, 144, 654–659. [Google Scholar] [CrossRef] [Green Version]
- Andersen, S.; Laurberg, P.; Hvingel, B.; Kleinschmidt, K.; Heickendorff, L.; Mosekilde, L. Vitamin D Status in Greenland Is Influenced by Diet and Ethnicity: A Population-Based Survey in an Arctic Society in Transition. Br. J. Nutr. 2013, 109, 928–935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, K.; Nashimoto, M.; Hori, Y.; Yamamoto, M. Serum 25-Hydroxyvitamin D Concentrations and Related Dietary Factors in Peri- and Postmenopausal Japanese Women. Am. J. Clin. Nutr. 2000, 71, 1161–1165. [Google Scholar] [CrossRef] [PubMed]
- Leiu, K.H.; Chin, Y.S.; Mohd Shariff, Z.; Arumugam, M.; Chan, Y.M. High Body Fat Percentage and Low Consumption of Dairy Products Were Associated with Vitamin D Inadequacy among Older Women in Malaysia. PLoS ONE 2020, 15, e0228803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alathari, B.E.; Aji, A.S.; Ariyasra, U.; Sari, S.R.; Tasrif, N.; Yani, F.F.; Sudji, I.R.; Lovegrove, J.A.; Lipoeto, N.I.; Vimaleswaran, K.S. Interaction between Vitamin D-Related Genetic Risk Score and Carbohydrate Intake on Body Fat Composition: A Study in Southeast Asian Minangkabau Women. Nutrients 2021, 13, 326. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Harris, S.S.; Lichtenstein, A.H.; Dolnikowski, G.; Palermo, N.J.; Rasmussen, H. Dietary Fat Increases Vitamin D-3 Absorption. J. Acad. Nutr. Diet 2015, 115, 225–230. [Google Scholar] [CrossRef]
- Gibson, R.S. Principles of Nutritional Assessment; Oxford University Press: New York, NY, USA; Oxford, UK, 2005; ISBN 978-0-19-517169-3. [Google Scholar]
- Obesity: Preventing and Managing the Global Epidemic. Report of a WHO Consultation. World Health Organ. Tech. Rep. Ser. 2000, 894, 1–253. [Google Scholar]
- Abouzid, M.; Karaźniewicz-Łada, M.; Pawlak, K.; Burchardt, P.; Kruszyna, Ł.; Główka, F. Measurement of Plasma 25-Hydroxyvitamin D2, 25-Hydroxyvitamin D3 and 3-Epi-25-Hydroxyvitamin D3 in Population of Patients with Cardiovascular Disease by UPLC-MS/MS Method. J. Chromatogr. B 2020, 1159, 122350. [Google Scholar] [CrossRef]
- Dimakopoulos, I.; Magriplis, E.; Mitsopoulou, A.-V.; Karageorgou, D.; Bakogianni, I.; Micha, R.; Michas, G.; Chourdakis, M.; Chrousos, G.P.; Roma, E.; et al. Intake and Contribution of Food Groups to Vitamin D Intake in a Representative Sample of Adult Greek Population. Nutrition 2020, 72, 110641. [Google Scholar] [CrossRef]
- Schmid, A.; Walther, B. Natural Vitamin D Content in Animal Products1. Adv. Nutr. 2013, 4, 453–462. [Google Scholar] [CrossRef]
- Ovesen, L.; Brot, C.; Jakobsen, J. Food Contents and Biological Activity of 25-Hydroxyvitamin D: A Vitamin D Metabolite to Be Reckoned With? Ann. Nutr. Metab. 2003, 47, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Hoseinzadeh, E.; Taha, P.; Wei, C.; Godini, H.; Ashraf, G.M.; Taghavi, M.; Miri, M. The Impact of Air Pollutants, UV Exposure and Geographic Location on Vitamin D Deficiency. Food Chem. Toxicol. 2018, 113, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Barrea, L.; Savastano, S.; Di Somma, C.; Savanelli, M.C.; Nappi, F.; Albanese, L.; Orio, F.; Colao, A. Low Serum Vitamin D-Status, Air Pollution and Obesity: A Dangerous Liaison. Rev. Endocr. Metab. Disord. 2017, 18, 207–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cashman, K.D.; Kiely, M. Towards Prevention of Vitamin D Deficiency and beyond: Knowledge Gaps and Research Needs in Vitamin D Nutrition and Public Health. Br. J. Nutr. 2011, 106, 1617–1627. [Google Scholar] [CrossRef] [PubMed]
- El Hayek, J.; Egeland, G.; Weiler, H. Older Age and Lower Adiposity Predict Better 25-Hydroxy Vitamin D Concentration in Inuit Adults: International Polar Year Inuit Health Survey, 2007–2008. Arch. Osteoporos. 2011, 6, 167–177. [Google Scholar] [CrossRef]
- Andersen, S.; Jakobsen, A.; Laurberg, P. Vitamin D Status in North Greenland Is Influenced by Diet and Season: Indicators of Dermal 25-Hydroxy Vitamin D Production North of the Arctic Circle. Br. J. Nutr. 2013, 110, 50–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rippin, H.L.; Hutchinson, J.; Greenwood, D.C.; Jewell, J.; Breda, J.J.; Martin, A.; Rippin, D.M.; Schindler, K.; Rust, P.; Fagt, S.; et al. Inequalities in Education and National Income Are Associated with Poorer Diet: Pooled Analysis of Individual Participant Data across 12 European Countries. PLoS ONE 2020, 15, e0232447. [Google Scholar] [CrossRef]
- Brouns, F. Overweight and Diabetes Prevention: Is a Low-Carbohydrate–High-Fat Diet Recommendable? Eur. J. Nutr. 2018, 57, 1301–1312. [Google Scholar] [CrossRef] [Green Version]
- Hall, K.D.; Bemis, T.; Brychta, R.; Chen, K.Y.; Courville, A.; Crayner, E.J.; Goodwin, S.; Guo, J.; Howard, L.; Knuth, N.D.; et al. Calorie for Calorie, Dietary Fat Restriction Results in More Body Fat Loss than Carbohydrate Restriction in People with Obesity. Cell Metab. 2015, 22, 427–436. [Google Scholar] [CrossRef] [Green Version]
- Hu, T.; Mills, K.T.; Yao, L.; Demanelis, K.; Eloustaz, M.; Yancy, W.S.; Kelly, T.N.; He, J.; Bazzano, L.A. Effects of Low-Carbohydrate Diets versus Low-Fat Diets on Metabolic Risk Factors: A Meta-Analysis of Randomized Controlled Clinical Trials. Am. J. Epidemiol. 2012, 176 (Suppl. S7), S44–S54. [Google Scholar] [CrossRef] [Green Version]
- Mansoor, N.; Vinknes, K.J.; Veierød, M.B.; Retterstøl, K. Effects of Low-Carbohydrate Diets v. Low-Fat Diets on Body Weight and Cardiovascular Risk Factors: A Meta-Analysis of Randomised Controlled Trials. Br. J. Nutr. 2016, 115, 466–479. [Google Scholar] [CrossRef]
- Bradley, U.; Spence, M.; Courtney, C.H.; McKinley, M.C.; Ennis, C.N.; McCance, D.R.; McEneny, J.; Bell, P.M.; Young, I.S.; Hunter, S.J. Low-Fat versus Low-Carbohydrate Weight Reduction Diets: Effects on Weight Loss, Insulin Resistance, and Cardiovascular Risk: A Randomized Control Trial. Diabetes 2009, 58, 2741–2748. [Google Scholar] [CrossRef] [Green Version]
- Pludowski, P.; Holick, M.F.; Grant, W.B.; Konstantynowicz, J.; Mascarenhas, M.R.; Haq, A.; Povoroznyuk, V.; Balatska, N.; Barbosa, A.P.; Karonova, T.; et al. Vitamin D Supplementation Guidelines. J. Steroid Biochem. Mol. Biol. 2018, 175, 125–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fock, K.M.; Khoo, J. Diet and Exercise in Management of Obesity and Overweight. J. Gastroenterol. Hepatol. 2013, 28, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Oh, R.; Gilani, B.; Uppaluri, K.R. Low Carbohydrate Diet. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Reyes, T.M. High-Fat Diet Alters the Dopamine and Opioid Systems: Effects across Development. Int. J. Obes. Suppl. 2012, 2, S25–S28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rohr, M.W.; Narasimhulu, C.A.; Rudeski-Rohr, T.A.; Parthasarathy, S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv. Nutr. 2020, 11, 77–91. [Google Scholar] [CrossRef] [Green Version]
- Seid, H.; Rosenbaum, M. Low Carbohydrate and Low-Fat Diets: What We Don’t Know and Why We Should Know It. Nutrients 2019, 11, 2749. [Google Scholar] [CrossRef] [Green Version]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.; Bianchi, M.L.; Stepan, J.; Fuleihan, G.E.-H.; Bouillon, R. Current Vitamin D Status in European and Middle East Countries and Strategies to Prevent Vitamin D Deficiency: A Position Statement of the European Calcified Tissue Society. Eur. J. Endocrinol. 2019, 180, P23–P54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased Bioavailability of Vitamin D in Obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Barrea, L.; Somma, C.D.; Laudisio, D.; Salzano, C.; Pugliese, G.; de Alteriis, G.; Colao, A.; Savastano, S. Sex Differences of Vitamin D Status across BMI Classes: An Observational Prospective Cohort Study. Nutrients 2019, 11, 3034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Women | Men | |||||
|---|---|---|---|---|---|---|
| EE (n = 179) | LCHF (n = 287) | p 2 | EE (n = 122) | LCHF (n = 229) | p 2 | |
| Age, y ς, % | 56.3 ± 4.8 1.25 | 59.7 ± 7.1 1.38 | NS | 57.6 ± 5.0 1.55 | 63.0 ± 9.7 2.01 | NS |
| Body height, cm ς, % | 163 ± 6 0.54 | 163 ± 5 0.33 | NS | 172 ± 6 0.67 | 173 ± 6 0.48 | NS |
| Bodyweight, kg ς, % | 67.2 ± 12.0 2.64 | 67.3 ± 6.6 1.14 | NS | 81.5 ± 13.2 2.90 | 77.2 ± 12.4 2.10 | NS |
| BMI, kg/m2 ς, % | 25.5 ± 4.6 2.63 | 25.3 ± 2.6 1.18 | NS | 27.7 ± 4.4 2.84 | 25.8 ± 3.2 1.61 | p < 0.05 |
| Macronutrient intake | ||||||
| Total energy, kcal/d ς (%) | 1768 ± 581 4.85 | 1688 ± 516 3.57 | NS | 2336 ± 807 6.19 | 2168 ± 724 4.37 | p < 0.05 |
| Protein, g/d ς (%) | 55.8 ± 19.5 5.16 | 53.9 ± 17.1 3.71 | NS | 72.0 ± 33.2 8.26 | 66.9 ± 22.2 4.46 | NS |
| Protein, % kcal ς (%) | 12.9 ± 3.4 3.83 | 13.2 ± 2.8 2.51 | NS | 12.3 ± 3.1 4.52 | 12.8 ± 3.0 3.06 | NS |
| Fat, g/d ς (%) | 64.0 ± 30.1 * 6.94 | 130 ± 44.4 * 4.02 | p < 0.05 | 88.6 ± 42.8 * 8.66 | 170 ± 62.4 * 4.80 | p < 0.05 |
| Fat, % kcal ς (%) | 32.2 ± 8.8 * 4.03 | 68.3 ± 8.5 * 1.45 | p < 0.05 | 33.4 ± 8.6 * 4.62 | 69.6 ± 8.7 * 1.63 | p < 0.05 |
| Carbohydrate, g/d ς (%) | 242 ± 88 * 5.39 | 80.0 ± 34 * 4.95 | p < 0.05 | 310 ± 105 * 6.07 | 97.4 ± 45 * 6.07 | p < 0.05 |
| Carbohydrate, % kcal ς (%) | 55.0 ± 10.0 * 2.68 | 19.6 ± 6.4 * 3.80 | p < 0.05 | 53.1 ± 9.2 * 3.09 | 18.3 ± 6.4 * 4.54 | p < 0.05 |
| Vitamin D intake, µg/d ς (%) | 3.22 ± 7.16 * 19.1 | 8.13 ± 6.52 * 9.37 | p < 0.05 | 5.90 ± 10.9 * 22.1 | 9.91 ± 8.61 * 11.4 | p < 0.05 |
| 25(OH)D3 | 25(OH)D2 | Total (D3 + D2) | ||||
|---|---|---|---|---|---|---|
| EE (n = 67) | LCHF (n = 41) | EE (n = 23) | LCHF (n = 14) | EE (n = 14) | LCHF (n = 14) | |
| Mean | 22.6 | 34.9 | 7.64 | 8.71 | 28.0 | 47.7 |
| SD | 12.1 | 15.9 | 2.42 | 3.51 | 10.7 | 17.7 |
| ς(%) | 7.07 | 6.08 | 4.33 | 5.75 | 5.45 | 5.29 |
| p value | 0.00004 * | 0.27987 | 0.00304 ** | |||
| Women | Men | |||||||
|---|---|---|---|---|---|---|---|---|
| 25(OH)D3 | 25(OH)D2 | 25(OH)D3 | 25(OH)D2 | |||||
| EE (n = 45) | LCHF (n = 16) | EE (n = 18) | LCHF (n = 7) | EE (n = 22) | LCHF (n = 25) | EE (n = 5) | LCHF (n = 7) | |
| Mea | 22.9 | 37.8 | 6.83 | 7.89 | 22.1 | 33.0 | 10.6 | 10.1 |
| SD | 11.9 | 16.2 | 1.79 | 4.33 | 12.8 | 15.9 | 2.24 | 1.78 |
| ς (%) | 6.90 | 6.03 | 3.65 | 8.85 | 7.92 | 6.56 | 3.86 | 2.84 |
| p value | 0.0003 * | 0.7853 | 0.0034 | 0.8709 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bolesławska, I.; Kowalówka, M.; Dobrzyńska, M.; Karaźniewicz-Łada, M.; Przysławski, J. Differences in the Concentration of Vitamin D Metabolites in Plasma Due to the Low-Carbohydrate-High-Fat Diet and the Eastern European Diet—A Pilot Study. Nutrients 2021, 13, 2774. https://doi.org/10.3390/nu13082774
Bolesławska I, Kowalówka M, Dobrzyńska M, Karaźniewicz-Łada M, Przysławski J. Differences in the Concentration of Vitamin D Metabolites in Plasma Due to the Low-Carbohydrate-High-Fat Diet and the Eastern European Diet—A Pilot Study. Nutrients. 2021; 13(8):2774. https://doi.org/10.3390/nu13082774
Chicago/Turabian StyleBolesławska, Izabela, Magdalena Kowalówka, Małgorzata Dobrzyńska, Marta Karaźniewicz-Łada, and Juliusz Przysławski. 2021. "Differences in the Concentration of Vitamin D Metabolites in Plasma Due to the Low-Carbohydrate-High-Fat Diet and the Eastern European Diet—A Pilot Study" Nutrients 13, no. 8: 2774. https://doi.org/10.3390/nu13082774
APA StyleBolesławska, I., Kowalówka, M., Dobrzyńska, M., Karaźniewicz-Łada, M., & Przysławski, J. (2021). Differences in the Concentration of Vitamin D Metabolites in Plasma Due to the Low-Carbohydrate-High-Fat Diet and the Eastern European Diet—A Pilot Study. Nutrients, 13(8), 2774. https://doi.org/10.3390/nu13082774









