Malnutrition in Older Adults—Recent Advances and Remaining Challenges
Abstract
:1. Introduction
2. Impact of Malnutrition in the Old
2.1. Role of Malnutrition in the Geriatric Syndromes Frailty, Fatigue, Sarcopenia
2.2. Prognostic Impact of Malnutrition in the Old on Mortality
3. Malnutrition: Definition and Types and How to Screen for Them
3.1. Macronutrient Deficiencies
3.2. Micronutrient Deficiencies
4. Prevalence of Malnutrition
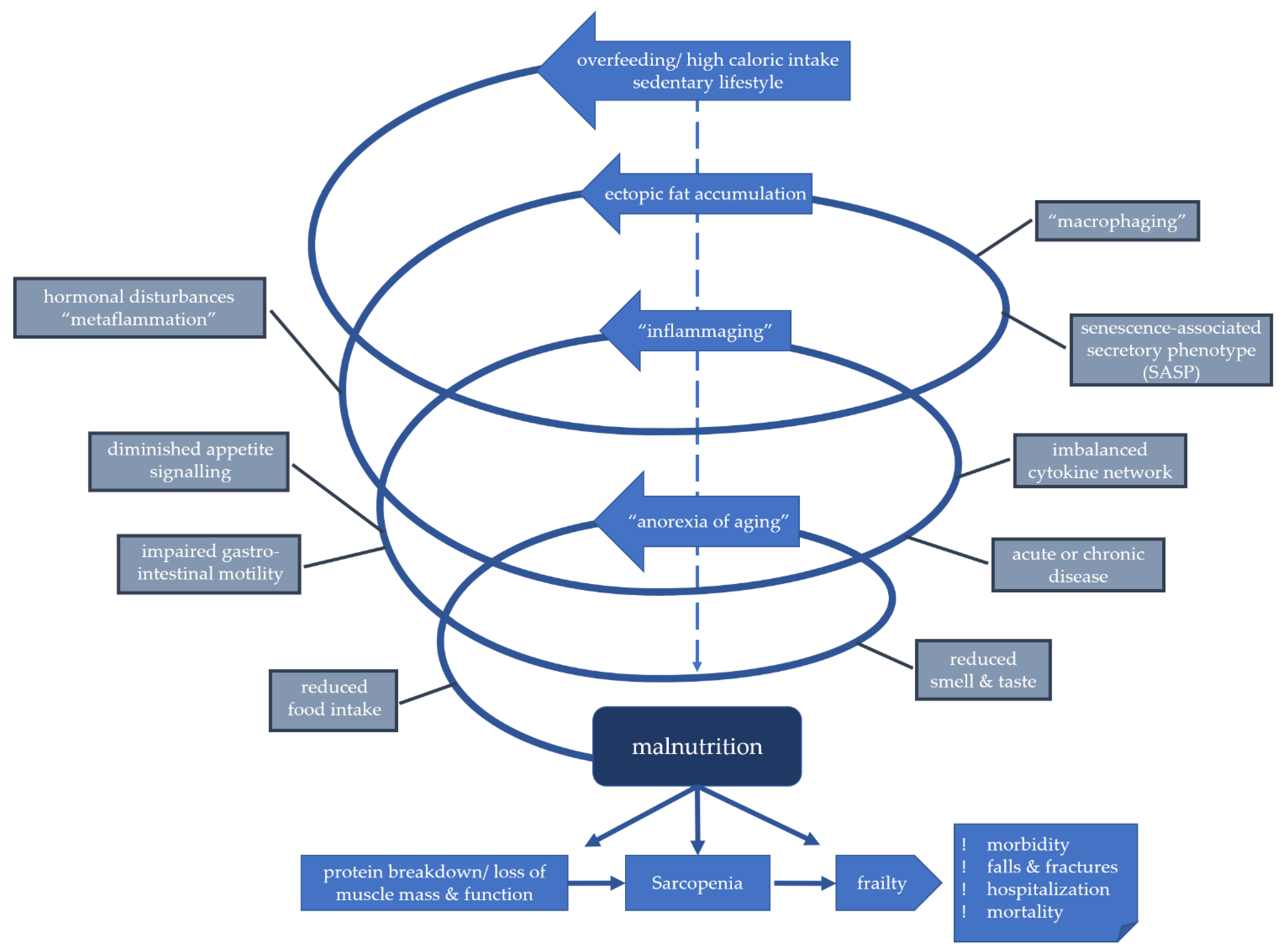
5. Determinants of Malnutrition
5.1. Modifiable Determinants of Malnutrition
5.2. Age-Associated Changes as Risk Factors
5.3. Inflammaging as a Risk Factor for Malnutrition
6. Treatment of Malnutrition
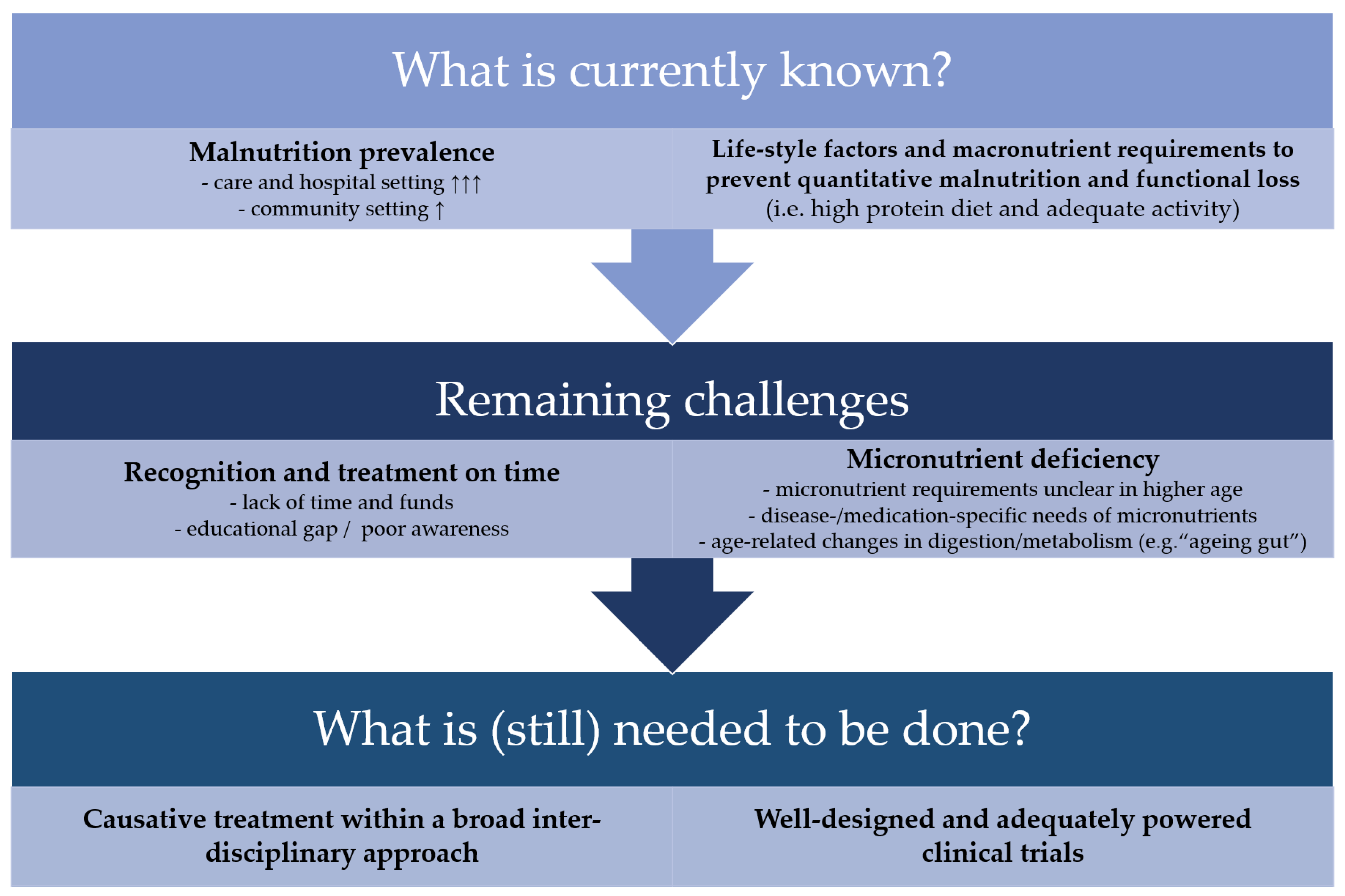
7. Remaining Challenges
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ASPEN | American Society for Parenteral and Enteral Nutrition |
| BMI | body mass index |
| CCK | cholecystokinin |
| CNS | central nervous system |
| COVID-19 | coronavirus disease 2019 |
| DoMAP | Determinants of Malnutrition in Aged Persons |
| DNA | deoxyribonucleic acid |
| ESPEN | European Society of Clinical Nutrition and Metabolism |
| Fe | iron |
| FELANPE | Federación Latinoamericana de Terapia Nutricional, Nutrición Clínica y Metabolismo |
| FGF21 | fibroblast growth factor 21 |
| GLIM | Global Leadership Initiative on Malnutrition |
| HIV/AIDS | human immunodeficiency virus/acquired immunodeficiency syndrome |
| ICU | intensive care unit |
| IL | interleukin |
| MNA | Mini Nutritional Assessment |
| PENSA | The Parenteral and Enteral Nutrition Society of Asia |
| PEM | protein-energy malnutrition |
| PEU | protein-energy undernutrition |
| (PG-) SGA | (Patient-Generated) Subjective Global Assessment |
| PYY | peptide YY |
| SASP | senescence-associated secretory phenotype |
| TNF-α | tumour necrosis factor alpha |
| WHO | World Health Organization |
| Zn | zinc |
References
- Rudnicka, E.; Napierała, P.; Podfigurna, A.; Męczekalski, B.; Smolarczyk, R.; Grymowicz, M. The World Health Organization (WHO) approach to healthy ageing. Maturitas 2020, 139, 6–11. [Google Scholar] [CrossRef] [PubMed]
- United Nations. World Population Prospects 2019: Highlights; Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2019. [Google Scholar]
- Niccoli, T.; Partridge, L. Ageing as a risk factor for disease. Curr. Biol. 2012, 22, R741–R752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkila, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Hickson, M. Malnutrition and ageing. Postgrad. Med. J. 2006, 82, 2–8. [Google Scholar] [CrossRef]
- Norman, K.; Pichard, C.; Lochs, H.; Pirlich, M. Prognostic impact of disease-related malnutrition. Clin. Nutr. 2008, 27, 5–15. [Google Scholar] [CrossRef]
- Cunha, A.I.L.; Veronese, N.; de Melo Borges, S.; Ricci, N.A. Frailty as a predictor of adverse outcomes in hospitalized older adults: A systematic review and meta-analysis. Ageing Res. Rev. 2019, 56, 100960. [Google Scholar] [CrossRef]
- Otten, L.; Stobäus, N.; Franz, K.; Genton, L.; Müller-Werdan, U.; Wirth, R.; Norman, K. Impact of sarcopenia on 1-year mortality in older patients with cancer. Age Ageing 2019, 48, 413–418. [Google Scholar] [CrossRef]
- Bardon, L.A.; Streicher, M.; Corish, C.A.; Clarke, M.; Power, L.C.; Kenny, R.A.; O’Connor, D.M.; Laird, E.; O’Connor, E.M.; Visser, M.; et al. Predictors of Incident Malnutrition in Older Irish Adults from the Irish Longitudinal Study on Ageing Cohort-A MaNuEL study. J. Gerontol. Ser. A 2020, 75, 249–256. [Google Scholar] [CrossRef]
- Dean, M.; Raats, M.M.; Grunert, K.G.; Lumbers, M. Factors influencing eating a varied diet in old age. Public Health Nutr. 2009, 12, 2421–2427. [Google Scholar] [CrossRef] [Green Version]
- Amarya, S.; Singh, K.; Sabharwal, M. Changes during aging and their association with malnutrition. J. Clin. Gerontol. Geriatr. 2015, 6, 78–84. [Google Scholar] [CrossRef] [Green Version]
- Gomes, F.; Schuetz, P.; Bounoure, L.; Austin, P.; Ballesteros-Pomar, M.; Cederholm, T.; Fletcher, J.; Laviano, A.; Norman, K.; Poulia, K.A.; et al. ESPEN guidelines on nutritional support for polymorbid internal medicine patients. Clin. Nutr. 2018, 37, 336–353. [Google Scholar] [CrossRef] [Green Version]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cereda, E.; Cruz-Jentoft, A.; Goisser, S.; de Groot, L.; Großhauser, F.; Kiesswetter, E.; Norman, K.; et al. Management of Malnutrition in Older Patients-Current Approaches, Evidence and Open Questions. J. Clin. Med. 2019, 8, 974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Den Broeke, C.; De Burghgraeve, T.; Ummels, M.; Gescher, N.; Deckx, L.; Tjan-Heijnen, V.; Buntinx, F.; van den Akker, M. Occurrence of Malnutrition and Associated Factors in Community-Dwelling Older Adults: Those with a Recent Diagnosis of Cancer Are at Higher Risk. J. Nutr. Health Aging 2018, 22, 191–198. [Google Scholar] [CrossRef]
- Eckert, C.; Gell, N.M.; Wingood, M.; Schollmeyer, J.; Tarleton, E.K. Malnutrition Risk, Rurality, and Falls among Community-Dwelling Older Adults. J. Nutr. Health Aging 2021, 25, 624–627. [Google Scholar] [CrossRef] [PubMed]
- Khalatbari-Soltani, S.; Marques-Vidal, P. The economic cost of hospital malnutrition in Europe; a narrative review. Clin. Nutr. ESPEN 2015, 10, e89–e94. [Google Scholar] [CrossRef] [Green Version]
- Lengelé, L.; Bruyère, O.; Beaudart, C.; Reginster, J.Y.; Locquet, M. Impact of Malnutrition Status on Muscle Parameter Changes over a 5-Year Follow-Up of Community-Dwelling Older Adults from the SarcoPhAge Cohort. Nutrients 2021, 13, 407. [Google Scholar] [CrossRef] [PubMed]
- Coin, A.; Sergi, G.; Benincà, P.; Lupoli, L.; Cinti, G.; Ferrara, L.; Benedetti, G.; Tomasi, G.; Pisent, C.; Enzi, G. Bone mineral density and body composition in underweight and normal elderly subjects. Osteoporos. Int. 2000, 11, 1043–1050. [Google Scholar] [CrossRef]
- Geinoz, G.; Rapin, C.H.; Rizzoli, R.; Kraemer, R.; Buchs, B.; Slosman, D.; Michel, J.P.; Bonjour, J.P. Relationship between bone mineral density and dietary intakes in the elderly. Osteoporos. Int. 1993, 3, 242–248. [Google Scholar] [CrossRef]
- Bonjour, J.P.; Schurch, M.A.; Rizzoli, R. Nutritional aspects of hip fractures. Bone 1996, 18, 139s–144s. [Google Scholar] [CrossRef]
- Schaible, U.E.; Kaufmann, S.H. Malnutrition and infection: Complex mechanisms and global impacts. PLoS Med. 2007, 4, e115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawakami, K.; Kadota, J.; Iida, K.; Shirai, R.; Abe, K.; Kohno, S. Reduced immune function and malnutrition in the elderly. Tohoku J. Exp. Med. 1999, 187, 157–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alam, I.; Almajwal, A.; Alam, W.; Alam, I.; Ullah, N.; Abulmeaaty, M.; Razak, S.; Khan, S.; Pawelec, G.; Paracha, P. The immune-nutrition interplay in aging facts and controversies. Nutr. Healthy Aging 2019, 5, 73–95. [Google Scholar] [CrossRef] [Green Version]
- Fitzpatrick, F.; Skally, M.; O’Hanlon, C.; Foley, M.; Houlihan, J.; Gaughan, L.; Smith, O.; Moore, B.; Cunneen, S.; Sweeney, E.; et al. Food for thought. Malnutrition risk associated with increased risk of healthcare-associated infection. J. Hosp. Infect. 2019, 101, 300–304. [Google Scholar] [CrossRef]
- Shpata, V.; Ohri, I.; Nurka, T.; Prendushi, X. The prevalence and consequences of malnutrition risk in elderly Albanian intensive care unit patients. Clin. Interv. Aging 2015, 10, 481–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barchitta, M.; Maugeri, A.; Favara, G.; Magnano San Lio, R.; Evola, G.; Agodi, A.; Basile, G. Nutrition and Wound Healing: An Overview Focusing on the Beneficial Effects of Curcumin. Int. J. Mol. Sci. 2019, 20, 1119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsson, M.; Järbrink, K.; Divakar, U.; Bajpai, R.; Upton, Z.; Schmidtchen, A.; Car, J. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair Regen. 2019, 27, 114–125. [Google Scholar] [CrossRef] [Green Version]
- Hébuterne, X.; Bermon, S.; Schneider, S.M. Ageing and muscle: The effects of malnutrition, re-nutrition, and physical exercise. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 295–300. [Google Scholar] [CrossRef]
- Inouye, S.K.; Studenski, S.; Tinetti, M.E.; Kuchel, G.A. Geriatric syndromes: Clinical, research, and policy implications of a core geriatric concept. J. Am. Geriatr. Soc. 2007, 55, 780–791. [Google Scholar] [CrossRef]
- Kane, R.L.; Shamliyan, T.; Talley, K.; Pacala, J. The association between geriatric syndromes and survival. J. Am. Geriatr. Soc. 2012, 60, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Rausch, C.; van Zon, S.K.R.; Liang, Y.; Laflamme, L.; Möller, J.; de Rooij, S.E.; Bültmann, U. Geriatric Syndromes and Incident Chronic Health Conditions Among 9094 Older Community-Dwellers: Findings from the Lifelines Cohort Study. J. Am. Med. Dir. Assoc. 2021. [Google Scholar] [CrossRef] [PubMed]
- Won, C.W.; Yoo, H.J.; Yu, S.H.; Kim, C.O.; Dumlao, L.C.I.; Dewiasty, E.; Rowland, J.; Chang, H.H.; Wang, J.; Akishita, M.; et al. Lists of geriatric syndromes in the Asian-Pacific geriatric societies. Eur. Geriatr. Med. 2013, 4, 335–338. [Google Scholar] [CrossRef]
- Tan, V.M.H.; Pang, B.W.J.; Lau, L.K.; Jabbar, K.A.; Seah, W.T.; Chen, K.K.; Ng, T.P.; Wee, S.L. Malnutrition and Sarcopenia in Community-Dwelling Adults in Singapore: Yishun Health Study. J. Nutr. Health Aging 2021, 25, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Juby, A.G.; Mager, D.R. A review of nutrition screening tools used to assess the malnutrition-sarcopenia syndrome (MSS) in the older adult. Clin. Nutr. ESPEN 2019, 32, 8–15. [Google Scholar] [CrossRef]
- Stenholm, S.; Harris, T.B.; Rantanen, T.; Visser, M.; Kritchevsky, S.B.; Ferrucci, L. Sarcopenic obesity: Definition, cause and consequences. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 693–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fougère, B.; Morley, J.E. Editorial: Weight Loss is a Major Cause of Frailty. J. Nutr. Health Aging 2017, 21, 933–935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boulos, C.; Salameh, P.; Barberger-Gateau, P. Malnutrition and frailty in community dwelling older adults living in a rural setting. Clin. Nutr. 2016, 35, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Jeejeebhoy, K.N. Malnutrition, fatigue, frailty, vulnerability, sarcopenia and cachexia: Overlap of clinical features. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 213–219. [Google Scholar] [CrossRef]
- Gingrich, A.; Volkert, D.; Kiesswetter, E.; Thomanek, M.; Bach, S.; Sieber, C.C.; Zopf, Y. Prevalence and overlap of sarcopenia, frailty, cachexia and malnutrition in older medical inpatients. BMC Geriatr. 2019, 19, 120. [Google Scholar] [CrossRef] [Green Version]
- Norazman, C.W.; Adznam, S.N.; Jamaluddin, R. Malnutrition as Key Predictor of Physical Frailty among Malaysian Older Adults. Nutrients 2020, 12, 1713. [Google Scholar] [CrossRef]
- Norazman, C.W.; Adznam, S.N.; Jamaluddin, R. Physical Frailty among Urban-Living Community-Dwelling Older Adults in Malaysia. Int. J. Environ. Res. Public Health 2020, 17, 6549. [Google Scholar] [CrossRef]
- Wei, K.; Nyunt, M.S.; Gao, Q.; Wee, S.L.; Yap, K.B.; Ng, T.P. Association of Frailty and Malnutrition With Long-term Functional and Mortality Outcomes Among Community-Dwelling Older Adults: Results From the Singapore Longitudinal Aging Study 1. JAMA Netw. Open 2018, 1, e180650. [Google Scholar] [CrossRef] [Green Version]
- Zengarini, E.; Ruggiero, C.; Pérez-Zepeda, M.U.; Hoogendijk, E.O.; Vellas, B.; Mecocci, P.; Cesari, M. Fatigue: Relevance and implications in the aging population. Exp. Gerontol. 2015, 70, 78–83. [Google Scholar] [CrossRef]
- Filler, K.; Lyon, D.; Bennett, J.; McCain, N.; Elswick, R.; Lukkahatai, N.; Saligan, L.N. Association of Mitochondrial Dysfunction and Fatigue: A Review of the Literature. BBA Clin. 2014, 1, 12–23. [Google Scholar] [CrossRef] [Green Version]
- Herpich, C.; Franz, K.; Klaus, S.; Müller-Werdan, U.; Ost, M.; Norman, K. Age-related fatigue is associated with reduced mitochondrial function in peripheral blood mononuclear cells. Exp. Gerontol. 2021, 144, 111177. [Google Scholar] [CrossRef]
- Franz, K.; Otten, L.; Müller-Werdan, U.; Doehner, W.; Norman, K. Severe Weight Loss and Its Association with Fatigue in Old Patients at Discharge from a Geriatric Hospital. Nutrients 2019, 11, 2415. [Google Scholar] [CrossRef] [Green Version]
- Azzolino, D.; Arosio, B.; Marzetti, E.; Calvani, R.; Cesari, M. Nutritional Status as a Mediator of Fatigue and Its Underlying Mechanisms in Older People. Nutrients 2020, 12, 444. [Google Scholar] [CrossRef] [Green Version]
- Haß, U.; Herpich, C.; Norman, K. Anti-Inflammatory Diets and Fatigue. Nutrients 2019, 11, 2315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogawa, S. Nutritional management of older adults with cognitive decline and dementia. Geriatr. Gerontol. Int. 2014, 14 (Suppl. 2), 17–22. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Yu, W.; Liu, X.; Wan, T.; Chen, C.; Xiong, L.; Zhang, W.; Lü, Y. Associations between malnutrition and cognitive impairment in an elderly Chinese population: An analysis based on a 7-year database. Psychogeriatrics 2021, 21, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, M.A.; Mesas, A.E.; Garcia, A.R.; de Andrade, S.M. Malnutrition and depression among community-dwelling elderly people. J. Am. Med. Dir. Assoc. 2007, 8, 582–584. [Google Scholar] [CrossRef] [PubMed]
- Smoliner, C.; Norman, K.; Wagner, K.H.; Hartig, W.; Lochs, H.; Pirlich, M. Malnutrition and depression in the institutionalised elderly. Br. J. Nutr. 2009, 102, 1663–1667. [Google Scholar] [CrossRef] [Green Version]
- Yoshimura, K.; Yamada, M.; Kajiwara, Y.; Nishiguchi, S.; Aoyama, T. Relationship between depression and risk of malnutrition among community-dwelling young-old and old-old elderly people. Aging Ment. Health 2013, 17, 456–460. [Google Scholar] [CrossRef] [Green Version]
- Gentile, S.; Lacroix, O.; Durand, A.C.; Cretel, E.; Alazia, M.; Sambuc, R.; Bonin-Guillaume, S. Malnutrition: A highly predictive risk factor of short-term mortality in elderly presenting to the emergency department. J. Nutr. Health Aging 2013, 17, 290–294. [Google Scholar] [CrossRef]
- Komici, K.; Vitale, D.F.; Mancini, A.; Bencivenga, L.; Conte, M.; Provenzano, S.; Grieco, F.V.; Visaggi, L.; Ronga, I.; Cittadini, A.; et al. Impact of Malnutrition on Long-Term Mortality in Elderly Patients with Acute Myocardial Infarction. Nutrients 2019, 11, 224. [Google Scholar] [CrossRef] [Green Version]
- Söderström, L.; Rosenblad, A.; Thors Adolfsson, E.; Bergkvist, L. Malnutrition is associated with increased mortality in older adults regardless of the cause of death. Br. J. Nutr. 2017, 117, 532–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teigen, L.M.; Kuchnia, A.J.; Nagel, E.M.; Price, K.L.; Hurt, R.T.; Earthman, C.P. Diagnosing clinical malnutrition: Perspectives from the past and implications for the future. Clin. Nutr. ESPEN 2018, 26, 13–20. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition–A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De van der Schueren, M.A.E.; Keller, H.; Cederholm, T.; Barazzoni, R.; Compher, C.; Correia, M.; Gonzalez, M.C.; Jager-Wittenaar, H.; Pirlich, M.; Steiber, A.; et al. Global Leadership Initiative on Malnutrition (GLIM): Guidance on validation of the operational criteria for the diagnosis of protein-energy malnutrition in adults. Clin. Nutr. 2020, 39, 2872–2880. [Google Scholar] [CrossRef] [PubMed]
- Yeung, S.S.Y.; Chan, R.S.M.; Kwok, T.; Lee, J.S.W.; Woo, J. Malnutrition According to GLIM Criteria and Adverse Outcomes in Community-Dwelling Chinese Older Adults: A Prospective Analysis. J. Am. Med. Dir. Assoc. 2020. [Google Scholar] [CrossRef]
- Cederholm, T.; Barazzoni, R.; Austin, P.; Ballmer, P.; Biolo, G.; Bischoff, S.C.; Compher, C.; Correia, I.; Higashiguchi, T.; Holst, M.; et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin. Nutr. 2017, 36, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E. Mini nutritional assessment. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 29–41. [Google Scholar] [CrossRef]
- Vandewoude, M.F.; Alish, C.J.; Sauer, A.C.; Hegazi, R.A. Malnutrition-sarcopenia syndrome: Is this the future of nutrition screening and assessment for older adults? J. Aging Res. 2012, 2012, 651570. [Google Scholar] [CrossRef] [PubMed]
- Kinosian, B.; Jeejeebhoy, K.N. What is malnutrition? Does it matter? Nutrition 1995, 11, 196–197. [Google Scholar]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznaric, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef] [Green Version]
- Hengeveld, L.M.; Wijnhoven, H.A.H.; Olthof, M.R.; Brouwer, I.A.; Harris, T.B.; Kritchevsky, S.B.; Newman, A.B.; Visser, M. Prospective associations of poor diet quality with long-term incidence of protein-energy malnutrition in community-dwelling older adults: The Health, Aging, and Body Composition (Health ABC) Study. Am. J. Clin. Nutr. 2018, 107, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Hengeveld, L.M.; Boer, J.M.A.; Gaudreau, P.; Heymans, M.W.; Jagger, C.; Mendonça, N.; Ocké, M.C.; Presse, N.; Sette, S.; Simonsick, E.M.; et al. Prevalence of protein intake below recommended in community-dwelling older adults: A meta-analysis across cohorts from the PROMISS consortium. J. Cachexia Sarcopenia Muscle 2020, 11, 1212–1222. [Google Scholar] [CrossRef]
- Conzade, R.; Koenig, W.; Heier, M.; Schneider, A.; Grill, E.; Peters, A.; Thorand, B. Prevalence and Predictors of Subclinical Micronutrient Deficiency in German Older Adults: Results from the Population-Based KORA-Age Study. Nutrients 2017, 9, 1276. [Google Scholar] [CrossRef] [Green Version]
- IOM (Institute of Medicine). Providing Healthy and Safe Foods as We Age: Workshop Summary; The National Academies Press: Washington, DC, USA, 2010. [Google Scholar]
- Hamer, D.H.; Sempértegui, F.; Estrella, B.; Tucker, K.L.; Rodríguez, A.; Egas, J.; Dallal, G.E.; Selhub, J.; Griffiths, J.K.; Meydani, S.N. Micronutrient deficiencies are associated with impaired immune response and higher burden of respiratory infections in elderly Ecuadorians. J. Nutr. 2009, 139, 113–119. [Google Scholar] [CrossRef] [Green Version]
- Wallace, T.C.; Frankenfeld, C.L.; Frei, B.; Shah, A.V.; Yu, C.R.; van Klinken, B.J.; Adeleke, M. Multivitamin/Multimineral Supplement Use is Associated with Increased Micronutrient Intakes and Biomarkers and Decreased Prevalence of Inadequacies and Deficiencies in Middle-Aged and Older Adults in the United States. J. Nutr. Gerontol. Geriatr. 2019, 38, 307–328. [Google Scholar] [CrossRef]
- Elmadfa, I.; Meyer, A.L. Present Knowledge in Nutrition. Chapter 5–Nutrition, Aging, and Requirements in the Elderly; Marriott, B.P., Birt, D.F., Stallings, V.A., Yates, A.A., Eds.; Academic Press: Cambridge, MA, USA, 2020; Volume 11. [Google Scholar]
- Maggini, S.; Pierre, A.; Calder, P.C. Immune Function and Micronutrient Requirements Change over the Life Course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef] [Green Version]
- Karadima, V.; Kraniotou, C.; Bellos, G.; Tsangaris, G.T. Drug-micronutrient interactions: Food for thought and thought for action. EPMA J. 2016, 7, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gröber, U.; Schmidt, J.; Kisters, K. Important drug-micronutrient interactions: A selection for clinical practice. Crit. Rev. Food Sci. Nutr. 2020, 60, 257–275. [Google Scholar] [CrossRef] [PubMed]
- Kehl-Fie, T.E.; Skaar, E.P. Nutritional immunity beyond iron: A role for manganese and zinc. Curr. Opin. Chem. Biol. 2010, 14, 218–224. [Google Scholar] [CrossRef] [Green Version]
- Mocchegiani, E.; Romeo, J.; Malavolta, M.; Costarelli, L.; Giacconi, R.; Diaz, L.E.; Marcos, A. Zinc: Dietary intake and impact of supplementation on immune function in elderly. Age 2013, 35, 839–860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, A.; Spira, D.; Steinhagen-Thiessen, E.; Demuth, I.; Norman, K. Zinc Deficiency Is associated With Depressive Symptoms-Results From the Berlin Aging Study II. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 72, 1149–1154. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Asakawa, A.; Li, J.B.; Tsai, M.; Amitani, H.; Ohinata, K.; Komai, M.; Inui, A. Zinc as an appetite stimulator–the possible role of zinc in the progression of diseases such as cachexia and sarcopenia. Recent Pat. Food Nutr. Agric. 2011, 3, 226–231. [Google Scholar] [CrossRef]
- Klotz, L.O.; Kröncke, K.D.; Buchczyk, D.P.; Sies, H. Role of copper, zinc, selenium and tellurium in the cellular defense against oxidative and nitrosative stress. J. Nutr. 2003, 133, 1448s–1451s. [Google Scholar] [CrossRef]
- Mocchegiani, E.; Malavolta, M. Zinc-gene interaction related to inflammatory/immune response in ageing. Genes Nutr. 2008, 3, 61–75. [Google Scholar] [CrossRef] [Green Version]
- Mocchegiani, E.; Basso, A.; Giacconi, R.; Piacenza, F.; Costarelli, L.; Pierpaoli, S.; Malavolta, M. Diet (zinc)-gene interaction related to inflammatory/immune response in ageing: Possible link with frailty syndrome? Biogerontology 2010, 11, 589–595. [Google Scholar] [CrossRef]
- Lauretani, F.; Semba, R.D.; Bandinelli, S.; Dayhoff-Brannigan, M.; Giacomini, V.; Corsi, A.M.; Guralnik, J.M.; Ferrucci, L. Low plasma carotenoids and skeletal muscle strength decline over 6 years. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 376–383. [Google Scholar] [CrossRef] [Green Version]
- Lauretani, F.; Semba, R.D.; Bandinelli, S.; Dayhoff-Brannigan, M.; Lauretani, F.; Corsi, A.M.; Guralnik, J.M.; Ferrucci, L. Carotenoids as protection against disability in older persons. Rejuvenation Res. 2008, 11, 557–563. [Google Scholar] [CrossRef]
- Bartali, B.; Frongillo, E.A.; Guralnik, J.M.; Stipanuk, M.H.; Allore, H.G.; Cherubini, A.; Bandinelli, S.; Ferrucci, L.; Gill, T.M. Serum micronutrient concentrations and decline in physical function among older persons. JAMA 2008, 299, 308–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cesari, M.; Pahor, M.; Bartali, B.; Cherubini, A.; Penninx, B.W.; Williams, G.R.; Atkinson, H.; Martin, A.; Guralnik, J.M.; Ferrucci, L. Antioxidants and physical performance in elderly persons: The Invecchiare in Chianti (InCHIANTI) study. Am. J. Clin. Nutr. 2004, 79, 289–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Rodríguez, D.; Annweiler, C.; Ronquillo-Moreno, N.; Tortosa-Rodríguez, A.; Guillén-Solà, A.; Vázquez-Ibar, O.; Escalada, F.; Muniesa, J.M.; Marco, E. Clinical application of the basic definition of malnutrition proposed by the European Society for Clinical Nutrition and Metabolism (ESPEN): Comparison with classical tools in geriatric care. Arch. Gerontol. Geriatr. 2018, 76, 210–214. [Google Scholar] [CrossRef] [Green Version]
- Leij-Halfwerk, S.; Verwijs, M.H.; van Houdt, S.; Borkent, J.W.; Guaitoli, P.R.; Pelgrim, T.; Heymans, M.W.; Power, L.; Visser, M.; Corish, C.A.; et al. Prevalence of protein-energy malnutrition risk in European older adults in community, residential and hospital settings, according to 22 malnutrition screening tools validated for use in adults >/=65 years: A systematic review and meta-analysis. Maturitas 2019, 126, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Crichton, M.; Craven, D.; Mackay, H.; Marx, W.; de van der Schueren, M.; Marshall, S. A systematic review, meta-analysis and meta-regression of the prevalence of protein-energy malnutrition: Associations with geographical region and sex. Age Ageing 2019, 48, 38–48. [Google Scholar] [CrossRef]
- Cereda, E.; Pedrolli, C.; Klersy, C.; Bonardi, C.; Quarleri, L.; Cappello, S.; Turri, A.; Rondanelli, M.; Caccialanza, R. Nutritional status in older persons according to healthcare setting: A systematic review and meta-analysis of prevalence data using MNA®. Clin. Nutr. 2016, 35, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Wolters, M.; Volkert, D.; Streicher, M.; Kiesswetter, E.; Torbahn, G.; O’Connor, E.M.; O’Keeffe, M.; Kelly, M.; O’Herlihy, E.; O’Toole, P.W.; et al. Prevalence of malnutrition using harmonized definitions in older adults from different settings–A MaNuEL study. Clin. Nutr. 2019, 38, 2389–2398. [Google Scholar] [CrossRef] [Green Version]
- Streicher, M.; van Zwienen-Pot, J.; Bardon, L.; Nagel, G.; Teh, R.; Meisinger, C.; Colombo, M.; Torbahn, G.; Kiesswetter, E.; Flechtner-Mors, M.; et al. Determinants of Incident Malnutrition in Community-Dwelling Older Adults: A MaNuEL Multicohort Meta-Analysis. J. Am. Geriatr. Soc. 2018, 66, 2335–2343. [Google Scholar] [CrossRef] [Green Version]
- O’Keeffe, M.; Kelly, M.; O’Herlihy, E.; O’Toole, P.W.; Kearney, P.M.; Timmons, S.; O’Shea, E.; Stanton, C.; Hickson, M.; Rolland, Y.; et al. Potentially modifiable determinants of malnutrition in older adults: A systematic review. Clin. Nutr. 2019, 38, 2477–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volkert, D.; Kiesswetter, E.; Cederholm, T.; Donini, L.M.; Eglseer, D.; Norman, K.; Schneider, S.M.; Ströbele-Benschop, N.; Torbahn, G.; Wirth, R.; et al. Development of a Model on Determinants of Malnutrition in Aged Persons: A MaNuEL Project. Gerontol. Geriatr. Med. 2019, 5, 2333721419858438. [Google Scholar] [CrossRef] [PubMed]
- Rémond, D.; Shahar, D.R.; Gille, D.; Pinto, P.; Kachal, J.; Peyron, M.A.; Dos Santos, C.N.; Walther, B.; Bordoni, A.; Dupont, D.; et al. Understanding the gastrointestinal tract of the elderly to develop dietary solutions that prevent malnutrition. Oncotarget 2015, 6, 13858–13898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, R.; Wilms, E.; Masclee, A.A.M.; Smidt, H.; Zoetendal, E.G.; Jonkers, D. Age-dependent changes in GI physiology and microbiota: Time to reconsider? Gut 2018, 67, 2213–2222. [Google Scholar] [CrossRef]
- Nagpal, R.; Mainali, R.; Ahmadi, S.; Wang, S.; Singh, R.; Kavanagh, K.; Kitzman, D.W.; Kushugulova, A.; Marotta, F.; Yadav, H. Gut microbiome and aging: Physiological and mechanistic insights. Nutr. Healthy Aging 2018, 4, 267–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van de Wouw, M.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. Microbiota-Gut-Brain Axis: Modulator of Host Metabolism and Appetite. J. Nutr. 2017, 147, 727–745. [Google Scholar] [CrossRef] [Green Version]
- Ticinesi, A.; Lauretani, F.; Milani, C.; Nouvenne, A.; Tana, C.; Del Rio, D.; Maggio, M.; Ventura, M.; Meschi, T. Aging Gut Microbiota at the Cross-Road between Nutrition, Physical Frailty, and Sarcopenia: Is There a Gut-Muscle Axis? Nutrients 2017, 9, 1303. [Google Scholar] [CrossRef] [Green Version]
- Moss, C.; Dhillo, W.S.; Frost, G.; Hickson, M. Gastrointestinal hormones: The regulation of appetite and the anorexia of ageing. J. Hum. Nutr. Diet. 2012, 25, 3–15. [Google Scholar] [CrossRef]
- Atalayer, D.; Astbury, N.M. Anorexia of aging and gut hormones. Aging Dis. 2013, 4, 264–275. [Google Scholar] [CrossRef]
- Morley, J.E.; Miller, D.K.; Perry, H.M., 3rd; Patrick, P.; Guigoz, Y.; Vellas, B. Anorexia of aging, leptin, and the Mini Nutritional Assessment. Nestle Nutr. Workshop Ser. Clin. Perform. Programme 1999, 1, 67–76. [Google Scholar] [CrossRef] [Green Version]
- Ueno, H.; Nakazato, M. Mechanistic relationship between the vagal afferent pathway, central nervous system and peripheral organs in appetite regulation. J. Diabetes Investig. 2016, 7, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Kirsz, K.; Zieba, D.A. Ghrelin-mediated appetite regulation in the central nervous system. Peptides 2011, 32, 2256–2264. [Google Scholar] [CrossRef]
- Pliquett, R.U.; Führer, D.; Falk, S.; Zysset, S.; von Cramon, D.Y.; Stumvoll, M. The effects of insulin on the central nervous system--focus on appetite regulation. Horm. Metab. Res. 2006, 38, 442–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serra-Prat, M.; Mans, E.; Palomera, E.; Clavé, P. Gastrointestinal peptides, gastrointestinal motility, and anorexia of aging in frail elderly persons. Neurogastroenterol. Motil. 2013, 25, 291-e245. [Google Scholar] [CrossRef]
- Bauer, J.M.; Haack, A.; Winning, K.; Wirth, R.; Fischer, B.; Uter, W.; Erdmann, J.; Schusdziarra, V.; Sieber, C.C. Impaired postprandial response of active ghrelin and prolonged suppression of hunger sensation in the elderly. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2010, 65, 307–311. [Google Scholar] [CrossRef] [Green Version]
- Morton, G.J.; Cummings, D.E.; Baskin, D.G.; Barsh, G.S.; Schwartz, M.W. Central nervous system control of food intake and body weight. Nature 2006, 443, 289–295. [Google Scholar] [CrossRef]
- Brown, W.E.; Bradford, B.J. Invited review: Mechanisms of hypophagia during disease. J. Dairy Sci. 2021. [Google Scholar] [CrossRef]
- Wysokinski, A.; Sobow, T.; Kloszewska, I.; Kostka, T. Mechanisms of the anorexia of aging—A review. Age 2015, 37, 9821. [Google Scholar] [CrossRef]
- Cox, N.J.; Ibrahim, K.; Sayer, A.A.; Robinson, S.M.; Roberts, H.C. Assessment and Treatment of the Anorexia of Aging: A Systematic Review. Nutrients 2019, 11, 144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giezenaar, C.; Chapman, I.; Luscombe-Marsh, N.; Feinle-Bisset, C.; Horowitz, M.; Soenen, S. Ageing Is Associated with Decreases in Appetite and Energy Intake--A Meta-Analysis in Healthy Adults. Nutrients 2016, 8, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, M.; Shatenstein, B.; Gaudreau, P.; Morais, J.A.; Payette, H. Seniors’ body weight dissatisfaction and longitudinal associations with weight changes, anorexia of aging, and obesity: Results from the NuAge Study. J. Aging Health 2015, 27, 220–238. [Google Scholar] [CrossRef]
- Morley, J.E. Anorexia of aging: A true geriatric syndrome. J. Nutr. Health Aging 2012, 16, 422–425. [Google Scholar] [CrossRef]
- Landi, F.; Liperoti, R.; Russo, A.; Giovannini, S.; Tosato, M.; Barillaro, C.; Capoluongo, E.; Bernabei, R.; Onder, G. Association of anorexia with sarcopenia in a community-dwelling elderly population: Results from the ilSIRENTE study. Eur. J. Nutr. 2013, 52, 1261–1268. [Google Scholar] [CrossRef]
- Tsutsumimoto, K.; Doi, T.; Makizako, H.; Hotta, R.; Nakakubo, S.; Makino, K.; Suzuki, T.; Shimada, H. The association between anorexia of aging and physical frailty: Results from the national center for geriatrics and gerontology’s study of geriatric syndromes. Maturitas 2017, 97, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Wall, B.T.; Gorissen, S.H.; Pennings, B.; Koopman, R.; Groen, B.B.; Verdijk, L.B.; van Loon, L.J. Aging Is Accompanied by a Blunted Muscle Protein Synthetic Response to Protein Ingestion. PLoS ONE 2015, 10, e0140903. [Google Scholar] [CrossRef]
- Dickinson, J.M.; Volpi, E.; Rasmussen, B.B. Exercise and nutrition to target protein synthesis impairments in aging skeletal muscle. Exerc. Sport Sci. Rev. 2013, 41, 216–223. [Google Scholar] [CrossRef]
- Drummond, M.J.; Dreyer, H.C.; Pennings, B.; Fry, C.S.; Dhanani, S.; Dillon, E.L.; Sheffield-Moore, M.; Volpi, E.; Rasmussen, B.B. Skeletal muscle protein anabolic response to resistance exercise and essential amino acids is delayed with aging. J. Appl. Physiol. 2008, 104, 1452–1461. [Google Scholar] [CrossRef]
- Markofski, M.M.; Dickinson, J.M.; Drummond, M.J.; Fry, C.S.; Fujita, S.; Gundermann, D.M.; Glynn, E.L.; Jennings, K.; Paddon-Jones, D.; Reidy, P.T.; et al. Effect of age on basal muscle protein synthesis and mTORC1 signaling in a large cohort of young and older men and women. Exp. Gerontol. 2015, 65, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Morais, J.A.; Jacob, K.W.; Chevalier, S. Effects of aging and insulin resistant states on protein anabolic responses in older adults. Exp. Gerontol. 2018, 108, 262–268. [Google Scholar] [CrossRef]
- Barclay, R.D.; Burd, N.A.; Tyler, C.; Tillin, N.A.; Mackenzie, R.W. The Role of the IGF-1 Signaling Cascade in Muscle Protein Synthesis and Anabolic Resistance in Aging Skeletal Muscle. Front. Nutr. 2019, 6, 146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, R.; Dalla Man, C.; Campioni, M.; Basu, A.; Klee, G.; Toffolo, G.; Cobelli, C.; Rizza, R.A. Effects of age and sex on postprandial glucose metabolism: Differences in glucose turnover, insulin secretion, insulin action, and hepatic insulin extraction. Diabetes 2006, 55, 2001–2014. [Google Scholar] [CrossRef] [Green Version]
- Herpich, C.; Haß, U.; Kochlik, B.; Franz, K.; Laeger, T.; Klaus, S.; Bosy-Westphal, A.; Norman, K. Postprandial dynamics and response of fibroblast growth factor 21 in older adults. Clin. Nutr. 2021, 40, 3765–3771. [Google Scholar] [CrossRef]
- Conte, M.; Ostan, R.; Fabbri, C.; Santoro, A.; Guidarelli, G.; Vitale, G.; Mari, D.; Sevini, F.; Capri, M.; Sandri, M.; et al. Human aging and longevity are characterized by high levels of mitokines. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 74, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Oost, L.J.; Kustermann, M.; Armani, A.; Blaauw, B.; Romanello, V. Fibroblast growth factor 21 controls mitophagy and muscle mass. J. Cachexia Sarcopenia Muscle 2019, 10, 630–642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tezze, C.; Romanello, V.; Desbats, M.A.; Fadini, G.P.; Albiero, M.; Favaro, G.; Ciciliot, S.; Soriano, M.E.; Morbidoni, V.; Cerqua, C.; et al. Age-Associated Loss of OPA1 in Muscle Impacts Muscle Mass, Metabolic Homeostasis, Systemic Inflammation, and Epithelial Senescence. Cell Metab. 2017, 25, 1374–1389.e1376. [Google Scholar] [CrossRef]
- Sun, H.; Sherrier, M.; Li, H. Skeletal Muscle and Bone–Emerging Targets of Fibroblast Growth Factor-21. Front. Physiol. 2021, 12, 625287. [Google Scholar] [CrossRef]
- Franz, K.; Ost, M.; Otten, L.; Herpich, C.; Coleman, V.; Endres, A.S.; Klaus, S.; Müller-Werdan, U.; Norman, K. Higher serum levels of fibroblast growth factor 21 in old patients with cachexia. Nutrition 2019, 63–64, 81–86. [Google Scholar] [CrossRef]
- Lin, Z.; Tian, H.; Lam, K.S.; Lin, S.; Hoo, R.C.; Konishi, M.; Itoh, N.; Wang, Y.; Bornstein, S.R.; Xu, A.; et al. Adiponectin mediates the metabolic effects of FGF21 on glucose homeostasis and insulin sensitivity in mice. Cell Metab. 2013, 17, 779–789. [Google Scholar] [CrossRef] [Green Version]
- Menzaghi, C.; Trischitta, V. The Adiponectin Paradox for All-Cause and Cardiovascular Mortality. Diabetes 2018, 67, 12–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herpich, C.; Kochlik, B.; Haß, U.; Weber, D.; Grune, T.; Norman, K. Altered Adiponectin Response in Older Women Following Dextrose and High-Fat Dietary Challenges. Mol. Nutr. Food Res. 2021, e2100487. [Google Scholar] [CrossRef]
- Franceschi, C.; Bonafè, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Meftahi, G.H.; Jangravi, Z.; Sahraei, H.; Bahari, Z. The possible pathophysiology mechanism of cytokine storm in elderly adults with COVID-19 infection: The contribution of “inflame-aging”. Inflamm. Res. 2020, 69, 825–839. [Google Scholar] [CrossRef]
- Omarjee, L.; Perrot, F.; Meilhac, O.; Mahe, G.; Bousquet, G.; Janin, A. Immunometabolism at the cornerstone of inflammaging, immunosenescence, and autoimmunity in COVID-19. Aging 2020, 12, 26263–26278. [Google Scholar] [CrossRef]
- Rothenberg, E. Coronavirus Disease 19 from the Perspective of Ageing with Focus on Nutritional Status and Nutrition Management-A Narrative Review. Nutrients 2021, 13, 1294. [Google Scholar] [CrossRef]
- Morrisette-Thomas, V.; Cohen, A.A.; Fulop, T.; Riesco, E.; Legault, V.; Li, Q.; Milot, E.; Dusseault-Belanger, F.; Ferrucci, L. Inflamm-aging does not simply reflect increases in pro-inflammatory markers. Mech. Ageing Dev. 2014, 139, 49–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minciullo, P.L.; Catalano, A.; Mandraffino, G.; Casciaro, M.; Crucitti, A.; Maltese, G.; Morabito, N.; Lasco, A.; Gangemi, S.; Basile, G. Inflammaging and Anti-Inflammaging: The Role of Cytokines in Extreme Longevity. Arch. Immunol. Ther. Exp. 2016, 64, 111–126. [Google Scholar] [CrossRef]
- Pinti, M.; Cevenini, E.; Nasi, M.; De Biasi, S.; Salvioli, S.; Monti, D.; Benatti, S.; Gibellini, L.; Cotichini, R.; Stazi, M.A.; et al. Circulating mitochondrial DNA increases with age and is a familiar trait: Implications for “inflamm-aging”. Eur. J. Immunol. 2014, 44, 1552–1562. [Google Scholar] [CrossRef]
- Conte, M.; Martucci, M.; Chiariello, A.; Franceschi, C.; Salvioli, S. Mitochondria, immunosenescence and inflammaging: A role for mitokines? In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar] [CrossRef]
- Onyango, I.G.; Jauregui, G.V.; Čarná, M.; Bennett, J.P., Jr.; Stokin, G.B. Neuroinflammation in Alzheimer’s Disease. Biomedicines 2021, 9, 524. [Google Scholar] [CrossRef]
- Liberale, L.; Montecucco, F.; Tardif, J.C.; Libby, P.; Camici, G.G. Inflamm-ageing: The role of inflammation in age-dependent cardiovascular disease. Eur. Heart J. 2020, 41, 2974–2982. [Google Scholar] [CrossRef]
- Livshits, G.; Kalinkovich, A. Inflammaging as a common ground for the development and maintenance of sarcopenia, obesity, cardiomyopathy and dysbiosis. Ageing Res. Rev. 2019, 56, 100980. [Google Scholar] [CrossRef]
- Prattichizzo, F.; De Nigris, V.; Spiga, R.; Mancuso, E.; La Sala, L.; Antonicelli, R.; Testa, R.; Procopio, A.D.; Olivieri, F.; Ceriello, A. Inflammageing and metaflammation: The yin and yang of type 2 diabetes. Ageing Res. Rev. 2018, 41, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, Bone, and Fat Crosstalk: The Biological Role of Myokines, Osteokines, and Adipokines. Curr. Osteoporos. Rep. 2020, 18, 388–400. [Google Scholar] [CrossRef]
- Marzetti, E.; Picca, A.; Marini, F.; Biancolillo, A.; Coelho-Junior, H.J.; Gervasoni, J.; Bossola, M.; Cesari, M.; Onder, G.; Landi, F.; et al. Inflammatory signatures in older persons with physical frailty and sarcopenia: The frailty “cytokinome” at its core. Exp. Gerontol. 2019, 122, 129–138. [Google Scholar] [CrossRef]
- Ostan, R.; Lanzarini, C.; Pini, E.; Scurti, M.; Vianello, D.; Bertarelli, C.; Fabbri, C.; Izzi, M.; Palmas, G.; Biondi, F.; et al. Inflammaging and cancer: A challenge for the Mediterranean diet. Nutrients 2015, 7, 2589–2621. [Google Scholar] [CrossRef] [Green Version]
- Olivieri, F.; Rippo, M.R.; Monsurrò, V.; Salvioli, S.; Capri, M.; Procopio, A.D.; Franceschi, C. MicroRNAs linking inflamm-aging, cellular senescence and cancer. Ageing Res. Rev. 2013, 12, 1056–1068. [Google Scholar] [CrossRef] [PubMed]
- Bonafè, M.; Storci, G.; Franceschi, C. Inflamm-aging of the stem cell niche: Breast cancer as a paradigmatic example: Breakdown of the multi-shell cytokine network fuels cancer in aged people. Bioessays 2012, 34, 40–49. [Google Scholar] [CrossRef]
- Calder, P.C.; Bosco, N.; Bourdet-Sicard, R.; Capuron, L.; Delzenne, N.; Dore, J.; Franceschi, C.; Lehtinen, M.J.; Recker, T.; Salvioli, S.; et al. Health relevance of the modification of low grade inflammation in ageing (inflammageing) and the role of nutrition. Ageing Res. Rev. 2017, 40, 95–119. [Google Scholar] [CrossRef]
- Yeh, S.S.; Schuster, M.W. Geriatric cachexia: The role of cytokines. Am. J. Clin. Nutr. 1999, 70, 183–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evans, W.J.; Morley, J.E.; Argiles, J.; Bales, C.; Baracos, V.; Guttridge, D.; Jatoi, A.; Kalantar-Zadeh, K.; Lochs, H.; Mantovani, G.; et al. Cachexia: A new definition. Clin. Nutr. 2008, 27, 793–799. [Google Scholar] [CrossRef]
- Tracey, K.J.; Wei, H.; Manogue, K.R.; Fong, Y.; Hesse, D.G.; Nguyen, H.T.; Kuo, G.C.; Beutler, B.; Cotran, R.S.; Cerami, A.; et al. Cachectin/tumor necrosis factor induces cachexia, anemia, and inflammation. J. Exp. Med. 1988, 167, 1211–1227. [Google Scholar] [CrossRef]
- Oldenburg, H.S.; Rogy, M.A.; Lazarus, D.D.; Van Zee, K.J.; Keeler, B.P.; Chizzonite, R.A.; Lowry, S.F.; Moldawer, L.L. Cachexia and the acute-phase protein response in inflammation are regulated by interleukin-6. Eur. J. Immunol. 1993, 23, 1889–1894. [Google Scholar] [CrossRef]
- Ershler, W.B. Interleukin-6: A cytokine for gerontologists. J. Am. Geriatr. Soc. 1993, 41, 176–181. [Google Scholar] [CrossRef]
- Boirie, Y. Physiopathological mechanism of sarcopenia. J. Nutr. Health Aging 2009, 13, 717–723. [Google Scholar] [CrossRef]
- Fatyga, P.; Pac, A.; Fedyk-Łukasik, M.; Grodzicki, T.; Skalska, A. The relationship between malnutrition risk and inflammatory biomarkers in outpatient geriatric population. Eur. Geriatr. Med. 2020, 11, 383–391. [Google Scholar] [CrossRef] [Green Version]
- Correa-Pérez, A.; Abraha, I.; Cherubini, A.; Collinson, A.; Dardevet, D.; de Groot, L.C.P.G.M. Efficacy of non-pharmacological interventions on nutritional status and clinical outcomes in older people with malnutrition or at risk of malnutrition: A systematic review. The SENATOR project ONTOP series and MaNuEL knowledge hub project. Aging Res. Rev. 2019, 49, 27–48. [Google Scholar] [CrossRef] [Green Version]
- Reinders, I.; Volkert, D.; de Groot, L.; Beck, A.M.; Feldblum, I.; Jobse, I.; Neelemaat, F.; de van der Schueren, M.A.E.; Shahar, D.R.; Smeets, E.; et al. Effectiveness of nutritional interventions in older adults at risk of malnutrition across different health care settings: Pooled analyses of individual participant data from nine randomized controlled trials. Clin. Nutr. 2019, 38, 1797–1806. [Google Scholar] [CrossRef] [Green Version]
- Höhn, A.; Weber, D.; Jung, T.; Ott, C.; Hugo, M.; Kochlik, B.; Kehm, R.; König, J.; Grune, T.; Castro, J.P. Happily (n)ever after: Aging in the context of oxidative stress, proteostasis loss and cellular senescence. Redox Biol. 2017, 11, 482–501. [Google Scholar] [CrossRef]
- Fisher, L. Food Science and Nutrition; ED-Tech Press: Waltham Abbey, UK, 2019. [Google Scholar]
- Ghosh, T.S.; Rampelli, S.; Jeffery, I.B.; Santoro, A.; Neto, M.; Capri, M.; Giampieri, E.; Jennings, A.; Candela, M.; Turroni, S.; et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: The NU-AGE 1-year dietary intervention across five European countries. Gut 2020, 69, 1218–1228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deutz, N.E.P.; Ashurst, I.; Ballesteros, M.D.; Bear, D.E.; Cruz-Jentoft, A.J.; Genton, L.; Landi, F.; Laviano, A.; Norman, K.; Prado, C.M. The Underappreciated Role of Low Muscle Mass in the Management of Malnutrition. J. Am. Med. Dir. Assoc. 2019, 20, 22–27. [Google Scholar] [CrossRef] [Green Version]
- Van der Pols-Vijlbrief, R.; Wijnhoven, H.A.H.; Bosmans, J.E.; Twisk, J.W.R.; Visser, M. Targeting the underlying causes of undernutrition. Cost-effectiveness of a multifactorial personalized intervention in community-dwelling older adults: A randomized controlled trial. Clin. Nutr. 2017, 36, 1498–1508. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norman, K.; Haß, U.; Pirlich, M. Malnutrition in Older Adults—Recent Advances and Remaining Challenges. Nutrients 2021, 13, 2764. https://doi.org/10.3390/nu13082764
Norman K, Haß U, Pirlich M. Malnutrition in Older Adults—Recent Advances and Remaining Challenges. Nutrients. 2021; 13(8):2764. https://doi.org/10.3390/nu13082764
Chicago/Turabian StyleNorman, Kristina, Ulrike Haß, and Matthias Pirlich. 2021. "Malnutrition in Older Adults—Recent Advances and Remaining Challenges" Nutrients 13, no. 8: 2764. https://doi.org/10.3390/nu13082764
APA StyleNorman, K., Haß, U., & Pirlich, M. (2021). Malnutrition in Older Adults—Recent Advances and Remaining Challenges. Nutrients, 13(8), 2764. https://doi.org/10.3390/nu13082764




