Creatine for Exercise and Sports Performance, with Recovery Considerations for Healthy Populations
Abstract
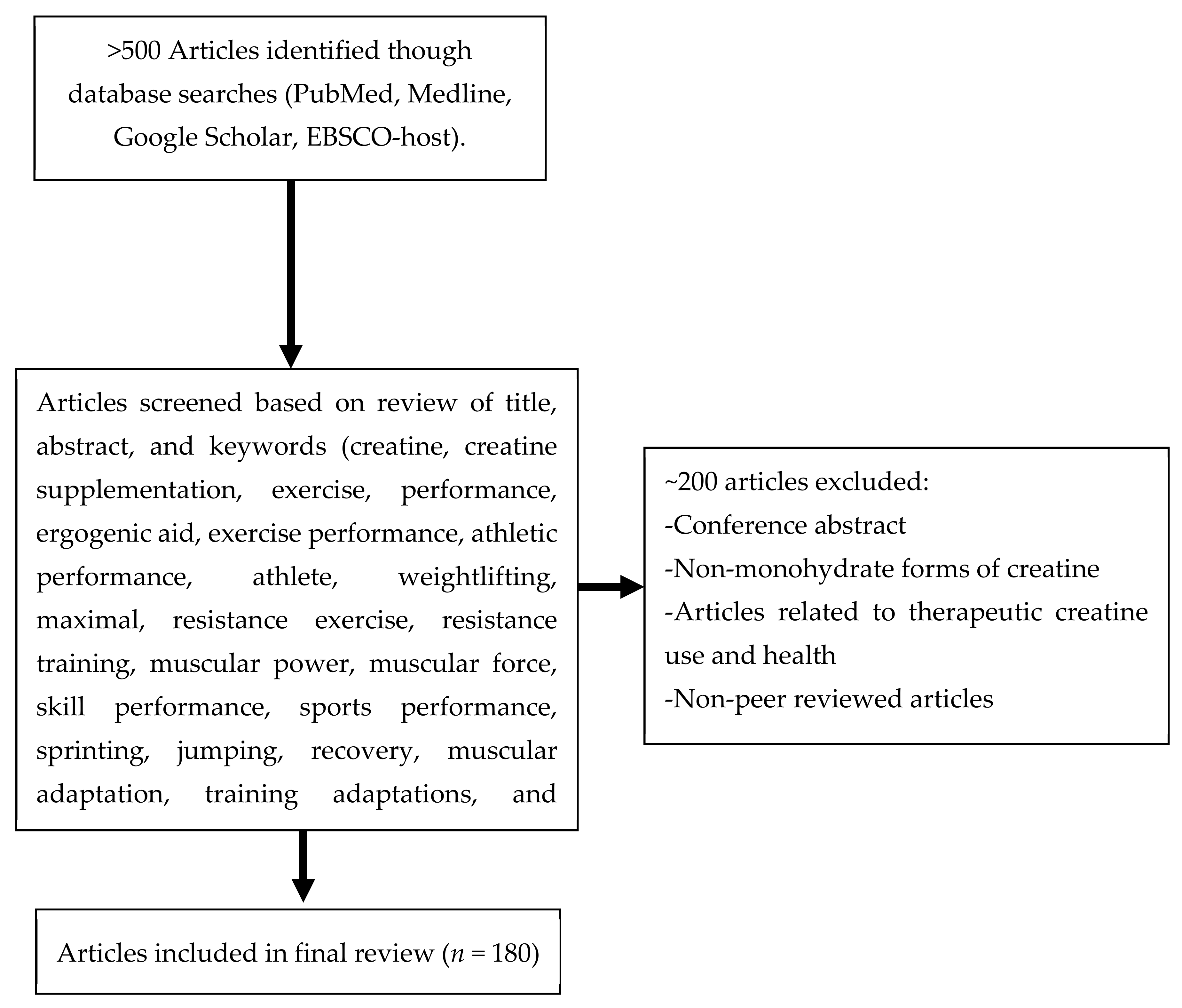
1. Introduction
2. Materials and Methods
3. Exercise and Sports Performance
3.1. Strength and Power
3.1.1. Short Term Benefits (<2 Weeks)
3.1.2. Longer-Term Training Adaptations (>2 Weeks)
3.1.3. Athletes
3.1.4. Untrained
3.2. Exercise Capacity/Prolonged High-Intensity Efforts
3.3. Sport-Specific Performance
3.3.1. Agility Performance
3.3.2. Sprint Performance
3.3.3. Jump Performance
3.3.4. Selected Competitive Athletes
American Football
Track and Field
Swimming
3.4. Endurance Performance
4. Recovery
4.1. Augmented Recovery Following Exercise
Augmented Recovery during Intermittent Activities
4.2. Loss of Force Production, Muscle Damage, Soreness, and Inflammation
4.3. Immobilization and Muscle Dysfunction
5. Other Considerations
6. Conclusions
- Creatine supplementation is safe during short- and long-term intervals for healthy males and females, as well as in younger and older individuals.
- Creatine supplementation, ingested at 0.3 g/kg/day for 3–5 consecutive days or 20 g/day for 5–7 successive days, has been shown to quickly increase intramuscular creatine, yielding immediate ergogenic benefits. Correspondingly, a regimen of 3–5 g/day over 4 weeks increased creatine stores, augmented muscle performance, mitigated recovery factors, and resulted in muscle accretion.
- Creatine supplementation intermixed with carbohydrates or carbohydrates and protein appears to be efficacious in increasing intramuscular creatine retention, although the additional benefits in terms of performance outcomes appear to be nebulous.
- Creatine supplementation appears to provide an ergogenic effect when assessing isolated or individual bouts of peak or maximal force production.
- Creatine supplementation facilitates more significant improvements in strength and FFM.
- Creatine supplementation provides benefits during single and repeated sprints and may increase agility and jumping performance.
- Creatine supplementation appears to provide ergogenic benefits to aerobic endurance bouts with positive physiological adaptations.
- Creatine supplementation may enhance recovery from intense exercise and possibly provide synergistic benefits during the post-injury rehabilitation period.
- Creatine supplementation provides positive benefits to both males and females, athletes and recreational fitness enthusiasts, as well as younger and older individuals.
- Creatine supplementation provides more significant augmentations of intramuscular creatine in vegans than omnivores, due to lower initial levels of creatine stores, with both groups receiving comparable ergogenic benefits.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.E.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN exercise & sports nutrition review update: Research & recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 38. [Google Scholar] [CrossRef]
- Williams, M.H. Facts and fallacies of purported ergogenic amino acid supplements. Clin. Sports Med. 1999, 18, 633–649. [Google Scholar] [CrossRef]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 1–18. [Google Scholar] [CrossRef]
- Antonio, J.; Candow, D.G.; Forbes, S.C.; Gualano, B.; Jagim, A.R.; Kreider, R.B.; Rawson, E.S.; Smith-Ryan, A.E.; VanDusseldorp, T.A.; Willoughby, D.S.; et al. Common questions and misconceptions about creatine supplementation: What does the scientific evidence really show? J. Int. Soc. Sports Nutr. 2021, 18, 1–17. [Google Scholar] [CrossRef]
- Fazio, C.; Elder, C.L.; Harris, M.M. Efficacy of alternative forms of creatine supplementation on improving performance and body composition in healthy subjects: A systematic review. J. Strength Cond. Res. 2021. [Google Scholar] [CrossRef]
- Harris, R.C.; Söderlund, K.; Hultman, E. Elevation of creatine in resting and exercised muscle of normal subjects by creatine supplementation. Clin. Sci. 1992, 83, 367–374. [Google Scholar] [CrossRef]
- Balsom, P.D.; Ekblom, B.; Söerlund, K.; Sjödln, B.; Hultman, E. Creatine supplementation and dynamic high-intensity intermittent exercise. Scand. J. Med. Sci. Sports 2007, 3, 143–149. [Google Scholar] [CrossRef]
- Greenhaff, P.L.; Casey, A.; Short, A.H.; Harris, R.; Soderlund, K.; Hultman, E. Influence of Oral Creatine Supplementation of Muscle Torque during Repeated Bouts of Maximal Voluntary Exercise in Man. Clin. Sci. 1993, 84, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B. Effects of creatine supplementation on performance and training adaptations. Mol. Cell. Biochem. 2003, 244, 89–94. [Google Scholar] [CrossRef]
- Rawson, E.S.; Volek, J.S. Effects of Creatine Supplementation and Resistance Training on Muscle Strength and Weightlifting Performance. J. Strength Cond. Res. 2003, 17, 822–831. [Google Scholar] [CrossRef]
- Stone, M.H.; Sanborn, K.; Smith, L.L.; O’Bryant, H.S.; Hoke, T.; Utter, A.C.; Johnson, R.L.; Boros, R.; Hruby, J.; Pierce, K.C.; et al. Effects of In-Season (5 Weeks) Creatine and Pyruvate Supplementation on Anaerobic Performance and Body Composition in American Football Players. Int. J. Sport Nutr. 1999, 9, 146–165. [Google Scholar] [CrossRef] [PubMed]
- Buford, T.W.; Kreider, R.B.; Stout, J.R.; Greenwood, M.; Campbell, B.; Spano, M.; Ziegenfuss, T.; Lopez, H.; Landis, J.; Antonio, J. International Society of Sports Nutrition position stand: Creatine supplementation and exercise. J. Int. Soc. Sports Nutr. 2007, 4, 6. [Google Scholar] [CrossRef]
- Cooper, R.; Naclerio, F.; Allgrove, J.; Jimenez, A. Creatine supplementation with specific view to exercise/sports performance: An update. J. Int. Soc. Sports Nutr. 2012, 9, 33. [Google Scholar] [CrossRef]
- Hultman, E.; Soderlund, K.; Timmons, J.A.; Cederblad, G.; Greenhaff, P.L. Muscle creatine loading in men. J. Appl. Physiol. 1996, 81, 232–237. [Google Scholar] [CrossRef]
- Greenhaff, P.L.; Bodin, K.; Soderlund, K.; Hultman, E. Effect of oral creatine supplementation on skeletal muscle phosphocreatine resynthesis. Am. J. Physiol. Metab. 1994, 266, E725–E730. [Google Scholar] [CrossRef]
- Law, Y.L.L.; Ong, W.S.; GillianYap, T.L.; Lim, S.C.J.; Von Chia, E. Effects of Two and Five Days of Creatine Loading on Muscular Strength and Anaerobic Power in Trained Athletes. J. Strength Cond. Res. 2009, 23, 906–914. [Google Scholar] [CrossRef]
- Sale, C.; Harris, R.C.; Florance, J.; Kumps, A.; Sanvura, R.; Poortmans, J.R. Urinary creatine and methylamine excretion following 4 × 5 g·day−1or 20 × 1 g·day−1of creatine monohydrate for 5 days. J. Sports Sci. 2009, 27, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Lanhers, C.; Pereira, B.; Naughton, G.; Trousselard, M.; Lesage, F.-X.; Dutheil, F. Creatine Supplementation and Lower Limb Strength Performance: A Systematic Review and Meta-Analyses. Sports Med. 2015, 45, 1285–1294. [Google Scholar] [CrossRef]
- Kraemer, W.J.; Volek, J.S. Creatine supplementation: Its Role in Human Performance. Clin. Sports Med. 1999, 18, 651–666. [Google Scholar] [CrossRef]
- Demant, T.W.; Rhodes, E. Effects of Creatine Supplementation on Exercise Performance. Sports Med. 1999, 28, 49–60. [Google Scholar] [CrossRef]
- Volek, J.S.; Duncan, N.D.; Mazzetti, S.A.; Staron, R.S.; Putukian, M.; Gómez, A.L.; Pearson, D.R.; Fink, W.J.; Kraemer, W.J. Performance and muscle fiber adaptations to creatine supplementation and heavy resistance training. Med. Sci. Sports Exerc. 1999, 31, 1147–1156. [Google Scholar] [CrossRef]
- Kreider, R.B.; Ferreira, M.; Wilson, M.; Grindstaff, P.; Plisk, S.; Reinardy, J.; Cantler, E.; Almada, A.L. Effects of creatine supplementation on body composition, strength, and sprint performance. Med. Sci. Sports Exerc. 1998, 30, 73–82. [Google Scholar] [CrossRef]
- Del Favero, S.; Roschel, H.; Artioli, G.; Ugrinowitsch, C.; Tricoli, V.; Costa, A.; Barroso, R.; Negrelli, A.L.; Otaduy, M.C.; Leite, C.D.C.; et al. Creatine but not betaine supplementation increases muscle phosphorylcreatine content and strength performance. Amino Acids 2011, 42, 2299–2305. [Google Scholar] [CrossRef]
- Atakan, M.; Karavelioğlu, M.; Harmancı, H.; Cook, M.; Bulut, S. Short term creatine loading without weight gain improves sprint, agility and leg strength performance in female futsal players. Sci. Sports 2019, 34, 321–327. [Google Scholar] [CrossRef]
- Stevenson, S.W.; Dudley, G.A. Creatine loading, resistance exercise performance, and muscle mechanics. J. Strength Cond. Res. 2001, 15, 413–419. [Google Scholar]
- Zuniga, J.M.; Housh, T.J.; Camic, C.L.; Hendrix, C.R.; Mielke, M.; Johnson, G.O.; Housh, D.J.; Schmidt, R.J. The Effects of Creatine Monohydrate Loading on Anaerobic Performance and One-Repetition Maximum Strength. J. Strength Cond. Res. 2012, 26, 1651–1656. [Google Scholar] [CrossRef] [PubMed]
- Lanhers, C.; Pereira, B.; Naughton, G.; Trousselard, M.; Lesage, F.-X.; Dutheil, F. Creatine Supplementation and Upper Limb Strength Performance: A Systematic Review and Meta-Analysis. Sports Med. 2016, 47, 163–173. [Google Scholar] [CrossRef]
- Kelly, V.G.; Jenkins, D.G. Effect of oral creatine supplementation on near-maximal strength and repeated sets of high-intensity bench press exercise. J. Strength Cond. Res. 1998, 12, 109–115. [Google Scholar]
- Antonio, J.; Ciccone, V. The effects of pre versus post workout supplementation of creatine monohydrate on body composition and strength. J. Int. Soc. Sports Nutr. 2013, 10, 36. [Google Scholar] [CrossRef]
- Peeters, B.M.; Lantz, C.D.; Mayhew, J.L. Effect of oral creatine monohydrate and creatine phosphate supplementation on maximal strength indices, body composition and blood pressure. J. Strength Cond. Res. 1999, 13, 3–9. [Google Scholar]
- Syrotuik, D.G.; Bell, G.J.; Burnham, R.; Sim, L.L.; Calvert, R.A.; Maclean, I.M. Absolute and relative strength performance following creatine monohydrate supplementation combined with periodized resistance training. J. Strength Cond. Res. 2000, 14, 182–190. [Google Scholar]
- Kreider, R.B.; Klesges, R.; Harmon, K.; Grindstaff, P.; Ramsey, L.; Bullen, D.; Wood, L.; Li, Y.; Almada, A. Effects of Ingesting Supplements Designed to Promote Lean Tissue Accretion on Body Composition during Resistance Training. Int. J. Sport Nutr. 1996, 6, 234–246. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Klesges, R.C.; Lotz, D.; Davis, M.; Cantler, E.; Grindstaff, P.; Ramsey, L.; Bullen, D.; Wood, L.; Almada, A.L. Effects of nutritional supplementation during off-season college football training on body composition and strength. J. Exerc. Physiol. 1999, 2, 24–39. [Google Scholar]
- Noonan, D.; Berg, K.; Latin, R.W.; Wagner, J.C.; Reimers, K. Effects of Varying Dosages of Oral Creatine Relative to Fat Free Body Mass on Strength and Body Composition. J. Strength Cond. Res. 1998, 12, 104. [Google Scholar] [CrossRef]
- Bemben, M.G.; Bemben, D.A.; Loftiss, D.D.; Knehans, A.W. Creatine supplementation during resistance training in college football athletes. Med. Sci. Sports Exerc. 2001, 33, 1667–1673. [Google Scholar] [CrossRef]
- Pearson, D.R.; Hambx, W.R.; Harris, T. Long-Term Effects of Creatine Monohdrate on Strength and Power. J. Strength Cond. Res. 1999, 13, 187–192. [Google Scholar]
- Brenner, M.; Rankin, J.W.; Sebolt, D. The effect of creatine supplementation during resistance training in women. J. Strength Cond. Res. 2000, 14, 207–213. [Google Scholar]
- Larson-Meyer, D.E.; Hunter, G.R.; Trowbridge, C.A.; Turk, J.C.; Ernest, J.M.; Torman, S.L.; Harbin, P.A. The effect of creatine supplementation on muscle strength and composition during off-season training in female soccery players. J. Strength Cond. Res. 2000, 14, 434–442. [Google Scholar]
- Vandenberghe, K.; Goris, M.; Van Hecke, P.; Van Leemputte, M.; Vangerven, L.; Hespel, P. Long-term creatine intake is beneficial to muscle performance during resistance training. J. Appl. Physiol. 1997, 83, 2055–2063. [Google Scholar] [CrossRef]
- Candow, D.G.; Chilibeck, P.D.; Burke, D.G.; Mueller, K.D.; Lewis, J.D. Effect of Different Frequencies of Creatine Supplementation on Muscle Size and Strength in Young Adults. J. Strength Cond. Res. 2011, 25, 1831–1838. [Google Scholar] [CrossRef]
- Herda, T.J.; Beck, T.W.; Smith-Ryan, A.; Smith, A.E.; Walter, A.A.; Hartman, M.J.; Stout, J.R.; Cramer, J.T. Effects of Creatine Monohydrate and Polyethylene Glycosylated Creatine Supplementation on Muscular Strength, Endurance, and Power Output. J. Strength Cond. Res. 2009, 23, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Birch, R.; Noble, D.; Greenhaff, P.L. The influence of dietary creatine supplementation on performance during repeated bouts of maximal isokinetic cycling in man. Graefe’s Arch. Clin. Exp. Ophthalmol. 1994, 69, 268–270. [Google Scholar] [CrossRef]
- Casey, A.; Constantin-Teodosiu, D.; Howell, S.; Hultman, E.; Greenhaff, P.L. Creatine ingestion favorably affects performance and muscle metabolism during maximal exercise in humans. Am. J. Physiol. Metab. 1996, 271, E31–E37. [Google Scholar] [CrossRef] [PubMed]
- Green, A.L.; Hultman, E.; Macdonald, I.A.; Sewell, D.A.; Greenhaff, P.L. Carbohydrate ingestion augments skeletal muscle creatine accumulation during creatine supplementation in humans. Am. J. Physiol. Content 1996, 271, 821–826. [Google Scholar] [CrossRef]
- Stout, J.R.; Eckerson, J.M.; Housh, T.J.; Ebersolevkt, K.T. The effects of creatine supplementation on anaerobic working capacity. J. Strength Cond. Res. 1999, 13, 135–138. [Google Scholar]
- Van Loon, L.J.C.; Oosterlaar, A.M.; Hartgens, F.; Hesselink, M.K.C.; Snow, R.J.; Wagenmakers, A.J.M. Effects of creatine loading and prolonged creatine supplementation on body composition, fuel selection, sprint and endurance performance in humans. Clin. Sci. 2003, 104, 153–162. [Google Scholar] [CrossRef]
- Barber, J.J.; McDermott, A.Y.; McGaughey, K.J.; Olmstead, J.D.; Hagobian, T.A. Effects of Combined Creatine and Sodium Bicarbonate Supplementation on Repeated Sprint Performance in Trained Men. J. Strength Cond. Res. 2013, 27, 252–258. [Google Scholar] [CrossRef]
- Fukuda, D.H.; Smith, A.E.; Kendall, K.L.; Dwyer, T.R.; Kerksick, C.; Beck, T.W.; Cramer, J.T.; Stout, J.R. The Effects of Creatine Loading and Gender on Anaerobic Running Capacity. J. Strength Cond. Res. 2010, 24, 1826–1833. [Google Scholar] [CrossRef]
- Branch, J.D. Effect of Creatine Supplementation on Body Composition and Performance: A Meta-analysis. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 198–226. [Google Scholar] [CrossRef] [PubMed]
- Cox, G.; Mujika, I.; Tumilty, D.; Burke, L. Acute Creatine Supplementation and Performance during a Field Test Simulating Match Play in Elite Female Soccer Players. Int. J. Sport Nutr. Exerc. Metab. 2002, 12, 33–46. [Google Scholar] [CrossRef]
- Ramírez-Campillo, R.; González-Jurado, J.A.; Martínez, C.; Nakamura, F.Y.; Peñailillo, L.; Meylan, C.M.; Caniuqueo, A.; Cañas-Jamet, R.; Moran, J.; Alonso-Martínez, A.M.; et al. Effects of plyometric training and creatine supplementation on maximal-intensity exercise and endurance in female soccer players. J. Sci. Med. Sport 2016, 19, 682–687. [Google Scholar] [CrossRef]
- Ostojic, S.M. Creatine Supplementation in Young Soccer Players. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Eijnde, B.O.; Vergauwen, L.; Hespel, P. Creatine Loading does not Impact on Stroke Performance in Tennis. Int. J. Sports Med. 2001, 22, 76–80. [Google Scholar] [CrossRef]
- Pluim, B.M. The effects of creatine supplementation on selected factors of tennis specific training *Commentary* Commentary. Br. J. Sports Med. 2006, 40, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Koçak, S.; Karli, U. Effects of high dose oral creatine supplementation on anaerobic capacity of elite wrestlers. J. Sports Med. Phys. Fit. 2003, 43, 488–492. [Google Scholar]
- Aedma, M.; Timpmann, S.; Lätt, E.; Ööpik, V. Short-term creatine supplementation has no impact on upper-body anaerobic power in trained wrestlers. J. Int. Soc. Sports Nutr. 2015, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- De Oca, R.M.-M.; Farfán-González, F.; Camarillo-Romero, S.; Tlatempa-Sotelo, P.; Francisco-Argüelles, C.; Kormanowski, A.; González-Gallego, J.; Alvear-Ordenes, I. Effects of creatine supplementation in taekwondo practitioners. Nutr. Hosp. 2013, 28, 391–399. [Google Scholar]
- Izquierdo, M.; Ibañez, J.; González-Badillo, J.J.; Gorostiaga, E.M. Effects of creatine supplementation on muscle power, endurance, and sprint performance. Med. Sci. Sports Exerc. 2002, 34, 332–343. [Google Scholar] [CrossRef]
- Stout, J.; Eckerson, J.; Noonan, D.; Moore, G.; Cullen, D. Effects of 8 weeks of creatine supplementation on exercise performance and fat-free weight in football players during training. Nutr. Res. 1999, 19, 217–225. [Google Scholar] [CrossRef]
- Jones, A.M.; Atter, T.; Georg, K.P. Oral creatine supplementation improves multiple sprint performance in elite ice-hockey players. J. Sports Med. Phys. Fit. 1999, 39, 189–196. [Google Scholar] [CrossRef]
- Mujika, I.; Padilla, S.; Ibañez, J.; Izquierdo, M.; Gorostiaga, E. Creatine supplementation and sprint performance in soccer players. Med. Sci. Sports Exerc. 2000, 32, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, A.S.; Cooke, C.B.; King, R.F.G.J.; Hood, C.; Denison, T.; Wainwright, B.G.; Havenetidis, K. The effect of longer-term creatine supplementation on elite swimming performance after an acute creatine loading. J. Sports Sci. 1999, 17, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Skare, O.-C.; Skadberg, O.; Wisnes, A.R. Creatine supplementation improves sprint performance in male sprinters. Scand. J. Med. Sci. Sports 2001, 11, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Delecluse, C.; Diels, R.; Goris, M. Effect of Creatine Supplementation on Intermittent Sprint Running Performance in Highly Trained Athletes. J. Strength Cond. Res. 2003, 17, 446. [Google Scholar] [CrossRef] [PubMed]
- Peyrebrune, M.C.; Nevill, M.E.; Donaldson, F.J.; Cosford, D.J. The effects of oral creatine supplementation on performance in single and repeated sprint swimming. J. Sports Sci. 1998, 16, 271–279. [Google Scholar] [CrossRef]
- Williams, J.; Abt, G.; Kilding, A.E. Effects of Creatine Monohydrate Supplementation on Simulated Soccer Performance. Int. J. Sports Physiol. Perform. 2014, 9, 503–510. [Google Scholar] [CrossRef]
- Glaister, M.; Lockey, R.A.; Abraham, C.S.; Staerck, A.; Goodwin, J.E.; McInnes, G. Creatine Supplementation and Multiple Sprint Running Performance. J. Strength Cond. Res. 2006, 20, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Lamontagne-Lacasse, M.; Nadon, R.; Goulet, E.D.B. Effect of Creatine Supplementation Jumping Performance in Elite Volleyball Players. Int. J. Sports Physiol. Perform. 2011, 6, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Mielgo-Ayuso, J.; Calleja-Gonzalez, J.; Marqués-Jiménez, D.; Caballero-García, A.; Córdova, A.; Fernández-Lázaro, D. Effects of Creatine Supplementation on Athletic Performance in Soccer Players: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 757. [Google Scholar] [CrossRef]
- Haff, G.; Kirksey, K.; Stone, M.; Warren, B.; Johnson, R.; Stone, M.; O’Bryant, H.; Proulx, C. The effects of 6 weeks of creatine monohydrate supplementation on dynamic rate of force development. J. Strength Cond. Res. 2000, 14, 426–433. [Google Scholar] [CrossRef]
- Kirksey, B.; Stone, M.; Warren, B.; Johnson, R.; Stone, M.; Haff, G.; Williams, F.; Proulx, C. The effects of 6 weeks of creatine monohydrate supplementation on performance measures and body composition in colleagiate track and field athletes. J. Strength Cond. Res. 1999, 13, 148–156. [Google Scholar]
- Lehmkuhl, M.; Malone, M.; Justice, B.; Trone, G.; Pistilli, E.; Vinci, D.; Haff, E.E.; Kilgore, J.L.; Haff, G.G. The Effects of 8 Weeks of Creatine Monohydrate and Glutamine Supplementation on Body Composition and Performance Measures. J. Strength Cond. Res. 2003, 17, 425. [Google Scholar] [CrossRef] [PubMed]
- Grindstaff, P.D.; Kreider, R.; Bishop, R.; Wilson, M.; Wood, L.; Alexander, C.; Almada, A. Effects of creatine supplementation on repetitive sprint performance and body composition in competitive swimmers. Int. J. Sport Nutr. 1997, 7, 330–346. [Google Scholar] [CrossRef]
- Papadimitriou, K. Effects of legal ergogenic supplements on swimmer’s performance: A bibliographic approach. Inq. Sport Phys. Educ. 2018, 16, 66–77. [Google Scholar]
- Rothschild, J.A.; Bishop, D.J. Effects of Dietary Supplements on Adaptations to Endurance Training. Sports Med. 2020, 50, 25–53. [Google Scholar] [CrossRef]
- Hawley, J.A.; Hargreaves, M.; Joyner, M.J.; Zierath, J.R. Integrative Biology of Exercise. Cell 2014, 159, 738–749. [Google Scholar] [CrossRef]
- Graef, J.L.; Smith, A.E.; Kendall, K.L.; Fukuda, D.H.; Moon, J.R.; Beck, T.W.; Cramer, J.T.; Stout, J.R. The effects of four weeks of creatine supplementation and high-intensity interval training on cardiorespiratory fitness: A randomized controlled trial. J. Int. Soc. Sports Nutr. 2009, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Barnett, C.; Hinds, M.; Jenkins, D.G. Effects of oral creatine supplementation on multiple sprint cycle performance. Aust. J. Sci. Med. Sport 1996, 28, 35–39. [Google Scholar] [PubMed]
- Cañete, S.; Juan, A.F.S.; Pérez, M.; Gómez-Gallego, F.; López-Mojares, L.M.; Earnest, C.; Fleck, S.J.; Lucia, A. Does Creatine Supplementation Improve Functional Capacity in Elderly Women? J. Strength Cond. Res. 2006, 20, 22–28. [Google Scholar] [CrossRef]
- Eijnde, B.O.; Van Leemputte, M.; Goris, M.; Labarque, V.; Taes, Y.; Verbessem, P.; Vanhees, L.; Ramaekers, M.; Eynde, B.V.; Van Schuylenbergh, R.; et al. Effects of creatine supplementation and exercise training on fitness in men 55–75 yr old. J. Appl. Physiol. 2003, 95, 818–828. [Google Scholar] [CrossRef] [PubMed]
- Syrotuik, D.G.; Game, A.B.; Gillies, E.M.; Bell, G.J. Effects of Creatine Monohydrate Supplementation during Combined Strength and High Intensity Rowing Training on Performance. Can. J. Appl. Physiol. 2001, 26, 527–542. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.C.; Sletten, N.; Durrer, C.; Myette-Côté, É.; Candow, D.; Little, J.P. Creatine Monohydrate Supplementation Does Not Augment Fitness, Performance, or Body Composition Adaptations in Response to Four Weeks of High-Intensity Interval Training in Young Females. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Engelhardt, M.; Neumann, G.; Berbalk, A.; Reuter, I. Creatine supplementation in endurance sports. Med. Sci. Sports Exerc. 1998, 30, 1123–1129. [Google Scholar] [CrossRef]
- Miura, A.; Kino, F.; Kajitani, S.; Sato, H.; Sato, H.; Fukuba, Y. The Effect of Oral Creatine Supplementation on the Curvature Constant Parameter of the Power-Duration Curve for Cycle Ergometry in Humans. Jpn. J. Physiol. 1999, 49, 169–174. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stroud, M.A.; Holliman, D.; Bell, D.; Green, A.L.; Macdonald, I.A.; Greenhaff, P.L. Effect of Oral Creatine Supplementation on Respiratory Gas Exchange and Blood Lactate Accumulation during Steady-State Incremental Treadmill Exercise and Recovery in Man. Clin. Sci. 1994, 87, 707–710. [Google Scholar] [CrossRef]
- Bellinger, B.; Bold, A.; Wilson, G.; Myburgh, K.; Noakes, T. Oral creatine supplementation decreases plasma markers of adenine nucleotide degradation during a 1-h cycle test. Acta Physiol. Scand. 2000, 170, 217–224. [Google Scholar] [CrossRef]
- Nemezio, K.M.D.A.; Bertuzzi, R.; Correia-Oliveira, C.R.; Gualano, B.; Bishop, D.; Lima-Silva, A. Effect of Creatine Loading on Oxygen Uptake during a 1-km Cycling Time Trial. Med. Sci. Sports Exerc. 2015, 47, 2660–2668. [Google Scholar] [CrossRef]
- Jones, A.M.; Carter, H.; Pringle, J.S.M.; Campbell, I.T. Effect of creatine supplementation on oxygen uptake kinetics during submaximal cycle exercise. J. Appl. Physiol. 2002, 92, 2571–2577. [Google Scholar] [CrossRef]
- McNaughton, L.R.; Dalton, B.; Tarr, J. The effects of creatine supplementation on high-intensity exercise performance in elite performers. Graefe’s Arch. Clin. Exp. Ophthalmol. 1998, 78, 236–240. [Google Scholar] [CrossRef]
- Rossiter, H.B.; Cannell, E.R.; Jakeman, P.M. The effect of oral creatine supplementation on the 1000-m performance of competitive rowers. J. Sports Sci. 1996, 14, 175–179. [Google Scholar] [CrossRef]
- Maganaris, C.; Maughan, R. Creatine supplementation enhances maximum voluntary isometric force and endurance capacity in resistance trained men. Acta Physiol. Scand. 1998, 163, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Prevost, M.C.; Nelson, A.G.; Morris, G.S. Creatine Supplementation Enhances Intermittent Work Performance. Res. Q. Exerc. Sport 1997, 68, 233–240. [Google Scholar] [CrossRef]
- Jakobi, J.M.; Rice, C.L.; Curtin, S.V.; Marsh, G.D. Contractile properties, fatigue and recovery are not influenced by short-term creatine supplementation in human muscle. Exp. Physiol. 2000, 85, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Vandebuerie, F.; Eynde, B.V.; Vandenberghe, K.; Hespel, P. Effect of Creatine Loading on Endurance Capacity and Sprint Power in Cyclists. Int. J. Sports Med. 1998, 19, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Chwalbiñska-Moneta, J. Effect of creatine supplementation on aerobic performance and anaerobic capacity in elite rowers in the course of endurance training. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 173–183. [Google Scholar] [CrossRef]
- Fernández-Landa, J.; Fernández-Lázaro, D.; Calleja-González, J.; Caballero-García, A.; Martínez, A.C.; León-Guereño, P.; Mielgo-Ayuso, J. Effect of Ten Weeks of Creatine Monohydrate Plus HMB Supplementation on Athletic Performance Tests in Elite Male Endurance Athletes. Nutrients 2020, 12, 193. [Google Scholar] [CrossRef] [PubMed]
- Kendall, K.L.; Smith, A.E.; Graef, J.L.; Fukuda, D.H.; Moon, J.R.; Beck, T.W.; Cramer, J.T.; Stout, J.R. Effects of Four Weeks of High-Intensity Interval Training and Creatine Supplementation on Critical Power and Anaerobic Working Capacity in College-Aged Men. J. Strength Cond. Res. 2009, 23, 1663–1669. [Google Scholar] [CrossRef]
- Mujika, I.; Padilla, S. Creatine Supplementation as an Ergogenic Aid for Sports Performance in Highly Trained Athletes: A Critical Review. Int. J. Sports Med. 1997, 18, 491–496. [Google Scholar] [CrossRef]
- Steenge, G.R.; Simpson, E.J.; Greenhaff, P.L. Protein- and carbohydrate-induced augmentation of whole body creatine retention in humans. J. Appl. Physiol. 2000, 89, 1165–1171. [Google Scholar] [CrossRef]
- Cooke, M.B.; Rybalka, E.; Williams, A.D.; Cribb, P.J.; Hayes, A. Creatine supplementation enhances muscle force recovery after eccentrically-induced muscle damage in healthy individuals. J. Int. Soc. Sports Nutr. 2009, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.B.; Rybalka, E.; Stathis, C.G.; Hayes, A. Myoprotective Potential of Creatine Is Greater than Whey Protein after Chemically-Induced Damage in Rat Skeletal Muscle. Nutrients 2018, 10, 553. [Google Scholar] [CrossRef]
- Santos, R.; Bassit, R.; Caperuto, E.; Rosa, L.C. The effect of creatine supplementation upon inflammatory and muscle soreness markers after a 30km race. Life Sci. 2004, 75, 1917–1924. [Google Scholar] [CrossRef]
- Volek, J.S.; Mazzetti, S.A.; Farquhar, W.B.; Barnes, B.R.; Gómez, A.L.; Kraemer, W.J. Physiological responses to short-term exercise in the heat after creatine loading. Med. Sci. Sports Exerc. 2001, 33, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.A.; Powers, M.E. Creatine supplementation does not impair the thermoregulatory response during a bout of exercise in the heat. J. Sports Med. Phys. Fit. 2006, 46, 555–563. [Google Scholar]
- Lopez, R.M.; Casa, D.J.; McDermott, B.P.; Ganio, M.S.; Armstrong, L.E.; Maresh, C.M. Does Creatine Supplementation Hinder Exercise Heat Tolerance or Hydration Status? A Systematic Review with Meta-Analyses. J. Athl. Train. 2009, 44, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Rawson, E.S.; Gunn, B.; Clarkson, P.M. The effects of creatine supplementation on exercise-induced muscle damage. J. Strength Cond. Res. 2001, 15, 178–184. [Google Scholar] [PubMed]
- Yquel, R.; Arsac, L.; Thiaudière, E.; Canioni, P.; Manier, G. Effect of creatine supplementation on phosphocreatine resynthesis, inorganic phosphate accumulation and pH during intermittent maximal exercise. J. Sports Sci. 2002, 20, 427–437. [Google Scholar] [CrossRef]
- Fitts, R. Cellular mechanisms of muscle fatigue. Physiol. Rev. 1994, 74, 49–94. [Google Scholar] [CrossRef]
- Okudan, N.; Belviranli, M.; Pepe, H.; Gökbel, H. The effects of beta alanine plus creatine administration on performance during repeated bouts of supramaximal exercise in sedentary men. J. Sports Med. Phys. Fit. 2014, 55, 1322–1328. [Google Scholar]
- Okudan, N.; Gokbel, H. The effects of creatine supplementation on performance during the repeated bouts of supramaximal exercise. J. Sports Med. Phys. Fit. 2005, 45, 507–511. [Google Scholar]
- Gill, N.D.; Hall, R.D.; Blazevich, A.J. Creatine Serum Is Not as Effective as Creatine Powder for Improving Cycle Sprint Performance in Competitive Male Team-Sport Athletes. J. Strength Cond. Res. 2004, 18, 272–275. [Google Scholar] [CrossRef]
- Griffen, C.; Rogerson, D.; Ranchordas, M.; Ruddock, A. Effects of Creatine and Sodium Bicarbonate Coingestion on Multiple Indices of Mechanical Power Output during Repeated Wingate Tests in Trained Men. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 298–306. [Google Scholar] [CrossRef]
- Crisafulli, D.L.; Buddhadev, H.H.; Brilla, L.R.; Chalmers, G.R.; Suprak, D.N.; Juan, J.G.S. Creatine-electrolyte supplementation improves repeated sprint cycling performance: A double blind randomized control study. J. Int. Soc. Sports Nutr. 2018, 15, 1–11. [Google Scholar] [CrossRef]
- Dawson, B.; Cutler, M.; Moody, A.; Lawrence, S.; Goodman, C.; Randall, N. Effects of oral creatine loading on single and repeated maximal short sprints. Aust. J. Sci. Med. Sport 1995, 27, 56–61. [Google Scholar]
- Cottrell, G.T.; Coast, J.R.; Herb, R.A. Effect of recovery interval on multiple-bout sprint cycling performance after acute creatine supplementation. J. Strength Cond. Res. 2002, 16, 109–116. [Google Scholar] [PubMed]
- Aaserud, R.; Gramvik, P.; Olsen, S.R.; Jensen, J. Creatine supplementation delays onset of fatigue during repeated bouts of sprint running. Scand. J. Med. Sci. Sports 1998, 8, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Deminice, R.; Rosa, F.T.; Franco, G.S.; Jordao, A.A.; de Freitas, E.C. Effects of creatine supplementation on oxidative stress and inflammatory markers after repeated-sprint exercise in humans. Nutrients 2013, 29, 1127–1132. [Google Scholar] [CrossRef]
- Green, J.; McLester, J.; Smith, J.; Mansfield, E. The effects of creatine supplementation on repeated upper- and lower-body Wingate performance. J. Strength Cond. Res. 2001, 15, 36–41. [Google Scholar]
- Ahmun, R.P.; Tong, R.J.; Grimshaw, P.N. The Effects of Acute Creatine Supplementation on Multiple Sprint Cycling and Running Performance in Rugby Players. J. Strength Cond. Res. 2005, 19, 92–97. [Google Scholar] [CrossRef]
- Deutekom, M.; Beltman, J.G.M.; De Ruiter, C.J.; De Koning, J.J.; De Haan, A. No acute effects of short-term creatine supplementation on muscle properties and sprint performance. Graefe’s Arch. Clin. Exp. Ophthalmol. 2000, 82, 223–229. [Google Scholar] [CrossRef]
- Rosene, J.; Matthews, T.; Ryan, C.; Belmore, K.; Bergsten, A.; Blaisdell, J.; Gaylord, J.; Love, R.; Marrone, M.; Ward, K.; et al. Short and longer-term effects of creatine supplementation on exercise induced muscle damage. J. Sports Sci. Med. 2009, 8, 89–96. [Google Scholar] [PubMed]
- Bassit, R.A.; Pinheiro, C.H.D.J.; Vitzel, K.F.; Sproesser, A.J.; Silveira, L.R.; Curi, R. Effect of short-term creatine supplementation on markers of skeletal muscle damage after strenuous contractile activity. Graefe’s Arch. Clin. Exp. Ophthalmol. 2009, 108, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Veggi, K.F.T.; Machado, M.; Koch, A.J.; Santana, S.C.; Oliveira, S.S.; Stec, M.J. Oral Creatine Supplementation Augments the Repeated Bout Effect. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 378–387. [Google Scholar] [CrossRef]
- Wang, C.-C.; Fang, C.-C.; Lee, Y.-H.; Yang, M.-T.; Chan, K.-H. Effects of 4-Week Creatine Supplementation Combined with Complex Training on Muscle Damage and Sport Performance. Nutrients 2018, 10, 1640. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.; Kim, S.; Yoon, D.; Kim, J.; Sung, D.J. Role of creatine supplementation in exercise-induced muscle damage: A mini review. J. Exerc. Rehabil. 2015, 11, 244–250. [Google Scholar] [CrossRef]
- Boychuk, K.E.; Lanovaz, J.L.; Krentz, J.R.; Lishchynsky, J.T.; Candow, D.G.; Farthing, J.P. Creatine supplementation does not alter neuromuscular recovery after eccentric exercise. Muscle Nerve 2016, 54, 487–495. [Google Scholar] [CrossRef]
- McKinnon, N.B.; Graham, M.T.; Tiidus, P.M. Effect of Creatine Supplementation on Muscle Damage and Repair following Eccentrically-Induced Damage to the Elbow Flexor Muscles. J. Sports Sci. Med. 2012, 11, 653–659. [Google Scholar]
- Rawson, E.S.; Conti, M.P.; Miles, M.P. Creatine Supplementation Does not Reduce Muscle Damage or Enhance Recovery from Resistance Exercise. J. Strength Cond. Res. 2007, 21, 1208–1213. [Google Scholar] [CrossRef]
- Warren, G.L.; Fennessy, J.M.; Millard-Stafford, M.L. Strength loss after eccentric contractions is unaffected by creatine supplementation. J. Appl. Physiol. 2000, 89, 557–562. [Google Scholar] [CrossRef]
- Lawler, J.M.; Barnes, W.S.; Wu, G.; Song, W.; Demaree, S. Direct Antioxidant Properties of Creatine. Biochem. Biophys. Res. Commun. 2002, 290, 47–52. [Google Scholar] [CrossRef]
- Sestili, P.; Martinelli, C.; Bravi, G.; Piccoli, G.; Curci, R.; Battistelli, M.; Falcieri, E.; Agostini, D.; Gioacchini, A.M.; Stocchi, V. Creatine supplementation affords cytoprotection in oxidatively injured cultured mammalian cells via direct antioxidant activity. Free Radic. Biol. Med. 2006, 40, 837–849. [Google Scholar] [CrossRef] [PubMed]
- Magne, H.; Savary-Auzeloux, I.; Rémond, D.; Dardevet, D. Nutritional strategies to counteract muscle atrophy caused by disuse and to improve recovery. Nutr. Res. Rev. 2013, 26, 149–165. [Google Scholar] [CrossRef]
- Greenhaff, P.L. The Molecular Physiology of Human Limb Immobilization and Rehabilitation. Exerc. Sport Sci. Rev. 2006, 34, 159–163. [Google Scholar] [CrossRef]
- Vazeille, E.; Codran, A.; Claustre, A.; Averous, J.; Listrat, A.; Béchet, D.; Taillandier, D.; Dardevet, D.; Attaix, D.; Combaret, L. The ubiquitin-proteasome and the mitochondria-associated apoptotic pathways are sequentially downregulated during recovery after immobilization-induced muscle atrophy. Am. J. Physiol. Metab. 2008, 295, E1181–E1190. [Google Scholar] [CrossRef]
- MacDougall, J.D.; Ward, G.R.; Sale, D.G.; Sutton, J.R. Biochemical adaptation of human skeletal muscle to heavy resistance training and immobilization. J. Appl. Physiol. 1977, 43, 700–703. [Google Scholar] [CrossRef]
- Luo, D.; Edwards, S.; Smeuninx, B.; McKendry, J.; Nishimura, Y.; Perkins, M.; Philp, A.; Joanisse, S.; Breen, L. Immobilization leads to alterations in intracellular phosphagen and creatine transporter content in human skeletal muscle. Am. J. Physiol. Physiol. 2020, 319, C34–C44. [Google Scholar] [CrossRef]
- Marshall, R.N.; Smeuninx, B.; Morgan, P.T.; Breen, L. Nutritional Strategies to Offset Disuse-Induced Skeletal Muscle Atrophy and Anabolic Resistance in Older Adults: From Whole-Foods to Isolated Ingredients. Nutrients 2020, 12, 1533. [Google Scholar] [CrossRef]
- Wall, B.T.; Morton, J.P.; Van Loon, L.J.C. Strategies to maintain skeletal muscle mass in the injured athlete: Nutritional considerations and exercise mimetics. Eur. J. Sport Sci. 2015, 15, 53–62. [Google Scholar] [CrossRef]
- Hespel, P.; Eijnde, B.O.; Van Leemputte, M.; Ursø, B.; Greenhaff, P.L.; Labarque, V.; Dymarkowski, S.; Van Hecke, P.; Richter, E.A. Oral creatine supplementation facilitates the rehabilitation of disuse atrophy and alters the expression of muscle myogenic factors in humans. J. Physiol. 2001, 536, 625–633. [Google Scholar] [CrossRef]
- Deldicque, L.; Louis, M.; Theisen, D.; Nielens, H.; Dehoux, M.; Thissen, J.-P.; Rennie, M.J.; Francaux, M. Increased IGF mRNA in Human Skeletal Muscle after Creatine Supplementation. Med. Sci. Sports Exerc. 2005, 37, 731–736. [Google Scholar] [CrossRef]
- Bodine, S.C. mTOR Signaling and the Molecular Adaptation to Resistance Exercise. Med. Sci. Sports Exerc. 2006, 38, 1950–1957. [Google Scholar] [CrossRef]
- Parise, G.; Mihic, S.; MacLennan, D.; Yarasheski, K.E.; Tarnopolsky, M.A. Effects of acute creatine monohydrate supplementation on leucine kinetics and mixed-muscle protein synthesis. J. Appl. Physiol. 2001, 91, 1041–1047. [Google Scholar] [CrossRef]
- Johnston, A.P.W.; Burke, D.G.; MacNeil, L.G.; Candow, D.G. Effect of Creatine Supplementation during Cast-Induced Immobilization on the Preservation of Muscle Mass, Strength, and Endurance. J. Strength Cond. Res. 2009, 23, 116–120. [Google Scholar] [CrossRef]
- Backx, E.M.P.; Hangelbroek, R.; Snijders, T.; Verscheijden, M.-L.; Verdijk, L.B.; De Groot, L.C.P.G.M.; Van Loon, L.J.C. Creatine Loading Does not Preserve Muscle Mass or Strength during Leg Immobilization in Healthy, Young Males: A Randomized Controlled Trial. Sports Med. 2017, 47, 1661–1671. [Google Scholar] [CrossRef]
- Roy, B.D.; De Beer, J.; Harvey, D.; Tarnopolsky, M.A. Creatine Monohydrate Supplementation Does not Improve Functional Recovery after total Knee Arthroplasty. Arch. Phys. Med. Rehabil. 2005, 86, 1293–1298. [Google Scholar] [CrossRef]
- Tyler, T.F.; Nicholas, S.J.; Hershman, E.B.; Glace, B.W.; Mullaney, M.J.; McHugh, M.P. The Effect of Creatine Supplementation on Strength Recovery after Anterior Cruciate Ligament (ACL) Reconstruction. Am. J. Sports Med. 2004, 32, 383–388. [Google Scholar] [CrossRef]
- Hamilton, K.L.; Meyers, M.C.; Skelly, W.A.; Marley, R.J. Oral creatine supplementation and upper extremity anaerobic response in females. Int. J. Sport Nutr. Exerc. Metab. 2000, 10, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Tarnopolsky, M.A.; MacLennan, D.P. Creatine Monohydrate Supplementation Enhances High-Intensity Exercise Performance in Males and Females. Int. J. Sport Nutr. Exerc. Metab. 2000, 10, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Kambis, K.W.; Pizzedaz, S.K. Short-term creatine supplementation improves maximum quadriceps contraction in women. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 87–96. [Google Scholar] [CrossRef]
- Candow, D.; Forbes, S.; Kirk, B.; Duque, G. Current Evidence and Possible Future Applications of Creatine Supplementation for Older Adults. Nutrients 2021, 13, 745. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.C.; Candow, D.G.; Ferreira, L.H.B.; Souza-Junior, T.P. Effects of Creatine Supplementation on Properties of Muscle, Bone, and Brain Function in Older Adults: A Narrative Review. J. Diet. Suppl. 2021, 10, 1–18. [Google Scholar]
- Candow, D.G.; Chilibeck, P.D.; Gordon, J.; Vogt, E.; Landeryou, T.; Kaviani, M.; Paus-Jensen, L. Effect of 12 months of creatine supplementation and whole-body resistance training on measures of bone, muscle and strength in older males. Nutr. Health 2021, 27, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.C.; Chilibeck, P.D.; Candow, D.G. Creatine Supplementation during Resistance Training Does not Lead to Greater Bone Mineral Density in Older Humans: A Brief Meta-Analysis. Front. Nutr. 2018, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Chilibeck, P.D.; Kaviani, M.; Candow, D.G.; Zello, G.A. Effect of creatine supplementation during resistance training on lean tissue mass and muscular strength in older adults: A meta-analysis. Open Access J. Sports Med. 2017, 8, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Vogt, E.; Johannsmeyer, S.; Forbes, S.C.; Farthing, J.P. Strategic creatine supplementation and resistance training in healthy older adults. Appl. Physiol. Nutr. Metab. 2015, 40, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Zello, G.A.; Ling, B.; Farthing, J.P.; Chilibeck, P.D.; McLeod, K.; Harris, J.; Johnson, S. Comparison of Creatine Supplementation before Versus after Supervised Resistance Training in Healthy Older Adults. Res. Sports Med. 2014, 22, 61–74. [Google Scholar] [CrossRef]
- Candow, D.G.; Little, J.P.; Chilibeck, P.D.; Abeysekara, S.; Zello, G.A.; Kazachkov, M.; Cornish, S.M.; Yu, P.H. Low-Dose Creatine Combined with Protein during Resistance Training in Older Men. Med. Sci. Sports Exerc. 2008, 40, 1645–1652. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Chilibeck, P.D.; Chad, K.E.; Chrusch, M.J.; Davison, K.S.; Burke, D.G. Effect of Ceasing Creatine Supplementation while Maintaining Resistance Training in Older Men. J. Aging Phys. Act. 2004, 12, 219–231. [Google Scholar] [CrossRef]
- Jäger, R.; Purpura, M.; Shao, A.; Inoue, T.; Kreider, R.B. Analysis of the efficacy, safety, and regulatory status of novel forms of creatine. Amino Acids 2011, 40, 1369–1383. [Google Scholar] [CrossRef] [PubMed]
- Kerksick, C.; Wilborn, C.D.; Campbell, W.I.; Harvey, T.M.; Marcello, B.M.; Roberts, M.D.; Parker, A.G.; Byars, A.G.; Greenwood, L.D.; Almada, A.L.; et al. The Effects of Creatine Monohydrate Supplementation With and Without D-Pinitol on Resistance Training Adaptations. J. Strength Cond. Res. 2009, 23, 2673–2682. [Google Scholar] [CrossRef] [PubMed]
- Spillane, M.; Schoch, R.; Cooke, M.B.; Harvey, T.; Greenwood, M.; Kreider, R.B.; Willoughby, D.S. The effects of creatine ethyl ester supplementation combined with heavy resistance training on body composition, muscle performance, and serum and muscle creatine levels. J. Int. Soc. Sports Nutr. 2009, 6, 6. [Google Scholar] [CrossRef]
- Greenwood, M. Differences in creatine retention among three nutritional formulations of oral creatine supplements. J. Exerc. Physiol. Online 2003, 6, 37–43. [Google Scholar]
- Jagim, A.R.; Oliver, J.M.; Sanchez, A.; Galvan, E.; Fluckey, J.; Riechman, S.; Greenwood, M.; Kelly, K.; Meininger, C.; Rasmussen, C.; et al. A buffered form of creatine does not promote greater changes in muscle creatine content, body composition, or training adaptations than creatine monohydrate. J. Int. Soc. Sports Nutr. 2012, 9, 43. [Google Scholar] [CrossRef]
- Stout, J.R.; Cramer, J.T.; Mielke, M.; O’Kroy, J.; Torok, D.J.; Zoeller, R.F. Effects of Twenty-Eight Days of Beta-Alanine and Creatine Monohydrate Supplementation on the Physical Working Capacity at Neuromuscular Fatigue Threshold. J. Strength Cond. Res. 2006, 20, 928–931. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.; Ratamess, N.; Kang, J.; Mangine, G.; Faigenbaum, A.; Stout, J. Effect of creatine and beta-alanine supplementation on performance and endocrine responses in strength/power athletes. Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 430–446. [Google Scholar] [CrossRef]
- Fernández-Landa, J.; Fernández-Lázaro, D.; Calleja-González, J.; Caballero-García, A.; Córdova, A.; León-Guereño, P.; Mielgo-Ayuso, J. Long-Term Effect of Combination of Creatine Monohydrate plus β-Hydroxy β-Methylbutyrate (HMB) on Exercise-Induced Muscle Damage and Anabolic/Catabolic Hormones in Elite Male Endurance Athletes. Biomolecules 2020, 10, 140. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Landa, J.; Calleja-González, J.; León-Guereño, P.; Caballero-García, A.; Córdova, A.; Mielgo-Ayuso, J. Effect of the Combination of Creatine Monohydrate Plus HMB Supplementation on Sports Performance, Body Composition, Markers of Muscle Damage and Hormone Status: A Systematic Review. Nutrients 2019, 11, 2528. [Google Scholar] [CrossRef]
- Mobley, C.B.; Fox, C.D.; Ferguson, B.S.; Amin, R.H.; Dalbo, V.J.; Baier, S.; Rathmacher, J.A.; Wilson, J.M.; Roberts, M.D. L-leucine, beta-hydroxy-beta-methylbutyric acid (HMB) and creatine monohydrate prevent myostatin-induced Akirin-1/Mighty mRNA down-regulation and myotube atrophy. J. Int. Soc. Sports Nutr. 2014, 11, 38. [Google Scholar] [CrossRef]
- O’Connor, D.M.; Crowe, M.J. Effects of Six Weeks of ?-Hydroxy-?-Methylbutyrate (HMB) and HMB/Creatine Supplementation on Strength, Power, and Anthropometry of Highly Trained Athletes. J. Strength Cond. Res. 2007, 21, 419–423. [Google Scholar] [CrossRef]
- Crowe, M.J.; O’Connor, D.M.; Lukins, J.E. The Effects of ß-Hydroxy-ß-Methylbutyrate (HMB) and HMB/Creatine Supplementation on Indices of Health in Highly Trained Athletes. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 184–197. [Google Scholar] [CrossRef]
- Jówko, E.; Ostaszewski, P.; Jank, M.; Sacharuk, J.; Zieniewicz, A.; Wilczak, J.; Nissen, S. Creatine and β-hydroxy-β-methylbutyrate (HMB) additively increase lean body mass and muscle strength during a weight-training program. Nutrients 2001, 17, 558–566. [Google Scholar] [CrossRef]
- Green, A.L.; Simpson, E.J.; Littlewood, J.J.; Macdonald, I.A.; Greenhaff, P.L. Carbohydrate ingestion augments creatine retention during creatine feeding in humans. Acta Physiol. Scand. 1996, 158, 195–202. [Google Scholar] [CrossRef]
- Roberts, P.A.; Fox, J.; Peirce, N.; Jones, S.; Casey, A.; Greenhaff, P.L. Creatine ingestion augments dietary carbohydrate mediated muscle glycogen supercompensation during the initial 24 h of recovery following prolonged exhaustive exercise in humans. Amino Acids 2016, 48, 1831–1842. [Google Scholar] [CrossRef] [PubMed]
- Kilduff, L.P.; Vidakovic, P.; Cooney, G.; Twycross-Lewis, R.; Amuna, P.; Parker, M.; Paul, L.; Pitsiladis, Y.P. Effects of creatine on isometric bench-press performance in resistance-trained humans. Med. Sci. Sports Exerc. 2002, 34, 1176–1183. [Google Scholar] [CrossRef] [PubMed]
- Syrotuik, D.G.; Bell, G.J. Acute Creatine Monohydrate Supplementation: A Descriptive Physiological Profile of Responders vs. Nonresponders. J. Strength Cond. Res. 2004, 18, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Kaviani, M.; Shaw, K.; Chilibeck, P.D. Benefits of Creatine Supplementation for Vegetarians Compared to Omnivorous Athletes: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 3041. [Google Scholar] [CrossRef]
- Venderley, A.M.; Campbell, W.W. Vegetarian Diets. Sports Med. 2006, 36, 293–305. [Google Scholar] [CrossRef]
- Shomrat, A.; Weinstein, Y.; Katz, A. Effect of creatine feeding on maximal exercise performance in vegetarians. Graefe’s Arch. Clin. Exp. Ophthalmol. 2000, 82, 321–325. [Google Scholar] [CrossRef]
- Watt, K.K.; Garnham, A.P.; Snow, R.J. Skeletal Muscle Total Creatine Content and Creatine Transporter Gene Expression in Vegetarians Prior to and Following Creatine Supplementation. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 517–531. [Google Scholar] [CrossRef]
- Lukaszuk, J.M.; Robertson, R.J.; Arch, J.E.; Moyna, N.M. Effect of a Defined Lacto-Ovo-Vegetarian Diet and Oral Creatine Monohydrate Supplementation on Plasma Creatine Concentration. J. Strength Cond. Res. 2005, 19, 735–740. [Google Scholar] [CrossRef]
| • Increased single and repetitive sprint performance |
| • Increased work performed during sets of maximal effort muscle contractions |
| • Increased muscle mass and strength adaptations during training |
| • Enhanced glycogen synthesis |
| • Increased anaerobic threshold |
| • Possible enhancement of aerobic capacity via greater shuttling of ATP from mitochondria |
| • Increased work capacity |
| • Enhanced recovery |
| • Greater training tolerance |
| Increased PCr |
| • Track sprints: 60–200 m |
| • Swim sprints: 50 m |
| • Pursuit cycling |
| Increased PCr Resynthesis |
| • Basketball |
| • Field hockey |
| • America Football |
| • Ice hockey |
| • Lacrosse |
| • Volleyball |
| Reduced Muscle Acidosis |
| • Downhill skiing |
| • Water Sports (e.g., Rowing, Canoeing, Kayaking, Stand-Up Paddling) |
| • Swim events: 100, 200 m |
| • Track events: 400, 800 m |
| • Combat Sports (e.g., MMA, Wrestling, Boxing, etc.) |
| Oxidative Metabolism |
| • Basketball |
| • Soccer |
| • Team handball |
| • Tennis |
| • Volleyball |
| • Interval Training in Endurance Athletes |
| Increased Body Mass/Muscle Mass |
| • American Football |
| • Bodybuilding |
| • Combat Sports (e.g., MMA, Wrestling, Boxing, etc.) |
| • Powerlifting |
| • Rugby |
| • Track/Field events (Shot put; Javelin; Discus; Hammer Throw) |
| • Olympic Weightlifting |
| Author and Year | Subjects | Duration | Dosing Protocol | Primary Variables | Results | Adverse Events |
|---|---|---|---|---|---|---|
| Peak Torque/Force Production | ||||||
| Greenhaff et al. 1993 | 12 healthy males | 5 days | 20/d for 5 days | isokinetic performance during repeated intervals | ↑ muscle torque ↓ plasma ammonia ↔ BLa | None reported |
| Casey et al. 1996 | 9 active males | 5 days | 20 g/day for 5 days | cycling sprint performance | ↑ total work | None reported |
| Gilliam et al. 2000 | 23 healthy males | 5 days | 20 g/day for 5 days | isokinetic performance of quadriceps | ↔ muscle torque | None reported |
| Rossouw et al. 2000 | 13 trained powerlifters | 5 days | 9 g/day for 5 days | isokinetic knee extension | ↑ peak torque ↑ average power, ↑ total work ↑ work output ↑ deadlift volume | None reported |
| Kilduff et al. 2002 | 32 trained males | 5 days | 20 g/day for 5 days | isometric bench press | ↑ peak force ↑ total force | None reported |
| Strength Performance | ||||||
| Birch et al. 1994 | 14 healthy males | 5 days | 20 g/day for 5 days | isokinetic cycling performance during repeated intervals | ↑ mean power output ↑ peak power output ↑ total power output ↓ plasma ammonia ↔ BLa | None reported |
| Barnett et al.1996 | 17 active males | 4 days | 280 mg/kg for 4 days | 10 s sprints on a cycle ergometer | ↔ mean power output ↔ peak power output ↔ blood pH ↔ BLa | None reported |
| Strength and Performance Outcomes | ||||||
| Edwards et al. 2000 | 21 active males | 6 days | 20 g/d for 6 days | anaerobic intervals on a treadmill | ↔ speed test performance ↔ BLa ↓ plasma ammonia | None reported |
| Rockwell et al. 2001 | 16 trained males | 4 days | 20 g/day for 4 days | repeated cycling sprints | ↔ total work ↔ maximal power ↔ work capacity | None reported |
| Volek et al. 2001 | 10 healthy males | 7 days | 0.3 g/kg for 7 days | repeated cycling sprints cardiovascular and thermoregulatory responses | ↔ cardiovascular or thermoregulatory responses ↑ peak power ↑ mean power | None reported |
| Finn et al. 2001 | 16 male triathletes | 5 days | 20 g/day for 5 days | repeated cycling sprints | ↔ peak power ↔ fatigue index ↔ mean power | None reported |
| Ziegenfuss et al. 2002 | 20 (10 male, 10 female) power athletes | 3 days | 0.35 g/kg of fat-free mass for 3 days | repeated cycling sprints | ↑ peak power ↑ total work | None reported |
| Yquel et al. 2002 | 9 healthy males | 6 days | 20 g/day for 6 days | repeated dynamic plantar flexion muscle pH | ↑ mean power ↔ muscle pH | None reported |
| Delecluse et al. 2003 | 12 (7 male, 5 female) competitive sprinters | 7 days | 0.35 g/day for 7 days | single 40-m sprint repeated 40-m sprints | ↔ single 40-m sprint ↔ repeated 40-m sprints | None reported |
| Kocak et al. 2003 | 20 elite male wrestlers | 5 days | 20 g/day for 5 days | average and peak power during wingate anaerobic power test | ↑ average power ↑ peak power | None reported |
| Selsby et al. 2004 | 31 trained males | 10 days | 2.5 g/day for 10 days | bench press strength bench press endurance | ↔ bench press strength ↔ bench press endurance | None reported |
| Zuniga et al. 2012 | 22 active males | 7 days | 20 g/day for 7 days | wingate cycling test lower body strength upper body strength | ↑ mean power output ↔ leg extension ↔ bench press | None reported |
| del Favero et al. 2012 | 34 untrained males | 10 days | 20 g/day for 10 days | lower body strength upper body strength | ↑ bench press ↑ squat | None reported |
| Wang et al. 2017 | 17 high school canoeists | 6 days | 20 g/day for 6 days | upper body strength overhead medicine ball throw Post-activation potentiation | ↑ upper body strength ↔ overhead medicine ball throw ↔ post-activation potentiation | None reported |
| Author and Year | Subjects | Duration | Dosing Protocol | Primary Variables | Results | Adverse Events |
|---|---|---|---|---|---|---|
| Peak Torque/Force Production | ||||||
| Stevenson et al. 2001 | 18 (17 males, 1 female) trained subjects | 9 weeks | 20 g/day for 7 days 5 g/day 8 weeks | maximal torque on isokinetic dynamometer quadriceps cross-sectional area | ↔ maximal torque ↑ cross-sectional area | None reported |
| Chrusch et al. 2001 | 30 healthy older males | 12 weeks | 0.03 g/kg/day for 5 days 0.07 g/kg/day for 11 weeks | lean tissue bench press strength leg press strength knee extension muscle endurance average power | ↑ lean tissue ↔ bench press ↑ leg press ↑ knee torque ↑ muscle endurance ↑ average power | None reported |
| Strength | ||||||
| Vandenberghe et al.1997 | 19 healthy female subjects | 11 weeks | 5 g/day for 4 days 2.5 g/day for 10 weeks | arm flexion on isokinetic dynamometer upper and lower body muscle strength body composition | ↑ arm torque ↑ leg press, leg extension, squat ↔ bench press, leg curl, shoulder press ↑ lean muscle mass | None reported |
| Kelly et al. 1998 | 18 male powerlifters | 26 days | 20 g/day for 5 days 5 g/day for 21 days | bench press strength bench press endurance total body mass | ↑ bench press strength ↑ bench press endurance ↑ body mass | None reported |
| Volek et al. 1999 | 19 trained males | 12 weeks | 25 g/day for 7 days 5 g/day for 11 weeks | body mass fat-free mass bench press strength squat strength quadriceps cross-sectional area | ↔ body mass ↑ fat-free mass ↑ bench press ↑ squat ↑ quadriceps cross-sectional area | None reported |
| Becque et al. 2000 | 23 trained males | 6 weeks | 20 g/day for 5 days 2 g/day for days 6–42 | upper body strength body composition | ↑ arm flexor strength ↑ body mass ↔ body fat ↑ fat-free mass ↑ upper arm muscle area | None reported |
| Brenner et al. 2000 | 20 female college lacrosse players | 5 weeks | 20 g/day for 7 days 2 g/day for days 8–32 | body composition bench press strength knee extension strength knee extension endurance BLa | ↔ body composition ↑ bench press strength ↔ knee extension strength ↔ knee extension endurance ↔ BLa | None reported |
| Larson-Meyer et al. 2000 | 14 female college soccer players | 13 weeks | 15 g/day for 5 days 5 g/day 5 days/week for 12 weeks | bench press strength squat strength vertical jump body composition | ↑ bench press strength ↑ squat strength ↔ vertical jump ↑ body mass ↑ lean mass ↔ body fat | 1 subject reported nausea |
| Bemben et al. 2001 | 25 male college football players | 9 weeks | 20 g/day for 5 days 5 g/day for 8 weeks | bench press strength squat strength power clean strength Wingate cycling test isokinetic strength body composition | ↑ bench press strength ↑ squat strength ↑ power clean strength ↑ anaerobic power, capacity and % decrement ↑ peak torque knee flexion ↔ peak torque knee extension ↑ lean body mass ↔ body fat | None reported |
| Burke et al. 2001 | 47 active male subjects | 21 days | 7.7 g/day for 21 days | bench press on a isokinetic dynamometer | ↑ peak force ↑ peak power ↑ time to fatigue | None reported |
| Chrusch et al. 2001 | 30 healthy older males | 12 weeks | 0.03 g/kg/day for 5 days 0.07 g/kg/day for 11 weeks | lean tissue bench press strength leg press strength knee extension muscle endurance average power | ↑ lean tissue ↔ bench press ↑ leg press ↑ knee torque ↑ muscle endurance ↑ average power | None reported |
| Wilder et al. 2001 | 25 male college football players | 10 weeks | 3 g/day for 10 week or 20 g/day for 7 days, then 5 g/day for rest of the study | squat strength body composition | ↔ squat strength ↔ lean body mass ↔ fat mass | None reported |
| Burke et al. 2003 | 49 (20 male, 29 female) active subjects | 8 weeks | 0.25 g/kg lean tissue/day for 7 days 0.0625 g/kg lean tissue/day for 49 days | bench press strength leg press strength isokinetic endurance quadriceps cross-sectional area body composition | ↑ bench press ↔ leg press ↑ total work ↑ body mass ↑ lean body mass ↑ cross-sectional area | None reported |
| Ferguson et al. 2006 | 26 trained females | 10 weeks | 0.3 g/kg for 7 days 0.03 g/kg for 9 weeks | bench press strength leg press strength body composition | ↔ bench press ↔ leg press ↔ total mass ↔ lean body mass ↔ fat mass | None reported |
| Kerksick et al. 2009 | 24 trained males | 4 weeks | 20 g/day for 5 days 5 g/day for 23 days | bench press strength leg press strength isokinetic knee extension Wingate cycling test body composition | ↑ bench press ↑ leg press ↔ peak torque ↔ peak power ↑ lean body mass ↑ fat-free mass | None reported |
| Camic et al. 2010 | 22 untrained males | 28 days | 5 g/day for 28 days | bench press strength leg extension strength Wingate cycling test | ↑ bench press ↔ leg extension ↔ mean power ↔ peak power | None reported |
| Hummer et al. 2019 | 22 (16 males, 6 females) active subjects | 6 weeks | 4 g/day for 6 weeks | bench press strength bench press endurance squat strength squat endurance | ↑ bench press strength ↑ bench press endurance ↑ squat strength ↔ squat endurance | None reported |
| Strength and Performance Outcomes | ||||||
| Kreider et al. 1998 | 25 college football players | 28 days | 15.75 g/day for 28 days | total work during sprints on a cycle ergometer bench press volume total volume | ↑ total work ↑ bench press volume ↑ total volume | None reported |
| Stone et al. 1999 | 42 college football players | 5 weeks | 0.22 g/kg/day for 5 weeks | bench press strength squat strength countermovement vertical jump static vertical jump body composition | ↑ bench press strength ↔ squat strength ↔ countermovement vertical jump ↔ static vertical jump ↑ body mass ↑ lean body mass | None reported |
| Chilibeck, et al. 2007 | 19 male union rugby players | 8 weeks | 0.7 g/kg/day for 8 weeks | bench press endurance leg press endurance body composition | ↔ bench press repetitions ↔ leg press repetitions ↑ when combining bench press and leg press scores ↔ total body mass ↔ lean tissue mass ↔ fat mass | None reported |
| Author Year | Subjects | Duration | Dosing Protocol | Primary Variables | Results | Adverse Events |
|---|---|---|---|---|---|---|
| Grindstaff et al. 1997 | 18 (7 male, 11 female) junior competitive swimmers | 9 days | 21 g/day for 9 days | 100-m sprint performance arm ergometer performance | ↑ sprint swimming performance | None reported |
| Kreider et al. 1998 | 25 college football players | 28 days | 15.75 g/day for 28 days | total work during sprints on a cycle ergometer bench press volume total volume | ↑ total work ↑ bench press volume ↑ total volume | None reported |
| Noonan et al. 1998 | 39 college football players | 9 weeks | 20 g/day for 5 days 100 or 300 mg/kg/fat-free mass for 8 weeks | bench press 40-yard dash % body fat fat-free mass vertical jump | ↑ bench press ↑ 40-yard dash ↔ % body fat ↔ fat-free mass ↔ vertical jump | None reported |
| Peyrebrune et al. 1998 | 14 male college swimmers | 5 days | 9 g/day for 5 days | single 50-m sprint time repetitive 50-m sprint time | ↔ single 50 m sprint time ↑ repetitive 50 m sprint time | None reported |
| Stout et al. 1999 | 24 college football players | 8 weeks | 21 g/day for 5 days 10 g/day thereafter | vertical jump 100-yard dash bench press strength | ↑ vertical jump ↑ 100-yard dash ↑ bench press strength | None reported |
| Jones et al. 1999 | 8 elite ice hockey players | 11 weeks | 20 g/day for 5 days 5 g/day for 10 weeks | 5 × 15 s skating sprints 6 timed 80-m skating sprints | ↑ 5 × 15 s skating sprints ↑ 6 timed 80 m skating sprints | None reported |
| Kirksey et al. 1999 | 36 (16 male, 20 female) track and field athletes | 6 weeks | 0.3 g/kg/day | countermovement vertical jump power and total work during sprints on a cycle ergometer | ↑ countermovement vertical jump ↑ peak power ↑ total work on cycle ergometer | None reported |
| Kreider et al. 1999 | 61 college football players | 12 weeks | 20–25 g/day | Bench press strength Bench press endurance Body composition | ↑ bench press strength ↑ bench press endurance ↑ body mass ↑ soft tissue lean mass | None reported |
| Mujika et al. 2000 | 17 trained soccer players | 10 weeks | 20 g/day for 5 days 5 g/day for 9 weeks | countermovement jump repeated sprint ability | ↔ countermovement jump ↑ repeated sprint ability | None reported |
| Haff et al. 2000 | 36 (16 male, 20 female) track and field athletes | 6 weeks | 0.3 g/kg/day | countermovement vertical jump | ↑ countermovement vertical jump | None reported |
| Skare et al. 2001 | 18 male competitive sprinters | 5 days | 20 g/day for 5 days | 100-m sprint time total sprint time (6 × 60 m) | ↑ 100 m sprint time ↑ total sprint time | None reported |
| Romer et al. 2001 | 9 competitive squash players | 5 days | 0.075 g/kg 4 times for 5 days | single sprint repetitive sprint performance | ↔ single sprint ↑ repetitive sprint performance | None reported |
| Izquierdo et al. 2002 | 19 male handball players | 5 days | 20 g/day for 5 days | countermovement vertical jump repetitive sprint performance | ↑ countermovement vertical jump ↑ 6 × 15 m sprints | None reported |
| Cox et al. 2002 | 12 elite female soccer players | 6 days | 20 g/day for 6 days | agility kick drill test agility race test repetitive sprint performance BLa | ↔ kick drill test ↑ agility run ↑ repetitive sprint performance ↓ BLa | None reported |
| Lehmkul et al. 2003 | 29 (17 male, 12 female) track and field athletes | 8 weeks | 0.3 g/kg/day for 7 days 0.03 g/kg/day for 7 weeks. | average and peak power during repeated sprints on a cycle ergometer | ↔ static vertical jump ↔ countermovement vertical jump ↔ average power ↔ peak power | None reported |
| Delecluse et al. 2003 | 12 (7 male, 5 female competitive sprinters | 7 days | 0.35 g/day for 7 days | single 40-m sprint repeated 40-m sprints | ↔ single 40 m sprint ↔ repeated 40 m sprints | None reported |
| Kocak et al. 2003 | 20 elite male wrestlers | 5 days | 20 g/day for 5 days | average and peak power during Wingate anaerobic power test | ↑ average power ↑ peak power | None reported |
| Ostojic et al. 2004 | 20 young male soccer players | 7 days | 30 g/day for 7 days | dribbling test sprint power countermovement jump | ↑ dribbling test ↑ sprint power ↑ countermovement vertical jump | None reported |
| Pluim et al. 2006 | 36 competitive tennis players | 32 days | 0.3 g/day for 6 days 0.03 g/day for 28 days | serve velocity groundstroke velocity repetitive sprints | ↔ serve velocity ↔ groundstroke velocity ↔ repetitive sprints | None reported |
| Glaister et al. 2006 | 42 active males | 5 days | 20 g/day for 5 days | repetitive sprint performance | ↔ repetitive sprint performance | None reported |
| Lamontagne-Lacasse et al. 2011 | 12 elite male volleyball players | 28 days | 20 g/day in days 1–4 10 g/day in days 5–6 5 g/day in days 7–28 | repeated block jump spike jump | ↔ repeated block jump ↔ spike jump | None reported |
| Ramierz-Campillo et al. 2016 | 30 amateur female soccer players | 6 weeks | 20 g/day for 7 days 5 g/day for 5 weeks | jump test repeated sprinting directional change | ↑ jump test ↑ repeated sprinting ↔ directional change | None reported |
| Author-Year | Subjects | Duration | Dosing Protocol | Primary Variables | Results | Adverse Events |
|---|---|---|---|---|---|---|
| Rossiter et al. 1996 | 38 (28 male, 10 female) competitive rowers | 5 days | 0.25 g/kg/day for 5 days | time trial performance during rowing ergometry | ↓ 2.3 s in 1000-m times | None reported |
| McNaughton et al. 1998 | 16 elite male paddlers | 5 days | 20 g/day for 5 days | total work, peak power, BLa during rowing ergometry | ↑ in total work during 90–300 s of rowing ergometry performance | None reported |
| Miura et al. 1999 | 8 healthy males | 5 days | 20 g/day for 5 days | critical power test during cycle ergometry | ↔ critical power ↑ anaerobic work capacity | None reported |
| Rico-Sanz et al. 2000 | 14 elite male cyclists | 5 days | 20 g/day for 5 days | oxygen consumption, time to exhaustion, BLa during maximal cycle ergometry | ↔ VO2 max ↑ time to exhaustion ↔ BLa | None reported |
| Syrotuik el al. 2001 | 22 (12 male, 10 female) competitive rowers | 6 weeks | 0.3 g/kg/day for 5 days 0.03 g/kg/day for 5 weeks | time trial performance during rowing ergometry | ↔ in 2000-m rowing times | None reported |
| Jones et al. 2002 | 9 active males | 5 days | 20 g/day for 5 days | VO2 kinetics during moderate and heavy submaximal cycle exercise | ↔ VO2 kinetics ↓ VO2 during heavy cycling exercise | None reported |
| Chwalbinska-Moneta 2003 | 16 elite male rowers | 5 days | 20 g/day for 5 days | maximal power output, time to exhaustion, Bla during rowing ergometry | ↔ maximal power output ↑ time to exhaustion ↔ BLa | None reported |
| Graef et al. 2009 | 43 active males | 30 days | 10 g/day for 20 days; only on training days (5 × week) | oxygen consumption, time to exhaustion, VT, total work, during maximal cycle ergometry | ↔ VO2 peak ↑ time to exhaustion ↑ ventilatory threshold ↔ Total work | None reported |
| Kendall et al. 2009 | 43 active males | 30 days | 10 g/day for 20 days; only on training days (5 × week) | critical power and anaerobic work capacity during cycle ergometry | ↑ Critical power ↔ Anaerobic work capacity | None reported |
| Hickner et al. 2010 | 12 endurance-trained males | 28 days | 3 g/day for 28 days | VO2peak, submaximal VO2, RER, Bla, 10 s sprints at 110% VO2peak during simulated cycling road race | ↔ VO2peak ↓ submaximal VO2 ↔ RER ↔ Bla, ↔ 10-s sprints at 110% VO2peak | 2 subjects reported muscle cramping at rest following supplementation |
| De Andrade Nemezio et al. 2015 | 24 male amateur cyclists | 5 days | 20 g/day for 5 days | time trial performance total O2 uptake, BLa during maximal cycle ergometry | ↔ 1000 m time ↓ total O2 uptake ↔ BLa | None reported |
| Forbes et al. 2017 | 17 active females | 28 days | 0.3 g/kg/day for 5 days 0.1 g/kg/day for 23 days | VO2peak, VT, peak workload, time trial performance during cycle ergometry | ↔ VO2peak ↔ VT ↔ 2000-m time ↔ peak workload | None reported |
| Fernandez-Landa et al. 2020 | 28 elite male rowers | 10 weeks | 0.04 g/kg/day for 10 weeks + 3 g HMB/day for 10 weeks | power output at AT, 4 mmol, 8 mmol Bla during rowing ergometry | ↑ power at AT for creatine-HMB and HMB only group ↑ power at 4 mmol BLa for creatine-HMB group ↑ power at 8 mmol BLa for creatine only, HMB only, and creatine-HMB groups | None reported |
| Author Year | Subjects | Duration | Dosing Protocol | Primary Variables | Results | Adverse Events |
|---|---|---|---|---|---|---|
| Oopik et al. 2002 | 5 well-trained male wrestlers | 17 hours | 30 g (7.5 g/serving) + 320 g glucose (80 g/serving) over 4 doses | isokinetic performance blood glucose blood lactate plasma ammonia plasma urea body mass | ↑ submaximal work ↔ blood glucose ↔ Bla ↓ plasma ammonia ↔ plasma urea ↔ body mass | None reported |
| Hespel et al. 2001 | 22 (13 males, 9 females) | 10 weeks | 20 g/day for 3 weeks (immobilization) 15 g/day for 3 weeks (10–8) 5 g/day for 7 weeks (7–1). | quadriceps cross-sectional area (CSA) knee extension isometric force | ↑ CSA ↑ knee torque ↑ isometric force | None reported |
| Tyler et al. 2004 | 60 ACL reconstruction patients (33 males, 27 females) | 6 months | 20 g/day for 7 days 5 g/day for 12 weeks | knee extension knee flexion hip flexion hip abduction hip adduction single leg hop | ↑ knee outcome measures comparing to baseline | None reported |
| Rawson et al. 2007 | 22 trained males | 10 days | 0.3 g/kg/day for 5 days 0.03 g/kg/day for 5 days | maximal strength range of Motion muscle Soreness blood lactate | ↔ strength ↔ ROM ↔ soreness ↔ Bla | None reported |
| Cooke et al. 2009 | 14 untrained males | 20 days | 0.3 g/kg/day + glucose (80 g/day) for 5 days 0.1 g/kg/day + glucose (0.4 g/day) for 14 days | isokinetic force isometric force | ↑ isokinetic force ↑ isometric force | None reported |
| Rosene et al. 2009 | 20 healthy males | 30 days | 20 g/day for 7 days 6 g/day for 23 days | isometric force knee range of motion muscle soreness creatine kinase blood lactate | ↑ isometric force | None reported |
| Johnston et al. 2009 | 7 healthy males | 30 days | Maltodextrin 20 g/day for 7 days (Day 1–7) 20 g/day for 7 days (Day 15–21) | fat free mass elbow flexor strength and endurance elbow extensor strength & endurance | ↑ lean tissue ↑ muscular strength ↑ muscular endurance | None reported |
| McKinnon et al. 2012 | 27 (15 male, 12 female) untrained subjects | 10 days | 40 g/day + CHO 40 g/day for 5 days 5 g/day + CHO 5 g/day for 5 days. | muscle force loss rate of recovery muscle soreness | ↔ force loss ↔ rate of recovery ↔ muscle soreness | None reported |
| Boychuk et al. 2016 | 14 healthy males | 48 hours | 0.3 d/kg | maximal voluntary contraction muscle thickness electromyography muscle soreness | ↔ strength ↔ EMG activation ↔ muscle soreness | None reported |
| Backx et al. 2017 | 30 healthy males | 12 days | 20 g/day for 5 days 5 g/day for 7 days | quadriceps cross-sectional area (CSA) leg 1 RM knee extensions | ↔ CSA ↔ 1 RM | None reported |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wax, B.; Kerksick, C.M.; Jagim, A.R.; Mayo, J.J.; Lyons, B.C.; Kreider, R.B. Creatine for Exercise and Sports Performance, with Recovery Considerations for Healthy Populations. Nutrients 2021, 13, 1915. https://doi.org/10.3390/nu13061915
Wax B, Kerksick CM, Jagim AR, Mayo JJ, Lyons BC, Kreider RB. Creatine for Exercise and Sports Performance, with Recovery Considerations for Healthy Populations. Nutrients. 2021; 13(6):1915. https://doi.org/10.3390/nu13061915
Chicago/Turabian StyleWax, Benjamin, Chad M. Kerksick, Andrew R. Jagim, Jerry J. Mayo, Brian C. Lyons, and Richard B. Kreider. 2021. "Creatine for Exercise and Sports Performance, with Recovery Considerations for Healthy Populations" Nutrients 13, no. 6: 1915. https://doi.org/10.3390/nu13061915
APA StyleWax, B., Kerksick, C. M., Jagim, A. R., Mayo, J. J., Lyons, B. C., & Kreider, R. B. (2021). Creatine for Exercise and Sports Performance, with Recovery Considerations for Healthy Populations. Nutrients, 13(6), 1915. https://doi.org/10.3390/nu13061915







