Does Protein Supplementation Support Adaptations to Arduous Concurrent Exercise Training? A Systematic Review and Meta-Analysis with Military Based Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
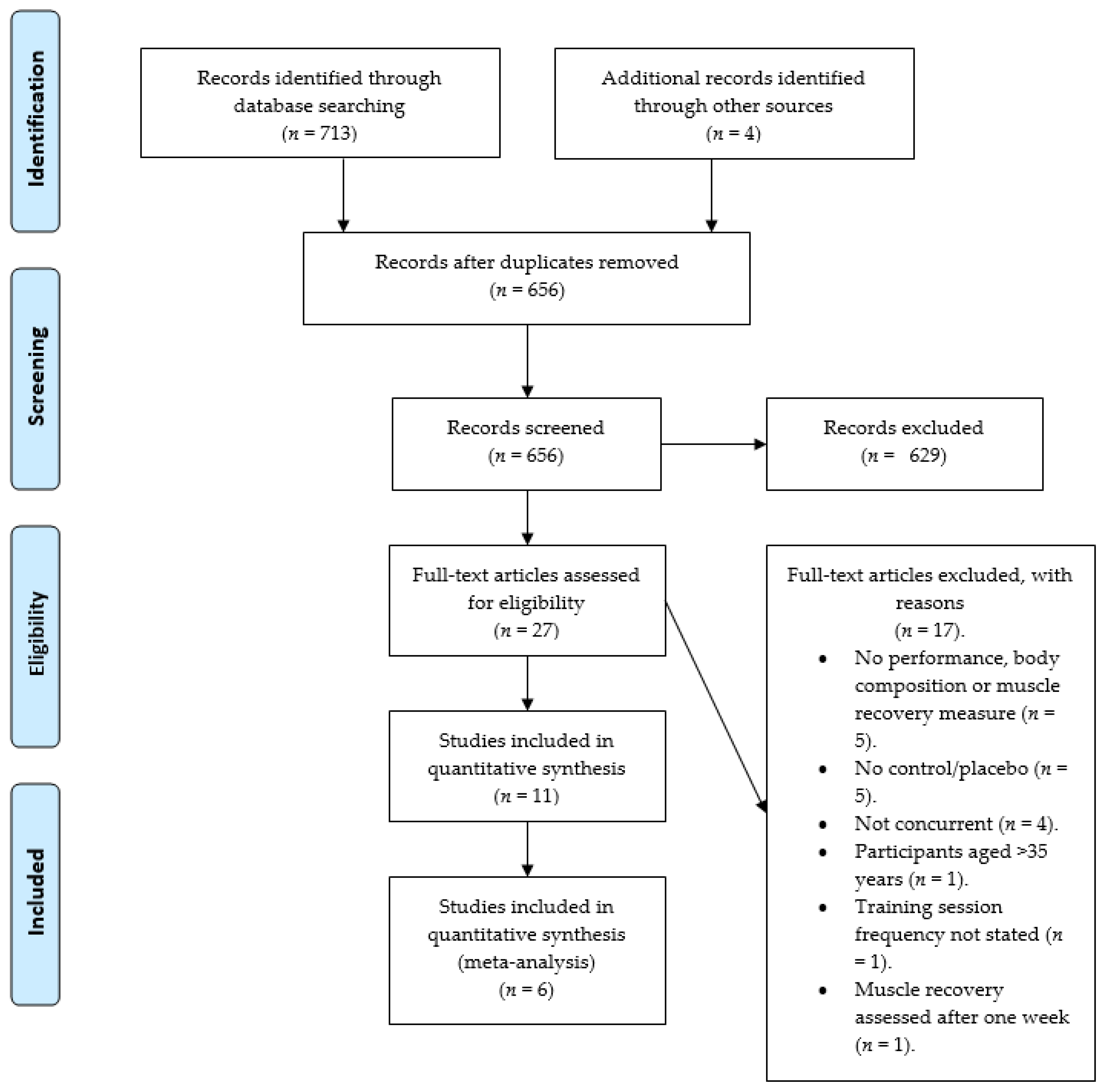
2.2. Search Strategy and Study Selection
2.3. Risk of Bias Assessment
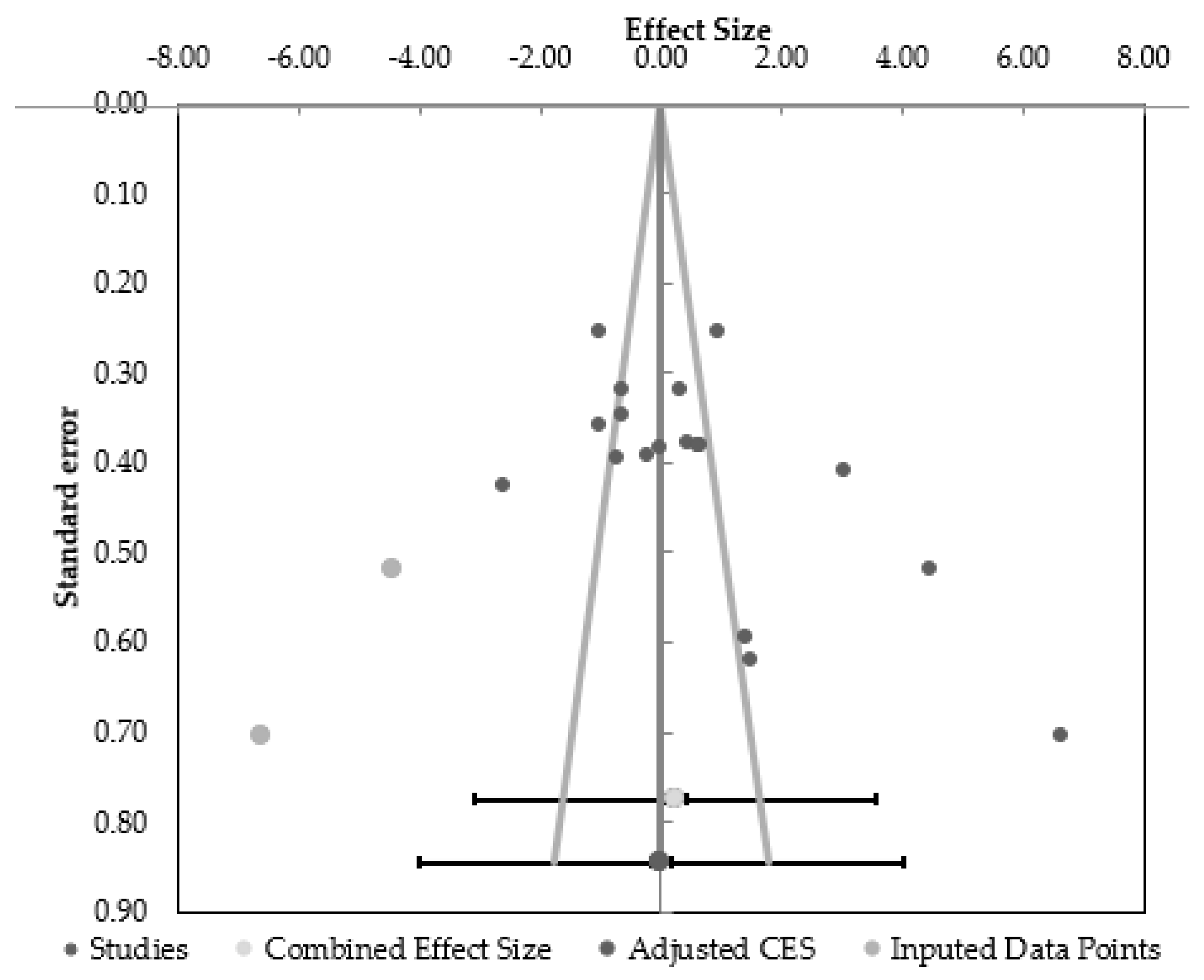
2.4. Data Synthesis and Analyses
3. Results
3.1. Study Quality and Risk of Bias Assessment
3.2. Participant Characteristics and Study Interventions
3.3. Protein Dose and Timing
3.4. Synthesis of Results
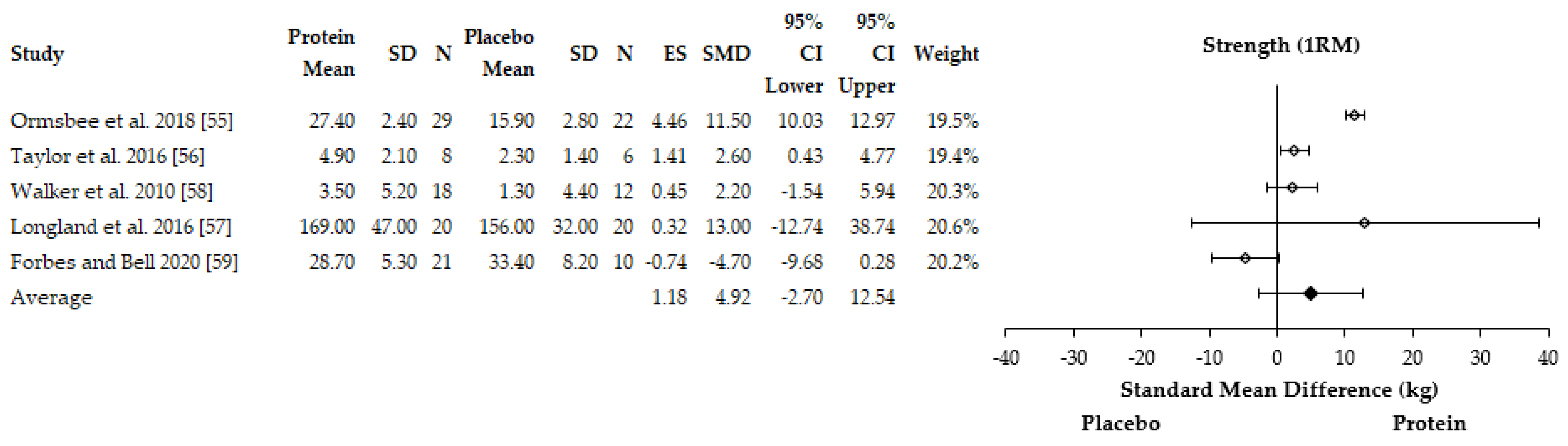
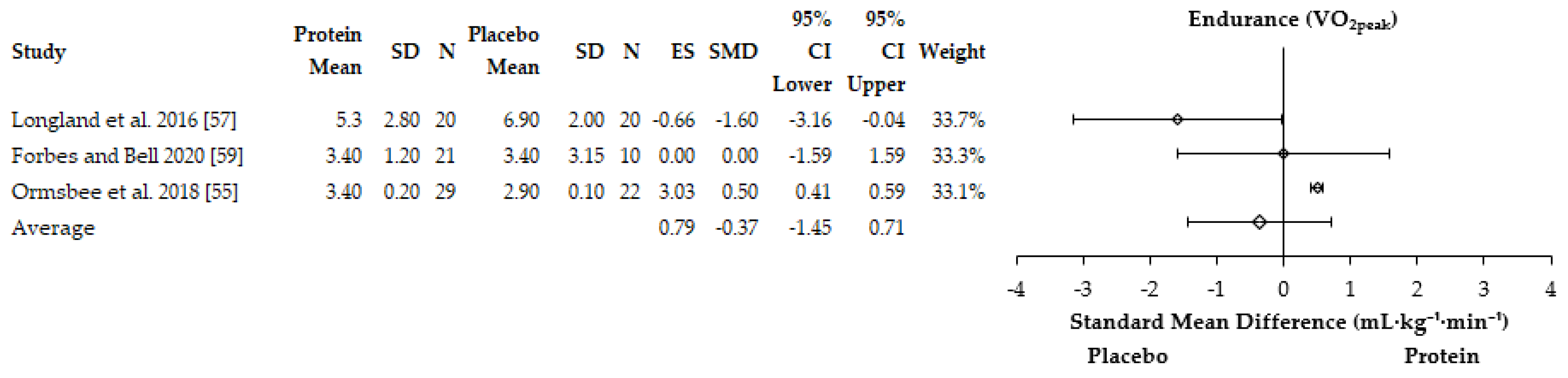
3.4.1. Performance Adaptations
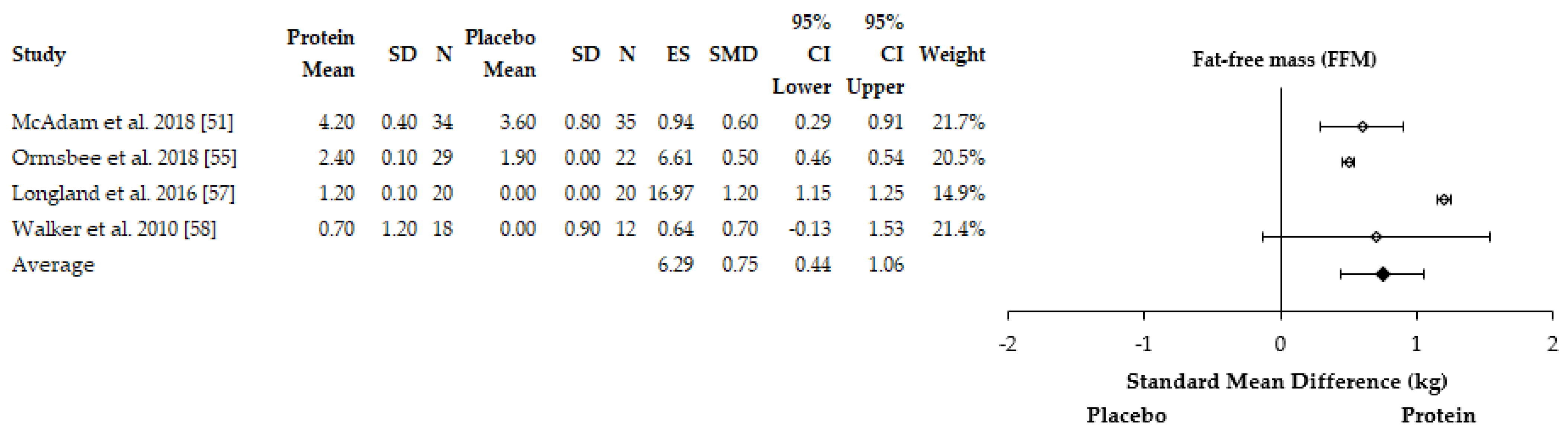
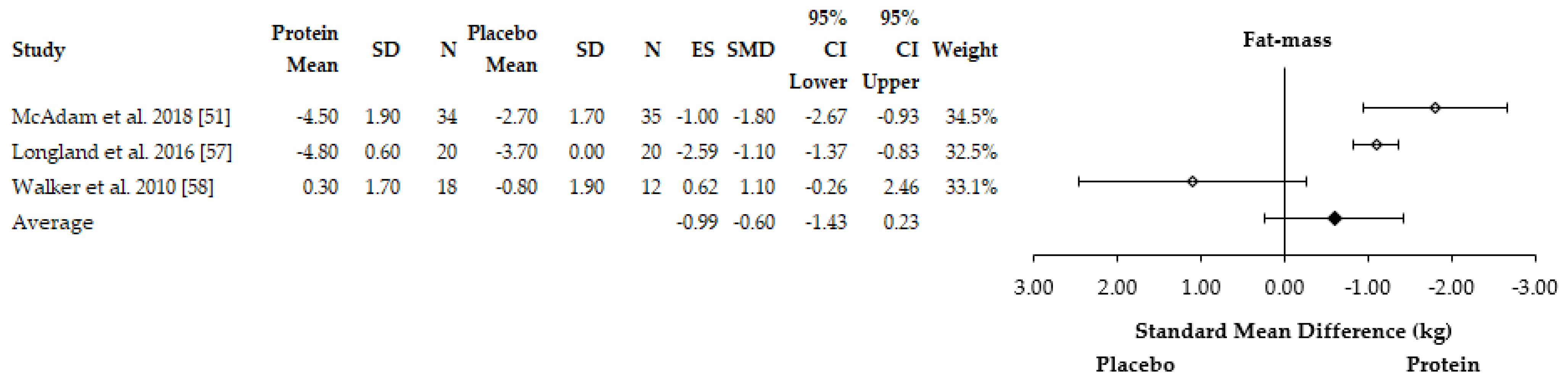
3.4.2. Body Composition Adaptations
3.4.3. Muscle Recovery Adaptations
4. Discussion
4.1. Muscle Strength and Body Composition Adaptations
4.2. Aerobic Adaptations
4.3. Muscle Recovery Adaptations
4.4. Limitations
4.5. Military Research Applications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wilson, J.M.; Marin, P.J.; Rhea, M.R.; Wilson, S.M.; Loenneke, J.P.; Anderson, J.C. Concurrent Training. J. Strength Cond. Res. 2012, 26, 2293–2307. [Google Scholar] [CrossRef]
- Nader, G.A. Concurrent Strength and Endurance Training. Med. Sci. Sports Exerc. 2006, 38, 1965–1970. [Google Scholar] [CrossRef]
- Claudino, J.G.; Gabbett, T.J.; Bourgeois, F.; Souza, H.D.S.; Miranda, R.C.; Mezêncio, B.; Soncin, R.; Filho, C.A.C.; Bottaro, M.; Hernandez, A.J.; et al. CrossFit Overview: Systematic Review and Meta-analysis. Sports Med.-Open 2018, 4, 1–14. [Google Scholar] [CrossRef]
- Bonnici, D.C.; Greig, M.; Akubat, I.; Sparks, S.A.; Bentley, D.; Mc Naughton, L.R. Nutrition in Soccer: A Brief Review of the Issues and Solutions. J. Sci. Sport Exerc. 2019, 1, 3–12. [Google Scholar] [CrossRef]
- Bradley, W.; Cavanagh, B.; Douglas, W.; Donovan, T.F.; Morton, J.P.; Close, G.L. Quantification of Training Load, Energy Intake, and Physiological Adaptations during a Rugby Preseason: A Case Study from an Elite European Rugby Union Squad. J. Strength Cond. Res. 2014, 12, 1–12. [Google Scholar] [CrossRef]
- Calleja-González, J.; Mielgo-Ayuso, J.; Sampaio, J.; Delextrat, A.; Ostojic, S.M.; Marques-Jiménez, D.; Arratibel, I.; Sánchez-Ureña, B.; Dupont, G.; Schelling, X.; et al. Brief ideas about evidence-based recovery in team sports. J. Exerc. Rehabil. 2018, 14, 545–550. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, T.; Saunders, S.; McGuire, S.; Kefyalew, S.; Venables, M.; Izard, R. Sex differences in physical performance and body composition adaptations to British Army basic military training. J. Sci. Med. Sport 2017, 20, S80. [Google Scholar] [CrossRef]
- Drain, J.R.; Sampson, J.A.; Billing, D.C.; Burley, S.D.; Linnane, D.M.; Groeller, H. The Effectiveness of Basic Military Training to Improve Functional Lifting Strength in New Recruits. J. Strength Cond. Res. 2015, 29, S173–S177. [Google Scholar] [CrossRef] [PubMed]
- Richmond, V.L.; Carter, J.M.; Wilkinson, D.M.; Horner, F.E.; Rayson, M.P.; Wright, A.; Bilzon, J.L. Comparison of the Physical Demands of Single-Sex Training for Male and Female Recruits in the British Army. Mil. Med. 2012, 177, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Richmond, V.L.; Horner, F.E.; Wilkinson, D.M.; Rayson, M.P.; Wright, A.; Izard, R. Energy Balance and Physical Demands During an 8-Week Arduous Military Training Course. Mil. Med. 2014, 179, 421–427. [Google Scholar] [CrossRef]
- Blacker, S.D.; Wilkinson, D.M.; Rayson, M.P. Gender Differences in the Physical Demands of British Army Recruit Training. Mil. Med. 2009, 174, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.; Heagerty, R.; Dalal, S.; Banerjee, B.; Booker, T. Risk Factors Associated With Musculoskeletal Injury: A Prospective Study of British Infantry Recruits. Curr. Rheumatol. Rev. 2018, 15, 50–58. [Google Scholar] [CrossRef]
- Wardle, S.L.; Greeves, J.P. Mitigating the risk of musculoskeletal injury: A systematic review of the most effective injury prevention strategies for military personnel. J. Sci. Med. Sport 2017, 20, S3–S10. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, T.J.; Wardle, S.L.; Greeves, J.P. Energy Deficiency in Soldiers: The Risk of the Athlete Triad and Relative Energy Deficiency in Sport Syndromes in the Military. Front. Nutr. 2020, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Chapman, S.; Rawcliffe, A.J.; Izard, R.; Jacka, K.; Tyson, H.; Smith, L.; Roberts, J. Dietary Intake and Nitrogen Balance in British Army Infantry Recruits Undergoing Basic Training. Nutrients 2020, 12, 2125. [Google Scholar] [CrossRef]
- Cermak, N.M.; Res, P.T.; De Groot, L.C.P.G.M.; Saris, W.H.M.; Van Loon, L.J.C. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1454–1464. [Google Scholar] [CrossRef]
- Lin, Y.-N.; Tseng, T.-T.; Knuiman, P.; Chan, W.P.; Wu, S.-H.; Tsai, C.-L.; Hsu, C.-Y. Protein supplementation increases adaptations to endurance training: A systematic review and meta-analysis. Clin. Nutr. 2020, 1–10. [Google Scholar] [CrossRef]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2018, 52, 376–384. [Google Scholar] [CrossRef]
- Davies, R.W.; Carson, B.P.; Jakeman, P.M. The Effect of Whey Protein Supplementation on the Temporal Recovery of Muscle Function Following Resistance Training: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 221. [Google Scholar] [CrossRef]
- Wilkinson, S.B.; Phillips, S.M.; Atherton, P.J.; Patel, R.; Yarasheski, K.E.; Tarnopolsky, M.A.; Rennie, M.J. Differential effects of resistance and endurance exercise in the fed state on signalling molecule phosphorylation and protein synthesis in human muscle. J. Physiol. 2008, 586, 3701–3717. [Google Scholar] [CrossRef]
- Egan, B.; Zierath, J.R. Exercise Metabolism and the Molecular Regulation of Skeletal Muscle Adaptation. Cell Metab. 2013, 17, 162–184. [Google Scholar] [CrossRef]
- Craig, B.W.; Lucas, J.; Pohlman, R.; Stelling, H. The Effects of Running, Weightlifting and a Combination of Both on Growth Hormone Release. J. Strength Cond. Res. 1991, 5, 198–203. [Google Scholar]
- Hennessy, L.C.; Watson, A.W.S. The Interference Effects of Training for Strength and Endurance Simultaneously. J. Strength Cond. Res. 1994, 8, 12–19. [Google Scholar]
- Kraemer, W.J.; Patton, J.F.; Gordon, S.E.; Harman, E.A.; Deschenes, M.R.; Reynolds, K.; Newton, R.U.; Triplett, N.T.; Dziados, J.E. Compatibility of high-intensity strength and endurance training on hormonal and skeletal muscle adaptations. J. Appl. Physiol. 1995, 78, 976–989. [Google Scholar] [CrossRef] [PubMed]
- Fyfe, J.J.; Bishop, D.J.; Bartlett, J.D.; Hanson, E.D.; Anderson, M.J.; Garnham, A.P.; Stepto, N.K. Enhanced skeletal muscle ribosome biogenesis, yet attenuated mTORC1 and ribosome biogenesis-related signalling, following short-term concurrent versus single-mode resistance training. Sci. Rep. 2018, 8, 560. [Google Scholar] [CrossRef] [PubMed]
- Fyfe, J.J.; Bartlett, J.D.; Hanson, E.D.; Stepto, N.K.; Bishop, D.J. Endurance Training Intensity Does Not Mediate Interference to Maximal Lower-Body Strength Gain during Short-Term Concurrent Training. Front. Physiol. 2016, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Baar, K. Training for Endurance and Strength: Lessons from Cell Signaling. Med. Sci. Sport Exerc. 2006, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Baar, K. Using molecular biology to maximize concurrent training. Sports Med. 2014, 44, S117–S125. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, T.R.; Fernandez-Gonzalo, R.; Gustafsson, T.; Tesch, P.A. Aerobic exercise does not compromise muscle hypertrophy response to short-term resistance training. J. Appl. Physiol. 2013, 114, 81–89. [Google Scholar] [CrossRef]
- De Souza, E.O.; Roschel, H.; Brum, P.C.; Bacurau, A.N.; Ferreira, J.B.; Aoki, M.S.; Neves, M., Jr.; Aihara, A.Y.; Ugrinowitsch, C.; Tricoli, V.; et al. Molecular Adaptations to Concurrent Training. Int. J. Sports Med. 2012, 34, 207–213. [Google Scholar] [CrossRef]
- Lundberg, T.R.; Fernandez-Gonzalo, R.; Tesch, P.A. Exercise-induced AMPK activation does not interfere with muscle hypertrophy in response to resistance training in men. J. Appl. Physiol. 2014, 116, 611–620. [Google Scholar] [CrossRef]
- Lantier, L.; Fentz, J.; Mounier, R.; Leclerc, J.; Treebak, J.T.; Pehmøller, C.; Sanz, N.; Sakakibara, I.; Saint-Amand, E.; Rimbaud, S.; et al. AMPK controls exercise endurance, mitochondrial oxidative capacity, and skeletal muscle integrity. FASEB J. 2014, 28, 3211–3224. [Google Scholar] [CrossRef]
- Macnaughton, L.S.; Wardle, S.L.; Witard, O.C.; McGlory, C.; Hamilton, D.L.; Jeromson, S.; Lawrence, C.E.; Wallis, G.A.; Tipton, K.D. The response of muscle protein synthesis following whole-body resistance exercise is greater following 40 g than 20 g of ingested whey protein. Physiol. Rep. 2016, 4, e12893. [Google Scholar] [CrossRef]
- Donges, C.E.; Burd, N.A.; Duffield, R.; Smith, G.C.; West, D.W.D.; Short, M.J.; MacKenzie, R.; Plank, L.D.; Shepherd, P.R.; Phillips, S.M.; et al. Concurrent resistance and aerobic exercise stimulates both myofibrillar and mitochondrial protein synthesis in sedentary middle-aged men. J. Appl. Physiol. 2012, 112, 1992–2001. [Google Scholar] [CrossRef]
- Camera, D.M.; West, D.W.D.; Phillips, S.M.; Rerecich, T.; Stellingwerff, T.; Hawley, J.A.; Coffey, V.G. Protein Ingestion Increases Myofibrillar Protein Synthesis after Concurrent Exercise. Med. Sci. Sports Exerc. 2015, 47, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M. Exercise and Amino Acid Anabolic Cell Signaling and the Regulation of Skeletal Muscle Mass. Nutrients 2012, 4, 740–758. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.; Zinchenko, A.; Suckling, C.; Smith, L.; Johnstone, J.; Henselmans, M. The short-term effect of high versus moderate protein intake on recovery after strength training in resistance-trained individuals. J. Int. Soc. Sports Nutr. 2017, 14, 44. [Google Scholar] [CrossRef]
- Tipton, K.D. Protein for adaptations to exercise training. Eur. J. Sport Sci. 2008, 8, 107–118. [Google Scholar] [CrossRef]
- Witard, O.C.; Wardle, S.L.; Macnaughton, L.S.; Hodgson, A.B.; Tipton, K.D. Protein Considerations for Optimising Skeletal Muscle Mass in Healthy Young and Older Adults. Nutrients 2016, 8, 181. [Google Scholar] [CrossRef] [PubMed]
- Tipton, K.D.; Sharp, C.P. The response of intracellular signaling and muscle-protein metabolism to nutrition and exercise. Eur. J. Sport Sci. 2005, 5, 107–121. [Google Scholar] [CrossRef]
- Tipton, K.D.; Wolfe, R.R. Protein and amino acids for athletes. J. Sports Sci. 2004, 22, 65–79. [Google Scholar] [CrossRef]
- Phillips, S.M.; Hartman, J.W.; Wilkinson, S.B. Dietary Protein to Support Anabolism with Resistance Exercise in Young Men. J. Am. Coll. Nutr. 2005, 24, 134S–139S. [Google Scholar] [CrossRef]
- Phillips, S.M.; Van Loon, L.J. Dietary protein for athletes: From requirements to optimum adaptation. J. Sports Sci. 2011, 29, S29–S38. [Google Scholar] [CrossRef]
- McLellan, T.M. Protein Supplementation for Military Personnel: A Review of the Mechanisms and Performance Outcomes. J. Nutr. 2013, 143, 1820S–1833S. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Methley, A.M.; Campbell, S.; Chew-Graham, C.; McNally, R.; Cheraghi-Sohi, S. PICO, PICOS and SPIDER: A comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv. Res. 2014, 14, 1–10. [Google Scholar] [CrossRef]
- Blacker, S.D.; Wilkinson, D.M.; Bilzon, J.L.; Rayson, M.P. Risk Factors for Training Injuries among British Army Recruits. Mil. Med. 2008, 173, 278–286. [Google Scholar] [CrossRef]
- Needleman, I.G. A guide to systematic reviews. J. Clin. Periodontol. 2002, 29, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Terwee, C.B.; Mokkink, L.B.; Knol, D.L.; Ostelo, R.W.J.G.; Bouter, L.M.; De Vet, H.C.W. Rating the methodological quality in systematic reviews of studies on measurement properties: A scoring system for the COSMIN checklist. Qual. Life Res. 2011, 21, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A power primer. Tutor. Quant. Methods Psychol. 2007, 3, 79. [Google Scholar] [CrossRef][Green Version]
- McAdam, J.S.; McGinnis, K.D.; Beck, D.T.; Haun, C.T.; Romero, M.A.; Mumford, P.W.; Roberson, P.A.; Young, K.C.; Lohse, K.R.; Lockwood, C.M.; et al. Effect of Whey Protein Supplementation on Physical Performance and Body Composition in Army Initial Entry Training Soldiers. Nutrients 2018, 10, 1248. [Google Scholar] [CrossRef]
- Eddens, L.; Browne, S.; Stevenson, E.J.; Sanderson, B.; Van Someren, K.; Howatson, G. The efficacy of protein supplementation during recovery from muscle-damaging concurrent exercise. Appl. Physiol. Nutr. Metab. 2017, 42, 716–724. [Google Scholar] [CrossRef]
- Crowe, M.J.; Weatherson, J.N.; Bowden, B.F. Effects of dietary leucine supplementation on exercise performance. Graefe’s Arch. Clin. Exp. Ophthalmol. 2005, 97, 664–672. [Google Scholar] [CrossRef]
- Forbes, S.C.; Bell, G.J. Whey protein isolate or concentrate combined with concurrent training does not augment performance, cardiorespiratory fitness, or strength adaptations. J. Sports Med. Phys. Fit. 2020, 60, 1–9. [Google Scholar] [CrossRef]
- Ormsbee, M.J.; Willingham, B.D.; Marchant, T.; Binkley, T.L.; Specker, B.L.; Vukovich, M.D. Protein Supplementation During a 6-Month Concurrent Training Program: Effect on Body Composition and Muscular Strength in Sedentary Individuals. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 619–628. [Google Scholar] [CrossRef]
- Taylor, L.W.; Wilborn, C.; Roberts, M.D.; White, A.J.P.; Dugan, M.K. Eight weeks of pre- and postexercise whey protein supplementation increases lean body mass and improves performance in Division III collegiate female basketball players. Appl. Physiol. Nutr. Metab. 2016, 41, 249–254. [Google Scholar] [CrossRef]
- Longland, T.M.; Oikawa, S.Y.; Mitchell, C.J.; Devries, M.C.; Phillips, S.M. Higher compared with lower dietary protein during an energy deficit combined with intense exercise promotes greater lean mass gain and fat mass loss: A randomized trial. Am. J. Clin. Nutr. 2016, 103, 738–746. [Google Scholar] [CrossRef]
- Walker, T.B.; Smith, J.; Herrera, M.; Lebegue, B.; Pinchak, A.; Fischer, J. The Influence of 8 Weeks of Whey-Protein and Leucine Supplementation on Physical and Cognitive Performance. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 409–417. [Google Scholar] [CrossRef]
- Jimenez-Flores, R.; Heick, J.; Davis, S.C.; Hall, K.G.; Schaffner, A. A Comparison of the Effects of a High Carbohydrate vs. a Higher Protein Milk Supplement Following Simulated Mountain Skirmishes. Mil. Med. 2012, 177, 723–731. [Google Scholar] [CrossRef]
- Blacker, S.D.; Williams, N.C.; Fallowfield, J.L.; Bilzon, J.L.; Willems, M.E. Carbohydrate vs protein supplementation for recovery of neuromuscular function following prolonged load carriage. J. Int. Soc. Sports Nutr. 2010, 7, 2. [Google Scholar] [CrossRef]
- Flakoll, P.J.; Judy, T.; Flinn, K.; Carr, C.; Flinn, S. Postexercise protein supplementation improves health and muscle soreness during basic military training in marine recruits. J. Appl. Physiol. 2004, 96, 951–956. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; McLellan, T.M.; Lieberman, H.R. The Effects of Protein Supplements on Muscle Mass, Strength, and Aerobic and Anaerobic Power in Healthy Adults: A Systematic Review. Sports Med. 2015, 45, 111–131. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; Garthe, I.; Phillips, S.M.; Philips, S.M. Dietary Protein for Training Adaptation and Body Composition Manipulation in Track and Field Athletes. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Kerksick, C.M.; Campbell, B.I.; Cribb, P.J.; Wells, S.D.; Skwiat, T.M.; Purpura, M.; Ziegenfuss, T.N.; Ferrando, A.A.; Arent, S.M.; et al. International Society of Sports Nutrition Position Stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2017, 14, 20. [Google Scholar] [CrossRef]
- Damas, F.; Phillips, S.M.; Libardi, C.A.; Vechin, F.C.; Lixandrão, M.E.; Jannig, P.R.; Costa, L.A.R.; Bacurau, A.V.; Snijders, T.; Parise, G.; et al. Resistance training-induced changes in integrated myofibrillar protein synthesis are related to hypertrophy only after attenuation of muscle damage. J. Physiol. 2016, 594, 5209–5222. [Google Scholar] [CrossRef]
- Pérez-Schindler, J.; Hamilton, D.L.; Moore, D.R.; Baar, K.; Philp, A. Nutritional strategies to support concurrent training. Eur. J. Sport Sci. 2015, 15, 41–52. [Google Scholar] [CrossRef]
- Areta, J.L.; Burke, L.M.; Ross, M.L.; Camera, D.M.; West, D.W.D.; Broad, E.M.; Jeacocke, N.A.; Moore, D.R.; Stellingwerff, T.; Phillips, S.M.; et al. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J. Physiol. 2013, 591, 2319–2331. [Google Scholar] [CrossRef]
- Mamerow, M.M.; Mettler, J.A.; English, K.L.; Casperson, S.L.; Arentson-Lantz, E.; Sheffield-Moore, M.; Layman, D.K.; Paddon-Jones, D. Dietary Protein Distribution Positively Influences 24-h Muscle Protein Synthesis in Healthy Adults. J. Nutr. 2014, 144, 876–880. [Google Scholar] [CrossRef]
- Trommelen, J.; Van Loon, L.J.C. Pre-Sleep Protein Ingestion to Improve the Skeletal Muscle Adaptive Response to Exercise Training. Nutrients 2016, 8, 763. [Google Scholar] [CrossRef]
- Antonio, J.; Candow, D.G.; Forbes, S.C.; Ormsbee, M.J.; Saracino, P.G.; Roberts, J. Effects of Dietary Protein on Body Composition in Exercising Individuals. Nutrients 2020, 12, 1890. [Google Scholar] [CrossRef]
- Reidy, P.T.; Rasmussen, B.B. Role of Ingested Amino Acids and Protein in the Promotion of Resistance Exercise–Induced Muscle Protein Anabolism. J. Nutr. 2016, 146, 155–183. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.E.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN exercise & sports nutrition review update: Research & recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 38. [Google Scholar] [CrossRef]
- Aragon, A.A.; Schoenfeld, B.J.; Wildman, R.; Kleiner, S.; VanDusseldorp, T.; Taylor, L.; Earnest, C.P.; Arciero, P.J.; Wilborn, C.; Kalman, D.S.; et al. International society of sports nutrition position stand: Diets and body composition. J. Int. Soc. Sports Nutr. 2017, 14, 1–19. [Google Scholar] [CrossRef]
- Kato, H.; Suzuki, K.; Bannai, M.; Moore, D.R. Protein Requirements Are Elevated in Endurance Athletes after Exercise as Determined by the Indicator Amino Acid Oxidation Method. PLoS ONE 2016, 11, e0157406. [Google Scholar] [CrossRef]
- Aguirre, N.; Baar, K. The Role of Amino Acids in Skeletal Muscle Adaptation to Exercise. Issues Complementary Feed. 2013, 76, 85–102. [Google Scholar] [CrossRef]
- Hulmi, J.J.; Kovanen, V.; Selänne, H.; Kraemer, W.J.; Häkkinen, K.; Mero, A.A. Acute and long-term effects of resistance exercise with or without protein ingestion on muscle hypertrophy and gene expression. Amino Acids 2008, 37, 297–308. [Google Scholar] [CrossRef]
- Huecker, M.; Sarav, M.; Pearlman, M.; Laster, J. Protein Supplementation in Sport: Source, Timing, and Intended Benefits. Curr. Nutr. Rep. 2019, 8, 382–396. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, A.; Costello, J.T. Realising the Potential of Urine and Saliva as Diagnostic Tools in Sport and Exercise Medicine. Sports Med. 2017, 47, 11–31. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.C.; Fragala, M.S.; Kavouras, S.A.; Queen, R.M.; Pryor, J.L.; Casa, D.J. Biomarkers in Sports and Exercise: Tracking Health, Performance, and Recovery in Athletes. J. Strength Cond. Res. 2017, 31, 2920–2937. [Google Scholar] [CrossRef] [PubMed]
- Markus, I.; Constantini, K.; Hoffman, J.R.; Bartolomei, S.; Gepner, Y. Exercise-induced muscle damage: Mechanism, assessment and nutritional factors to accelerate recovery. Graefe’s Arch. Clin. Exp. Ophthalmol. 2021, 121, 969–992. [Google Scholar] [CrossRef]
- Clarkson, P.M.; Hubal, M.J. Exercise-Induced Muscle Damage in Humans. Am. J. Phys. Med. Rehabil. 2002, 81, S52–S69. [Google Scholar] [CrossRef]
- O’Leary, T.J.; Saunders, S.C.; McGuire, S.J.; Izard, R.M. Sex differences in neuromuscular fatigability in response to load carriage in the field in British Army recruits. J. Sci. Med. Sport 2018, 21, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.B.; Aragon, A.A.; Krieger, J.W. The effect of protein timing on muscle strength and hypertrophy: A meta-analysis. J. Int. Soc. Sports Nutr. 2013, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Moran, D.S.; Heled, Y.; Arbel, Y.; Israeli, E.; Finestone, A.S.; Evans, R.K.; Yanovich, R. Dietary intake and stress fractures among elite male combat recruits. J. Int. Soc. Sports Nutr. 2012, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Wentz, L.; Liu, P.-Y.; Haymes, E.; Ilich, J.Z. Females Have a Greater Incidence of Stress Fractures Than Males in Both Military and Athletic Populations: A Systemic Review. Mil. Med. 2011, 176, 420–430. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, T.J.; Walsh, N.P.; Casey, A.; Izard, R.M.; Tang, J.C.Y.; Fraser, W.D.; Greeves, J.P. Supplementary Energy Increases Bone Formation during Arduous Military Training. Med. Sci. Sports Exerc. 2021, 53, 394–403. [Google Scholar] [CrossRef]
- Sale, C.; Elliott-Sale, K.J. Nutrition and Athlete Bone Health. Sports Med. 2019, 49, 139–151. [Google Scholar] [CrossRef]
- O’Leary, T.J.; Wardle, S.L.; Rawcliffe, A.J.; Chapman, S.; Mole, J.; Greeves, J.P. Understanding the musculoskeletal injury risk of women in combat: The effect of infantry training and sex on musculoskeletal injury incidence during British Army basic training. BMJ Mil. Health 2020, 1–5. [Google Scholar] [CrossRef]
| Study | Reviewer 1 | Reviewer 2 | Mean | Included |
|---|---|---|---|---|
| McAdam et al. [51] | 4.00 | 3.70 | 3.85 | Y |
| Eddens et al. [52] | 3.60 | 3.30 | 3.45 | Y |
| Crowe, Weatherson and Bowden [53] | 4.00 | 3.40 | 3.70 | Y |
| Forbes and Bell [54] | 3.80 | 3.20 | 3.50 | Y |
| Ormsbee et al. [55] | 3.80 | 3.40 | 3.60 | Y |
| Taylor et al. [56] | 4.00 | 4.00 | 4.00 | Y |
| Longland et al. [57] | 4.00 | 3.50 | 3.75 | Y |
| Walker et al. [58] | 4.00 | 3.90 | 3.95 | Y |
| Jimenez-Flores et al. [59] | 3.30 | 2.90 | 3.10 | Y |
| Blacker et al. [60] | 3.40 | 3.00 | 3.20 | Y |
| Flakoll et al. [61] | 4.00 | 3.80 | 3.90 | Y |
| Study | Sample | Age | Total Protein Intake | Intervention | Supplement Type & Dose |
|---|---|---|---|---|---|
| McAdam et al. [51] | 69 male U.S. Army recruits. | 19 ± 1 years | 2.8 ± 0.5 & 1.6 ± 0.4 g·kg−1·d−1 in PRO and PLA. | 8-week U.S. Army Initial Entry Training. | 38.6 g WP or isocaloric PLA post-exercise in AM & prior to sleep. |
| Crowe, Weatherson and Bowden [53] | 10 male, 3 female trained canoeists. | 32 ± 2 years | 0.85 ± 0.06 & 0.85 ± 0.05 g·kg−1·day−1 in PRO & PLA. | 6-weeks endurance & resistance training. | 45 mg·kg−1·day−1 leucine or PLA. |
| Ormsbee et al. [55] | 26 sedentary men and 25 sedentary women. | 21± 1 years & 20 ± 1 years in PRO & PLA. | 2.2 ± 0.1 & 1.1 ± 0.1 g·kg−1·day−1 for the PRO & PLA groups. | 6-month endurance & resistance training. | 42 g PRO or isocaloric PLA consumed immediately post-exercise & 8–12 h later. |
| Taylor et al. [56] | 16 female intermittent sport athletes. | 20 ± 2 years | Not measured. | 8-week endurance & resistance training. | 24 g pre-and-post-exercise. |
| Longland et al. [57] | 40 recreationally active men. | 23 ± 2 years | 2.4 & 1.2 g·kg−1·day−1 for the PRO & PLA groups. | 4-weeks endurance & resistance training with an energy deficit | 50 g WP or CHO drink given post-exercise to PRO & PLA groups. |
| Walker et al. [56] | 30 U.S. Air force men. | 26 ± 9 years | Not measured. | 8-week U.S. Air force training. | 20 g WP or isocaloric PLA post-exercise. |
| Forbes and Bell [54] | 15 healthy women & 16 men. | Women: 27 ± 4 years, men: 26 ± 3 years | PLA (men = 1.4 ± 0.4 g·kg−1·day−1, women= 1.2 ± 0.2 g·kg−1·day−1), PRO (men = 3.8 ± 0.4 g·kg−1·day−1, women= 3.2 ± 0.3 g·kg−1·day−1). | 6-weeks endurance & resistance training. | 2.0 and 2.4 g·kg−1·day−1 WP for women & men. |
| Study | Sample | Age | Total Protein Intake | Intervention | Supplement Type & Dose |
|---|---|---|---|---|---|
| Eddens et al. [52] | 24 male cyclists. | PRO = 27 ± 3 years; PLA = 28 ± 5 years; CHO = 26 ± 5 years | PRO = 1.2 ± 0.6 g·kg−1·day−1; PLA = 1.2 ± 0.6 g·kg−1·day−1; CHO = 1.2 ± 0.7 g·kg−1·day−1 | Single concurrent exercise event (high-intensity cycling followed by 100 box jumps). | 20 g WP, isocaloric CHO or low-calorific PLA post-exercise. |
| Jimenez-Flores et al. [59] | 33 healthy men and 2 healthy women. | 21 ± 1 years & 21 ± 1 years in PLA & PRO groups | Not measured. | 4-day loaded (13.2–26.4 kg) mountain skirmish. | 25 g protein bar or isocaloric CHO bar post-exercise. |
| Blacker et al. [60] | 10 healthy men. | 28 ± 9 years | * 0.9 ± 0.3 g·kg−1·day−1, in the PLA, CHO & PRO. | 3 days post-load (25 kg) carriage exercise. | 36 g PRO, 32 g CHO or low-calorie PLA post-exercise. |
| Flakoll et al. [61] | 387 male U.S. Marine recruits. | 19 ± 1 years | Not measured. | Single day loaded march hike. | PLA = 0 g CHO, 0 g PRO, 0 g fat; CON = 0 g PRO, 8 g CHO and 3 g fat; PRO = 10 g PRO, 8 g CHO and 3 g fat. Participants who weighed <81.8 kg received one portion and those weighing >81.8 kg received two portions post-exercise. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chapman, S.; Chung, H.C.; Rawcliffe, A.J.; Izard, R.; Smith, L.; Roberts, J.D. Does Protein Supplementation Support Adaptations to Arduous Concurrent Exercise Training? A Systematic Review and Meta-Analysis with Military Based Applications. Nutrients 2021, 13, 1416. https://doi.org/10.3390/nu13051416
Chapman S, Chung HC, Rawcliffe AJ, Izard R, Smith L, Roberts JD. Does Protein Supplementation Support Adaptations to Arduous Concurrent Exercise Training? A Systematic Review and Meta-Analysis with Military Based Applications. Nutrients. 2021; 13(5):1416. https://doi.org/10.3390/nu13051416
Chicago/Turabian StyleChapman, Shaun, Henry C. Chung, Alex J. Rawcliffe, Rachel Izard, Lee Smith, and Justin D. Roberts. 2021. "Does Protein Supplementation Support Adaptations to Arduous Concurrent Exercise Training? A Systematic Review and Meta-Analysis with Military Based Applications" Nutrients 13, no. 5: 1416. https://doi.org/10.3390/nu13051416
APA StyleChapman, S., Chung, H. C., Rawcliffe, A. J., Izard, R., Smith, L., & Roberts, J. D. (2021). Does Protein Supplementation Support Adaptations to Arduous Concurrent Exercise Training? A Systematic Review and Meta-Analysis with Military Based Applications. Nutrients, 13(5), 1416. https://doi.org/10.3390/nu13051416









