Role of Probiotics in Modulating Human Gut Microbiota Populations and Activities in Patients with Colorectal Cancer—A Systematic Review of Clinical Trials
Abstract
1. Introduction
2. Materials and Methods
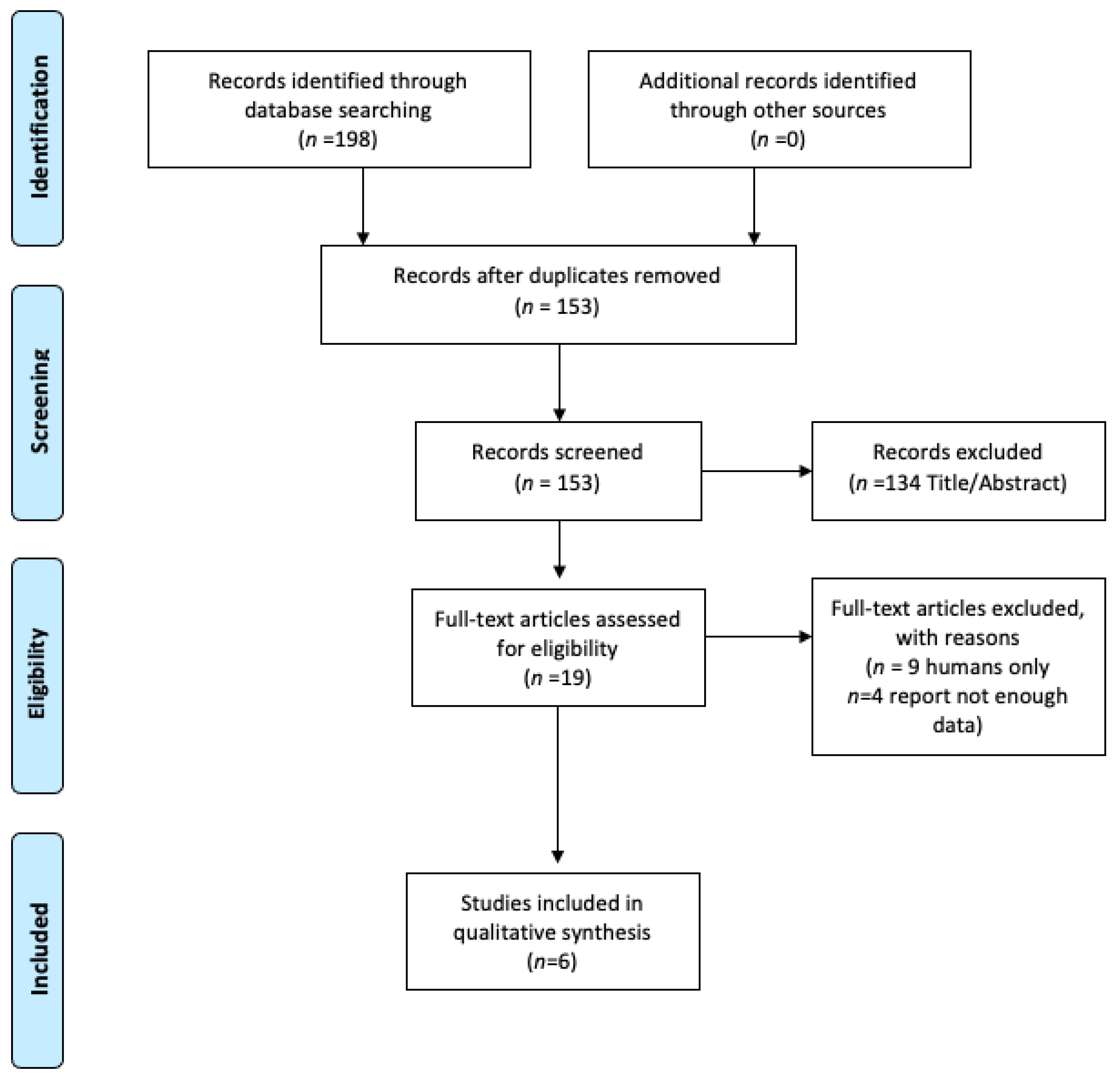
2.1. Search Strategy and Study Selection
2.2. Data Extraction
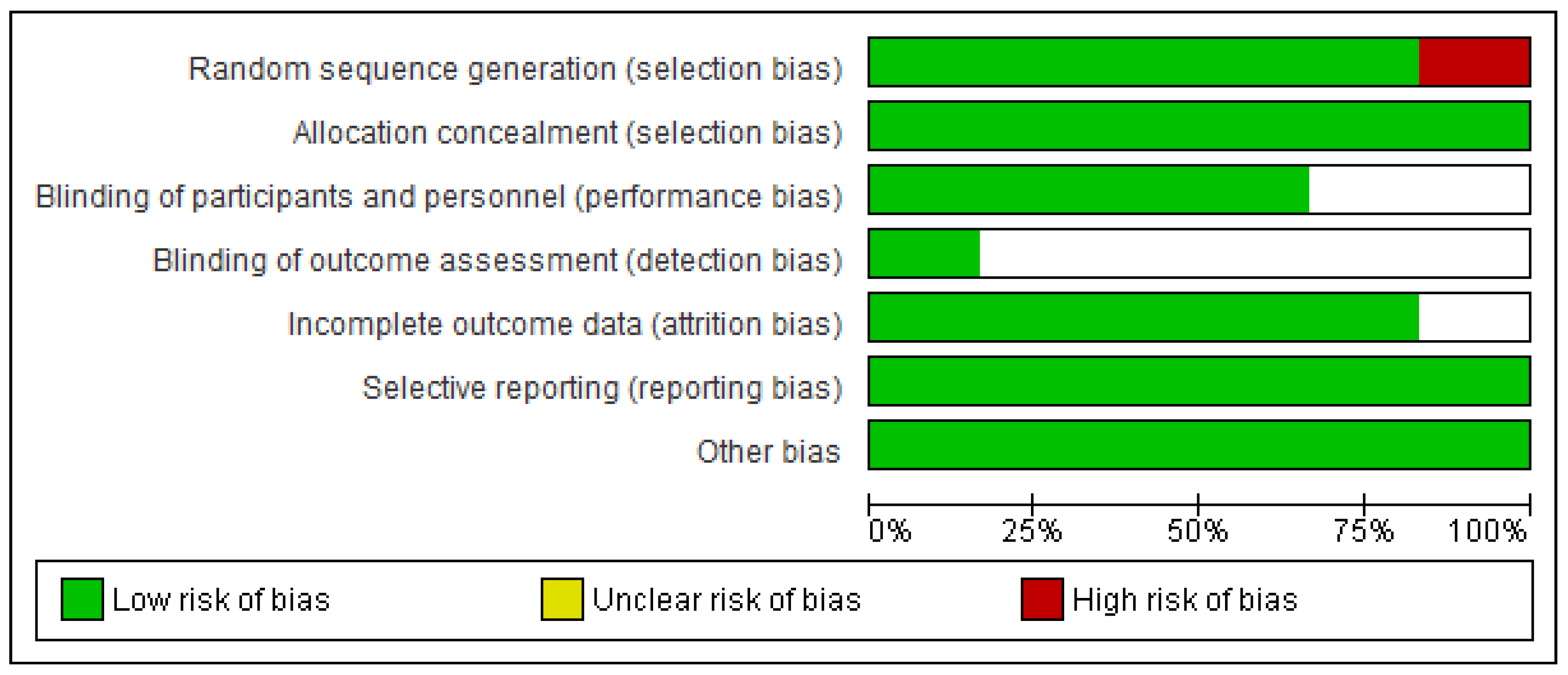
2.3. Risk of Bias Tool
3. Results
3.1. Study Selection and Characteristics of Study Population
3.2. Effect of Probiotics on Changes in Gut Microbiota and Postoperative Outcome
3.3. Risk of Bias Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Goding Sauer, A.; Fedewa, S.A.; Butterly, L.F.; Anderson, J.C.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal Cancer Statistics, 2020. CA Cancer J. Clin. 2020, 70, 145–164. [Google Scholar] [CrossRef]
- Sinicrope, F.A. DNA Mismatch Repair and Adjuvant Chemotherapy in Sporadic Colon Cancer. Nat. Rev. Clin. Oncol. 2010, 7, 174–177. [Google Scholar] [CrossRef]
- Schell, M.J.; Yang, M.; Teer, J.K.; Lo, F.Y.; Madan, A.; Coppola, D.; Monteiro, A.N.A.; Nebozhyn, M.V.; Yue, B.; Loboda, A.; et al. A Multigene Mutation Classification of 468 Colorectal Cancers Reveals a Prognostic Role for APC. Nat. Commun. 2016, 7, 11743. [Google Scholar] [CrossRef] [PubMed]
- Clinton, S.K.; Giovannucci, E.L.; Hursting, S.D. The World Cancer Research Fund/American Institute for Cancer Research Third Expert Report on Diet, Nutrition, Physical Activity, and Cancer: Impact and Future Directions. J. Nutr. 2020, 150, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Thun, M.; Linet, M.S.; Cerhan, J.R.; Haiman, C.A.; Schottenfeld, D. (Eds.) Cancer Epidemiology and Prevention; Oxford University Press: New York, NY, USA, 2017; Volume 1, ISBN 978-0-19-023866-7. [Google Scholar]
- Song, M.; Garrett, W.S.; Chan, A.T. Nutrients, Foods, and Colorectal Cancer Prevention. Gastroenterology 2015, 148, 1244–1260.e16. [Google Scholar] [CrossRef]
- Sears, C.L.; Garrett, W.S. Microbes, Microbiota, and Colon Cancer. Cell Host Microbe 2014, 15, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Tjalsma, H.; Boleij, A.; Marchesi, J.R.; Dutilh, B.E. A Bacterial Driver–Passenger Model for Colorectal Cancer: Beyond the Usual Suspects. Nat. Rev. Microbiol. 2012, 10, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, A.; Totino, V.; Cacciotti, F.; Iebba, V.; Neroni, B.; Bonfiglio, G.; Trancassini, M.; Passariello, C.; Pantanella, F.; Schippa, S. Rebuilding the Gut Microbiota Ecosystem. Int. J. Environ. Res. Public Health 2018, 15, 1679. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Interactions between the Microbiota and Pathogenic Bacteria in the Gut. Available online: https://pubmed.ncbi.nlm.nih.gov/27383983/ (accessed on 9 August 2020).
- Gensollen, T.; Iyer, S.S.; Kasper, D.L.; Blumberg, R.S. How Colonization by Microbiota in Early Life Shapes the Immune System. Science 2016, 352, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Natividad, J.M.M.; Verdu, E.F. Modulation of Intestinal Barrier by Intestinal Microbiota: Pathological and Therapeutic Implications. Pharmacol. Res. 2013, 69, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Mego, M.; Chovanec, J.; Vochyanova-Andrezalova, I.; Konkolovsky, P.; Mikulova, M.; Reckova, M.; Miskovska, V.; Bystricky, B.; Beniak, J.; Medvecova, L.; et al. Prevention of Irinotecan Induced Diarrhea by Probiotics: A Randomized Double Blind, Placebo Controlled Pilot Study. Complement. Ther. Med. 2015, 23, 356–362. [Google Scholar] [CrossRef]
- Delia, P. Use of Probiotics for Prevention of Radiation-Induced Diarrhea. World J. Gastroenterol. 2007, 13, 912. [Google Scholar] [CrossRef]
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, Prebiotics and Synbiotics—A Review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert Consensus Document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.M.; Finlay, B.B. Gut Microbiota in Health and Disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Healthcare Interventions: Explanation and Elaboration. BMJ 2009, 339. [Google Scholar] [CrossRef]
- Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation. Available online: https://www.ncbi.nlm.nih.gov/nlmcatalog/101617803 (accessed on 1 September 2020).
- Andersson, H.; Asp, N.-G.; Bruce, Å.; Roos, S.; Wadström, T.; Wold, A.E. Health Effects of Probiotics and Prebiotics A Literature Review on Human Studies. Näringsforskning 2001, 45, 58–75. [Google Scholar] [CrossRef]
- Shuster, J.J. Review: Cochrane Handbook for Systematic Reviews for Interventions, Version 5.1.0, Published 3/2011. Julian P.T. Higgins and Sally Green, Editors. Res. Synth. Methods 2011, 2, 126–130. [Google Scholar] [CrossRef]
- Mizuta, M.; Endo, I.; Yamamoto, S.; Inokawa, H.; Kubo, M.; Udaka, T.; Sogabe, O.; Maeda, H.; Shirakawa, K.; Okazaki, E.; et al. Perioperative Supplementation with Bifidobacteria Improves Postoperative Nutritional Recovery, Inflammatory Response, and Fecal Microbiota in Patients Undergoing Colorectal Surgery: A Prospective, Randomized Clinical Trial. Biosci. Microbiota Food Health 2016, 35, 77–87. [Google Scholar] [CrossRef]
- Aisu, N.; Tanimura, S.; Yamashita, Y.; Yamashita, K.; Maki, K.; Yoshida, Y.; Sasaki, T.; Takeno, S.; Hoshino, S. Impact of Perioperative Probiotic Treatment for Surgical Site Infections in Patients with Colorectal Cancer. Exp. Ther. Med. 2015, 10, 966–972. [Google Scholar] [CrossRef]
- Liu, Z.; Qin, H.; Yang, Z.; Xia, Y.; Liu, W.; Yang, J.; Jiang, Y.; Zhang, H.; Yang, Z.; Wang, Y.; et al. Randomised Clinical Trial: The Effects of Perioperative Probiotic Treatment on Barrier Function and Post-Operative Infectious Complications in Colorectal Cancer Surgery—A Double-Blind Study: Randomised Clinical Trial: Perioperative Probiotics on Colon Cancer. Aliment. Pharmacol. Ther. 2011, 33, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Gianotti, L. A Randomized Double-Blind Trial on Perioperative Administration of Probiotics in Colorectal Cancer Patients. World J. Gastroenterol. 2010, 16, 167. [Google Scholar] [CrossRef]
- Hibberd, A.A.; Lyra, A.; Ouwehand, A.C.; Rolny, P.; Lindegren, H.; Cedgård, L.; Wettergren, Y. Intestinal Microbiota Is Altered in Patients with Colon Cancer and Modified by Probiotic Intervention. BMJ Open Gastroenterol. 2017, 4, e000145. [Google Scholar] [CrossRef] [PubMed]
- Rafter, J.; Bennett, M.; Caderni, G.; Clune, Y.; Hughes, R.; Karlsson, P.C.; Klinder, A.; O’Riordan, M.; O’Sullivan, G.C.; Pool-Zobel, B.; et al. Dietary Synbiotics Reduce Cancer Risk Factors in Polypectomized and Colon Cancer Patients. Am. J. Clin. Nutr. 2007, 85, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Tojo, R. Intestinal Microbiota in Health and Disease: Role of Bifidobacteria in Gut Homeostasis. World J. Gastroenterol. 2014, 20, 15163. [Google Scholar] [CrossRef] [PubMed]
- Mańkowska-Wierzbicka, D.; Stelmach-Mardas, M.; Gabryel, M.; Tomczak, H.; Skrzypczak-Zielińska, M.; Zakerska-Banaszak, O.; Sowińska, A.; Mahadea, D.; Baturo, A.; Wolko, Ł.; et al. The Effectiveness of Multi-Session FMT Treatment in Active Ulcerative Colitis Patients: A Pilot Study. Biomedicines 2020, 8, 268. [Google Scholar] [CrossRef]
- Nowak, A.; Paliwoda, A.; Błasiak, J. Anti-Proliferative, pro-Apoptotic and Anti-Oxidative Activity of Lactobacillus and Bifidobacterium Strains: A Review of Mechanisms and Therapeutic Perspectives. Crit. Rev. Food Sci. Nutr. 2019, 59, 3456–3467. [Google Scholar] [CrossRef]
- Moore, W.E.; Moore, L.H. Intestinal Floras of Populations That Have a High Risk of Colon Cancer. Appl. Environ. Microbiol. 1995, 61, 3202–3207. [Google Scholar] [CrossRef]
- Bachrach, G.; Ianculovici, C.; Naor, R.; Weiss, E.I. Fluorescence Based Measurements of Fusobacterium Nucleatum Coaggregation and of Fusobacterial Attachment to Mammalian Cells. FEMS Microbiol. Lett. 2005, 248, 235–240. [Google Scholar] [CrossRef]
- Flynn, K.J.; Baxter, N.T.; Schloss, P.D. Metabolic and Community Synergy of Oral Bacteria in Colorectal Cancer. Msphere 2016, 1, e00102-16. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Guo, B.; Gao, R.; Zhu, Q.; Qin, H. Microbiota Disbiosis Is Associated with Colorectal Cancer. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- He, D.; Wang, H.-Y.; Feng, J.-Y.; Zhang, M.-M.; Zhou, Y.; Wu, X.-T. Use of Pro-/Synbiotics as Prophylaxis in Patients Undergoing Colorectal Resection for Cancer: A Meta-Analysis of Randomized Controlled Trials. Clin. Res. Hepatol. Gastroenterol. 2013, 37, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Huycke, M.M.; Abrams, V.; Moore, D.R. Enterococcus Faecalis Produces Extracellular Superoxide and Hydrogen Peroxide That Damages Colonic Epithelial Cell DNA. Carcinogenesis 2002, 23, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Guo, B.; Gao, R.; Zhu, Q.; Wu, W.; Qin, H. Probiotics Modify Human Intestinal Mucosa-Associated Microbiota in Patients with Colorectal Cancer. Mol. Med. Rep. 2015, 12, 6119–6127. [Google Scholar] [CrossRef]
- Zhang, M.; Fan, X.; Fang, B.; Zhu, C.; Zhu, J.; Ren, F. Effects of Lactobacillus Salivarius Ren on Cancer Prevention and Intestinal Microbiota in 1, 2-Dimethylhydrazine-Induced Rat Model. J. Microbiol. 2015, 53, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.M.; Versalovic, J. Probiotics-Host Communication: Modulation of Signaling Pathways in the Intestine. Gut Microbes 2010, 1, 148–163. [Google Scholar] [CrossRef] [PubMed]
- Rautava, S.; Arvilommi, H.; Isolauri, E. Specific Probiotics in Enhancing Maturation of IgA Responses in Formula-Fed Infants. Pediatr. Res. 2006, 60, 221–224. [Google Scholar] [CrossRef]
- Takeda, K.; Suzuki, T.; Shimada, S.-I.; Shida, K.; Nanno, M.; Okumura, K. Interleukin-12 Is Involved in the Enhancement of Human Natural Killer Cell Activity by Lactobacillus Casei Shirota. Clin. Exp. Immunol. 2006, 146, 109–115. [Google Scholar] [CrossRef]
- Oh, N.S.; Joung, J.Y.; Lee, J.Y.; Kim, Y. Probiotic and Anti-Inflammatory Potential of Lactobacillus Rhamnosus 4B15 and Lactobacillus Gasseri 4M13 Isolated from Infant Feces. PLoS ONE 2018, 13, e0192021. [Google Scholar] [CrossRef]
- Tien, M.-T.; Girardin, S.E.; Regnault, B.; Le Bourhis, L.; Dillies, M.-A.; Coppée, J.-Y.; Bourdet-Sicard, R.; Sansonetti, P.J.; Pédron, T. Anti-Inflammatory Effect of Lactobacillus Casei on Shigella-Infected Human Intestinal Epithelial Cells. J. Immunol. 2006, 176, 1228–1237. [Google Scholar] [CrossRef] [PubMed]
- Bai, A.-P.; Ouyang, Q.; Xiao, X.-R.; Li, S.-F. Probiotics Modulate Inflammatory Cytokine Secretion from Inflamed Mucosa in Active Ulcerative Colitis: PROBIOTICS MODULATE INFLAMMATORY CYTOKINE SECRETION. Int. J. Clin. Pract. 2006, 60, 284–288. [Google Scholar] [CrossRef]
- Resta–Lenert, S.; Barrett, K.E. Probiotics and Commensals Reverse TNF-α– and IFN-γ–Induced Dysfunction in Human Intestinal Epithelial Cells. Gastroenterology 2006, 130, 731–746. [Google Scholar] [CrossRef] [PubMed]
- Reid, G. The Scientific Basis for Probiotic Strains OfLactobacillus. Appl. Environ. Microbiol. 1999, 65, 3763–3766. [Google Scholar] [CrossRef]
- Johnson, D.A.; Barkun, A.N.; Cohen, L.B.; Dominitz, J.A.; Kaltenbach, T.; Martel, M.; Robertson, D.J.; Boland, C.R.; Giardello, F.M.; Lieberman, D.A.; et al. Optimizing Adequacy of Bowel Cleansing for Colonoscopy: Recommendations From the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology 2014, 147, 903–924. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.K.; Said, S.; Rajandram, R.; Wang, Z.; Roslani, A.C.; Chin, K.F. Pre-Surgical Administration of Microbial Cell Preparation in Colorectal Cancer Patients: A Randomized Controlled Trial. World J. Surg. 2016, 40, 1985–1992. [Google Scholar] [CrossRef] [PubMed]
- Pitsouni, E.; Alexiou, V.; Saridakis, V.; Peppas, G.; Falagas, M.E. Does the Use of Probiotics/Synbiotics Prevent Postoperative Infections in Patients Undergoing Abdominal Surgery? A Meta-Analysis of Randomized Controlled Trials. Eur. J. Clin. Pharmacol. 2009, 65, 561–570. [Google Scholar] [CrossRef]
- Zhang, J.-W.; Du, P.; Yang, B.-R.; Gao, J.; Fang, W.-J.; Ying, C.-M. Preoperative Probiotics Decrease Postoperative Infectious Complications of Colorectal Cancer. Am. J. Med. Sci. 2012, 343, 199–205. [Google Scholar] [CrossRef]
- Yang, Y.; Xia, Y.; Chen, H.; Hong, L.; Feng, J.; Yang, J.; Yang, Z.; Shi, C.; Wu, W.; Gao, R.; et al. The Effect of Perioperative Probiotics Treatment for Colorectal Cancer: Short-Term Outcomes of a Randomized Controlled Trial. Oncotarget 2016, 7, 8432–8440. [Google Scholar] [CrossRef]
- Okazaki, M.; Matsukuma, S.; Suto, R.; Miyazaki, K.; Hidaka, M.; Matsuo, M.; Noshima, S.; Zempo, N.; Asahara, T.; Nomoto, K. Perioperative Synbiotic Therapy in Elderly Patients Undergoing Gastroenterological Surgery: A Prospective, Randomized Control Trial. Nutrition 2013, 29, 1224–1230. [Google Scholar] [CrossRef]
- Vizoso Pinto, M.G.; Schuster, T.; Briviba, K.; Watzl, B.; Holzapfel, W.H.; Franz, C.M.A.P. Adhesive and Chemokine Stimulatory Properties of Potentially Probiotic Lactobacillus Strains. J. Food Prot. 2007, 70, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Zaharuddin, L.; Mokhtar, N.M.; Muhammad Nawawi, K.N.; Raja Ali, R.A. A Randomized Double-Blind Placebo-Controlled Trial of Probiotics in Post-Surgical Colorectal Cancer. BMC Gastroenterol. 2019, 19, 131. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Wang, C.; Ye, L.; Yang, W.; Huang, H.; Meng, F.; Shi, S.; Ding, Z. Anti-Tumour Immune Effect of Oral Administration of Lactobacillus Plantarum to CT26 Tumour-Bearing Mice. J. Biosci. 2015, 40, 269–279. [Google Scholar] [CrossRef] [PubMed]
| Source, Year | Trial Type | Sample Size (n) | Age (years) | Sex (M/F n) | BMI (kg/cm2) | Stage | Location | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control Group | Probiotic Group | Control Group | Probiotic Group | Control Group | Probiotic Group | Control Group | Probiotic Group | Control Group | Probiotic Group | Control Group | Probiotic Group | |||||||
| Mizuta et al., 2016 | PRCL | n = 29 | n = 31 | 71.2 ± 9.5 | 68.9 ± 10.4 | 15/14 | 20/11 | 24.1 ± 3.4 | 22.4 ± 3.7 | n/A | n/A | Colon 12 Rectum 13 Others 4 *** | Colon 11 Rectum 19 Others 0 | |||||
| Aisu et al., 2014 | RCT | n = 81 | n = 75 | 69.1 ± 11.3 | 68.0 ± 13.8 | 44/37 | 47/28 | 23.3 ± 3.8 | 21.7 ± 2.7 | I 29 II 32 IIIA 11 IIIB 3 IV 6 * | I 31 II 16 IIIA 10 IIIB 3 IV 8 * | Colon 3 Ascending colon 8 Transverse colon 6 Descending colon 3 Sigmoid colon 25 Rectum 22 | Colon 5 Ascending colon 14 Transverse colon 5 Descending colon 0 Sigmoid colon 16 Rectum 8 | |||||
| Hibberd et al., 2017 | RCT | n = 21 | n = 15 | 63 (55–73) | 77 (68–75) | 4/17 | 6/9 | n/A | 24.1 (22.5–24.8) | n/A | I 2 II 6 III 7 * | n/A | n/A | |||||
| Liu et al., 2010 | RDBT | n = 50 | n = 50 | 65.7 ± 9.9 | 65.3 ± 11.0 | 31/19 | 28/22 | 22.6 ± 2.0 | 22.8 ± 1.8 | A 12 B 29 C 9 ** | A 11 B 30 C 9 ** | Transverse Colon 8 Descending Colon 10 Sigmoid colon 21 Rectum 11 | Transverse Colon 7 Descending Colon 5 Sigmoid colon 25 Rectum 13 | |||||
| Gianotti et al., 2010 | RDBT | n = 10 | Low dose | High dose | 63.3 ± 10.2 | Low dose | High dose | 7/3 | Low dose | High dose | 25.6 ± 2.6 | Low dose | High dose | n/A | n/A | Left colon 4 Right colon 3 Rectum 3 | Low dose | High dose |
| n = 11 | n = 10 | 64.7 ± 4.8 | 62.7 ± 7.8 | 8/3 | 7/3 | 26.5 ± 4.1 | 24.4 ± 3.7 | Left colon 6 Right colon 2 Rectum 3 | Left colon 5 Right colon 2 Rectum 3 | |||||||||
| Rafter et al., 2007 | RDBPCT | n = 40 | n = 34 | 57.0 ± 9.75 | 61.1 ± 5.55 | 22/18 | 21/13 | n/A | n/A | n/A | n/A | n/A | n/A | |||||
| Scheme | Probiotic Intervention Dose (g) | Duration (Days) | Key Results |
|---|---|---|---|
| Mizuta et al., 2016 | 2 g of Bifidobacterium longum BB536 powder (approximately 5 × 1010 CFU/2 g) | 21–28 | ↑ anti-inflammatory response(↓ high sensitive C-reactive proteins, ↑ postoperative levels of erythrocytes, hemoglobin, lymphocytes, total protein, and albumin) ↓ duration of hospital stay |
| Aisu et al., 2014 | 2 mg Enterococcus faecalis T110, 0.01 g Clostridium butyricum TO-A and 0.01 g Bacillus mesentericus TO-A 6 × 109 CFU/d | 15 | ↓ incidence of postoperative complications (↓ time of flatus, ↓ time of meal intake, ↓ superficial incisional infections |
| Hibberd et al., 2017 | 1.4 × 101⁰ CFUs Bifidobacterium lactis Bl-04 (ATCC SD5219), 7 × 10⁹ CFUs Lactobacillus acidophilus NCFM (ATCC 700396) and 0.63 g inulin. | 8–78 | ↑ anti-inflammatory response ↑ microbial diversity: α- diversity and β-diversity |
| Liu et al., 2010 | Lactobacillus plantarum (CGMCC No. 1258, cell count ≥ 1011 CFU/g), Lactobacillus acidophilus (LA-11, cell count ≥ 7.0 × 101⁰ CFU/g) and Bifidobacterium longum (BL-88, cell count ≥ 5.0 × 101⁰ CFU⁄ g) | 16 | ↓ incidence of postoperative complications (↓ abdominal cramping, ↓ abdominal distention, ↓ duration of pyrexia ↓ time to first defecation) ↓ incidence of diarrhea ↑ microbial diversity: α- diversity and β-diversity |
| Gianotti et al., 2010 | 2 × 10⁷ CFU/d of a mixture of Bifidobacterium longum (BB536) and Lactobacillus johnsonii (La1) | 6 | ↑ anti-inflammatory response (↑ CD3, CD4, CD8, dendritic phenotypes CD83-123, ↓ CD83-HLA DR, CD83-11c) |
| Rafter et al., 2007 | Oligofructose enriched inulin (SYN1) and Lactobacillus rhamnosus GG (LGG) and Bifidobacterium lactis Bb12 (BB12), 12 g SYN1 together with the probiotic capsule > log10 CFU/g | 42 | ↑ anti-inflammatory response (↑ interferon γ, ↓ interleukin (IL) 2) ↓ proliferation rate of colorectal cells |
| Source, Year | Bacteria | Effect of Probiotic | Probiotic Group (Mean ± SD) | Control Group (Mean ± SD) | Method Used for Bacterial DNA Isolation | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phylum | Genus | Before | After | Before | After | |||||
| Mizuta et al., 2016 | Actinobacteria | n/A | Increase | 0.24–1.90 | 0.36–3.09 *** | 0.32–4.89 | 0.21–2.60 | PCR amplificationof the V3-V4 region of bacterial 16S rRNA genes obtained from fecal samples | ||
| Bacteroidetes | n/A | No change | 18.88–32.89 | 24.76–32.87 | 18.32–32.01 | 27.17–40.60 *** | ||||
| Firmicutes | n/A | Decrease | 52.34–72.98 | 48.46–64.15 *** | 57.18–75.96 | 46.77–64.24 | ||||
| Proteobacteria | n/A | No change | 1.54–5.06 | 2.27–9.75 *** | 1.50–2.16 | 2.90–5.84 | ||||
| Aisu et al., 2014 | Actinobacteria | Bifidobacterium * | Increase | 4.6% | 9.1% *** | 7% | 5.8% | PCR amplification of the 16S rDNA genes obtained from fecal samples | ||
| Hibberd et al., 2017 | Firmicutes Firmicutes | Eubacterium * Peptostreptococcus * | Increase Increase | n/A 2.1 ± 2.6 | 2.9 ± 2.7 *** 0.04 ± 0.06 *** | n/A 0.00 ± 0.00 | 0.86 ± 1.8 0.42 ± 0.71 *** | PCR amplification of the V4 variable region of the 16S rRNA gene obtained from mucosa and tumor tissue as well as from fecal samples | ||
| Fusobacteria Bacteroidetes | Fusobacterium ** Porphyromonas ** | Increase Increase | 7.6 ± 7.8 n/A | 0.03 ± 0.05 *** 0.00 ± 0.00 | 0.23 ± 0.60 n/A | 0.81 ± 0.87 0.43 ± 0.56 *** | ||||
| Faecalibacterium Firmicutes Firmicutes Actinobacteria | n/A | Decrease | n/A | 6.5 ± 2.0 *** | n/A | 3.2 ± 2.6 | ||||
| Clostridium | Increase | 3.1 ± 2.6 | 8.5 ± 4.1 *** | 1.6 ± 1.6 | 3.5 ± 3.1 | |||||
| Erysipelothrix | Increase | n/A | 1.3 ± 1.0 *** | n/A | 0.42 ± 0.59 | |||||
| Coriobacterium | Decrease | 0.30 ± 0.25 | 1.3 ± 0.75 *** | 0.25 ± 0.46 | 0.49 ± 0.46 | |||||
| Liu et al., 2010 | Actinobacteria Firmicutes Firmicutes | Bifidobacterium * Lactobacillus * Bacillus * | Increase Increase No change | 9.6 ± 1.2 5.6 ± 2.3 3.0 ± 1.9 | 10.8 ± 0.4 *** 7.4 ± 1.0 2.9 ± 1.3 | 9.7 ± 1.1 6.3 ± 1.8 2.7 ± 1.1 | 8.8 ± 2.4 6.0 ± 1.7 2.8 ± 1.2 | PCR amplification of the V2-V3 region of the 16S rDNA gene obtained from fecal samples | ||
| Proteobacteria Firmicutes | Pseudomonas ** Enterococcus ** | Decrease No change | 2.6 ± 1.5 9.8 ± 1.2 | 2.1 ± 0.4 *** 10.5 ± 0.7 *** | 2.5 ± 1.2 10.4 ± 0.7 | 2.7 ± 1.3 10.5 ± 0.5 | ||||
| Bacteroidetes Firmicutes Firmicutes | n/A n/A Staphylococcus | Increase Decrease No change | 7.9 ± 1.5 7.6 ± 1.1 3.8 ± 1.5 | 8.9 ± 0.7 6.4 ± 1.2 3.6 ± 1.0 | 8.0 ± 1.3 7.5 ± 1.0 3.5 ± 1.3 | 8.7 ± 1.1 8.3 ± 1.0 3.5 ± 1.2 | ||||
| Gianotti et al., 2010 | Proteobacteria | n/A | Decrease | Low dose | High dose | Low dose | High dose | n/A | 4.5 ± 0.2 | PCR amplification, material was obtained from fecal samples |
| n/A | n/A | 4.6 ± 0.6 | 2.4 ± 0.3 *** | |||||||
| Firmicutes | Enterococcus ** | Decrease | n/A | n/A | 4.1 ± 0.4 | 3.4 ± 0.7 *** | n/A | 4.3 ± 0.5 | ||
| Rafter et al., 2007 | Actinobacteria Firmicutes | Bifidobacterium * Lactobacillus * | Increase Increase | 7.52 ± 1.58 5.68 ± 1.51 | 8.76 ± 0.90 *** 6.79 ± 1.39 *** | 7.67 ± 0.93 7.39 ± 0.89 | 8.08 ± 0.96 7.74 ± 1.38 | standard plate count techniques, material obtained from fecal samples | ||
| Firmicuites | Enterococcus ** | No change | 5.26 ± 1.02 | 6.44 ± 1.13 | 6.74 ±1.07 | 6.11 ± 1.23 | ||||
| Firmicuites Coliforms Bacteroidetes | Clostridium n/A Bacteroides | Decrease Decrease No change | 4.01 ± 2.22 5.33 ± 1.37 7.12 ± 1.10 | 3.79 ± 2.69 *** 5.63 ± 1.35 7.24 ± 1.34 | 3.90 ± 2.49 6.08 ± 1.19 7.47 ± 1.10 | 3.03 ± 2.30 6.10 ± 1.00 7.92 ± 1.32 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wierzbicka, A.; Mańkowska-Wierzbicka, D.; Mardas, M.; Stelmach-Mardas, M. Role of Probiotics in Modulating Human Gut Microbiota Populations and Activities in Patients with Colorectal Cancer—A Systematic Review of Clinical Trials. Nutrients 2021, 13, 1160. https://doi.org/10.3390/nu13041160
Wierzbicka A, Mańkowska-Wierzbicka D, Mardas M, Stelmach-Mardas M. Role of Probiotics in Modulating Human Gut Microbiota Populations and Activities in Patients with Colorectal Cancer—A Systematic Review of Clinical Trials. Nutrients. 2021; 13(4):1160. https://doi.org/10.3390/nu13041160
Chicago/Turabian StyleWierzbicka, Adrianna, Dorota Mańkowska-Wierzbicka, Marcin Mardas, and Marta Stelmach-Mardas. 2021. "Role of Probiotics in Modulating Human Gut Microbiota Populations and Activities in Patients with Colorectal Cancer—A Systematic Review of Clinical Trials" Nutrients 13, no. 4: 1160. https://doi.org/10.3390/nu13041160
APA StyleWierzbicka, A., Mańkowska-Wierzbicka, D., Mardas, M., & Stelmach-Mardas, M. (2021). Role of Probiotics in Modulating Human Gut Microbiota Populations and Activities in Patients with Colorectal Cancer—A Systematic Review of Clinical Trials. Nutrients, 13(4), 1160. https://doi.org/10.3390/nu13041160






