Pre-Pregnancy Diet Quality Is Associated with Lowering the Risk of Offspring Obesity and Underweight: Finding from a Prospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
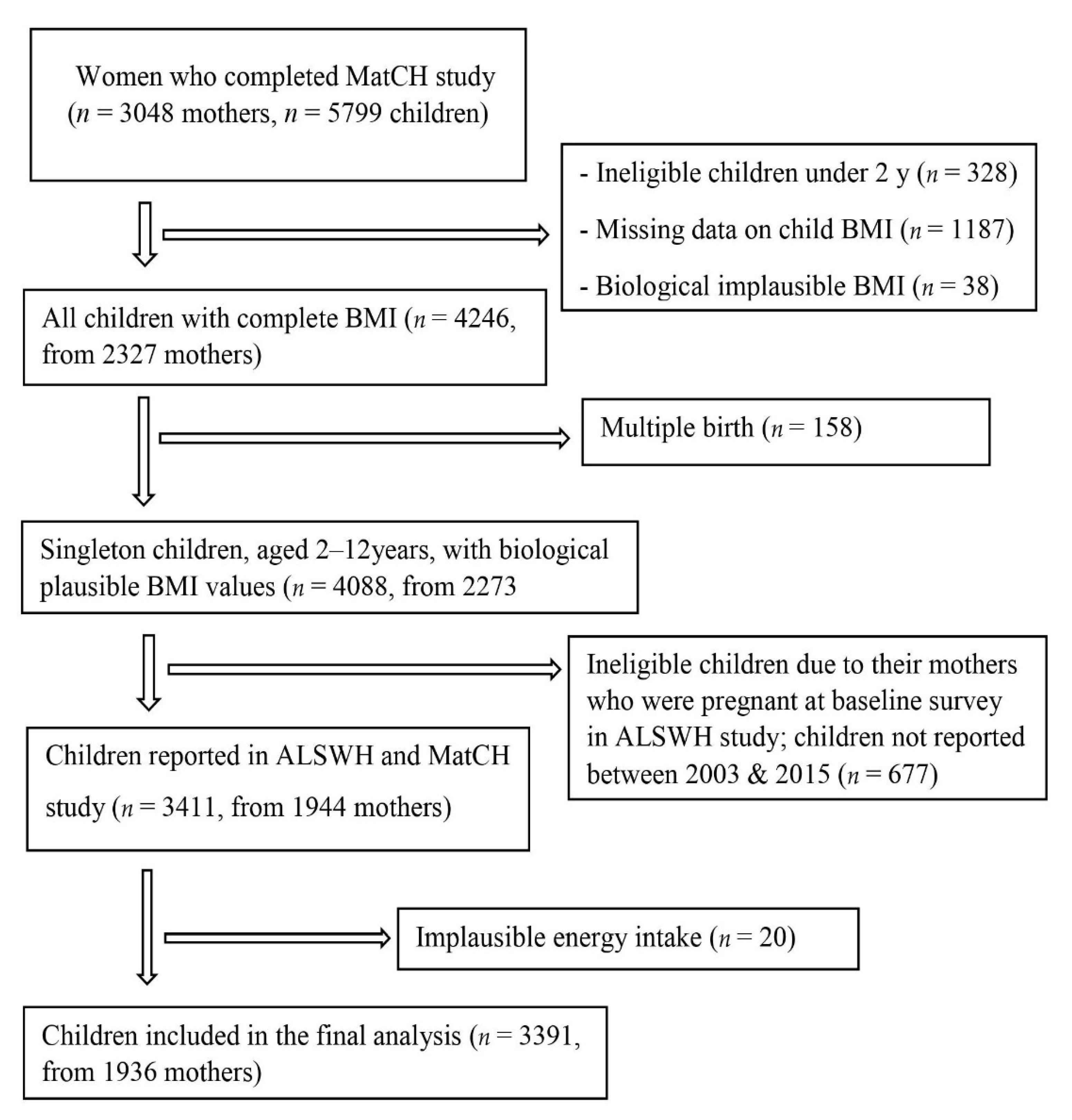
2.1. Study Design and Populations
2.2. Dietary Assessment
2.3. Assessment of Offspring BMI
2.4. Assessment of Confounders and Covariates
2.5. Statistical Analyses
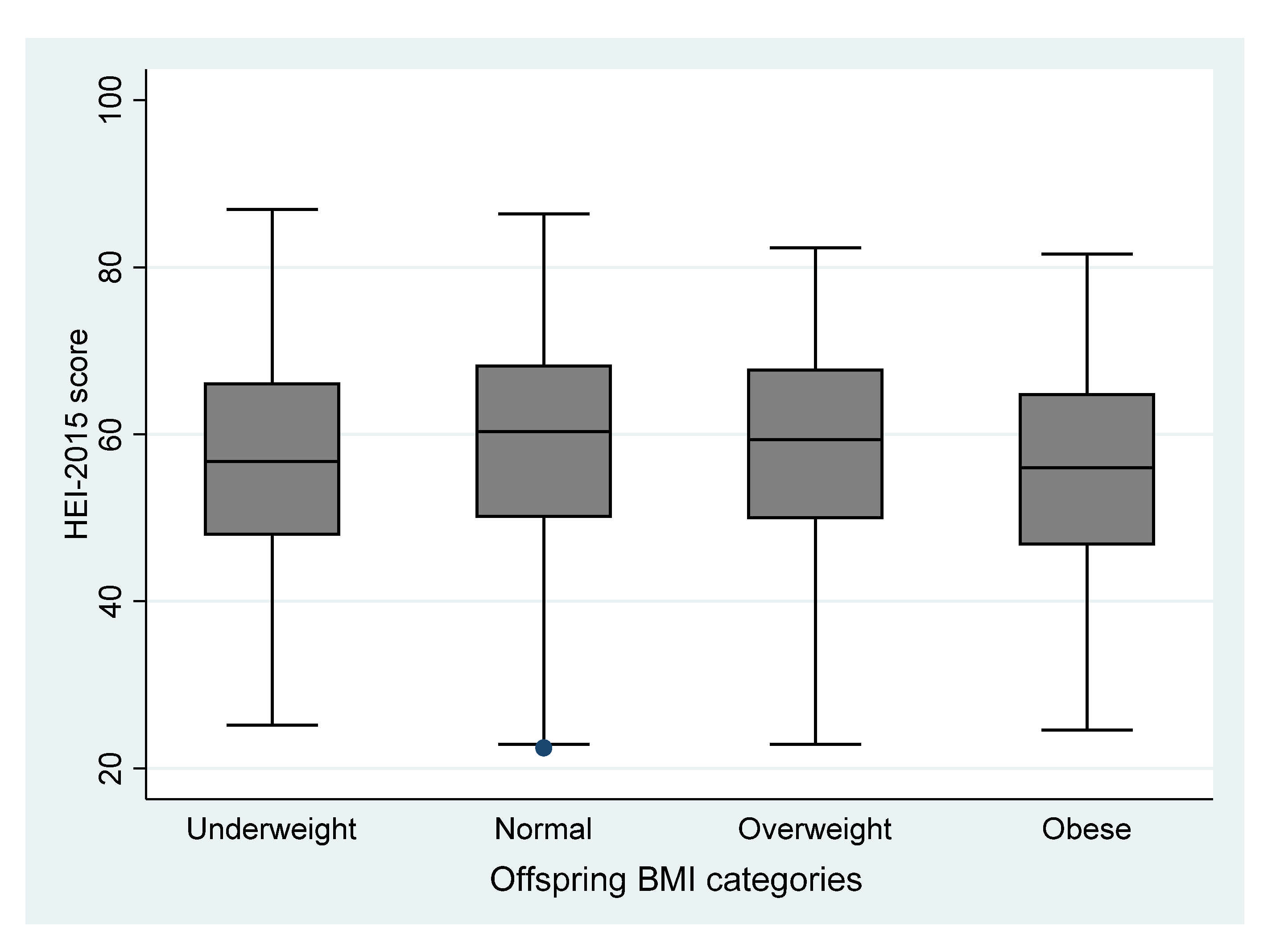
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALSWH | Australian Longitudinal Study on Women’s Health |
| ANOVA | analysis of variance |
| BMI | body mass index |
| CDQ | children’s dietary questionnaire |
| FFQ | food frequency questionnaire |
| GDM | gestational diabetes mellitus |
| HDP | hypertensive disorder in pregnancy |
| HEI-2015 | healthy eating index |
| LBW | low birth weight |
| MET | metabolic equivalent |
| MatCH | Mothers and their Children’s Health |
| RRR | relative risk ratio |
| TEI | total energy intake |
References
- World Health Organization. Obesity and Overweight. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 12 November 2020).
- Australian Institute of Health and Welfare. Australia’s Children. Overweight and Obesity. 2020. Available online: https://www.aihw.gov.au/reports/children-youth/australias-children/contents/health/overweight-and-obesity (accessed on 12 November 2020).
- Abarca-Gómez, L.; Abdeen, Z.A.; Hamid, Z.A.; Abu-Rmeileh, N.M.; Acosta-Cazares, B.; Acuin, C.; Adams, R.J.; Aekpla-korn, W.; Afsana, K.; Aguilar-Salinas, C.A. Worldwide trends in body-mass index, underweight, overweight, and obesi-ty from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 1289 million children, ado-lescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef]
- Singh, A.S.; Mulder, C.; Twisk, J.W.; Van Mechelen, W.; Chinapaw, M.J. Tracking of childhood overweight into adult-hood: A systematic review of the literature. Obes. Rev. 2008, 9, 474–488. [Google Scholar] [CrossRef] [PubMed]
- Daniels, S.R. The consequences of childhood overweight and obesity. Future Child. 2006, 16, 47–67. [Google Scholar] [CrossRef] [PubMed]
- Dietz, W.H. Health consequences of obesity in youth: Childhood predictors of adult disease. Pediatrics 1998, 101, 518–525. [Google Scholar] [PubMed]
- Caballero, B. A nutrition paradox—Underweight and obesity in developing countries. N. Engl. J. Med. 2005, 352, 1514–1516. [Google Scholar] [CrossRef]
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; De Onis, M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Li, Y. Epigenetic mechanisms link maternal diets and gut microbiome to obesity in the offspring. Front. Genet. 2018, 9, 342. [Google Scholar] [CrossRef]
- Murrin, C.; Shrivastava, A.; Kelleher, C. Maternal macronutrient intake during pregnancy and 5 years postpartum and associations with child weight status aged five. Eur. J. Clin. Nutr. 2013, 67, 670. [Google Scholar] [CrossRef]
- Howie, G.; Sloboda, D.; Kamal, T.; Vickers, M. Maternal nutritional history predicts obesity in adult offspring inde-pendent of postnatal diet. J. Physiol. 2009, 587, 905–915. [Google Scholar] [CrossRef]
- American Congress of Obstetricians and Gynecologists. Prenatal Development: How Your Baby Grows during Pregnancy: Women’s Health Care Physicians. Available online: https://www.acog.org/Patients/FAQs/How-Your-Fetus-Grows-During-Pregnancy?IsMobileSet=false (accessed on 20 September 2020).
- Stephenson, J.; Heslehurst, N.; Hall, J.; Schoenaker, D.A.; Hutchinson, J.; Cade, J.E.; Poston, L.; Barrett, G.; Crozier, S.R.; Barker, M. Before the beginning: Nutrition and lifestyle in the preconception period and its importance for future health. Lancet 2018, 391, 1830–1841. [Google Scholar] [CrossRef]
- My Virtual Medical Center. Importance of Healthy Eating Before and During Pregnancy. Available online: https://www.myvmc.com/pregnancy/importance-of-healthy-eating-before-and-during-pregnancy/ (accessed on 20 November 2020).
- Hu, Z.; Tylavsky, F.A.; Kocak, M.; Fowke, J.H.; Han, J.C.; Davis, R.L.; LeWinn, K.Z.; Bush, N.R.; Sathyanarayana, S.; Karr, C.J. Effects of Maternal Dietary Patterns during Pregnancy on Early Childhood Growth Trajectories and Obesity Risk: The CANDLE Study. Nutrients 2020, 12, 465. [Google Scholar] [CrossRef]
- Okubo, H.; Crozier, S.R.; Harvey, N.C.; Godfrey, K.M.; Inskip, H.M.; Cooper, C.; Robinson, S.M. Maternal dietary glycemic index and glycemic load in early pregnancy are associated with offspring adiposity in childhood: The Southampton Women’s Survey. Am. J. Clin. Nutr. 2014, 100, 676–683. [Google Scholar] [CrossRef]
- Van den Broek, M.; Leermakers, E.T.; Jaddoe, V.W.; Steegers, E.A.; Rivadeneira, F.; Raat, H.; Hofman, A.; Franco, O.H.; Kiefte-de Jong, J.C. Maternal dietary patterns during pregnancy and body composition of the child at age 6 y: The Generation R Study, 2. Am. J. Clin. Nutr. 2015, 102, 873–880. [Google Scholar] [CrossRef]
- Poon, A.K.; Yeung, E.; Boghossian, N.; Albert, P.S.; Zhang, C. Maternal dietary patterns during third trimester in association with birthweight characteristics and early infant growth. Scientifica 2013, 2013, 786409. [Google Scholar] [CrossRef]
- Strohmaier, S.; Bogl, L.H.; Eliassen, A.H.; Massa, J.; Field, A.E.; Chavarro, J.E.; Ding, M.; Tamimi, R.M.; Schernhammer, E. Maternal healthful dietary patterns during peripregnancy and long-term overweight risk in their offspring. Eur. J. Epidemiol. 2020, 35, 283–293. [Google Scholar] [CrossRef]
- Dobson, A.J.; Hockey, R.; Brown, W.J.; Byles, J.E.; Loxton, D.J.; McLaughlin, D.; Tooth, L.R.; Mishra, G.D. Cohort Profile Update: Australian Longitudinal Study on Women’s Health. Int. J. Epidemiol. 2015, 44, 1547. [Google Scholar] [CrossRef]
- Mishra, G.D.; Moss, K.; Loos, C.; Dobson, A.J.; Davies, P.S.; Loxton, D.; Hesketh, K.D.; Koupil, I.; Bower, C.; Sly, P. MatCH (Mothers and their Children’s Health) Profile: Offspring of the 1973-78 Cohort of the Australian Longitudinal Study on Women’s Health. Longitud. Life Course Stud. 2018, 9, 351–375. [Google Scholar] [CrossRef]
- Willett, W. Nutritional Epidemiology; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Centers for Disease Control and Prevention. Division of Nutrition, Physical Activity, and Obesity. Extreme or Biologi-Cally Implausible Values. Available online: https://www.cdc.gov/nccdphp/dnpao/growthcharts/resources/sas.htm#values (accessed on 16 September 2020).
- Lewis, J.; Milligan, G.C.; Hunt, A. Nuttab95: Nutrient Data Table for Use in Australia; Australian Government Publishing Service: Canberra, NSW, Australia, 1995. [Google Scholar]
- Ireland, P.; Jolley, D.; Giles, G.; O’Dea, K.; Powles, J.; Rutishauser, I.; Wahlqvist, M.L.; Williams, J. Development of the Melbourne FFQ: A food frequency questionnaire for use in an Australian prospective study involving an ethnically diverse cohort. Asia Pac. J. Clin. Nutr. 1994, 3, 19–31. [Google Scholar]
- Hodge, A.; Patterson, A.J.; Brown, W.J.; Ireland, P.; Giles, G. The Anti Cancer Council of Victoria FFQ: Relative validity of nutrient intakes compared with weighed food records in young to middle-aged women in a study of iron supplementation. Aust. N. Z. J. Public Health 2000, 24, 576–583. [Google Scholar] [CrossRef]
- Krebs-Smith, S.M.; Pannucci, T.E.; Subar, A.F.; Kirkpatrick, S.I.; Lerman, J.L.; Tooze, J.A.; Wilson, M.M.; Reedy, J. Update of the Healthy Eating Index: HEI-2015. J. Acad. Nutr. Diet 2018, 118, 1591–1602. [Google Scholar] [CrossRef]
- Reedy, J.; Lerman, J.L.; Krebs-Smith, S.M.; Kirkpatrick, S.I.; Pannucci, T.E.; Wilson, M.M.; Subar, A.F.; Kahle, L.L.; Tooze, J.A. Evaluation of the Healthy Eating Index-2015. J. Acad. Nutr. Diet 2018, 118, 1622–1633. [Google Scholar] [CrossRef]
- Cole, T.J.; Bellizzi, M.C.; Flegal, K.M.; Dietz, W.H. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ 2000, 320, 1240–1243. [Google Scholar] [CrossRef]
- Department of Health and Aged Care (GISCA). Measuring Remoteness: Accessibility/Remoteness Index of Australia (ARIA), Revised edn; Commonwealth Department of Health and Aged Care: Canberra, NSW, Australia, 2001. [Google Scholar]
- National Health and Medical Research Council. Australian Alcohol Guidelines: Health Risks and Benefits. 2001. Available online: https://books.google.com.au/books?id=D7-mAAAACAAJ (accessed on 20 June 2019).
- Brown, W.J.; Burton, N.W.; Marshall, A.L.; Miller, Y.D. Reliability and validity of a modified self-administered version of the Active Australia physical activity survey in a sample of mid-age women. Aust. N. Z. J. Public Health 2008, 32, 535–541. [Google Scholar] [CrossRef]
- Magarey, A.; Golley, R.; Spurrier, N.; Goodwin, E.; Ong, F. Reliability and validity of the Children’s Dietary Questionnaire: A new tool to measure children’s dietary patterns. Int. J. Pediatr. Obes. IJPO Off. J. Int. Assoc. Stud. Obes. 2009, 4, 257–265. [Google Scholar] [CrossRef]
- Stuebe, A.M.; Forman, M.R.; Michels, K.B. Maternal-recalled gestational weight gain, pre-pregnancy body mass index, and obesity in the daughter. Int. J. Obes. 2009, 33, 743–752. [Google Scholar] [CrossRef]
- Martin, C.L.; Siega-Riz, A.M.; Sotres-Alvarez, D.; Robinson, W.R.; Daniels, J.L.; Perrin, E.M.; Stuebe, A.M. Maternal Dietary Patterns during Pregnancy Are Associated with Child Growth in the First 3 Years of Life–3. J. Nutr. 2016, 146, 2281–2288. [Google Scholar] [CrossRef] [PubMed]
- Murrin, C.M.; Heinen, M.M.; Kelleher, C.C. Are Dietary Patterns of Mothers during Pregnancy Related to Children’s Weight Status? Evidence from the Lifeways Cross- Generational Cohort Study. AIMS Public Health 2015, 2, 274–296. [Google Scholar] [CrossRef]
- Fernández-Barrés, S.; Romaguera, D.; Valvi, D.; Martínez, D.; Vioque, J.; Navarrete-Muñoz, E.; Amiano, P.; Gonzalez-Palacios, S.; Guxens, M.; Pereda, E. Mediterranean dietary pattern in pregnant women and offspring risk of overweight and abdominal obesity in early childhood: The INMA birth cohort study. Pediatr. Obes. 2016, 11, 491–499. [Google Scholar] [CrossRef]
- Chen, L.-W.; Aris, I.; Bernard, J.; Tint, M.-T.; Chia, A.; Colega, M.; Gluckman, P.; Shek, L.; Saw, S.-M.; Chong, Y.-S. Associations of maternal dietary patterns during pregnancy with offspring adiposity from birth until 54 months of age. Nutrients 2017, 9, 2. [Google Scholar] [CrossRef]
- Dhana, K.; Haines, J.; Liu, G.; Zhang, C.; Wang, X.; Field, A.E.; Chavarro, J.E.; Sun, Q. Association between maternal adherence to healthy lifestyle practices and risk of obesity in offspring: Results from two prospective cohort studies of mother-child pairs in the United States. BMJ 2018, 362, k2486. [Google Scholar] [CrossRef]
- Aagaard-Tillery, K.M.; Grove, K.; Bishop, J.; Ke, X.; Fu, Q.; McKnight, R.; Lane, R.H. Developmental origins of disease and determinants of chromatin structure: Maternal diet modifies the primate fetal epigenome. J. Mol. Endocrinol. 2008, 41, 91. [Google Scholar] [CrossRef] [PubMed]
- Paul, H.A.; Bomhof, M.R.; Vogel, H.J.; Reimer, R.A. Diet-induced changes in maternal gut microbiota and metabolomic profiles influence programming of offspring obesity risk in rats. Sci. Rep. 2016, 6, 20683. [Google Scholar] [CrossRef] [PubMed]
- Hardy, T.M.; Tollefsbol, T.O. Epigenetic diet: Impact on the epigenome and cancer. Epigenomics 2011, 3, 503–518. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tollefsbol, T.O. Impact on DNA methylation in cancer prevention and therapy by bioactive dietary components. Curr. Med. Chem. 2010, 17, 2141–2151. [Google Scholar] [CrossRef]
- Jaclyn Chasse, N.D. Epigenetics and Preconception: The Ultimate Preventive Medicine. Available online: https://divcomplatform.s3.amazonaws.com/ip.divcomstaging.com/images/6c098f92dcd09563a0a9b79fa51c3f00.pdf (accessed on 16 April 2019).
- Rodríguez-Bernal, C.L.; Rebagliato, M.; Iñiguez, C.; Vioque, J.; Navarrete-Muñoz, E.M.; Murcia, M.; Bolumar, F.; Marco, A.; Ballester, F. Diet quality in early pregnancy and its effects on fetal growth outcomes: The Infancia y Medio Ambiente (Childhood and Environment) Mother and Child Cohort Study in Spain. Am. J. Clin. Nutr. 2010, 91, 1659–1666. [Google Scholar] [CrossRef]
- Oh, S.W.; Koo, H.S.; Han, K.H.; Han, S.Y.; Chin, H.J. Associations of sodium intake with obesity, metabolic disorder, and albuminuria according to age. PLoS ONE 2017, 12, e0188770. [Google Scholar] [CrossRef]
- Lee, J.; Hwang, Y.; Kim, K.-N.; Ahn, C.; Sung, H.K.; Ko, K.-P.; Oh, K.-H.; Ahn, C.; Park, Y.J.; Kim, S. Associations of urinary sodium levels with overweight and central obesity in a population with a sodium intake. BMC Nutr. 2018, 4, 47. [Google Scholar] [CrossRef]
- Fonseca-Alaniz, M.H.; Takada, J.; Andreotti, S.; De Campos, T.B.; Campaña, A.B.; Borges-Silva, C.N.; Lima, F.B. High sodium intake enhances insulin-stimulated glucose uptake in rat epididymal adipose tissue. Obesity 2008, 16, 1186–1192. [Google Scholar] [CrossRef]
- Libuda, L.; Kersting, M.; Alexy, U. Consumption of dietary salt measured by urinary sodium excretion and its association with body weight status in healthy children and adolescents. Public Health Nutr. 2012, 15, 433–441. [Google Scholar] [CrossRef]
- He, F.J.; Marrero, N.M.; MacGregor, G.A. Salt Intake, Hypertension, and Obesity in Children. Hypertension 2008, 51, 629–634. [Google Scholar] [CrossRef]
- Hosomi, R.; Yoshida, M.; Fukunaga, K. Seafood consumption and components for health. Glob. J. Health Sci. 2012, 4, 72. [Google Scholar] [CrossRef]
- Krajcovicova-Kudlackova, M.; Babinska, K.; Valachovicova, M. Health benefits and risks of plant proteins. Bratisl. Lekarske Listy 2005, 106, 231. [Google Scholar]

| Characteristics | All Children | Offspring BMI Category | p-Value b | |||
|---|---|---|---|---|---|---|
| n = 3391 | Underweight | Normal | Overweight | Obese | ||
| HEI-2015 score (%) of mothers | 0.01 | |||||
| Quartile 1 | 25 | 13.9 | 69.3 | 12.3 | 4.5 | |
| Quartile 2 | 25 | 12.5 | 71.2 | 13 | 3.3 | |
| Quartile 3 | 25 | 9.9 | 74.1 | 12.6 | 3.4 | |
| Quartile 4 | 25 | 9.8 | 76.1 | 12.2 | 1.9 | |
| Maternal age (years), mean (SD) | 33.1 (2.9) | 32.9 (2.9) | 33.1 (2.9) | 33.2 (3.1) | 33.2 (2.8) | 0.41 |
| Area of residence (%) c | 0.24 | |||||
| Urban | 61.4 | 10.6 | 73.5 | 12.7 | 3.2 | |
| Rural/remote | 37.4 | 12.8 | 71.3 | 12.4 | 3.5 | |
| Marital status (%) | 0.52 | |||||
| Married | 55.3 | 11.4 | 72.9 | 12.1 | 3.6 | |
| De facto/separated/divorced | 26.3 | 12.1 | 73.2 | 12.3 | 2.3 | |
| Single | 18.3 | 10.9 | 71.3 | 14 | 3.7 | |
| Educational status (%) c | 0.002 | |||||
| Up to year 12 or equivalent | 15.5 | 11.9 | 68.9 | 14.2 | 4.9 | |
| Trade/apprenticeship/certificate/diploma | 20.3 | 12.1 | 69.3 | 13.9 | 4.6 | |
| University/higher degree | 63.7 | 11.3 | 74.7 | 11.6 | 2.4 | |
| Smoking status (%) | 0.02 | |||||
| Never smoked | 65.1 | 11.2 | 74.3 | 11.8 | 2.6 | |
| Ex-smoker | 19.5 | 11.9 | 70.4 | 13 | 4.7 | |
| Current smoker | 15.3 | 12.3 | 68.6 | 14.8 | 4.2 | |
| Alcohol intake (%) | 0.7 | |||||
| Non-drinker | 6.2 | 11.5 | 71.8 | 13.4 | 3.3 | |
| Rarely-drinker | 19.9 | 10.6 | 71.7 | 13.3 | 4.3 | |
| Low-risk drinker | 70.6 | 11.7 | 73.1 | 12.2 | 2.9 | |
| Risky-drinker | 3.3 | 12.6 | 70.3 | 11.7 | 5.4 | |
| Physical activity (%) c | 0.09 | |||||
| Sedentary/low, <600 MET min/week | 39.4 | 12 | 70.6 | 13.8 | 3.7 | |
| Moderate, 600 to 1200 MET min/week | 26.4 | 11.7 | 75.1 | 11.2 | 2 | |
| High, ≥1200 MET min/week | 33.6 | 11 | 73.3 | 12.2 | 3.7 | |
| Pre-pregnancy BMI (%) c | <0.0001 | |||||
| Healthy weight, < 25 kg/m2 | 67.7 | 13 | 74.8 | 10.4 | 1.7 | |
| Overweight, 25–30 kg/m2 | 21.5 | 9.1 | 71.7 | 15 | 4.3 | |
| Obese, ≥30 kg/m2 | 10.5 | 7.3 | 61.1 | 20.5 | 11.2 | |
| Total energy intake (KJ/day), mean (SD) | 6752.9 (2238.3) | 6799.8 (2209.1) | 6702.3 (2215.3) | 6826.5 (2334.5) | 7430.9 (2378.8) | 0.007 |
| Household income (weekly) (%) c | 0.009 | |||||
| ≤ 999$ | 16.5 | 13.1 | 69.3 | 12.2 | 5.4 | |
| 1000$–1499$ | 19.7 | 11.8 | 71.3 | 12.7 | 4.2 | |
| ≥1500$ | 51 | 10.9 | 75 | 11.8 | 2.3 | |
| Don’t know/don’t want to answer | 6.2 | 14.3 | 68.4 | 12.9 | 4.3 | |
| Living alone | 4.9 | 9.7 | 70.3 | 17.6 | 2.4 | |
| Gestational diabetes mellitus (%) c | 0.33 | |||||
| Yes | 4.6 | 15.4 | 69.2 | 10.9 | 4.5 | |
| No | 95 | 11.4 | 72.9 | 12.5 | 3.2 | |
| Hypertensive disorder in pregnancy (%) c | 0.4 | |||||
| Yes | 5.4 | 11.4 | 70.6 | 12.5 | 5.4 | |
| No | 94.2 | 11.5 | 72.9 | 12.5 | 3.1 | |
| Preterm birth (%) c | 0.32 | |||||
| Yes | 4.5 | 12.3 | 66.9 | 16.2 | 4.5 | |
| No | 95.2 | 11.5 | 73 | 12.3 | 3.2 | |
| LBW (%) c | 0.05 | |||||
| Yes | 3.2 | 18.3 | 69.7 | 7.3 | 4.6 | |
| No | 96.6 | 11.3 | 72.8 | 12.7 | 3.2 | |
| Child age (years), mean (SD) | 7.3 (2.9) | 7.6 (2.9) | 7.3 (2.8) | 7.3 (3.1) | 7.4 (2.9) | 0.16 |
| Child sex (%) | 0.27 | |||||
| Male | 53 | 10.9 | 74.1 | 11.9 | 3.1 | |
| Female | 47 | 12.2 | 71.1 | 13.2 | 3.5 | |
| Breastfeeding status (%) c | 0.04 | |||||
| Never received | 3.6 | 13.2 | 62.8 | 17.4 | 6.6 | |
| Received | 94.6 | 11.5 | 73 | 12.2 | 3.2 | |
| Child diets c, mean (SD) | ||||||
| Fruits and vegetables | 12.0 (3.6) | 12.0 (3.7) | 12.0 (3.6) | 11.7 (3.6) | 11.8 (4.4) | 0.37 |
| Sweetened beverages | 0.6 (0.8) | 0.6 (0.8) | 0.5 (0.7) | 0.5 (0.7) | 0.8 (1.0) | 0.001 |
| Fat from dairy | 3.0 (2.0) | 3.0 (1.9) | 3.0 (1.9) | 3.1 (2.0) | 3.1 (2.6) | 0.84 |
| Non-core foods | 2.3 (1.0) | 2.3 (1.0) | 2.3 (1.0) | 2.3 (0.9) | 2.3 (1.1) | 0.64 |
| HEI-2015 Score | |||||
|---|---|---|---|---|---|
| Characteristics | Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | p-Value b |
| (n = 484) | (n = 484) | (n = 484) | (n = 484) | ||
| Maternal age (years), mean (SD) | 31.6 (2.8) | 32.4 (3.1) | 32.2 (2.9) | 32.7 (2.9) | <0.0001 |
| Area of residence (%) | <0.0001 | ||||
| Urban | 21.8 | 24.8 | 26.9 | 26.5 | |
| Rural/remote | 30.7 | 25.3 | 22 | 22 | |
| Marital status (%) | 0.2 | ||||
| Married | 26.4 | 26.2 | 24.5 | 22.9 | |
| De facto/separated/divorced | 24.1 | 25 | 25.4 | 25.4 | |
| Single | 22.9 | 22.2 | 25.7 | 29.2 | |
| Educational status (%) | <0.0001 | ||||
| Up to year 12 or equivalent | 40.1 | 22.2 | 21.6 | 16.2 | |
| Trade/apprenticeship/certificate/diploma | 31.1 | 24.3 | 22.6 | 22.1 | |
| University/higher degree | 18.7 | 26.1 | 26.9 | 28.3 | |
| Smoking (%) | <0.0001 | ||||
| Never smoked | 23.6 | 25.1 | 27 | 24.4 | |
| Ex-smoker | 21.4 | 24.7 | 25.2 | 28.8 | |
| Current smoker | 34.2 | 25 | 17.6 | 23.2 | |
| Alcohol intake (%) | 0.001 | ||||
| Non-drinker | 34.3 | 28.6 | 24.8 | 12.4 | |
| Rarely-drinker | 30.7 | 26.6 | 22.7 | 20 | |
| Low-risk drinker | 22.7 | 24.2 | 25.5 | 27.6 | |
| Risky-drinker | 25 | 26.5 | 27.9 | 20.6 | |
| Physical activity (%) | <0.0001 | ||||
| Sedentary/low, <600 MET min/week | 31.7 | 26 | 22.7 | 19.7 | |
| Moderate, 600 to 1200 MET min/week | 21.7 | 26.6 | 27.1 | 24.6 | |
| High, ≥1200 MET min/week | 19.9 | 22.7 | 25.9 | 31.5 | |
| Pre-pregnancy BMI (%) | 0.08 | ||||
| Healthy weight, < 25 kg/m2 | 24.6 | 24.5 | 24.6 | 26.3 | |
| Overweight, 25–30 kg/m2 | 23.1 | 24.6 | 28.5 | 23.8 | |
| Obese, ≥30 kg/m2 | 30.5 | 28.6 | 21.1 | 19.7 | |
| Total energy intake (KJ/day), mean (SD) | 7727.9 (2540.9) | 68.94.2 (2143.6) | 6247.5 (1880.3) | 5912.5 (1842.5) | <0.0001 |
| Household income (weekly) (%) | <0.0001 | ||||
| ≤ 999$ | 37.5 | 27.8 | 17.9 | 16.8 | |
| 1000$–1499$ | 28 | 27.5 | 25.1 | 19.4 | |
| ≥1500$ | 20.3 | 23.6 | 26.6 | 29.5 | |
| Don’t know/don’t want to answer | 21.4 | 23 | 27.8 | 27.8 | |
| Living alone | 21.7 | 22.5 | 28.3 | 27.5 | |
| Offspring BMI Category | HEI-2015 Score | |||
|---|---|---|---|---|
| Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | |
| Underweight | ||||
| Model 1 | 1 | 0.87 (0.64, 1.19) | 0.66 (0.48, 0.92) | 0.64 (0.46, 0.89) |
| Model 2 b | 1 | 0.89 (0.65, 1.21) | 0.71 (0.50, 0.99) | 0.64 (0.46, 0.90) |
| Model 3 c | 1 | 0.93 (0.67, 1.28) | 0.76 (0.55, 1.07) | 0.69 (0.49, 0.97) |
| Model 4 d | 1 | 0.94 (0.68, 1.29) | 0.77 (0.55, 1.08) | 0.68 (0.49, 0.96) |
| Normal e | 1 | 1 | 1 | 1 |
| Overweight | ||||
| Model 1 | 1 | 1.03 (0.76, 1.39) | 0.96 (0.70, 1.31) | 0.90 (0.66, 1.24) |
| Model 2 b | 1 | 1.09 (0.80, 1.49) | 1.04 (0.75, 1.43) | 0.99 (0.70, 1.38) |
| Model 3 c | 1 | 1.15 (0.84, 1.59) | 1.05 (0.75, 1.47) | 0.98 (0.69, 1.38) |
| Model 4 d | 1 | 1.17 (0.85, 1.62) | 1.08 (0.77, 1.52) | 1.03 (0.72, 1.47) |
| Obese | ||||
| Model 1 | 1 | 0.72 (0.42, 1.23) | 0.71 (0.42, 1.21) | 0.38 (0.20, 0.74) |
| Model 2 b | 1 | 0.89 (0.51, 1.56) | 0.97 (0.56, 1.67) | 0.47 (0.24, 0.93) |
| Model 3 c | 1 | 0.98 (0.56, 1.72) | 1.10 (0.64, 1.91) | 0.49 (0.24, 0.98) |
| Model 4 d | 1 | 0.99 (0.56, 1.76) | 1.12 (0.64, 1.97) | 0.54 (0.26, 1.11) |
| Components | Standard for Maximum Score | Standard for Minimum Score of Zero | Maximum Points | Offspring BMI Categories | Unadjusted RRR (95% CI) a | Adjusted RRR (95% CI) b |
|---|---|---|---|---|---|---|
| Adequacy: | ||||||
| Total Fruits | ≥0.8 C eq./1,000 kcal | No fruit | 5 | Underweight | 0.99 (0.90, 1.09) | 1.06 (0.95, 1.18) |
| Overweight | 0.90 (0.83, 0.99) | 0.92 (0.83, 1.01) | ||||
| Obese | 0.93 (0.78, 1.10) | 1.13 (0.93, 1.37) | ||||
| Whole Fruits | ≥0.4 C | No whole fruit | 5 | Underweight | 0.98 (0.88, 1.09) | 1.06 (0.93, 1.21) |
| eq./1000 kcal | Overweight | 0.94 (0.85, 1.03) | 0.99 (0.89, 1.11) | |||
| Obese | 0.89 (0.74, 1.06) | 1.14 (0.90, 1.43) | ||||
| Total Vegetables | ≥1.1 C eq./1000 kcal | No vegetables | 5 | Underweight | 0.98 (0.89, 1.07) | 1.05 (0.94, 1.17) |
| Overweight | 0.96 (0.87, 1.05) | 0.97 (0.87, 1.07) | ||||
| Obese | 0.93 (0.80, 1.09) | 1.07 (0.87, 1.30) | ||||
| Greens and Beans | ≥0.2 C eq./1000 kcal | No Greens and Beans | 5 | Underweight | 1.10 (0.93, 1.30) | 1.19 (0.99, 1.44) |
| Overweight | 0.95 (0.81, 1.10) | 1.02 (0.85, 1.22) | ||||
| Obese | 1.05 (0.79, 1.39) | 1.36 (0.96, 1.94) | ||||
| Whole Grains | ≥1.5 oz. eq./1000 kcal | No whole grains | 10 | Underweight | 0.97 (0.94, 0.99) | 0.98 (0.94, 1.01) |
| Overweight | 1.00 (0.98, 1.03) | 1.02 (0.98, 1.05) | ||||
| Obese | 0.94 (0.89, 0.99) | 0.97 (0.91, 1.03) | ||||
| Dairy | ≥1.3 C eq./1000 kcal | No dairy | 10 | Underweight | 0.97 (0.94, 1.01) | 1.03 (0.99, 1.08) |
| Overweight | 0.99 (0.96, 1.02) | 1.00 (0.95, 1.05) | ||||
| Obese | 0.94 (0.89, 1.00) | 0.99 (0.92, 1.09) | ||||
| Total Protein | ≥2.5 oz. eq./1000 kcal | No protein foods | 5 | Underweight | 0.82 (0.67, 1.01) | 0.86 (0.69, 1.07) |
| Overweight | 0.96 (0.74, 1.24) | 0.93 (0.71, 1.22) | ||||
| Obese | 0.89 (0.65, 1.20) | 0.91 (0.64, 1.29) | ||||
| Seafood and Plant Proteins | ≥0.8 C eq./1000 kcal | No seafood or plant proteins | 5 | Underweight | 0.85 (0.74, 0.97) | 0.84 (0.73, 0.99) |
| Overweight | 0.99 (0.89, 1.10) | 1.03 (0.91, 1.18) | ||||
| Obese | 0.99 (0.80, 1.22) | 1.14 (0.89, 1.47) | ||||
| Fatty Acids | (PUFA + MUFA)/SFA (g/day) ≥ 2.5 | (PUFA + MUFA)/SFA (g/day) ≤1.2 | 10 | Underweight | 0.97 (0.94, 1.01) | 0.98 (0.94, 1.02) |
| Overweight | 1.00 (0.97, 1.04) | 1.01 (0.97, 1.05) | ||||
| Obese | 0.96 (0.90, 1.03) | 0.98 (0.91, 1.05) | ||||
| Moderation: | ||||||
| Refined Grains | ≤1.8 oz. eq./1000 kcal | ≥4.3 oz. eq./1000 kcal | 10 | Underweight | 0.97 (0.94, 1.00) | 0.99 (0.95, 1.03) |
| Overweight | 0.99 (0.96, 1.03) | 0.99 (0.95, 1.03) | ||||
| Obese | 0.99 (0.93, 1.06) | 0.99 (0.93, 1.07) | ||||
| Sodium | ≤1.1 g/1000 kcal | ≥2.0 g/1000 kcal | 10 | Underweight | 1.05 (0.87, 1.26) | 1.08 (0.89, 1.32) |
| Overweight | 0.96 (0.78, 1.20) | 0.18 (0.14, 0.23) | ||||
| Obese | 0.23 (0.18, 0.30) | 0.21 (0.17, 0.26) | ||||
| Added Sugars | ≤6.5% of energy | ≥26% of energy | 10 | Underweight | 0.97 (0.90, 1.04) | 1.00 (0.93, 1.08) |
| Overweight | 0.98 (0.91, 1.07) | 0.99 (0.91, 1.08) | ||||
| Obese | 0.93 (0.83, 1.04) | 0.96 (0.85, 1.07) | ||||
| Saturated Fats | ≤8% of energy | ≥16% of energy | 10 | Underweight | 0.97 (0.95, 1.00) | 1.00 (0.97, 1.05) |
| Overweight | 0.99 (0.96, 1.01) | 0.99 (0.96, 1.03) | ||||
| Obese | 0.94 (0.90, 0.99) | 0.99 (0.93, 1.07) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gete, D.G.; Waller, M.; Mishra, G.D. Pre-Pregnancy Diet Quality Is Associated with Lowering the Risk of Offspring Obesity and Underweight: Finding from a Prospective Cohort Study. Nutrients 2021, 13, 1044. https://doi.org/10.3390/nu13041044
Gete DG, Waller M, Mishra GD. Pre-Pregnancy Diet Quality Is Associated with Lowering the Risk of Offspring Obesity and Underweight: Finding from a Prospective Cohort Study. Nutrients. 2021; 13(4):1044. https://doi.org/10.3390/nu13041044
Chicago/Turabian StyleGete, Dereje G., Michael Waller, and Gita D. Mishra. 2021. "Pre-Pregnancy Diet Quality Is Associated with Lowering the Risk of Offspring Obesity and Underweight: Finding from a Prospective Cohort Study" Nutrients 13, no. 4: 1044. https://doi.org/10.3390/nu13041044
APA StyleGete, D. G., Waller, M., & Mishra, G. D. (2021). Pre-Pregnancy Diet Quality Is Associated with Lowering the Risk of Offspring Obesity and Underweight: Finding from a Prospective Cohort Study. Nutrients, 13(4), 1044. https://doi.org/10.3390/nu13041044