A Pilot Study Comparing the Effects of Consuming 100% Orange Juice or Sucrose-Sweetened Beverage on Risk Factors for Cardiometabolic Disease in Women
Abstract
1. Introduction
2. Materials and Methods
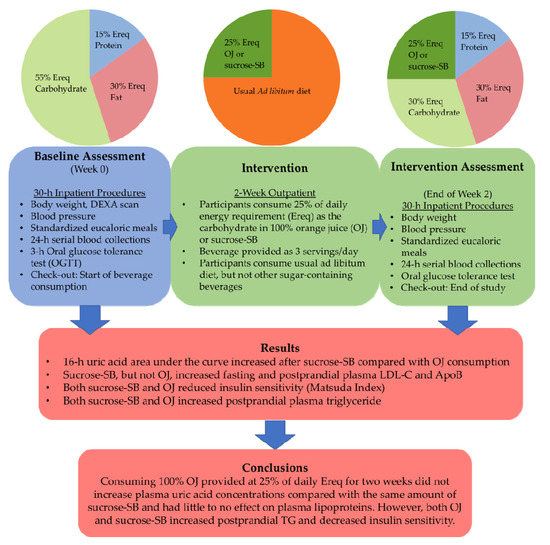
2.1. Study Design
2.2. Study Participants
2.3. Study Beverages
Standardized Isocaloric Meals
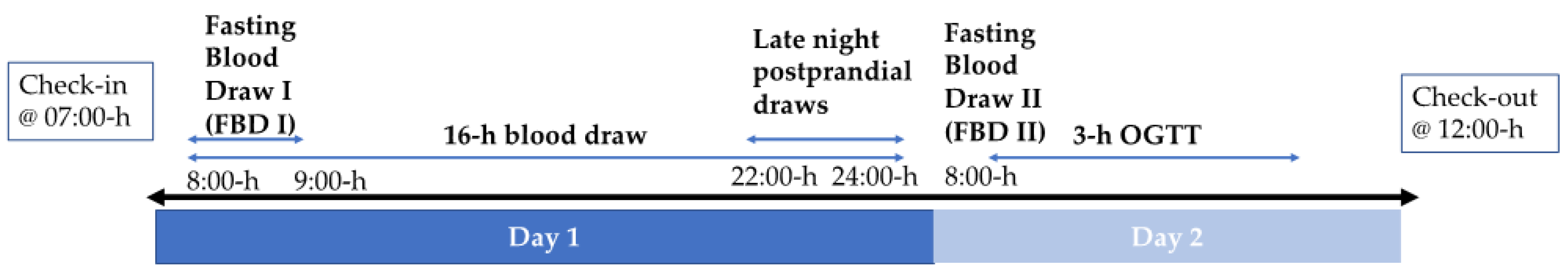
2.4. Metabolic Testing Procedures and Sample Collection
2.5. Statistical Analyses
3. Results
3.1. Anthropometric Measures
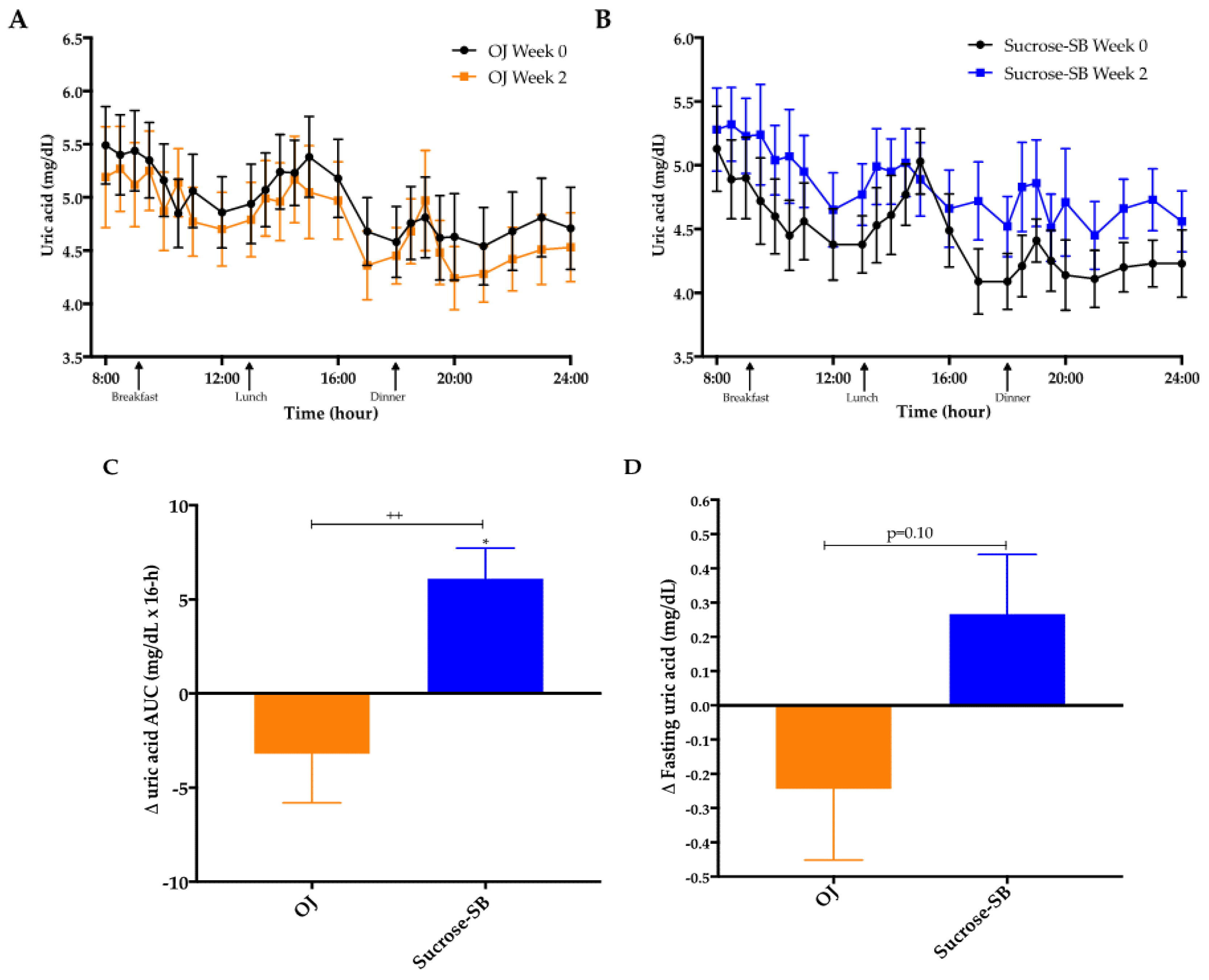
3.2. Uric Acid
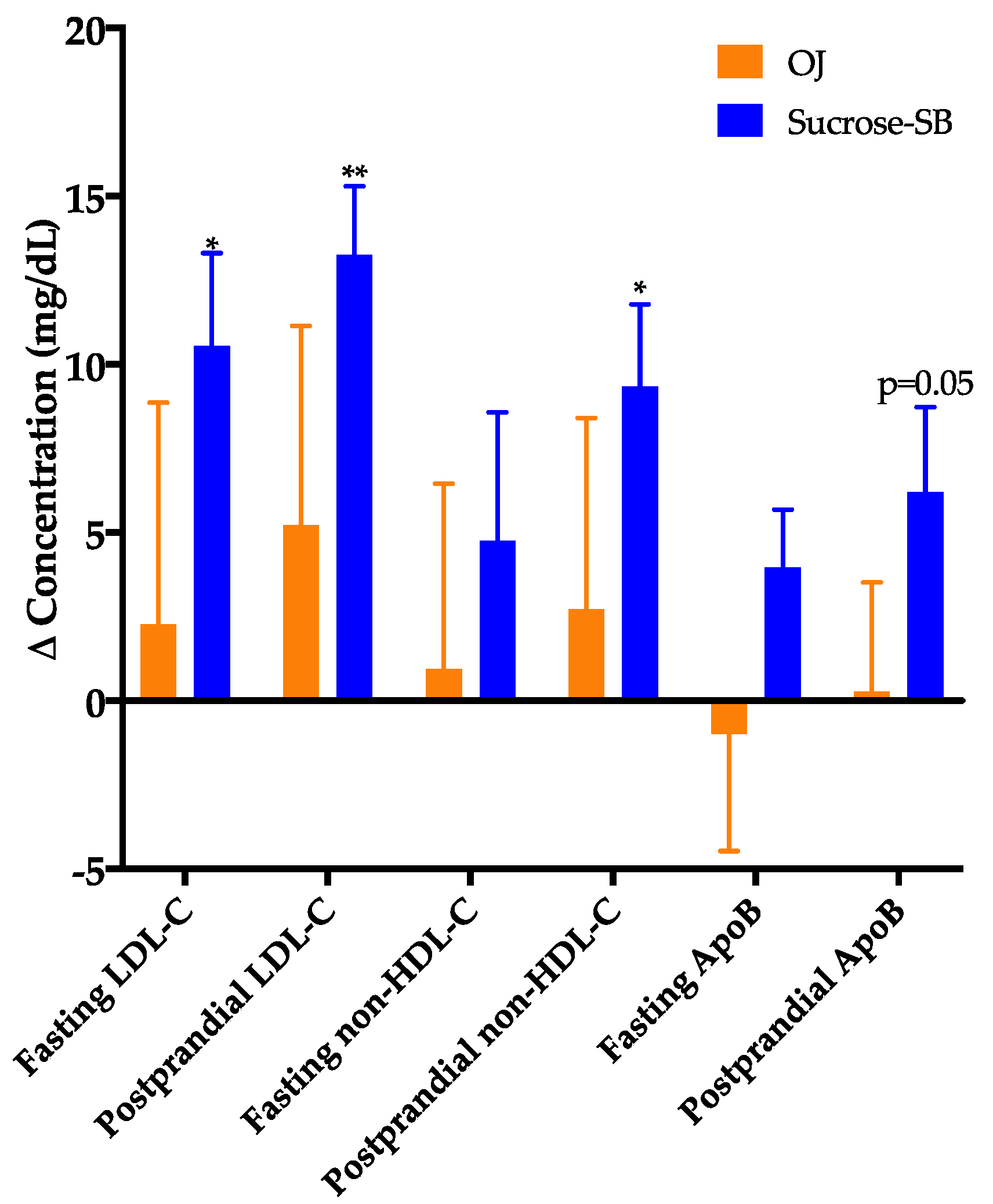
3.3. Plasma Cholesterol, Lipoproteins and Apolipoproteins
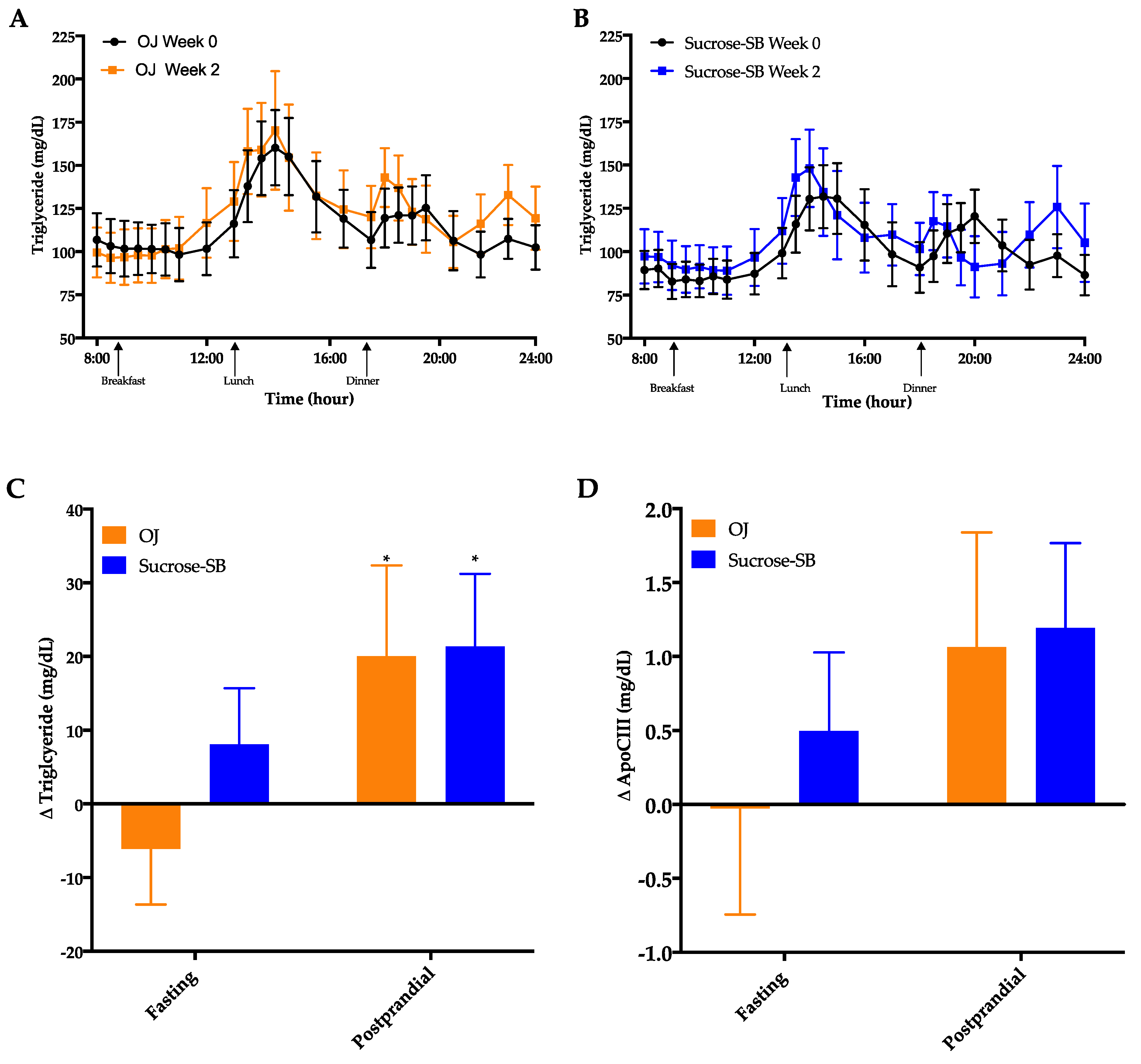
3.4. Circulating Triglycerides (TG) and Apolipoprotein CIII (ApoCIII)
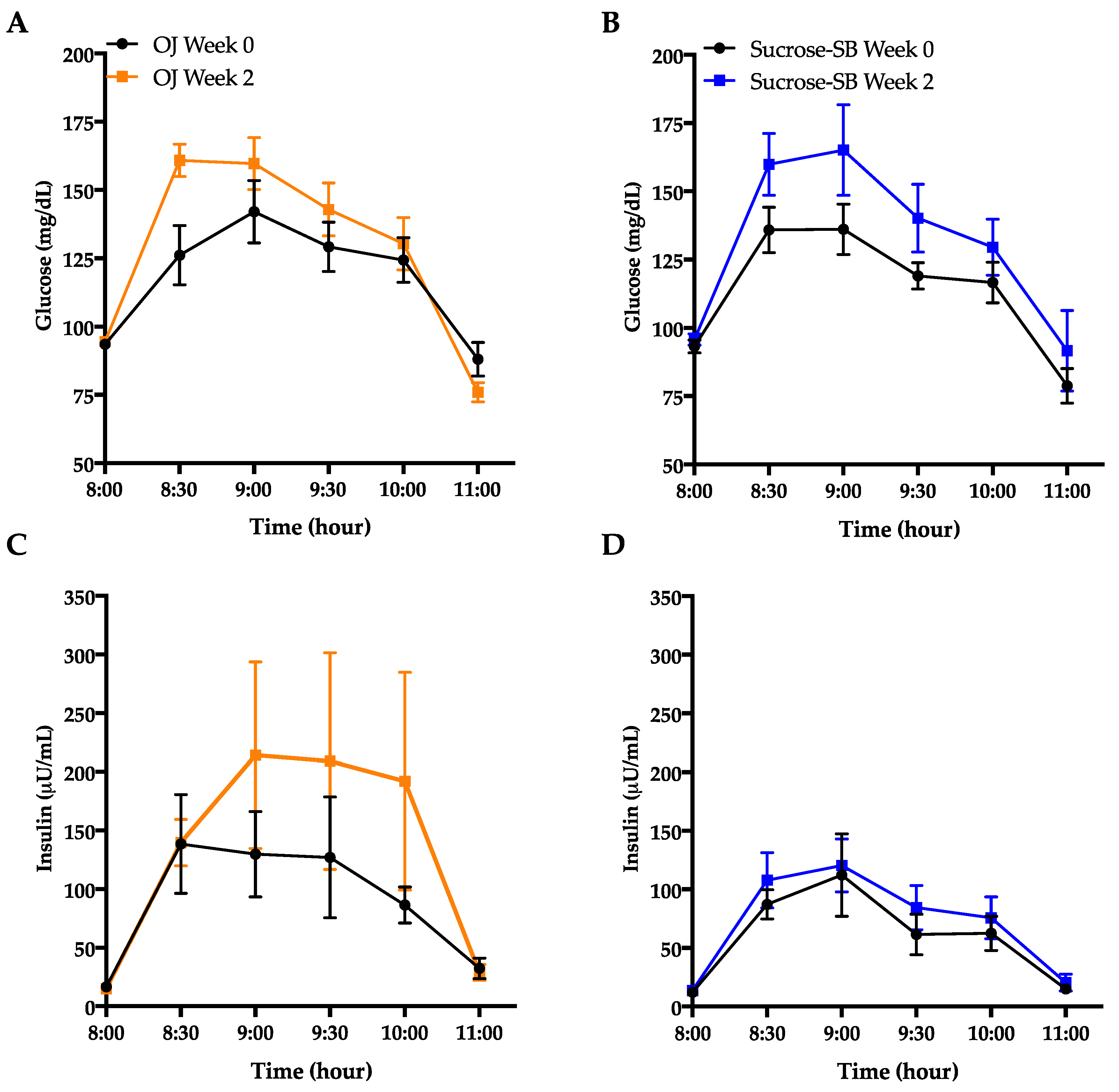
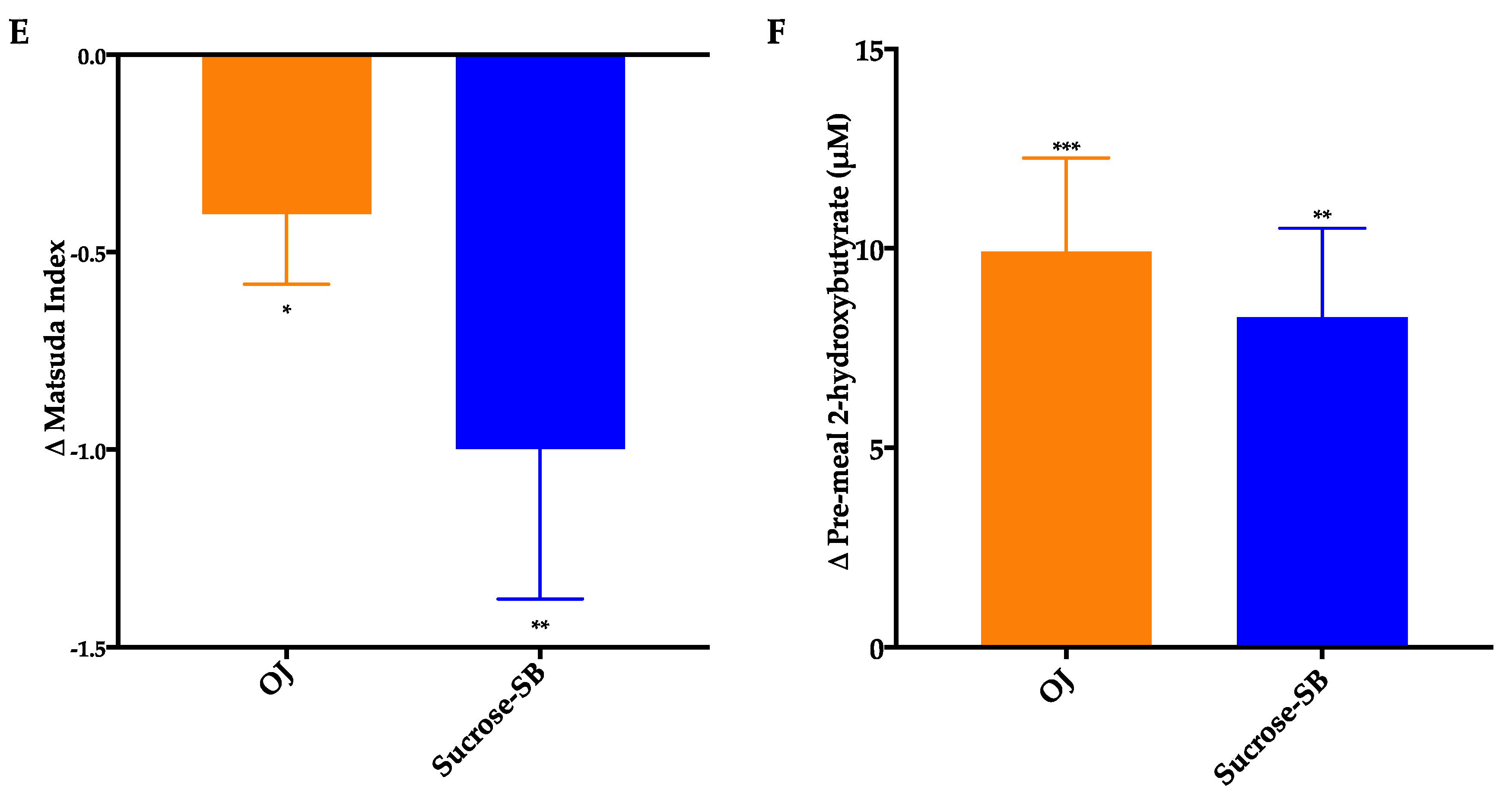
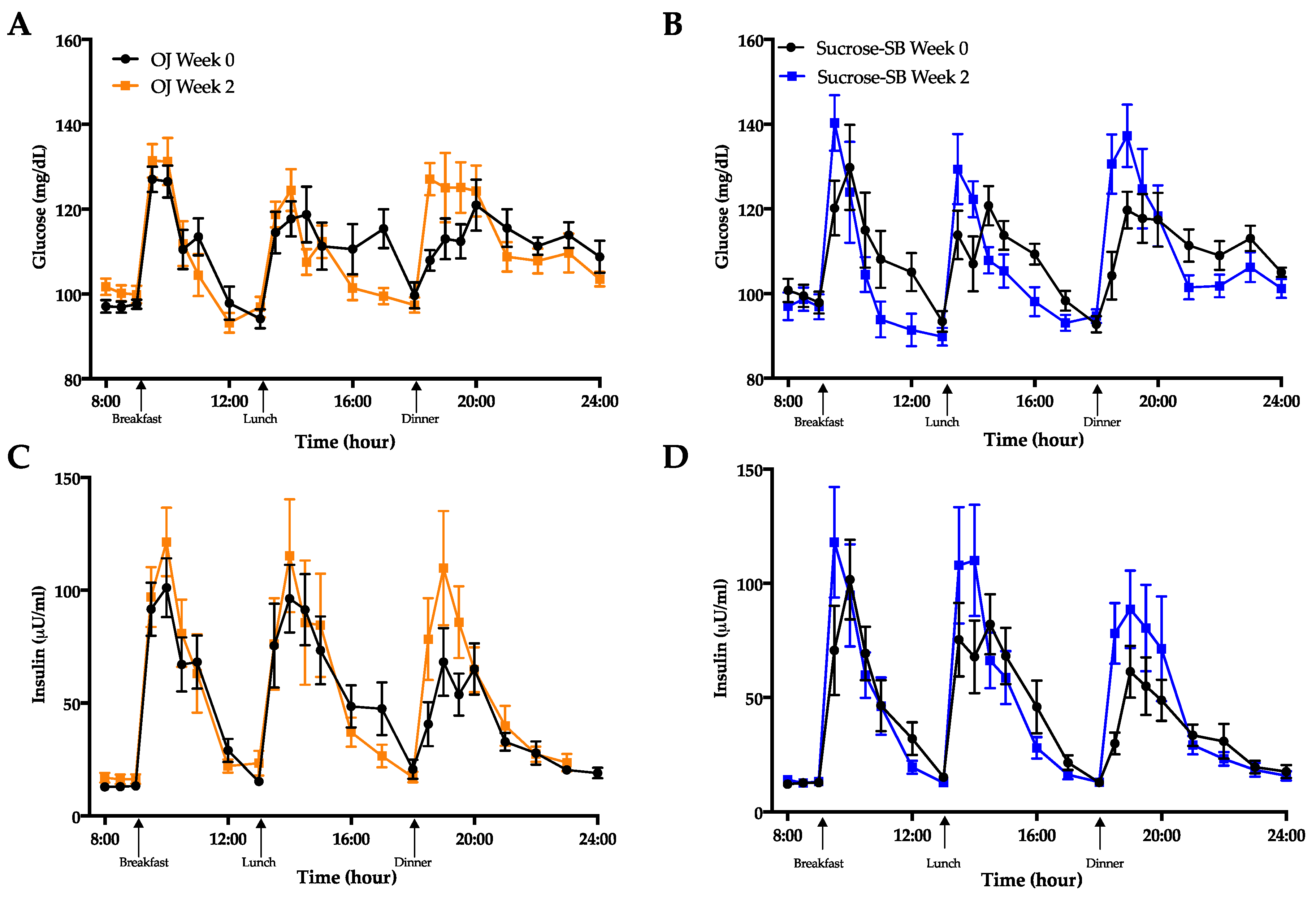
3.5. Indices of Insulin Sensitivity and Post-Meal Glycemic Response
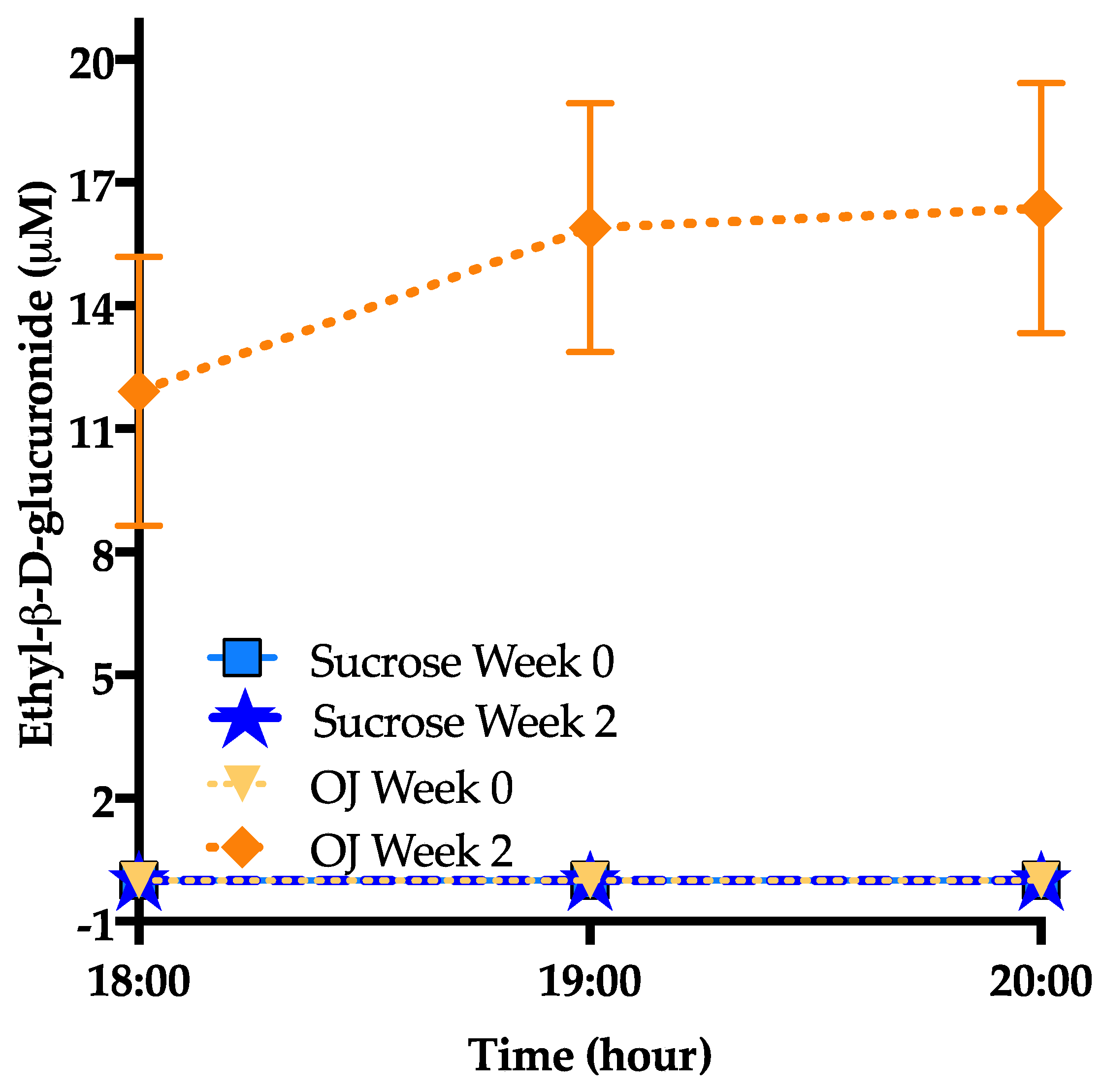
3.6. Ethyl-β-d-Glucuronide
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stanhope, K.L. Sugar consumption, metabolic disease and obesity: The state of the controversy. Crit. Rev. Clin. Lab. Sci. 2016, 53, 52–67. [Google Scholar] [CrossRef]
- Stanhope, K.L.; Goran, M.I.; Bosy-Westphal, A.; King, J.C.; Schmidt, L.A.; Schwarz, J.-M.; Stice, E.; Sylvetsky, A.C.; Turnbaugh, P.J.; Bray, G.A.; et al. Pathways and mechanisms linking dietary components to cardiometabolic disease: Thinking beyond calories. Obes. Rev. 2018, 19, 1205–1235. [Google Scholar] [CrossRef]
- Softic, S.; Stanhope, K.L.; Boucher, J.; Divanovic, S.; Lanaspa, M.A.; Johnson, R.J.; Kahn, C.R. Fructose and hepatic insulin resistance. Crit. Rev. Clin. Lab. Sci. 2020, 57, 308–322. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Schwarz, J.M.; Keim, N.L.; Griffen, S.C.; Bremer, A.A.; Graham, J.L.; Hatcher, B.; Cox, C.L.; Dyachenko, A.; Zhang, W.; et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J. Clin. Investig. 2009, 119, 1322–1334. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Medici, V.; Bremer, A.A.; Lee, V.; Lam, H.D.; Nunez, M.V.; Chen, G.X.; Keim, N.L.; Havel, P.J. A dose-response study of consuming high-fructose corn syrup–sweetened beverages on lipid/lipoprotein risk factors for cardiovascular disease in young adults. Am. J. Clin. Nutr. 2015, 101, 1144–1154. [Google Scholar] [CrossRef] [PubMed]
- Adiels, M.; Taskinen, M.-R.; Packard, C.; Caslake, M.J.; Soro-Paavonen, A.; Westerbacka, J.; Vehkavaara, S.; Häkkinen, A.; Olofsson, S.-O.; Yki-Järvinen, H.; et al. Overproduction of large VLDL particles is driven by increased liver fat content in man. Diabetologia 2006, 49, 755–765. [Google Scholar] [CrossRef]
- Batt, C.; Phipps-Green, A.J.; Black, A.M.; Cadzow, M.; Merriman, E.M.; Topless, R.; Gow, P.; Harrison, A.; Highton, J.; Jones, P.; et al. Sugar-sweetened beverage consumption: A risk factor for prevalent gout withSLC2A9genotype-specific effects on serum urate and risk of gout. Ann. Rheum. Dis. 2013, 73, 2101–2106. [Google Scholar] [CrossRef]
- Sánchez-Lozada, L.G.; Lanaspa, M.A.; Cristóbal-García, M.; García-Arroyo, F.; Soto, V.; Cruz-Robles, D.; Nakagawa, T.; Yu, A.M.; Kang, D.-H.; Johnson, R.J. Uric Acid-Induced Endothelial Dysfunction Is Associated with Mitochondrial Alterations and Decreased Intracellular ATP Concentrations. Nephron 2013, 121, e71–e78. [Google Scholar] [CrossRef]
- Soltani, Z.; Rasheed, K.; Kapusta, D.R.; Reisin, E. Potential Role of Uric Acid in Metabolic Syndrome, Hypertension, Kidney Injury, and Cardiovascular Diseases: Is It Time for Reappraisal? Curr. Hypertens. Rep. 2013, 15, 175–181. [Google Scholar] [CrossRef]
- Dehghan, A.; Van Hoek, M.; Sijbrands, E.J.; Hofman, A.; Witteman, J.C. High Serum Uric Acid as a Novel Risk Factor for Type 2 Diabetes. Diabetes Care 2007, 31, 361–362. [Google Scholar] [CrossRef]
- Walker, R.W.; Dumke, K.A.; Goran, M.I. Fructose content in popular beverages made with and without high-fructose corn syrup. Nutrients 2014, 30, 928–935. [Google Scholar] [CrossRef]
- Pereira-Caro, G.; Borges, G.; Van Der Hooft, J.; Clifford, M.N.; Del Rio, D.; Lean, M.E.; Roberts, S.A.; Kellerhals, M.B.; Crozier, A. Orange juice (poly)phenols are highly bioavailable in humans. Am. J. Clin. Nutr. 2014, 100, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Mullen, W.; Archeveque, M.-A.; Edwards, C.A.; Matsumoto, H.; Crozier, A. Bioavailability and Metabolism of Orange Juice Flavanones in Humans: Impact of a Full-Fat Yogurt. J. Agric. Food Chem. 2008, 56, 11157–11164. [Google Scholar] [CrossRef] [PubMed]
- Eshak, E.S.; Iso, H.; Mizoue, T.; Inoue, M.; Noda, M.; Tsugane, S. Soft drink, 100% fruit juice, and vegetable juice intakes and risk of diabetes mellitus. Clin. Nutr. 2013, 32, 300–308. [Google Scholar] [CrossRef]
- Fulgoni, V., III; Pereira, M.A. Consumption of 100% Fruit Juice and Risk of Obesity and Metabolic Syndrome: Findings from the National Health and Nutrition Examination Survey 1999–2004. FASEB J. 2009, 23, LB506. [Google Scholar]
- O’Neil, C.E.; Nicklas, T.A.; Rampersaud, G.C.; Fulgoni, V.L., 3rd. 100% orange juice consumption is associated with better diet quality, improved nutrient adequacy, decreased risk for obesity, and improved biomarkers of health in adults: National Health and Nutrition Examination Survey, 2003–2006. Nutr. J. 2012, 11, 107. [Google Scholar]
- Scheffers, F.R.; Wijga, A.H.; Verschuren, W.M.M.; Van Der Schouw, Y.T.; Sluijs, I.; Smit, A.H.; Boer, A.J.M. Pure Fruit Juice and Fruit Consumption Are Not Associated with Incidence of Type 2 Diabetes after Adjustment for Overall Dietary Quality in the European Prospective Investigation into Cancer and Nutrition–Netherlands (EPIC-NL) Study. J. Nutr. 2020, 150, 1470–1477. [Google Scholar] [CrossRef]
- Imamura, F.; O’Connor, L.; Ye, Z.; Mursu, J.; Hayashino, Y.; Bhupathiraju, S.N.; Forouhi, N.G. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: Systematic review, meta-analysis, and estimation of population attributable fraction. Br. J. Sports Med. 2016, 50, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Muraki, I.; Imamura, F.; Manson, E.J.; Hu, F.B.; Willett, W.C.; Van Dam, R.M.; Sun, Q. Fruit consumption and risk of type 2 diabetes: Results from three prospective longitudinal cohort studies. BMJ 2013, 347, f5001. [Google Scholar] [CrossRef]
- Semnani-Azad, Z.; Khan, T.A.; Blanco Mejia, S.; de Souza, R.J.; Leiter, L.A.; Kendall, C.W.C. Association of Major Food Sources of Fructose-Containing Sugars With Incident Metabolic Syndrome: A Systematic Review and Meta-analysis. JAMA Netw. Open. 2020, 3, e209993. [Google Scholar] [CrossRef] [PubMed]
- Pepin, A.; Stanhope, K.L.; Imbeault, P. Are Fruit Juices Healthier Than Sugar-Sweetened Beverages? A Review. Nutrients 2019, 11, 1006. [Google Scholar] [CrossRef]
- Dohadwala, M.M.; Hamburg, N.M.; Holbrook, M.; Kim, B.H.; Duess, M.-A.; Levit, A.; Titas, M.; Chung, W.B.; Vincent, F.B.; Caiano, T.L.; et al. Effects of Concord grape juice on ambulatory blood pressure in prehypertension and stage 1 hypertension. Am. J. Clin. Nutr. 2010, 92, 1052–1059. [Google Scholar] [CrossRef]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.M.; Shenouda, S.M.; Chung, W.B.; Titas, M.; Kluge, A.M.; Wang, N.; Palmisano, J.; Milbury, E.P.; et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef] [PubMed]
- Morand, C.; DuBray, C.; Milenkovic, D.; Lioger, D.; Martin, J.F.; Scalbert, A.; Mazur, A. Hesperidin contributes to the vascular protective effects of orange juice: A randomized crossover study in healthy volunteers. Am. J. Clin. Nutr. 2011, 93, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Simpson, L.; Mendis, B.; Macdonald, I.A. Orange juice consumption and its effect on blood lipid profile and indices of the metabolic syndrome; a randomised, controlled trial in an at-risk population. Food Funct. 2016, 7, 1884–1891. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guideline: Sugars Intake for Adults and Children. 2015. Available online: http://apps.who.int/iris/bitstream/10665/149782/1/9789241549028_eng.pdf (accessed on 5 June 2020).
- United States Department of Health and Human Services; US Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.December 2015. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 5 June 2020).
- Mifflin, M.D.; Jeor, S.T.S.; Hill, A.L.; Scott, B.J.; Daugherty, A.S.; Koh, O.Y. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [CrossRef]
- Gao, X.; Qi, L.; Qiao, N.; Choi, H.K.; Curhan, G.C.; Tucker, K.L.; Ascherio, A. Intake of Added Sugar and Sugar-Sweetened Drink and Serum Uric Acid Concentration in US Men and Women. Hypertension 2007, 50, 306–312. [Google Scholar] [CrossRef]
- Teff, K.L.; Grudziak, J.; Townsend, R.R.; Dunn, T.N.; Grant, R.W.; Adams, S.H.; Keim, N.L.; Cummings, B.P.; Stanhope, K.L.; Havel, P.J. Endocrine and Metabolic Effects of Consuming Fructose- and Glucose-Sweetened Beverages with Meals in Obese Men and Women: Influence of Insulin Resistance on Plasma Triglyceride Responses. J. Clin. Endocrinol. Metab. 2009, 94, 1562–1569. [Google Scholar] [CrossRef]
- American Diabetes Association. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes. Diabetes Care 2019, 42 (Suppl. 1), S13–S28. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services National Institutes of Health National Heart L, and Blood Institute. High Blood Cholesterol: What You Need to Know; National Cholesterol Education Program: Washington, DC, USA, 2015; (NIH Publication No. 05-3290).
- Zeballos, E.; Jessica, E.; Brandon, R. Frequency and Time of Day That Americans Eat: A Comparison of Data From the American Time Use Survey and the National Health and Nutrition Examination Survey; United States Department of Agriculture: Washington, DC, USA, 2019. [Google Scholar]
- Matsuda, M.; DeFronzo, R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing: Comparison with the euglycemic insulin clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Orozco, J.S.; Hertz-Picciotto, I.; Abbeduto, L.; Slupsky, C.M. Metabolomics analysis of children with autism, idiopathic-developmental delays, and Down syndrome. Transl. Psychiatry 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Gall, W.E.; Beebe, K.; Lawton, K.A.; Adam, K.-P.; Mitchell, M.W.; Nakhle, P.J.; Ryals, J.A.; Milburn, M.V.; Nannipieri, M.; Camastra, S.; et al. α-Hydroxybutyrate Is an Early Biomarker of Insulin Resistance and Glucose Intolerance in a Nondiabetic Population. PLoS ONE 2010, 5, e10883. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E.; Natali, A.; Camastra, S.; Nannipieri, M.; Mari, A.; Adam, K.-P.; Milburn, M.V.; Kastenmüller, G.; Adamski, J.; Tuomi, T.; et al. Early Metabolic Markers of the Development of Dysglycemia and Type 2 Diabetes and Their Physiological Significance. Diabetes 2012, 62, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Van de Luitgaarden, I.A.T.; Beulens, J.W.J.; Schrieks, I.C.; Kieneker, L.M.; Touw, D.J.; van Ballegooijen, A.J. Urinary Ethyl Glucuronide can be Used as a Biomarker of Habitual Alcohol Consumption in the General Population. J. Nutr. 2019, 149, 2199–2205. [Google Scholar] [CrossRef]
- Bruun, J.M.; Maersk, M.; Belza, A.; Astrup, A.; Richelsen, B. Consumption of sucrose-sweetened soft drinks increases plasma levels of uric acid in overweight and obese subjects: A 6-month randomised controlled trial. Eur. J. Clin. Nutr. 2015, 69, 949–953. [Google Scholar] [CrossRef]
- Cox, C.L.; Stanhope, K.L.; Schwarz, J.M.; Graham, J.L.; Hatcher, B.; Griffen, S.C.; Bremer, A.A.; Berglund, A.L.; McGahan, J.P.; Havel, P.J.; et al. Consumption of fructose-sweetened beverages for 10 weeks reduces net fat oxidation and energy expenditure in overweight/obese men and women. Eur. J. Clin. Nutr. 2011, 66, 201–208. [Google Scholar] [CrossRef]
- Perez-Pozo, S.E.; Nakagawa, T.; Sanchez-Lozada, L.G.; Johnson, R.J.; Lillo, J.L. Excessive fructose intake induces the features of metabolic syndrome in healthy adult men: Role of uric acid in the hypertensive response. Int. J. Obes. 2010, 34, 454–461. [Google Scholar] [CrossRef]
- Raivio, K.O.; Becker, M.A.; Meyer, L.J.; Greene, M.L.; Nuki, G.; Seegmiller, J. Stimulation of human purine synthesis de novo by fructose infusion. Metabolism 1975, 24, 861–869. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Kong, L.; Tang, Z.-Z.; Zhang, Y.-M.; Liu, Y.; Wang, T.-Y.; Liu, Y.-W. Hesperetin ameliorates diabetic nephropathy in rats by activating Nrf2/ARE/glyoxalase 1 pathway. Biomed. Pharmacother. 2019, 111, 1166–1175. [Google Scholar] [CrossRef]
- Haidari, F.; Keshavarz, S.A.; Rashidi, M.R.; Mohammadshahi, M. Orange Juice and Hesperetin Supplementation to Hyperuricemic Rats Alter Oxidative Stress Markers and Xanthine Oxidoreductase Activity. J. Clin. Biochem. Nutr. 2009, 45, 285–291. [Google Scholar] [CrossRef]
- Liu, K.; Wang, W.; Guo, B.-H.; Gao, H.; Liu, Y.; Liu, X.-H.; Yao, H.-L.; Cheng, K. Chemical Evidence for Potent Xanthine Oxidase Inhibitory Activity of Ethyl Acetate Extract of Citrus aurantium L. Dried Immature Fruits. Molecules 2016, 21, 302. [Google Scholar] [CrossRef]
- Ota-Kontani, A.; Hirata, H.; Ogura, M.; Tsuchiya, Y.; Harada-Shiba, M. Comprehensive analysis of mechanism underlying hypouricemic effect of glucosyl hesperidin. Biochem. Biophys. Res. Commun. 2020, 521, 861–867. [Google Scholar] [CrossRef] [PubMed]
- So, M.W.; Lim, D.H.; Kim, S.H.; Lee, S. Dietary and nutritional factors associated with hyperuricemia: The seventh Korean National Health and Nutrition Examination Survey. Asia Pac. J. Clin. Nutr. 2020, 29, 609–617. [Google Scholar]
- Sánchez-Moreno, C.; Cano, M.P.; De Ancos, B.; Plaza, L.; Olmedilla-Alonso, B.; Granado, F.; Martín, A. Effect of orange juice intake on vitamin C concentrations and biomarkers of antioxidant status in humans. Am. J. Clin. Nutr. 2003, 78, 454–460. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Miller, E.R., 3rd; Gelber, A.C. Effect of oral vitamin C supplementation on serum uric acid: A meta-analysis of randomized controlled trials. Arthritis Care Res. 2011, 63, 1295–1306. [Google Scholar] [CrossRef] [PubMed]
- Di Angelantonio, E.; Sarwar, N.; Perry, P.; Kaptoge, S.; Ray, K.K.; Thompson, A. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 2009, 302, 1993–2000. [Google Scholar] [PubMed]
- Aeberli, I.; Hochuli, M.; Gerber, P.A.; Sze, L.; Murer, S.B.; Tappy, L.; Spinas, G.A.; Berneis, K.; Espeland, M.A.; Bryan, R.N.; et al. Moderate Amounts of Fructose Consumption Impair Insulin Sensitivity in Healthy Young Men: A randomized controlled trial. Diabetes Care 2012, 36, 150–156. [Google Scholar] [CrossRef]
- Black, R.N.; Spence, M.; McMahon, R.O.; Cuskelly, G.J.; Ennis, C.N.; McCance, D.R. Effect of eucaloric high- and low-sucrose-SBdiets with identical macronutrient profile on insulin resistance and vascular risk: A randomized controlled trial. Diabetes 2006, 55, 3566–3572. [Google Scholar] [CrossRef] [PubMed]
- Reiser, S.; Bickard, M.C.; Hallfrisch, J.; Michaelis, E.O.; Prather, E.S. Blood lipids and their distribution in lipoproteins in hyperinsulinemic subjects fed three different levels of sucrose. J. Nutr. 1981, 111, 1045–1057. [Google Scholar] [CrossRef]
- Lima, A.C.D.; Cecatti, C.; Fidélix, M.P.; Adorno, M.A.T.; Sakamoto, I.K.; Cesar, T.B.; Sivieri, K. Effect of Daily Consumption of Orange Juice on the Levels of Blood Glucose, Lipids, and Gut Microbiota Metabolites: Controlled Clinical Trials. J. Med. Food 2019, 22, 202–210. [Google Scholar] [CrossRef]
- Fidélix, M.; Milenkovic, D.; Sivieri, K.; Cesar, T. Microbiota modulation and effects on metabolic biomarkers by orange juice: A controlled clinical trial. Food Funct. 2020, 11, 1599–1610. [Google Scholar] [CrossRef]
- Azzini, E.; Venneria, E.; Ciarapica, D.; Foddai, M.S.; Intorre, F.; Zaccaria, M.; Maiani, F.; Palomba, L.; Barnaba, L.; Tubili, C.; et al. Effect of Red Orange Juice Consumption on Body Composition and Nutritional Status in Overweight/Obese Female: A Pilot Study. Oxidative Med. Cell. Longev. 2017, 2017, 1672567. [Google Scholar] [CrossRef]
- Cesar, T.B.; Aptekmann, N.P.; Araujo, M.P.; Vinagre, C.C.; Maranhão, R.C. Orange juice decreases low-density lipoprotein cholesterol in hypercholesterolemic subjects and improves lipid transfer to high-density lipoprotein in normal and hypercholesterolemic subjects. Nutr. Res. 2010, 30, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Borradaile, N.M.; Carroll, K.K.; Kurowska, E.M. Regulation of HepG2 cell apolipoprotein B metabolism by the citrus flavanones hesperetin and naringenin. Lipids 1999, 34, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Allister Price, C.; Stanhope, K.L. Understanding the Impact of Added Sugar Consumption on Risk for Type 2 Diabetes. J. Calif. Dent. Assoc. 2016, 44, 619–626. [Google Scholar] [PubMed]
- Shaibi, G.Q.; Davis, J.N.; Weigensberg, M.J.; Goran, M.I. Improving insulin resistance in obese youth: Choose your measures wisely. Pediatr. Obes. 2011, 6, e290–e296. [Google Scholar] [CrossRef]
- Ghanim, H.; Mohanty, P.; Pathak, R.; Chaudhuri, A.; Sia, C.L.; Dandona, P. Orange Juice or Fructose Intake Does Not Induce Oxidative and Inflammatory Response. Diabetes Care 2007, 30, 1406–1411. [Google Scholar] [CrossRef]
- Ghanim, H.; Sia, C.L.; Upadhyay, M.; Korzeniewski, K.; Viswanathan, P.; Abuaysheh, S.; Mohanty, P.; Dandona, P. Orange juice neutralizes the proinflammatory effect of a high-fat, high-carbohydrate meal and prevents endotoxin increase and Toll-like receptor expression. Am. J. Clin. Nutr. 2010, 91, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Li, J.M.; Che, C.T.; Lau, C.B.; Leung, P.S.; Cheng, C.H. Inhibition of intestinal and renal Na+-glucose cotransporter by naringenin. Int. J. Biochem. Cell Biol. 2006, 38, 985–995. [Google Scholar] [CrossRef]
- Gorgus, E.; Hittinger, M.; Schrenk, D. Estimates of Ethanol Exposure in Children from Food not Labeled as Alcohol-Containing. J. Anal. Toxicol. 2016, 40, 537–542. [Google Scholar] [CrossRef]
- Neumann, T.; Helander, A.; Dahl, H.; Holzmann, T.; Neuner, B.; Weiss-Gerlach, E.; Müller, C.; Spies, C.; Weiß-Gerlach, E. Value of Ethyl Glucuronide in Plasma as a Biomarker for Recent Alcohol Consumption in the Emergency Room. Alcohol Alcohol. 2008, 43, 431–435. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lundquist, F.; Tygstrup, N.; Winkler, K.; Mellemgaard, K.; Munck-Petersen, S. Ethanol metabolism and production of free acetate in the human liver. J. Clin. Investig. 1962, 41, 955–961. [Google Scholar] [CrossRef] [PubMed]
| OJ | Sucrose | Effect of Group | |
|---|---|---|---|
| (n = 10) | (n = 10) | P | |
| Age (years) | 33.2 ± 1.2 | 31.2 ± 1.7 | 0.35 |
| Body weight (kg) | 80.1 ± 3.8 | 87.4 ± 3.8 | 0.44 |
| Body mass index (kg/m2) | 30.2 ± 1.0 | 31.0 ± 1.0 | 0.98 |
| % Body fat | 40.5 ± 2.1 | 40.2 ± 1.4 | 0.88 |
| Waist circumference (cm) | 88.3 ± 4.5 | 86.3 ± 2.4 | 0.70 |
| OJ (n = 10) | Sucrose (n = 10) | Effect of Beverage | |||
|---|---|---|---|---|---|
| Baseline Week 0 | Intervention Week 2 | Baseline Week 0 | Intervention Week 2 | ||
| Mean ± SEM | Mean ± SEM | Mean ± SEM | Mean ± SEM | P | |
| Body weight (kg) | 80.1 ± 3.8 | 80.8 ± 3.6 | 87.4 ± 3.8 | 88.2 ± 3.8 * | 0.44 |
| Systolic blood pressure (mmHg) | 111.6 ± 2.1 | 115.3 ± 3.1 | 121.7 ± 4.2 | 121.8 ± 2.2 | 0.47 |
| Diastolic blood pressure (mmHg) | 71.5 ± 2.4 | 70.2 ± 2.7 | 69.7 ± 3.2 | 70.3 ± 2.4 | 0.88 |
| Fasting uric acid (mgdL) | 5.4 ± 0.4 | 5.2 ± 0.4 | 5.0 ± 0.3 | 5.3 ± 0.3 | 0.10 |
| AUC uric acid (mg/dL × 16-h) 2 | 78.5 ± 5.6 | 75.3 ± 5.3 | 70.3 ± 3.8 | 76.5 ± 4.1 * | 0.008 |
| Fasting total cholesterol (mg/dL) 2 | 172.7 ± 8.7 | 174.7 ± 8.3 | 167.5 ± 12.3 | 174.9 ± 9.2 | 0.48 |
| Postprandial total cholesterol (mg/dL) 2 | 163.5 ± 8.2 | 168.5 ± 7.1 | 158.8 ± 10.2 | 169.3 ± 9.4 * | 0.40 |
| Fasting LDL-C (mg/dL) 2 | 123.3 ± 9.9 | 126.8 ± 10.1 | 110.3 ± 8.3 | 120.8 ± 7.3 * | 0.47 |
| Postprandial LDL-C (mg/dL) 2 | 118.2 ± 9.5 | 124.3 ± 9.8 | 105.7 ± 6.6 | 118.9 ± 7.2 ** | 0.32 |
| Fasting non-HDL-C (mg/dL) | 133.7 ± 8.7 | 130.3 ± 7.9 | 119.4 ± 9.0 | 124.1 ± 6.9 | 0.62 |
| Postprandial non-HDL-C (mg/dL) 1 | 122.2 ± 8.6 | 127.9 ± 6.9 | 112.2 ± 7.2 | 121.55 ± 7.4 * | 0.71 |
| Fasting ApoB (mg/dL) 2 | 63.2 ± 5.8 | 62.7 ± 5.9 | 54.0 ± 5.7 | 57.9 ± 4.9 | 0.35 |
| Postprandial ApoB (mg/dL) 2,3 | 61.4 ± 6.2 | 62.6 ± 5.0 | 54.2 ± 5.4 | 60.4 ± 5.7 * | 0.31 |
| Fasting HDL (mg/dL) | 43.4 ± 2.1 | 44.4 ± 2.4 | 48.2 ± 4.6 | 50.8 ± 4.4 | 0.42 |
| Postprandial HDL (mg/dL) | 41.3 ± 2.3 | 41.5 ± 2.6 | 46.6 ± 4.2 | 47.8 ± 3.8 | 0.43 |
| Fasting TG (mg/dL) | 103.9 ± 15.7 | 97.8 ± 15.0 | 87.6 ± 33.5 | 95.7 ± 14.8 | 0.24 |
| Postprandial TG (mg/dL) 1,2 | 102.8 ± 12.3 | 122.8 ± 17.3 * | 92.2 ± 12.7 | 113.6 ± 21.5 * | 0.82 |
| TG AUC (mg/dL × 16-h) | 1855.4 ± 257.8 | 1971.0 ± 313.4 | 1606.11 ± 220.6 | 1713.0 ± 279.3 | 0.89 |
| Fasting ApoCIII (mg/dL) 2 | 8.4 ± 1.0 | 8.4 ± 1.2 | 7.7 ± 1.2 | 8.1 ± 1.3 | 0.65 |
| Postprandial ApoCIII (mg/dL) 2 | 7.6 ± 0.8 | 8.7 ± 1.2 | 7.5 ± 1.3 | 8.7 ± 1.5 | 0.97 |
| OJ (n = 10) | Sucrose (n = 10) | ||||
|---|---|---|---|---|---|
| Baseline | Intervention | Baseline | Intervention | Effect of Beverage | |
| Week 0 | Week 2 | Week 0 | Week 2 | ||
| Mean ± SEM | Mean ± SEM | Mean ± SEM | Mean ± SEM | P | |
| Matsuda Index 1 | 2.8 ± 0.4 | 2.5 ± 0.4 * | 4.4 ± 1.1 | 3.4 ± 0.8 ** | 0.53 |
| OGTT Glucose AUC (mg/dL × 180 min) | 359.3 ± 16.6 | 391.0 ± 16.3 | 345.7 ± 11.7 | 399.5 ± 27.0 * | 0.44 |
| OGTT Insulin AUC (µU/mL × 180 min) | 282.9 ± 78.8 | 443.6 ± 166.9 * | 188.1 ± 40.5 | 226.9 ± 40.7 | 0.53 |
| Glucose, insulin and HOMA-IR from FBD I on day 1 of the 24-h blood collection period | |||||
| Fasting glucose (mg/dL) | 97.1 ± 1.5 | 101.7 ± 2.0 * | 100.8 ± 2.8 | 97.0 ± 3.2 | 0.006 |
| Fasting insulin (µU/mL) | 13.1 ± 0.9 | 16.5 ± 2.2 * | 12.5 ± 1.2 | 13.3 ± 1.5 | 0.16 |
| HOMA-IR 2 | 3.1 ± 0.2 | 4.2 ± 0.6 ** | 3.1 ± 0.3 | 3.2 ± 0.4 | 0.04 |
| Glucose, insulin and HOMA-IR from FBD II on day 2 of the 24-h blood collection period | |||||
| Fasting glucose (mg/dL) | 98.7 ± 1.3 | 98.7 ± 1.5 | 97.1 ± 2.3 | 97.8 ± 2.6 | 0.70 |
| Fasting insulin (µU/mL) | 16.3 ± 1.3 | 16.1 ± 1.8 | 11.8 ± 1.5 | 14.0 ± 1.7 | 0.39 |
| HOMA-IR 2 | 4.0 ± 0.3 | 4.0 ± 0.5 | 2.9 ± 0.4 | 3.4 ± 0.4 | 0.41 |
| 2-hydroxybutyrate (µM) | 26.6 ± 2.3 | 37.1 ± 2.4 ** | 26.8 ± 3.2 | 36.4 ± 3.9 ** | 0.66 |
| 16-h Glucose AUC (mg/dL × 16-h) | 1769.3 ± 35.0 | 1750.4 ± 35.0 | 1740.9 ± 39.4 | 1702.0 ± 39.4 | 0.38 |
| 16-h Insulin AUC (µU/mL × 16-h) | 757.0 ± 10.91 | 820.8 ± 141.2 | 658.7 ± 99.3 | 711.7 ± 112.5 | 0.87 |
| Mean Glucose AMP (mg/dL) | 37.2 ± 1.4 | 41.7 ± 1.8 | 39.4 ± 3.4 | 55.0 ± 5.8 *** | 0.04 |
| Mean Insulin AMP (µU/mL) | 85.1 ± 10.5 | 108.3 ± 18.4 ** | 79.4 ± 14.3 | 114.9 ± 20.6 *** | 0.20 |
| OJ | Sucrose | Effect of Beverage | |
|---|---|---|---|
| Mean ± SEM | Mean ± SEM | P | |
| Glucose post-breakfast AMP (mg/dL) | 3.0 ± 42 | 8.9± 6.1 * | 0.14 |
| Glucose post-lunch AMP (mg/dL) | 0.75 ± 5.8 | 17.2 ± 8.9 * | 0.20 |
| Glucose post-dinner AMP (mg/dL) | 9.6 ± 4.7 | 20.6 ± 6.7 ** | 0.16 |
| Mean Glucose AMP (mg/dL) | 4.5 ± 2.0 | 15.6 ± 4.4 *** | 0.04 |
| Insulin post-breakfast AMP (µU/mL) | 20.8 ± 5.0 *** | 26.7 ± 7.9 ** | 0.52 |
| Insulin post-lunch AMP (µU/mL) | 7.5 ± 12.4 | 31.8 ± 19.5 | 0.23 |
| Insulin post-dinner AMP (µU/mL) | 41.4 ± 18.0 * | 48.1 ± 15.9 * | 0.76 |
| Mean Insulin AMP (µU/mL) | 31.2 ± 12.8 ** | 42.5 ± 10.7 *** | 0.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Price, C.A.; Medici, V.; Nunez, M.V.; Lee, V.; Sigala, D.M.; Benyam, Y.; Keim, N.L.; Mason, A.E.; Chen, S.-Y.; Parenti, M.; et al. A Pilot Study Comparing the Effects of Consuming 100% Orange Juice or Sucrose-Sweetened Beverage on Risk Factors for Cardiometabolic Disease in Women. Nutrients 2021, 13, 760. https://doi.org/10.3390/nu13030760
Price CA, Medici V, Nunez MV, Lee V, Sigala DM, Benyam Y, Keim NL, Mason AE, Chen S-Y, Parenti M, et al. A Pilot Study Comparing the Effects of Consuming 100% Orange Juice or Sucrose-Sweetened Beverage on Risk Factors for Cardiometabolic Disease in Women. Nutrients. 2021; 13(3):760. https://doi.org/10.3390/nu13030760
Chicago/Turabian StylePrice, Candice Allister, Valentina Medici, Marinelle V. Nunez, Vivien Lee, Desiree M. Sigala, Yanet Benyam, Nancy L. Keim, Ashley E. Mason, Shin-Yu Chen, Mariana Parenti, and et al. 2021. "A Pilot Study Comparing the Effects of Consuming 100% Orange Juice or Sucrose-Sweetened Beverage on Risk Factors for Cardiometabolic Disease in Women" Nutrients 13, no. 3: 760. https://doi.org/10.3390/nu13030760
APA StylePrice, C. A., Medici, V., Nunez, M. V., Lee, V., Sigala, D. M., Benyam, Y., Keim, N. L., Mason, A. E., Chen, S.-Y., Parenti, M., Slupsky, C., Epel, E. S., Havel, P. J., & Stanhope, K. L. (2021). A Pilot Study Comparing the Effects of Consuming 100% Orange Juice or Sucrose-Sweetened Beverage on Risk Factors for Cardiometabolic Disease in Women. Nutrients, 13(3), 760. https://doi.org/10.3390/nu13030760