Vitamin D and Lung Outcomes in Elderly COVID-19 Patients
Abstract
1. Introduction
2. Materials and Methods
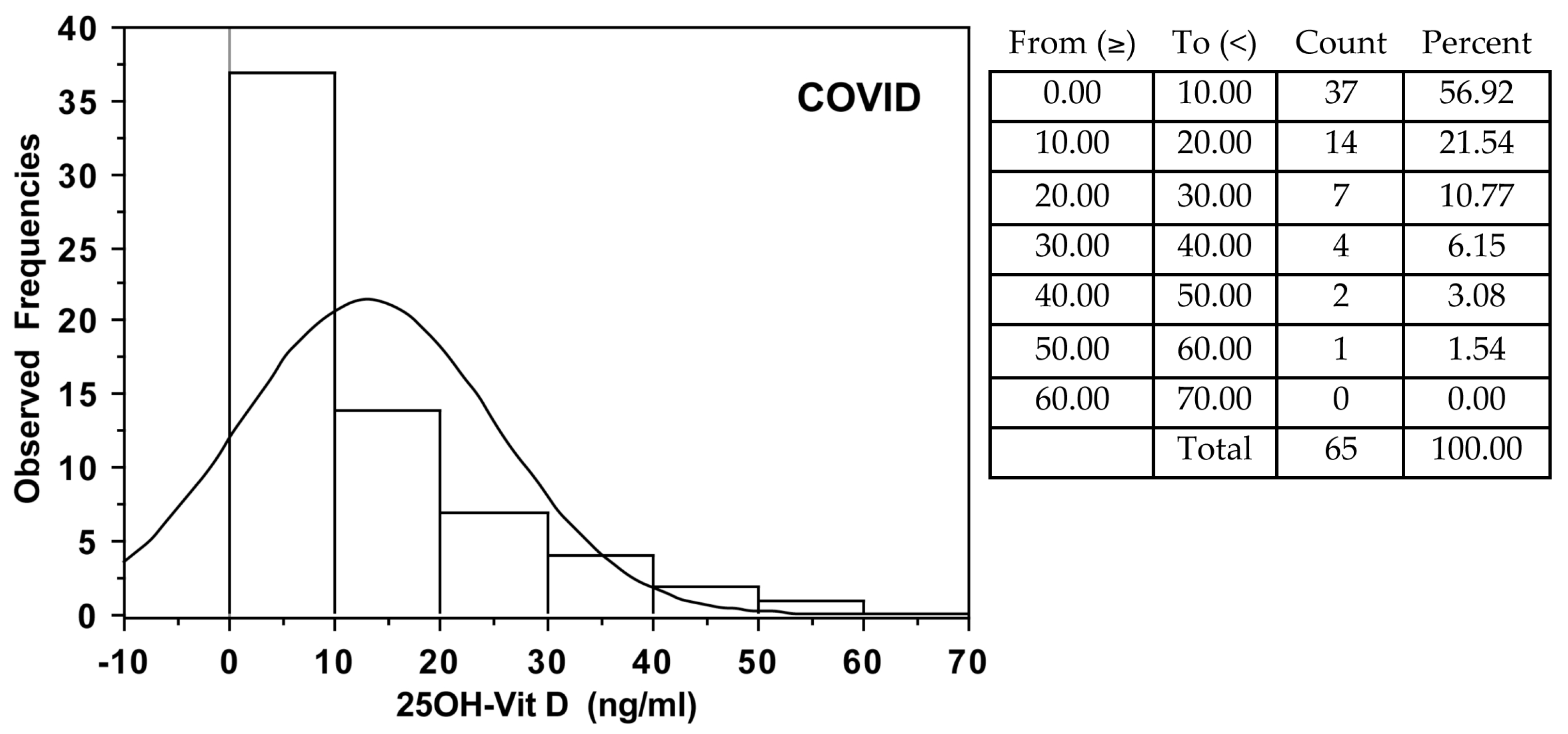
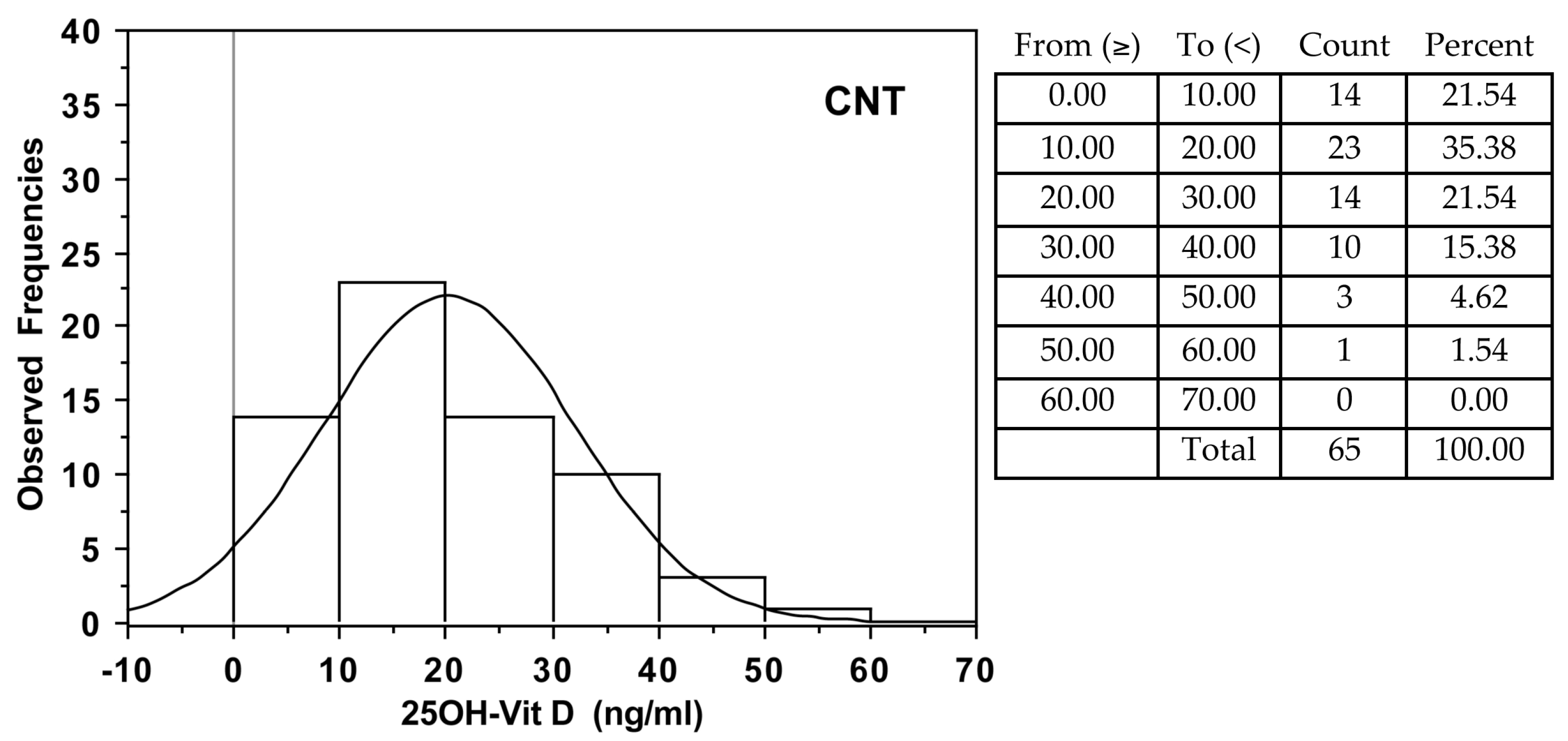
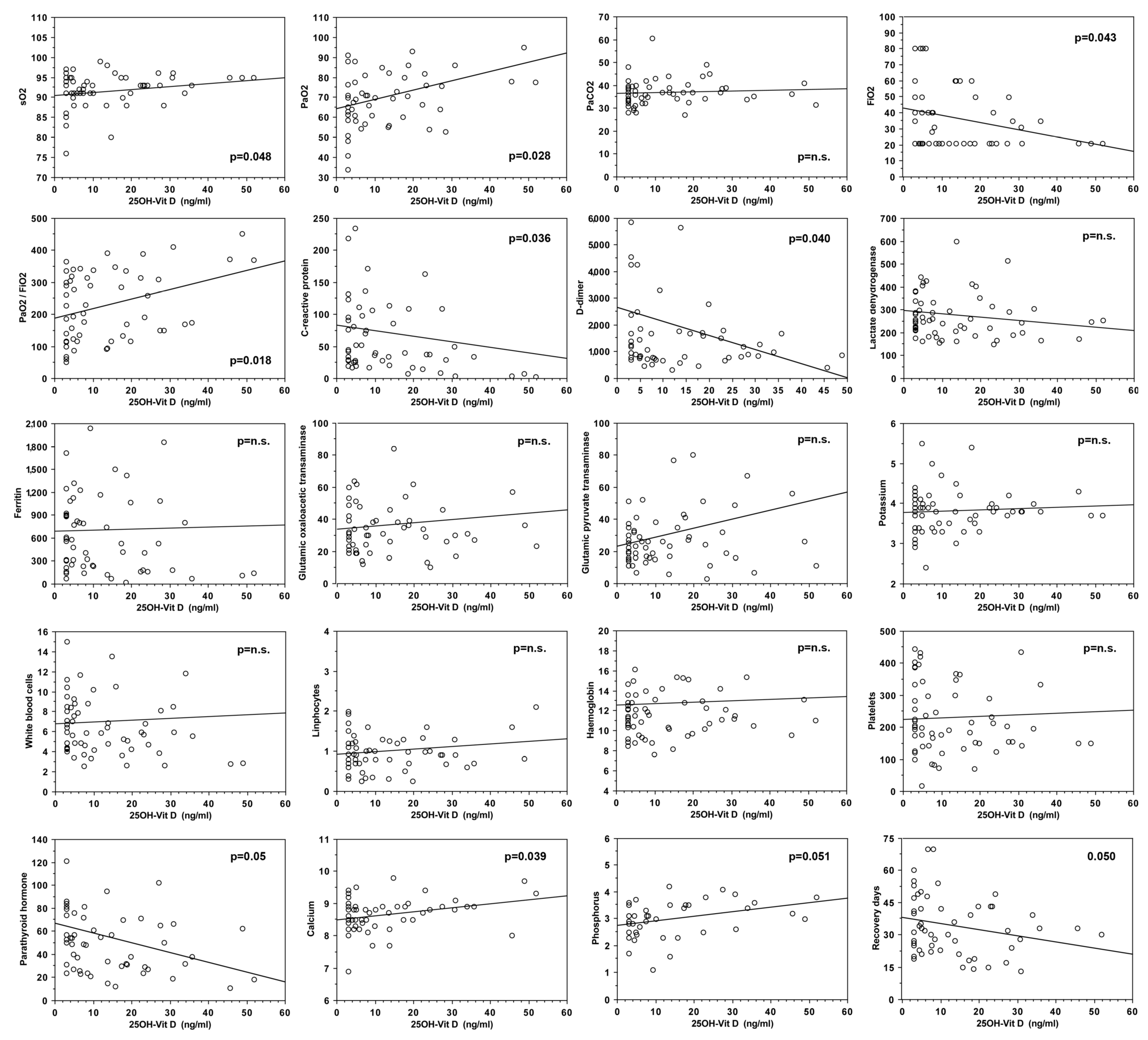
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cutolo, M.; Pizzorni, C.; Sulli, A. Vitamin D endocrine system involvement in autoimmune rheumatic diseases. Autoimmun. Rev. 2011, 11, 84–87. [Google Scholar] [CrossRef]
- Cutolo, M.; Plebani, M.; Shoenfeld, Y.; Adorini, L.; Tincani, A. Vitamin D endocrine system and the immune response in rheumatic diseases. Vitam. Horm. 2011, 86, 327–351. [Google Scholar] [PubMed]
- Adorini, L.; Penna, G. Control of autoimmune diseases by the vitamin D endocrine system. Nat. Clin. Pract. Rheumatol. 2008, 4, 404–412. [Google Scholar] [CrossRef]
- Dall’Ara, F.; Cutolo, M.; Andreoli, L.; Tincani, A.; Paolino, S. Vitamin D and systemic lupus erythematous: A review of immunological and clinical aspects. Clin. Exp. Rheumatol. 2018, 36, 153–162. [Google Scholar]
- Cutolo, M.; Paolino, S.; Sulli, A.; Smith, V.; Pizzorni, C.; Seriolo, B. Vitamin D, steroid hormones, and autoimmunity. Ann. N. Y. Acad. Sci. 2014, 1317, 39–46. [Google Scholar] [CrossRef]
- Zittermann, A.; Pilz, S.; Hoffmann, H.; März, W. Vitamin D and airway infections: A European perspective. Eur. J. Med. Res. 2016, 21, 14. [Google Scholar] [CrossRef]
- Weir, E.K.; Thenappan, T.; Bhargava, M.; Chen, Y. Does vitamin D deficiency increase the severity of COVID-19? Clin. Med. 2020, 20, e107–e108. [Google Scholar] [CrossRef]
- Munshi, R.; Hussein, M.H.; Toraih, E.A.; Elshazli, R.M.; Jardak, C.; Sultana, N.; Youssef, M.R.; Omar, M.; Attia, A.S.; Fawzy, M.S.; et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J. Med. Virol. 2021, 93, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.; Binkley, N.; Bischoff-Ferrari, H.A.; Gordon, C.; Hanley, D.; Heaney, R.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Bilezikian, J.P.; Bikle, D.; Hewison, M.; Lazaretti-Castro, M.; Formenti, A.M.; Gupta, A.; Madhavan, M.V.; Nair, N.; Babalyann, V.; Hutchings, N.; et al. Mechanisms in Endocrinology: Vitamin D and COVID-19. Eur. J. Endocrinol. 2020, 183, R133–R147. [Google Scholar] [CrossRef] [PubMed]
- Vojinovic, J.; Tincani, A.; Sulli, A.; Soldano, S.; Andreoli, L.; Dall’Ara, F.; Ionescu, R.; Pasalic, K.S.; Balcune, I.; Ferraz-Amaro, I.; et al. European multicentre pilot survey to assess vitamin D status in rheumatoid arthritis patients and early development of a new Patient Reported Outcome questionnaire (D-PRO). Autoimmun. Rev. 2017, 16, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Cutolo, M. Further emergent evidence for the vitamin D endocrine system involvement in autoimmune rheumatic disease risk and prognosis. Ann. Rheum. Dis. 2013, 72, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Orgaz-Molina, J.; Buendia-Eisman, A.; Arrabal-Polo, M.A.; Ruiz, J.C.; Arias-Santiago, S. Deficiency of serum concentration of 25-hydroxyvitamin D in psoriatic patients: A case-control study. J. Am. Acad. Derm. 2012, 67, 931–938. [Google Scholar] [CrossRef]
- Trombetta, A.C.; Smith, V.; Gotelli, E.; Ghio, M.; Paolino, S.; Pizzorni, C.; Vanhaecke, A.; Ruaro, B.; Sulli, A.; Cutolo, M. Vitamin D deficiency and clinical correlations in systemic sclerosis patients: A retrospective analysis for possible future developments. PLoS ONE 2017, 12, e0179062. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients. 2020, 12, 2097. [Google Scholar] [CrossRef]
- Science, M.; Maguire, J.L.; Russell, M.L.; Smieja, M.; Walter, S.D.; Loeb, M. Low serum 25-hydroxyvitamin D level and risk of upper respiratory tract infection in children and adolescents. Clin. Infect Dis. 2013, 57, 392–397. [Google Scholar] [CrossRef]
- Berry, D.J.; Hesketh, K.; Power, C.; Hypponen, E. Vitamin D status has a linear association with seasonal infections and lung function in British adults. Br. J. Nutr. 2011, 106, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Zhang, J.; Ma, C.; Yue, Y.; Zou, Z.; Yu, C.; Yin, F. Link between community-acquired pneumonia and vitamin D levels in older patients. Z. Gerontol. Geriatr. 2018, 51, 435–439. [Google Scholar] [CrossRef]
- Aibana, O.; Huang, C.C.; Aboud, S.; Arnedo-Pena, A.; Becerra, M.C.; Bellido-Blasco, J.B.; Bhosale, R.; Calderon, R.; Chiang, S.; Contreras, C.; et al. Vitamin D status and risk of incident tuberculosis disease: A nested case-control study, systematic review, and individual-participant data meta-analysis. PLoS Med. 2019, 16, e1002907. [Google Scholar] [CrossRef]
- Wang, M.X.; Koh, J.; Pang, J. Association between micronutrient deficiency and acute respiratory infections in healthy adults: A systematic review of observational studies. Nutr. J. 2019, 18, 80. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Im, J.H.; Je, Y.S.; Baek, J.; Chung, M.H.; Kwon, H.Y.; Lee, J.S. Nutritional status of patients with coronavirus disease 2019 (COVID-19). Int. J. Infect Dis. 2020, 100, 390–393. [Google Scholar] [CrossRef] [PubMed]
- Giustina, A.; Formenti, A.M. Preventing a covid-19 pandemic Can high prevalence of severe hypovitaminosis D play a role in the high impact of Covid infection in Italy? BMJ 2020, 368, m810. [Google Scholar]
- Mitchell, F. Vitamin-D and COVID-19: Do deficient risk a poorer outcome? Lancet Diabetes Endocrinol. 2020, 8, 570. [Google Scholar] [CrossRef]
- Hastie, C.E.; Mackay, D.F.; Ho, F.; Celis-Morales, C.A.; Katikireddi, S.V.; Niedzwiedz, C.L.; Jani, B.D.; Welsh, P.; Mair, F.S.; Gray, S.R.; et al. Vitamin D concentrations and COVID-19 infection in UK Biobank Diabetes. Metab. Syndr. 2020, 14, 561–565. [Google Scholar] [CrossRef]
- Panagiotou, G.; Tee, S.A.; Ihsan, Y.; Athar, W.; Marchitelli, G.; Kelly, D.; Boot, C.S.; Stock, N.; Macfarlane, J.; Martineau, A.R.; et al. Low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity. Clin. Endocrinol. 2020, 93, 508–511. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Griffin, G.; Kenny, R.A. Perspective: Vitamin D deficiency and COVID-19 severity-plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J. Intern. Med. 2020. [Google Scholar] [CrossRef]
- Kühn, J.; Trotz, P.; Stangl, G.I. Prevalence of vitamin D insufficiency and evidence for disease prevention in the older population. Z. Gerontol. Geriatr. 2018, 51, 567–572. [Google Scholar] [CrossRef]
- Hill, T.R.; Granic, A.; Davies, K.; Collerton, J.; Martin-Ruiz, C.; Siervo, M.; Mathers, J.C.; Adamson, A.J.; Francis, R.M.; Pearce, S.H.; et al. Serum 25-hydroxyvitamin D concentration and its determinants in the very old: The Newcastle 85+ Study. Osteoporos. Int. 2016, 27, 1199–1208. [Google Scholar] [CrossRef]
- ARDS Definition Task Force; Ranieri, V.M.; Rubenfeld, G.D.; Thompson, B.T.; Ferguson, N.D.; Caldwell, E.; Fan, E.; Camporota, L.; Slutsky, A.S. Acute respiratory distress syndrome: The Berlin Definition. JAMA 2012, 307, 2526–2533. [Google Scholar]
- Hardie, J.A.; Vollmer, W.M.; Buist SAEllingsen, I.; Mørkve, O. Reference values for arterial blood gases in the elderly. Chest. 2004, 125, 2053–2060. [Google Scholar] [CrossRef]
- Zhou, S.; Yang, Y.; Zhang, X.; Li, Z.; Liu, X.; Hu, C.; Chen, C.; Wang, D.; Peng, Z. Clinical Course of 195 Critically ILL COVID-19 Patients, A Retrospective Multi-Center Study. Shock 2020, 54, 644–651. [Google Scholar] [CrossRef]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Capozza, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J. Endocrinol. Invest. 2020. [Google Scholar] [CrossRef]
- Beard, J.A.; Bearden, A.; Striker, R. Vitamin D and the anti-viral state. J. Clin. Virol. 2011, 50, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Malek Mahdavi, A. A brief review of interplay between vitamin D and angiotensin-converting enzyme 2: Implications for a potential treatment for COVID-19. Rev. Med. Virol. 2020, 30, e2119. [Google Scholar] [CrossRef]
- Xu, X.; Chen, P.; Wang, J.; Feng, J.; Zhou, H.; Li, X.; Zhong, W.; Hao, P. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci. China Life Sci. 2020, 63, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Guo, Y.; Pan, Y.; Zhao, Z.J. Structure analysis of the receptor binding of 2019-nCoV. Biochem. Biophys. Res. Commun. 2020, 525, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet. 2020, 395, 565–574. [Google Scholar] [CrossRef]
- Kuba, K.; Imai, Y.; Rao, S.; Gao, H.; Guo, F.; Guan, B.; Huan, Y.; Yang, P.; Zhang, Y.; Deng, W.; et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat. Med. 2005, 11, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C. Molecular mechanism of vitamin D in the cardiovascular system. J. Investig. Med. 2011, 59, 868–871. [Google Scholar] [CrossRef]
- Machado, C.D.S.; Ferro Aissa, A.; Ribeiro, D.L.; Antunes, L.M.G. Vitamin D supplementation alters the expression of genes associated with hypertension and did not induce DNA damage in rats. J. Toxicol. Environ. Health A. 2019, 82, 299–313. [Google Scholar] [CrossRef]
- Bourgonje, A.R.; Abdulle, A.E.; Timens, W.; Hillebrands, J.L.; Navis, G.J.; Gordijn, S.J.; Bolling, M.C.; Dijkstra, G.; Voors, A.A.; Osterhaus, A.D.; et al. Angiotensin-converting enzyme 2 (ACE2), SARS-CoV-2 and the pathophysiology of coronavirus disease 2019 (COVID-19). J. Pathol. 2020, 251, 228–248. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Zhu, X.; Shi, Y.; Liu, T.; Chen, Y.; Bhan, I.; Zhao, Q.; Thadhani, R.; Li, Y.C. VDR attenuates acute lung injury by blocking Ang-2-Tie-2 pathway and renin-angiotensin system. Mol. Endocrinol. 2013, 27, 2116–2125. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.J. Thoughts on the alveolar phase of COVID-19. Am. J. Physiol. Lung Cell Mol. Physiol. 2020, 31, L115–L120. [Google Scholar] [CrossRef]
- Rehan, V.K.; Torday, J.S.; Peleg, S.; Gennaro, L.; Vouros, P.; Padbury, J.; Rao, D.S.; Reddy, G.S. 1Alpha,25-dihydroxy-3-epi-vitamin D3, a natural metabolite of 1alpha,25dihydroxy vitamin D3: Production and biological activity studies in pulmonary alveolar type II cells. Mol. Genet. Metab. 2002, 76, 46–56. [Google Scholar] [CrossRef]
- Phokela, S.S.; Peleg, S.; Moya, F.R.; Alcorn, J.L. Regulation of human pulmonary surfactant protein gene expression by 1alpha,25-dihydroxyvitamin D3. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 289, L617–L626. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wilson, R.; Bennett, E.; Zosky, G.R. Identification of vitamin D sensitive pathways during lung development. Respir. Res. 2016, 17, 47. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Meza, C.A.; Clarke, H.; Kim, J.S.; Hickner, R.C. Vitamin D and Endothelial Function. Nutrients 2020, 12, 575. [Google Scholar] [CrossRef]
- Mohammad, S.; Mishra, A.; Ashraf, M.Z. Emerging role of vitamin D and its associated molecules in pathways related to pathogenesis of thrombosis. Biomolecules 2019, 9, 649. [Google Scholar] [CrossRef]
- Kommoss, F.K.F.; Schwab, C.; Tavernar, L.; Schreck, J.; Wagner, W.L.; Merle, U.; Jonigk, D.; Schirmacher, P.; Longerich, T. The Pathology of Severe COVID-19-Related Lung Damage. Dtsch. Arztebl. Int. 2020, 117, 500–506. [Google Scholar]
- McGonagle, D.; O’Donnell, J.S.; Sharif, K.; Emery, P.; Bridgewood, C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol. 2020, 2, e437–e445. [Google Scholar] [CrossRef]
- Adam, S.S.; Key, N.S.; Greenberg, C.S. D-dimer antigen: Current concepts and future prospects. Blood 2009, 113, 2878–2887. [Google Scholar] [CrossRef] [PubMed]
- Sakka, M.; Connors, J.M.; Hékimian, G.; Martin-Toutain, I.; Crichi, B.; Colmegna, I.; Bonnefont-Rousselot, D.; Farge, D.; Frere, C. Association between D-Dimer levels and mortality in patients with coronavirus disease 2019 (COVID-19): A systematic review and pooled analysis. J. Med. Vasc. 2020, 45, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Baktash, V.; Hosack, T.; Patel, N.; Shah, S.; Kandiah, P.; Van den Abbeele, K.; Mandal, A.K.J.; Missouris, C.G. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med. J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cutolo, M.; Paolino, S.; Smith, V. Evidences for a protective role of vitamin D in COVID-19. RMD Open. 2020, 6, e001454. [Google Scholar] [CrossRef]
- Sassi, F.; Tamone, C.; D’Amelio, P. Vitamin D: Nutrient, hormone, and immunomodulator. Nutrients 2018, 10, 1656. [Google Scholar] [CrossRef] [PubMed]
- Waldron, J.L.; Ashby, H.L.; Cornes, M.P.; Bechervaise, J.; Razavi, C.; Thomas, O.L.; Chugh, S.; Deshpande, S.; Ford, C.; Gama, R. Vitamin D: A negative acute phase reactant. J. Clin. Pathol. 2013, 66, 620–622. [Google Scholar] [CrossRef]
- Ohaegbulam, K.C.; Swalih, M.; Patel, P.; Smith, M.A.; Perrin, R. Vitamin D Supplementation in COVID-19 Patients: A Clinical Case Series. Am. J Ther. 2020, 27, e485–e490. [Google Scholar] [CrossRef]
- Kaminetzky, M.; Moore, W.; Fansiwala, K.; Babb, J.S.; Kaminetzky, D.; HorwitzL, I.; McGuinness, G.; Knoll, A.; Ko, J.P. Pulmonary Embolism on CTPA in COVID-19 Patients. Radiol. Cardiothorac. Imaging. 2020, 2, e200308. [Google Scholar] [CrossRef]
- Grosse, C.; Grosse, A.; Salzer, H.J.F.; Dünser, M.W.; Motz, R.; Langer, R. Analysis of cardiopulmonary findings in COVID-19 fatalities: High incidence of pulmonary artery thrombi and acute suppurative bronchopneumonia. Cardiovasc. Pathol. 2020, 49, 107263. [Google Scholar] [CrossRef]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed]
- Cianferotti, L.; Bertoldo, F.; Bischoff-Ferrari, H.A.; Bruyere, O.; Cooper, C.; Cutolo, M.; Kanis, J.A.; Kaufman, J.M.; Reginster, J.Y.; Rizzoli, R.; et al. Vitamin D supplementation in the prevention and management of major chronic diseases not related to mineral homeostasis in adults: Research for evidence and a scientific statement from the European society for clinical and economic aspects of osteoporosis and osteoarthritis (ESCEO). Endocrine 2017, 56, 245–261. [Google Scholar] [PubMed]
- Pereira, M.; Dantas Damascena, A.; Galvão Azevedo, L.M.; de Almeida Oliveira, T.; da Mota Santana, J. Vitamin D deficiency aggravates COVID-19: Systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]

| COVID-19 | CONTROLS | p | |
|---|---|---|---|
| Age (years, mean ± SD) | 76 ± 13 | 76 ± 13 | 0.814 |
| Disease duration (days, mean ± SD) | 13.1 ± 13 | - | - |
| Male/Female | 30/35 | 30/35 | - |
| Body mass index (kg/m2) | 26.3 | 25.4 | 0.634 |
| Ethnicity: Caucasian | 65/65 | 65/65 | 0.931 |
| Died during hospitalization | 10/65 | - | - |
| Recovery time (days, mean ± SD) | 33 ± 14 | - | - |
| Smoking status | 4/65 | 4/65 | 0.891 |
| Arterial hypertension | 35/65 (54%) | 36/65 (55%) | 0.281 |
| Previous cardiac/cerebral ischemic vasculopathy | 14/65 (21%) | 6/65 (11%) | 0.357 |
| Neoplasms | 11/65 (16%) | 5/65 (8%) | 0.091 |
| Recent hip or vertebral fracture | 12/65 (18%) | 11/65 (17%) | 0.916 |
| Diabetes | 10/65 (15%) | 9/65 (14%) | 0.737 |
| Chronic atrial fibrillation | 11/65 (17%) | 11/65 (17%) | 0.492 |
| Chronic obstructive pulmonary disease | 8/65(12%) | 3/65 (3%) | 0.213 |
| Chronic kidney disease | 5/65 (8%) | 4/65 (6%) | 0.652 |
| Dysthyroidism | 4/65 (6%) | 5/65 (8%) | 0.458 |
| Colic diverticulosis | 5/65 (8%) | 2/65 (3%) | 0.223 |
| Chronic arthritis (rheumatoid or psoriatic) | 2/65 (3%) | 7/65 (11%) | 0.049 |
| Epilepsy | 2/65 (3%) | 4/65 (6%) | 0.296 |
| Allergic asthma | 1/65 (2%) | 2/65 (3%) | 0.414 |
| Liver cirrhosis | 1/65 (2%) | 3/65 (5%) | 0.243 |
| Hepatitis B infection | 1/65 (2%) | 1/65 (2%) | 0.892 |
| No comorbidities | 7/65 (11%) | 10/65 (15%) | 0.490 |
| One comorbidity | 17/65 (26%) | 19/65 (29%) | 0.749 |
| Two comorbidities | 24/65 (37%) | 22/65 (34%) | 0.647 |
| Three comorbidities | 13/65 (20%) | 11/65 (17) | 0.547 |
| Four or more comorbidities | 4/65 (6%) | 3/65 (5%) | 0.616 |
| Vitamin D supplementation | 22/65 (34%) | 44/65 (68%) | 0.015 |
| COVID-19 | Normal Range | |
|---|---|---|
| SO2 (%) | 92 (4) | 95–99 |
| PaO2 (mmHg) | 69 (20) | 83–108 |
| PaCO2 (mmHg) | 37 (7) | 35–48 |
| FiO2 (%) | 31 (36) | 21 |
| PaO2/FiO2 | 211 (199) | >300 |
| D-dimer (mcg/L) | 1078 (1071) | 0–500 |
| C-reactive protein (mg/L) | 39 (78) | 0–5 |
| Ferritin (mcg/L) | 562 (746) | 30–400 |
| LDH—lactate dehydrogenase (UI/L) | 259 (117) | 135–225 |
| White blood cells (×109/L) | 5.9 (4.2) | 4.5–9.8 |
| Lymphocytes (×109/L) | 0.9 (0.6) | 1.1–4.8 |
| Haemoglobin (g/L) | 11.3 (2.9) | 12–17.5 |
| Platelets (×109/L) | 218 (166) | 130–430 |
| GOT—glutamic oxaloacetic transaminase (UI/L) | 34 (21) | 0–40 |
| GPT—glutamic-pyruvate transaminase (UI/L) | 29 (21) | 0–40 |
| Creatinine (mg/dL) | 0.9 (0.4) | 0.6–1.0 |
| Calcium (mg/dL) | 8.7 (0.5) | 8.5–11.0 |
| Phosphorus (mg/dL) | 3.2 (1.1) | 2.5–4.5 |
| PTH—parathyroid hormone (ng/L) | 51 (40) | 6.5–36.8 |
| COVID-19 | CNT | Statistical Significance | |
|---|---|---|---|
| All subjects | 7.9 (15) | 16.3 (19) | p = 0.001 |
| Died/Survived | 3.0 (8)/8.4 (18) | - | p = 0.046 |
| Male | 7.0 (12) * | 13.6 (20) * | p = 0.021 |
| Female | 9.3 (20) * | 18.3 (19) * | p = 0.0049 |
| Outcome | Predictors | Beta (95% CI) | p Value |
|---|---|---|---|
| PaO2/FiO2 (log) | 25OHD (ng/mL) (log) | 0.17 (0.01, 0.33) | 0.033 |
| Sex (female) | −0.02 (−0.31, 0.27) | 0.889 | |
| Age (years) | −0.00 (−0.01, 0.01) | 0.812 | |
| Comorbidities | −0.07 (−0.21, 0.06) | 0.272 | |
| SO2 (%) | 25OHD (ng/mL) (log) | 0.97 (−0.66, 2.60) | 0.240 |
| Sex (female) | −2.38 (−5.37, 0.61) | 0.116 | |
| Age (years) | 0.02 (−0.09, 0.13) | 0.713 | |
| Comorbidities | −1.08 (−2.48, 0.31) | 0.126 | |
| PaO2 (mmHg) | 25OHD (ng/mL) (log) | 5.18 (0.83, 9.52) | 0.021 |
| Sex (female) | −10.3 (−18.2, −2.3) | 0.012 | |
| Age (years) | −0.03 (−0.33, 0.27) | 0.823 | |
| Comorbidities | −1.37 (−5.06, 2.32) | 0.459 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulli, A.; Gotelli, E.; Casabella, A.; Paolino, S.; Pizzorni, C.; Alessandri, E.; Grosso, M.; Ferone, D.; Smith, V.; Cutolo, M. Vitamin D and Lung Outcomes in Elderly COVID-19 Patients. Nutrients 2021, 13, 717. https://doi.org/10.3390/nu13030717
Sulli A, Gotelli E, Casabella A, Paolino S, Pizzorni C, Alessandri E, Grosso M, Ferone D, Smith V, Cutolo M. Vitamin D and Lung Outcomes in Elderly COVID-19 Patients. Nutrients. 2021; 13(3):717. https://doi.org/10.3390/nu13030717
Chicago/Turabian StyleSulli, Alberto, Emanuele Gotelli, Andrea Casabella, Sabrina Paolino, Carmen Pizzorni, Elisa Alessandri, Marco Grosso, Diego Ferone, Vanessa Smith, and Maurizio Cutolo. 2021. "Vitamin D and Lung Outcomes in Elderly COVID-19 Patients" Nutrients 13, no. 3: 717. https://doi.org/10.3390/nu13030717
APA StyleSulli, A., Gotelli, E., Casabella, A., Paolino, S., Pizzorni, C., Alessandri, E., Grosso, M., Ferone, D., Smith, V., & Cutolo, M. (2021). Vitamin D and Lung Outcomes in Elderly COVID-19 Patients. Nutrients, 13(3), 717. https://doi.org/10.3390/nu13030717






