Baby-Led Weaning: What Role Does It Play in Obesity Risk during the First Years? A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Intervention
2.4. Outcome Measures
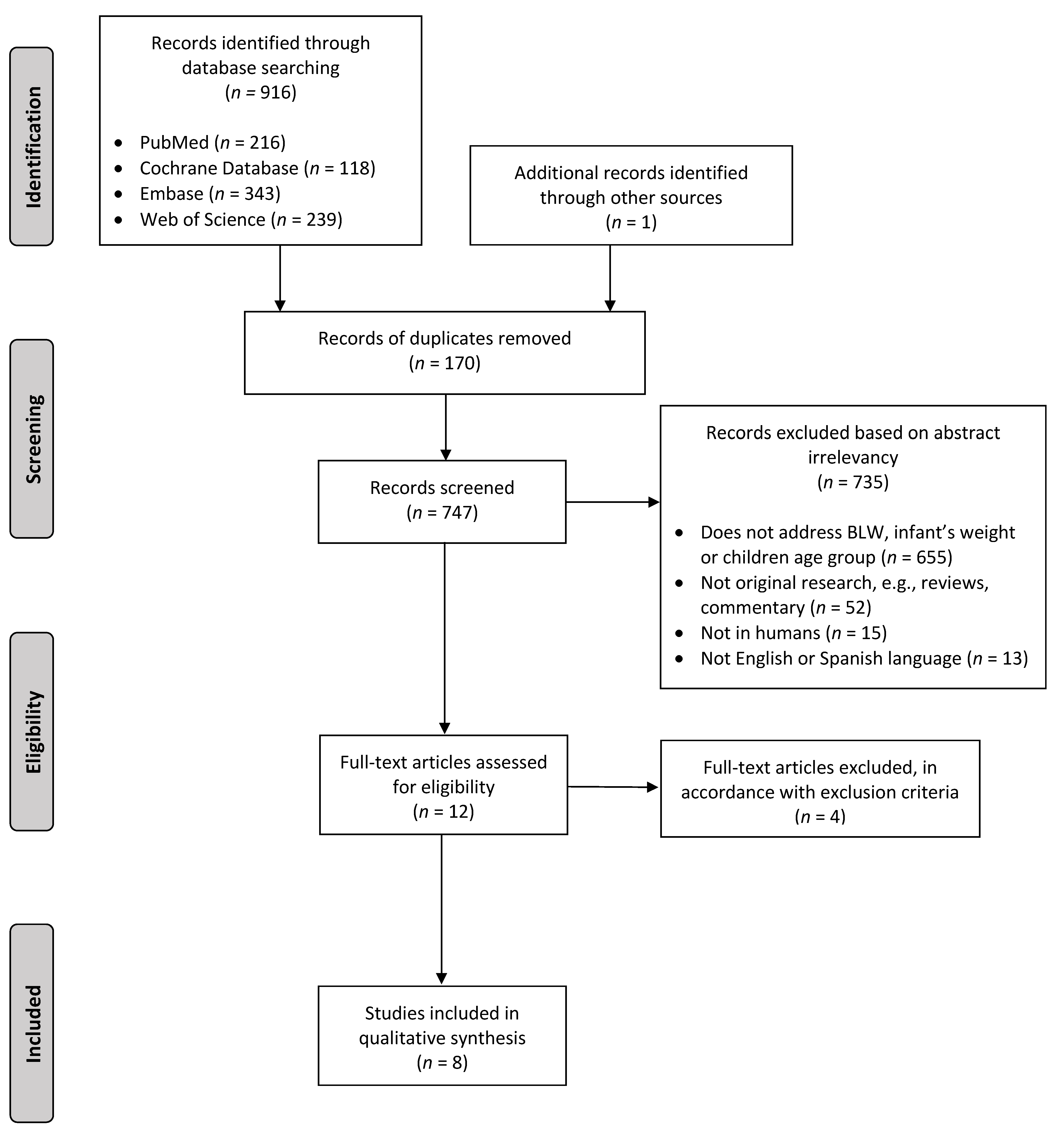
2.5. Study Selection
2.6. Data Extraction
2.7. Assessment of Risk of Bias
3. Results
3.1. Study Characteristics
3.2. Method of Weaning
3.3. Weight and Body Mass Index
3.4. Risk-of-Bias Assessment
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 1 November 2020).
- Latorre-Millán, M.; Rupérez, A.I.; González-Gil, E.M.; Santaliestra-Pasías, A.; Vázquez-Cobela, R.; Gil-Campos, M.; Aguilera, C.M.; Gil, A.; Moreno, L.A.; Leis, R.; et al. Dietary patterns and their association with body composition and cardiometabolic markers in children and adolescents: Genobox cohort. Nutrients 2020, 12, 3424. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.M.; Collado, M.C.; Larqué, E.; Leis, R.; Saenz, M.; Moreno, L.A. The first 1000 days: An opportunity to reduce the burden of noncommunicable diseases. Nutr Hosp. 2019, 36, 218–232. [Google Scholar]
- Plaza-Díaz, J.; Molina-Montes, E.; Soto-Méndez, M.J.; Madrigal, C.; Hernández-Ruiz, A.; Valero, T.; Villoslada, F.L.; Leis, R.; Martínez de Victoria, E.; Moreno, J.M.; et al. Clustering of dietary patterns and lifestyles among Spanish children in the EsNuPI Study. Nutrients 2020, 12, 2536. [Google Scholar] [CrossRef] [PubMed]
- Leis, R.; de Lamas, C.; de Castro, M.J.; Picáns, R.; Gil-Campo, M.; Couce, M.L. Effects of nutritional education interventions on metabolic risk in children and adolescents: A systematic review of controlled trials. Nutrients. 2020, 12, 31. [Google Scholar] [CrossRef] [PubMed]
- Onge, T.P.; Guest, P.C. Nutritional programming effects on development of metabolic disorders in later life. Methods Mol Biol. 2018, 1735, 3–17. [Google Scholar]
- World Health Organization. Complementary Feeding: Report of the Global Consultation and Summary of Guiding Principles. Available online: https://www.who.int/nutrition/publications/Complementary_Feeding.pdf (accessed on 1 November 2020).
- Papoutsou, S.; Savva, S.C.; Hunsberger, M.; Jilani, H.; Michels, N.; Ahrens, W.; Tornaritis, M.; Veidebaum, T.; Molnár, D.; Siani, A.; et al. Timing of solid food introduction and association with later childhood overweight and obesity: The IDEFICS study. Matern. Child Nutr. 2018, 14, e12471. [Google Scholar] [CrossRef]
- Moorcroft, K.E.; Marshall, J.L.; McCormick, F.M. Association between timing of introducing solid foods and obesity in infancy and childhood: A systematic review. Matern Child Nutr. 2011, 7, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Gingras, V.; Aris, I.M.; Rifas-Shiman, S.L.; Switkowski, K.M.; Oken, E.; Hivert, M. Timing of complementary feeding introduction and adiposity throughout childhood. Pediatrics 2019, 144, e20191320. [Google Scholar] [CrossRef]
- Smith-Brown, P.; Morrison, M.; Krause, L.; Davies, P.S.W. Microbiota and body composition during the period of complementary feeding. J. Pediatric Gastroenterol. Nutr. 2019, 69, 726–732. [Google Scholar] [CrossRef]
- World Health Organization (WHO). 55th World Health Assembly. Infant and Young Child Nutrition. 2002. Available online: https://www.who.int/nutrition/topics/WHA55.25_iycn_en.pdf (accessed on 1 November 2020).
- Fewtrell, M.; Bronsky, J.; Campoy, C.; Domellöf, M.; Embleton, N.; Mis, N.F.; Iva, H.; Hulst, J.M.; Flavia, I. Complementary feeding: A position paper by the European Society for Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) Committee on Nutrition. J. Pediatric Gastroenterol. Nutr. 2017, 64, 119–132. [Google Scholar] [CrossRef]
- American Academy of Pediatrics, Committee on Nutrition. Complementary feeding. In Pediatric Nutrition, 7th ed.; Kleinman, R.E., Freer, F.R., Eds.; American Academy of Pediatrics, Committee on Nutrition: Elk Grove Village, IL, USA, 2014; pp. 23–139. [Google Scholar]
- Martinón, N.; Picáns, R.; Leis, R. Complementary feeding according ESPGHAN, AAP and AEP Nutrition Committees recomendations. Acta Pediatr Esp. 2020, 78, 48–53. [Google Scholar]
- Rapley, G. Baby-led weaning. In Maternal and Infant Nutrition and Nurture: Controversies and Challenges; Hall, M.V., Dykes, F., Eds.; Quay Books: London, UK, 2005; pp. 275–298. [Google Scholar]
- Morison, B.J.; Taylor, R.W.; Haszard, J.J.; Schramm, C.J.; Erickson, L.W.; Fangupo, L.J.; Fleming, E.A.; Luciano, A.; Heath, A.M. How different are baby-led weaning and conventional complementary feeding? A cross-sectional study of infants aged 6–8 months. BMJ Open. 2016, 6, e010665. [Google Scholar] [CrossRef]
- Cameron, S.L.; Heath, A.M.; Taylor, R.W. Healthcare professionals’ and mothers’ knowledge of, attitudes to and experiences with, baby-led weaning: A content analysis study. BMJ Open 2012, 2, e001542. [Google Scholar] [CrossRef] [PubMed]
- Cameron, S.L.; Taylor, R.W.; Heath, A.L. Development and pilot testing of baby-led introduction to SolidS-a version of baby-led weaning modified to address concerns about iron deficiency, growth faltering and choking. BMC Pediatrics 2015, 15, 99. [Google Scholar] [CrossRef] [PubMed]
- Carter, R.C.; Jacobson, J.L.; Burden, M.J.; Armony-Sivan, R.; Dodge, N.C.; Angelilli, M.L.; Lozoff, B.; Jacobson, S.W. Iron deficiency anemia and cognitive function in infancy. Pediatrics 2010, 126, e427–e434. [Google Scholar] [CrossRef]
- Fangupo, L.J.; Heath, A.L.M.; Williams, S.M.; Erickson Williams, L.W.; Morison, B.J.; Fleming, E.A.; Taylor, B.J. Wheeler, B.J.; Taylor, R.W. A baby-led approach to eating solids and risk of chocking. Pediatrics 2016, 138, e20160772. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M. PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inform. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef]
- Kahraman, A.; Gümüs, M.; Yaz, S.B.; Basbakkal, Z. Baby-led weaning versus traditional weaning: The assessment of nutritional status in early childhood and maternal feeding practices in Turkey. Early Child Dev. Care. 2020, 190, 615–624. [Google Scholar] [CrossRef]
- Jones, S.W.; Lee, M.; Brown, A. Spoonfeeding is associated with increased infant weight but only amongst formula-fed infants. Matern. Child Nutr. 2020, 16, e12941. [Google Scholar] [CrossRef]
- Dogan, E.; Yilmaz, G.; Caylan, N.; Turgut, M.; Gokcay, G.; Oguz, M.M. Baby-led complementary feeding: A randomized controlled study. Pediatrics Int. 2018, 60, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Conlon, C.A.; Haszard, J.J.; Beck, K.L.; von Hurst, P.R.; Taylor, R.W.; Heath, A.-L.M. Food fussiness and early feeding characteristics of infants following baby-led weaning and traditional spoon-feeding in New Zealand: An internet survey. Appetite 2018. [Google Scholar] [CrossRef]
- Taylor, R.W.; Williams, S.M.; Fangupo, L.J.; Wheeler, B.J.; Taylor, B.J.; Daniels, L.; Fleming, E.A.; McArthur, J.; Morison, B.; Erickson, L.W.; et al. Effect of a baby-led approach to complementary feeding on infant growth and overweight: A randomized clinical trial. JAMA Pediatrics 2017, 171, 838–846. [Google Scholar] [CrossRef]
- Brown, A.; Lee, M.D. Early influences on child satiety-responsiveness: The role of weaning style. Pediatrics Obes. 2015, 10, 57–66. [Google Scholar] [CrossRef]
- Townsend, E.; Pitchford, N.J. Baby knows best? The impact of weaning style on food preferences and body mass index in early childhood in a case-controlled sample. BMJ Open 2012, 2, e000298. [Google Scholar] [CrossRef]
- Brown, A.; Lee, M.D. Maternal control of child feeding during the weaning period: Differences between mothers following a baby-led or standard weaning approach. Matern. Child Health J. 2011, 15, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Savović, J.; Page, M.J.; Elbers, R.G.; Sterne, J.A.C. Chapter 8: Assessing risk of bias in a randomized trial. In Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; John Wiley & Sons: Chichester, UK, 2019; pp. 205–228. [Google Scholar]
- Sterne, J.A.C.; Hernán, M.A.; McAleenan, A.; Reeves, B.C.; Higgins, J.P.T. Chapter 25: Assessing risk of bias in a non-randomized study. In Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; John Wiley & Sons: Chichester, UK, 2019; pp. 621–642. [Google Scholar]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomized studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed]
- Daniels, L.; Heath, A.M.; Williams, S.M.; Cameron, S.L.; Fleming, E.A.; Taylor, B.J.; Wheeler, B.J.; Gibson, R.S.; Taylor, R.W. Baby-led Introduction to SolidS (BLISS) study: A randomised controlled trial of a baby-led approach to complementary feeding. BMC Pediatrics 2015, 15, 179. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The WHO Child Growth Standards. 2006. Available online: https://www.who.int/childgrowth/en/ (accessed on 1 November 2020).
- Rogers, S.L.; Blissett, J. Breastfeeding duration and its relation to weight gain, eating behaviors and positive maternal feeding practices in infancy. Appetite 2017, 109, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Harder, T.; Bergmann, R.; Kallischnigg, G.; Plagemann, A. Duration of breastfeeding and risk of overweight: A meta-analysis. Am. J. Epidemiol. 2005, 162, 397–403. [Google Scholar] [CrossRef]
- Brown, A.; Lee, M. A descriptive study investigating the use and nature of baby-led weaning in a UK sample of mothers. Matern. Child Nutr. 2011, 7, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Cameron, S.L.; Heath, A.L.; Taylor, R.W. How feasible is baby-led weaning as an approach to infant feeding? A review of the evidence. Nutrients 2012, 4, 1575–1609. [Google Scholar] [CrossRef]
- Taylor, R.W.; Grant, A.M.; Goulding, A.; Williams, S.M. Early adiposity rebound: Review of papers linking this to subsequent obesity in children and adults. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Lamb, K.E.; Grimes, C.; Laws, R.; Bolton, K.; Ong, K.K.; Campbell, K. Rapid weight gain during infancy and subsequent adiposity: A systematic review and meta-analysis of evidence. Obes. Rev. 2018, 19, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Shin, J. The timing of rapid infant weight gain in relation to childhood obesity. J. Obes. Metab.Syndr. 2019, 28, 213–215. [Google Scholar] [CrossRef]
| Parameter | Inclusion Criteria |
|---|---|
| Population | Infants and children (no limitation for age) |
| Intervention | Baby-led weaning approach |
| Comparison | Standard or spoon-fed weaning approach |
| Outcome | Weight, body mass index and prevalence of overweight |
| Study type | Controlled trial and observational studies |
| Reference | Location | Type of Study | N (Number of Subjects) Recruitment (Dates) | Age of Infants (Months) | Outcome Measures | Definition of BLW | Intervention | Results and Conclusions (Regarding Weight or Body Mass Index) |
|---|---|---|---|---|---|---|---|---|
| Kahraman et al. (2020) [24] | Turkey | Observational (cross-sectional) | 485 mothers (137 BLW, 246 partial BLW, 102 TSF) October 2017–February 2018. | 6–24 | Self-reported infant weight and length. Calculation of WAZ, LAZ and BMIZ (Turkish children reference data). | (No definition in the text) | None | Less overweight in BLW (BMIZ > 2SD: 5.1% BLW vs. 14.7% TSF; p = 0.000). |
| Jones et al. (2019) [25] | United Kingdom | Observational (comparative cross-sectional and longitudinal) | Cross-sectional study: 269 infants (109 BLW, 160 TSF). Longitudinal study: 101 infants (30 BLW, 71 TSF). February 2016–November 2017. | 3–12 | Infant weight and length at one time point (cross sectional data set) and ≥16 weeks later (longitudinal data set). Calculation of WAZ, LAZ, BMIZ and WAZV (WHO reference data). | Predominantly self-fed (self-fed always and often). | None | No significant differences in WAZ among BLW regardless of the type of breastfeeding (WAZ, mean (SD): =−0.07 (1.35) BLW and formula vs. 0.06 (1.00) BLW and any breastfeeding; p = 0.84) TSF and fully formula fed (both simultaneously) significantly heavier than those who had received any breasfeeding (WAZ, mean (SD): 0.38 (0.81) TSF and formula vs. 0.17 (0.98) TSF and any breastfeeding; p = 0.04) Lower increases in WAZ and BMIZ in BLW, but not statistically significant (WAZ change: +0.13 BLW vs. +0.29 TSF, p = 0.86; BMIZ change: +0.17 BLW vs. +0.42 TSF, p = 0.07) |
| Dogan et al. (2018) [26] | Turkey | RCT | 280 breast-fed infants (142 BLW, 138 Control (TSF). January 2014–April 2016. | 5/6–12 | Infant weight, length and head circumference (at 6 and 12 months). Calculation of weight for length percentiles (at 12 months; WHO reference data). | BLISS | Control (TSF): standard well childcare. BLW group: 4 additional group training meetings and 6 home visits for support and education in BLISS until 11 months of age. | Control (TSF) significantly heavier at 12 months (11.1 ± 0.5 kg Control (TSF) vs. 10.4 ± 0.9 kg BLW; p < 0.001). No overweight in BLW at 12 months (BMIZ > 2SD: 17% in Control (TSF) vs. 0% in BLW). No underweight in Control (TSF) at 12 months (BMIZ ≤ 2SD: 0% in Control (TSF) vs. 2% in BLW). |
| Fu et al. (2018) [27] | New Zaeland | Observational (cross-sectional) | 876 parents (155 BLW, 93 partial BLW, 628 TSF) June 2017. | 6–36 | Infant weight at 6–7 months as recorded by a health professional. Calculation of WAZ. | Mostly self-fed or self-fed | None | No differences in WAZ between full BLW and TSF at 6–8 months. (WAZ, mean (SD): −0.3 (0.9) BLW vs. −0.4 (1.3) TSF; p = 0.874). |
| Taylor et al. (2017) [28] | New Zealand | RCT | 206 women in late pregnancy (105 BLW, 101 Control (TSF)). 166 infants at 24 months (88 BLW, 78 Control (TSF)). December 2012–March 2014. | 0–24 | Infant weight (at 6, 7, 8, 9, 12, and 24 months) and length (6, 12 and 24 months). Calculation of BMI and BMIZ (at 12 and 24 months; WHO reference data). | BLISS | Control (TSF): government-funded routine midwifery and well-childcare. BLW group: 8 additional contacts (6 face-to-face, 2 telephone) for lactation and BLISS support from birth to 9 months of age. | No significant differences in BMIZ between BLW and Control (TSF) at 12 months (adjusted difference, 0.23; 95% CI, −0.06 to 0.52) and at 24 months (adjusted difference, 0.15; 95% CI, −0.12 to 0.45). Higher risk (no statistically significant) of overweight in BLW at 12 months (RR 2.5; 95% CI, 0.9 to 6.9) and at 24 months (RR 1.8; 95% CI, 0.6 to 5.7). |
| Brown and Lee (2015) [29] | United Kingdom | Observational (longitudinal) | 298 children from a previous study [31] (163 BLW, 135 TSF). | 18–24 | Self-reported infant weight and length at the time of the recruitment. Calculation of WAZ (WHO reference data). | ≤10% spoon-feeding and purees. | None | TSF significantly heavier at 18–24 months. (Weight, kg (SD): 12.86 kg (3.73) in TSF vs. 11.79 kg (3.53) in BLW; p = 0.005). Less overweight in BLW at 18–24 months (8.1% BLW vs. 19.2% TSF). More underweight in BLW at 18–24 months (5.4% BLW vs. 2.5% TSF). |
| Townsend and Pitchford (2012) [30] | United Kingdom | Observational (case-control) | 155 parents (92 BLW, 63 TSF). June 2006–January 2009. | 20–78 | Self-reported infant weight and length. Calculation of BMI, BMIZ (WHO reference data) and BMI percentile rank (CDC, NHS). | Self-reported as BLW (and checked by specific questions about their weaning practices). | None | Lower BMI in BLW (CDC percentile rank: 48.46 BLW vs. 61.44 TSF; p = 0.009). Less obesity in BLW (BMIZ > 2: 1.6% BLW vs. 12.7% TSF). More underweight in BLW (BMIZ ≤ 2: 4.8% BLW vs. 0% TSF). |
| Brown and Lee (2011) [31] | United Kingdom | Observational (cross-sectional) | 604 mothers (351 BLW, 253 TSF). | 6–12 | Self-reported estimates of the infant weight (at 6 months and at the time of recruitment). Self-reported perception of infants’ growth (at 6 months). | ≤10% spoon-feeding and purees. | None | No significant differences for estimated weight at 6 months or estimated current weight (statistics not available). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinón-Torres, N.; Carreira, N.; Picáns-Leis, R.; Pérez-Ferreirós, A.; Kalén, A.; Leis, R. Baby-Led Weaning: What Role Does It Play in Obesity Risk during the First Years? A Systematic Review. Nutrients 2021, 13, 1009. https://doi.org/10.3390/nu13031009
Martinón-Torres N, Carreira N, Picáns-Leis R, Pérez-Ferreirós A, Kalén A, Leis R. Baby-Led Weaning: What Role Does It Play in Obesity Risk during the First Years? A Systematic Review. Nutrients. 2021; 13(3):1009. https://doi.org/10.3390/nu13031009
Chicago/Turabian StyleMartinón-Torres, Nazareth, Nathalie Carreira, Rosaura Picáns-Leis, Alexandra Pérez-Ferreirós, Anton Kalén, and Rosaura Leis. 2021. "Baby-Led Weaning: What Role Does It Play in Obesity Risk during the First Years? A Systematic Review" Nutrients 13, no. 3: 1009. https://doi.org/10.3390/nu13031009
APA StyleMartinón-Torres, N., Carreira, N., Picáns-Leis, R., Pérez-Ferreirós, A., Kalén, A., & Leis, R. (2021). Baby-Led Weaning: What Role Does It Play in Obesity Risk during the First Years? A Systematic Review. Nutrients, 13(3), 1009. https://doi.org/10.3390/nu13031009









