Longitudinal Weight Gain and Related Risk Behaviors during the COVID-19 Pandemic in Adults in the US
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Questionnaire
2.3. Measures
2.4. Data Analysis
3. Results
3.1. Change in Health Behaviors between Peak-Lockdown Period and Post-Lockdown Period
3.2. Change in Health Behaviors between Peak-Lockdown Period and Post-Lockdown Period by Weight Change Categories
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BMI | Body mass index |
| Mturk | Amazon Mechanical Turk |
| PA | Physical Activity |
| SSS | Stanford Sleepiness Scale |
| CoEQ | The Control of Eating Questionnaire |
References
- Zachary, Z.; Brianna, F.; Brianna, L.; Garrett, P.; Jade, W.; Alyssa, D.; Mikayla, K. Self-quarantine and weight gain related risk factors during the COVID-19 pandemic. Obes. Res. Clin. Pract. 2020, 14, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Schoeller, D.A. The effect of holiday weight gain on body weight. Physiol. Behav. 2014, 134, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Bhutani, S.; Cooper, J.A. COVID-19 related home confinement in adults: Weight gain risks and opportunities. Obesity 2020. [Google Scholar] [CrossRef] [PubMed]
- Bhutani, S.; Wells, N.; Finlayson, G.; Schoeller, D.A. Change in eating pattern as a contributor to energy intake and weight gain during the winter holiday period in obese adults. Int. J. Obes. 2020, 44, 1586–1595. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.A.; Tokar, T. A prospective study on vacation weight gain in adults. Physiol. Behav. 2016, 156, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, J.L.; Krishnan, S.; Stoner, M.A.; Goktas, Z.; Cooper, J.A. Effects of exercise during the holiday season on changes in body weight, body composition and blood pressure. Eur. J. Clin. Nutr. 2013, 67, 944–949. [Google Scholar] [CrossRef]
- Flanagan, E.W.; Beyl, R.A.; Fearnbach, S.N.; Altazan, A.D.; Martin, C.K.; Redman, L.M. The impact of COVID-19 stay-at-home orders on health behaviors in adults. Obesity 2020. [Google Scholar] [CrossRef]
- Bhutani, S.; Cooper, J.A.; vanDellen, M.R. Self-reported changes in energy balance behaviors during COVID-19 related home confinement: A Cross-Sectional Study. medRxiv 2020. [Google Scholar] [CrossRef]
- Ammar, A.; Brach, M.; Trabelsi, K.; Chtourou, H.; Boukhris, O.; Masmoudi, L.; Bouaziz, B.; Bentlage, E.; How, D.; Ahmed, M.; et al. Effects of COVID-19 Home Confinement on Eating Behaviour and Physical Activity: Results of the ECLB-COVID19 International Online Survey. Nutrients 2020, 12, 1583. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.A.; vanDellen, M.; Bhutani, S. Self-weighing Practices and Associated Health Behaviors during COVID-19. Am. J. Health Behav. 2021, 45, 17–30. [Google Scholar] [CrossRef]
- Wang, Y.; Beydoun, M.A.; Min, J.; Xue, H.; Kaminsky, L.A.; Cheskin, L.J. Has the prevalence of overweight, obesity and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int. J. Epidemiol. 2020, 49, 810–823. [Google Scholar] [CrossRef]
- Mason, W.; Suri, S. Conducting behavioral research on Amazon’s Mechanical Turk. Behav. Res. Methods 2012, 44, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Paolacci, G.; Chandler, J. Inside the Turk:Understanding Mechanical Turk as a Participant Pool. Curr. Dir. Psychol. Sci. 2014, 23, 184–188. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Levy, R.B.; Moubarac, J.C.; Louzada, M.L.; Rauber, F.; Khandpur, N.; Cediel, G.; Neri, D.; Martinez-Steele, E.; et al. Ultra-processed foods: What they are and how to identify them. Public. Health Nutr. 2019, 22, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Kurtze, N.; Rangul, V.; Hustvedt, B.E. Reliability and validity of the international physical activity questionnaire in the Nord-Trondelag health study (HUNT) population of men. BMC Med. Res. Methodol. 2008, 8, 63. [Google Scholar] [CrossRef] [PubMed]
- Cerin, E.; Cain, K.L.; Oyeyemi, A.L.; Owen, N.; Conway, T.L.; Cochrane, T.; Van Dyck, D.; Schipperijn, J.; Mitas, J.; Toftager, M.; et al. Correlates of Agreement between Accelerometry and Self-reported Physical Activity. Med. Sci. Sports Exerc. 2016, 48, 1075–1084. [Google Scholar] [CrossRef]
- Dalton, M.; Finlayson, G.; Hill, A.; Blundell, J. Preliminary validation and principal components analysis of the Control of Eating Questionnaire (CoEQ) for the experience of food craving. Eur. J. Clin. Nutr. 2015, 69, 1313–1317. [Google Scholar] [CrossRef]
- Hoddes, E.D.; Zarcone, V. The development and use of the stanford sleepiness scale (SSS). Psychophysiology 1972, 9, 150. [Google Scholar]
- Hunter, J.A.; Dyer, K.J.; Cribbie, R.A.; Eastwood, J.D. Exploring the utility of the Multidimensional State Boredom Scale. Eur. J. Psychol. Assess. 2016, 32, 241. [Google Scholar] [CrossRef]
- Rothman, K.J. No adjustments are needed for multiple comparisons. Epidemiology 1990, 1, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Saville, D.J. Multiple Comparison Procedures: The Practical Solution. Am. Stat. 1990, 44, 174–180. [Google Scholar] [CrossRef]
- Bellisle, F. Meals and snacking, diet quality and energy balance. Physiol. Behav. 2014, 134, 38–43. [Google Scholar] [CrossRef]
- America Gets Cooking. HUNTER: Food Study Special Report; USA. Available online: https://www.hunterpr.com/foodstudy_coronavirus/ (accessed on 20 December 2020).
- Cellini, N.; Canale, N.; Mioni, G.; Costa, S. Changes in sleep pattern, sense of time and digital media use during COVID-19 lockdown in Italy. J. Sleep Res. 2020, e13074. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.Y.; Wang, J.; Ou-Yang, X.Y.; Miao, Q.; Chen, R.; Liang, F.X.; Zhang, Y.P.; Tang, Q.; Wang, T. The immediate impact of the 2019 novel coronavirus (COVID-19) outbreak on subjective sleep status. Sleep Med. 2020. [Google Scholar] [CrossRef]
- Salari, N.; Hosseinian-Far, A.; Jalali, R.; Vaisi-Raygani, A.; Rasoulpoor, S.; Mohammadi, M.; Rasoulpoor, S.; Khaledi-Paveh, B. Prevalence of stress, anxiety, depression among the general population during the COVID-19 pandemic: A systematic review and meta-analysis. Glob. Health 2020, 16, 57. [Google Scholar] [CrossRef] [PubMed]
- Moynihan, A.B.; van Tilburg, W.A.; Igou, E.R.; Wisman, A.; Donnelly, A.E.; Mulcaire, J.B. Eaten up by boredom: Consuming food to escape awareness of the bored self. Front. Psychol. 2015, 6, 369. [Google Scholar] [CrossRef]
- Chao, A.; Grilo, C.M.; White, M.A.; Sinha, R. Food cravings mediate the relationship between chronic stress and body mass index. J. Health Psychol. 2015, 20, 721–729. [Google Scholar] [CrossRef]
- Van Tilburg, W.A.P.; Igou, E.R. On boredom: Lack of challenge and meaning as distinct boredom experiences. Motiv. Emot. 2012, 36, 181–194. [Google Scholar] [CrossRef]
- Chao, A.M.; Jastreboff, A.M.; White, M.A.; Grilo, C.M.; Sinha, R. Stress, cortisol, and other appetite-related hormones: Prospective prediction of 6-month changes in food cravings and weight. Obesity 2017, 25, 713–720. [Google Scholar] [CrossRef]
- Scourboutakos, M.J.; L’Abbe, M.R. Restaurant menus: Calories, caloric density, and serving size. Am. J. Prev. Med. 2012, 43, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Skues, J.W.B.; Oldmeadow, J.; Wise, L. The effects of boredom, loneliness, and distress tolerance on problem internet use among university students. Int. J. Ment. Health Addict. Vol. 2016, 14, 167–180. [Google Scholar] [CrossRef]
- Whiting, A.; Wiliams, D. Why people use social media: A uses and gratifications approach. Qual. Market. Res. Int. J. 2013, 16, 362–369. [Google Scholar] [CrossRef]
- Dunton, G.W.S.; Do, B.; Coutney, J. Early Effects of the COVID-19 Pandemic on Physical Activity in US Adults. Camb. Open Engag. 2020. [Google Scholar] [CrossRef]
- Thompson, T.; Rodebaugh, T.L.; Bessaha, M.L.; Sabbath, E.L. The Association Between Social Isolation and Health: An Analysis of Parent-Adolescent Dyads from the Family Life, Activity, Sun, Health, and Eating Study. Clin. Soc. Work J. 2020, 48, 18–24. [Google Scholar] [CrossRef]
- Rocha, J.; Paxman, J.; Dalton, C.; Winter, E.; Broom, D.R. Effects of a 12-week aerobic exercise intervention on eating behaviour, food cravings, and 7-day energy intake and energy expenditure in inactive men. Appl. Physiol. Nutr. Metab. 2016, 41, 1129–1136. [Google Scholar] [CrossRef]
- Cecchetto, C.; Aiello, M.; Gentili, C.; Ionta, S.; Osimo, S.A. Increased emotional eating during COVID-19 associated with lockdown, psychological and social distress. Appetite 2021, 160, 105122. [Google Scholar] [CrossRef] [PubMed]
- Fearnbach, S.N.; Flanagan, E.W.; Hochsmann, C.; Beyl, R.A.; Altazan, A.D.; Martin, C.K.; Redman, L.M. Factors Protecting against a Decline in Physical Activity during the COVID-19 Pandemic. Med. Sci. Sports Exerc. 2021. [Google Scholar] [CrossRef] [PubMed]
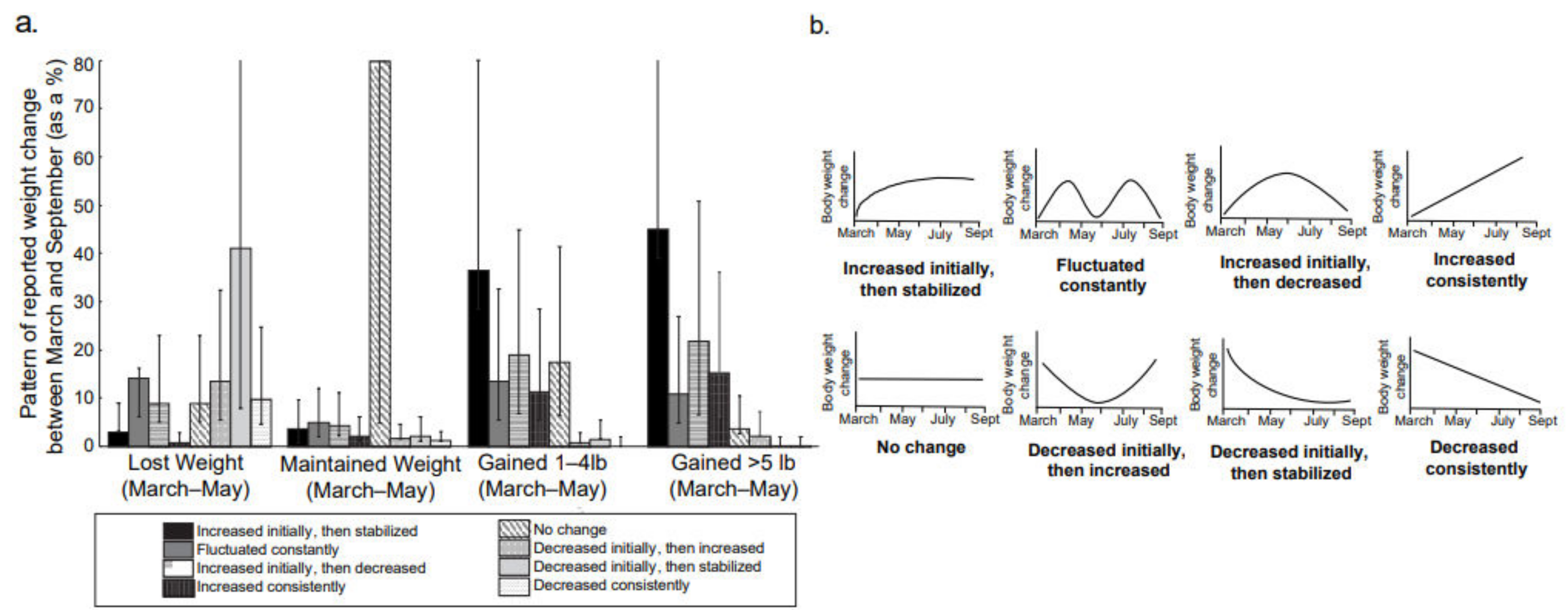
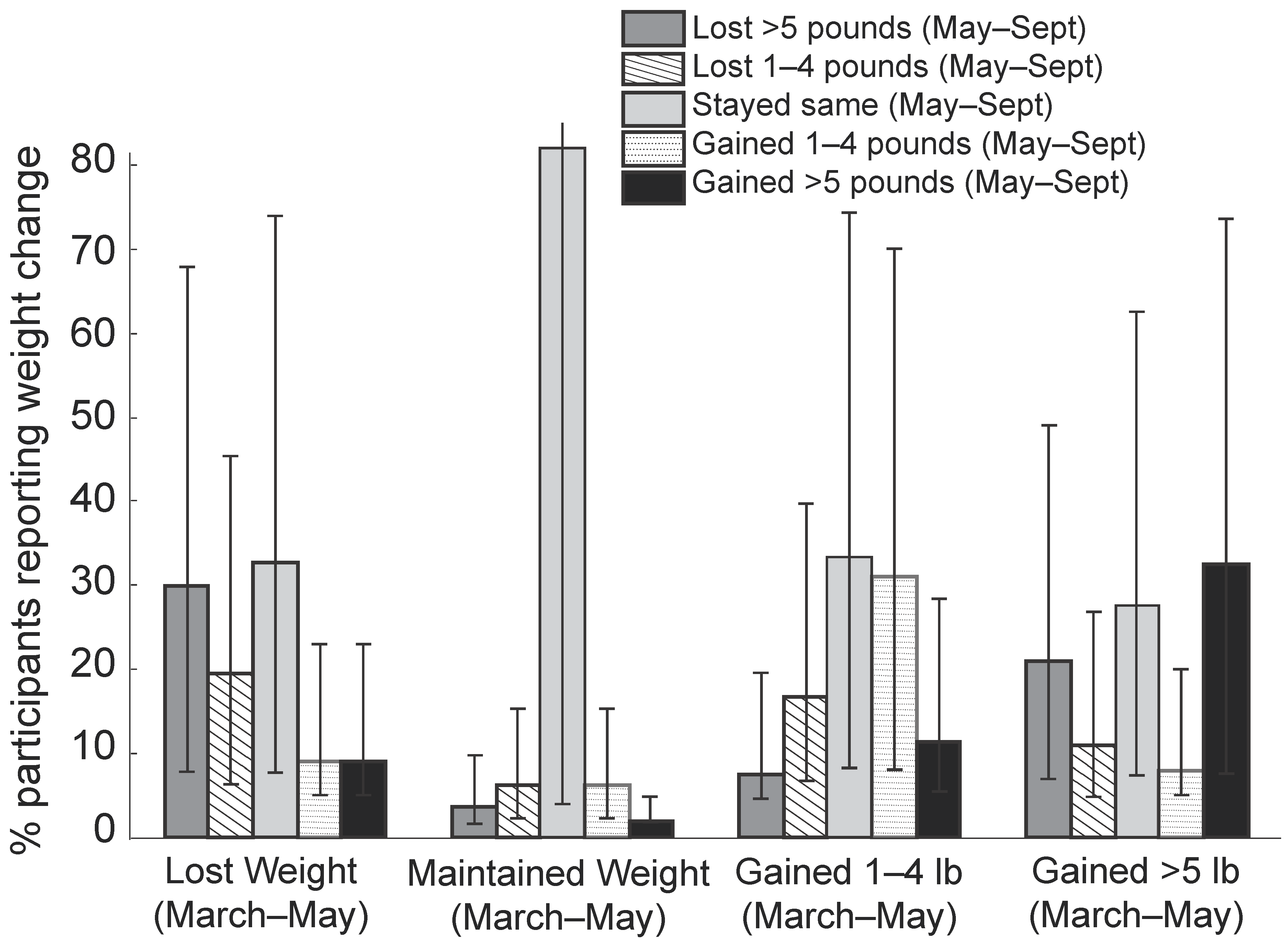
| Behavioral Variables | Peak-Lockdown Period (March–May) | Post-Lockdown Period May–September) |
|---|---|---|
| Body Weight (kg) * | 76.70 (19.79) | 77.32 (20.01) |
| Body Mass Index (kg/m2) ** | 26.12 (5.81) | 26.38 (5.98) |
| Diet variables | ||
| Takeout/Restaurant Frequency *** | 2.94 (1.41) | 3.36 (1.31) |
| Alcohol Frequency | 4.11 (1.43) | 4.01 (1.28) |
| Vegetable Frequency | 5.83 (1.43) | 5.88 (1.40) |
| Fruit Frequency | 5.62 (1.52) | 5.48 (1.52) |
| Non-diet drinks (all SSBs) Frequency | 3.50 (2.34) | 3.54 (2.24) |
| Diet soda or other diet drinks Frequency | 2.47 (2.14) | 2.55 (2.15) |
| Processed foods (NOVA) Frequency * | 4.66 (1.80) | 4.52 (1.59) |
| Ultra-processed foods (NOVA) Frequency | 3.79 (1.76) | 3.71 (1.70) |
| Physical activity variables | ||
| Weekly MET *** | 2100.66 (2287.28) | 2712.71 (2898.18) |
| Walking | 644.93 (856.65) | 647.77 (831.93) |
| Moderate Activity *** | 482.31 (759.20) | 583.98 (874.43) |
| Vigorous Activity | 973.41 (1468.77) | 953.89 (1424.85) |
| Sitting Time ** | 410.74 (290.27) | 378.44 (250.90) |
| TV Time Frequency *** | 3.87 (1.42) | 3.60 (1.33) |
| Leisure Screen Time Frequency ** | 3.68 (1.40) | 3.55 (1.33) |
| Psychological State variables | ||
| Stress Rating | 4.48 (2.59) | 4.36 (2.54) |
| Sleep (hours) | 7.21 (1.32) | 7.08 (1.29) |
| Sleepiness Rating | 2.78 (1.42) | 2.76 (1.39) |
| Boredom Rating *** | 3.49 (1.52) | 3.24 (1.53) |
| Self-Weighing Frequency *** | 2.48 (1.33) | 2.62 (1.19) |
| Importance of Eating Healthily | 2.96 (0.87) | 3.00 (0.85) |
| Mean Cravings *** | 5.78 (2.38) | 6.20 (2.41) |
| Lost Weight in March−May N = 134 (18.4%) | Maintained Weight in March−May N = 323 (44.4%) | Gained 1–4 lbs in March−May N = 132 (18.2%) | Gained > 5 lbsin March−May N = 138 (19.0%) | p# | Time * Group Interaction | ||
|---|---|---|---|---|---|---|---|
| Body Mass Index (kg/m2) | T1 | 26.57 (5.62) ab | 25.21(5.51) a | 25.58 (5.07) a | 28.32 (6.69) bc | *** | 9.90 *** |
| T2 | 26.38 (5.69) a | 25.22(5.44) a | 26.03 (5.32) a | 29.40 (7.00) b | *** | ||
| Weight (kg) | T1 | 77.66 (19.14) a | 74.41 (19.02) a | 73.32 (17.78) a | 84.34 (32.00) b | *** | 10.18 *** |
| T2 | 76.91 (18.92) a | 74.33(18.57) a | 74.50 (18.44) a | 87.36 (22.53) b | *** | ||
| Diet variables Frequency | |||||||
| Vegetables Frequency | T1 | 5.88 (1.41) a | 5.91 (1.46) a | 5.60 (1.51) a | 5.80 (1.27) a | ns | 0.83 |
| T2 | 5.88 (1.43) a | 6.01 (1.42) a | 5.67 (1.49) a | 5.80 (1.24) a | ns | ||
| Fruits Frequency | T1 | 5.68 (1.56) a | 5.61 (1.52) a | 5.64 (1.60) a | 5.57 (1.42 a | ns | 2.47 |
| T2 | 5.54 (1.49) a | 5.58 (1.53) a | 5.30 (1.51) a | 5.33 (1.52) a | ns | ||
| Non-diet drinks (all SSBs) Frequency | T1 | 3.40 (2.29) ab | 3.24 (2.29) a | 3.86 (2.35) ab | 3.86(2.42) b | * | 0.71 |
| T2 | 3.64 (2.25) ab | 3.28 (2.27) a | 3.70 (2.00) ab | 3.91(2.34) b | * | ||
| Diet soda or diet drinks Frequency | T1 | 2.44 (2.08) a | 2.37 (2.06) a | 2.29 (2.16) a | 2.91(2.31) a | * | 1.56 |
| T2 | 2.75 (2.19) ab | 2.37 (2.08) a | 2.32 (2.04) a | 2.99 (2.30) b | * | ||
| Processed foods (NOVA) Frequency | T1 | 4.40 (1.81) a | 4.56 (1.79) a | 4.95 (1.61) a | 4.86 (1.94) a | * | ns |
| T2 | 4.48 (1.48) a | 4.39 (1.81) a | 4.67 (1.50) a | 4.73 (1.73) a | ns | ns | |
| Ultra-processed foods (NOVA) Frequency | T1 | 3.60 (1.84) a | 3.63 (1.68) a | 3.98 (1.72) ab | 4.17 (1.84) b | ** | ns |
| T2 | 3.66 (1.79) a | 3.50 (1.68) a | 3.70 (1.50) a | 4.28 (1.75) b | *** | ns | |
| Snacking frequency | T1 | 3.38 (1.31) a | 3.30 (1.32) a | 2.75 (1.29) b | 2.85 (1.26) b | *** | * |
| T2 | 3.12 (1.23) a | 3.30 (1.22) a | 3.14 (1.25) a | 3.04 (1.12) a | ns | * | |
| Alcohol Frequency | T1 | 3.76 (1.30) a | 4.19 (1.50) b | 4.09 (1.53) b | 4.28 (1.26) b | * | * |
| T2 | 3.12 (1.23) a | 3.30 (1.22) a | 3.14 (1.25) a | 3.04 (1.12) a | ns | * | |
| Takeout/Restaurant Frequency | T1 | 2.95 (1.38) a | 2.81 (1.41) a | 3.18 (1.42) a | 3.02 (1.42) a | ns | * |
| T2 | 3.53 (1.26) a | 3.14 (1.28) b | 3.50 (1.25) a | 3.55 (1.40) a | *** | ns | |
| Physical activity variables | |||||||
| Weekly Kcal MET | T1 | 2514.65 (2627.80) a | 2051.19 (2191.27) a | 1859.81 (2174.49) a | 2044.82 (2231.74) a | ns | 0.43 |
| T2 | 3327.72 (3360.22) a | 2609.33 (2813.11) ab | 2339.12 (2270.61) b | 2710.61 (3077.10) ab | * | ||
| Vigorous Activity | T1 | 1304.84 (1798.59) a | 936.07 (1429.47) a | 854.79 (1283.79) a | 852.46 (1331.88) a | * | 0.26 |
| T2 | 1278.21 (1798.94) a | 957.67 (1445.37) ab | 768.55 (1056.40) b | 804.96 (1212.78) b | * | ||
| Moderate Activity | T1 | 480.46 (786.98) a | 527.55 (802.72) a | 399.55 (649.82) a | 457.39 (723.64) a | ns | 0.32 |
| T2 | 646.15 (976.04) a | 609.55 (880.78) a | 521.98 (843.42) a | 522.19 (780.55) a | ns | ||
| Walking Time | T1 | 729.35 (879.01) a | 587.56 (745.67) a | 605.48 (771.32) a | 734.97 (1067.44) a | ns | 2.21 |
| T2 | 778.46 (971.36) a | 637.31 (798.67) a | 629.65 (831.24) a | 561.96 (752.45) a | ns | ||
| Sitting Time | T1 | 406.38 a | 406.38 a | 403.40 a | 426.93 a | ns | 0.57 |
| T2 | 359.33 a | 367.88 a | 396.41 a | 404.82 a | ns | ||
| TV time | T1 | 3.67 (1.48) a | 3.79 (1.40) ab | 4.01 (1.36) ab | 4.14 (1.42) b | * | 2.74 * |
| T2 | 3.51 (1.40) a | 3.58 (1.35) a | 3.71 (1.26) a | 3.62 (1.27) a | ns | ||
| Leisure Screen time | T1 | 3.67 (1.38) ab | 3.51 (1.37) a | 3.93 (1.39) b | 3.83 (1.44) ab | * | 0.78 |
| T2 | 3.63 (1.44) ab | 3.42 (1.27) a | 3.77 (1.34) b | 3.55 (1.33) ab | ns | ||
| Lost Weight in March–May N = 134 (18.4%) | Maintained Weight in March–May N = 323 (44.4%) | Gained 1–4 lbs in March–May N = 132 (18.2%) | Gained >5 lbs in March–May N = 138 (19.0%) | p# | Time * Group Interaction | ||
|---|---|---|---|---|---|---|---|
| Stress rating | T1 | 4.61 a | 3.87 b | 4.73 a | 4.92 a | *** | 1.60 |
| T2 | 4.44 ab | 3.99 b | 5.12a | 5.07 a | *** | ||
| Sleepiness rating | T1 | 2.72 (1.43) ab | 2.54 (1.39) b | 3.02 (1.36) ac | 3.18 (1.44)c | *** | 3.30 * |
| T2 | 2.57 (1.38) a | 2.63 (1.38) a | 3.11 (1.49) b | 2.89 (1.23)ab | ** | ||
| Sleep time (h) | T1 | 7.03 (1.40) a | 7.28 (1.15)a | 7.14 (1.44) a | 7.31 (1.48)a | ns | 1.27 |
| T2 | 7.02 (1.38) a | 7.10 (1.23)a | 7.05 (1.49) a | 7.10 (1.20)a | ns | ||
| Boredom rating | T1 | 3.72 (1.48) a | 3.16 (1.55) b | 3.78 (1.36) a | 3.78 (1.51) a | *** | 0.41 |
| T2 | 3.40 (1.49) a | 2.92 (1.55) b | 3.63 (1.50) b | 3.49 (1.43) b | *** | ||
| Craving Control rating | T1 | 6.00 (2.12) a | 6.39 (2.26) a | 5.16 (2.20) b | 4.53 (2.50) b | *** | 0.77 |
| T2 | 6.27 (2.33) a | 6.87 (2.20) ab | 5.83 (2.37) ab | 4.95 (2.41) c | *** | ||
| Sweet Cravings rating | T1 | 3.99 (2.21) ab | 3.46 (2.21) b | 4.64 (2.17) ac | 4.76 (2.30) c | *** | 0.80 |
| T2 | 3.66 (2.21) a | 2.94 (2.16) b | 3.83 (2.00) a | 4.14 (2.40) a | *** | ||
| Savory Cravings rating | T1 | 4.16 (1.90) a | 4.07 (2.08) a | 4.47 (1.89) ab | 5.03 (2.10) b | *** | 1.22 |
| T2 | 4.12 (1.89) a | 3.65 (2.05) a | 3.99 (2.02) a | 4.81 (2.09) b | *** | ||
| Positive Mood rating | T1 | 5.42 (1.90) a | 6.10 (2.07) b | 5.24 (1.89) a | 5.29 (2.20) a | *** | 0.55 |
| T2 | 5.72 (2.02) a | 6.30 (2.09) b | 5.41 (1.94) a | 5.65 (2.22) a | *** | ||
| Daily self-weighing frequency | T1 | 2.78 (1.44) a | 2.40 (1.26) b | 2.45 (1.34) b | 2.43 (1.33) b | * | 0.39 |
| T2 | 2.88 (1.16) a | 2.58 (1.19) a | 2.52 (1.19) a | 2.54 (1.17) a | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhutani, S.; vanDellen, M.R.; Cooper, J.A. Longitudinal Weight Gain and Related Risk Behaviors during the COVID-19 Pandemic in Adults in the US. Nutrients 2021, 13, 671. https://doi.org/10.3390/nu13020671
Bhutani S, vanDellen MR, Cooper JA. Longitudinal Weight Gain and Related Risk Behaviors during the COVID-19 Pandemic in Adults in the US. Nutrients. 2021; 13(2):671. https://doi.org/10.3390/nu13020671
Chicago/Turabian StyleBhutani, Surabhi, Michelle R. vanDellen, and Jamie A. Cooper. 2021. "Longitudinal Weight Gain and Related Risk Behaviors during the COVID-19 Pandemic in Adults in the US" Nutrients 13, no. 2: 671. https://doi.org/10.3390/nu13020671
APA StyleBhutani, S., vanDellen, M. R., & Cooper, J. A. (2021). Longitudinal Weight Gain and Related Risk Behaviors during the COVID-19 Pandemic in Adults in the US. Nutrients, 13(2), 671. https://doi.org/10.3390/nu13020671





