Effects of a Rice Bran Dietary Intervention on the Composition of the Intestinal Microbiota of Adults with a High Risk of Colorectal Cancer: A Pilot Randomised-Controlled Trial
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Study Settings and Participants
2.3. Randomisation and Masking
2.4. Intervention
2.5. Outcomes
2.6. Data Collection Procedures
2.7. Procedures
2.7.1. Stool Sample Collection and Processing
2.7.2. 16S Metagenomic Sequencing
2.8. Data Analysis
2.9. Ethical Considerations
3. Results
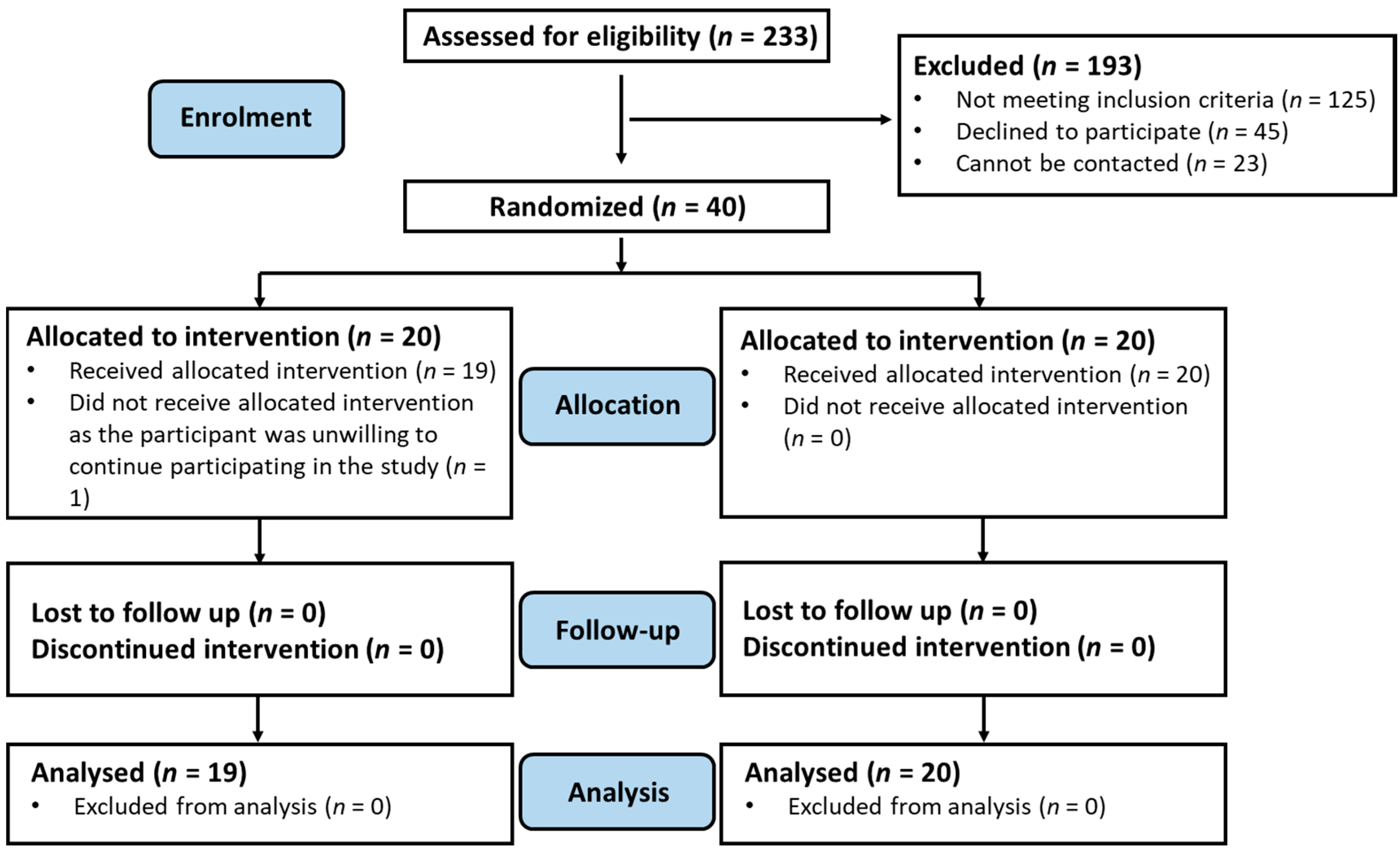
3.1. Subject Recruitment and Retention Rate
3.2. Effect of Rice Bran Intake on Intestinal Microbiota
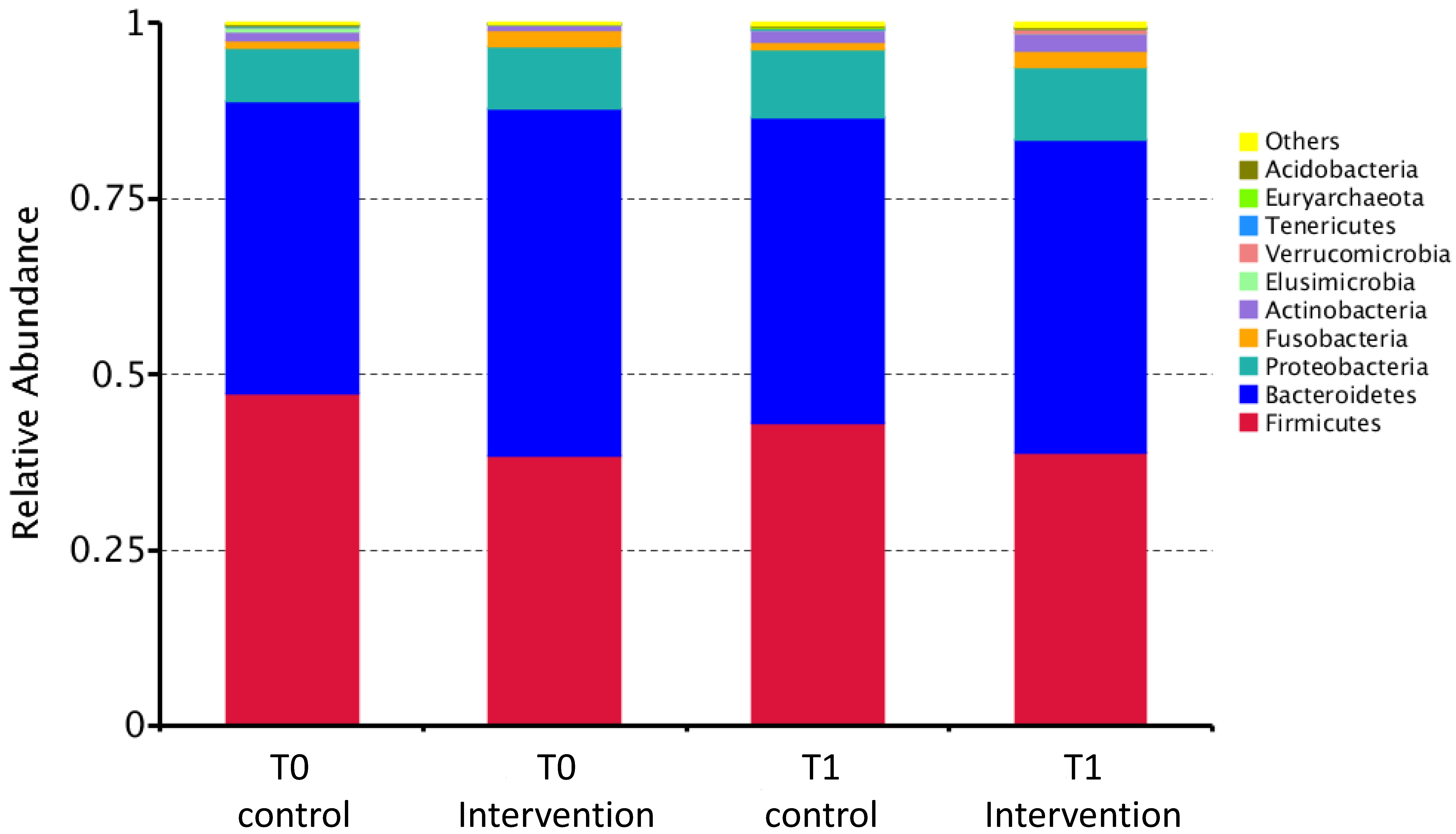
3.2.1. Composition of Intestinal Microbiota
3.2.2. F/B Ratio
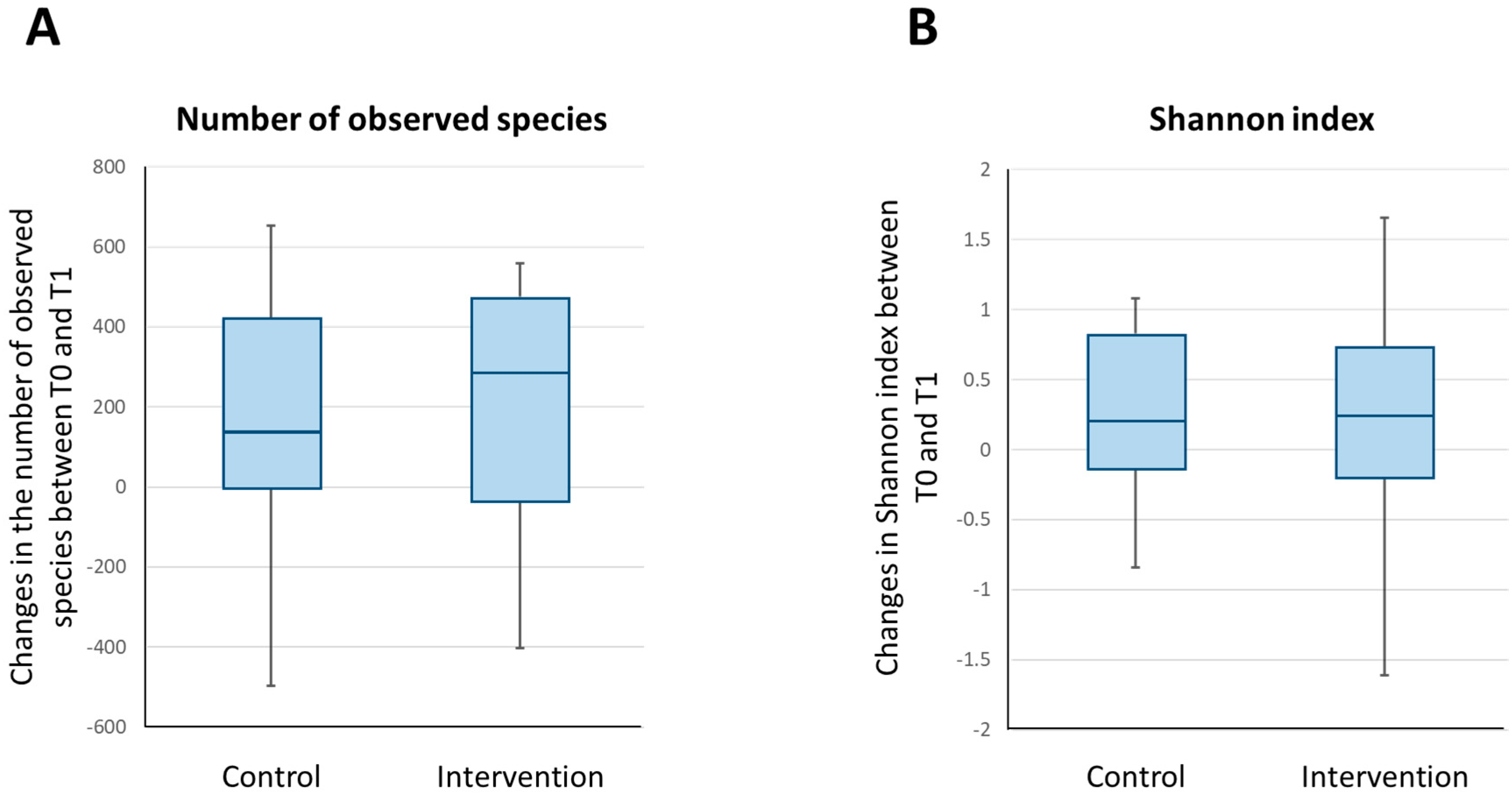
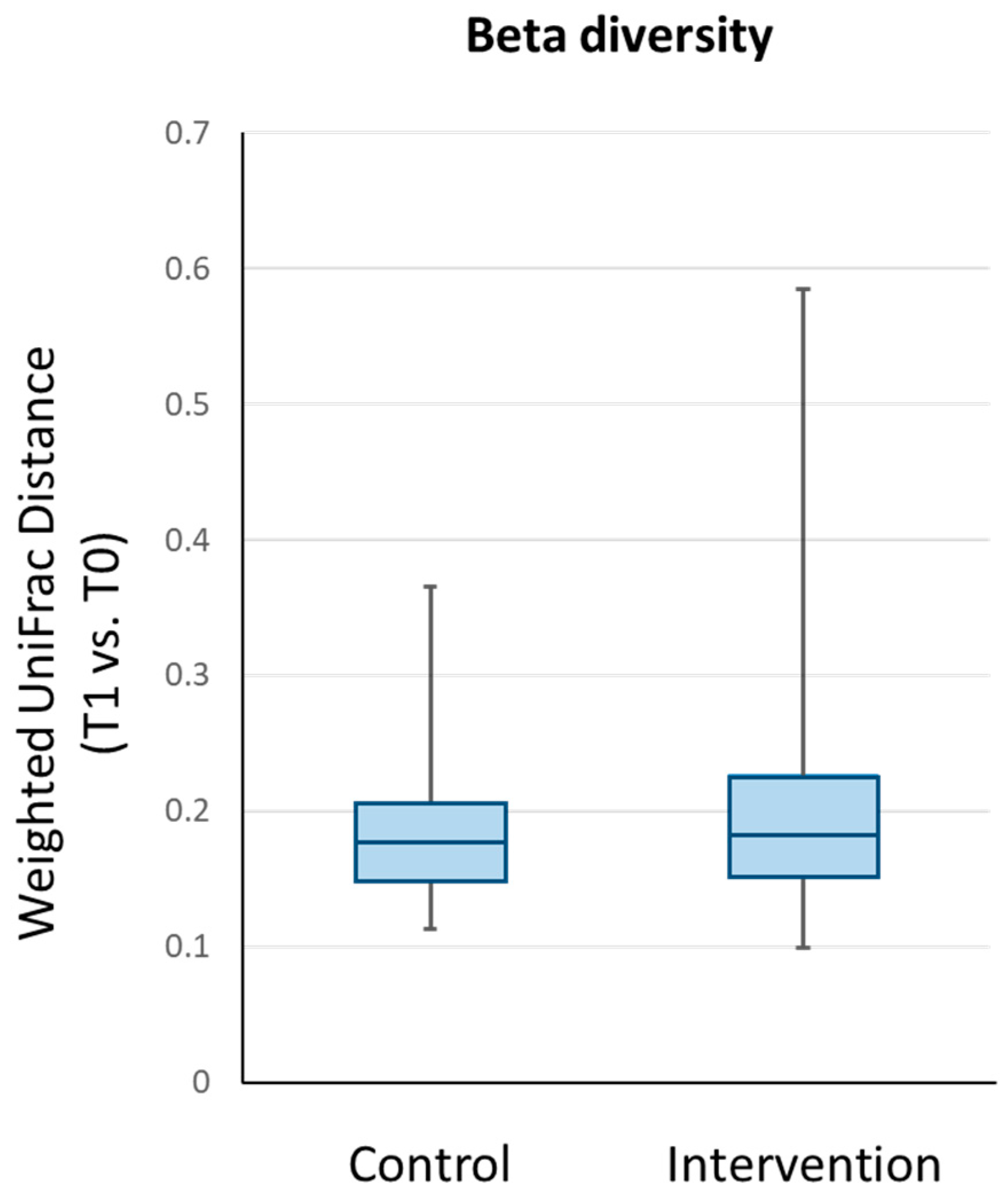
3.2.3. Bacterial Diversity
3.3. Compliance Rate
3.4. Adverse Events Caused by Rice Bran Intake
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, M.A.; Soteriades, E.S.; King, J.; Govender, R.; Hashim, M.J.; Masood-Husain, S.; Javaid, S.F.; Al Darei, S.D.M.S.; Al Sheryani, S.D.; Nauman, J. Global Trends and Forecast of the Burden of Adverse Effects of Medical Treatment: Epidemiological Analysis Based on the Global Burden of Disease Study. Cureus 2020, 12, e7250. [Google Scholar] [CrossRef]
- Rajamanickam, S.; Agarwal, R. Natural products and colon cancer: Current status and future prospects. Drug Dev. Res. 2008, 69, 460–471. [Google Scholar] [CrossRef] [PubMed]
- Tieri, M.; Ghelfi, F.; Vitale, M.; Vetrani, C.; Marventano, S.; Lafranconi, A.; Godos, J.; Titta, L.; Gambera, A.; Alonzo, E.; et al. Whole grain consumption and human health: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2020, 71, 668–677. [Google Scholar] [CrossRef]
- Henderson, A.J.; Ollila, C.A.; Kumar, A.; Borresen, E.C.; Raina, K.; Agarwal, R.; Ryan, E.P. Chemopreventive properties of dietary rice bran: Current status and future prospects. Adv. Nutr. 2012, 3, 643–653. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E. Scientific Evidence of Rice By-Products for Cancer Prevention: Chemopreventive Properties of Waste Products from Rice Milling on Carcinogenesis In Vitro and In Vivo. Biomed Res. Int. 2017, 2017, 9017902. [Google Scholar] [CrossRef] [PubMed]
- Law, B.M.H.; Waye, M.M.Y.; So, W.K.W.; Chair, S.Y. Hypotheses on the Potential of Rice Bran Intake to Prevent Gastrointestinal Cancer through the Modulation of Oxidative Stress. Int. J. Mol. Sci. 2017, 18, 1352. [Google Scholar]
- So, W.K.; Law, B.M.; Law, P.T.; Chan, C.W.; Chair, S.Y. Current Hypothesis for the Relationship between Dietary Rice Bran Intake, the Intestinal Microbiota and Colorectal Cancer Prevention. Nutrients 2016, 8, 569. [Google Scholar] [CrossRef]
- Gao, Z.; Guo, B.; Gao, R.; Zhu, Q.; Qin, H. Microbiota disbiosis is associated with colorectal cancer. Front. Microbiol. 2015, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Gagnière, J.; Raisch, J.; Veziant, J.; Barnich, N.; Bonnet, R.; Buc, E.; Bringer, M.A.; Pezet, D.; Bonnet, M. Gut microbiota imbalance and colorectal cancer. World J. Gastroenterol. 2016, 22, 501–518. [Google Scholar] [CrossRef]
- Bishehsari, F.; Engen, P.A.; Preite, N.Z.; Tuncil, Y.E.; Naqib, A.; Shaikh, M.; Rossi, M.; Wilber, S.; Green, S.J.; Hamaker, B.R.; et al. Dietary Fiber Treatment Corrects the Composition of Gut Microbiota, Promotes SCFA Production, and Suppresses Colon Carcinogenesis. Genes 2018, 9, 102. [Google Scholar] [CrossRef]
- Lu, Y.; Chen, J.; Zheng, J.; Hu, G.; Wang, J.; Huang, C.; Lou, L.; Wang, X.; Zeng, Y. Mucosal adherent bacterial dysbiosis in patients with colorectal adenomas. Sci. Rep. 2016, 6, 26337. [Google Scholar] [CrossRef]
- Kumar, A.; Henderson, A.; Forster, G.M.; Goodyear, A.W.; Weir, T.L.; Leach, J.E.; Dow, S.W.; Ryan, E.P. Dietary rice bran promotes resistance to Salmonella enterica serovar Typhimurium colonization in mice. BMC Microbiol. 2012, 12, 71. [Google Scholar] [CrossRef]
- Henderson, A.J.; Kumar, A.; Barnett, B.; Dow, S.W.; Ryan, E.P. Consumption of rice bran increases mucosal immunoglobulin A concentrations and numbers of intestinal Lactobacillus spp. J. Med. Food 2012, 15, 469–475. [Google Scholar] [CrossRef]
- Yang, X.; Twitchell, E.; Li, G.; Wen, K.; Weiss, M.; Kocher, J.; Lei, S.; Ramesh, A.; Ryan, E.P.; Yuan, L. High protective efficacy of rice bran against human rotavirus diarrhea via enhancing probiotic growth, gut barrier function, and innate immunity. Sci. Rep. 2015, 5, 15004. [Google Scholar] [CrossRef] [PubMed]
- Sheflin, A.M.; Borresen, E.C.; Wdowik, M.J.; Rao, S.; Brown, R.J.; Heuberger, A.L.; Broeckling, C.D.; Weir, T.L.; Ryan, E.P. Pilot dietary intervention with heat-stabilized rice bran modulates stool microbiota and metabolites in healthy adults. Nutrients 2015, 7, 1282–1300. [Google Scholar] [CrossRef] [PubMed]
- Sheflin, A.M.; Borresen, E.C.; Kirkwood, J.S.; Boot, C.M.; Whitney, A.K.; Lu, S.; Brown, R.J.; Broeckling, C.D.; Ryan, E.P.; Weir, T.L. Dietary supplementation with rice bran or navy bean alters gut bacterial metabolism in colorectal cancer survivors. Mol. Nutr. Food Res. 2017, 61, 1500905. [Google Scholar] [CrossRef] [PubMed]
- So, W.K.W.; Law, B.M.H.; Law, P.T.W.; Choi, K.C.; Chan, C.W.H. A pilot study to compare two types of heat-stabilized rice bran in modifying compositions of intestinal microbiota among healthy Chinese adults. Adv. Mod. Oncol. Res. 2018, 4, 253. [Google Scholar]
- Zambrana, L.E.; McKeen, S.; Ibrahim, H.; Zarei, I.; Borresen, E.C.; Doumbia, L.; Boré, A.; Cissoko, A.; Douyon, S.; Koné, K.; et al. Rice bran supplementation modulates growth, microbiota and metabolome in weaning infants: A clinical trial in Nicaragua and Mali. Sci. Rep. 2019, 9, 13919. [Google Scholar] [CrossRef]
- Brooks, A.W.; Priya, S.; Blekhman, R.; Bordenstein, S.R. Gut microbiota diversity across ethnicities in the United States. PLoS Biol. 2018, 16, e2006842. [Google Scholar] [CrossRef]
- Goodrich, J.K.; Waters, J.L.; Poole, A.C.; Sutter, J.L.; Koren, O.; Blekhman, R.; Beaumont, M.; Van Treuren, W.; Knight, R.; Bell, J.T.; et al. Human genetics shape the gut microbiome. Cell 2014, 159, 789–799. [Google Scholar] [CrossRef]
- Conlon, M.A.; Bird, A.R. The impact of diet and lifestyle on gut microbiota and human health. Nutrients 2014, 7, 17–44. [Google Scholar] [CrossRef]
- Lancaster, G.A.; Dodd, S.; Williamson, P.R. Design and analysis of pilot studies: Recommendations for good practice. J. Eval. Clin. Pract. 2004, 10, 307–312. [Google Scholar] [CrossRef]
- Yeoh, K.G.; Ho, K.Y.; Chiu, H.M.; Zhu, F.; Ching, J.Y.; Wu, D.C.; Matsuda, T.; Byeon, J.S.; Lee, S.K.; Goh, K.L.; et al. Asia-Pacific Working Group on Colorectal Cancer. The Asia-Pacific Colorectal Screening score: A validated tool that stratifies risk for colorectal advanced neoplasia in asymptomatic Asian subjects. Gut 2011, 60, 1236–1241. [Google Scholar] [CrossRef]
- O’Donnell, L.J.; Virjee, J.; Heaton, K.W. Detection of pseudodiarrhoea by simple clinical assessment of intestinal transit rate. BMJ 1990, 300, 439–440. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liu, F.; Ling, Z.; Tong, X.; Xiang, C. Human intestinal lumen and mucosa-associated microbiota in patients with colorectal cancer. PLoS ONE 2012, 7, e39743. [Google Scholar] [CrossRef]
- Wang, T.; Cai, G.; Qiu, Y.; Fei, N.; Zhang, M.; Pang, X.; Jia, W.; Cai, S.; Zhao, L. Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers. ISME J. 2012, 6, 320–329. [Google Scholar] [CrossRef]
- Stidham, R.W.; Higgins, P.D.R. Colorectal Cancer in Inflammatory Bowel Disease. Clin. Colon Rectal Surg. 2018, 31, 168–178. [Google Scholar]
- Kabeerdoss, J.; Jayakanthan, P.; Pugazhendhi, S.; Ramakrishna, B.S. Alterations of mucosal microbiota in the colon of patients with inflammatory bowel disease revealed by real time polymerase chain reaction amplification of 16S ribosomal ribonucleic acid. Indian J. Med. Res. 2015, 142, 23–32. [Google Scholar]
- Leeming, E.R.; Johnson, A.J.; Spector, T.D.; Le Roy, C.I. Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration. Nutrients 2019, 11, 2862. [Google Scholar] [CrossRef] [PubMed]
- Haro, C.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Gómez-Delgado, F.; Pérez-Martínez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Landa, B.B.; Navas-Cortés, J.A.; Tena-Sempere, M.; et al. Intestinal Microbiota Is Influenced by Gender and Body Mass Index. PLoS ONE 2016, 11, e0154090. [Google Scholar] [CrossRef] [PubMed]
| Nutrients | Rice Bran (per 30 g Serving) | Rice Powder (per 30 g Serving) |
|---|---|---|
| Calories | 111 kcal | 120 kcal |
| Protein | 3.2 g | 2.0 g |
| Total fats | 2.1 g | 1.0 g |
| Saturated fats | 0.4 g | 0.0 g |
| Trans fats | 0.0 g | 0.0 g |
| Total carbohydrates | 19.7 g | 24.0 g |
| Of which sugars | 1.0 g | <2.0 g |
| Dietary fibre | 3.1 g | 0.0 g |
| Sodium | 1.2 mg | 10 mg |
| Characteristics | Control (n = 20) | Intervention (n = 20) | p-Value |
|---|---|---|---|
| Age (years) † | 65.9 (5.4) | 65.3 (4.7) | 0.663 a |
| Body mass index (kg/m2) † | 24.0 (2.6) | 24.0 (2.7) | 0.961 a |
| Sex | |||
| Male | 10 (50.0%) | 17 (85.0%) | 0.018 b |
| Female | 10 (50.0%) | 3 (15.0%) | |
| Educational level | |||
| No formal education/primary | 4 (20.0%) | 6 (30.0%) | 0.693 b |
| Secondary | 9 (45.0%) | 9 (45.0%) | |
| Post-secondary or above | 7 (35.0%) | 5 (25.0%) | |
| Marital status | |||
| Married | 14 (70.0%) | 18 (90.0%) | 0.235 c |
| Single/divorced/widowed | 6 (30.0%) | 2 (10.0%) | |
| Employment status | |||
| Employed | 6 (30.0%) | 9 (45.0%) | 0.327 b |
| Unemployed/retired | 14 (70.0%) | 11 (55.0%) | |
| Current smoker | |||
| No | 18 (90.0%) | 13 (68.4%) | 0.127 c |
| Yes | 2 (10.0%) | 6 (31.6%) | |
| Regular alcohol drinker | |||
| No | 17 (85.0%) | 13 (68.4%) | 0.273 c |
| Yes | 3 (15.0%) | 6 (31.6%) | |
| Average daily fruit intake | |||
| 0–1 portion | 12 (60.0%) | 13 (65.0%) | 0.744 b |
| ≥2 portions | 8 (40.0%) | 7 (35.0%) | |
| Average daily vegetables intake | |||
| 0–1 portion | 10 (50.0%) | 12 (60.0%) | 0.525 b |
| ≥2 portions | 10 (50.0%) | 8 (40.0%) | |
| Average daily intake of meat, fish, egg and alternatives | |||
| 0–4 tael | 12 (60.0%) | 14 (70.0%) | 0.507 b |
| ≥5 tael | 8 (40.0%) | 6 (30.0%) | |
| Regular meal time | |||
| Yes | 14 (70.0%) | 13 (65.0%) | 0.736 b |
| No | 6 (30.0%) | 7 (35.0%) |
| Control Group (n = 20) | Intervention Group (n = 19) | |||
|---|---|---|---|---|
| T0 (Baseline) | T1 (24 Weeks) | T0 (Baseline) | T1 (24 Weeks) | |
| Absolute abundance (number of reads) | ||||
| Phylum | ||||
| Firmicutes | 19,211 (14,696, 21,670) | 17,224 (14,512, 19,996) | 15,375 (12,747, 16,761) | 16,209 (13,531, 18,752) |
| Bacteroidetes | 16,396 (15,074, 19,594) | 17,674 (14,340, 20,421) | 18,987 (14,466, 22,206) | 19,812 (17,450, 22,988) |
| Proteobacteria | 2703 (1904, 3589) | 3568 (2386, 5157) | 2526 (1926, 5051) | 3954 (2040, 6645) |
| Actinobacteria | 229 (153, 560) | 366 (182, 1018) | 199 (141, 307) | 876 (358, 1336) |
| Fusobacteria | 61 (33, 200) | 52 (15, 136) | 99 (23, 953) | 54 (24, 1613) |
| Firmicutes/Bacteroidetes ratio | 1.190 (0.866, 1.588) | 1.018 (0.816, 1.239) | 0.794 (0.639, 0.979) | 0.775 (0.656, 0.989) |
| Class | ||||
| Bacteroidia | 16,396 (15,074, 19,594) | 17,674 (14,336, 20,420) | 18,987 (14,466, 22,206) | 19,801 (17,450, 22,988) |
| Clostridia | 16,714 (12,641, 20,710) | 15,230 (12,924, 18,568) | 12,133 (10,841, 15,110) | 13,532 (10,966, 15,929) |
| Negativicutes | 1190 (602, 1510) | 1489 (946, 1866) | 834 (634, 1415) | 1652 (971, 2117) |
| Bacilli | 82 (40, 292) | 79 (28, 134) | 108 (43, 357) | 148 (95, 196) |
| Gammaproteobacteria | 2278 (1298, 2863) | 2436 (1836, 4682) | 2254 (1602, 4908) | 2879 (1976, 6249) |
| Order | ||||
| Bacteroidales | 16,394 (15,073, 19,583) | 17,674 (14,230, 20,346) | 18,986 (14,448, 22,206) | 19,699 (17,396, 22,988) |
| Clostridiales | 16,711 (12,640, 20,708) | 15,228 (12,924, 18,567) | 12,130 (10,841, 15,110) | 13,531 (10,966, 15,929) |
| Selenomonadales | 1190 (602, 1510) | 1489 (946, 1866) | 834 (634, 1415) | 1652 (971, 2117) |
| Lactobacillales | 38 (24, 74) | 36 (26, 76) | 36 (19, 53) | 73 (25, 99) |
| Enterobacteriales | 423 (226, 1190) | 976 (557, 3221) | 337 (117, 965) | 454 (216, 865) |
| Family | ||||
| Bacteroidaceae | 13,433 (8114, 16,306) | 12,500 (8170, 17,482) | 10,808 (5397, 15,592) | 12,226 (4698, 15,899) |
| Prevotellaceae | 602 (366, 1484) | 548 (456, 1750) | 1021 (548, 11,807) | 971 (553, 10,564) |
| Clostridiaceae_1 | 50 (30, 83) | 22 (12, 36) | 43 (18, 136) | 33 (15, 59) |
| Lachnospiraceae | 6294 (5660, 8950) | 6947 (5388, 9041) | 6937 (5385, 8577) | 6638 (4647, 8269) |
| Veillonellaceae | 694 (260, 1266) | 812 (340, 1474) | 306 (168, 1075) | 542 (219, 1874) |
| Genus | ||||
| Bacteroides | 13,433 (8114, 16,306) | 12,500 (8170, 17,482) | 10,808 (5397, 15,592) | 12,226 (4698, 15,899) |
| Prevotella_9 | 400 (227, 1000) | 441 (304, 1031) | 472 (369, 9805) | 491 (310, 9629) |
| Clostridium_sensu_stricto_1 | 50 (30, 83) | 22 (12, 34) | 43 (18, 136) | 32 (15, 51) |
| Veillonella | 10 (7, 30) | 41 (14, 95) | 11 (3, 35) | 18 (12, 91) |
| Lactobacillus | 0 (0, 18) | 1 (0, 18) | 0 (0, 31) | 36 (0, 62) |
| Bifidobacterium | 122 (70, 198) | 129 (63, 197) | 63 (30, 124) | 162 (93, 395) |
| Faecalibacterium | 2734 (1470, 3684) | 2270 (1780, 3014) | 2410 (1307, 3467) | 2326 (728, 3324) |
| Escherichia-Shigella | 206 (122, 416) | 421 (273, 1354) | 83 (64, 283) | 206 (133, 322) |
| Blautia | 142 (102, 192) | 108 (87, 204) | 133 (100, 202) | 113 (74, 199) |
| Roseburia | 498 (290, 1160) | 514 (334, 926) | 614 (292, 941) | 605 (371, 1638) |
| Fusobacterium | 52 (30, 200) | 52 (15, 136) | 99 (17, 953) | 54 (24, 1613) |
| Ruminococcus_1 | 224 (133, 529) | 216 (161, 346) | 183 (78, 345) | 174 (84, 262) |
| Ruminococcus_2 | 384 (238, 619) | 508 (282, 762) | 351 (145, 631) | 506 (197, 709) |
| Species | ||||
| Faecalibacterium prausnitzii | 5 (2, 8) | 6 (3, 10) | 2 (0, 6) | 4 (1, 7) |
| Bifidobacterium longum | 18 (5, 32) | 19 (12, 36) | 8 (3, 29) | 20 (12, 38) |
| Clostridium butyricum | 12 (0, 42) | 0 (0, 2) | 0 (0, 38) | 0 (0, 3) |
| Bacteroides fragilis | 45 (17, 192) | 68 (26, 223) | 42 (15, 102) | 68 (23, 126) |
| Change from T0 to T1 (T1–T0) | ||||
|---|---|---|---|---|
| Control Group (n = 20) | Intervention Group (n = 19) | p-Value (Unadjusted) | p-Value (Sex-Adjusted) | |
| Absolute abundance (number of reads) | ||||
| Phylum | ||||
| Firmicutes | −2160 (−4778, 1964) | 1473 (−1351, 5285) | 0.022 | 0.018 |
| Bacteroidetes | 899 (−2052, 4177) | −48 (−3987, 5346) | 0.564 | 0.525 |
| Proteobacteria | 1046 (−512, 2320) | 682 (−1313, 2184) | 0.797 | 0.304 |
| Actinobacteria # | 98 (−82, 698) | 697 (72, 1168) | 0.143 | 0.129 |
| Fusobacteria # | −2 (−40, 30) | −13 (−46, 100) | 0.362 | 0.382 |
| Firmicutes/Bacteroidetes ratio # | −0.131 (−0.447, 0.017) | 0.059 (−0.152, 0.241) | 0.079 | 0.054 |
| Class | ||||
| Bacteroidia | 897 (−2052, 4177) | −48 (−3987, 5335) | 0.564 | 0.525 |
| Clostridia | −2700 (−5066, 2203) | 752 (−2631, 2811) | 0.249 | 0.262 |
| Negativicutes # | 310 (10, 684) | 759 (14, 1474) | 0.266 | 0.289 |
| Bacilli # | −28 (−212, 60) | 31 (−199, 111) | 0.153 | 0.179 |
| Gammaproteobacteria | 984 (−462, 1728) | 469 (−1286, 2175) | 0.810 | 0.662 |
| Order | ||||
| Bacteroidales | 794 (−2063, 4156) | −36 (−3989, 5251) | 0.561 | 0.522 |
| Clostridiales | −2696 (−5065, 2202) | 752 (−2628, 2811) | 0.248 | 0.262 |
| Selenomonadales # | 310 (10, 684) | 759 (14, 1474) | 0.266 | 0.289 |
| Lactobacillales # | −2 (−22, 24) | 38 (−9, 65) | 0.096 | 0.146 |
| Enterobacteriales # | 234 (−39, 1408) | 42 (−125, 380) | 0.563 | 0.754 |
| Family | ||||
| Bacteroidaceae | 480 (−2570, 2966) | 553 (−2703, 2882) | 0.942 | 0.660 |
| Prevotellaceae | 42 (−288, 788) | −389 (−2707, 485) | 0.186 | 0.281 |
| Clostridiaceae_1 | −26 (−48, −3) | −12 (−100, 34) | 0.323 | 0.457 |
| Lachnospiraceae | −88 (−2448, 1773) | −341 (−1757, 1461) | 0.744 | 0.641 |
| Veillonellaceae # | 154 (−170, 402) | 121 (9, 1210) | 0.253 | 0.255 |
| Genus | ||||
| Bacteroides | 480 (−2570, 2966) | 553 (−2703, 2882) | 0.942 | 0.660 |
| Prevotella_9 | 100 (−266, 536) | −159 (−362, 359) | 0.117 | 0.160 |
| Clostridium_sensu_stricto_1 | −28 (−50, −4) | −11 (−100, 32) | 0.342 | 0.481 |
| Veillonella # | 18 (2, 61) | 10 (−2, 67) | 0.841 | 0.831 |
| Lactobacillus | 0 (0, 4) | 19 (0, 43) | 0.026 | 0.033 |
| Bifidobacterium # | 28 (−120, 92) | 91 (12, 162) | 0.125 | 0.131 |
| Faecalibacterium | −486 (−983, 948) | −240 (−1506, 955) | 0.862 | 0.582 |
| Escherichia-Shigella | 190 (20, 898) | 64 (−6, 225) | 0.386 | 0.914 |
| Blautia | −26 (−100, 64) | −5 (−99, 50) | 0.527 | 0.611 |
| Roseburia # | 38 (−196, 420) | 143 (−551, 421) | 0.376 | 0.656 |
| Fusobacterium | 2 (−40, 36) | −3 (−46, 120) | 0.815 | 0.840 |
| Ruminococcus_1 # | 7 (−208, 108) | 29 (−148, 153) | 0.879 | 0.962 |
| Ruminococcus_2 # | 111 (−62, 536) | −16 (−215, 347) | 0.820 | 0.543 |
| Species | ||||
| Faecalibacterium prausnitzii * | 2 (−4, 6) | 1 (0, 4) | 0.841 | 0.682 |
| Bifidobacterium longum * | 10 (−14, 22) | 12 (−3, 27) | 0.365 | 0.261 |
| Clostridium butyricum * | −12 (−39, 0) | 0 (−36, 0) | 0.571 | 0.660 |
| Bacteroides fragilis * | 44 (−16, 84) | 16 (−62, 69) | 0.476 | 0.647 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
So, W.K.W.; Chan, J.Y.W.; Law, B.M.H.; Choi, K.C.; Ching, J.Y.L.; Chan, K.L.; Tang, R.S.Y.; Chan, C.W.H.; Wu, J.C.Y.; Tsui, S.K.W. Effects of a Rice Bran Dietary Intervention on the Composition of the Intestinal Microbiota of Adults with a High Risk of Colorectal Cancer: A Pilot Randomised-Controlled Trial. Nutrients 2021, 13, 526. https://doi.org/10.3390/nu13020526
So WKW, Chan JYW, Law BMH, Choi KC, Ching JYL, Chan KL, Tang RSY, Chan CWH, Wu JCY, Tsui SKW. Effects of a Rice Bran Dietary Intervention on the Composition of the Intestinal Microbiota of Adults with a High Risk of Colorectal Cancer: A Pilot Randomised-Controlled Trial. Nutrients. 2021; 13(2):526. https://doi.org/10.3390/nu13020526
Chicago/Turabian StyleSo, Winnie K. W., Judy Y. W. Chan, Bernard M. H. Law, Kai Chow Choi, Jessica Y. L. Ching, Kam Leung Chan, Raymond S. Y. Tang, Carmen W. H. Chan, Justin C. Y. Wu, and Stephen K. W. Tsui. 2021. "Effects of a Rice Bran Dietary Intervention on the Composition of the Intestinal Microbiota of Adults with a High Risk of Colorectal Cancer: A Pilot Randomised-Controlled Trial" Nutrients 13, no. 2: 526. https://doi.org/10.3390/nu13020526
APA StyleSo, W. K. W., Chan, J. Y. W., Law, B. M. H., Choi, K. C., Ching, J. Y. L., Chan, K. L., Tang, R. S. Y., Chan, C. W. H., Wu, J. C. Y., & Tsui, S. K. W. (2021). Effects of a Rice Bran Dietary Intervention on the Composition of the Intestinal Microbiota of Adults with a High Risk of Colorectal Cancer: A Pilot Randomised-Controlled Trial. Nutrients, 13(2), 526. https://doi.org/10.3390/nu13020526







