The “Fat but Fit” Paradigm and Bone Health in Young Adults: A Cluster Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. The Study Design and Participants
2.2. Study Variables
2.3. Statistical Analysis
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
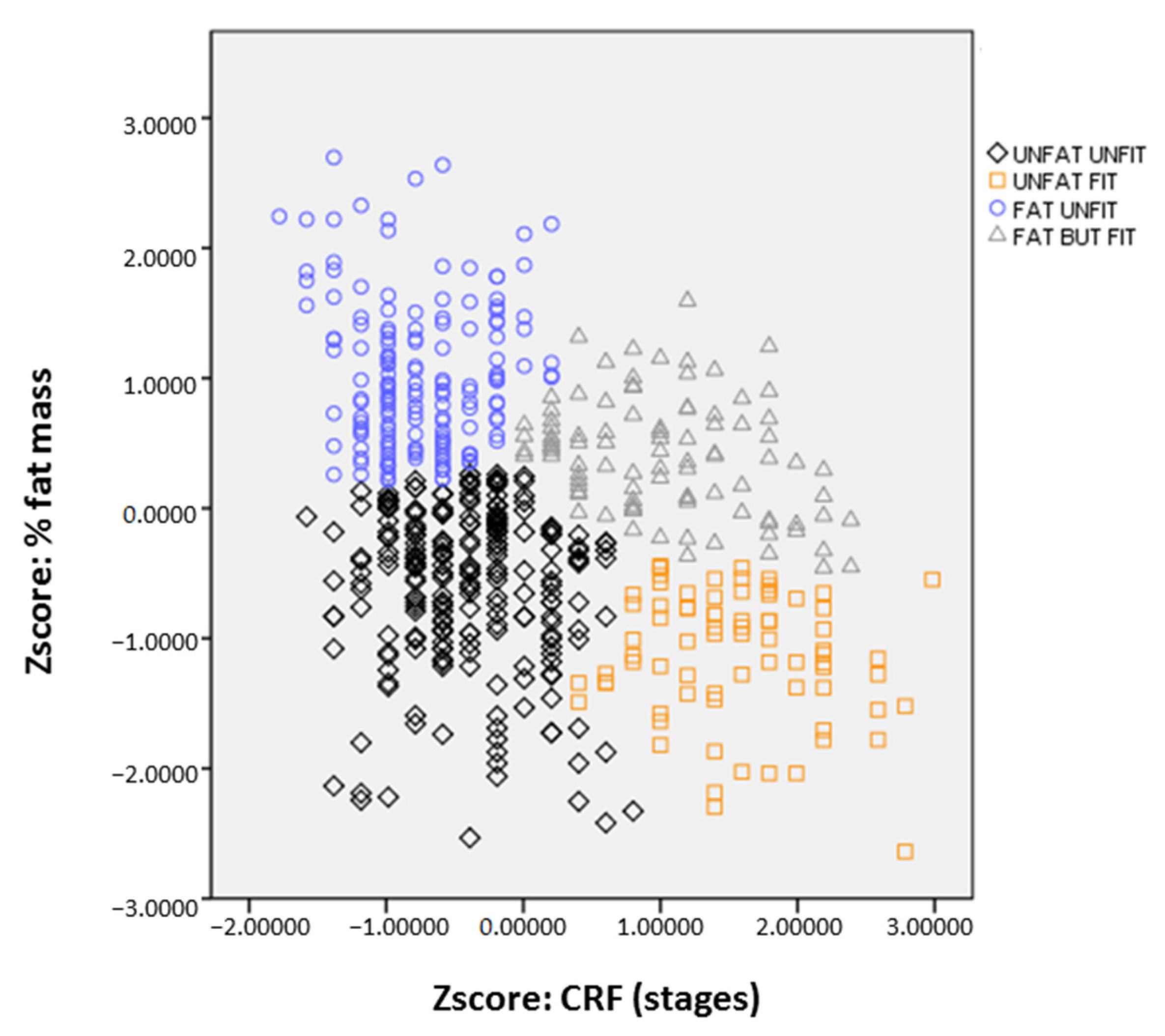
| Cluster (Body Fat %, CRF) | Pairwise Comparisons | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| FU | UU | FF | UF | p Value | 1–2 | 1–3 | 1–4 | 2–3 | 2–4 | 3–4 | |
| n | 142 | 208 | 79 | 70 | |||||||
| Body composition | |||||||||||
| BMI | 0.68 ± 1.10 | −0.56 ± 0.70 | 0.42 ± 0.69 | −0.30 ± 0.49 | <0.001 | > | > | > | < | > | |
| Waist circumference | 0.71 ± 1.01 | −0.48 ± 0.56 | 0.64 ± 0.89 | −0.30 ± 0.54 | <0.001 | > | > | > | < | < | > |
| Body fat % | 0.96 ± 0.56 | −0.01 ± 0.47 | −0.56 ± 0.83 | −1.37 ± 0.54 | <0.001 | > | > | > | ˂ | > | > |
| Total lean mass | −0.13 ± 0.79 | −0.45 ± 0.59 | 1.33 ± 0.90 | 1.17 ± 0.88 | <0.001 | < | < | < | < | < | |
| Total body BMC | −0.09 ± 0.89 | −0.40 ± 0.68 | 1.13 ± 1.19 | 0.91 ± 1.00 | <0.001 | > | ˂ | ˂ | > | > | |
| Total body BMD | −0.02 ± 0.96 | −0.34 ± 0.78 | 0.82 ± 1.21 | 0.61 ± 0.97 | 0.001 | > | ˂ | ˂ | > | > | |
| Physical fitness | |||||||||||
| CRF | −0.75 ± 0.42 | −0.36 ± 0.47 | 1.08 ± 0.63 | 1.59 ± 0.61 | <0.001 | < | < | > | < | ||
| Handgrip strength | −0.30 ± 0.76 | −0.42 ± 0.65 | 1.13 ± 0.87 | 0.95 ± 0.89 | <0.001 | < | < | < | < | ||
| Nutrients | |||||||||||
| Calcium | −0.23 ± 0.63 | −0.04 ± 0.74 | −0.02 ± 0.55 | 0.13 ± 0.83 | 0.048 | < | |||||
| Magnesium | −0.17 ± 0.52 | −0.03 ± 0.74 | −0.02 ± 0.50 | 0.29 ± 1.02 | 0.030 | < | |||||
| Phosphorus | −0.19 ± 0.52 | −0.05 ± 0.78 | −0.03 ± 0.47 | 0.27 ± 0.99 | 0.022 | < | |||||
| Potassium | −0.17 ± 0.56 | −0.02 ± 0.80 | −0.03 ± 0.56 | 0.38 ± 1.10 | 0.023 | < | |||||
| Vitamin D | −0.19 ± 0.80 | −0.02 ± 0.96 | 0.09 ± 0.67 | 0.02 ± 0.87 | 0.401 | ||||||
| Proteins | −0.20 ± 0.49 | −0.04 ± 0.83 | −0.07 ± 0.46 | 0.36 ± 1.10 | 0.012 | < | < | ||||
References
- Ortega, F.B.; Ruiz, J.R.; Labayen, I.; Lavie, C.J.; Blair, S.N. The fat but fit paradox: What we know and don’t know about it. BMJ Publ. Gr. Ltd. Br. Assoc. Sport Exersise Med. 2018, 52, 151–153. [Google Scholar] [CrossRef]
- Ruiz, J.R.; Cavero-Redondo, I.; Ortega, F.B.; Welk, G.J.; Andersen, L.B.; Martinez-Vizcaino, V. Cardiorespiratory fitness cut points to avoid cardiovascular disease risk in children and adolescents; what level of fitness should raise a red flag? A systematic review and meta-analysis. Br. J. Sports Med. 2016, 50, 1451–1458. [Google Scholar] [CrossRef]
- Heaney, R.; Abrams, S.; Dawson-Hughes, B.; Looker, A.; Marcus, R.; Matkovic, V.; Weaver, C. Peak bone mass. Osteoporos. Int. 2000, 11, 985. [Google Scholar] [CrossRef] [PubMed]
- Baxter-Jones, A.D.; Faulkner, R.A.; Forwood, M.R.; Mirwald, R.L.; Bailey, D.A. Bone mineral accrual from 8 to 30 years of age: An estimation of peak bone mass. J. Bone Miner. Res. 2011, 26, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Teegarden, D.; Proulx, W.R.; Martin, B.R.; Zhao, J.; McCabe, G.P.; Lyle, R.M.; Peacock, M.; Slemenda, C.; Johnston, C.C.; Weaver, C.M. Peak bone mass in young women. J. Bone Miner. Res. 1995, 10, 711–715. [Google Scholar] [CrossRef]
- Bland, V.; Heatherington-Rauth, M.; Howe, C.; Going, S.; Bea, J. Association of objectively measured physical activity and bone health in children and adolescents: A systematic review and narrative synthesis. Osteop. Int. 2020, 31, 1865–1894. [Google Scholar] [CrossRef]
- Maillane-Vanegas, S.; Agostinete, R.R.; Lynch, K.R.; Ito, I.H.; Luiz-de-Marco, R.; Rodrigues-Junior, M.A.; Fernandes, R.A. Bone mineral density and sports participation. J. Clin. Dens. 2020, 23, 294–302. [Google Scholar] [CrossRef]
- Atkin, A.J.; Sharp, S.J.; Corder, K.; van Sluijs, E.M.; International Children’s Accelerometry Database (ICAD) Collaborators. Prevalence and correlates of screen time in youth: An international perspective. Am. J. Prev. Med. 2014, 47, 803–807. [Google Scholar] [CrossRef]
- Gracia-Marco, L.; Vicente-Rodriguez, G.; Casajus, J.A.; Molnar, D.; Castillo, M.J.; Moreno, L.A. Effect of fitness and physical activity on bone mass in adolescents: The HELENA Study. Eur. J. Appl. Physiol. 2011, 111, 2671–2680. [Google Scholar] [CrossRef]
- Kindler, J.M.; Pollock, N.K.; Ross, H.L.; Modlesky, C.M.; Singh, H.; Laing, E.M.; Lewis, R.D. Obese versus normal-weight late-adolescent females have inferior trabecular bone microarchitecture: A pilot case-control study. Calcif. Tissue Int. 2017, 101, 479–488. [Google Scholar] [CrossRef]
- Klein, K.O.; Larmore, K.A.; De Lancey, E.; Brown, J.M.; Considine, R.V.; Hassink, S.G. Effect of obesity on estradiol level, and its relationship to leptin, bone maturation, and bone mineral density in children. J. Clin. Endocrinol. Metab. 1998, 83, 3469–3475. [Google Scholar] [CrossRef]
- Kawai, M.; de Paula, F.J.; Rosen, C.J. New insights into osteoporosis: The bone–fat connection. J. Int. Med. 2012, 272, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Pollock, N.K.; Laing, E.; Hamrick, M.W.; Baile, C.; Hall, D.; Lewis, R.D. Bone and fat relationships in postadolescent black females: A pQCT study. Osteoporos. Int. 2011, 22, 655–665. [Google Scholar] [CrossRef]
- Palacios, C. The role of nutrients in bone health, from A to Z. Crit. Rev. Food Sci. Nutr. 2006, 46, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Teegarden, D. Calcium intake and reduction in weight or fat mass. J. Nutr. 2003, 133, 249S–251S. [Google Scholar] [CrossRef] [PubMed]
- Ortega, F.B.; Artero, E.G.; Ruiz, J.R.; Vicente-Rodriguez, G.; Bergman, P.; Hagstromer, M.; Ottevaere, C.; Nagy, E.; Konsta, O.; Rey-López, J.P.; et al. Reliability of health-related physical fitness tests in European adolescents. The HELENA Study. Int. J. Obes. 2008, 32 (Suppl. 5), S49–S57. [Google Scholar] [CrossRef]
- Vicente-Rodriguez, G.; Ara, I.; Perez-Gomez, J.; Serrano-Sanchez, J.A.; Dorado, C.; Calbet, J.A. High femoral bone mineral density accretion in prepubertal soccer players. Med. Sci. Sports Exerc. 2004, 36, 1789–1795. [Google Scholar] [CrossRef]
- Kemper, H.C.; Twisk, J.W.; Van Mechelen, W.; Post, G.B.; Roos, J.C.; Lips, P.T. A fifteen-year longitudinal study in young adults on the relation of physical activity and fitness with the development of the bone mass: The Amsterdam growth and health longitudinal study. Bone 2000, 27, 847–853. [Google Scholar] [CrossRef]
- Smith, J.J.; Eather, N.; Morgan, P.J.; Plotnikoff, R.C.; Faigenbaum, A.D.; Lubans, D.R. The health benefits of muscular fitness for children and adolescents: A systematic review and meta-analysis. Sports Med. 2014, 44, 1209–1223. [Google Scholar] [CrossRef]
- Torres-Costoso, A.; López-Muñoz, P.; Martínez-Vizcaíno, V.; Álvarez-Bueno, C.; Cavero-Redondo, I. Association between muscular strength and bone health from children to young adults: A systematic review and meta-analysis. Sports Med. 2020, 50, 1163–1190. [Google Scholar] [CrossRef]
- Nordstrom, P.; Nordstrom, G.; Thorsen, K.; Lorentzon, R. Local bone mineral density, muscle strength, and exercise in adolescent boys: A comparative study of two groups with different muscle strength and exercise levels. Calcif. Tissue Int. 1996, 58, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, I.; Yamaguchi, T.; Yamauchi, M.; Yamamoto, M.; Kurioka, S.; Yano, S.; Sugimoto, T. Adiponectin is associated with changes in bone markers during glycemic control in type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 2009, 94, 3031–3037. [Google Scholar] [CrossRef]
- Gil-Cosano, J.J.; Gracia-Marco, L.; Ubago-Guisado, E.; Labayen, I.; Adelantado-Renau, M.; Cadenas-Sanchez, C.; Mora-Gonzalez, J.; Plaza-Florido, A.; Aguilera, C.M.; Gómez-Vida, J.; et al. Inflammatory markers and bone mass in children with overweight/obesity: The role of muscular fitness. Pediatr. Res. 2020, 87, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Vizcaino, V.; Alvarez-Bueno, C.; Sanchez-Lopez, M.; Jimenez-Lopez, E.; Soriano-Cano, A.; Ortega, F.B.; Cavero-Redondo, I.; Garrido-Miguel, M. A cluster mediation analysis confirms the validity of the ‘fat but fit’paradigm in children’s cognitive function and academic achievement. J. Pediatr. 2020, 26. [Google Scholar] [CrossRef]
- Torres-Costoso, A.; López-Muñoz, P.; Ferri-Morales, A.; Bravo-Morales, E.; Martínez-Vizcaíno, V.; Garrido-Miguel, M. Body mass index, lean mass, and body fat percentage as mediators of the relationship between milk consumption and bone health in young adults. Nutrients 2019, 11, 2500. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ballart, J.D.; Piñol, J.L.; Zazpe, I.; Corella, D.; Carrasco, P.; Toledo, E.; Pérez-Baurer, M.; Martínez-González, M.A.; Salas-Salvadó, J.; Martín-Moreno, J.M. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br. J. Nutr. 2010, 103, 1808–1816. [Google Scholar] [CrossRef]
- Moreiras, O.; Carbajal, A.; Cabrera, L. Tablas de Composicion de Alimentos, 9th ed.; Editorial Piramide: Madrid, Spain, 2005. [Google Scholar]
- Tinsley, H.E.; Brown, S.D. Handbook of Applied Multivariate Statistics and Mathematical Modeling; Academic Press: Cambridge, MA, USA, 2000. [Google Scholar]
- Miguel-Berges, M.L.; Zachari, K.; Santaliestra-Pasias, A.M.; Mouratidou, T.; Androutsos, O.; Iotova, V.; Galcheva, S.; De Craemer, M.; Cardon, G.; Koletzko, B.; et al. Clustering of energy balance-related behaviours and parental education in European preschool children: The ToyBox study. Br. J. Nutr. 2017, 118, 1089–1096. [Google Scholar] [CrossRef]
- Jaacks, L.M.; Vandevijvere, S.; Pan, A.; McGowan, C.J.; Wallace, C.; Imamura, F.; Mozaffarian, D.; Swinburn, B.; Ezzati, M. The obesity transition: Stages of the global epidemic. Lancet Diab. Endocrinol. 2019, 7, 231–240. [Google Scholar] [CrossRef]
- Lavie, C.J.; McAuley, P.A.; Church, T.S.; Milani, R.V.; Blair, S.N. Obesity and cardiovascular diseases: Implications regarding fitness, fatness, and severity in the obesity paradox. J. Am. Coll. Cardiol. 2014, 63, 1345–1354. [Google Scholar] [CrossRef]
- Elagizi, A.; Kachur, S.; Lavie, C.J.; Carbone, S.; Pandey, A.; Ortega, F.B.; Milani, R.V. An overview and update on obesity and the obesity paradox in cardiovascular diseases. Prog. Cardiovasc. Dis. 2018, 61, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Felson, D.T.; Zhang, Y.; Hannan, M.T.; Anderson, J.J. Effects of weight and body mass index on bone mineral density in men and women: The Framingham study. J. Bone Miner. Res. 1993, 8, 567–573. [Google Scholar] [CrossRef]
- Cao, J.J. Effects of obesity on bone metabolism. J. Orthop. Surg. Res. 2011, 6, 30. [Google Scholar] [CrossRef]
- Torres-Costoso, A.; Gracia-Marco, L.; Sanchez-Lopez, M.; Garcia-Prieto, J.C.; Garcia-Hermoso, A.; Diez-Fernandez, A.; Martínez-Vizcaíno, V. Lean mass as a total mediator of the influence of muscular fitness on bone health in schoolchildren: A mediation analysis. J. Sports Sci. 2015, 33, 817–830. [Google Scholar] [CrossRef]
- Carbone, S.; Canada, J.M.; Billingsley, H.E.; Siddiqui, M.S.; Elagizi, A.; Lavie, C.J. Obesity paradox in cardiovascular disease: Where do we stand? Vasc. Health Risk Manag. 2019, 15, 89. [Google Scholar] [CrossRef] [PubMed]
- Torres-Costoso, A.; Gracia-Marco, L.; Sanchez-Lopez, M.; Notario-Pacheco, B.; Arias-Palencia, N.; Martinez-Vizcaino, V. Physical activity and bone health in schoolchildren: The mediating role of fitness and body fat. PLoS ONE 2015, 10, e0123797. [Google Scholar] [CrossRef] [PubMed]
- Zemel, M.B. Regulation of adiposity and obesity risk by dietary calcium: Mechanisms and implications. J. Am. Coll. Nutr. 2020, 21, 146S–151S. [Google Scholar] [CrossRef]
- Jacobsen, R.; Lorenzen, J.K.; Toubro, S.; Krog-Mikkelsen, I.; Astrup, A. Effect of short-term high dietary calcium intake on 24 h energy expenditure, fat oxidation, and fecal fat excretion. Int. J. Obes. 2005, 29, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Densitometry. 2019 ISCD Official Positions–Adult. Available online: https://wwwiscdorg/official-positions/2019-iscd-official-positions-adult (accessed on 20 October 2020).
| Male n = 169 | Female n = 330 | p Value | |
|---|---|---|---|
| Age (years) | 20.53 ± 2.71 | 20.02 ± 2.79 | 0.209 |
| Weight (kg) | 72.92 ± 11.35 | 59.74 ± 10.50 | 0.026 |
| Height (cm) | 175.66 ± 7.23 | 162.04 ± 12.96 | 0.743 |
| BMI (kg/m2) | 23.59 ± 3.17 | 22.51 ± 3.70 | 0.044 |
| Waist circumference (cm) | 83.53 ± 8.14 | 78.18 ± 8.70 | 0.820 |
| Body fat % | 20.55 ± 6.68 | 31.93 ± 7.02 | 0.979 |
| Total body lean mass (kg) | 54.11 ± 6.78 | 37.50 ± 4.52 | <0.001 |
| CRF (stages) | 8.07 ± 2.13 | 4.13 ± 1.40 | <0.001 |
| Handgrip strength (kg) | 40.12 ± 7.23 | 24.68 ± 4.13 | <0.001 |
| Total body BMC (g) | 2904.66 ± 482.13 | 2243.55 ± 279.95 | <0.001 |
| Total body BMD (g·cm−2) | 1.23 ± 0.12 | 1.12 ± 0.09 | <0.001 |
| Calcium(mg/d) | 1277.73 ± 1042.99 | 1179.91 ± 571.67 | 0.327 |
| Magnesium (mg/day) | 499.69 ± 485.61 | 452.41 ± 230.55 | 0.290 |
| Phosphorus (mg/day) | 2160.89 ± 179.13 | 1963.65 ± 881.70 | 0.237 |
| Potassium (mg/day) | 5351.02 ± 4955.63 | 4894.47 ± 2615.83 | 0.330 |
| Vitamin D (µg/day) | 8.64 ± 6.34 | 7.77 ± 5.99 | 0.254 |
| Proteins (kcal/day) | 510.78 ± 424.64 | 454.88 ± 209.42 | 0.157 |
| Body Fat % | Total Body Lean Mass | CRF | Handgrip | Total Body BMD | Total Body BMC | Calcium | Magnesium | Phosphorus | Potassium | Vitamin D | Proteins | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Height | 0.046 | 0.392 ** | 0.419 ** | 0.222 ** | 0.176 ** | 0.404 ** | −0.012 | 0.019 | 0.001 | 0.013 | 0.010 | 0.025 |
| Body fat % | - | 0.095 * | −0.668 ** | −0.460 ** | 0.157 ** | 0.128 * | −0.149 ** | −0.148 ** | −0.146 ** | −0.154 ** | −0.116 | −0.159 * |
| Total body lean mass | - | 0.648 ** | 0.837 ** | 0.481 ** | 0.775 ** | −0.006 | 0.012 | 0.015 | 0.024 | 0.074 | 0.006 | |
| CRF | - | 0.633 ** | 0.330 ** | 0.483 ** | 0.129 | 0.164 * | 0.149 * | 0.153 * | 0.111 | 0.154 * | ||
| Handgrip | - | 0.533 ** | 0.753 ** | 0.090 | 0.119 | 0.108 | 0.113 | 0.105 | 0.116 | |||
| Total body BMD | - | 0.802 ** | 0.090 | 0.114 | 0.122 | 0.121 | 0.034 | 0.038 | ||||
| Total body BMC | - | 0.105 | 0.152 | 0.153 * | 0.156 * | 0.080 | 0.032 | |||||
| Calcium | - | 0.881 ** | 0.921 ** | 0.857 ** | 0.477 ** | 0.855 ** | ||||||
| Magnesium | - | 0.932 ** | 0.972 ** | 0.550 ** | 0.905 ** | |||||||
| Phosphorus | - | 0.929 ** | 0.551 ** | 0.969 ** | ||||||||
| Potassium | . | 0.541 ** | 0.897 ** | |||||||||
| Vitamin D | 0.574 ** |
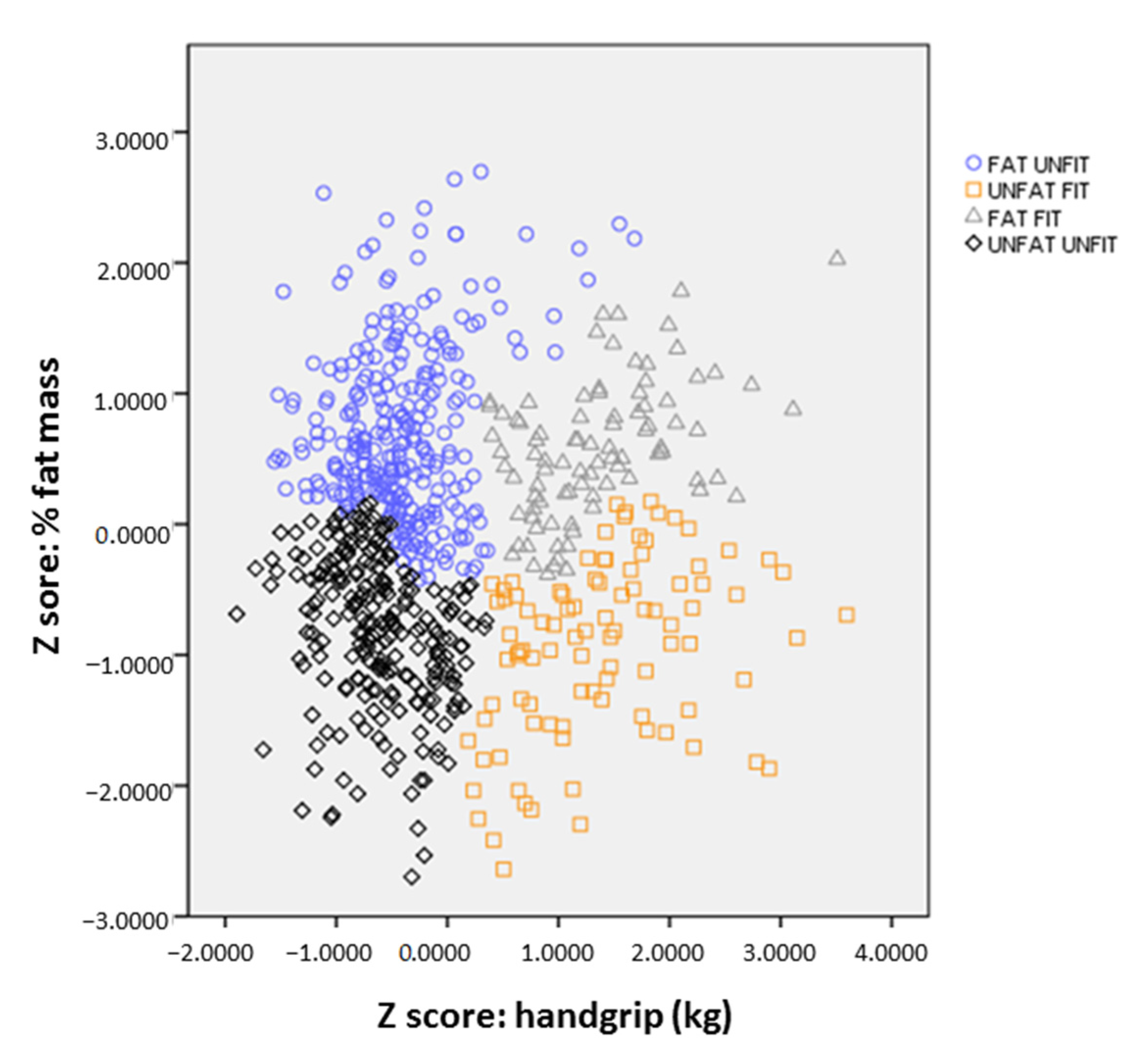
| Cluster (Body Fat %, Handgrip) | Pairwise Comparisons | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| FU | UU | FF | UF | p Value | 1–2 | 1–3 | 1–4 | 2–3 | 2–4 | 3–4 | |
| n | 193 | 156 | 72 | 78 | |||||||
| Body composition | |||||||||||
| BMI | 0.44 ± 1.03 | −0.73 ± 0.49 | 0.51 ± 0.85 | −0.14 ± 0.51 | <0.001 | > | > | < | |||
| Waist circumference | 0.43 ± 0.92 | −0.71 ± 0.56 | 0.84 ± 0.89 | 0.01 ± 0.75 | <0.001 | > | > | < | < | > | |
| Body fat % | 0.81 ± 0.67 | −0.34 ± 0.48 | −0.45 ± 0.54 | −1.51 ± 0.53 | <0.001 | > | > | ˂ | > | ||
| Total lean mass | −0.19 ± 0.77 | −0.53 ± 0.75 | 1.17 ± 0.60 | 1.16 ± 0.59 | <0.001 | < | < | < | |||
| Total body BMC | −0.16 ± 0.79 | −0.49 ± 0.80 | 1.10 ± 0.71 | 1.00 ± 0.74 | <0.001 | ˂ | ˂ | > | > | ||
| Total body BMD | −0.06 ± 0.88 | −0.33 ± 0.89 | 0.82 ± 0.96 | 0.66 ± 0.91 | 0.001 | ˂ | ˂ | > | > | ||
| Physical fitness | |||||||||||
| CRF | −0.59 ± 0.59 | −0.26 ± 0.75 | 0.67 ± 0.74 | 1.32 ± 0.84 | <0.001 | < | < | > | < | ||
| Handgrip strength | −0.41 ± 0.49 | −0.61 ± 0.44 | 1.35 ± 0.73 | 1.38 ± 0.76 | <0.001 | < | < | < | < | ||
| Nutrients | |||||||||||
| Calcium | −0.16 ± 0.70 | 0.04 ± 0.80 | −0.06 ± 0.62 | 0.53 ± 2.47 | 0.048 | < | |||||
| Magnesium | −0.11 ± 0.62 | 0.02 ± 0.76 | −0.08 ± 0.53 | 0.69 ± 2.73 | 0.019 | < | < | ||||
| Phosphorus | −0.11 ± 0.70 | −0.06 ± 0.75 | −0.07 ± 0.49 | 0.67 ± 2.74 | 0.026 | < | |||||
| Potassium | −0.10 ± 0.67 | 0.04 ± 0.85 | −0.07 ± 0.58 | 0.73 ± 2.74 | 0.017 | < | < | ||||
| Vitamin D | −0.14 ± 0.82 | −0.04 ± 0.94 | 0.06 ± 0.73 | 0.17 ± 1.21 | 0.404 | ||||||
| Proteins | −0.13 ± 0.65 | −0.01 ± 0.80 | −0.08 ± 0.50 | 0.72 ± 2.72 | 0.011 | < | < | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres-Costoso, A.; Garrido-Miguel, M.; Gracia-Marco, L.; López-Muñoz, P.; Reina-Gutiérrez, S.; Núñez de Arenas-Arroyo, S.; Martínez-Vizcaíno, V. The “Fat but Fit” Paradigm and Bone Health in Young Adults: A Cluster Analysis. Nutrients 2021, 13, 518. https://doi.org/10.3390/nu13020518
Torres-Costoso A, Garrido-Miguel M, Gracia-Marco L, López-Muñoz P, Reina-Gutiérrez S, Núñez de Arenas-Arroyo S, Martínez-Vizcaíno V. The “Fat but Fit” Paradigm and Bone Health in Young Adults: A Cluster Analysis. Nutrients. 2021; 13(2):518. https://doi.org/10.3390/nu13020518
Chicago/Turabian StyleTorres-Costoso, Ana, Miriam Garrido-Miguel, Luis Gracia-Marco, Purificación López-Muñoz, Sara Reina-Gutiérrez, Sergio Núñez de Arenas-Arroyo, and Vicente Martínez-Vizcaíno. 2021. "The “Fat but Fit” Paradigm and Bone Health in Young Adults: A Cluster Analysis" Nutrients 13, no. 2: 518. https://doi.org/10.3390/nu13020518
APA StyleTorres-Costoso, A., Garrido-Miguel, M., Gracia-Marco, L., López-Muñoz, P., Reina-Gutiérrez, S., Núñez de Arenas-Arroyo, S., & Martínez-Vizcaíno, V. (2021). The “Fat but Fit” Paradigm and Bone Health in Young Adults: A Cluster Analysis. Nutrients, 13(2), 518. https://doi.org/10.3390/nu13020518









