Greater Loss of Central Adiposity from Low-Carbohydrate versus Low-Fat Diet in Middle-Aged Adults with Overweight and Obesity
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Recruitment
2.2. Diet Prescriptions
2.2.1. Low-Carbohydrate High-Fat Diet (LCHF)
2.2.2. Low-Calorie Low-Fat Diet (LF)
2.2.3. Diet Selection and Adherence
2.3. Study Outcomes and Tests
2.3.1. Body Composition
2.3.2. Resting Energy Expenditure
2.3.3. Glucose, Insulin and Lipids
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
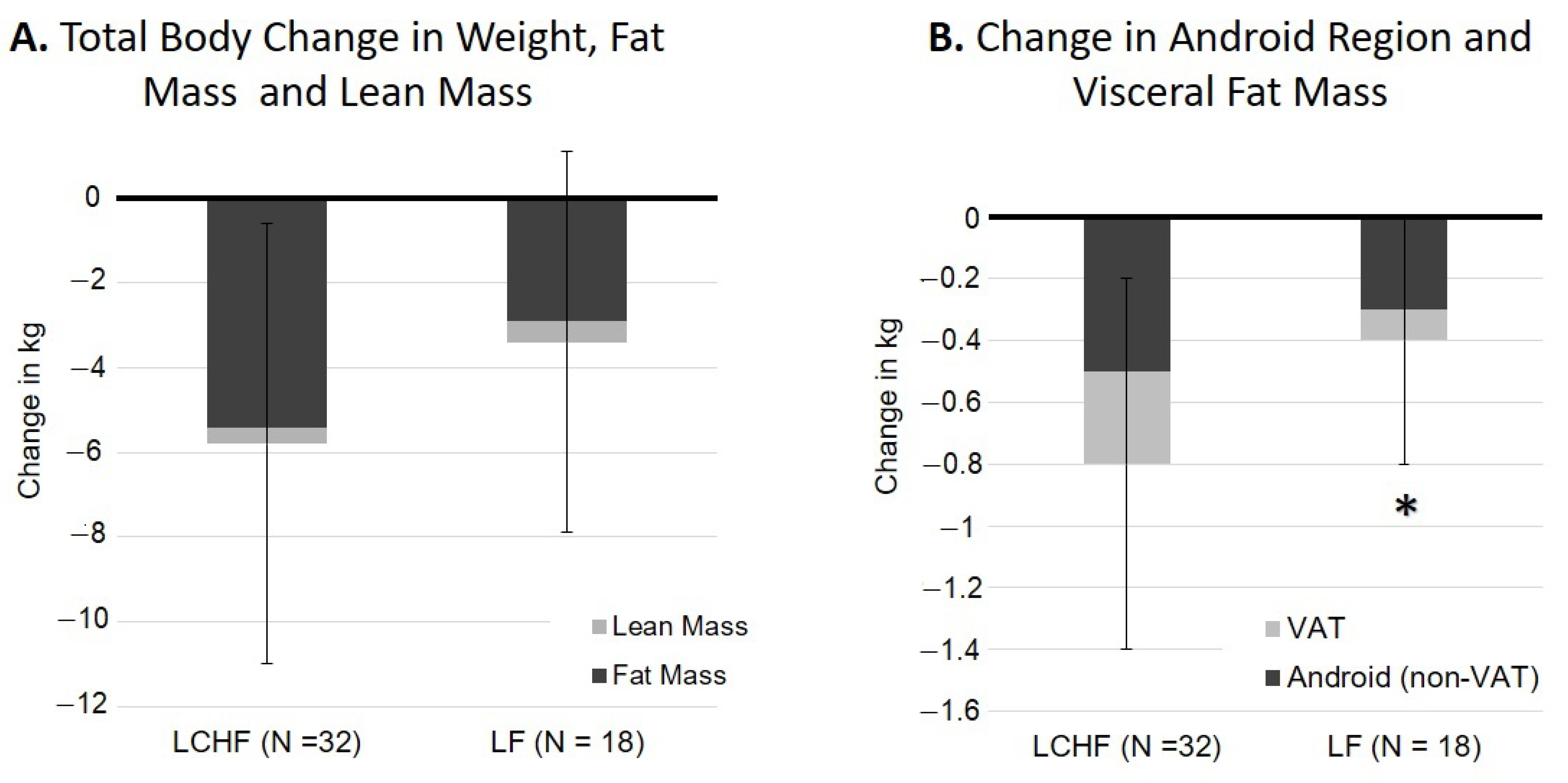
3.2. Total Fat and Lean Mass Loss
3.3. Regional Fat and Lean Mass Loss
3.4. Serum Measures
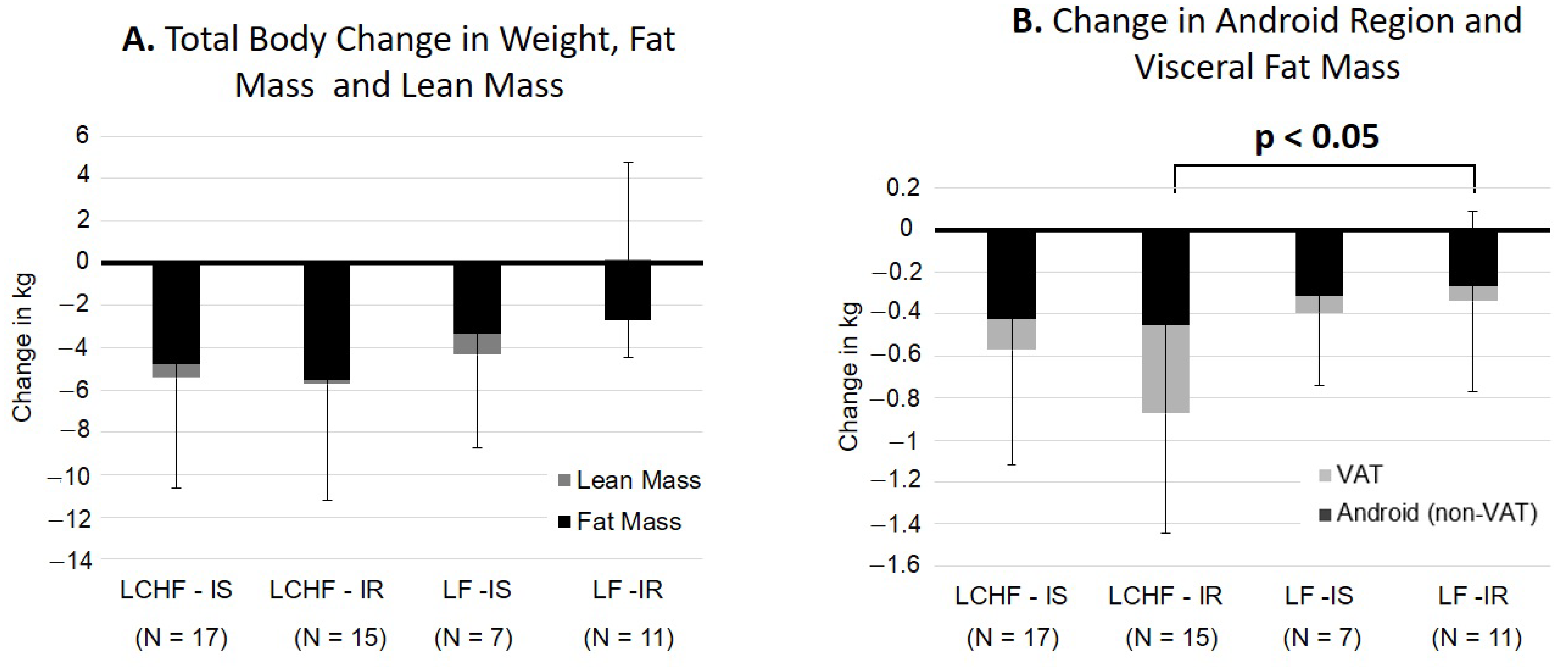
3.5. Subgroup Analysis by Insulin Resistance Status
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jura, M.; Kozak, L.P. Obesity and related consequences to ageing. AGE 2016, 38. [Google Scholar] [CrossRef]
- Locher, J.L.; Goldsby, T.U.; Goss, A.M.; Kilgore, M.L.; Gower, B.; Ard, J.D. Calorie restriction in overweight older adults: Do benefits exceed potential risks? Exp. Gerontol. 2016, 86, 4–13. [Google Scholar] [CrossRef]
- Hunter, G.R.; Gower, B.A.; Kane, B.L. Age Related Shift in Visceral Fat. Int. J. Body Compos. Res. 2010, 8, 103–108. [Google Scholar]
- Brinkworth, G.D.; Wycherley, T.P.; Noakes, M.; Buckley, J.D.; Clifton, P.M. Long-term effects of a very-low-carbohydrate weight-loss diet and an isocaloric low-fat diet on bone health in obese adults. Nutrition 2016, 32, 1033–1036. [Google Scholar] [CrossRef]
- Bravata, D.M.; Sanders, L.; Huang, J.; Krumholz, H.M.; Olkin, I.; Gardner, C.D.; Bravata, D.M. Efficacy and safety of low-carbohydrate diets: A systematic review. JAMA 2003, 289, 1837–1850. [Google Scholar] [CrossRef]
- Buyken, A.E.; Mela, D.J.; Dussort, P.; Johnson, I.T.; Macdonald, I.A.; Stowell, J.D.; Brouns, F.J.P.H. Dietary carbohydrates: A review of international recommendations and the methods used to derive them. Eur. J. Clin. Nutr. 2018, 72, 1625–1643. [Google Scholar] [CrossRef]
- Gower, B.A.; Goss, A.M. A Lower-Carbohydrate, Higher-Fat Diet Reduces Abdominal and Intermuscular Fat and Increases Insulin Sensitivity in Adults at Risk of Type 2 Diabetes. J. Nutr. 2015, 145, 177S–183S. [Google Scholar] [CrossRef]
- Gardner, C.D.; Trepanowski, J.F.; Del Gobbo, L.C. Effect of low-fat vs low-carbohydrate diet on 12-month weight loss in overweight adults and the association with genotype pattern or insulin secretion: The dietfits randomized clinical trial. JAMA 2018, 319, 667–679. [Google Scholar] [CrossRef]
- Goss, A.M.; Goree, L.L.; Ellis, A.C.; Chandler-Laney, P.C.; Casazza, K.; Lockhart, M.E.; Gower, B.A. Effects of diet macronutrient composition on body composition and fat distribution during weight maintenance and weight loss. Obesity 2013, 21, 1139–1142. [Google Scholar] [CrossRef]
- Goss, A.M.; Gower, B.; Soleymani, T.; Stewart, M.; Pendergrass, M.; Lockhart, M.; Krantz, O.; Dowla, S.; Bush, N.; Barry, V.G. Effects of weight loss during a very low carbohydrate diet on specific adipose tissue depots and insulin sensitivity in older adults with obesity: A randomized clinical trial. Nutr. Metab. 2020, 17, 1–12. [Google Scholar]
- Lee, J.-K.; Wu, C.-K.; Lin, L.-Y.; Cheng, C.-L.; Lin, J.-W.; Hwang, J.-J.; Chiang, F.-T. Insulin resistance in the middle-aged women with “Tigerish Back and Bearish Waist”. Diabetes Res. Clin. Pract. 2010, 90, e85–e87. [Google Scholar] [CrossRef]
- Piché, M.-E.; Poirier, P.; Lemieux, I.; Després, J.-P. Overview of Epidemiology and Contribution of Obesity and Body Fat Distribution to Cardiovascular Disease: An Update. Prog. Cardiovasc. Dis. 2018. [Google Scholar] [CrossRef]
- Hjorth, M.F.; Ritz, C.; Blaak, E.E.; Saris, W.H.; Langin, D.; Poulsen, S.K.; Larsen, T.M.; Sørensen, T.I.; Zohar, Y.; Astrup, A. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: Results from 3 randomized clinical trials. Am. J. Clin. Nutr. 2017, 106, 499–505. [Google Scholar] [CrossRef]
- Ryan, A.S.; Ortmeyer, H.K.; Sorkin, J.D. Exercise with calorie restriction improves insulin sensitivity and glycogen synthase activity in obese postmenopausal women with impaired glucose tolerance. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E145–E152. [Google Scholar] [CrossRef]
- Pittas, A.G.; Das, S.K.; Hajduk, C.L.; Golden, J.; Saltzman, E.; Stark, P.C.; Greenberg, A.S.; Roberts, S.B. A Low-Glycemic Load Diet Facilitates Greater Weight Loss in Overweight Adults With High Insulin Secretion but Not in Overweight Adults With Low Insulin Secretion in the CALERIE Trial. Diabetes Care 2005, 28, 2939–2941. [Google Scholar] [CrossRef]
- Waters, D.L.; Ward, A.L.; Villareal, D.T. Weight loss in obese adults 65years and older: A review of the controversy. Exp. Gerontol. 2013, 48, 1054–1061. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar]
- Goran, M.I.; Poehlman, E.T. Total energy expenditure and energy requirements in healthy elderly persons. Metabolism 1992, 41, 744–753. [Google Scholar] [CrossRef]
- Stults-Kolehmainen, M.A.; Stanforth, P.R.; Bartholomew, J.B.; Lu, T.; Abolt, C.J.; Sinha, R. DXA estimates of fat in abdominal, trunk and hip regions varies by ethnicity in men. Nutr. Diabetes 2013, 3, e64. [Google Scholar] [CrossRef]
- Micklesfield, L.K.; Goedecke, J.H.; Punyanitya, M.; Wilson, K.E.; Kelly, T.L. Dual-Energy X-Ray Performs as Well as Clinical Computed Tomography for the Measurement of Visceral Fat. Obesity 2012, 20, 1109–1114. [Google Scholar] [CrossRef]
- Hind, K.; Oldroyd, B. In-vivo precision of the GE Lunar iDXA densitometer for the measurement of appendicular and trunk lean and fat mass. Eur. J. Clin. Nutr. 2013, 67, 1331–1333. [Google Scholar] [CrossRef]
- Hind, K.; Oldroyd, B.; Truscott, J.G. In vivo precision of the GE Lunar iDXA densitometer for the measurement of total body composition and fat distribution in adults. Eur. J. Clin. Nutr. 2011, 65, 140–142. [Google Scholar] [CrossRef]
- Kaul, S.; Rothney, M.P.; Peters, D.M.; Wacker, W.K.; Davis, C.E.; Shapiro, M.D.; Ergun, D.L. Dual-Energy X-Ray Absorptiometry for Quantification of Visceral Fat. Obesity 2012, 20, 1313–1318. [Google Scholar] [CrossRef]
- Wallace, T.M.; Matthews, D.R. The assessment of insulin resistance in man. Diabet. Med. 2002, 19, 527–534. [Google Scholar] [CrossRef]
- Qu, H.-Q.; Li, Q.; Rentfro, A.R.; Fisher-Hoch, S.P.; McCormick, J.B. The Definition of Insulin Resistance Using HOMA-IR for Americans of Mexican Descent Using Machine Learning. PLoS ONE 2011, 6, e21041. [Google Scholar] [CrossRef]
- Association, A.D. Glycemic Targets. Diabetes Care 2017, 40, S48–S56. [Google Scholar] [CrossRef]
- Freedland, E.S. Role of a critical visceral adipose tissue threshold (CVATT) in metabolic syndrome: Implications for controlling dietary carbohydrates: A review. Nutr. Metab. 2004, 1, 1–12. [Google Scholar] [CrossRef]
- Magkos, F.; Fraterrigo, G.; Yoshino, J.; Luecking, C.; Kirbach, K.; Kelly, S.C.; De Las Fuentes, L.; He, S.; Okunade, A.L.; Patterson, B.W. Effects of moderate and subsequent progressive weight loss on metabolic function and adipose tissue biology in humans with obesity. Cell Metab. 2016, 23, 591–601. [Google Scholar]
- Feinman, R.D.; Pogozelski, W.K.; Astrup, A.; Bernstein, R.K.; Fine, E.J.; Westman, E.C.; Accurso, A.; Frassetto, L.; Gower, B.A.; McFarlane, S.I.; et al. Dietary carbohydrate restriction as the first approach in diabetes management: Critical review and evidence base. Nutrition 2015, 31, 1–13. [Google Scholar] [CrossRef]
- Willoughby, D.; Hewlings, S.; Kalman, D. Body Composition Changes in Weight Loss: Strategies and Supplementation for Maintaining Lean Body Mass, a Brief Review. Nutrients 2018, 10, 1876. [Google Scholar] [CrossRef]
- Walsh, C.O.; Ebbeling, C.B.; Swain, J.F.; Markowitz, R.L.; Feldman, H.A.; Ludwig, D.S. Effects of diet composition on postprandial energy availability during weight loss maintenance. PLoS ONE 2013, 8, e58172. [Google Scholar]
- Scott, R.V.; Bloom, S.R. Problem or solution: The strange story of glucagon. Peptides 2018, 100, 36–41. [Google Scholar] [CrossRef]
- Iannuzzi-Sucich, M.; Prestwood, K.M.; Kenny, A.M. Prevalence of Sarcopenia and Predictors of Skeletal Muscle Mass in Healthy, Older Men and Women. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2002, 57, M772–M777. [Google Scholar] [CrossRef]
- Foster, M.T.; Pagliassotti, M.J. Metabolic alterations following visceral fat removal and expansion: Beyond anatomic location. Adipocyte 2012, 1, 192–199. [Google Scholar] [CrossRef]
- Schoeller, D.A.; Buchholz, A.C. Energetics of obesity and weight control: Does diet composition matter? J. Am. Diet. Assoc. 2005, 105, S24–S28. [Google Scholar] [CrossRef]
- Ruth, M.R.; Port, A.M.; Shah, M.; Bourland, A.C.; Istfan, N.W.; Nelson, K.P.; Gokce, N.; Apovian, C.M. Consuming a hypocaloric high fat low carbohydrate diet for 12weeks lowers C-reactive protein, and raises serum adiponectin and high density lipoprotein-cholesterol in obese subjects. Metabolism 2013, 62, 1779–1787. [Google Scholar] [CrossRef]
- Tighe, P.; Duthie, G.; Vaughan, N.; Brittenden, J.; Simpson, W.G.; Duthie, S.; Mutch, W.; Wahle, K.; Horgan, G.; Thies, F. Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: A randomized controlled trial. Am. J. Clin. Nutr. 2010, 92, 733–740. [Google Scholar] [CrossRef]
- Volek, J.S.; Sharman, M.J.; Gomez, A.L.; DiPasquale, C.; Roti, M.; Pumerantz, A.; Kraemer, W.J. Comparison of a very low-carbohydrate and low-fat diet on fasting lipids, LDL subclasses, insulin resistance, and postprandial lipemic responses in overweight women. J. Am. Coll. Nutr. 2004, 23, 177–184. [Google Scholar]
- Hu, T.; Mills, K.T.; Yao, L.; Demanelis, K.; Eloustaz, M.; Yancy Jr, W.S.; Kelly, T.N.; He, J.; Bazzano, L.A. Effects of Low-Carbohydrate Diets Versus Low-Fat Diets on Metabolic Risk Factors: A Meta-Analysis of Randomized Controlled Clinical Trials. Am. J. Epidemiol. 2012, 176, S44–S54. [Google Scholar]
- Farquhar, J.W.; Frank, A.; Gross, R.C.; Reaven, G.M. Glucose, insulin, and triglyceride responses to high and low carbohydrate diets in man. J. Clin. Investig. 1966, 45, 1648–1656. [Google Scholar] [CrossRef]
- Parks, E.J.; Krauss, R.M.; Christiansen, M.P.; Neese, R.A.; Hellerstein, M.K. Effects of a low-fat, high-carbohydrate diet on VLDL-triglyceride assembly, production, and clearance. J. Clin. Investig. 1999, 104, 1087–1096. [Google Scholar] [CrossRef]
- Volek, J.S.; Fernandez, M.L.; Feinman, R.D.; Phinney, S.D. Dietary carbohydrate restriction induces a unique metabolic state positively affecting atherogenic dyslipidemia, fatty acid partitioning, and metabolic syndrome. Prog. Lipid Res. 2008, 47, 307–318. [Google Scholar]
- Yancy, W.S.; Mayer, S.B.; Coffman, C.J.; Smith, V.A.; Kolotkin, R.L.; Geiselman, P.J.; McVay, M.A.; Oddone, E.Z.; Voils, C.I. Effect of Allowing Choice of Diet on Weight Loss. A Randomized Trial. Ann. Intern. Med. 2015, 162, 805–814. [Google Scholar] [CrossRef]
- McVay, M.A.; Voils, C.I.; Coffman, C.J.; Geiselman, P.J.; Kolotkin, R.L.; Mayer, S.B.; Smith, V.A.; Gaillard, L.; Turner, M.J.; Yancy, W.S. Factors associated with choice of a low-fat or low-carbohydrate diet during a behavioral weight loss intervention. Appetite 2014, 83, 117–124. [Google Scholar] [CrossRef]
- Dalle Grave, R.; Calugi, S.; Compare, A.; El Ghoch, M.; Petroni, M.L.; Tomasi, F.; Mazzali, G.; Marchesini, G. Weight Loss Expectations and Attrition in Treatment-Seeking Obese Women. Obes. Facts 2015, 8, 311–318. [Google Scholar] [CrossRef]
| LCHF Diet (n = 32) | LF Diet (n = 18) | p-Value | |
|---|---|---|---|
| Age, years | 52.8 (2.1) | 48.4 (10.3) | 0.69 |
| BMI, kg/m2 | 36.3 (7.6) | 37.4 (6.9) | 0.61 |
| REE, kcal/day | 1562.6 (275.8) | 1613.7 (339.6) | 0.33 |
| Gender, % | |||
| Female | 81.3 | 83.3 | 0.85 |
| Male | 18.8 | 16.7 | |
| Race, % | |||
| European American | 81.3 | 61.1 | 0.12 |
| African American | 18.8 | 38.9 | |
| Insulin Resistance, % | |||
| HOMA-IR < 2.6 (IS) | 70.8 | 57.7 | 0.39 |
| HOMA-IR ≥ 2.6 (IR) | 29.1 | 42.3 | |
| Diabetes, % | |||
| HbA1C > 6.5 | 87.5 | 83.33 | 0.69 |
| HbA1C ≤ 6.5 | 12.5 | 16.37 | |
| Regular Exercise, % | |||
| Yes | 42.9 | 31.3 | 0.53 |
| No | 57.1 | 68.8 | |
| Dropouts, % | 31.9 | 43.8 | 0.28 |
| LCHF Diet (n = 32) | LF Diet (n = 18) | p-Value | |||
|---|---|---|---|---|---|
| Baseline | Follow-Up | Baseline | Follow-Up | ||
| Demographics | |||||
| Weight, kg | 100.1 (24.7) | 94.0 (24.1) *** | 105.3 (18.9) | 102.1 (19.4) ** | 0.1615 |
| BMI, kg/m2 | 36.1 (7.7) | 33.9 (7.4) *** | 37.4 (6.9) | 36.3 (7.1) ** | 0.1713 |
| Fat, % | 45.3 (7.4) | 42.6 (7.7) *** | 45.8 (5.2) | 44.3 (5.3) *** | 0.0864 |
| REE, kcal/day | 1562.6 (275.8) | 1531 (285.7) | 1613.7 (339.6) | 1556.3 (304.9) | 0.1199 |
| Region Fat Mass—DXA | |||||
| Total Fat, kg | 46.1 (15.8) | 40.7(15.0) *** | 48.7 (13.2) | 45.8 (12.9) ** | 0.055 |
| Appendicular Fat, kg | 20.3 (8.7) | 23.6 (6.4) *** | 21.7 (6.8) | 20.6 (7.1) ** | 0.193 |
| Trunk Fat, kg | 25.3 (9.1) | 21.9 (8.1) *** | 26.0(7.7) | 24.2 (7.2) ** | 0.049 |
| Android Fat, kg | 4.6 (1.8) | 3.9 (1.6) *** | 4.7 (1.4) | 4.3 (1.4) *** | 0.027 |
| Gynoid Fat, kg | 7.8 (3.0) | 6.8 (2.9) *** | 7.9 (2.7) | 7.4 (2.5) ** | 0.068 |
| Visceral Fat, kg | 1.6 (1.3) | 1.4 (1.0) *** | 1.8 (0.9) | 1.6 (0.9) | 0.019 |
| Region Lean Mass—DXA | |||||
| Total lean, kg | 50.6 (11.4) | 50.1 (1.2) | 52.7 (7.2) | 52.4 (7.7) | 0.997 |
| Trunk lean, kg | 23.7 (5.5) | 23.3 (5.7) * | 24.0 (3.1) | 23.8 (3.3) | 0.804 |
| Android lean, kg | 3.8 (1.0) | 3.7 (1.0) ** | 3.8 (0.6) | 3.8 (0.6) | 0.542 |
| Gynoid lean, kg | 8.1 (1.9) | 7.8 (1.9) ** | 8.1 (1.1) | 8.1(1.2) | 0.250 |
| Appendicular lean, kg | 23.7 (5.8) | 23.6 (6.4) | 25.3 (4.3) | 25.3 (4.5 | 0.996 |
| Serum Analytes | |||||
| Glucose, mg/dl | 109.4 (39.6) | 103.4 (12.6) | 107.7 (33.5) | 116.7 (59.4) | 0.2529 |
| Insulin, µU/mL | 14.0 (9.1) | 12.3(8.2) | 18.0 (11.1) | 21.5 (18.2) | 0.1230 |
| HOMA-IR | 4.0 (3.4) | 3.2 (2.4) *** | 5.4 (5.7) | 6.3 (5.9) * | 0.0627 |
| Triglycerides, mg/dl | 121.0 (69.1) | 96.0 (61.1) * | 134.9 (75.3) | 132.3 (83.7) | 0.0875 |
| HDL, mg/dl | 66.1 (16.6) | 65.3 (16.2) | 58.1(16.2) | 58.5 (12.5) | 0.9360 |
| LDL, mg/dl | 113.0 (26.6) | 113.5 (30.5) | 101.3 (36.3) | 104.4 (30.1) | 0.3278 |
| Total cholesterol, mg/dl | 203.3 (31.1) | 198.0 (35.8) | 186.4 (35.8) | 184.3 (30.8) | 0.6262 |
| LCHF Diet (n = 32) | LF Diet (n=18) | Within Diet Contrasts | Between Diet Contrasts | |||||
|---|---|---|---|---|---|---|---|---|
| LCHF Diet | LF Diet | IR | IS | |||||
| Insulin Resistant (n = 17) | Insulin Sensitive (n = 15) | Insulin Resistant (n = 11) | Insulin Sensitive (n = 7) | IR vs. IS | IR vs IS | LCHF vs. LF | LCHF vs. LF | |
| Demographics | p-Value | p-Value | p-Value | p-Value | ||||
| Weight, kg | −6.3 (5.3) | −6.0 (5.2) | −2.5 (4.6) | −4.2 (4.4) | 0.7730 | 0.4432 | 0.0761 | 0.3207 |
| BMI, kg/m2 | −2.2 (1.9) | −2.2 (2.0) | −0.9 (1.8) | −1.4 (1.4) | 0.7479 | 0.5400 | 0.0896 | 0.2694 |
| Fat, % | −2.7 (3.0) | −2.8 (2.7) | −1.4 (1.1) | −1.6 (1.9) | 0.9672 | 0.8715 | 0.1682 | 0.2785 |
| REE, kcal/day | −46.2 (223.4) | 3.9 (134.3) | −92.4 (14.6) | −38.2 (140) | 0.7855 | 0.5209 | 0.2949 | 0.6271 |
| Serum Analytes | ||||||||
| Glucose, mg/dL | −17.3 (45.3) | 3.9 (7.6) | 13.6 (39.5) | 1.6 (12.3) | 0.1702 | 0.3926 | 0.1093 | 0.7917 |
| Insulin, µU/mL | −6.4 (8.8) | 2.5 (7.5) | 2.5 (19.9) | 5 (10.9) | 0.1016 | 0.7273 | 0.0827 | 0.7624 |
| HOMA−IR | −2.4 (3.8) | 0.7 (1.9) | 0.8 (6.1) | 1.2 (2.5) | 0.8911 | 0.8608 | 0.0267 * | 0.0895 |
| Triglycerides, mg/dL | −42.6 (79.2) | −9.6 (27.9) | −5.6 (64.9) | 2.1 (21.8) | 0.2057 | 0.8310 | 0.0614 | 0.7537 |
| HDL, mg/dL | −2.4 (8.6) | 0.6 (10.2) | 3.6 (8.1) | −4.7 (9.4) | 0.0448 * | 0.5996 | 0.3598 | 0.1342 |
| LDL, mg/dL | 1.5 (35.4) | −0.4 (22.2) | −0.3 (10.5) | −3.4 (26.0) | 0.5551 | 0.7674 | 0.9664 | 0.3967 |
| Total cholesterol, mg/dl | −9.5 (34.5) | −1.7 (25.9) | −1.5 (15.3) | −7.7 (21.8) | 0.1132 | 0.5854 | 0.3945 | 0.2539 |
| Region Fat Mass—DXA | ||||||||
| Total Fat, kg | −6.0 (3.4) | −4.8 (3.8) | −2.7 (3.1) | −3.4 (3.5) | 0.7579 | 0.6532 | 0.1341 | 0.3413 |
| Appendicular Fat, kg | −3 (4.0) | −1.8 (1.4) | −0.7 (1.0) | −1.5 (1.8) | 0.1523 | 0.5187 | 0.0298 * | 0.9288 |
| Trunk Fat, kg | −4.1 (2.4) | −2.9 (2.5) | −1.9 (2.7) | −1.8 (1.8) | 0.5034 | 0.9746 | 0.0844 | 0.0844 |
| Android Fat, kg | −0.9 (0.5) | −0.6 (0.5) | −0.4 (0.4) | −0.4 (0.3) | 0.3810 | 0.9198 | 0.0367 * | 0.2895 |
| Gynoid Fat, kg | −1 (0.7) | −0.9 (0.7) | −0.4 (0.7) | −0.8 (0.6) | 0.8396 | 0.2321 | 0.0395 * | 0.6548 |
| Visceral Fat, kg | −0.4 (0.4) | −0.2 (0.2) | 0.0 (0.3) | −0.1 (0.2) | 0.1606 | 0.6264 | 0.0182 * | 0.6133 |
| Region Lean Mass—DXA | ||||||||
| Total lean, kg | −0.3 (2.4) | −0.6 (2.0) | 0.2 (1.6) | −1 (2.5) | 0.7220 | 0.2214 | 0.5537 | 0.2876 |
| Trunk lean, kg | 0.2 (1.3) | −0.3 (1.3) | 0.2 (1.6) | −0.3 (1.3) | 0.6762 | 0.4284 | 0.9850 | 0.6177 |
| Android lean, kg | −0.5 (1.2) | −0.3 (1.0) | 0 (1.1) | −0.6 (1.3) | 0.2428 | 0.2961 | 0.2210 | 0.3196 |
| Gynoid lean, kg | −0.2 (0.3) | −0.1 (0.2) | 0 (0.1) | −0.1 (0.3) | 0.2888 | 0.4527 | 0.0558 | 0.9607 |
| Appendicular lean, kg | −0.2 (0.4) | −0.2 (0.3) | −0.1 (0.4) | −0.1 (0.3) | 0.7326 | 0.7984 | 0.3111 | 0.7242 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garr Barry, V.; Stewart, M.; Soleymani, T.; Desmond, R.A.; Goss, A.M.; Gower, B.A. Greater Loss of Central Adiposity from Low-Carbohydrate versus Low-Fat Diet in Middle-Aged Adults with Overweight and Obesity. Nutrients 2021, 13, 475. https://doi.org/10.3390/nu13020475
Garr Barry V, Stewart M, Soleymani T, Desmond RA, Goss AM, Gower BA. Greater Loss of Central Adiposity from Low-Carbohydrate versus Low-Fat Diet in Middle-Aged Adults with Overweight and Obesity. Nutrients. 2021; 13(2):475. https://doi.org/10.3390/nu13020475
Chicago/Turabian StyleGarr Barry, Valene, Mariah Stewart, Taraneh Soleymani, Renee A. Desmond, Amy M. Goss, and Barbara A. Gower. 2021. "Greater Loss of Central Adiposity from Low-Carbohydrate versus Low-Fat Diet in Middle-Aged Adults with Overweight and Obesity" Nutrients 13, no. 2: 475. https://doi.org/10.3390/nu13020475
APA StyleGarr Barry, V., Stewart, M., Soleymani, T., Desmond, R. A., Goss, A. M., & Gower, B. A. (2021). Greater Loss of Central Adiposity from Low-Carbohydrate versus Low-Fat Diet in Middle-Aged Adults with Overweight and Obesity. Nutrients, 13(2), 475. https://doi.org/10.3390/nu13020475






