Breastfeeding Duration and Development of Dysglycemia in Women Who Had Gestational Diabetes Mellitus: Evidence from the GUSTO Cohort Study
Abstract
1. Introduction
2. Materials and Methods
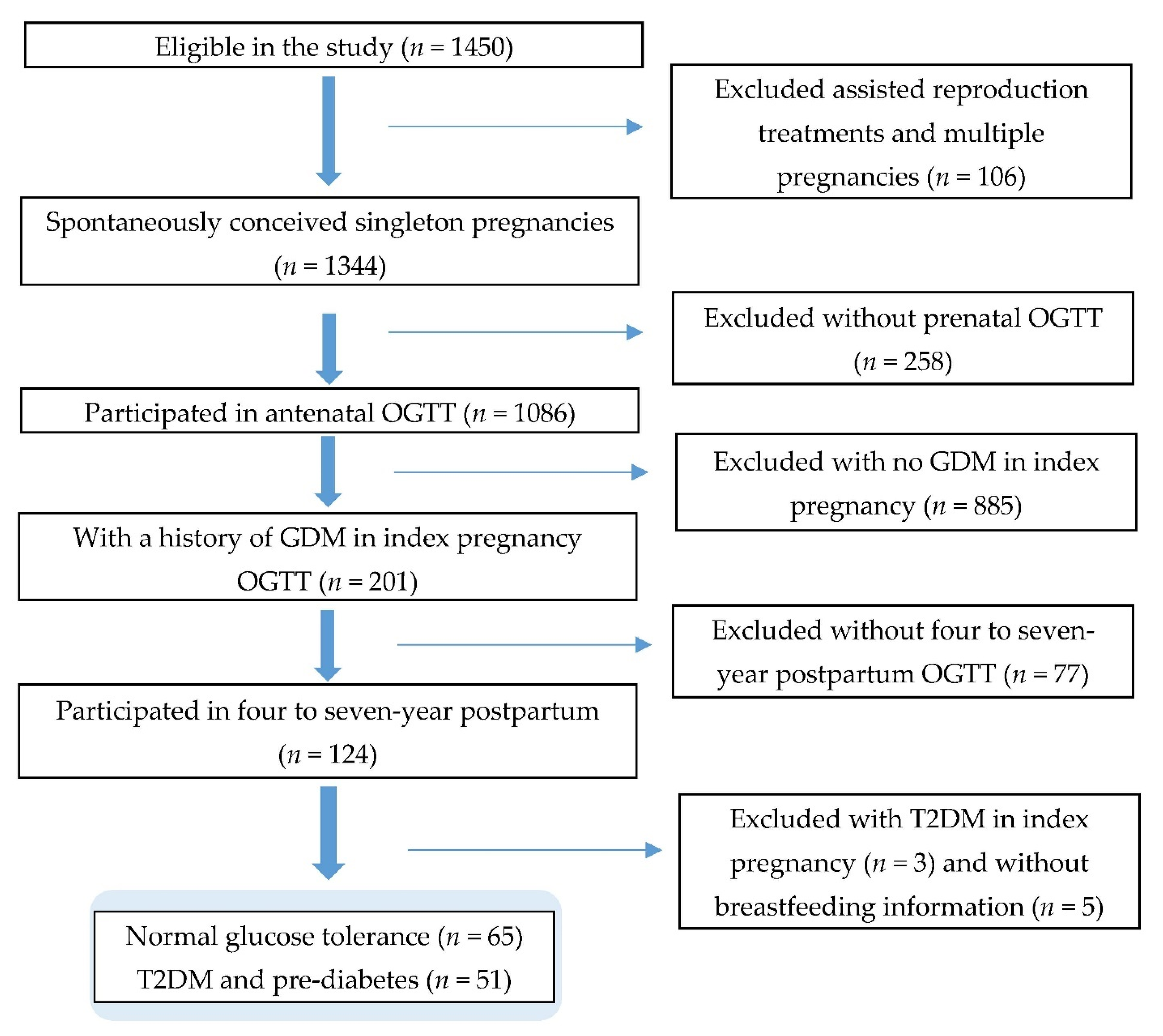
2.1. Study Population
2.2. Data Collection
2.2.1. Ascertainment of T2DM and Prediabetes
2.2.2. Assessment of Breastfeeding Duration
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, C.; Newton, K.M.; Knopp, R.H. Gestational Diabetes and the Incidence of Type 2 Diabetes. A. Syst. Rev. 2002, 25, 1862–1868. [Google Scholar]
- Song, C.; Lyu, Y.; Li, C.; Liu, P.; Li, J.; Ma, R.; Yang, X.J.O.R. Long-term risk of diabetes in women at varying durations after gestational diabetes: A systematic review and meta-analysis with more than 2 million women. Obesity. Rev. 2018, 19, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, T.A.; Xiang, A.; Kjos, S.L.; Watanabe, R. What Is Gestational Diabetes? Diabetes Care 2007, 30 (Suppl. 2), S105–S111. [Google Scholar] [CrossRef]
- Yuen, L.; Wong, V.W. Gestational diabetes mellitus: Challenges for different ethnic groups. World J. Diabetes 2015, 6, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Girgis, C.M.; Gunton, J.E.; Cheung, N.W. The influence of ethnicity on the development of type 2 diabetes mellitus in women with gestational diabetes: A prospective study and review of the literature. ISRN Endocrinol. 2012, 2012, 341638. [Google Scholar] [CrossRef]
- Chong, Y.-S.; Cai, S.; Lin, H.; Soh, S.E.; Lee, Y.-S.; Leow, M.K.-S.; Chan, Y.-H.; Chen, L.; Holbrook, J.D.; Tan, K.-H.; et al. Ethnic differences translate to inadequacy of high-risk screening for gestational diabetes mellitus in an Asian population: A cohort study. BMC Pregnancy Childbirth 2014, 14, 345. [Google Scholar] [CrossRef]
- Casagrande, S.S.; Linder, B.; Cowie, C.C. Prevalence of gestational diabetes and subsequent Type 2 diabetes among U.S. women. Diabetes Res. Clin. Pract. 2018, 141, 200–208. [Google Scholar] [CrossRef]
- International Diabetes Federation IDF DIABETES ATLAS; International Diabetes Federation: Brussels, Belgium, 2019.
- Horta, B.L.; Victora, C.G. Long-Term Effects of Breastfeeding-A Systematic Review; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Eidelman, A.I. Breastfeeding and the use of human milk: An analysis of the American Academy of Pediatrics 2012 Breastfeeding Policy Statement. Breastfeed. Med. 2012, 7, 323–324. [Google Scholar] [CrossRef]
- James, D.; Lessen, R. Position of the American Dietetic Association: Promoting and supporting breastfeeding. J. Am. Diet. Assoc. 2009, 109, 1926–1942. [Google Scholar]
- Lubold, A.M. The effect of family policies and public health initiatives on breastfeeding initiation among 18 high-income countries: A qualitative comparative analysis research design. Int. Breastfeed. J. 2017, 12, 34. [Google Scholar] [CrossRef]
- Gunderson, E.P.; Jacobs, D.R., Jr.; Chiang, V.; Lewis, C.E.; Feng, J.; Quesenberry, C.P., Jr.; Sidney, S. Duration of lactation and incidence of the metabolic syndrome in women of reproductive age according to gestational diabetes mellitus status: A 20-Year prospective study in CARDIA (Coronary Artery Risk Development in Young Adults). Diabetes 2010, 59, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Breastfeeding and the maternal risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Nutr. Metab. Cardiovasc. Dis. NMCD 2014, 24, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and breastfeeding: Collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50302 women with breast cancer and 96973 women without the disease. Lancet 2002, 360, 187–195.
- Gunderson, E.P. Impact of Breastfeeding on Maternal Metabolism: Implications for Women with Gestational Diabetes. Curr. Diabetes Rep. 2014, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gouveri, E.; Papanas, N.; Hatzitolios, A.I.; Maltezos, E. Breastfeeding and Diabetes. Curr. Diabetes Rev. 2011, 7, 135–142. [Google Scholar] [CrossRef]
- Chouinard-Castonguay, S.; Weisnagel, S.J.; Tchernof, A.; Robitaille, J. Relationship between lactation duration and insulin and glucose response among women with prior gestational diabetes. Eur. J. Endocrinol. 2013, 168, 515–523. [Google Scholar] [CrossRef]
- Gunderson, E.P.; Hurston, S.R.; Ning, X.; Lo, J.C.; Crites, Y.; Walton, D.; Dewey, K.G.; Azevedo, R.A.; Young, S.; Fox, G.; et al. Lactation and Progression to Type 2 Diabetes Mellitus After Gestational Diabetes Mellitus: A Prospective Cohort Study. Ann. Intern. Med. 2015, 163, 889–898. [Google Scholar] [CrossRef]
- Kjos, S.L.; Henry, O.; Lee, R.M.; Buchanan, T.A.; Mishell, D.R., Jr. The effect of lactation on glucose and lipid metabolism in women with recent gestational diabetes. Obstet. Gynecol. 1993, 82, 451–455. [Google Scholar]
- Yasuhi, I.; Soda, T.; Yamashita, H.; Urakawa, A.; Izumi, M.; Kugishima, Y.; Umezaki, Y. The effect of high-intensity breastfeeding on postpartum glucose tolerance in women with recent gestational diabetes. Int. Breastfeed. J. 2017, 12, 32. [Google Scholar] [CrossRef]
- Ziegler, A.-G.; Wallner, M.; Kaiser, I.; Rossbauer, M.; Harsunen, M.H.; Lachmann, L.; Maier, J.; Winkler, C.; Hummel, S. Long-term protective effect of lactation on the development of type 2 diabetes in women with recent gestational diabetes mellitus. Diabetes 2012, 61, 3167–3171. [Google Scholar] [CrossRef]
- Hamman, R.F.; Wing, R.R.; Edelstein, S.L.; Lachin, J.M.; Bray, G.A.; Delahanty, L.; Hoskin, M.; Kriska, A.M.; Mayer-Davis, E.J.; Pi-Sunyer, X.J.D. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 2006, 29, 2102–2107. [Google Scholar] [CrossRef]
- Butte, N.F.; Wong, W.W.; Hopkinson, J.M. Energy requirements of lactating women derived from doubly labeled water and milk energy output. J. Nutr. 2001, 131, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Gunderson, E.P.; Kim, C.; Quesenberry, C.P., Jr.; Marcovina, S.; Walton, D.; Azevedo, R.A.; Fox, G.; Elmasian, C.; Young, S.; Salvador, N. Lactation intensity and fasting plasma lipids, lipoproteins, non-esterified free fatty acids, leptin and adiponectin in postpartum women with recent gestational diabetes mellitus: The SWIFT cohort. Metabolism 2014, 63, 941–950. [Google Scholar] [CrossRef] [PubMed]
- McManus, R.M.; Cunningham, I.; Watson, A.; Harker, L.; Finegood, D.T. Beta-cell function and visceral fat in lactating women with a history of gestational diabetes. Metab. Clin. Exp. 2001, 50, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Tigas, S.; Sunehag, A.; Haymond, M.W. Metabolic adaptation to feeding and fasting during lactation in humans. J. Clin. Endocrinol. Metab. 2002, 87, 302–307. [Google Scholar] [CrossRef]
- Baker, J.L.; Gamborg, M.; Heitmann, B.L.; Lissner, L.; Sorensen, T.I.; Rasmussen, K.M. Breastfeeding reduces postpartum weight retention. Am. J. Clin. Nutr. 2008, 88, 1543–1551. [Google Scholar] [CrossRef] [PubMed]
- Dewey, K.G.; Heinig, M.J.; Nommsen, L.A. Maternal weight-loss patterns during prolonged lactation. Am. J. Clin. Nutr. 1993, 58, 162–166. [Google Scholar] [CrossRef]
- Ben-Jonathan, N.; Hugo, E.R.; Brandebourg, T.D.; LaPensee, C.R. Focus on prolactin as a metabolic hormone. Trends Endocrinol. Metab. 2006, 17, 110–116. [Google Scholar] [CrossRef]
- Wang, T.; Lu, J.; Xu, Y.; Li, M.; Sun, J.; Zhang, J.; Xu, B.; Xu, M.; Chen, Y.; Bi, Y.J.D. Circulating prolactin associates with diabetes and impaired glucose regulation: A population-based study. Diabetes Care 2013, 36, 1974–1980. [Google Scholar] [CrossRef]
- Wang, T.; Xu, Y.; Xu, M.; Ning, G.; Lu, J.; Dai, M.; Xu, B.; Sun, J.; Sun, W.; Lai, S.; et al. Circulating Prolactin and Risk of Type 2 Diabetes: A Prospective Study. Am. J. Epidemiol. 2016, 184, 295–301. [Google Scholar] [CrossRef]
- Stuebe, A.M.; Rich-Edwards, J.W. The reset hypothesis: Lactation and maternal metabolism. Am. J. Perinatol. 2009, 26, 081–088. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.S.; Kacmar, J.E.; Nothnagle, M.; Lawrence, R.A. A systematic review of the literature associating breastfeeding with type 2 diabetes and gestational diabetes. J. Am. Coll. Nutr. 2005, 24, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Tanase-Nakao, K.; Arata, N.; Kawasaki, M.; Yasuhi, I.; Sone, H.; Mori, R.; Ota, E. Potential protective effect of lactation against incidence of type 2 diabetes mellitus in women with previous gestational diabetes mellitus: A systematic review and meta-analysis. Diabetes Metab. Res. Rev. 2017, 33, e2875. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Hu, S.; Liang, H.; Xiao, Y.; Tan, H. Metabolic effects of breastfeed in women with prior gestational diabetes mellitus: A systematic review and meta-analysis. Diabetes Metab Res. Rev. 2019, 35, e3108. [Google Scholar] [CrossRef] [PubMed]
- Soh, S.-E.; Tint, M.T.; Gluckman, P.D.; Godfrey, K.M.; Rifkin-Graboi, A.; Chan, Y.H.; Stünkel, W.; Holbrook, J.D.; Kwek, K.; Chong, Y.-S.; et al. Cohort Profile: Growing Up in Singapore Towards healthy Outcomes (GUSTO) birth cohort study. Int. J. Epidemiol. 2014, 43, 1401–1409. [Google Scholar] [CrossRef]
- World Health Organization. Diagnostic Criteria and Classification of Hyperglycaemia First Detected in Pregnancy; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- World Health Organization. Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycemia: Report of a WHO/IDF Consultation; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Pang, W.W.; Aris, I.M.; Fok, D.; Soh, S.E.; Chua, M.C.; Lim, S.B.; Saw, S.M.; Kwek, K.; Gluckman, P.D.; Godfrey, K.M.; et al. Determinants of Breastfeeding Practices and Success in a Multi-Ethnic Asian Population. Birth 2016, 43, 68–77. [Google Scholar] [CrossRef]
- Schwarz, E.B.; Brown, J.S.; Creasman, J.M.; Stuebe, A.; McClure, C.K.; Van Den Eeden, S.K.; Thom, D. Lactation and maternal risk of type 2 diabetes: A population-based study. Am. J. Med. 2010, 123, 863.e1–863.e6. [Google Scholar] [CrossRef]
- World Health Organization. Indicators for Assessing Breast-Feeding Practices: Report of an Informal Meeting, 11–12 June 1991; World Health Organization: Geneva, Switzerland, 1991. [Google Scholar]
- Stuebe, A.M.; Rich-Edwards, J.W.; Willett, W.C.; Manson, J.E.; Michels, K.B. Duration of lactation and incidence of type 2 diabetes. JAMA 2005, 294, 2601–2610. [Google Scholar] [CrossRef]
- Villegas, R.; Gao, Y.T.; Yang, G.; Li, H.L.; Elasy, T.; Zheng, W.; Shu, X.O. Duration of breast-feeding and the incidence of type 2 diabetes mellitus in the Shanghai Women’s Health Study. Diabetologia 2008, 51, 258–266. [Google Scholar] [CrossRef]
- Department of Statistics Household Income. Available online: https://www.singstat.gov.sg/find-data/search-by-theme/households/household-income/latest-data (accessed on 22 May 2019).
- Han, C.Y.; Colega, M.; Quah, E.P.L.; Chan, Y.H.; Godfrey, K.M.; Kwek, K.; Saw, S.-M.; Gluckman, P.D.; Chong, Y.-S.; Chong, M.F.-F.; et al. A healthy eating index to measure diet quality in pregnant women in Singapore: A cross-sectional study. BMC Nutr. 2015, 1, 39. [Google Scholar] [CrossRef]
- IPAQ Research Committee. Guidelines for Data Processing and Analysis of the International Physical Activity Questionnaire (IPAQ)-Short and Long Forms. Available online: http://www.ipaq.ki.se/ (accessed on 27 January 2021).
- Padmapriya, N.; Bernard, J.Y.; Liang, S.; Loy, S.L.; Cai, S.; Zhe, I.S.; Kwek, K.; Godfrey, K.M.; Gluckman, P.D.; Saw, S.M.; et al. Associations of physical activity and sedentary behavior during pregnancy with gestational diabetes mellitus among Asian women in Singapore. BMC Pregnancy Childbirth 2017, 17, 364. [Google Scholar] [CrossRef] [PubMed]
- Zou, G. A Modified Poisson Regression Approach to Prospective Studies with Binary Data. Am. J. Epidemiol. 2004, 159, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Baptiste-Roberts, K.; Barone, B.B.; Gary, T.L.; Golden, S.H.; Wilson, L.M.; Bass, E.B.; Nicholson, W.K. Risk Factors for Type 2 Diabetes Among Women with Gestational Diabetes: A Systematic Review. Am. J. Med. 2009, 122, 207–214.e4. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, W.K.; Asao, K.; Brancati, F.; Coresh, J.; Pankow, J.S.; Powe, N.R. Parity and Risk of Type 2 Diabetes. Atheroscler. Risk Communities Study 2006, 29, 2349–2354. [Google Scholar]
- InterAct Consortium; Scott, R.A.; Langenberg, C.; Sharp, S.J.; Franks, P.W.; Rolandsson, O.; Drogan, D.; van der Schouw, Y.T.; Ekelund, U.; Kerrison, N.D.; et al. The link between family history and risk of type 2 diabetes is not explained by anthropometric, lifestyle or genetic risk factors: The EPIC-InterAct study. Diabetologia 2013, 56, 60–69. [Google Scholar] [PubMed]
- Pallardo, F.; Herranz, L.; Garcia-Ingelmo, T.; Grande, C.; Martin-Vaquero, P.; Jañez, M.; Gonzalez, A. Early postpartum metabolic assessment in women with prior gestational diabetes. Diabetes Care 1999, 22, 1053–1058. [Google Scholar] [CrossRef]
- Spijkerman, A.M.W.; van der A, D.L.; Nilsson, P.M.; Ardanaz, E.; Gavrila, D.; Agudo, A.; Arriola, L.; Balkau, B.; Beulens, J.W.; Boeing, H.; et al. Smoking and Long-Term Risk of Type 2 Diabetes: The EPIC-InterAct Study in European Populations. Diabetes Care 2014, 37, 3164–3171. [Google Scholar]
- Feng, L.; Xu, Q.; Hu, Z.; Pan, H. Lactation and progression to type 2 diabetes in patients with gestational diabetes mellitus: A systematic review and meta-analysis of cohort studies. J. Diabetes Investig. 2018, 9, 1360–1369. [Google Scholar] [CrossRef]
- Much, D.; Beyerlein, A.; Kindt, A.; Krumsik, J.; Rossbauer, M.; Hofelich, A.; Hivner, S.; Herbst, M.; Römisch-Margl, W.; Prehn, C.; et al. Lactation is associated with altered metabolomic signatures in women with gestational diabetes. Diabetologia 2016, 59, S187. [Google Scholar] [CrossRef]
- Buchanan, T.A.; Xiang, A.; Kjos, S.L.; Lee, W.P.; Trigo, E.; Nader, I.; Bergner, E.A.; Palmer, J.P.; Peters, R.K. Gestational diabetes: Antepartum characteristics that predict postpartum glucose intolerance and type 2 diabetes in Latino women. Diabetes 1998, 47, 1302–1310. [Google Scholar] [CrossRef]
- Saucedo, R.; Basurto, L.; Galván, R.; Sánchez, J.; Puello, E.; Zárate, A. Duration of lactation is associated with lower leptin levels in patients with gestational diabetes mellitus. Salud Cienc. 2014, 20, 581–585. [Google Scholar]
- Benhalima, K.; Jegers, K.; Devlieger, R.; Verhaeghe, J.; Mathieu, C. Glucose Intolerance after a Recent History of Gestational Diabetes Based on the 2013 WHO Criteria. PLoS ONE 2016, 11, e0157272. [Google Scholar] [CrossRef] [PubMed]
- Mattei, L.; Colatrella, A.; Bitterman, O.; Bianchi, P.; Giuliani, C.; Roma, G.; Festa, C.; Merola, G.; Toscano, V.; Napoli, A. Long lasting effects of breastfeeding on metabolism in women with prior gestational diabetes. J. Diabetes Mellit. 2014, 4, 257. [Google Scholar] [CrossRef]
- Bentley-Lewis, R.; Xiong, G.; Lee, H.; Yang, A.; Huynh, J.; Kim, C. Metabolomic analysis reveals amino-acid responses to an oral glucose tolerance test in women with prior history of gestational diabetes mellitus. J. Clin. Transl. Endocrinol. 2014, 1, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kim, M.Y.; Yang, J.H.; Park, S.Y.; Yim, C.H.; Han, K.O.; Yoon, H.K.; Park, S. Nutritional risk factors of early development of postpartum prediabetes and diabetes in women with gestational diabetes mellitus. Nutrition 2011, 27, 782–788. [Google Scholar] [CrossRef]
- Page, K.A.; Buchanan, T.A. The vicious cycle of maternal diabetes and obesity: Moving from “what” to “how” and “why”. J. Pediatrics 2011, 158, 872. [Google Scholar] [CrossRef]
- Ministry of Health Resources and Statistics, Disease Burden. Available online: https://www.moh.gov.sg/resources-statistics/singapore-health-facts/disease-burden (accessed on 11 May 2020).
| Characteristics | All (n = 116) | NGT (n = 65) | Dysglycemia (n = 51) | p-Value * |
|---|---|---|---|---|
| Glycemic activity * | ||||
| Antenatal fasting glucose, mmol/L, (median, IQR) | 4.5 (4.2, 4.9) | 4.4 (4.1, 4.8) | 4.5 (4.2, 4.7) | 0.26 |
| Antenatal 2-h glucose, mmol/L, (median, IQR) | 8.4 (8.1, 9.1) | 8.3 (8.1, 9.1) | 8.4 (8.1, 9.1) | 0.84 |
| Postpartum fasting glucose, mmol/L, (median, IQR) | 4.9 (4.7, 5.3) | 4.8 (4.6, 5.0) | 5.2 (4.9, 5.4) | <0.05 |
| Postpartum 2-h glucose, mmol/L, (median, IQR) | 7.4 (6.4, 9.1) | 6.5 (5.9, 6.9) | 9.2 (8.4, 10.7) | <0.05 |
| Socio-demographics | ||||
| Maternal age at delivery, years, (mean, SD) | 33.3 (±4.8) | 32.8 (±4.5) | 33.9 (±5.2) | 0.25 |
| Ethnicity, n (%) | ||||
| Chinese | 73 (62.9) | 44 (67.7) | 29 (56.9) | 0.25 |
| Malay | 16 (13.8) | 6 (9.2) | 10 (19.6) | |
| Indian | 27 (23.3) | 15 (23.1) | 12 (23.5) | |
| Education level, n (%) | ||||
| Secondary or below | 24 (20.7) | 11 (16.9) | 13 (25.5) | 0.33 |
| Diploma/Technical education | 43 (37.1) | 23 (35.4) | 20 (39.2) | |
| University or higher | 49 (42.2) | 31 (47.7) | 18 (35.3) | |
| Household monthly income at recruitment, n (%) | ||||
| ≤S$6000 | 73 (67.0) | 39 (62.9) | 34 (72.3) | 0.30 |
| >S$6000 | 36 (33.0) | 23 (37.1) | 13 (27.7) | |
| Parity at recruitment, n (%) | ||||
| 0 | 45 (38.8) | 27 (41.5) | 18 (35.3) | 0.06 |
| 1 | 50 (43.1) | 31 (47.7) | 19 (37.2) | |
| ≥2 | 21 (18.1) | 7 (10.8) | 14 (27.5) | |
| Health | ||||
| Family history of diabetes at recruitment, n (%) | ||||
| No | 101 (87.1) | 61 (93.8) | 40 (78.4) | 0.01 |
| Yes | 15 (12.9) | 4 (6.2) | 11 (21.6) | |
| Treatment of GDM, n (%) | 0.14 | |||
| Diet only | 102 (88.0) | 59 (90.8) | 43 (84.3) | |
| Insulin | 7 (6.0) | 2 (3.1) | 5 (9.8) | |
| Unknown or none | 7 (6.0) | 4 (6.1) | 3 (5.9) | |
| GDM in a previous pregnancy, n (%) | 0.37 | |||
| Nulliparous | 45 (38.8) | 27 (41.5) | 18 (35.3) | |
| No or missing | 60 (51.7) | 34 (52.3) | 26 (51.0) | |
| Yes | 11 (9.5) | 4 (6.2) | 8 (13.7) | |
| Anthropometric measurements | ||||
| Pre-pregnancy BMI, kg/m2 (mean, SD) | 23.7 (±4.2) | 22.7 (±3.8) | 25.0 (±4.3) | <0.05 |
| Postpartum BMI, kg/m2 at 18-month visit, n (%) | <0.01 | |||
| <23 | 40 (42.6) | 27 (51.9) | 13 (31.0) | |
| 23 to <27.5 | 34 (36.2) | 20 (38.5) | 14 (33.3) | |
| ≥27.5 | 20 (21.3) | 5 (9.6) | 15 (35.7) | |
| Postpartum waist circumference at 48-month visit, n (%) | 0.01 | |||
| <80 cm | 31 (32.0) | 23 (42.6) | 8 (18.6) | |
| ≥80 cm | 66 (68.0) | 31 (57.4) | 35 (81.4) | |
| Lifestyle | ||||
| Healthy eating index for Singapore pregnant women (HEI-SGP), (mean, SD) | 57.3 (±13.7) | 58.3 (±12.4) | 56.0 (±15.3) | 0.37 |
| Physical activity before index pregnancy, n (%) | 0.62 | |||
| <600 MET/week | 11 (12.1) | 6 (11.5) | 5 (12.8) | |
| 600–3000 MET/week | 61 (67.0) | 37 (71.2) | 24 (61.5) | |
| >3000 MET/week | 19 (20.9) | 9 (17.3) | 10 (25.7) | |
| Perinatal tobacco smoking exposure, n (%) | 0.49 | |||
| Exposed | 38 (32.8) | 23 (35.4) | 15 (29.4) | |
| Not exposed | 78 (67.2) | 42 (64.6) | 36 (70.6) | |
| Breastfeeding behaviors | ||||
| Any breastfeeding, n (%) | 0.03 | |||
| No or <1 month | 21 (18.1) | 9 (13.9) | 12 (23.5) | |
| ≥1 to <6 months | 50 (43.1) | 24 (36.9) | 26 (51.0) | |
| ≥6 months | 45 (38.8) | 32 (49.2) | 13 (25.5) |
| Variables | Unadjusted IRR (95% CI) | p-Value | Age-Adjusted IRR (95% CI) | p-Value | Covariate-Adjusted IRR * (95% CI) | p-Value |
|---|---|---|---|---|---|---|
| Any breastfeeding | ||||||
| No or <1 month | Reference | Reference | Reference | |||
| ≥1 to <6 months | 0.91 (0.57, 1.43) | 0.687 | 0.90 (0.57, 1.44) | 0.682 | 0.67 (0.41, 1.10) | 0.117 |
| ≥6 months | 0.50 (0.27, 0.91) | 0.024 | 0.51 (0.28, 0.93) | 0.030 | 0.42 (0.22, 0.79) | 0.008 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hewage, S.S.; Koh, X.Y.H.; Soh, S.E.; Pang, W.W.; Fok, D.; Cai, S.; Müller-Riemenschneider, F.; Yap, F.; Tan, K.H.; Chua, M.C.; et al. Breastfeeding Duration and Development of Dysglycemia in Women Who Had Gestational Diabetes Mellitus: Evidence from the GUSTO Cohort Study. Nutrients 2021, 13, 408. https://doi.org/10.3390/nu13020408
Hewage SS, Koh XYH, Soh SE, Pang WW, Fok D, Cai S, Müller-Riemenschneider F, Yap F, Tan KH, Chua MC, et al. Breastfeeding Duration and Development of Dysglycemia in Women Who Had Gestational Diabetes Mellitus: Evidence from the GUSTO Cohort Study. Nutrients. 2021; 13(2):408. https://doi.org/10.3390/nu13020408
Chicago/Turabian StyleHewage, Sumali S., Xin Yu Hazel Koh, Shu E. Soh, Wei Wei Pang, Doris Fok, Shirong Cai, Falk Müller-Riemenschneider, Fabian Yap, Kok Hian Tan, Mei Chien Chua, and et al. 2021. "Breastfeeding Duration and Development of Dysglycemia in Women Who Had Gestational Diabetes Mellitus: Evidence from the GUSTO Cohort Study" Nutrients 13, no. 2: 408. https://doi.org/10.3390/nu13020408
APA StyleHewage, S. S., Koh, X. Y. H., Soh, S. E., Pang, W. W., Fok, D., Cai, S., Müller-Riemenschneider, F., Yap, F., Tan, K. H., Chua, M. C., Lim, S. B., Godfrey, K. M., Colega, M. T., Chong, Y.-S., Chan, S.-Y., Yoong, J., & Chong, M. F. F. (2021). Breastfeeding Duration and Development of Dysglycemia in Women Who Had Gestational Diabetes Mellitus: Evidence from the GUSTO Cohort Study. Nutrients, 13(2), 408. https://doi.org/10.3390/nu13020408







