An In Vitro Approach to Studying the Microbial Community and Impact of Pre and Probiotics under Anorexia Nervosa Related Dietary Restrictions
Abstract
:1. Introduction
2. Materials and Methods
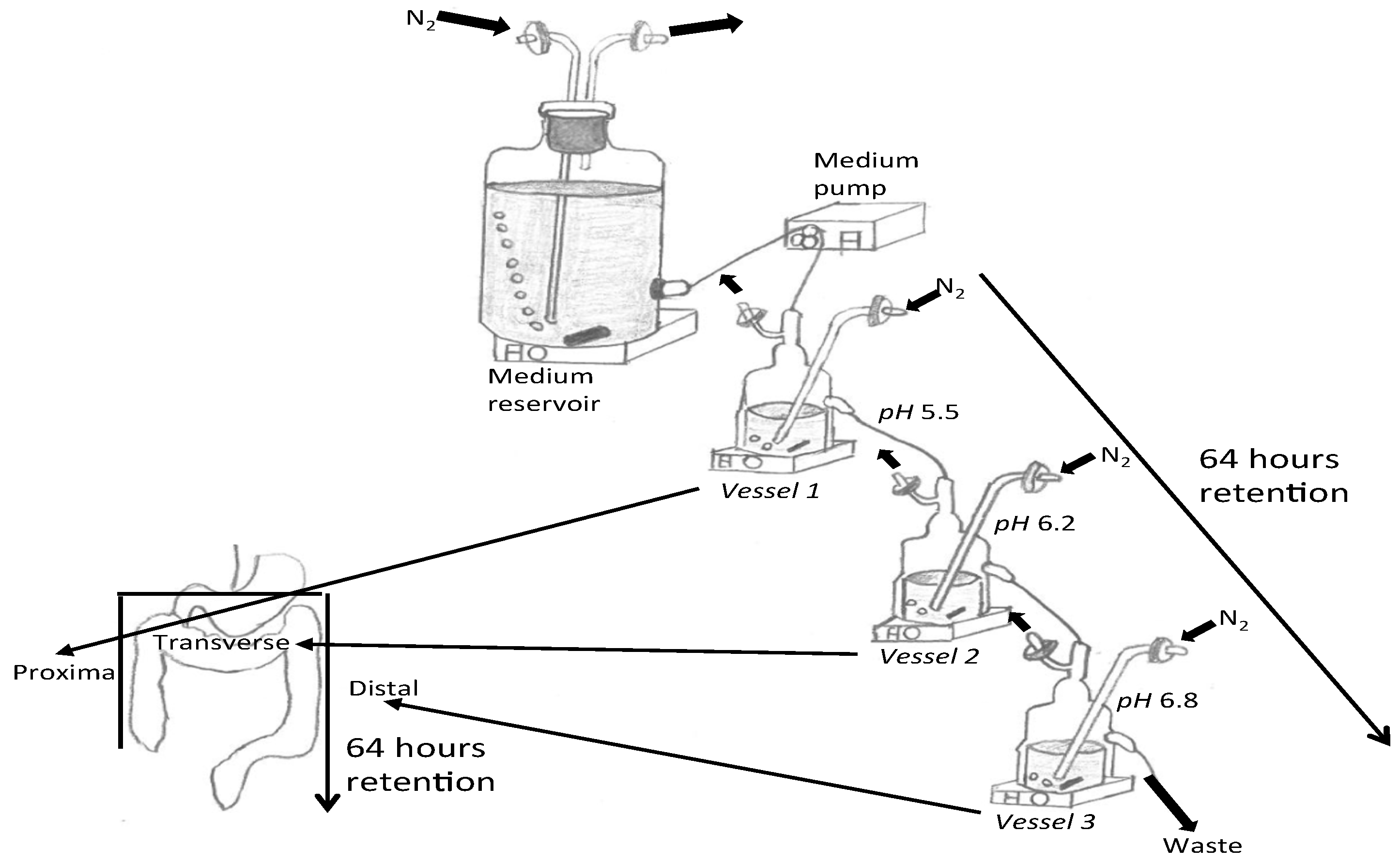
2.1. Three-Stage Compound Continuous Culture System
2.2. Gut Model Medium Determination in HC, AN, and AN with Pre and Probiotic Feeding
2.3. Treatments
2.4. Preparation of the Samples for SCFA/BCFA Analysis, Neurotransmitter Analysis and Bacterial Community Analysis
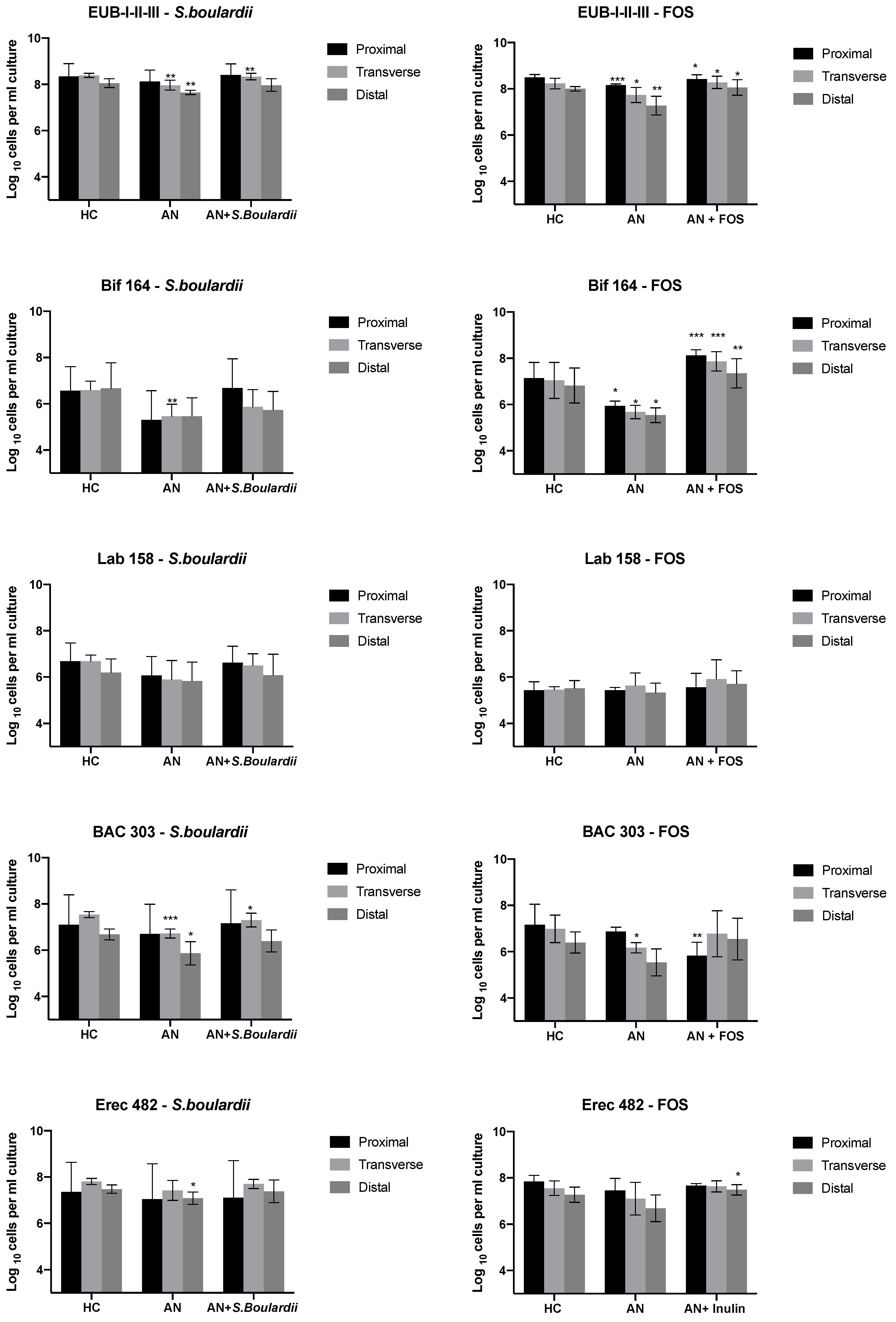
2.5. In Vitro Enumeration of Bacterial Population by Flow-Fluorescent In Situ Hybridisation (FISH)
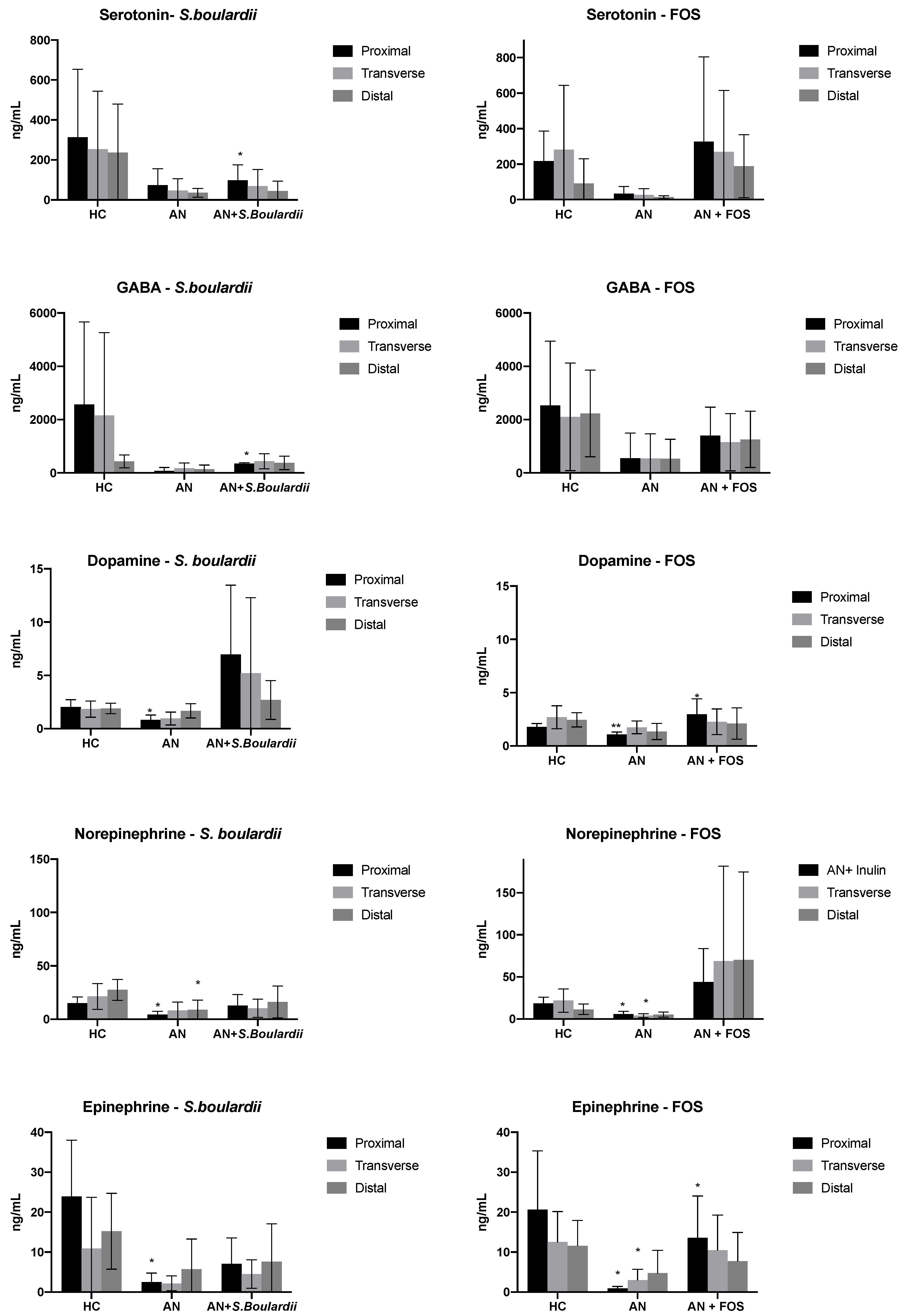
2.6. Neurotransmitter Analysis by Liquid Chromatography Mass Spectrometry (LCMS)
2.6.1. Reagents and Chemicals
2.6.2. Stock Solutions, Calibration Standards and Sample Preparation
2.6.3. LCMS System
2.6.4. Liquid Chromatography (LC) Conditions
2.6.5. Mass Spectrometry (MS) Conditions
2.6.6. Quantification of Samples
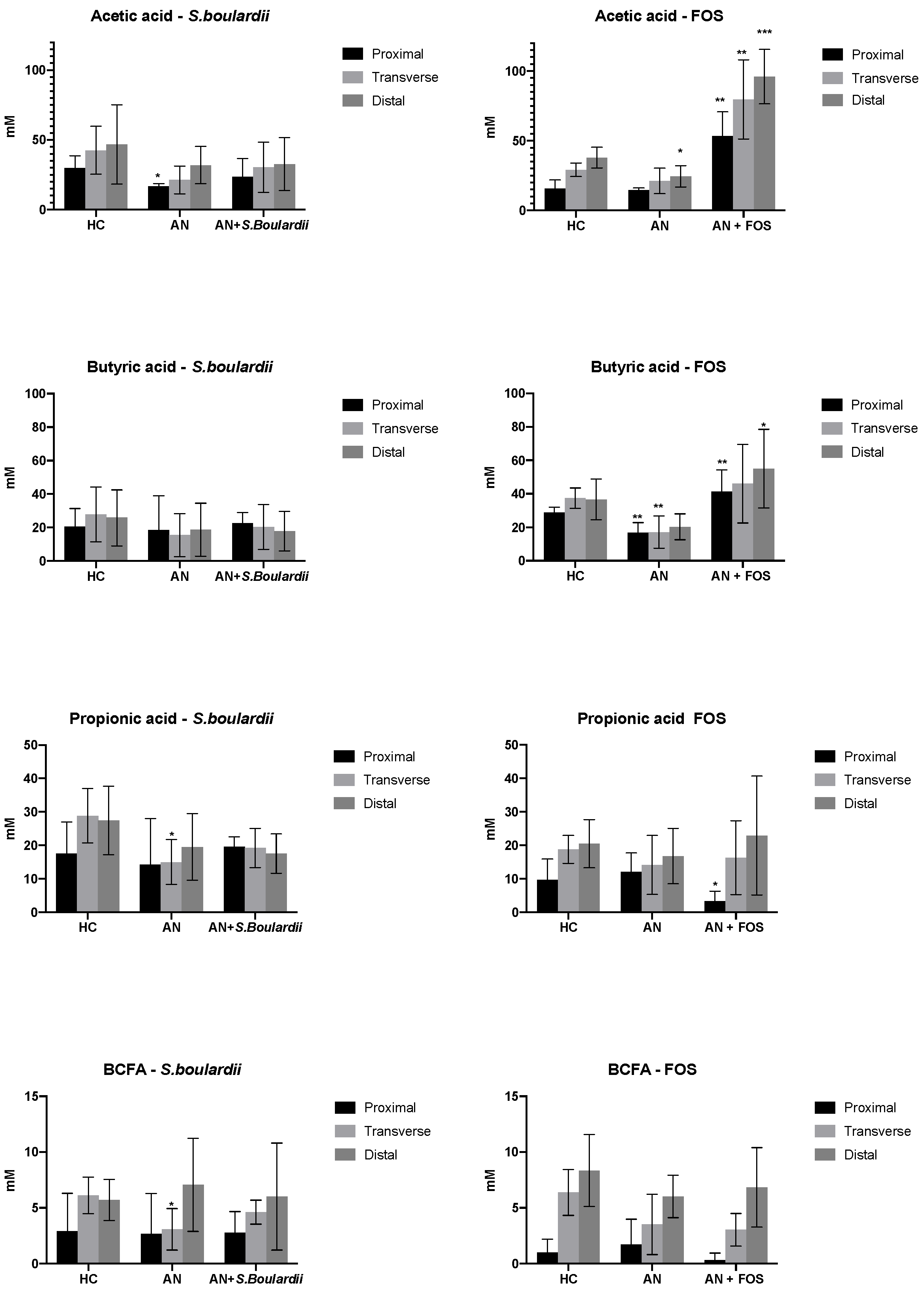
2.7. Short Chain and Branched Chain Fatty Acid Analysis by Gas Chromatography
2.8. Statistical Analysis
3. Results
3.1. Bacterial Enumeration
3.2. Neurotransmitter Production
3.3. SCFA and BCFA Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Association, A.P. Desk Reference to the Diagnostic Criteria from Dsm-5 (R); American Psychiatric Publishing: Washington, DC, USA, 2013. [Google Scholar]
- Borgo, F.; Riva, A.; Benetti, A.; Casiraghi, M.C.; Bertelli, S.; Garbossa, S.; Anselmetti, S.; Scarone, S.; Pontiroli, A.E.; Morace, G.; et al. Microbiota in anorexia nervosa: The triangle between bacterial species, metabolites and psychological tests. PLoS ONE 2017, 12, e0179739. [Google Scholar] [CrossRef] [PubMed]
- Kleiman, S.C.; Watson, H.J.; Bulik-Sullivan, E.C.; Huh, E.Y.; Tarantino, L.M.; Bulik, C.M.; Carroll, I.M. The Intestinal Microbiota in Acute Anorexia Nervosa and During Renourishment: Relationship to Depression, Anxiety, and Eating Disorder Psychopathology. Psychosom. Med. 2015, 77, 969–981. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morita, C.; Tsuji, H.; Hata, T.; Gondo, M.; Takakura, S.; Kawai, K.; Yoshihara, K.; Ogata, K.; Nomoto, K.; Miyazaki, K.; et al. Gut Dysbiosis in Patients with Anorexia Nervosa. PLoS ONE 2015, 10, e0145274. [Google Scholar] [CrossRef] [Green Version]
- Mack, I.; Cuntz, U.; Grämer, C.; Niedermaier, S.; Pohl, C.; Schwiertz, A.; Zimmermann, K.; Zipfel, S.; Enck, P.; Penders, J. Weight gain in anorexia nervosa does not ameliorate the faecal microbiota, branched chain fatty acid profiles and gastrointestinal complaints. Sci. Rep. 2016, 6, 26752. [Google Scholar] [CrossRef] [Green Version]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S.; et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017, 17, 120. [Google Scholar] [CrossRef] [Green Version]
- Sweeney, T.E.; Morton, J.M. The Human Gut Microbiome: A Review of the Effect of Obesity and Surgically Induced Weight Loss. JAMA Surg 2013, 148, 563–569. [Google Scholar] [CrossRef] [Green Version]
- Armougom, F.; Henry, M.; Vialettes, B.; Raccah, D.; Raoult, D. Monitoring Bacterial Community of Human Gut Microbiota Reveals an Increase in Lactobacillus in Obese Patients and Methanogens in Anorexic Patients. PLoS ONE 2009, 4, e7125. [Google Scholar] [CrossRef]
- Million, M.; Angelakis, E.; Maraninchi, M.; Henry, M.; Giorgi, R.; Valero, R.; Vialettes, B.; Raoult, D. Correlation between body mass index and gut concentrations of Lactobacillus reuteri, Bifidobacterium animalis, Methanobrevibacter smithii and Escherichia coli. Int. J. Obes. 2013, 37, 1460–1466. [Google Scholar] [CrossRef] [Green Version]
- Jalanka, J.; Salonen, A.; Salojärvi, J.; Ritari, J.; Immonen, O.; Marciani, L.; Gowland, P.; Hoad, C.; Garsed, K.; Lam, C.; et al. Effects of bowel cleansing on the intestinal microbiota. Gut 2014, 64, 1562–1568. [Google Scholar] [CrossRef]
- Shehata, T.E.; Marr, A.G. Effect of Nutrient Concentration on the Growth of Escherichia Coli. J. Bacteriol. 1971, 107, 210–216. [Google Scholar] [CrossRef] [Green Version]
- Herpertz-Dahlmann, B.; Seitz, J.; Baines, J. Food matters: How the microbiome and gut–brain interaction might impact the development and course of anorexia nervosa. Eur. Child. Adolesc. Psychiatry 2017, 26, 1031–1041. [Google Scholar] [CrossRef] [Green Version]
- Mack, I.; Penders, J.; Cook, J.; Dugmore, J.; Mazurak, N.; Enck, P. Is the Impact of Starvation on the Gut Microbiota Specific or Unspecific to Anorexia Nervosa? A Narrative Review Based on a Systematic Literature Search. Curr. Neuropharmacol. 2018, 16, 1131–1149. [Google Scholar] [CrossRef]
- Neuman, H.; Debelius, J.; Knight, R.; Koren, O. Microbial endocrinology: The interplay between the microbiota and the endocrine system. FEMS Microbiol. Rev. 2015, 39, 509–521. [Google Scholar] [CrossRef] [Green Version]
- Seitz, J.; Belheouane, M.; Schulz, N.; Dempfle, A.; Baines, J.F.; Herpertz-Dahlmann, B. The Impact of Starvation on the Microbiome and Gut-Brain Interaction in Anorexia Nervosa. Front. Endocrinol. 2019, 10, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seitz, J.; Trinh, S.; Herpertz-Dahlmann, B. The Microbiome and Eating Disorders. Psychiatr. Clin. North. Am. 2019, 42, 93. [Google Scholar] [CrossRef]
- Roubalová, R.; Procházková, P.; Papežová, H.; Smitka, K.; Bilej, M.; Tlaskalová-Hogenová, H. Anorexia nervosa: Gut microbiota-immune-brain interactions. Clin. Nutr. 2019, 39, 676–684. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, J.; Su, W.; Rahat-Rozenbloom, S.; Wolever, T.M.S.; Comelli, E.M. Adiposity, gut microbiota and faecal short chain fatty acids are linked in adult humans. Nutr. Diabetes 2014, 4, e121. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef]
- Braniste, V.; Al-Asmakh, M.; Kowal, C.; Anuar, F.; Abbaspour, A.; Tóth, M.; Korecka, A.; Bakocevic, N.; Ng, L.G.; Kundu, P.; et al. The gut microbiota influences blood-brain barrier permeability in mice. Sci. Transl. Med. 2014, 6, 263ra158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stilling, R.M.; Dinan, T.G.; Cryan, J.F. Microbial genes, brain & behaviour—Epigenetic regulation of the gut-brain axis. Genes Brain Behav. 2014, 13, 69–86. [Google Scholar] [CrossRef]
- Sherwin, E.; Rea, K.; Dinan, T.; Cryan, J.F. A gut (microbiome) feeling about the brain. Curr. Opin. Gastroenterol. 2016, 32, 96–102. [Google Scholar] [CrossRef]
- Lyte, M. Probiotics function mechanistically as delivery vehicles for neuroactive compounds: Microbial endocrinology in the design and use of probiotics. BioEssays 2011, 33, 574–581. [Google Scholar] [CrossRef]
- Gershon, M.D.; Tack, J. The Serotonin Signaling System: From Basic Understanding To Drug Development for Functional GI Disorders. Gastroenterology 2007, 132, 397–414. [Google Scholar] [CrossRef]
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell 2015, 163, 258. [Google Scholar] [CrossRef] [Green Version]
- Kaye, W.H.; Bulik, C.M.; Thornton, L.; Barbarich, N.; Masters, K.; Group PFC. Comorbidity of Anxiety Disorders With Anorexia and Bulimia Nervosa. Am. J. Psychiatry 2004, 161, 2215–2221. [Google Scholar] [CrossRef]
- Weltzin, T.; Fernstrom, M.H.; Hansen, D.; McConaha, C.; Kaye, W.H. Abnormal caloric requirements for weight maintenance in patients with anorexia and bulimia nervosa. Am. J. Psychiatry 1991, 148, 1675–1682. [Google Scholar] [CrossRef]
- Kaye, W.H.; Gwirtsman, H.E.; George, D.T.; Ebert, M.H. Altered Serotonin Activity in Anorexia-Nervosa after Long-Term Weight Restoration—Does Elevated Cerebrospinal-Fluid 5-Hydroxyindoleacetic Acid Level Correlate with Rigid and Obsessive Behavior. Arch. Gen. Psychiatry 1991, 48, 556–562. [Google Scholar] [CrossRef]
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the Manipulation of Bacteria-Gut-Brain Signals. Trends Neurosci. 2016, 39, 763–781. [Google Scholar] [CrossRef] [Green Version]
- Dinan, T.G.; Stanton, C.; Cryan, J.F. Psychobiotics: A Novel Class of Psychotropic. Biol. Psychiatry 2013, 74, 720–726. [Google Scholar] [CrossRef]
- Egrenham, S.; Clarke, G.; Cryan, J.F.; Dinan, T.G. Brain-Gut-Microbe Communication in Health and Disease. Front. Physiol. 2011, 2, 94. [Google Scholar] [CrossRef] [Green Version]
- Mayer, E.A.; Knight, R.; Mazmanian, S.K.; Cryan, J.F.; Tillisch, K. Gut Microbes and the Brain: Paradigm Shift in Neuroscience. J. Neurosci. 2014, 34, 15490–15496. [Google Scholar] [CrossRef] [Green Version]
- Savignac, H.M.; Corona, G.; Mills, H.; Chen, L.; Spencer, J.P.; Tzortzis, G.; Burnet, P.W. Prebiotic feeding elevates central brain derived neurotrophic factor, N-methyl-d-aspartate receptor subunits and d-serine. Neurochem. Int. 2013, 63, 756–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macfarlane, G.; Macfarlane, S.; Gibson, G. Validation of a Three-Stage Compound Continuous Culture System for Investigating the Effect of Retention Time on the Ecology and Metabolism of Bacteria in the Human Colon. Microb. Ecol. 1998, 35, 180–187. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Gibson, G.R. Metabolic Activities of the Normal Colonic Flora. In Human Health—The Contribution of Microorganisms; Gibson, S.A.W., Ed.; Springer: London, UK, 1994; pp. 17–52. [Google Scholar]
- Macfarlane, G.T.; Gibson, G.R. Microbiological Aspects of Short Chain Fatty Acid Production in the Large Bowel. In Physiological and Clinical Aspects of Short Chain Fatty Acid Metabolism; Cummings, J.H., Rombeau, J.L., Sakata, T., Eds.; Cambridge University Press: Cambridge, UK, 1995; pp. 87–105. [Google Scholar]
- Kamal, N.; Chami, T.; Andersen, A.; Rosell, F.A.; Schuster, M.M.; Whitehead, W.E. Delayed gastrointestinal transit times in anorexia nervosa and bulimia nervosa. Gastroenterology 1991, 101, 1320–1324. [Google Scholar] [CrossRef]
- Jauregui Lobera, I.; Bolanos Rios, P. Choice of diet in patients with anorexia nervosa. Nutr. Hosp. 2009, 24, 682–687. [Google Scholar]
- Raatz, S.K.; Jahns, L.; Johnson, L.K.; Crosby, R.; Mitchell, J.E.; Crow, S.; Peterson, C.; Le Grange, D.; Wonderlich, S.A. Nutritional Adequacy of Dietary Intake in Women with Anorexia Nervosa. Nutrients 2015, 7, 3652–3665. [Google Scholar] [CrossRef] [Green Version]
- British Nutrition Foundation Nutrition Requirements. Available online: https://www.nutrition.org.uk/bnfevents/events/158-healthyliving/resources/907-nutritionrequirements.html (accessed on 26 October 2019).
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association: Arlington, TX, USA, 2013. [Google Scholar]
- Wallner, G.; Amann, R.; Beisker, W. Optimizing Fluorescent in Situ Hybridization with Rrna-Targeted Oligonucleotide Probes for Flow Cytometric Identification of Microorganisms. Cytometry 1993, 14, 136–143. [Google Scholar] [CrossRef]
- Daims, H.; Brühl, A.; Amann, R.; Schleifer, K.-H.; Wagner, M. The Domain-specific Probe EUB338 is Insufficient for the Detection of all Bacteria: Development and Evaluation of a more Comprehensive Probe Set. Syst. Appl. Microbiol. 1999, 22, 434–444. [Google Scholar] [CrossRef]
- Langendijk, P.S.; Schut, F.; Jansen, G.J.; Raangs, G.C.; Kamphuis, G.R.; Wilkinson, M.H.; Welling, G.W. Quantitative Fluorescence in Situ Hybridization of Bifidobacterium Spp. With Genus-Specific 16s Rrna-Targeted Probes and Its Application in Fecal Samples. Appl. Environ. Microbiol. 1995, 61, 3069–3075. [Google Scholar] [CrossRef] [Green Version]
- Hermie, J.; Harmsen, M.; Elfferich, P.; Schut, F.; Welling, G.W. A 16s Rrna-Targeted Probe for Detection of Lactobacilli and Enterococci in Faecal Samples by Fluorescent in Situ Hybridization. Microb. Ecol. Health Dis. 1999, 11, 3–12. [Google Scholar]
- Manz, W.; Amann, R.; Ludwig, W.; Vancanneyt, M.; Schleifer, K.H. Application of a Suite of 16s Rrna-Specific Oligonucleotide Probes Designed to Investigate Bacteria of the Phylum Cytophaga-Flavobacter-Bacteroides in the Natural Environment. Microbiology 1996, 142 Pt 5, 1097–1106. [Google Scholar] [CrossRef] [Green Version]
- Franks, A.H.; Harmsen, H.J.; Raangs, G.C.; Jansen, G.J.; Schut, F.; Welling, G.W. Variations of Bacterial Populations in Human Feces Measured by Fluorescent in Situ Hybridization with Group-Specific 16s Rrna-Targeted Oligonucleotide Probes. Appl. Environ. Microbiol. 1998, 64, 3336–3345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, A.W.; Duncan, S.H.; McWilliam Leitch, E.C.; Child, M.W.; Flint, H.J. pH and Peptide Supply Can Radically Alter Bacterial Populations and Short-Chain Fatty Acid Ratios within Microbial Communities from the Human Colon. Appl. Environ. Microbiol. 2005, 71, 3692–3700. [Google Scholar] [CrossRef] [Green Version]
- Harmsen, H.J.; Wildeboer-Veloo, A.C.; Grijpstra, J.; Knol, J.; Degener, J.E.; Welling, G.W. Development of 16s Rrna-Based Probes for the Coriobacterium Group and the Atopobium Cluster and Their Application for Enumeration of Coriobacteriaceae in Human Feces from Volunteers of Different Age Groups. Appl. Environ. Microbiol. 2000, 66, 4523–4527. [Google Scholar] [CrossRef] [Green Version]
- Hold, G.L.; Schwiertz, A.; Aminov, R.I.; Blaut, M.; Flint, H.J. Oligonucleotide Probes That Detect Quantitatively Significant Groups of Butyrate-Producing Bacteria in Human Feces. Appl. Environ. Microbiol. 2003, 69, 4320–4324. [Google Scholar] [CrossRef] [Green Version]
- Devereux, R.; Kane, M.D.; Winfrey, J.; Stahl, D.A. Genus- and Group-Specific Hybridization Probes for Determinative and Environmental Studies of Sulfate-Reducing Bacteria. Syst. Appl. Microbiol. 1992, 15, 601–609. [Google Scholar] [CrossRef]
- Harmsen, H.J.; Raangs, G.C.; He, T.; Degener, J.E.; Welling, G.W. Extensive Set of 16s Rrna-Based Probes for Detection of Bacteria in Human Feces. Appl. Environ. Microbiol. 2002, 68, 2982–2990. [Google Scholar] [CrossRef] [Green Version]
- Stoffels, M.; Amann, R.; Ludwig, W.; Hekmat, D.; Schleifer, K.H. Bacterial Community Dynamics During Start-up of a Trickle-Bed Bioreactor Degrading Aromatic Compounds. Appl. Environ. Microbiol. 1998, 64, 930–939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Audie, J.P.; Janin, A.; Porchet, N.; Copin, M.C.; Gosselin, B.; Aubert, J.P. Expression of human mucin genes in respiratory, digestive, and reproductive tracts ascertained by in situ hybridization. J. Histochem. Cytochem. 1993, 41, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Scientific Working Group for Forensic Toxicology (Swgtox) Standard Practices for Method Validation in Forensic Toxicology. J. Anal. Toxicol. 2013, 37, 452–474. [CrossRef] [PubMed]
- Richardson, A.; Calder, A.; Stewart, C.; Smith, A. Simultaneous determination of volatile and non-volatile acidic fermentation products of anaerobes by capillary gas chromatography. Lett. Appl. Microbiol. 1989, 9, 5–8. [Google Scholar] [CrossRef]
- Liu, Y.; Gibson, G.R.; Walton, G.E. An In Vitro Approach to Study Effects of Prebiotics and Probiotics on the Faecal Microbiota and Selected Immune Parameters Relevant to the Elderly. PLoS ONE 2016, 11, e0162604. [Google Scholar] [CrossRef]
- Kelly, J.R.; Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G.; Hyland, N.P. Breaking down the barriers: The gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front. Cell. Neurosci. 2015, 9, 392. [Google Scholar] [CrossRef] [Green Version]
- Petra, A.I.; Panagiotidou, S.; Hatziagelaki, E.; Stewart, J.M.; Conti, P.; Theoharides, T.C. Gut-Microbiota-Brain Axis and Its Effect on Neuropsychiatric Disorders With Suspected Immune Dysregulation. Clin. Ther. 2015, 37, 984–995. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, A.; Harty, S.; Lehto, S.; Moeller, A.H.; Dinan, T.G.; Dunbar, R.I.; Cryan, J.F.; Burnet, P. The Microbiome in Psychology and Cognitive Neuroscience. Trends Cogn. Sci. 2018, 22, 611–636. [Google Scholar] [CrossRef]
- Tlaskalova-Hogenova, H.; Stepankova, R.; Kozakova, H.; Hudcovic, T.; Vannucci, L.; Tuckova, L. The role of gut microbiota (commensal bacteria) and the mucosal barrier in the pathogenesis of inflammatory and autoimmune diseases and cancer: Contribution of germ-free and gnotobiotic animal models of human diseases. Cell Mol. Immunol. 2011, 8, 110–120. [Google Scholar] [CrossRef]
- Prochazkova, P.; Roubalova, R.; Dvorak, J.; Tlaskalova-Hogenova, H.; Cermakova, M.; Tomasova, P.; Sediva, B.; Kuzma, M.; Bulant, J.; Bilej, M.; et al. Microbiota, Microbial Metabolites, and Barrier Function in A Patient with Anorexia Nervosa after Fecal Microbiota Transplantation. Microorganisms 2019, 7, 338. [Google Scholar] [CrossRef] [Green Version]
- Corrêa-Oliveira, R.; Fachi, J.L.; Vieira, A.; Sato, F.T.; Vinolo, M.A.R. Regulation of immune cell function by short-chain fatty acids. Clin. Transl. Immunol. 2016, 5, e73. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, D.R.; Garge, N.; Zhang, X.; Sun, W.; O’Connell, T.M.; Bunger, M.K.; Bultman, S.J. The Microbiome and Butyrate Regulate Energy Metabolism and Autophagy in the Mammalian Colon. Cell Metab. 2011, 13, 517–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imhann, F.; Vila, A.V.; Bonder, M.J.; Fu, J.; Gevers, D.; Visschedijk, M.C.; Spekhorst, L.M.; Alberts, R.; Franke, L.; Van Dullemen, H.M.; et al. Interplay of host genetics and gut microbiota underlying the onset and clinical presentation of inflammatory bowel disease. Gut 2016, 67, 108–119. [Google Scholar] [CrossRef]
- Genton, L.; Cani, P.; Schrenzel, J. Alterations of gut barrier and gut microbiota in food restriction, food deprivation and protein-energy wasting. Clin. Nutr. 2015, 34, 341–349. [Google Scholar] [CrossRef] [PubMed]
- van de Wouw, M.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. Microbiota-Gut-Brain Axis: Modulator of Host Metabolism and Appetite. J. Nutr. 2017, 147, 727–745. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Koga, K.; Osuga, Y.; Cardenas, I.; Izumi, G.; Takamura, M.; Hirata, T.; Yoshino, O.; Hirota, Y.; Harada, M.; et al. Toll-Like Receptor-3 Ligation-Induced Indoleamine 2, 3-Dioxygenase Expression in Human Trophoblasts. Endocrinology 2011, 152, 4984–4992. [Google Scholar] [CrossRef] [Green Version]
- Frost, G.; Sleeth, M.L.; Sahuri-Arisoylu, M.; Lizarbe, B.; Cerdan, S.; Brody, L.; Anastasovska, J.; Ghourab, S.; Hankir, M.; Zhang, S.; et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 2014, 5, 3611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puertollano, E.; Kolida, S.; Yaqoob, P. Biological significance of short-chain fatty acid metabolism by the intestinal microbiome. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 139–144. [Google Scholar] [CrossRef]
- Collins, S.M.; Surette, M.; Bercik, P. The interplay between the intestinal microbiota and the brain. Nat. Rev. Microbiol. 2012, 10, 735–742. [Google Scholar] [CrossRef]
- Boulangé, C.L.; Neves, A.L.; Chilloux, J.; Nicholson, J.K.; Dumas, M.-E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 2016, 8, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Byrne, C.; Chambers, E.S.; Alhabeeb, H.; Chhina, N.; Morrison, D.J.; Preston, T.; Tedford, C.; Fitzpatrick, J.; Irani, C.; Busza, A.; et al. Increased colonic propionate reduces anticipatory reward responses in the human striatum to high-energy foods. Am. J. Clin. Nutr. 2016, 104, 5–14. [Google Scholar] [CrossRef] [Green Version]
- Perry, R.J.; Peng, L.; Barry, N.A.; Cline, G.W.; Zhang, D.; Cardone, R.L.; Petersen, K.F.; Kibbey, R.G.; Goodman, A.L.; Shulman, G.I. Acetate Mediates a Microbiome-Brain-Beta-Cell Axis to Promote Metabolic Syndrome. Nature 2016, 534, 213–217. [Google Scholar] [CrossRef] [Green Version]
- Santonicola, A.; Gagliardi, M.; Guarino, M.P.L.; Siniscalchi, M.; Ciacci, C.; Iovino, P. Eating Disorders and Gastrointestinal Diseases. Nutrients 2019, 11, 3038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Speranza, E.; Cioffi, I.; Santarpia, L.; Del Piano, C.; De Caprio, C.; Naccarato, M.; Marra, M.; De Filippo, E.; Contaldo, F.; Pasanisi, F. Fecal Short Chain Fatty Acids and Dietary Intake in Italian Women With Restrictive Anorexia Nervosa: A Pilot Study. Front. Nutr. 2018, 5, 119. [Google Scholar] [CrossRef] [Green Version]
- Bailey, M.T.; Dowd, S.; Galley, J.D.; Hufnagle, A.R.; Allen, R.G.; Lyte, M. Exposure to a social stressor alters the structure of the intestinal microbiota: Implications for stressor-induced immunomodulation. Brain Behav. Immun. 2011, 25, 397–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cowen, P.J.; Clifford, E.M.; Walsh, A.E.S.; Williams, C.; Fairburn, C.G. Moderate dieting causes 5-HT 2C receptor supersensitivity. Psychol. Med. 1996, 26, 1155–1159. [Google Scholar] [CrossRef]
- Goodwin, G.M.; Fairburn, C.G.; Cowen, P.J. Dieting Changes Serotonergic Function in Women, Not Men: Implications for the Aetiology of Anorexia Nervosa? Psychol. Med. 1987, 17, 839–842. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, G.M.; Cowen, P.J.; Fairburn, C.G.; Parry-Billings, M.; Calder, P.C.; Newsholme, E. Plasma concentrations of tryptophan and dieting. BMJ 1990, 300, 1499–1500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petty, F.; Davis, L.L.; Kabel, D.; Kramer, G.L. Serotonin dysfunction disorders: A behavioral neurochemistry perspective. J. Clin. Psychiatry 1996, 57, 11–16. [Google Scholar]
- Bailer, U.F.; Kaye, W.H. Serotonin: Imaging Findings in Eating Disorders. Behav. Neurobiol. Eat. Disord. 2010, 6, 59–79. [Google Scholar] [CrossRef] [Green Version]
- Kaye, W.H.; Frank, G.; McConaha, C. Altered Dopamine Activity after Recovery from Restricting-Type Anorexia Nervosa. Neuropsychopharmacology 1999, 21, 503–506. [Google Scholar] [CrossRef] [Green Version]
- Bailer, U.F.; Frank, G.; Henry, S.E.; Price, J.C.; Meltzer, C.C.; Weissfeld, L.; Mathis, C.A.; Drevets, W.C.; Wagner, A.; Hoge, J.; et al. Altered Brain Serotonin 5-HT1A Receptor Binding After Recovery From Anorexia Nervosa Measured by Positron Emission Tomography and [Carbonyl11C]WAY-100635. Arch. Gen. Psychiatry 2005, 62, 1032–1041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dugdale, D.C. Catecholamine Blood Test. Available online: https://medlineplus.gov/ency/article/003561.htm (accessed on 26 October 2021).
- Haldeman, C.; Turley, K.; Foley, M. Catecholamines (Urine). Available online: https://www.urmc.rochester.edu/encyclopedia/content.aspx?contenttypeid=167&contentid=catecholamines_urine (accessed on 26 October 2021).
- Mittal, R.; Debs, L.H.; Patel, A.P.; Nguyen, D.; Patel, K.; O’Connor, G.; Grati, M.; Mittal, J.; Yan, D.; Eshraghi, A.A.; et al. Neurotransmitters: The Critical Modulators Regulating Gut–Brain Axis. J. Cell. Physiol. 2016, 232, 2359–2372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirst, G.D.; Silinsky, E.M. Some Effects of 5-Hydroxytryptamine, Dopamine and Noradrenaline on Neurones in the Submucous Plexus of Guinea-Pig Small Intestine. J. Physiol. 1975, 251, 817–832. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Nutrients | BNF level | Quartile 1 (Restricting AN n = 12) | Comparison % | Representative Medium | Healthy Gut Model Medium (g/L) SS1 | Anorexic Medium (g/L) SS2 and SS3 | |
|---|---|---|---|---|---|---|---|
| M A C R O N U T R I E N T S | Carbohydrate (g) | 260 | 110 | 42% | Starch | 5 g | 2.1 g |
| Protein (g) | 50 | 33 | 66% | Peptone water Tryptone Yeast extract Casein | 5 g 5 g 4.5 g 3 g | 3.3 g 3.3 g 2.97 g 1.98 g | |
| Dietary fibre (g) | 30 | 12 | 40% | Guar Gum Inulin Pectin Arabinogalactan Xylan | 1 g 1 g 2 g 2 g 2 g | 0.4 g 0.4 g 0.8 g 0.8 g 0.8 g | |
| M I C R O N U T R I E N T S | Potassium (mg) | 3500 | 2660 | 76% | KCI | 4.5 g | 3.28 g |
| Chloride (mg) | 2500 | 1825 | 73% | NaCI | 4.5 g | 3.28 g | |
| Sodium (mg) | 1600 | 1168 | 73% | NaHCO3 | 1.5 g | 1.095 g | |
| Magnesium (mg) | 270 | 227 | 84% | MgSO4·7H2O | 1.25 g | 1.05 g | |
| Phosphorus (mg) | 550 | 607 | 110% | KH2PO4 K2HPO4 | 0.5 g 0.5 g | 0.55 g 0.55 g | |
| Calcium (mg) | 700 | 545 | 78% | CaCl2·6H2O | 0.15 g | 0.117 g | |
| Iron (mg) | 14.8 | 7.9 | 53% | Hemin FeSO4.7H2O | 0.5 g 0.005 g | 0.0265 g 0.00795 g | |
| Vitamin K (μg) | - | 68% | Vitamin K | 10 μL | 6.68 μL |
| Probe Name | Sequence (5′ to 3′) | Target Species | Reference |
|---|---|---|---|
| Non Eub | ACTCCTACGGGAGGCAGC | Control probe complementary to EUB338 | Wallner et al. (1993) [43] |
| Eub338I+ | GCTGCCTCCCGTAGGAGT | Most Bacteria | Daims et al. (1999) [44] |
| Eub338II+ | GCAGCCACCCGTAGGTGT | Planctomycetales | Daims et al. (1999) [44] |
| Eub338III+ | GCTGCCACCCGTAGGTGT | Verrucomicrobiales | Daims et al. (1999) [44] |
| Bif164 | CATCCGGCATTACCACCC | Bifidobacterium spp. | Langendijk et al. (1995) [45] |
| Lab158 | GGTATTAGCAYCTGTTTCCA | Lactobacillus and Enterococcus | Harmsen et al. (1999) [46] |
| Bac303 | CCAATGTGGGGGACCTT | Most Bacteroidaceae and Prevotellaceae, some Porphyromonadaceae | Manz et al. (1996) [47] |
| Erec482 | GCTTCTTAGTCARGTACCG | Most of the Clostridium coccoides-Eubacterium rectale group (Clostridium cluster XIVa and XIVb) | Franks et al. (1998) [48] |
| Rrec584 | TCAGACTTGCCGYACCGC | Roseburia genus | Walker et al. (2005) [49] |
| Ato291 | GGTCGGTCTCTCAACCC | Atopobium cluster | Harmsen et al. (2000) [50] |
| Prop853 | ATTGCGTTAACTCCGGCAC | Clostridium cluster IX | Walker et al. (2005) [49] |
| Fprau655 | CGCCTACCTCTGCACTAC | Feacalibacterium prausnitzii and relatives | Hold et al. (2003) [51] |
| DSV687 | TACGGATTTCACTCCT | Desulfovibrio genus | Devereux et al. (1992) [52] |
| Chis150 | TTATGCGGTATTAATCTYCCTTT | Most of the Clostridium histolyticum group (Clostridium cluster I and II) | Franks et al. (1998) [48] |
| Phasco741 | TCAGCGTCAGACACAGTC | Phascolartobacterium faecium, Acidaminococcus fermentans, Succiniclasticum ruminis | Harmsen et al. (2002) [53] |
| SUBU1237 | CCCTCTGTTCCGACCATT | Burkholderia spp. | Stoffels et al. (1998) [54] |
| Muc1437 | CCTTGCGGTTGGCTTCAGAT | Akkermansia muciniphila | Audie et al. (1993) [55] |
| Compound Name | Precursor Ion (m/z) | Product Ion (m/z) | Retention Time (min) | Classification |
|---|---|---|---|---|
| 5-HT | 177.10 | 160.10 | 10.527 | Amino acid derivative |
| DA | 154.10 | 91.05 | 8.078 | Amino acid derivative |
| NE | 170.10 | 152.15 | 4.988 | Catecholamine |
| EPI | 184.10 | 166.10 | 7.164 | Catecholamine |
| GABA | 104.10 | 87.05 | 3.690 | Organic acid |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Poveda, C.; Jenkins, P.E.; Walton, G.E. An In Vitro Approach to Studying the Microbial Community and Impact of Pre and Probiotics under Anorexia Nervosa Related Dietary Restrictions. Nutrients 2021, 13, 4447. https://doi.org/10.3390/nu13124447
Liu L, Poveda C, Jenkins PE, Walton GE. An In Vitro Approach to Studying the Microbial Community and Impact of Pre and Probiotics under Anorexia Nervosa Related Dietary Restrictions. Nutrients. 2021; 13(12):4447. https://doi.org/10.3390/nu13124447
Chicago/Turabian StyleLiu, Litai, Carlos Poveda, Paul E. Jenkins, and Gemma E. Walton. 2021. "An In Vitro Approach to Studying the Microbial Community and Impact of Pre and Probiotics under Anorexia Nervosa Related Dietary Restrictions" Nutrients 13, no. 12: 4447. https://doi.org/10.3390/nu13124447
APA StyleLiu, L., Poveda, C., Jenkins, P. E., & Walton, G. E. (2021). An In Vitro Approach to Studying the Microbial Community and Impact of Pre and Probiotics under Anorexia Nervosa Related Dietary Restrictions. Nutrients, 13(12), 4447. https://doi.org/10.3390/nu13124447






