Habitual Choline Intakes across the Childbearing Years: A Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Databases
2.2. Data Extraction
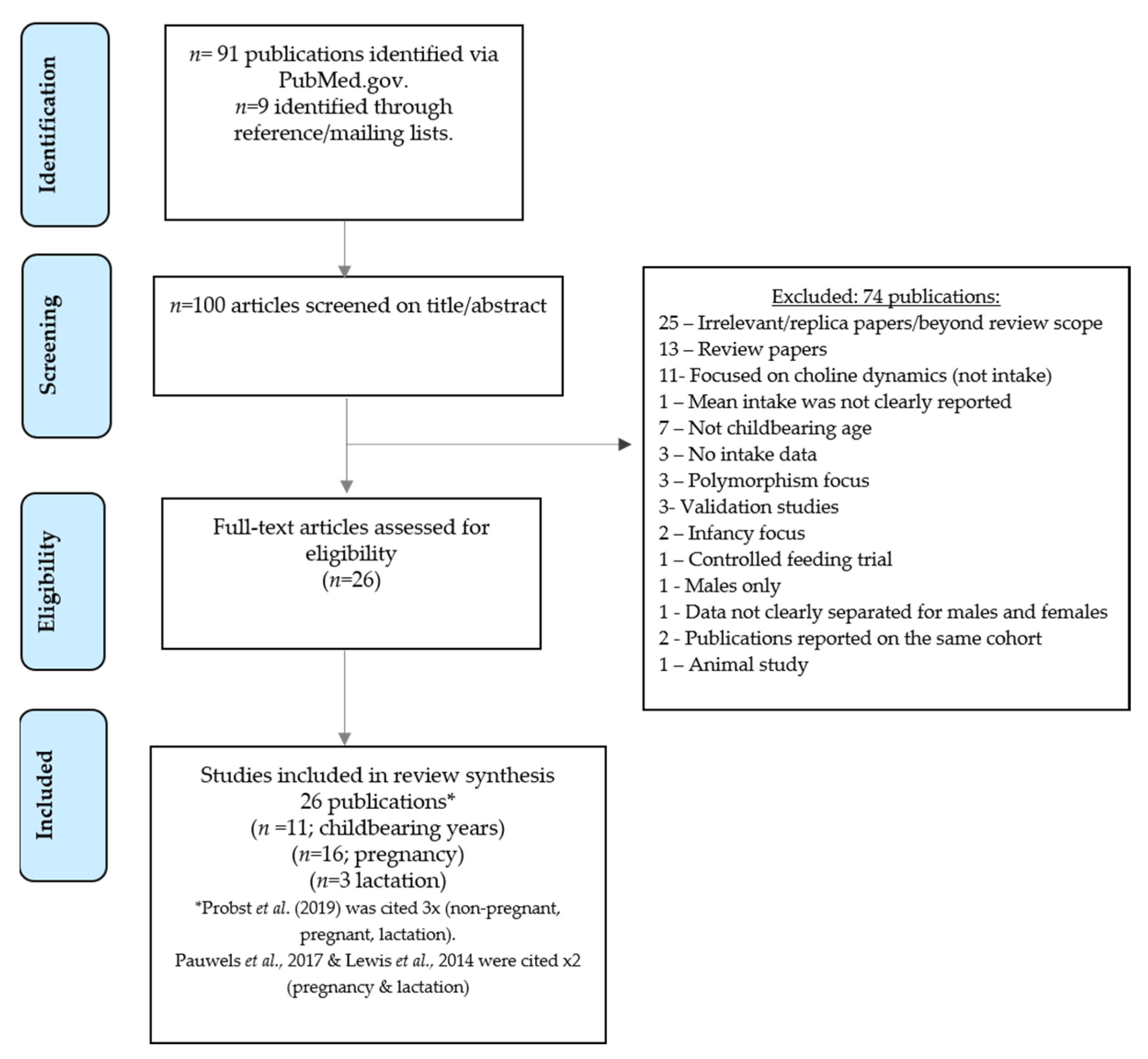
3. Results
3.1. Search Results
3.2. Childbearing Age
3.3. Pregnancy
3.4. Lactation
4. Discussion
Limitations in the Evidence and Gaps in Knowledge Surrounding Choline Intake
5. Final Evaluation and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Blusztajn, J.K.; Slack, B.E.; Mellott, T.J. Neuroprotective Actions of Dietary Choline. Nutrients 2017, 9, 815. [Google Scholar] [CrossRef] [Green Version]
- Radziejewska, A.; Chmurzynska, A. Folate and choline absorption and uptake: Their role in fetal development. Biochimie 2019, 158, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Korsmo, H.W.; Jiang, X.; Caudill, M.A. Choline: Exploring the Growing Science on Its Benefits for Moms and Babies. Nutrients 2019, 11, 1823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Derbyshire, E.; Obeid, R. Choline, Neurological Development and Brain Function: A Systematic Review Focusing on the First 1000 Days. Nutrients 2020, 12, 1731. [Google Scholar] [CrossRef]
- Wallace, T.C.; Blusztajn, J.K.; Caudill, M.A.; Klatt, K.C.; Zeisel, S.H. Choline: The Neurocognitive Essential Nutrient of Interest to Obstetricians and Gynecologists. J. Diet Suppl. 2020, 17, 733–752. [Google Scholar] [CrossRef] [PubMed]
- AAP. American Academy of Paediatrics. Food for Thought: AAP Aims to Ensure Kids Get Key Nutrients for Brain Development. 2018. Available online: https://www.aap.org/en-us/about-the-aap/aap-press-room/Pages/Food-for-Thought-AAP-Aims-to-Ensure-Kids-Get-Key-Nutrients-for-Brain-Development.aspx (accessed on 12 April 2021).
- Berg, S. American Medical Association. AMA backs Global Health Experts in Calling Infertility a Disease. 2017. Available online: https://www.ama-assn.org/delivering-care/public-health/ama-backs-global-health-experts-calling-infertility-disease (accessed on 10 April 2021).
- Bekdash, R.A. Neuroprotective Effects of Choline and Other Methyl Donors. Nutrients 2019, 11, 2995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheatham, C.L. Nutritional Factors in Fetal and Infant Brain Development. Ann. Nutr. Metab. 2019, 75 (Suppl. 1), 20–32. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. The fetal origins of memory: The role of dietary choline in optimal brain development. J. Pediatr. 2006, 149, 131–136. [Google Scholar] [CrossRef] [Green Version]
- Blusztajn, J.K.; Mellott, T.J. Choline nutrition programs brain development via DNA and histone methylation. Cent. Nerv. Syst. Agents Med. Chem. 2012, 12, 82–94. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Dietary Reference Values for Choline. EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). EFSA J. 2016, 14, 4484. [Google Scholar]
- IoM. Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin and Choline; National Academy Press: Washington, DC, USA, 1998. [Google Scholar]
- Wallace, T.C.; Blusztajn, J.K.; Caudill, M.A.; Klatt, K.C.; Natker, E.; Zeisel, S.H.; Zelman, K.M. Choline: The Underconsumed and Underappreciated Essential Nutrient. Nutr. Today 2018, 53, 240–253. [Google Scholar] [CrossRef] [PubMed]
- Derbyshire, E. Could we be overlooking a potential choline crisis in the United Kingdom? BMJ Nutr. Prev. Health 2019, 2, 86–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bailey, R.L.; Pac, S.G.; Fulgoni, V.L., 3rd; Reidy, K.C.; Catalano, P.M. Estimation of Total Usual Dietary Intakes of Pregnant Women in the United States. JAMA Netw. Open 2019, 2, e195967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, T.C.; Fulgoni, V.L., 3rd. Assessment of Total Choline Intakes in the United States. J. Am. Coll. Nutr. 2016, 35, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Fulgoni, V.L. Usual Choline Intakes Are Associated with Egg and Protein Food Consumption in the United States. Nutrients 2017, 9, 839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Probst, Y.; Guan, V.; Neale, E. Development of a Choline Database to Estimate Australian Population Intakes. Nutrients 2019, 11, 913. [Google Scholar] [CrossRef] [Green Version]
- Mygind, V.L.; Evans, S.E.; Peddie, M.C.; Miller, J.C.; Houghton, L.A. Estimation of usual intake and food sources of choline and betaine in New Zealand reproductive age women. Asia Pac. J. Clin. Nutr. 2013, 22, 319–324. [Google Scholar] [CrossRef]
- Vennemann, F.B.; Ioannidou, S.; Valsta, L.M.; Dumas, C.; Ocke, M.C.; Mensink, G.B.; Lindtner, O.; Virtanen, S.M.; Tlustos, C.; D’Addezio, L.; et al. Dietary intake and food sources of choline in European populations. Br. J. Nutr. 2015, 114, 2046–2055. [Google Scholar] [CrossRef] [Green Version]
- Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary Choline Intake: Current State of Knowledge Across the Life Cycle. Nutrients 2018, 10, 1513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- USDA. The USDA National Nutrient Databank: Acquision, Evaluation & Compilation of Food Composiion Data. Available online: https://fdc.nal.usda.gov/ (accessed on 6 December 2021).
- Derbyshire, E. Nutrition in the Childbearing Years; John Wiley and Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Lavery, A.M.; Brender, J.D.; Zhao, H.; Sweeney, A.; Felkner, M.; Suarez, L.; Canfield, M.A. Dietary intake of choline and neural tube defects in Mexican Americans. Birth Defects Res. A Clin. Mol. Teratol. 2014, 100, 463–471. [Google Scholar] [CrossRef]
- Petersen, J.M.; Parker, S.E.; Crider, K.S.; Tinker, S.C.; Mitchell, A.A.; Werler, M.M. One-Carbon Cofactor Intake and Risk of Neural Tube Defects Among Women Who Meet Folic Acid Recommendations: A Multicenter Case-Control Study. Am. J. Epidemiol. 2019, 188, 1136–1143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oyen, J.; Gjesdal, C.G.; Karlsson, T.; Svingen, G.F.; Tell, G.S.; Strand, E.; Drevon, C.A.; Vinknes, K.J.; Meyer, K.; Ueland, P.M.; et al. Dietary Choline Intake Is Directly Associated with Bone Mineral Density in the Hordaland Health Study. J. Nutr. 2017, 147, 572–578. [Google Scholar] [CrossRef] [Green Version]
- Gao, X.; Wang, Y.; Randell, E.; Pedram, P.; Yi, Y.; Gulliver, W.; Sun, G. Higher Dietary Choline and Betaine Intakes Are Associated with Better Body Composition in the Adult Population of Newfoundland, Canada. PLoS ONE 2016, 11, e0155403. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.E.; Jacques, P.F.; Dougherty, L.; Selhub, J.; Giovannucci, E.; Zeisel, S.H.; Cho, E. Are dietary choline and betaine intakes determinants of total homocysteine concentration? Am. J. Clin. Nutr. 2010, 91, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Mar, M.H.; Howe, J.C.; Holden, J.M. Concentrations of choline-containing compounds and betaine in common foods. J. Nutr. 2003, 133, 1302–1307. [Google Scholar] [CrossRef]
- Chiuve, S.E.; Giovannucci, E.L.; Hankinson, S.E.; Zeisel, S.H.; Dougherty, L.W.; Willett, W.C.; Rimm, E.B. The association between betaine and choline intakes and the plasma concentrations of homocysteine in women. Am. J. Clin. Nutr. 2007, 86, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.M.; Scearce, J.A.; Mar, M.H.; Patel, J.R.; Blanchard, R.T.; Macintosh, B.A.; Busby, M.G.; Zeisel, S.H. Ad libitum choline intake in healthy individuals meets or exceeds the proposed adequate intake level. J. Nutr. 2005, 135, 826–829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, G.M.; Carmichael, S.L.; Yang, W.; Selvin, S.; Schaffer, D.M. Periconceptional dietary intake of choline and betaine and neural tube defects in offspring. Am. J. Epidemiol. 2004, 160, 102–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fawcett, K.; Sims, C.; Gaitliff, R. Associations Between Maternal Dietary Choline Intake During Pregnancy and Developmental Outcomes in Toddlers. Curr. Dev. Nutr. 2020, 4, 980. [Google Scholar] [CrossRef]
- Moore, C.J.; Perreault, M.; Mottola, M.F.; Atkinson, S.A. Diet in Early Pregnancy: Focus on Folate, Vitamin B12, Vitamin D, and Choline. Can. J. Diet. Pract. Res. 2020, 81, 58–65. [Google Scholar] [CrossRef]
- Zhu, J.; Liu, Y.H.; He, X.L.; Kohlmeier, M.; Zhou, L.L.; Shen, L.W.; Yi, X.X.; Tang, Q.Y.; Cai, W.; Wang, B. Dietary Choline Intake during Pregnancy and PEMT rs7946 Polymorphism on Risk of Preterm Birth: A Case-Control Study. Ann. Nutr. Metab. 2020, 76, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, S.; Ghosh, M.; Duca, R.C.; Bekaert, B.; Freson, K.; Huybrechts, I.; Langie, S.A.S.; Koppen, G.; Devlieger, R.; Godderis, L. Maternal intake of methyl-group donors affects DNA methylation of metabolic genes in infants. Clin. Epigenet. 2017, 9, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Groth, S.W.; Stewart, P.A.; Ossip, D.J.; Block, R.C.; Wixom, N.; Fernandez, I.D. Micronutrient Intake Is Inadequate for a Sample of Pregnant African-American Women. J. Acad. Nutr. Diet 2017, 117, 589–598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masih, S.P.; Plumptre, L.; Ly, A.; Berger, H.; Lausman, A.Y.; Croxford, R.; Kim, Y.I.; O’Connor, D.L. Pregnant Canadian Women Achieve Recommended Intakes of One-Carbon Nutrients through Prenatal Supplementation but the Supplement Composition, Including Choline, Requires Reconsideration. J. Nutr. 2015, 145, 1824–1834. [Google Scholar] [CrossRef] [Green Version]
- Goon, S.; Dey, S.R. A 24-hour dietary recall for assessing the intake pattern of choline among Bangladeshi pregnant women at their third trimester of pregnancy. Cent. Asian J. Glob. Health 2014, 3, 72. [Google Scholar] [CrossRef] [Green Version]
- Lewis, E.D.; Subhan, F.B.; Bell, R.C.; McCargar, L.J.; Curtis, J.M.; Jacobs, R.L.; Field, C.J.; APrON team. Estimation of choline intake from 24 h dietary intake recalls and contribution of egg and milk consumption to intake among pregnant and lactating women in Alberta. Br. J. Nutr. 2014, 112, 112–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, B.T.; Dyer, R.A.; King, D.J.; Richardson, K.J.; Innis, S.M. Early second trimester maternal plasma choline and betaine are related to measures of early cognitive development in term infants. PLoS ONE 2012, 7, e43448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villamor, E.; Rifas-Shiman, S.L.; Gillman, M.W.; Oken, E. Maternal intake of methyl-donor nutrients and child cognition at 3 years of age. Paediatr. Perinat. Epidemiol. 2012, 26, 328–335. [Google Scholar] [CrossRef] [Green Version]
- EFSA. European Food Safety Authority. Use of the EFSA Comprehensive European Food Consumption Database in exposure assessment. EFSA J. 2011, 9, 2097. [Google Scholar]
- Gossell-Williams, M.; Fletcher, H.; McFarlane-Anderson, N.; Jacob, A.; Patel, J.; Zeisel, S. Dietary intake of choline and plasma choline concentrations in pregnant women in Jamaica. West Indian Med. J. 2005, 54, 355–359. [Google Scholar] [CrossRef]
- Visentin, C.E.; Masih, S.; Plumptre, L.; Malysheva, O.; Nielsen, D.E.; Sohn, K.J.; Ly, A.; Lausman, A.Y.; Berger, H.; Croxford, R.; et al. Maternal Choline Status, but Not Fetal Genotype, Influences Cord Plasma Choline Metabolite Concentrations. J. Nutr. 2015, 145, 1491–1497. [Google Scholar] [CrossRef] [Green Version]
- Fisher, M.C.; Zeisel, S.H.; Mar, M.H.; Sadler, T.W. Inhibitors of choline uptake and metabolism cause developmental abnormalities in neurulating mouse embryos. Teratology 2001, 64, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Zeisel, S.H.; Mar, M.H.; Sadler, T.W. Perturbations in choline metabolism cause neural tube defects in mouse embryos in vitro. FASEB J. 2002, 16, 619–621. [Google Scholar] [CrossRef] [Green Version]
- Lewis, E.D.; Kosik, S.J.; Zhao, Y.Y.; Jacobs, R.L.; Curtis, J.M.; Field, C.J. Total choline and choline-containing moieties of commercially available pulses. Plant Foods Hum. Nutr. 2014, 69, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Zhao, Y.Y.; Richard, C.; Bruce, H.L.; Jacobs, R.L.; Field, C.J.; Curtis, J.M. Measurement of the abundance of choline and the distribution of choline-containing moieties in meat. Int. J. Food Sci. Nutr. 2015, 66, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Richard, C.; Lewis, E.; Zhao, Y.; Asomaning, J.; Jacobs, R.; Field, C.J.; Curtis, J. Measurement of the total choline content in 48 commercial dairy products or dairy alternatives. J. Food Compost. Anal. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Lecorguille, M.; Lioret, S.; de Lauzon-Guillain, B.; de Gavelle, E.; Forhan, A.; Mariotti, F.; Charles, M.A.; Heude, B. Association between Dietary Intake of One-Carbon Metabolism Nutrients in the Year before Pregnancy and Birth Anthropometry. Nutrients 2020, 12, 838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radjursoga, M.; Lindqvist, H.M.; Pedersen, A.; Karlsson, B.G.; Malmodin, D.; Ellegard, L.; Winkvist, A. Nutritional Metabolomics: Postprandial Response of Meals Relating to Vegan, Lacto-Ovo Vegetarian, and Omnivore Diets. Nutrients 2018, 10, 1063. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrin, M.T.; Pawlak, R.; Allen, L.H.; Hampel, D. Total Water-Soluble Choline Concentration Does Not Differ in Milk from Vegan, Vegetarian, and Nonvegetarian Lactating Women. J. Nutr. 2020, 150, 512–517. [Google Scholar] [CrossRef]
- Wiedeman, A.M.; Whitfield, K.C.; March, K.M.; Chen, N.N.; Kroeun, H.; Sokhoing, L.; Sophonneary, P.; Dyer, R.A.; Xu, Z.; Kitts, D.D.; et al. Concentrations of Water-Soluble Forms of Choline in Human Milk from Lactating Women in Canada and Cambodia. Nutrients 2018, 10, 381. [Google Scholar] [CrossRef] [Green Version]
- Wennberg, A.L.; Isaksson, U.; Sandstrom, H.; Lundqvist, A.; Hornell, A.; Hamberg, K. Swedish women’s food habits during pregnancy up to six months post-partum: A longitudinal study. Sex Reprod. Healthc. 2016, 8, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Van der Pligt, P.; Olander, E.K.; Ball, K.; Crawford, D.; Hesketh, K.D.; Teychenne, M.; Campbell, K. Maternal dietary intake and physical activity habits during the postpartum period: Associations with clinician advice in a sample of Australian first time mothers. BMC Pregnancy Childbirth 2016, 16, 27. [Google Scholar] [CrossRef] [Green Version]
- Harvard, T. Harvard Willett Food Frequency Questionnaire. Available online: https://snaped.fns.usda.gov/library/materials/harvard-willett-food-frequency-questionnaire (accessed on 6 December 2021).
- Carter, R.C.; Jacobson, S.W.; Booley, S.; Najaar, B.; Dodge, N.C.; Bechard, L.J.; Meintjes, E.M.; Molteno, C.D.; Duggan, C.P.; Jacobson, J.L.; et al. Development and validation of a quantitative choline food frequency questionnaire for use with drinking and non-drinking pregnant women in Cape Town, South Africa. Nutr. J. 2018, 17, 108. [Google Scholar] [CrossRef]
- Ortega, R.M.; Perez-Rodrigo, C.; Lopez-Sobaler, A.M. Dietary assessment methods: Dietary records. Nutr. Hosp. 2015, 31 (Suppl. 3), 38–45. [Google Scholar] [CrossRef] [PubMed]
- Shaneshin, M.; Jessri, M.; Rashidkhani, B. Validity of energy intake reports in relation to dietary patterns. J. Health Popul. Nutr. 2014, 32, 36–45. [Google Scholar] [PubMed]
- Wiedeman, A.M.; Dyer, R.A.; Innis, S.M. Variability in Plasma Free Choline and its Relation with Diet and Potential Plasma Biomarkers. FASEB J. 2015, 29, 919–922. [Google Scholar] [CrossRef]
- Romano, K.A.; Martinez-Del Campo, A.; Kasahara, K.; Chittim, C.L.; Vivas, E.I.; Amador-Noguez, D.; Balskus, E.P.; Rey, F.E. Metabolic, Epigenetic, and Transgenerational Effects of Gut Bacterial Choline Consumption. Cell Host Microbe 2017, 22, 279–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goh, Y.Q.; Cheam, G.; Wang, Y. Understanding Choline Bioavailability and Utilization: First Step Toward Personalizing Choline Nutrition. J. Agric. Food Chem. 2021, 69, 37. [Google Scholar] [CrossRef]
- Schwarzenberg, S.J.; Georgieff, M. Advocacy for Improving Nutrition in the First 1000 Days to Support Childhood Development and Adult Health. Committee on Nutrition. Paediatrics 2018, 141, e20173716. [Google Scholar] [CrossRef] [Green Version]
| First Author, Country | Age, Years | Study Design, Cohort Name | Sample Size | Method and Duration/Time Period | Database Used to Estimate the Intake | Source(s) of Choline | Choline Intake (mg/day) |
|---|---|---|---|---|---|---|---|
| Petersen et al. (2019), United States [26] | Range <25 years (12.4%) 25–34 years (68.3%) ≥35 years (19.4%) | Case–control study (the Slone Birth Defect study 1975–2014) | Mothers of n = 164 NTD cases (live births, foetal deaths, elective terminations), and n = 2831 control infants | SQ FFQ (the Willett FFQ) to capture the intake in the past 6 months | Harvard T.H. Chan School of Public Health Nutrition Department’s Food Composition Table. | F+S | Mean (SD) = Controls: 275 (68) Cases: 273 (67) Choline intake data were based on women with folic acid intake >400 µg/day |
| Probst et al. (2019), Australia [19] | Range 16–44 years | Cross-sectional study based on The Australian Health Survey 2011–13 | n = 2210 | 2 day intake data | Sourced studies and global food composition databases and compared these with data from Australian foods (from AUSNUT 2011–13 Australian Food) | FO | Median (IQR) = 233 (103) |
| Oyen et al. (2017), Norway [27] | Range 46–49 years | Cross-sectional study based on The Hordaland Health study | n = 1600 | FFQ (habitual diet from past year) validated: 169 items; frequency of consumption specified per day, week, or month | Choline intakes estimated according to the 2008 USDA choline database | F+S | Median (IQR) = 255 (63) |
| Gao et al. (2016), Canada [28] | Mean = 43.7 years | Cross-sectional CODING study (Complex Diseases in the Newfoundland population: Environment and Genetics) study | n = 2232–2295 (sample size range) | SQ FFQ (the Willett FFQ) 124 items; data from past 12 months | NutriBase Clinical Nutrition Manager | FO | Mean (SD) = 292 (213) |
| Wallace et al. (2016), United States [17] | Range 19–30 years Range 31–50 years | Cross-sectional study based on the National Health and Nutrition Examination Survey (2009–2012) | n = 1096 (19–30 years) n = 1794 (31–50 years) | Two 24 h dietary recalls with trained interviewers | Various USDA food composition databases | F+S | Mean (SD) = 19–30 years: 250 (166); 31–50 years: 278 (169) |
| Vennemann et al. (2015), Europe [21] | Range 18–≤65 years | Cross-sectional dietary surveys in each country | Finland n = 710; France n = 1340; Ireland n = 640; Italy n = 1245; Netherlands n = 1034; Sweden n = 807; UK n = 706 | 3 day dietary record 7 day dietary record 4 day dietary record 3 day dietary record 24 h dietary recall 4 day web record 4 day dietary record | USDA Database (2013) | FO | Mean (5th, 95th percentiles) = Finland: 344 (177, 578); France: 291 (162, 440); Ireland: 318 (166, 485); Italy: 293 (153, 463); Netherlands: 334 (185, 543); Sweden: 374 (186, 631); UK: 294 (145, 478) |
| Lavery et al. (2014), Mexican Americans [25] | Range <20 years (24.7%) 20–24 years (34.2%) 25–29 years (24%) >30 years (35%) | Case–control study | Mothers of n = 184 cases with NTDs and n = 225 controls | FFQ: 98 items ascertaining food frequency for 3 months before their conception date to 3 months postpartum | USDA database containing 630 food items and six choline metabolites. | FO | Median (IQR) = Controls: 760 (456); Cases: 722 (689) |
| Lee et al. (2010), United States [29] | Range 29–86 years | Cross=sectional study in the sixth examination (1995–1998) of the Framingham Offspring Study | n = 1407 | FFQ, validated: 130 items; frequency of consumption over the past year | Choline composition data values published by Zeisel et al. [30] and from the USDA 2008 Database | F+S | Energy adjusted mean (SD) = 308 (56) |
| Chiuve et al. (2007), United States [31] | Range 30–55 years | Cross-sectional study based on the Nurses’ Health Study | n = 1477 | SQ FFQ, validated: undertaken every 4-years | Choline composition data values published by Zeisel et al. [30] and from the USDA 2008 Database | FO | Energy adjusted median (range of 3rd quintile) = 323 (311–334) |
| Fischer et al. (2005), United States [32] | Range 18–67 years | Ad libitum dietary intake in the study centre | n = 16 | Choline content was measured in the foods and compared to estimates from a pre-study 3 day food record | The Food Processor SQL program using USDA Nutrient Database and Zeisel et al. [30] data | FO | Mean (SD) = 443 (88) |
| Shaw et al. (2004), United States [33] | Women recruited after delivery, age not reported | Case–control study | Mothers of n = 424 cases with NTDs, mothers of n = 440 controls | FFQ: 100 items used to assess frequency and portion size consumed 3 months before conception | Used choline values published by Zeisel et al. [30] | FO | Mean (SD) = Controls: 409 (179); Cases: 377 (176) |
| First Author, Country | Age, Years | Study Design | Sample Size | Method and Duration/ Time Period | Database Used to Estimate the Intake | Source(s) of Choline | Choline Intake (mg/day) |
|---|---|---|---|---|---|---|---|
| Fawcet et al. (2020), United States [34] | Not reported. | Prospective longitudinal study | n = 251 | 3 day food records completed during the 1st, 2nd, and 3rd trimester | Nutrient Data System for Research | FO | Mean (SD not reported) = 281 |
| Moore et al. (2020), United Kingdom [35] | Mean (SD) = 31.4 (4) years | Be Healthy in Pregnancy (B-HIP) study, baseline data of a RCT | n = 232 | 3 day weighed diet record (2 week days and 1 weekend day) completed at 12–17 weeks gestation | Nutritionist Pro™ diet analysis software | F+S | Median (min, max) = 338 (120, 1016) |
| Zhu et al. (2020), China [36] | Mean (SD) = 28 (4) years | Case–control study (SQ FFQ retrospective collecting dietary intake during pregnancy) | Mothers of n = 157 term controls | SQ FFQ, validated, 120 food items including the most common foods in the Chinese diet conducted no later than 3 days after parturition | China Food Composition and the USDA Food Composition Database | FO | Energy-adjusted choline intake in the controls Median (IQR) = 255 (70) |
| Probst et al. (2019), Australia [19] | Range 19–50 years | Cross-sectional study based on data from the Australian National Nutrition and Physical Activity Survey 2011–12 | n = 116 | 2 day intake data (data filtered for women who were pregnant at the time of the survey) | Sourced studies and global food composition databases, compared with data for Australian foods to create a choline database | FO | Median (IQR) = 251 (111) |
| Bailey et al. (2019), United States [16] | Range 20–40 years | Cross-sectional study based on data from NHANES (2001–2014) | n = 533 | 24 h dietary recall ×2 (taking part in the What We Eat in America survey) | USDA Food Composition Database 2019 | FO | Mean (SD) = 321 (231) |
| Pauwels et al. (2017), Belgium [37] | Range 25–41 years | A longitudinal Maternal Nutrition and Offspring’s Epigenome study (MANOE) | 1st trimester: n = 94 2nd trimester: n = 85 3rd trimester: n = 82 | FFQ, validated: 51 items completed at 11–13, 18–22, and 30–34 weeks of pregnancy | Not reported | F+S | Mean (SD) = 1st trimester: 274 (72); 2nd trimester: 268 (68); 3rd trimester: 280 (78) |
| Wallace et al. (2017), United States [18] | Range 13–44 years | Cross-sectional study based on data from the 2009–2014 and 2005–2014 NHANES (2009–2014 and 2005–2014) datasets | n = 593 | 24 h dietary recalls ×2 | Various USDA food composition databases used | F+S | Mean (SD) = 319 (241) |
| Groth et al. (2017), United States [38] | Range 18–36 years | Prospective observational study, secondary analysis of the Limiting the Phenotypic Effect of Pregnancy-Related Weight Gain | 1st trimester: n = 90 2nd trimester: n = 68 3rd trimester: n = 67 | 24 h dietary recalls ×3 at three timepoints: early (<22 weeks), mid (24–29 weeks), and late (32–37 weeks) pregnancy. | Nutrition Data System for Research software 2009 | FO | Mean (SD) = 1st trimester: 318 (68); 2nd trimester: 289 (28); 3rd trimester: 306 (28) |
| Masih et al. (2015), Canada [39] | Mean (SD) = 32 (5) years | Prospective observational study, the Prenatal Folic Acid Exposure on DNA Methylation in the newborn infant study | n = 290 | SQ FFQ, validated: 110 items; recall of habitual intakes between 0–16 and 23–27 weeks gestation | Nutrition Quest used the nutrient composition data primarily from version 1.0 of the USDA Food and Nutrient Database | F+S | Mean (SD) = 1st trimester: 306 (127); 3rd trimester: 302 (122) |
| Goon et al. (2014), Bangladesh [40] | Range 21–25 years | Cross-sectional study | n = 103 | 24 h dietary recall in the 7th, 8th, or 9th months of pregnancy | USDA Food Composition database | FO | Mean (SD) = 190 (98) |
| Lewis et al. (2014), Canada [41] | Range 17–30 years (45.8%) 31–45 years (54.2%) | Prospective cohort study, the Alberta Pregnancy Outcomes and Nutrition (APrON) cohort study | 1st trimester: n = 123; 2nd trimester: n = 562; 3rd trimester: n = 493 | 24 h dietary recall using the multiple-pass method in each trimester | The Alberta database used the USDA Database for the Choline Content of Common Foods, Release 2 (634 foods) | F+S | Mean (SD) = 1st trimester: 340 (148); 2nd trimester: 349 (154); 3rd trimester: 353 (144) |
| Mygind et al. (2013), New Zealand [20] | Range 18–40 years | Baseline part of dietary data collection for a folate intervention study | n = 125 | 3 day weighed food record Two weekdays and one weekend day | USDA Choline Content of Common Foods, Release 2, 2008 | FO | Median (IQR) = 310 (87) |
| Wu et al. (2012), Canada [42] | Not reported | Prospective study | n = 154 | FFQ, women enrolled from 16 weeks gestation | USDA Choline Content of Common Foods, Release 2, 2008 | FO | Mean (SD) = 383 (99) |
| Villamor et al. (2012), United States [43] | Mean (SD) = 33 (5) years | Project Viva longitudinal study | 1st trimester: n = 1148 2nd trimester: n = 1083 | FFQ in the first and second trimester (26–28 weeks gestation) | Harvard nutrient composition database | F+S | Mean (SD) = 1st trimester: 332 (63); 2nd trimester: 325 (64) |
| EFSA (2016), Latvia [12,44] | Range 15–45 years | Food Consumption data from the EFSA European Comprehensive Food Consumption Database | n = 990 | 24 h dietary recall in pregnancy | Not reported | FO | Pregnant adolescents: Mean = 336; Pregnant women: Mean (5th, 95th percentiles) = 356 (200, 592) |
| Gossell-Williams et al. (2005), Jamaica [45] | Range 18–32 years | Observational study | n = 16 | FFQ (Jamaican foods) recruited at 10–15 weeks gestation | USDA Food Composition database | FO | Mean (SD) = 279 (116) |
| First Author, Country | Age, Years | Study Design | Sample Size | Method and Duration/Time Period | Database Used to Estimate the Intake | Source(s) of Choline | Choline Intake (mg/day) |
|---|---|---|---|---|---|---|---|
| Probst et al. (2019), Australia [19] | Range 19–50 years | Cross-sectional study based on data from the Australian National Nutrition and Physical Activity Survey 2011–12 | n = 110 | 2 day intake data analysed from ‘lactating mothers’ | Sourced studies and global food composition databases, compared these with data for Australian foods to create a choline database | FO | Median (IQR) = 257 (100) |
| Pauwels et al. (2017), Belgium [37] | Range 25–41 years | The Maternal and Offspring’s Epigenome prospective, observational cohort study | 6–8 weeks PP: n = 79; 6 months PP: n = 60 | FFQ, validated 51-items conducted at 6–8 weeks and 6 months postpartum | Not reported | F+S | Mean (SD) = 6–8 weeks PP: 278 (76); 6 months PP: 268 (60) |
| Lewis et al. (2014), Canada [41] | Range 17–30 years (45.8%) 31–45 years (54.2%) | Alberta Pregnancy Outcomes and Nutrition (APrON) cohort study | n = 488 | 24 h dietary recall using the multiple-pass method | The Alberta database was developed using the USDA Database for the Choline Content of Common Foods, Release 2 (634 foods) | F+S | Mean (SD) = 346 (151) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Derbyshire, E.; Obeid, R.; Schön, C. Habitual Choline Intakes across the Childbearing Years: A Review. Nutrients 2021, 13, 4390. https://doi.org/10.3390/nu13124390
Derbyshire E, Obeid R, Schön C. Habitual Choline Intakes across the Childbearing Years: A Review. Nutrients. 2021; 13(12):4390. https://doi.org/10.3390/nu13124390
Chicago/Turabian StyleDerbyshire, Emma, Rima Obeid, and Christiane Schön. 2021. "Habitual Choline Intakes across the Childbearing Years: A Review" Nutrients 13, no. 12: 4390. https://doi.org/10.3390/nu13124390
APA StyleDerbyshire, E., Obeid, R., & Schön, C. (2021). Habitual Choline Intakes across the Childbearing Years: A Review. Nutrients, 13(12), 4390. https://doi.org/10.3390/nu13124390








