Exercise Effects on Bone Mineral Density in Men
Abstract
:1. Introduction
2. Materials and Methods
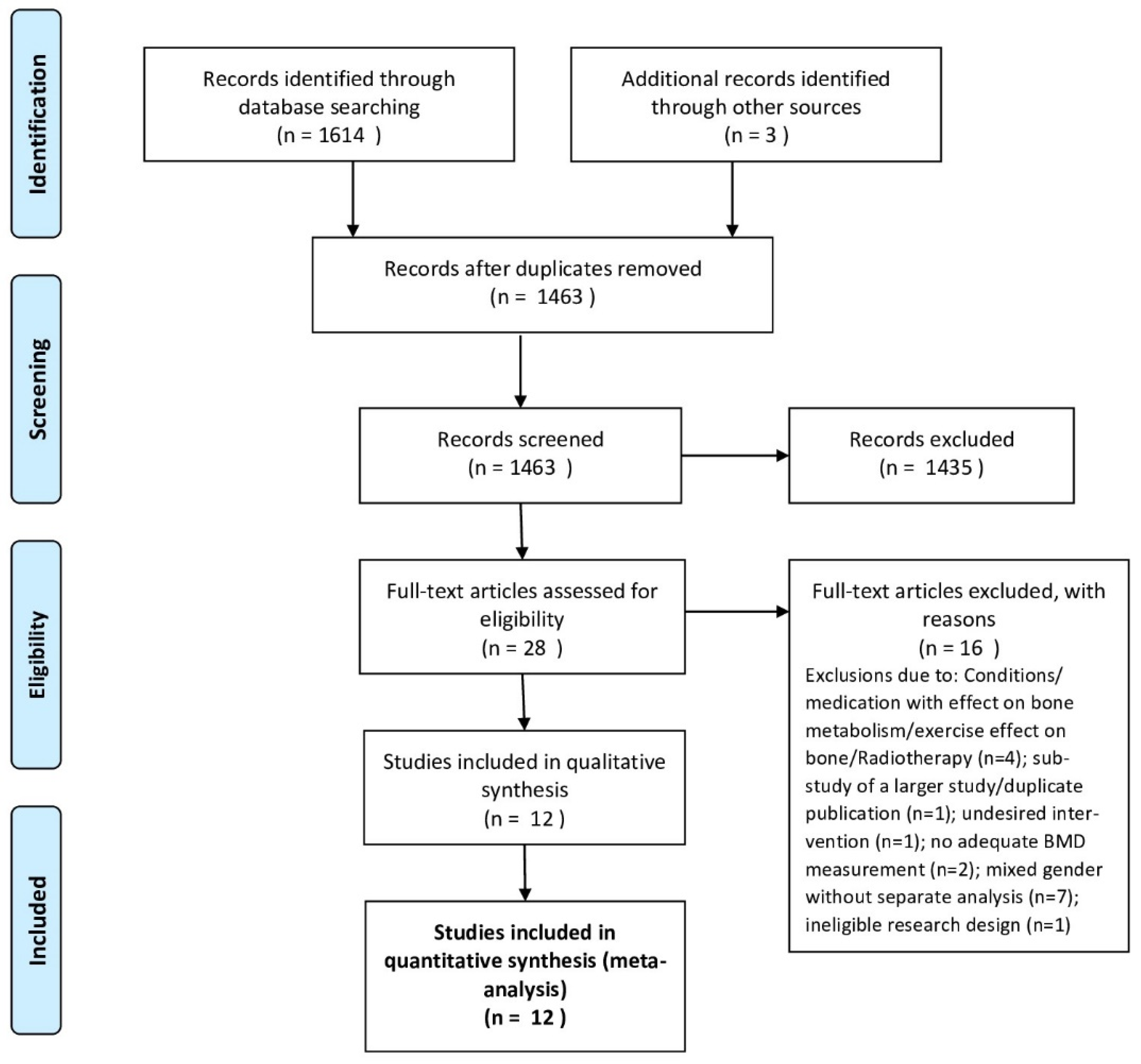
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Outcome Measures
2.5. Quality Assessment
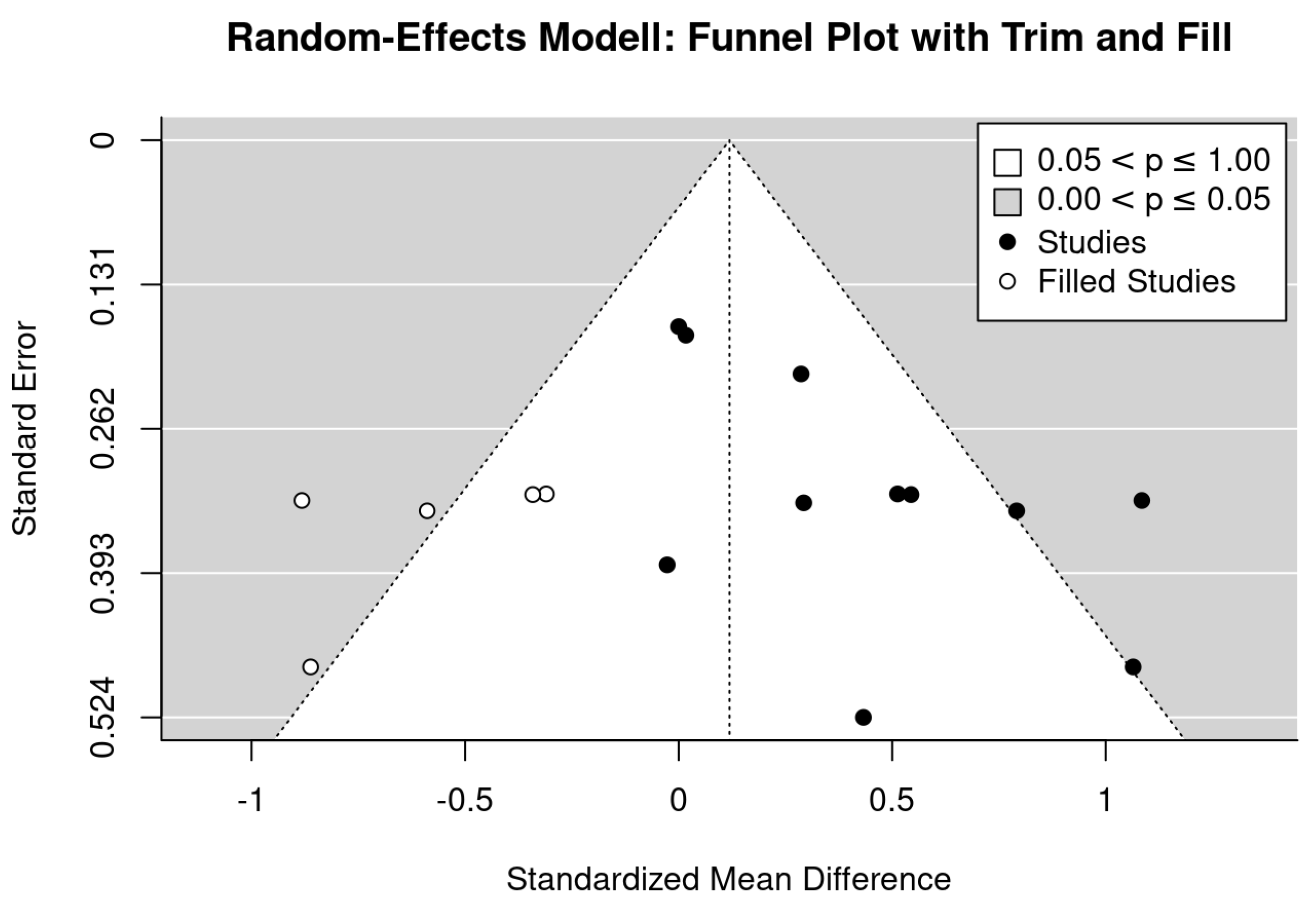
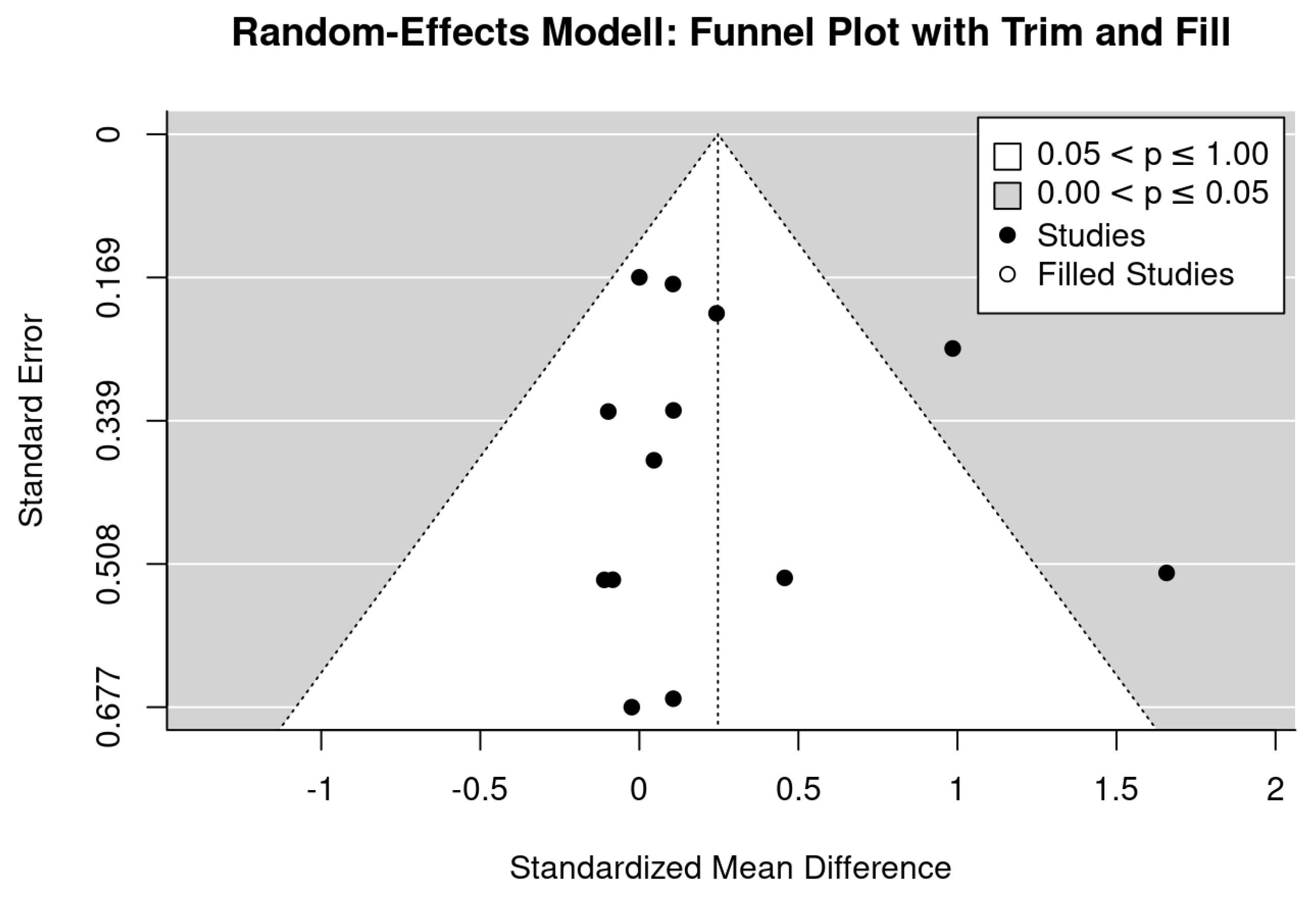
2.6. Data Synthesis
2.7. Statistical Analysis
3. Results
3.1. Study Characteristics and Quality Assessment
3.2. Intervention Characteristics
3.2.1. Protein, Vitamin-D and Calcium Supplementation; Nutrition
3.2.2. Exercise Characteristics
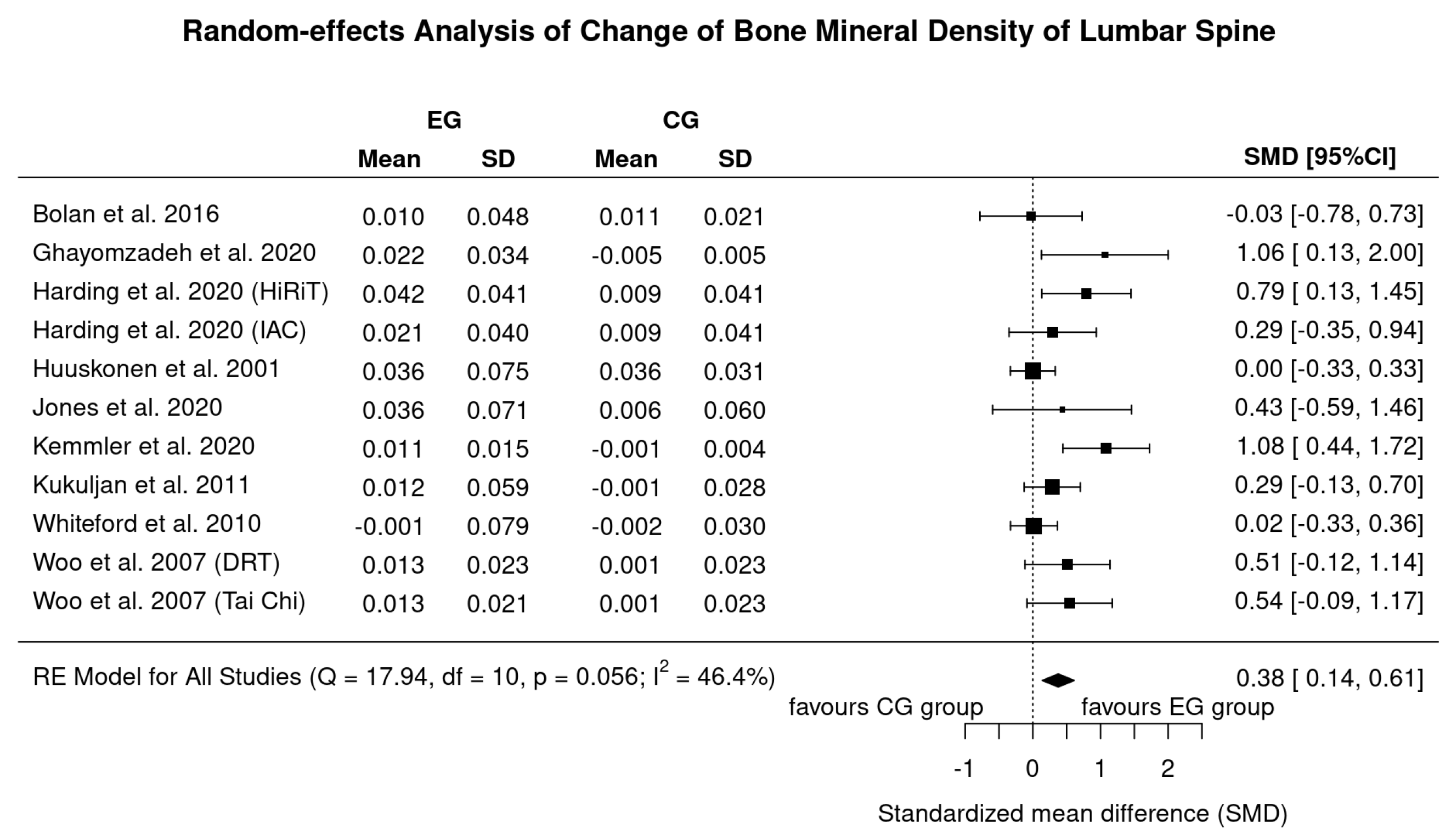
3.3. Results on BMD at the Lumbar Spine (LS)-ROI
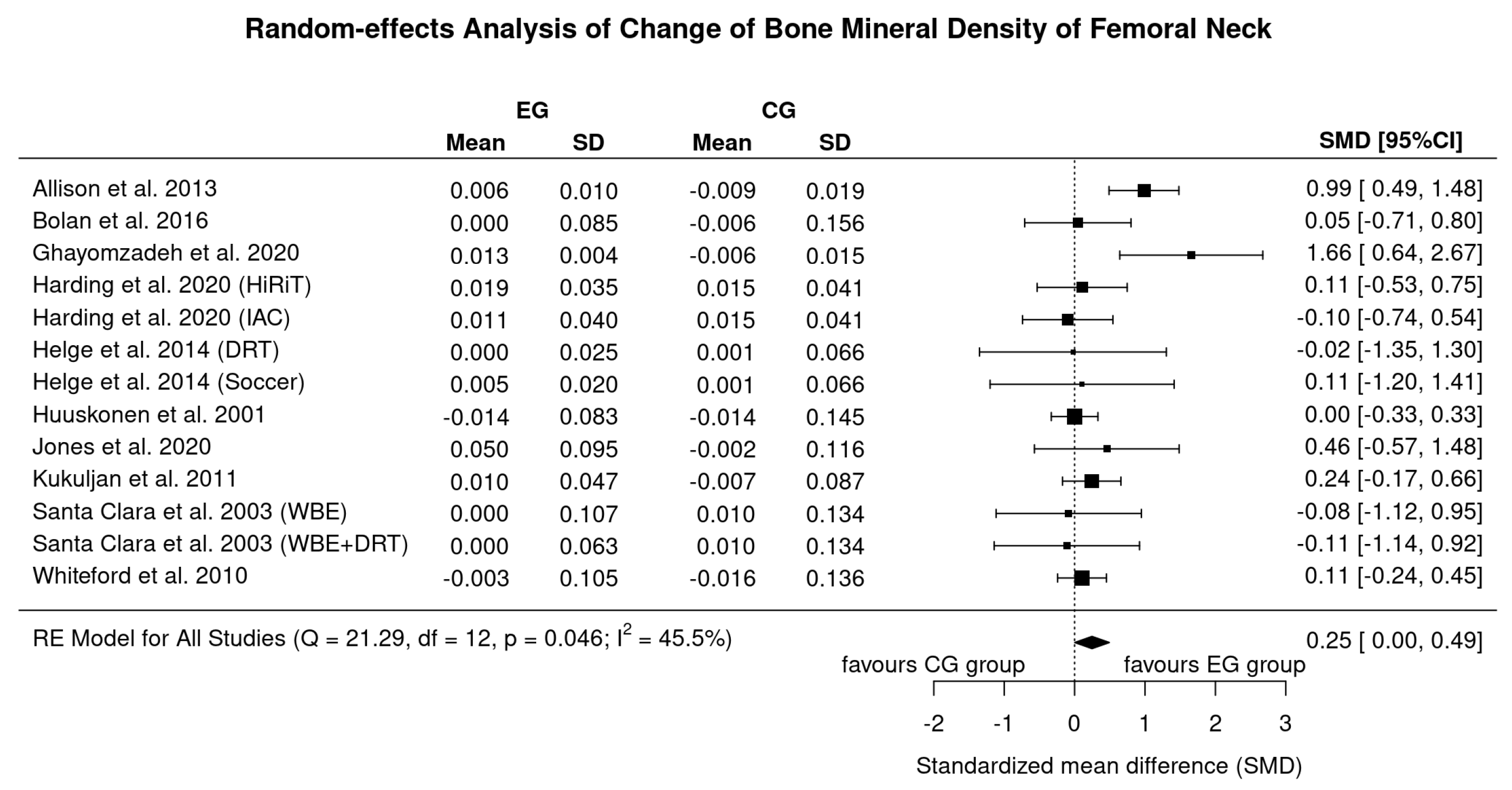
3.4. Results on BMD at the Proximal Femur
3.5. Subgroup Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Steering Group. Best Practice Guidelines to Prevent and Manage Osteoporosis and Fractures; National Council on Aging and Older People: Dublin, Ireland, 2008. [Google Scholar]
- RACGP. Osteoporosis Prevention, Diagnosis and Management in Postmenopausal Women and Men over 50 Years of Age; The Royal Australian College of General Practitioners and Osteoporosis Australia: East Melbourne, Australia, 2017. [Google Scholar]
- SIGN. Management of Osteoporosis and the Prevention of Fragility Fractures; A National Clinical Guideline; Scottish Intercollegiate Guidelines Network: Edinburgh, Scotland, 2020. [Google Scholar]
- Shojaa, M.; Von Stengel, S.; Kohl, M.; Schoene, D.; Kemmler, W. Effects of dynamic resistance exercise on bone mineral density in postmenopausal women: A systematic review and meta-analysis with special emphasis on exercise parameters. Osteoporos. Int. 2020, 31, 1427–1444. [Google Scholar] [CrossRef] [PubMed]
- Shojaa, M.; Von Stengel, S.; Schoene, D.; Kohl, M.; Barone, G.; Bragonzoni, L.; Dallolio, L.; Marini, S.; Murphy, M.H.; Stephenson, A.; et al. Effect of Exercise Training on Bone Mineral Density in Post-menopausal Women: A Systematic Review and Meta-Analysis of Intervention Studies. Front. Physiol. 2020, 11, 652. [Google Scholar] [CrossRef]
- Ashe, M.C.; dos Santos, I.K.; Edwards, N.Y.; Burnett, L.A.; Barnes, R.; Fleig, L.; Puyat, J.H.; Sale, J.E.M.; McKay, H.A.; Giangregorio, L.M. Physical Activity and Bone Health in Men: A Systematic Review and Meta-Analysis. J. Bone Metab. 2021, 28, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Bolam, K.A.; Van Uffelen, J.G.Z.; Taaffe, D.R. The effect of physical exercise on bone density in middle-aged and older men: A systematic review. Osteoporos. Int. 2013, 24, 2749–2762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamilton, B.R.; Staines, K.A.; Kelley, G.A.; Kelley, K.S.; Kohrt, W.M.; Pitsiladis, Y.; Guppy, F.M. The Effects of Exercise on Bone Mineral Density in Men: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Calcif. Tissue Int. 2021, 109, 1–16. [Google Scholar] [CrossRef]
- Kemmler, W.; Shojaa, M.; Kohl, M.; Von Stengel, S. Exercise effects on bone mineral density in older men: A systematic review with special emphasis on study interventions. Osteoporos. Int. 2018, 29, 1493–1504. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Jones, K.; Baker, K.; Speight, R.A.; Thompson, N.P.; Tew, G.A. Randomised clinical trial: Combined impact and resistance training in adults with stable Crohn’s disease. Aliment. Pharmacol. Ther. 2020, 52, 964–975. [Google Scholar] [CrossRef]
- Allison, S.; Folland, J.P.; Rennie, W.J.; Summers, G.D.; Brooke-Wavell, K. High impact exercise increased femoral neck bone mineral density in older men: A randomised unilateral intervention. Bone 2013, 53, 321–328. [Google Scholar] [CrossRef] [Green Version]
- Bolam, K.A.; Skinner, T.; Jenkins, D.; Taaffe, D. The Osteogenic Effect of Impact-Loading and Resistance Exercise on Bone Mineral Density in Middle-Aged and Older Men: A Pilot Study. Gerontology 2015, 62, 22–32. [Google Scholar] [CrossRef] [Green Version]
- Ghayomzadeh, M.; Earnest, C.P.; Hackett, D.; Alinaghi, S.S.; Navalta, J.W.; Gholami, M.; Rouzbahani, N.H.; Mohraz, M.; Voltarelli, F.A. Combination of resistance and aerobic exercise for six months improves bone mass and physical function in HIV infected individuals: A randomized controlled trial. Scand. J. Med. Sci. Sports 2021, 31, 720–732. [Google Scholar] [CrossRef]
- Harding, A.T.; Weeks, B.; Lambert, C.; Watson, S.L.; Weis, L.J.; Beck, B.R. A Comparison of Bone-Targeted Exercise Strategies to Reduce Fracture Risk in Middle-Aged and Older Men with Osteopenia and Osteoporosis: LIFTMOR-M Semi-Randomized Controlled Trial. J. Bone Miner. Res. 2020, 35, 1404–1414. [Google Scholar] [CrossRef]
- Helge, E.W.; Andersen, T.R.; Schmidt, J.F.; Jørgensen, N.R.; Hornstrup, T.; Krustrup, P.; Bangsbo, J. Recreational football improves bone mineral density and bone turnover marker profile in elderly men. Scand. J. Med. Sci. Sports 2014, 24, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Huuskonen, J.; Väisänen, S.B.; Kröger, H.; Jurvelin, J.S.; Alhava, E.; Rauramaa, R. Regular Physical Exercise and Bone Mineral Density: A Four-Year Controlled Randomized Trial in Middle-aged Men. The DNASCO Study. Osteoporos. Int. 2001, 12, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Kohl, M.; Jakob, F.; Engelke, K.; Von Stengel, S. Effects of High Intensity Dynamic Resistance Exercise and Whey Protein Supplements on Osteosarcopenia in Older Men with Low Bone and Muscle Mass. Final Results of the Randomized Controlled FrOST Study. Nutrients 2020, 12, 2341. [Google Scholar] [CrossRef] [PubMed]
- Kukuljan, S.; Nowson, C.; Sanders, K.; Nicholson, G.; Seibel, M.; Salmon, J.; Daly, R.M. Independent and Combined Effects of Calcium-Vitamin D3 and Exercise on Bone Structure and Strength in Older Men: An 18-Month Factorial Design Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2011, 96, 955–963. [Google Scholar] [CrossRef] [Green Version]
- Santa-Clara, H.; Fernhall, B.; Baptista, F.; Mendes, M.; Sardinha, L.B. Effect of a one-year combined exercise training program on body composition in men with coronary artery disease. Metababolism 2003, 52, 1413–1417. [Google Scholar] [CrossRef]
- Whiteford, J.; Ackland, T.R.; Dhaliwal, S.S.; James, A.P.; Woodhouse, J.J.; Price, R.; Prince, R.L.; Kerr, D.A. Effects of a 1-year randomized controlled trial of resistance training on lower limb bone and muscle structure and function in older men. Osteoporos. Int. 2010, 21, 1529–1536. [Google Scholar] [CrossRef] [Green Version]
- Woo, J.; Hong, A.; Lau, E.; Lynn, H. A randomised controlled trial of Tai Chi and resistance exercise on bone health, muscle strength and balance in community-living elderly people. Age Ageing 2007, 36, 262–268. [Google Scholar] [CrossRef] [Green Version]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro Scale for Rating Quality of Randomized Controlled Trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [CrossRef] [Green Version]
- Smart, N.A.; Waldron, M.; Ismail, H.; Giallauria, F.; Vigorito, C.; Cornelissen, V.; Dieberg, G. Validation of a new tool for the assessment of study quality and reporting in exercise training studies: TESTEX. Int. J. Evid. Based Healthc. 2015, 13, 9–18. [Google Scholar] [CrossRef] [PubMed]
- De Avila, V.R.; Bento, T.; Gomes, W.; Leitao, J.; De Sousa, N.F. Functional Outcomes and Quality of Life After Ankle Fracture Surgically Treated: A Systematic Review. J. Sport Rehabil. 2018, 27, 274–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Viechtbauer, W. Conducting Meta-Analyses in R with the metafor Package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef] [Green Version]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Doi, S.A.; Barendregt, J.J.; Khan, S.; Thalib, L.; Williams, G. Advances in the meta-analysis of heterogeneous clinical trials I: The inverse variance heterogeneity model. Contemp. Clin. Trials 2015, 45, 130–138. [Google Scholar] [CrossRef] [Green Version]
- Duval, S.; Tweedie, R. A Nonparametric “Trim and Fill” Method of Accounting for Publication Bias in Meta-Analysis. J. Am. Stat. Assoc. 2000, 95, 89. [Google Scholar] [CrossRef]
- Furuya-Kanamori, L.; Barendregt, J.J.; Doi, S.A. A new improved graphical and quantitative method for detecting bias in meta-analysis. Int. J. Evid. Based Health 2018, 16, 195–203. [Google Scholar] [CrossRef]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.C.; Stehle, P.; Teta, D.; et al. Evidence-Based Recommendations for Optimal Dietary Protein Intake in Older People: A Position Paper From the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Bjerre, E.D.; Brasso, K.; Jørgensen, A.B.; Petersen, T.H.; Eriksen, A.R.; Tolver, A.; Christensen, J.F.; Poulsen, M.H.; Madsen, S.S.; Østergren, P.B.; et al. Football Compared with Usual Care in Men with Prostate Cancer (FC Prostate Community Trial): A Pragmatic Multicentre Randomized Controlled Trial. Sports Med. 2019, 49, 145–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.H.; Seong, D.H.; Yoon, S.M.; Choi, Y.D.; Choi, E.; Song, Y.; Song, H. The Effect on Bone Outcomes of Home-based Exercise Intervention for Prostate Cancer Survivors Receiving Androgen Deprivation Therapy: A Pilot Randomized Controlled Trial. Cancer Nurs. 2018, 41, 379–388. [Google Scholar] [CrossRef]
- Uth, J.; Hornstrup, T.; Christensen, J.F.; Christensen, K.B.; Jørgensen, N.R.; Helge, E.W.; Schmidt, J.F.; Brasso, K.; Helge, J.; Jakobsen, M.D.; et al. Football training in men with prostate cancer undergoing androgen deprivation therapy: Activity profile and short-term skeletal and postural balance adaptations. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 116, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.J.; Koo, K.C. Pathophysiology of Bone Loss in Patients with Prostate Cancer Receiving Androgen-Deprivation Therapy and Lifestyle Modifications for the Management of Bone Health: A Comprehensive Review. Cancers 2020, 12, 1529. [Google Scholar] [CrossRef]
- McCartney, N.; Hicks, A.L.; Martin, J.; Webber, C.E. Long-term Resistance Training in the Elderly: Effects on Dynamic Strength, Exercise Capacity, Muscle, and Bone. J. Gerontol. Ser. A Boil. Sci. Med Sci. 1995, 50, B97–B104. [Google Scholar] [CrossRef]
- Kemmler, W. Meta-analysis and exercise related sports medicine [Meta-Analysen im trainingswissenschaftlichen und sportmedizinischen Spannungsfeld]. Dt. Ztschr. Sportmedizin. 2013, 64, 96–98. [Google Scholar]
- Kemmler, W.; Stengel, V. The Role of Exercise on Fracture Reduction and Bone Strengthening; Avademic Press: London, UK, 2019. [Google Scholar]
- Gomez-Bruton, A.; Gonzalez-Aguero, A.; Gómez-Cabello, A.; Casajús, J.A.; Vicente-Rodríguez, G. Is Bone Tissue Really Affected by Swimming? A Systematic Review. PLoS ONE 2013, 8, e70119. [Google Scholar] [CrossRef] [Green Version]
- Olmedillas, H.; Gonzalez-Aguero, A.; Moreno, L.A.; Casajus, J.A.; Vicente-Rodríguez, G. Cycling and bone health: A systematic review. BMC Med. 2012, 10, 168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- James, M.M.-S.; Carroll, S. Meta-analysis of walking for preservation of bone mineral density in postmenopausal women. Bone 2008, 43, 521–531. [Google Scholar] [CrossRef]
- James, M.M.-S.; Caroll, S. High intensity resistance training and postmenopausal bone loss: A meta-analysis. Osteoporos Int. 2006, 17, 1225–1240. [Google Scholar] [CrossRef] [PubMed]
- Furuya-Kanamori, L.; Thalib, L.; Barendregt, J. Meta-analysis in evidence-based healthcare: A paradigm shift away from random effects is overdue. Int. J. Evid. 2017, 15, 152–160. [Google Scholar]
- Shojaa, M.; Von Stengel, S.; Schoene, D.; Kohl, M.; Kemmler, W. Effect of different types of exercise on bone mineral density in postmenopausal women: A systematic review and meta-analysis. Calcif. Tissue Int. 2020, 107, 409–439. [Google Scholar] [CrossRef]
- Weineck, J. Optimales Training; Spitta: Erlangen, Germany, 2019. [Google Scholar]
- Gentil, P.; Arruda, A.; Souza, D.; Giessing, J.; Paoli, A.; Fisher, J.; Steele, J. Is There Any Practical Application of Meta-Analytical Results in Strength Training? Front. Physiol. 2017, 8, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Von Stengel, S.; Kemmler, W.; Lauber, D.; Weineck, J.; Kalender, W.A.; Engelke, K. Power Training is more Effective than Strength Training to Maintain Bone Mineral Density in Postmenopausal Woman. J. Appl. Physiol. 2005, 99, 181–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Souza, D.; Barbalho, M.; Ramirez-Campillo, R.; Martins, W.; Gentil, P. High and low-load resistance training produce similar effects on bone mineral density of middle-aged and older people: A systematic review with meta-analysis of randomized clinical trials. Exp. Gerontol. 2020, 138, 110973. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year, Origin (Country) | Initial Sample Size (n) | Drop Out, Loss to FU (%) | Age (Years) | BMI (kg/m²) | Health Status, Bone Status | Medication with Impact on Bone |
|---|---|---|---|---|---|---|
| Allison, 2013 UK [12] | EG: 50 | 30 b | 69.9 + 4.0 b | 26.2 + 2.3 b | Healthy, no BMD restriction | n.g |
| CG: 50 | ||||||
| Bolam, 2015 Australia [13] | HI-EG: 13 | 23 | 62.1 + 6.9 | 25.8 + 2.8 | Healthy, no osteoporosis | n.g |
| CG: 14 | 7 | 58.7 + 7.4 | 26.6 + 3.4 | |||
| Ghayomzadeh *, 2020 Iran [14] | EG: 10 | 10 | 36.2 + 6.7 | 26.5 + 3.3 | HIV-infected men, osteopenia or osteoporosis at hip and/or LS | No medication known to relevantly affect bone metabolism |
| CG: 10 | 5 | 38.3 + 5.6 | 25.2 + 3.0 | |||
| Harding, 2020 Australia [15] | HiRIT-EG: 34 | 12 | 64.9 + 8.6 | 27.2 + 3.5 | Healthy, osteopenia or osteoporosis at the hip and/or LS | Apart from 2 men in the HiRIT- and IAC-EG each, no medication known to relevantly affect bone metabolism |
| IAC-EG: 33 | 9 | 69.0 + 6.8 | 26.6 + 4.0 | |||
| CG: 26 | 19 | 67.4 + 6.3 | 26.3 + 2.8 | |||
| Helge, 2014 Denmark [16] | Soccer-EG: 9 | 0 | 68.0 + 4.0 | 26.1 + 3.9 | Healthy, no BMD restriction | n.g |
| RT-EG: 9 | 11 | 69.1 + 3.1 | 27.4 + 2.8 | |||
| CG: 8 | 25 | 67.4 + 2.7 | 27.9 + 4.6 | |||
| Huuskonen, 2001 Finland [17] | EG: 70 | 6 | 58.1 + 2.9 | 27.1 c | n.g, n.g. (probably healthy without BMD restrictions) | n.g |
| CG: 70 | 58.2 + 2.9 | 27.2 c | ||||
| Jones *, 2020 UK [11] | EG: 7 | 4 | 46.1 + 11.9 | 26.0 + 3.1 | Quiescent or mildly-active Crohns disease, no BMD restictrion | No medication known to relevantly affect bone metabolism |
| CG: 8 | 13 | 52.3 + 13.6 | 27.1 + 5.1 | |||
| Kemmler, 2020 Germany [18] | EG: 21 | 10 | 77.8 + 3.6 | 25.0 + 3.0 | Sarcopenia, osteopenia or osteo-porosis at the hip and/or LS | No medication known to relevantly affect bone metabolism |
| CG: 22 | 5 | 79.2 + 4.7 | 24.5 + 1.9 | |||
| Kukuljan, 2011 Australia [19] | EG: 46 | 4 | 60.7 + 7.1 | 28.1 + 3.3 | n.g., partially osteopenia or osteoporosis at the hip and/or LS | No medication known to relevantly affect bone metabolism |
| CG: 44 | 4 | 59.9 + 7.4 | 26.7 + 2.9 | |||
| Santa Clara, 2003 Portugal [20] | AE-EG: 13 | n.g | 57 + 11 | 28.1 + 4.2 | Coronary artery diseases, no BMD restriction | No medication known to relevantly affect bone metabolism |
| AE + RT-EG: 13 | 55 + 10 | 27.2 + 2.3 | ||||
| CG: 10 | 57 + 11 | 26.0 + 3.3 | ||||
| Whiteford, 2010 Australia [21] | RT-EG: 73 | 11 | 64 + 6 | 26.4 + 3.1 | Healthy, no osteoporosis | No medication known to relevantly affect bone metabolism |
| CG: 70 | 4 | 64 + 6 | 26.3 + 3.0 | |||
| Woo, 2007 a Hong Kong [22] | RT-EG: 30 | 2 | 68.6 + 3.0 | 24.1 + 3.4 | Healthy, no BMD restriction | n.g |
| TaiChi-EG: 30 | 68.2 + 2.4 | 23.6 + 3.4 | ||||
| CG: 30 | 68.1 + 2.7 | 23.9 + 3.1 |
| PEDro-Criteria | Additional TESTEX Criteria ¹ | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Author, Year | Eligibility Criteria | Random Allocation | Allocation Concealment | Inter Group Homogeneity | Blinding Subjects | Blinding Personnel | Blinding Assessors | Participation ≥ 85% | Intention to Treat Analysis ² | Between Group Comparison | Measure of Variability | Total Score PEDro | Adverse Effects Reported | Attendance Reported | Activity Monitoring in Control Groups | Relative Exercise Intensity Constant | Exercise Volume/Energy Expended | Total Score TESTEX |
| Allison et al. 2013 [12] | y | + | n.a. | + | - | - | + | - | + | + | + | 6 | + | + | + | n.a. | + | 11 |
| Bolam et al. 2016 [13] | y | + | + | + | - | - | - | + | + | + | + | 7 | + | + | - | + | + | 13 |
| Ghayomzadeh et al. 2020 [14] | y | + | - | + | - | - | + | + | + | + | + | 7 | + | + | - | + | + | 12 |
| Harding et al. 2020 [15] | y | + | + | - | - | - | + | + | + | + | + | 7 | + | + | + | + | + | 14 |
| Helge et al. 2014 [22] | y | + | - | + | - | - | - | + | - | + | + | 5 | - | + | - | + | + | 10 |
| Huuskonen et al. 2001 [17] | y | + | - | + | - | - | - | + | - | + | + | 5 | - | - | - | + | + | 8 |
| Jones et al. 2020 [11] | y | + | + | - | - | - | + | + | + | + | + | 7 | - | + | - | + | + | 13 |
| Kemmler et al. 2020 [18] | y | + | + | - | - | - | + | + | + | + | + | 7 | + | + | + | + | + | 14 |
| Kukuljan et al. 2011 [19] | y | + | + | + | - | - | - | + | + | + | + | 7 | - | + | + | + | + | 11 |
| Santa Clara et al. 2003 [20] | y | - | - | + | - | - | - | - | - | + | + | 3 | - | + | - | - | + | 7 |
| Whiteford et al. 2010 [21] | y | + | - | + | - | - | - | + | + | + | + | 6 | + | + | - | + | + | 12 |
| Woo et al. 2007 [22] | y | + | + | + | - | - | + | + | + | + | + | 8 | + | + | - | - | - | 10 |
| Author, Year [Ref] | Exercise Status | Study Length (Months) | Progression of Intensity? | Type of Exercise, Methods | Setting/ Supervision | Intervention, Exercise Composition | Attendance | Site Specificity |
|---|---|---|---|---|---|---|---|---|
| Allison, 2013 [12] | Untrained | 12 | Yes | Unilateral “hops” Unloaded leg: CG | IE/NS | Seven session/week, five sets of ten multi-directional, unilateral hops with peak GRF of ≈3x body weight; 15 s rest between sets | 91% | LS: Yes TH: Yes |
| Bolam, 2016 1 [13] | Untrained | 9 | Yes | DRT (upper body) on machines and with free weights; multi-directional jumps with high GRF | JE/S IE/NS | Four sessions/week: 2 × 60 min/week: jumping (see below) and upper body DRT with four exercises. Two sets of 12 reps at 60% 1RM; two jumping sessions/week with three exercises, two–four sets, 5–18 reps and GRF: 4.6–5.8x body weight; 1 min rest between sets High volume jumping group (HV): 80 jumps/session Low volume jumping group (LV): 40 jumps/session | HVJ:53% LVJ: 65% | LS: Yes TH: Yes |
| Ghayomzadeh, 2020 [14] | Untrained | 6 | Yes | DRT (all main muscle groups) on machines and with free weights; WBE: treadmill walking/running | JE/S | Three sessions/week, eight exercises; four–twenty reps at 60–85% 1RM (i.e., first session 80–85%; second session 60–80%; third session 50–65% 1RM); each session ≈23 min of walking/running at up to ≈70%HRmax | 85% | LS: Yes TH: Yes |
| Harding, 2020 [15] | No RT | 8 | Yes | DRT (deadlift, squat, and overhead press) and “jumping chin-ups” | JE/S | Two sessions/week; three exercises (deadlift, squat, and overhead press), five sets of five repetitions with 80–85% 1RM (RPE ≥ 16), five sets of five repetitions jumping chin-ups with “flat footed landing” | 78% | LS: Yes TH: Yes |
| Yes | Isometric-Axial-Comp-ression (IAC) at machines | JE/S | Two sessions/week, four exercises (chest press, leg press, core pull, vertical lift; bioDensity device), near-maximal 5-s isometric contraction (RPE ≥ 16) | 79% | LS: Yes TH: Yes | |||
| Helge, 2014 [16] | Not given | 12 | Yes | Soccer (on natural grass) | JE/S | Two–three sessions/week, four set ×15 min FB at 65–90% HRmax, 2 min rest between sets. | 66% | LS: Yes TH: Yes |
| Yes | DRT (all main muscle groups) on machines and with free weights | JE/S | Two–three sessions/week; five–seven exercises (leg press, leg extension, leg curl, pull-down, and lateral raises, lunges, seated row) four sets at 8RM (i.e., eight reps at ≈75% 1RM), explosive concentric movement | 73% | LS: Yes TH: Yes | |||
| Huuskonen, 2001 [17] | Not given | 48 | Yes | Brisk walking | IE/NS | Five sessions/week 60 min of brisk walking at 40–60% of VO2max (aerobic threshold pace) | Not given | LS: Yes TH: Yes |
| Jones, 2020 [11] | RT < 2 s/w. | 6 | Yes | DRT (all main muscle groups) with own body weight and elastic bands; rope skipping, multi-directional jumps | IE/ mainly NS | Three sessions/week, 5 min rope skipping, two–three sets of 10–15 reps of five different jumps (e.g., squat, broad, scissor jump); eight–ten RT exercises, two–three sets of 10–15 repetitions with “moderate-hard effort” (i.e., ≈65–75% 1RM) | 62% | LS: Yes TH: yes |
| Kemmler, 2020 [18] | RT ≤ 45 min /w. | 18 | Yes | DRT (all main muscle groups) on machines | JE/S | Two sessions/week, periodized single set RT with periods of high intensity (up to 85% 1RM), high effort (by RM, supersets, drop sets) and high velocity (explosive concentric movement) | 95% | LS: Yes TH: Yes |
| Kukuljan, 2011 [19] | Untrained | 18 | Yes | DRT (all main muscle groups) on machines and with free weights and jumps (IE) | JE/S IE/S | Three sessions/week, periodized RT with up to 85% 1RM and explosive velocity during the concentric phase (last 6 month), and two–three sets of different jumps with 20 reps with peak GRF of 1.5–9.7x body weight | 63% | LS: Yes TH: Yes |
| Santa Clara, 2003 [20] | Not given | 12 | Yes | WBE: walking/ running on treadmill | JE/n.g. | Three sessions/week 50 min treadmill walking/running at 60–70% HRR | 85% | LS: Yes TH: Yes |
| Yes | DRT (all main muscle groups) on machines and treadmill walking/running | JE/n.g. | Three sessions/week 30 min treadmill walking/running at 60–70% HRR and DRT: eight exercises, two sets of eight–twelve reps at 40–50% 1RM; 2 × 20 reps of abdominal exercises; 2 × 10 reps of back exercises (intensity n.g.) | 82% | LS: Yes TH:Yes | |||
| Whiteford, 2010 2 [21] | ≤2 s/w. ≤moderate intensity | 12 | Yes | DRT (all main muscle groups) on machines and with free weights | JE/S | Three sessions/week, 10 exercises, three sets at 8RM (i.e., eight reps at ≈75% 1RM) | 71% | LS: Yes TH: Yes |
| Woo, 2007 [22] | Untrained | 12 | No | Tai Chi (Yang Style) | n.g. | Three session/week, 24 Forms of Yang Style, intensity n.g. | 81% | LS: ? TH: yes |
| No | DRT with elastic bands | n.g. | Three sessions/week, six exercises (arm lifting, hip abduction, heel raise, hip flexion, extension, ankle dorsiflexion), 30 reps with an elastic band of low–moderate strength; intensity n.g. (presumably low) | 76% | LS: Yes TH: Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mages, M.; Shojaa, M.; Kohl, M.; von Stengel, S.; Becker, C.; Gosch, M.; Jakob, F.; Kerschan-Schindl, K.; Kladny, B.; Klöckner, N.; et al. Exercise Effects on Bone Mineral Density in Men. Nutrients 2021, 13, 4244. https://doi.org/10.3390/nu13124244
Mages M, Shojaa M, Kohl M, von Stengel S, Becker C, Gosch M, Jakob F, Kerschan-Schindl K, Kladny B, Klöckner N, et al. Exercise Effects on Bone Mineral Density in Men. Nutrients. 2021; 13(12):4244. https://doi.org/10.3390/nu13124244
Chicago/Turabian StyleMages, Michelle, Mahdieh Shojaa, Matthias Kohl, Simon von Stengel, Clemens Becker, Markus Gosch, Franz Jakob, Katharina Kerschan-Schindl, Bernd Kladny, Nicole Klöckner, and et al. 2021. "Exercise Effects on Bone Mineral Density in Men" Nutrients 13, no. 12: 4244. https://doi.org/10.3390/nu13124244
APA StyleMages, M., Shojaa, M., Kohl, M., von Stengel, S., Becker, C., Gosch, M., Jakob, F., Kerschan-Schindl, K., Kladny, B., Klöckner, N., Lange, U., Middeldorf, S., Peters, S., Schoene, D., Sieber, C. C., Tholen, R., Thomasius, F. E., Uder, M., & Kemmler, W. (2021). Exercise Effects on Bone Mineral Density in Men. Nutrients, 13(12), 4244. https://doi.org/10.3390/nu13124244








