Latin American Considerations for Infant and Young Child Formulae
Abstract
1. Introduction
2. Methods
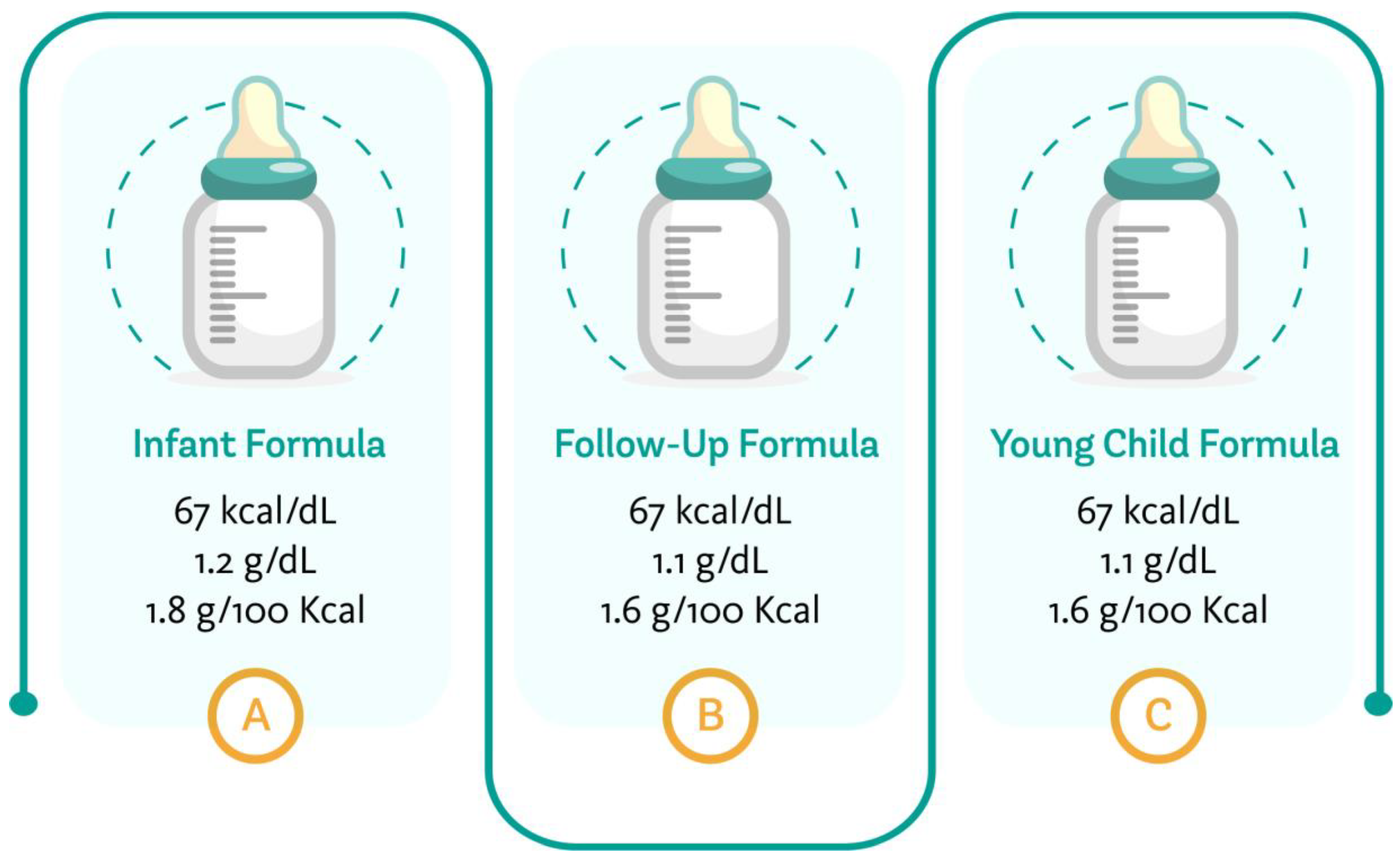
3. Infant Formulae
4. Follow-Up Formulae
5. Young Child Formula
6. Presentation, Storage and Preparation of Formulae
7. Marketing of Formulae for Infants and Young Children
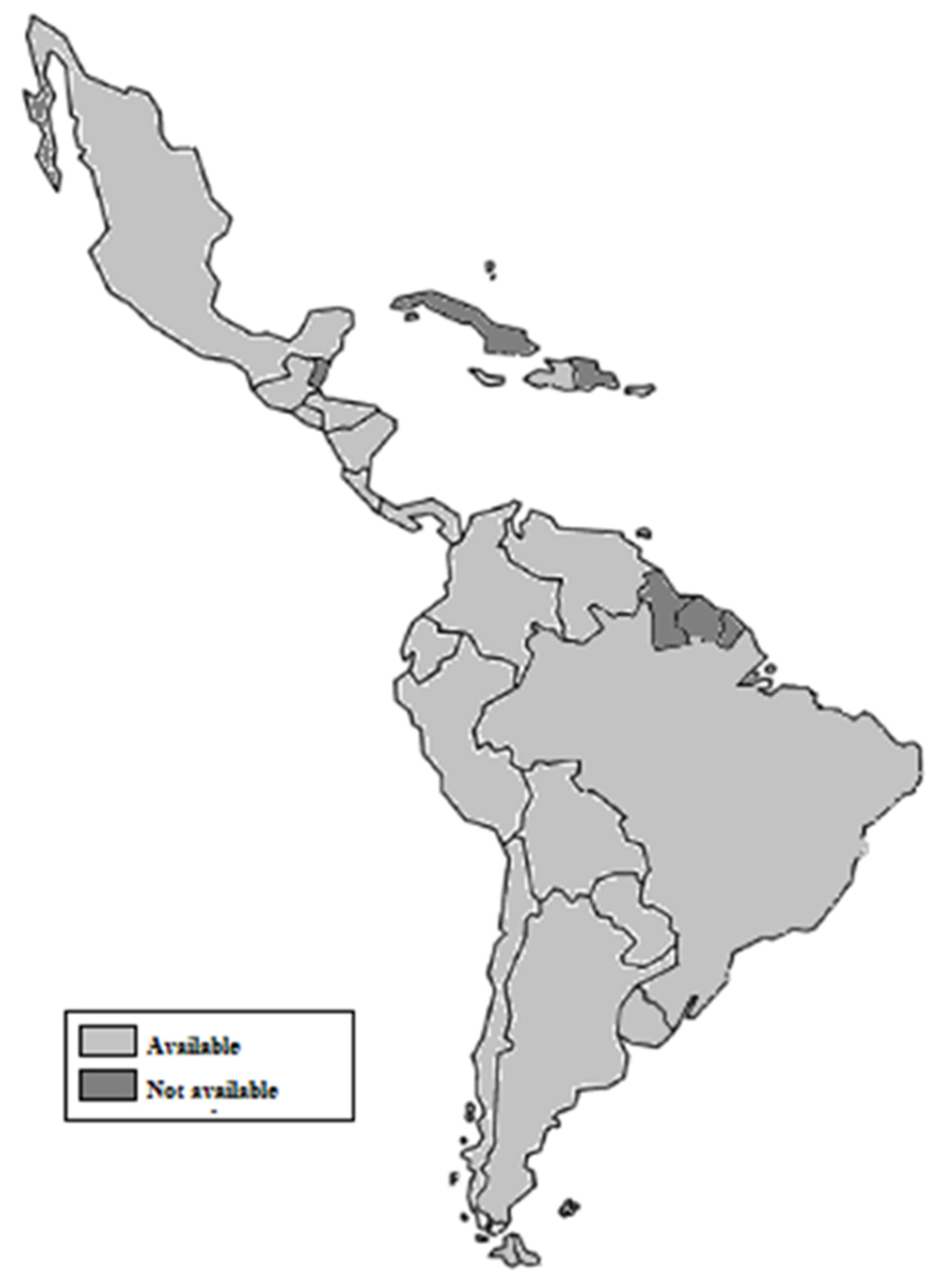
7.1. Regulations on Marketing of Infant Formula
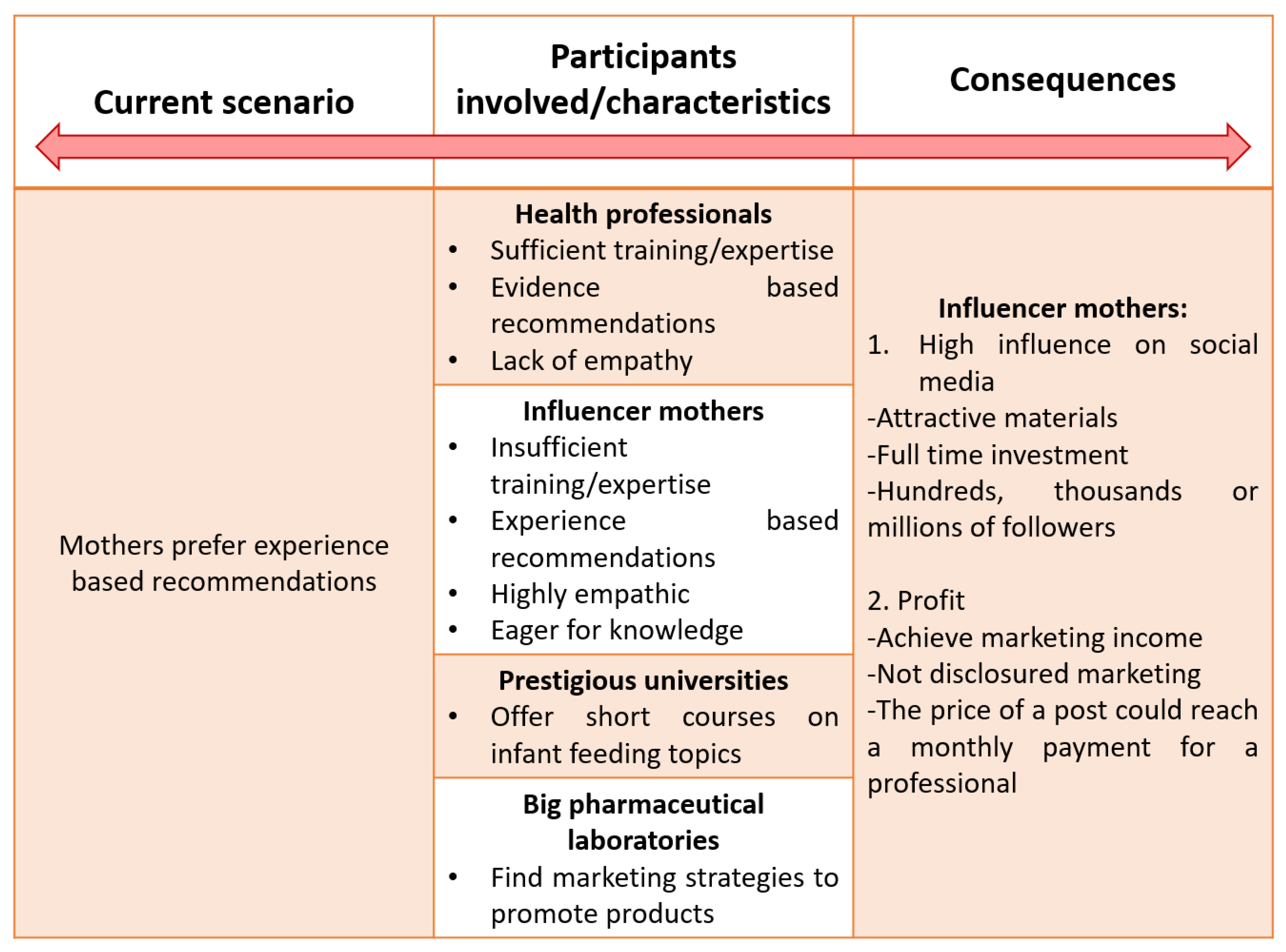
7.2. New Marketing Strategies
7.3. The Role of Influencers
8. Feeding and Infant Health
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Essential Nutrition Actions: Improving Maternal, Newborn, Infant and Young Child Health and Nutrition; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific Panel on Nutrition, Novel Foods and Food allergen. In Minutes of the 43rd Meeting of the Working Group on Food Allergies; EFSA: Parma, Italy, 2019. [Google Scholar]
- Koletzko, B. Formula Feeding. World Rev. Nutr. Diet. 2015, 113, 97–103. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. Standard for Infant Formulas and Formulas for Medical PURPOSES Intended for Infants; Report No.: CXS 72–1981; Codex Alimentarius Commission: Rome, Italy, 2007. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific Opinion on the Essential Composition of Infant and Follow-On Formulae; EFSA: Parma, Italy, 2014. [Google Scholar]
- European Union. Commission Delegated Regulation (EU) 2016/127 of 25 September 2015 Supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as Regards the Specific Compositional and Information Requirements for Infant Formula and Follow-On Formula and as Regards Requirements on Information Relating to Infant and Young Child Feeding; Official Journal of the European Union; European Union: Maastricht, The Netherlands, 2016; p. L25. [Google Scholar]
- Federal Food and Drugs Administration. CA4T2. Code of Federal Regulations; Federal Food and Drugs Administration: Silver Spring, MD, USA, 2014; pp. 106–107.
- American Academy of Pediatrics Committee on Nutrition. Formula Feeding of Term Infants. In Pediatric Nutrition, 8th ed.; Kleinman, R.E., Ed.; American Academy of Pediatrics Committee on Nutrition: Itasca, IL, USA, 2019; pp. 79–104. [Google Scholar]
- Koletzko, B.; Baker, S.; Cleghorn, G.; Neto, U.F.; Gopalan, S.; Hernell, O.; Hock, Q.S.; Jirapinyo, P.; Lonnerdal, B.; Pencharz, P.; et al. Global standard for the composition of infant formula: Recommendations of an ESPGHAN coordinated international expert group. J. Pediatr. Gastroenterol. Nutr. 2005, 41, 584–599. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, B.; Demmelmair, H.; Grote, V.; Totzauer, M. Optimized protein intakes in term infants support physiological growth and promote long-term health. Semin. Perinatol. 2019, 43, 151153. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.; Grote, V.; Closa-Monasterolo, R.; Escribano, J.; Langhendries, J.P.; Dain, E.; Giovannini, M.; Verduci, E.; Gruszfeld, D.; Socha, P.; et al. Lower protein content in infant formula reduces BMI and obesity risk at school age: Follow-up of a randomized trial. Am. J. Clin. Nutr. 2014, 99, 1041–1051. [Google Scholar] [CrossRef]
- Francavilla, R.; Calasso, M.; Calace, L.; Siragusa, S.; Ndagijimana, M.; Vernocchi, P.; Brunetti, L.; Mancino, G.; Tedeschi, G.; Guerzoni, E.; et al. Effect of lactose on gut microbiota and metabolome of infants with cow’s milk allergy. Pediatric Allergy Immunol. 2012, 23, 420–427. [Google Scholar] [CrossRef]
- Szlagyi, A. Review article: Lactose—A potential prebiotic. Aliment. Pharmacol. Ther. 2002, 16, 1591–1602. [Google Scholar] [CrossRef]
- Koletzko, B. Human Milk Lipids. Ann. Nutr. Metab. 2016, 69 (Suppl. 2), 28–40. [Google Scholar] [CrossRef]
- Koletzko, B.; Bergmann, K.; Brenna, J.T.; Calder, P.C.; Campoy, C.; Clandinin, M.T.; Colombo, J.; Daly, M.; Decsi, T.; Demmelmair, H.; et al. Should formula for infants provide arachidonic acid along with DHA? A position paper of the European Academy of Paediatrics and the Child Health Foundation. Am. J. Clin. Nutr. 2020, 111, 10–16. [Google Scholar] [PubMed]
- Koletzko, B.V.; Shamir, R. Infant formula: Does one size fit all? Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 205–207. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius. CODEX Standard Follow-Up Formula 156–1987; Codex Alimentarius: Berlin, Germany, 2013; pp. 1–44. Available online: https://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCXS%2B156-1987%252FCXS_156e.pdf (accessed on 3 May 2021).
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA); Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; et al. Scientific Opinion on the safety and suitability for use by infants of follow-on formulae with a protein content of at least 1.6 g/100 kcal. EFSA J. 2017, 11, 15. [Google Scholar]
- Koletzko, B. Why breastfeeding? In Breastfeeding and Breast Milk—From Biochemistry to Impact. Stuttgart, Family-Larsson-Rosenquist-Foundation; Thieme: New York, NY, USA, 2018; pp. 78–89. [Google Scholar]
- Koletzko, B.; Bhutta, Z.A.; Cai, W.; Cruchet, S.; El Guindi, M.; Fuchs, G.J.; Goddard, E.A.; van Goudoever, J.B.; Quak, S.H.; Kulkarni, B.; et al. Compositional requirements of follow-up formula for use in infancy: Recommendations of an international expert group coordinated by the Early Nutrition Academy. Ann. Nutr. Metab. 2013, 62, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Suthutvoravut, U.; Abiodun, P.O.; Chomtho, S.; Chongviriyaphan, N.; Cruchet, S.; Davies, P.S.; Fuchs, G.J.; Gopalan, S.; van Goudoever, J.B.; Nel Ede, L.; et al. Composition of Follow-Up Formula for Young Children Aged 12–36 Months: Recommendations of an International Expert Group Coordinated by the Nutrition Association of Thailand and the Early Nutrition Academy. Ann. Nutr. Metab. 2015, 67, 119–132. [Google Scholar] [CrossRef] [PubMed]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies. Scientific Opinion on nutrient requirements and dietary intakes of infants and young children in the European Union. EFSA J. 2013, 11, 3408. [Google Scholar]
- Hojsak, I.; Bronsky, J.; Campoy, C.; Domellöf, M.; Embleton, N.; Fidler Mis, N.; Hulst, J.; Indrio, F.; Lapillonne, A.; Mølgaard, C.; et al. Young Child Formula: A Position Paper by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2018, 66, 177–185. [Google Scholar] [CrossRef]
- UNICEF. From the First Hour of Life; UNICEF: New York, NY, USA, 2016. [Google Scholar]
- Dipasquale, V.; Serra, G.; Corsello, G.; Romano, C. Standard and Specialized Infant Formulas in Europe: Making, Marketing, and Health Outcomes. Nutr. Clin. Pract. 2020, 35, 273–281. [Google Scholar] [CrossRef]
- World Health Organization; Food and Agriculture Organization of the United Nations. Safe Preparation, Storage and Handling of Powdered Infant Formula Guidelines; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- FAO-OMS; Organización de las Naciones Unidas para la Agricultura y la Alimentación. Directrices, Preparación, Almacenamiento y Manipulación en Condiciones Higiénicas de Sucedáneos en Polvo para Lactantes en Entornos Asistenciales; OMS Ediciones: Geneva, Switzerland, 2007. [Google Scholar]
- Luque, V.; Escribano, J.; Mendez-Riera, G.; Schiess, S.; Koletzko, B.; Verduci, E.; Stolarczyk, A.; Martin, F.; Closa-Monasterolo, R. Methodological approaches for dietary intake assessment in formula-fed infants. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 320–327. [Google Scholar] [CrossRef]
- Márquez, M.; Ladino, L. Menos polvo, más volumen: Reducción en los costos de preparación de las fórmulas lácteas infantiles de inicio. Rev. Salud Bosque 2017, 7, 43–48. [Google Scholar] [CrossRef][Green Version]
- Ladino, L.; Velásquez, O. Características e Indicaciones de las Fórmulas Lácteas. In Nutridatos Manual de Nutrición Clínica, 3rd ed.; Health Book’s: Medellín, Colombia, 2021; pp. 283–296. [Google Scholar]
- Agostoni, C.; Axelsson, I.; Goulet, O.; Koletzko, B.; Michaelsen, K.F.; Puntis, J.W.; Rigo, J.; Shamir, R.; Szajewska, H.; Turck, D.; et al. Preparation and handling of powdered infant formula: A commentary by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 320–322. [Google Scholar] [CrossRef] [PubMed]
- World-Health-Assembly. WHA Resolution 34.22. International Code of Marketing of Breast-Milk Substitutes; World Health Organisation: Geneva, Switzerland, 1981. [Google Scholar]
- World Health Organization. The International Code of Marketing of Breast-Milk Substitutes: Frequently Asked Questions on the Roles and Responsibilities of Health Workers; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Bognar, Z.; De Luca, D.; Domellöf, M.; Hadjipanayis, A.; Haffner, D.; Johnson, M.; Kolacek, S.; Koletzko, B.; Saenz de Pipaon, M.; Shingadia, D.; et al. Promoting Breastfeeding and Interaction of Pediatric Associations with Providers of Nutritional Products. Front. Pediatr. 2020, 25, 562870. [Google Scholar] [CrossRef]
- Ministerio de Salud Pública de Ecuador. Ley 101/1995 Ley de Fomento Apoyo y Protección a la lactancia Materna. Available online: https://www.salud.gob.ec/wp-content/uploads/downloads/2014/02/Ley-de-LM-Reglamento.pdf (accessed on 5 May 2021).
- SITEAL UNESCO. Decreto Supremo 009/2006, Reglamento de Alimendación Infantil. Available online: https://siteal.iiep.unesco.org/sites/default/files/sit_accion_files/siteal_peru_0119.pdf (accessed on 5 May 2021).
- Invima. Decreto Número 1397 de 1992. Available online: https://www.invima.gov.co/documents/20143/441790/decreto_1397_1992.pdf/29a2e93f-068d-04c8–8f83-fd2a05ca5b54 (accessed on 5 May 2021).
- Ministerio de Salud de Nicaragua. Ley 295 Ley de Promoción, Protección y Mantenimiento de la Lactancia Materna y Regulación de la Comercialización de Sucedáneos de la Leche Materna. Available online: https://docs.bvsalud.org/leisref/2018/03/276/ley_no_295.pdf (accessed on 5 May 2021).
- República de Panamá Órgano Judicial. Ley No. 50 del 23 de Noviembre de 1995 Protege y Fomenta la Lactancia Materna. Available online: https://www.organojudicial.gob.pa/uploads/wp_repo/blogs.dir/cendoj/ley-50-de-1995-nov-27-1995.pdf (accessed on 5 May 2021).
- World Health Organization. Policy Decreto Ejecutivo No.1457 (de 30 de Octubre de 2012) que Reglamenta la Ley No.50 de 23 de Noviembre de 1995, por la cual se Protege y Fomenta la Lactancia Materna. Available online: https://extranet.who.int/nutrition/gina/es/node/15037 (accessed on 5 May 2021).
- Diario Oficial de la Federación de México. Proyecto de Norma Oficial Méxicana 050/2018 del 2 de Mayo. Available online: https://www.dof.gob.mx/nota_detalle.php?codigo=5521251&fecha=02/05/2018 (accessed on 5 May 2021).
- Gobierno de México. NORMA Oficial Mexicana NOM-131-SSA1-2012, Productos y Servicios. Fórmulas para Lactantes, de Continuación y para Necesidades Especiales de Nutrición. Alimentos y Bebidas no Alcohólicas para Lactantes y Niños de Corta Edad. Disposiciones y Especificaciones Sanitarias y Nutrimentales. Etiquetado y Métodos de Prueba. Available online: http://sitios1.dif.gob.mx/alimentacion/docs/NOM-131-SSA1-2012Formulas_y_alimentos%20para%20lactantes.pdf (accessed on 5 May 2021).
- SITEAL UNESCO. Ley Nº 8 (y su Decreto Reglamentario Nº 31/1996)/1995. Declara Como Prioridad Nacional la Promoción y Fomento de la Lactancia Materna. Available online: https://siteal.iiep.unesco.org/sites/default/files/sit_accion_files/do_0323.pdf (accessed on 5 May 2021).
- Reglamento para Aplicación de la ley que Declara Como Prioridad Nacional la Promoción y Fomento de la Lactancia Materna. Available online: https://www.resumendesalud.net/images/Documentos/Decreto-142-18.pdf (accessed on 5 May 2021).
- Gobierno de Argentina. Código Alimentario Argentino Capítulo XVIII Alimentos de Regimen o Dietéticos. Available online: https://www.argentina.gob.ar/sites/default/files/anmat-capitulo_xvii_dieteticosactualiz_2018-12.pdf (accessed on 5 May 2021).
- Argentina. Ley 26.873/2013 del 5 de Agosto, Lactancia Materna Promoción y Conscientización Pública. Available online: https://oig.cepal.org/sites/default/files/2013_ley26873_arg.pdf (accessed on 5 May 2021).
- Biblioteca del Congreso Nacional de Chile. Decreto 977 Aprueba Reglamento Sanitario de Alimentos, Titulo XXVIII De los Alimentos para Regímenes Especiales, Párrafo II, Artículo 497. Available online: https://www.bcn.cl/leychile/navegar?idNorma=71271 (accessed on 5 May 2021).
- Biblioteca del Congreso Nacional de Chile. Ley 21155 Establece Medidas de Protección a la Lactancia Materna y su Ejercicio. Available online: https://www.bcn.cl/leychile/navegar?idNorma=1131064 (accessed on 5 May 2021).
- Observatorio de Igualdad de Género CEPAL. Ley de Fomento a la Lactancia Materna y Comercialización de sus Sucedáneos. Available online: https://oig.cepal.org/sites/default/files/2006_ley3460_bol.pdf (accessed on 5 May 2021).
- Legal Office FAO LEX. Ley de Comercialiacion de los Sucedaneos de la Leche Materna y su Reglamento Decreto ley Numero 66–83. Available online: http://extwprlegs1.fao.org/docs/pdf/gua200153.pdf (accessed on 5 May 2021).
- SITEAL UNESCO. Decreto Legislativo Nº 404/2013. Ley de Promoción, Protección y Apoyo a la Lactancia Materna. Available online: https://siteal.iiep.unesco.org/sites/default/files/sit_accion_files/sv_0185.pdf (accessed on 5 May 2021).
- Biblioteca de Archivo Central del Congreso de la Nación. Ley Nº 1478/Comercialización de Sucedáneos de la Leche Materna. Available online: https://www.bacn.gov.py/leyes-paraguayas/5345/comercializacion-de-sucedaneos-de-la-leche-materna (accessed on 5 May 2021).
- Ministerio de Salud de Costa Rica. Ley 7430 de Fomento a la Lactancia Materna. Available online: https://www.ministeriodesalud.go.cr/gestores_en_salud/lactancia/legislacion/CLM_ley_7430_fomento_lactancia_materna.pdf (accessed on 5 May 2021).
- SITEAL UNESCO. Decreto 231/2013 Ley de Fomento y Protección a la Lactancia Materna. Available online: https://siteal.iiep.unesco.org/sites/default/files/sit_accion_files/hn_6026.pdf (accessed on 5 May 2021).
- Planalto. Decreto nº 9.579 de Novembro, do Lactente, da Criança e do Adolescente e do Aprendiz, e Sobre o Conselho Nacional dos Direitos da Criança e do Adolescente, o Fundo Nacional para a Criança e o Adolescente e os Programas Federais da Criança e do Adolescente, e dá Outras Providências. Available online: https://www.planalto.gov.br/ccivil_03/_ato2015-2018/2018/decreto/d9579.htm (accessed on 27 May 2021).
- República Bolivariana de Venezuela. Ley de Promocion, Protección y Apoyo a la Lactancia Materna/2007 del 12 de Julio. Available online: http://www.ilo.org/dyn/travail/docs/1911/breastfeeding-Gace- (accessed on 5 May 2021).
- Comisión de Salud Pública y Asistencia Social Repartido nº 702 Junio de 2017 Carpeta nº 2062 de 2017 Comercialización de Sucedáneos de Leche Materna. Available online: https://1library.co/document/ye86d5ey-comision-publica-asistencia-repartido-carpeta-comercializacion-sucedaneos-materna.html (accessed on 27 May 2021).
- Oxford Dictionary. Influencer. Available online: https://www.oxfordlearnersdictionaries.com/definition/english/influencer (accessed on 5 May 2021).
- Coates, A.E.; Hardman, C.A.; Halford, J.C.G.; Christiansen, P.; Boyland, E.J. Social Media Influencer Marketing and Children’s Food Intake: A Randomized Trial. Pediatrics 2019, 143, e20182554. [Google Scholar] [CrossRef]
- American Academy of Pediatrics Committee on Nutrition. Complementary Feeding. In Pediatric Nutrition, 7th ed.; Kleinman, R.E., Greer, F.R., Eds.; American Academy of Pediatrics: Itasca, IL, USA, 2013; pp. 123–134. [Google Scholar]
- McInnes, R.J.; Chambers, J.A. Supporting breastfeeding mothers: Qualitative synthesis. J Adv Nurs. 2008, 62, 407–427. [Google Scholar] [CrossRef] [PubMed]
- WHO. Complementary Feeding. Available online: https://www.who.int/health-topics/complementary-feeding#tab=tab_1 (accessed on 15 July 2021).
- The Long-Term Effects of Breastfeeding: A Systematic Review; World Health Organization: Geneva, Switzerland, 2013.
- Fan, Y.; Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 2021, 19, 55–71. [Google Scholar] [CrossRef]
- Chen, L.; Garmaeva, S.; Zhernakova, A.; Fu, J.; Wijmenga, C. A system biology perspective on environment-host-microbe interactions. Hum. Mol. Genet. 2018, 27, R187–R194. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, M.; Bronsky, J.; Campoy, C.; Domellöf, M.; Embleton, N.; Fidler Mis, N.; Hojsak, I.; Hulst, J.M.; Indrio, F.; Lapillonne, A.; et al. Complementary Feeding: A Position Paper by the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Ladino, L. Nutrición Desde la Preconcepción Hasta la Etapa Preescolar. In Alimentación y Nutrición Aplicada; Rodríguez, G., Ed.; Editorial Universidad El Bosque: Bogotá, Colombia, 2018; pp. 406–421. Available online: https://repositorio.unbosque.edu.co/bitstream/handle/20.500.12495/3359/9789587391350.pdf?sequence=1&isAllowed=y (accessed on 5 May 2021).
- Rytter, M.J.H.; Kolte, L.; Briend, A.; Friis, H.; Christensen, V.B. The Immune System in Children with Malnutrition—A Systematic Review. PLoS ONE 2014, 9, e105017. [Google Scholar] [CrossRef]
- UNICEF DATA. Diarrhoea. 2021. Available online: https://data.unicef.org/topic/child-health/diarrhoeal-disease/ (accessed on 15 July 2021).
- UNICEF DATA. Pneumonia. 2021. Available online: https://data.unicef.org/topic/child-health/pneumonia/ (accessed on 15 July 2021).
- UNICEF. Pneumonia and Diarrhoea Tackling the Deadliest Diseases for the World’s Poorest Children; UNICEF: New York, NY, USA, 2012. [Google Scholar]
| Component/Unit | Mínimum | Máximum |
|---|---|---|
| Energy kcal/100 mL | 60 | 70 |
| Proteins g/100 kcal | ||
| Cow’s milk protein | 1.8 | 3 |
| Soy protein isolates | 2.25 | 3 |
| Hydrolyzed cow’s milk protein | 1.8 | 3 |
| Fats | ||
| Total fat, g/100 kcal | 4.4 | 6 |
| Linoleic acid, g/100 kcal | 0.3 | 1.2 |
| α-Linolenic acid, mg/100 kcal | 50 | n.s. |
| Ratio linoleic/α-Linolenic acids | 5:1 | 15:1 |
| Lauric + myristic acids, % of fat | n.s. | 20 |
| Trans fatty acids, % of fat | n.s. | 3 |
| Erucic acids, % of fat | n.s. | 1 |
| Carbohydrates | ||
| Total carbohydrates, g/100 kcal | 9 | 14 |
| Vitamins | ||
| Vitamin A, µg RE/100 kcal | 60 | 180 |
| Vitamin D3, µg/100 kcal | 1 | 2.5 |
| Vitamin E, mg α-TE/100 kcal | 0.5 | 5 |
| Vitamin K, µg/100 kcal | 4 | 25 |
| Thiamin, µg/100 kcal | 60 | 300 |
| Riboflavin, µg/100 kcal | 80 | 400 |
| Niacin, µg/100 kcal | 300 | 1,500 |
| Vitamin B6, µg/100 kcal | 35 | 175 |
| Vitamin B12, µg/100 kcal | 0.1 | 0.5 |
| Pantothenic acid, µg/100 kcal | 400 | 2,000 |
| Folic acid, µg/100 kcal | 10 | 50 |
| Vitamin C, mg/100 kcal | 10 | 30 |
| Biotin, µg/100 kcal | 1.5 | 7.5 |
| Mineral trace elements | ||
| Iron (formula based on cow’s milk protein and protein hydrolysate mg/100 kcal) | 0.3 | 1.3 |
| Iron (formula based on soy protein isolate) mg/100 kcal | 0.45 | 2 |
| Calcium mg/100 kcal | 50 | 140 |
| Phosphorus (formula based on cow’s milk protein and protein hydrolysate, mg/100 kcal) | 25 | 90 |
| Phosphorus (formula based on soy protein isolate) mg/100 kcal | 30 | 100 |
| Ratio calcium/phosphorus mg/mg | 1:1 | 2:1 |
| Magnesium mg/100 kcal | 5 | 15 |
| Sodium mg/100 kcal | 20 | 60 |
| Chloride mg/100 kcal | 50 | 160 |
| Potassium mg/100 kcal | 60 | 160 |
| Manganese µg/100 kcal | 1 | 50 |
| Fluoride µg/100 kcal | n.s. | 60 |
| Iodine µg/100 kcal | 10 | 50 |
| Selenium µg/100 kcal | 1 | 9 |
| Copper µg/100 kcal | 35 | 80 |
| Zinc mg/100 kcal | 0.5 | 1.5 |
| Other substances | ||
| Choline mg/100 kcal | 7 | 50 |
| Myo-inositol mg/100 kcal | 4 | 40 |
| L-Carnitine mg/100 kcal | 1.2 | n.s. |
| 100 mL | 100 Kcal | |
|---|---|---|
| Kcal | 60–70 | 100 |
| Protein g | 1.1–2.1 | 1.8–3.0 |
| Fat g | 2.6–4.2 | 4.4–6.0 |
| Carbohydrates g | 5.4–9.8 | 9.0–14.0 |
| Component/Unit | Minimum | Maximum | Guidance Upper Level |
|---|---|---|---|
| Energy kcal/100 mL | 60 | 70 | |
| Proteins | |||
| Cow’s milk protein g/100 kcal | 1.7 | 2.5 | |
| Soy protein isolates g/100 kcal | 2.1 | 2.5 | |
| Fats | |||
| Total fat, g/100 kcal | 4.4 | 6.0 | |
| Linoleic acid, g/100 kcal | 0.3 | 1.4 | |
| α-Linolenic acid, mg/100 kcal | 50 | n.s. | |
| Ratio linoleic/α-Linolenic acids | 5:1 | 15:1 | |
| Lauric + myristic acids, % of fat | n.s. | 20 | |
| Trans fatty acids, % of fat | n.s. | 3 | |
| Erucic acids, % of fat | n.s. | 1 | |
| Phospholipids mg/100 kcal | 550 | ||
| Carbohydrates | |||
| Total carbohydrates | 9 | 14 | |
| Vitamins | |||
| Vitamin A, µg RE/100 kcal | 60 | 180 | |
| Vitamin D3, µg/100 kcal | 1 | 4.5 | |
| Vitamin E, mg α-TE/100 kcal | 0.5 | 5 | |
| Vitamin K, µg/100 kcal | 4 | 27 | |
| Thiamin, µg/100 kcal | 60 | 300 | |
| Riboflavin, µg/100 kcal | 80 | 500 | |
| Niacin, µg/100 kcal | 300 | 1,500 | |
| Vitamin B6, µg/100 kcal | 35 | 175 | |
| Vitamin B12, µg/100 kcal | 0.1 | 1.5 | |
| Folic acid, µg/100 kcal | 10 | 50 | |
| Pantothenic acid, µg/100 kcal | 400 | 2000 | |
| Vitamin C, mg/100 kcal | 10 | 70 | |
| Biotin, µg/100 kcal | 1.5 | 10 | |
| Minerals and trace elements | |||
| Iron (formula based on cow’s milk protein), mg/100 kcal | 1.1 | 1.9 | |
| Iron (formula based on soy protein isolates), mg/100 kcal | 1.3 | 2.5 | |
| Calcium, mg/100 kcal | 50 | 180 | |
| Phosphorus, mg/100 kcal | 25 | n.s. | |
| Magnesium, mg/100 kcal | 5 | 15 | |
| Sodium, mg/100 kcal | 20 | 60 | |
| Chloride, mg/100 kcal | 50 | 160 | |
| Potassium, mg/100 kcal | 60 | 180 | |
| Manganese, µg/100 kcal | 100 | ||
| Iodine, µg/100 kcal | 10 | 60 | |
| Selenium, µg/100 kcal | 1 | 9 | |
| Copper, µg/100 kcal | 35 | 250 | |
| Zinc, mg/100 kcal | 0.5 | 1.5 | |
| Other substances | |||
| Choline, mg/100 kcal | 7 | 150 | |
| L-Carnitine, mg/100 kcal | 1.2 | n.s. | |
| Taurine, mg/100 kcal | n.s. | 12 | |
| Total added nucleotides, mg/100 kcal | 0 | 10.8 |
| Component | Minimum | Maximum | Guidance Upper Level |
|---|---|---|---|
| Energy, kcal/100 mL | 45 | 70 | |
| Proteins g/100 kcal | |||
| Cow’s milk protein, g/100 kcal | 1.6 | 2.7 | |
| Soy protein isolate, g/100 kcal | 2 | 2.7 | |
| Fats | |||
| Total fat. g/100 kcal | 4.4 | 6.0 | |
| Linoleic acid, mg/100 kcal | 500 | NS | |
| Alpha-linolenic acid, mg/100 kcal | 50 | NS | |
| Trans fatty acids, % of fat | NS | 2 | |
| DHA 1, % of fat | 0.3 | NS | |
| Carbohydrates | |||
| Total carbohydrates, g/100 kcal | 9 | 14 | |
| Vitamins | |||
| Vitamin A 2, mcg RE/100 kcal | 60 | 180 | |
| Vitamin D, mcg/100 kcal | 1.5 | 4.5 | |
| Vitamin B12, mcg/100 kcal | 0.15 | 0.75 | |
| Folic acid, mcg/100 kcal | 20 | 100 | |
| Vitamin C, mg/100 kcal | 4.5 | 22.5 | |
| Minerals and trace elements | |||
| Iron 3 (formula based on cow’s milk protein), mg/100 kcal | 1 | 3 | |
| Iron (formula based on soy protein isolates), mg/100 kcal | NS | NS | |
| Calcium, mg/100 kcal | 200 | NS | |
| Sodium, mg/100 kcal | 25 | 75 | |
| Iodine, mcg/100 kcal | 12 | 36 | |
| Zinc, mg/100 kcal | 0.6 | 1.8 |
| Water (mL/ounces) | Powder (g) | Displaced Water (mL *) | Final Volume (mL) | Powder Energy (kcal) |
|---|---|---|---|---|
| 30/1 | 4.3 | 3.3 | 33 | 22 |
| 60/2 | 8.6 | 6.6 | 66 | 44 |
| 90/3 | 12.9 | 9.9 | 99 | 66 |
| Country | Year | Name and Number | Institution |
|---|---|---|---|
| Ecuador [35] | 1995 | Law 101, Official Register No. 814 | Health Ministry |
| Perú [36] | 2006 | Supreme Decrete N 009 | Health Ministry |
| Colombia [37] | 1992 | Decrete 1397 | Health Ministry |
| Nicaragua [38] | 1999 | Law 295 | Health Ministry |
| Panamá [39,40] | 1995 2012 | Law 50 Decrete 1457 | Health Ministry |
| Estados de Unidos de México [41,42] | 2012 2018 | Norm131 Bill 050 | Executive power Health Secretary |
| República Dominicana [43,44] | 1996 1995 | Law No 8 Decrete 31 | Congress of the republic Congress of the republic |
| Argentina [45,46] | 2013 1969 | Law 26.873 National Law 18284 | Congress Public Health Secretary |
| Chile [47,48] | 1996 2019 | Decrete 977 Law 21155 | Health Ministry Ministry of woman and gender equity |
| Bolivia [49] | 2006 | Law 3460 | Congress |
| Guatemala [50] | 1983 | Law 66–83 | Health and social assistance Ministry |
| El Salvador [51] | 2013 | Decrete 404 | Legislative branch |
| Paraguay [52] | 1999 | Law 1.478 | Congress |
| Costa Rica [53] | 1995 | Law 7430 | Health Ministry |
| Honduras [54] | 2013 | Decrete 231 | Congress |
| Brazil [55] | 2018 | Decrete Nº 9.579 | Executive branch |
| Venezuela [56] | 2007 | Law for protection, promotion and support of breastfeeding | National assembly Permanent commision for family, woman and youth |
| Uruguay [57] | 2017 | File 2062 | House of representatives |
| Country | Label: State Human Milk Is Superior and the Ideal Feeding Option | Label: State Proper Manipulation, Storage and Preparation | Label: State Risk of Inappropriate Preparation or Non-Hygienic Conditions | Label: Pictures or Graph That Suggest Formula Is an Ideal Feeding Method | Label: Forbids the Use of the Terms Humanized or Maternalized Milks | Label: Forbids the Use of Pictures of Children, Feeding Bottles, Toys or Animals (Except for Preparation Directions) | Label: Official Language and Native Language Adaptations | Label: Logo or Brand Name <20% of Space in Label | Label: It Should Only Be Used under Medical Prescription | Approximate Cost for Using the Product for 6 Months | Label: Risks for Using Feeding Bottles | Label: Describing Best Way to Feed (Cup, Spoon) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ecuador [35] | X | X | X | X | X | X | X | --- | --- | --- | --- | --- |
| Perú [36] | X | X | --- | X | X | X | X | --- | --- | --- | --- | --- |
| Colombia [37] | X | X | X | X | X | X | --- | --- | --- | --- | --- | X |
| Nicaragua [38] | X | X | X | X | X | X | X | --- | --- | --- | --- | --- |
| Panamá [39,40] | X | X | --- | X | X | X | X | X | --- | --- | --- | |
| Estados de Unidos de México [41,42] | X | X | X | X | X | --- | --- | --- | X | --- | --- | --- |
| República Dominicana [43,44] | X | X | X | X | X | X | X | --- | X | X | X | --- |
| Argentina [45,46] | X | X | X | X | X | X | X | --- | X | --- | --- | --- |
| Chile [47,48] | X | X | --- | X | X | X | ---- | ---- | X | --- | --- | --- |
| Bolivia [49] | X | X | --- | X | X | X | X | --- | --- | X | --- | --- |
| Guatemala [50] | X | X | --- | X | X | X | --- | --- | --- | --- | --- | --- |
| El Salvador [51] | --- | --- | --- | X | X | X | X | --- | --- | --- | --- | --- |
| Paraguay [52] | X | X | Risk of feeding wrong age group | --- | X | --- | X | --- | --- | --- | --- | --- |
| Costa Rica [53] | X | X | X | X | X | X | X | --- | X | --- | --- | --- |
| Honduras [54] | X | X | X | X | X | X | --- | --- | --- | --- | --- | --- |
| Brazil [55] | X | X | X | X | X | X | --- | --- | X | --- | --- | --- |
| Venezuela [56] | X | X | X | X | X | X | X | --- | X | --- | --- | X |
| Uruguay [57] | X | X | X | X | X | X | X | --- | X | X | X | --- |
| Pre-pregnancy | 500 days |
| Pregnancy | 270 days |
| First two years of age | 730 days |
| Preschooler years | 500 days |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ladino, L.; Sánchez, N.; Vázquez-Frias, R.; Koletzko, B. Latin American Considerations for Infant and Young Child Formulae. Nutrients 2021, 13, 3942. https://doi.org/10.3390/nu13113942
Ladino L, Sánchez N, Vázquez-Frias R, Koletzko B. Latin American Considerations for Infant and Young Child Formulae. Nutrients. 2021; 13(11):3942. https://doi.org/10.3390/nu13113942
Chicago/Turabian StyleLadino, Liliana, Nathalia Sánchez, Rodrigo Vázquez-Frias, and Berthold Koletzko. 2021. "Latin American Considerations for Infant and Young Child Formulae" Nutrients 13, no. 11: 3942. https://doi.org/10.3390/nu13113942
APA StyleLadino, L., Sánchez, N., Vázquez-Frias, R., & Koletzko, B. (2021). Latin American Considerations for Infant and Young Child Formulae. Nutrients, 13(11), 3942. https://doi.org/10.3390/nu13113942







