Are Nutritional Patterns among Polish Hashimoto Thyroiditis Patients Differentiated Internally and Related to Ailments and Other Diseases?
Abstract
:1. Introduction
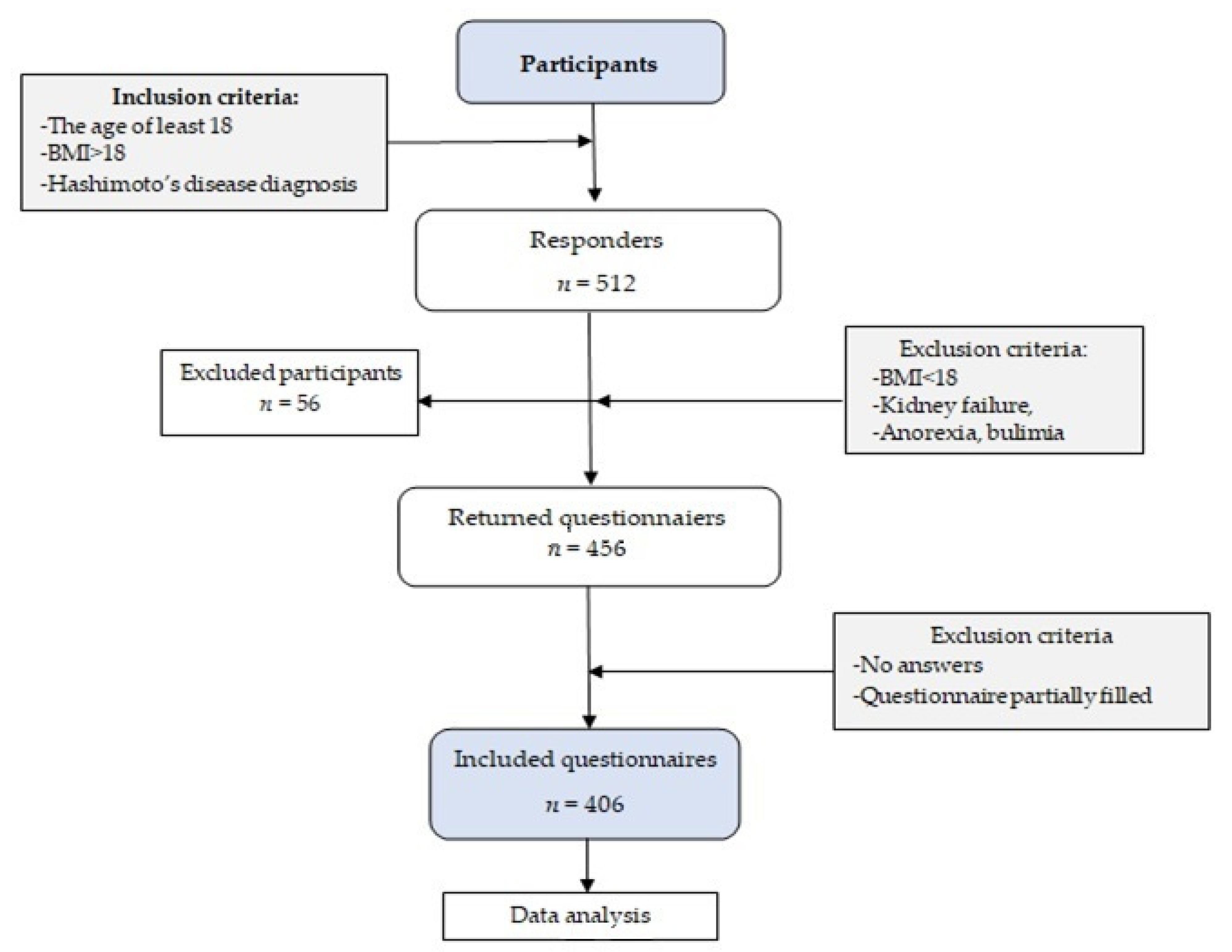
2. Materials and Methods
2.1. Study Design and Sample Collection
2.2. Eating Habits, Health Status, and Lifestyle
2.3. Socio-Demographic Variables
2.4. Statistical Analysis
3. Results
3.1. Sample and Patterns Characteristics
3.2. Dieting Experiences
3.3. Disease and Health-Related Complaints Occurrence
3.4. Nutritional Behavior and Self-Assessment of the Nutrition
3.5. Nutritional Knowledge
3.6. Smoking Habits
3.7. Predictors of the Dietery Patterns
4. Discussion
4.1. Socio-Demographic Data
4.2. BMI, Obesity, and Dieting Experiences
4.3. Nutritional Patterns and Characteristic of Patterns
4.4. Strenghts, Limitations, and Future Perspective
4.5. From the Point of View of a Clinical Dietitian Working with HT Patients
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Garmendia Madariaga, A.; Santos Palacios, S.; Guillén-Grima, F.; Galofré, J.C. The incidence and prevalence of thyroid dysfunction in europe: A meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 923–931. [Google Scholar] [CrossRef] [Green Version]
- Mendes, D.; Alves, C.; Silverio, N.; Batel Marques, F. Prevalence of Undiagnosed Hypothyroidism in Europe: A Systematic Review and Meta-Analysis. Eur. Thyroid. J. 2019, 8, 130–143. [Google Scholar] [CrossRef] [PubMed]
- GUS. Health Status of the Population in Poland in 2019. Available online: http://stat.gov.pl/cps/rde/xbcr/gus/ZO_stan_zdrowia_2009.pdf (accessed on 10 April 2019). (In Polish)
- Jóźków, P.; Lwow, F.; Słowińska-Lisowska, M.; Mędraś, M. Trends in the prevalence of autoimmune thyroiditis in the leading private health-care provider in Poland. Adv. Clin. Exp. Med. 2017, 26, 497–503. [Google Scholar] [CrossRef] [Green Version]
- Ihnatowicz, P.; Drywień, M.; Wątor, P.; Wojsiat, J. The importance of nutritional factors and dietary management of Hashimoto’s thyroiditis. Ann. Agric. Environ. Med. 2020, 27, 184–193. [Google Scholar] [CrossRef]
- WHO. Global Status Report on Non-Communicable Diseases. 2014. Available online: https://ncdalliance.org/sites/default/files/resource_files/Global%20status%20report%20on%20NCDs%202014.pdf?utm_source=NCD+Alliance+Combined+List&utm_campaign=0cd3b9d080-NCDA+E-Alert+22+January+2015&utm_medium=email&utm_term=0_1750ef6b4b-0cd3b9d080-64374793 (accessed on 29 September 2020).
- Krusińska, B.; Hawrysz, I.; Słowinska, M.; Wadołowska, L.; Biernacki, M.; Czerwińska, A.; Gołota, J. Dietary patterns and breast or lung cancer risk: A pooled analysis of 2 case-control studies in north-eastern Poland. Adv. Clin. Exp. Med. 2017, 26, 1367–1375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schulte, E.M.; Avena, N.M.; Gearhardt, A.N. Which Foods May Be Addictive? The Roles of Processing, Fat Content, and Glycemic Load. PLoS ONE 2015, 10, e0117959. Available online: https://pubmed.ncbi.nlm.nih.gov/25692302/ (accessed on 15 May 2021). [CrossRef] [PubMed]
- Carter, A.; Hendrikse, J.; Lee, N.; Yücel, M.; Verdejo-Garcia, A.; Andrews, Z.B.; Hall, W. The Neurobiology of “Food Addiction” and Its Implications for Obesity Treatment and Policy. Annu. Rev. Nutr. 2016, 36, 105–128. [Google Scholar] [CrossRef] [PubMed]
- Middleton, K.R.; Anton, S.D.; Perri, M. Long-Term Adherence to Health Behavior Change. Am. J. Lifestyle Med. 2013, 7, 395–404. [Google Scholar] [CrossRef] [Green Version]
- Czarnocinska, J.; Wadolowska, L.; Lonnie, M.; Kowalkowska, J.; Jezewska-Zychowicz, M.; Babicz-Zielinska, E. Regional and socioeconomic variations in dietary patterns in a representative sample of young polish females: A cross-sectional study (GEBaHealth project). Nutr. J. 2020, 19, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, R.M.; Vicchio, T.M.; Cristani, M.; Certo, R.; Caccamo, D.; Alibrandi, A.; Giovinazzo, S.; Saija, A.; Campennì, A.; Trimarchi, F.; et al. Oxidative Stress and Advanced Glycation End Products in Hashimoto’s Thyroiditis. Thyroid 2016, 26, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Bettermann, E.L.; Hartman, T.J.; Easley, K.; Ferranti, E.P.; Jones, D.P.; Quyyumi, A.A.; Vaccarino, V.; Ziegler, T.R.; Alvarez, J.A. Higher Mediterranean Diet Quality Scores and Lower Body Mass Index Are Associated with a Less-Oxidized Plasma Glutathione and Cysteine Redox Status in Adults. J. Nutr. 2018, 148, 245–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asemi, Z.; Samimi, M.; Tabassi, Z.; Shakeri, H.; Sabihi, S.-S.; Esmaillzadeh, A. Effects of DASH diet on lipid profiles and biomarkers of oxidative stress in overweight and obese women with polycystic ovary syndrome: A randomized clinical trial. Nutrition 2014, 30, 1287–1293. [Google Scholar] [CrossRef] [PubMed]
- Whalen, K.A.; McCullough, M.L.; Flanders, W.D.; Hartman, T.J.; Judd, S.; Bostick, R.M. Paleolithic and Mediterranean Diet Pattern Scores Are Inversely Associated with Biomarkers of Inflammation and Oxidative Balance in Adults. J. Nutr. 2016, 146, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Gould, R.L.; Pazdro, R. Impact of Supplementary Amino Acids, Micronutrients, and Overall Diet on Glutathione Homeostasis. Nutrients 2019, 11, 1056. [Google Scholar] [CrossRef] [Green Version]
- Jiang, S.; Liu, H.; Li, C. Dietary Regulation of Oxidative Stress in Chronic Metabolic Diseases. Foods 2021, 10, 1854. [Google Scholar] [CrossRef]
- Ihnatowicz, P.; Wątor, P.; Drywień, M.E. The importance of gluten exclusion in the management of Hashimoto’s thyroiditis. Ann. Agric. Environ. Med. 2021. [Google Scholar] [CrossRef]
- Trofimiuk-Muldner, M.; Czubek, E.; Sztorc, J.; Skalniak, A.; Hubalewska-Dydejczyk, A. MON-013 Nutritional Approach To Autoimmune Thyroiditis (AIT)—The Patients’ And Medical Professionals’ View. J. Endocr. Soc. 2019, 3, MON-013. [Google Scholar] [CrossRef]
- Markiewicz-Żukowska, R.; Naliwajko, S.K.; Bartosiuk, E.; Sawicka, E.; Omeljaniuk, W.J.; Borawska, M.H. The content of vitamins in diets of patients with Hashimoto disease. Bromat. Chem. Toksykol. 2011, XLIV, 539–543. Available online: https://silo.tips/download/zawarto-witamin-w-dietach-kobiet-z-chorob-hashimoto (accessed on 15 August 2021). (In Polish).
- Omeljaniuk, W.J.; Dziemanowicz, M.; Naliwajko, S.; Bartosiuk, E.; Markiewicz-Żukowska, M.; Borawska, M.H. Ocena Sposobu żywienia Pacjentek z Chorobą Hashimoto. Bromat. Chem. Toksykol. 2011, XLIV, 428–433. Available online: https://www.ptfarm.pl/wydawnictwa/czasopisma/bromatologia-i-chemia-toksykologiczna/117/-/13973 (accessed on 15 August 2021). (In Polish).
- Szostak-Węgierek, D.; Bednarczuk, T.; Respondek, W.; Traczyk, I.; Cukrowska, B.; Ostrowska, L.; Włodarek, D.; Jeznach-Steinhagen, A.; Bierła, J.; Lange, E.; et al. The Validity of Gluten-Free Diet in Hashimoto’s Thyroiditis: Statement of the Expert Committee of the Section of Medical Dietetics of the Polish Society for Parenteral, Enteral Nutrition and Me-tabolism (POLSPEN). Postęp. Żywienia Klin. 2018, 14, 33–47. Available online: https://zdk.wum.edu.pl/sites/zdk.wum.edu.pl/files/czerwiec_2018_stanowisko_polspen.pdf (accessed on 20 May 2020). (In Polish).
- Ott, J.; Promberger, R.; Kober, F.; Neuhold, N.; Tea, M.; Huber, J.C.; Hermann, M. Hashimoto’s Thyroiditis Affects Symptom Load and Quality of Life Unrelated to Hypothyroidism: A Prospective Case–Control Study in Women Undergoing Thyroidectomy for Benign Goiter. Thyroid 2011, 21, 161–167, Erratum in 2011, 21, 467. Available online: https://pubmed.ncbi.nlm.nih.gov/21186954/ (accessed on 20 May 2020). [CrossRef]
- Watt, T.; Hegedüs, L.; Bjorner, J.B.; Groenvold, M.; Bonnema, S.J.; Rasmussen, Å.K.; Feldt-Rasmussen, U. Is Thyroid Autoimmunity per se a Determinant of Quality of Life in Patients with Autoimmune Hypothyroidism? Eur. Thyroid. J. 2012, 1, 186–192. [Google Scholar] [CrossRef] [Green Version]
- Ruggeri, R.M.; Giovinazzo, S.; Barbalace, M.C.; Cristani, M.; Alibrandi, A.; Vicchio, T.M.; Giuffrida, G.; Aguennouz, M.H.; Malaguti, M.; Angeloni, C.; et al. Influence of Dietary Habits on Oxidative Stress Markers in Hashimoto’s Thyroiditis. Thyroid 2021, 31, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Kaličanin, D.; Brčić, L.; Ljubetić, K.; Barić, A.; Gračan, S.; Brekalo, M.; Lovrić, V.T.; Kolčić, I.; Polašek, O.; Zemunik, T.; et al. Differences in food consumption between patients with Hashimoto’s thyroiditis and healthy individuals. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wojtas, N.; Wadolowska, L.; Bandurska-Stankiewicz, E. Evaluation of Qualitative Dietary Protocol (Diet4Hashi) Application in Dietary Counseling in Hashimoto Thyroiditis: Study Protocol of a Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2019, 16, 4841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matana, A.; Torlak, V.; Brdar, D.; Popović, M.; Lozić, B.; Barbalić, M.; Perica, V.B.; Punda, A.; Polašek, O.; Hayward, C.; et al. Dietary Factors Associated with Plasma Thyroid Peroxidase and Thyroglobulin Antibodies. Nutrients 2017, 9, 1186. [Google Scholar] [CrossRef] [Green Version]
- Wadolowska, L.; Hamulka, J.; Kowalkowska, J.; Kostecka, M.; Wadolowska, K.; Biezanowska-Kopec, R.; Czarniecka-Skubina, E.; Kozirok, W.; Piotrowska, A. Prudent-Active and Fast-Food-Sedentary Dietary-Lifestyle Patterns: The Association with Adiposity, Nutrition Knowledge and Sociodemographic Factors in Polish Teenagers—The ABC of Healthy Eating Project. Nutrients 2018, 10, 1988. [Google Scholar] [CrossRef] [Green Version]
- Jezewska-Zychowicz, M.; Gębski, J.; Kobylińska, M. Food Involvement, Eating Restrictions and Dietary Patterns in Polish Adults: Expected Effects of Their Relationships (LifeStyle Study). Nutrients 2020, 12, 1200. [Google Scholar] [CrossRef]
- Długosz, A. Wzory Żywienia, Niepożądane Skutki Zdrowotne, Sytuacja Społeczno-Ekonomiczna i Styl Życia Młodzieży z Mniej Zurbanizowanych Regionów Polski [Dietary Patterns, Adverse Health Outcomes, Socio-Economic Situation and Lifestyle of Adolescents from Less Urbanized Regions of Poland]; UWM Press: Olsztyn, Poland, 2017. [Google Scholar]
- PAN. Kwestionariusz Do Badania Poglądów i Zwyczajów Żywieniowych Oraz Procedura Opracowania Danych (KomPAN®): Wersja Polskojezyczna [Dietary Habits and Nutrition Beliefs Questionnaire and the Manual for Developing of Nutritional Data (KomPAN)]; Committee of Human Nutrition Science, Polish Academy of Science: Warsaw, Poland, 2014; Available online: http://www.knozc.pan.pl/ (accessed on 25 May 2017).
- Kowalkowska, J.; Wadolowska, L.; Czarnocinska, J.; Człapka-Matyasik, M.; Galiński, G.; Jezewska-Zychowicz, M.; Bron-kowska, M.; Dlugosz, A.; Loboda, D.; Wyka, J. Reproducibility of a Questionnaire for Dietary Habits, Lifestyle and Nutrition Knowledge Assessment (KomPAN®) in Polish Adolescents and Adults. Nutrients 2018, 10, 1845. [Google Scholar] [CrossRef] [Green Version]
- WHO. The Challenge of Obesity in the WHO European Region and the Strategies for Response: Summary; Branca, F., Nikogosian, H., Lobstein, T., Eds.; World Health Organization: Copenhagen, Denmark, 2007; Available online: https://www.euro.who.int/__data/assets/pdf_file/0010/74746/E90711.pdf (accessed on 18 April 2019).
- Ashwell, M.; Gibson, S. Waist-to-height ratio as an indicator of ‘early health risk’: Simpler and more predictive than using a ‘matrix’ based on BMI and waist circumference. BMJ Open 2016, 6, e010159. [Google Scholar] [CrossRef] [Green Version]
- Christmann, A.; Van Aelst, S. Robust estimation of Cronbach’s alpha. J. Multivar. Anal. 2006, 97, 1660–1674. [Google Scholar] [CrossRef] [Green Version]
- Vaske, J.J.; Beaman, J.; Sponarski, C.C. Rethinking Internal Consistency in Cronbach’s Alpha. Leis. Sci. 2017, 39, 163–173. [Google Scholar] [CrossRef]
- Borowiec, A.A.; Aranowska, A.E. Nutritional Styles of Poles and Their Socio-Demographic Determinants. Pomeranian J. Life Sci. 2018, 64, 93–98. Available online: https://ojs.pum.edu.pl/pomjlifesci/article/view/413 (accessed on 20 May 2020). (In Polish) [CrossRef]
- Mincer, D.L.; Jialal, I. Hashimoto Thyroiditis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://pubmed.ncbi.nlm.nih.gov/29083758/ (accessed on 20 August 2021).
- Sanyal, D.; Raychaudhuri, M. Hypothyroidism and obesity: An intriguing link. Indian J. Endocrinol. Metab. 2016, 20, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Stoffel, N.U.; El-Mallah, C.; Herter-Aeberli, I.; Bissani, N.; Wehbe, N.; Obeid, O.; Zimmermann, M.B. The effect of central obesity on inflammation, hepcidin, and iron metabolism in young women. Int. J. Obes. 2020, 44, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Solmonson, A.; Mills, E.M. Uncoupling Proteins and the Molecular Mechanisms of Thyroid Thermogenesis. Endocrinology 2015, 157, 455–462. [Google Scholar] [CrossRef] [Green Version]
- Mancuso, P. The role of adipokines in chronic inflammation. ImmunoTargets Ther. 2016, 5, 47–56. [Google Scholar] [CrossRef] [Green Version]
- Treadway, M.T.; Cooper, J.; Miller, A.H. Can’t or Won’t? Immunometabolic Constraints on Dopaminergic Drive. Trends Cogn. Sci. 2019, 23, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Van Bussel, B.C.; Soedamah-Muthu, S.S.; Henry, R.M.; Schalkwijk, C.G.; Ferreira, I.; Chaturvedi, N.; Toeller, M.; Fuller, J.H.; Stehouwer, C.D.; EURODIAB Prospective Complications Study Group. Unhealthy dietary patterns associated with inflammation and endothelial dysfunction in type 1 diabetes: The EURODIAB study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 758–764. [Google Scholar] [CrossRef]
- Chung, W.-S.; Shin, K.O.; Bae, J.Y. Gender Differences in Body Image Misperception According to Body Mass Index, Physical Activity, and Health Concern among Korean University Students. J. Men’s Health 2019, 15, e1–e9. [Google Scholar] [CrossRef] [Green Version]
- Galinski, G.; Lonnie, M.; Kowalkowska, J.; Wadolowska, L.; Czarnocińska, J.; Jezewska-Zychowicz, M.; Babicz-Zielinska, E. Self-Reported Dietary Restrictions and Dietary Patterns in Polish Girls: A Short Research Report (GEBaHealth Study). Nutrients 2016, 8, 796. [Google Scholar] [CrossRef] [Green Version]
- Krysiak, R.; Szkróbka, W.; Okopień, B. The Effect of Gluten-Free Diet on Thyroid Autoimmunity in Drug-Naïve Women with Hashimoto’s Thyroiditis: A Pilot Study. Exp. Clin. Endocrinol. Diabetes 2018, 127, 417–422. [Google Scholar] [CrossRef] [Green Version]
- Asik, M.; Gunes, F.; Binnetoglu, E.; Eroglu, M.; Bozkurt, N.; Sen, H.; Akbal, E.; Bakar, C.; Beyazit, Y.; Ukinc, K. Decrease in TSH levels after lactose restriction in Hashimoto’s thyroiditis patients with lactose intolerance. Endocrine 2014, 46, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Heckl, S.; Reiners, C.; Buck, A.; Schäfer, A.; Dick, A.; Scheurlen, M. Evidence of impaired carbohydrate assimilation in euthyroid patients with Hashimoto’s thyroiditis. Eur. J. Clin. Nutr. 2016, 70, 222–228. [Google Scholar] [CrossRef]
- Chang, B.W.; Chua, K.S.; Lin, E.; Chang, C.; Pimentel, M. Mo1864 Understanding the Significant Interaction Between Hydrogen and Methane in the Performance and Interpretation of Breath Testing. Gastroenterology 2015, 148, S-729. [Google Scholar] [CrossRef]
- Waśkiewicz, A.; Szcześniewska, D.; Szostak-Węgierek, D.; Kwaśniewska, M.; Pająk, A.; Stepaniak, U.; Kozakiewicz, K.; Tykarski, A.; Zdrojewski, T.; Zujko, M.; et al. Are dietary habits of the Polish population consistent with the recommendations for prevention of cardiovascular disease?—WOBASZ II project. Kardiol. Pol. 2016, 74, 969–977. [Google Scholar] [CrossRef] [Green Version]
- Muthukumar, J.; Selvasekaran, P.; Lokanadham, M.; Chidambaram, R. Food and food products associated with food allergy and food intolerance—An overview. Food Res. Int. 2020, 138, 109780. [Google Scholar] [CrossRef]
- Moncayo, R.; Moncayo, H. The WOMED model of benign thyroid disease: Acquired magnesium deficiency due to physical and psychological stressors relates to dysfunction of oxidative phosphorylation. BBA Clin. 2014, 3, 44–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rousseau, S.; Kyomugasho, C.; Celus, M.; Hendrickx, M.E.G.; Grauwet, T. Barriers impairing mineral bioaccessibility and bioavailability in plant-based foods and the perspectives for food processing. Crit. Rev. Food Sci. Nutr. 2020, 60, 826–843. [Google Scholar] [CrossRef] [PubMed]
- Srour, B.; Fezeu, L.K.; Kesse-Guyot, E.; Allès, B.; Méjean, C.; Andrianasolo, R.M.; Chazelas, E.; Deschasaux, M.; Hercberg, S.; Galan, P.; et al. Ultra-processed food intake and risk of cardiovascular disease: Prospective cohort study (NutriNet-Santé). BMJ 2019, 365, l1451. [Google Scholar] [CrossRef] [Green Version]
- Nieman, K.M.; Anderson, B.D.; Cifelli, C.J. The Effects of Dairy Product and Dairy Protein Intake on Inflammation: A Systematic Review of the Literature. J. Am. Coll. Nutr. 2021, 40, 571–582. [Google Scholar] [CrossRef]
- Towery, P.; Guffey, J.S.; Doerflein, C.; Stroup, K.; Saucedo, S.; Taylor, J. Chronic musculoskeletal pain and function improve with a plant-based diet. Complement. Ther. Med. 2018, 40, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Nanda, N.; Bobby, Z.; Hamide, A. Oxidative stress and protein glycation in primary hypothyroidism. Male/female difference. Clin. Exp. Med. 2008, 8, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Pasupathi, P.; Latha, R. Free Radical Activity and Antioxidant Defense Mechanisms in Patients with Hypothyroidism. Thyroid. Sci. 2008, 3, CLS1-6. Available online: https://www.researchgate.net/publication/202072862_Free_radical_activity_and_antioxidant_defense_mechanisms_in_patients_with_Hypothyroidism (accessed on 20 August 2021).
- Reddy, V.S.; Gouroju, S.; Suchitra, M.M.; Suresh, V.; Sachan, A.; Rao, P.V.L.N.S.; Bitla, A.R. Antioxidant Defense in Overt and Subclinical Hypothyroidism. Horm. Metab. Res. 2013, 45, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Rostami, R.; Aghasi, M.; Mohammadi, A.; Nourooz-Zadeh, J. Enhanced oxidative stress in Hashimoto’s thyroiditis: Inter-relationships to biomarkers of thyroid function. Clin. Biochem. 2013, 46, 308–312. [Google Scholar] [CrossRef]
- Minich, D.M.; Brown, B.I. A Review of Dietary (Phyto)Nutrients for Glutathione Support. Nutrients 2019, 11, 2073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muhlheim, L.S.; Allison, D.; Heshka, S.; Heymsfield, S.B. Do unsuccessful dieters intentionally underreport food intake? Int. J. Eat. Disord. 1998, 24, 259–266. [Google Scholar] [CrossRef]
- Kessler, K.; Pivovarova-Ramich, O. Meal Timing, Aging, and Metabolic Health. Int. J. Mol. Sci. 2019, 20, 1911. [Google Scholar] [CrossRef] [Green Version]
- Qian, F.; Riddle, M.C.; Wylie-Rosett, J.; Hu, F.B. Red and Processed Meats and Health Risks: How Strong Is the Evidence? Diabetes Care 2020, 43, 265–271. [Google Scholar] [CrossRef] [Green Version]
- Farvid, M.S.; Sidahmed, E.; Spence, N.D.; Angua, K.M.; Rosner, B.A.; Barnett, J.B. Consumption of red meat and processed meat and cancer incidence: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2021, 36, 937–951. [Google Scholar] [CrossRef]
- Hamulka, J.; Wadolowska, L.; Hoffmann, M.; Kowalkowska, J.; Gutkowska, K. Effect of an Education Program on Nutrition Knowledge, Attitudes toward Nutrition, Diet Quality, Lifestyle, and Body Composition in Polish Teenagers. The ABC of Healthy Eating Project: Design, Protocol, and Methodology. Nutrients 2018, 10, 1439. [Google Scholar] [CrossRef] [Green Version]
- Zielińska, M.; Hamułka, J.; Gajda, K. Family influences on breakfast frequency and quality among primary school pupils in Warsaw and its surrounding areas. Rocz. Państw. Zak. Hig. 2015, 66, 143–149. [Google Scholar]
- Wüenstel, J.W.; Kowalkowska, J.; Wądołowska, L.; Słowińska, M.; Niedźwiedzka, E.; Kurp, L. Habitual eating of breakfast, consumption frequency of selected food and overweight prevalence in adolescents from various age groups. Dev. Period Med. 2015, 19, 193–201. [Google Scholar]
- Kołłątaj, W.; Sygit, K.; Sygit, M.; Karwat, I.D.; Kołłątaj, B. Eating habits of children and adolescents from rural regions de-pending on gender, education, and economic status of parents. Ann. Agric. Environ. Med. 2011, 18, 393–397. [Google Scholar]
- Zakrzewski, J.K.; Gillison, F.B.; Cumming, S.; Church, T.S.; Katzmarzyk, P.T.; Broyles, S.T.; Champagne, C.M.; Chaput, J.-P.; Denstel, K.D.; Fogelholm, M.; et al. Associations between breakfast frequency and adiposity indicators in children from 12 countries. Int. J. Obes. Suppl. 2015, 5 (Suppl. 2), S80–S88. [Google Scholar] [CrossRef] [Green Version]
- Brug, J.; van Stralen, M.M.; Te Velde, S.J.T.; Chinapaw, M.J.M.; De Bourdeaudhuij, I.; Lien, N.; Bere, E.; Maskini, V.; Singh, A.S.; Maes, L.; et al. Differences in weight status and energy-balance related behaviors among schoolchildren across europe: The energy-project. PLoS ONE 2012, 7, e34742. [Google Scholar] [CrossRef] [Green Version]
- Hallström, L.; Vereecken, C.A.; Labayen, I.; Ruiz, J.R.; Le Donne, C.; García, M.C.; Gilbert, C.C.; Martínez, S.G.; Grammatikaki, E.; Huybrechts, I.; et al. Breakfast habits among European adolescents and their association with sociodemographic factors: The HELENA (Healthy Lifestyle in Europe by Nutrition in Adolescence) study. Public Health Nutr. 2012, 15, 1879–1889. [Google Scholar] [CrossRef] [Green Version]
- Kowalkowska, J.; Slowinska, M.A.; Slowinski, D.; Dlugosz, A.; Niedzwiedzka, E.; Wadolowska, L. Comparison of a full food-frequency questionnaire with the three-day unweighted food records in young polish adult women: Implications for dietary assessment. Nutrients 2013, 5, 2747–2776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forrestal, S.G. Energy intake misreporting among children and adolescents: A literature review. Matern. Child Nutr. 2011, 7, 112–127. [Google Scholar] [CrossRef]
- Czarnocińska, J.; Jeżewska-Zychowicz, M.; Babicz-Zielińska, E.; Kowalkowska, J.; Wądołowska, L. Postawy Względem Żywności, Żywienia i Zdrowia a Zachowania Żywieniowe Dziewcząt i Młodych Kobiet w Polsce. [Attitudes towards Food, Nutrition and Health and Dietary Behaviours of Girls and Young Women in Poland]; UWM Press: Olsztyn, Poland, 2013; Available online: http://wydawnictwo.uwm.edu.pl/uploads/documents/czytelnia/monografie/Czarnocinska-Postawy-wzgledem-zywnosci.pdf (accessed on 30 September 2021).
- Drywień, M.E.; Hamulka, J.; Jezewska-Zychowicz, M. Perceived Nutrition and Health Concerns: Do They Protect against Unhealthy Dietary Patterns in Polish Adults? Nutrients 2021, 13, 170. [Google Scholar] [CrossRef]
- Abbott, R.D.; Sadowski, A.; Alt, A.G. Efficacy of the Autoimmune Protocol Diet as Part of a Multi-disciplinary, Supported Lifestyle Intervention for Hashimoto’s Thyroiditis. Cureus 2019, 11, e4556. [Google Scholar] [CrossRef] [Green Version]
- Ihnatowicz, P.; Wątor, P.; Drywień, M. Supplementation in Autoimmune Thyroid Hashimoto’s Disease. Vitamin D and Selenium. J. Food Nutr. Res. 2019, 7, 584–591. [Google Scholar] [CrossRef] [Green Version]
- Janowska-Miasik, E.; Waśkiewicz, A.; Witkowska, A.M.; Drygas, W.; Markhus, M.W.; Zujko, M.E.; Kjellevold, M. Diet quality in the population of Norway and Poland: Differences in the availability and consumption of food considering national nutrition guidelines and food market. BMC Public Health 2021, 21, 319. [Google Scholar] [CrossRef] [PubMed]
- Głąbska, D.; Włodarek, D.; Kołota, A.; Czekajło, A.; Drozdzowska, B.; Pluskiewicz, W. Assessment of mineral intake in the diets of Polish postmenopausal women in relation to their BMI—The RAC-OST-POL study. J. Health Popul. Nutr. 2016, 35, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eveleigh, E.R.; Coneyworth, L.J.; Avery, A.; Welham, S.J.M. Vegans, Vegetarians, and Omnivores: How Does Dietary Choice Influence Iodine Intake? A Systematic Review. Nutrients 2020, 12, 1606. [Google Scholar] [CrossRef] [PubMed]
- Krela-Kaźmierczak, I.; Czarnywojtek, A.; Skoracka, K.; Rychter, A.M.; Ratajczak, A.E.; Szymczak-Tomczak, A.; Ruchała, M.; Dobrowolska, A. Is There an Ideal Diet to Protect against Iodine Deficiency? Nutrients 2021, 13, 513. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Farwell, A.P. Euthyroid Sick Syndrome. Compr. Physiol. 2016, 6, 1071–1080. [Google Scholar] [CrossRef]
- Ganesan, K.; Wadud, K. Euthyroid Sick Syndrome. 2 November 2020. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://pubmed.ncbi.nlm.nih.gov/29489255/ (accessed on 12 October 2021).
| Product | Highly Processed Food Products | Dairy Products | Meat and Meat Products | Different Not Advised | Vegetables and Fruits | Vegetables and Fruit Juices | Water |
|---|---|---|---|---|---|---|---|
| Powder soups | 0.780 | - | - | - | - | - | - |
| Canned meat | 0.752 | - | - | - | - | - | - |
| Ready-made soups (e.g., in cardboard packages) | 0.711 | - | - | - | - | - | - |
| Fast food | 0.678 | - | - | - | - | - | - |
| Energy drinks | 0.675 | - | - | - | - | - | - |
| Alcoholic drinks | 0.574 | - | - | - | - | - | - |
| Sweetened drinks | 0.546 | - | - | - | - | - | - |
| Canned and marinated vegetables | 0.526 | - | - | - | - | - | - |
| Yogurt, kefir | - | 0.825 | - | - | - | - | - |
| Cottage cheese | - | 0.824 | - | - | - | - | - |
| Milk | - | 0.722 | - | - | - | - | - |
| Yellow cheeses | - | 0.649 | - | - | - | - | - |
| Wholegrain bread | - | 0.469 | - | - | - | - | - |
| Butter | - | 0.452 | - | - | - | - | - |
| Homogenized cheese | - | 0.428 | - | - | - | - | - |
| Red meat dishes | - | - | 0.791 | - | - | - | - |
| White meat dishes (poultry, rabbit) | - | - | 0.722 | - | - | - | - |
| Sausages or frankfurters | - | - | 0.721 | - | - | - | - |
| Fried foods (e.g., meat or flour based) | - | - | 0.486 | - | - | - | - |
| Fish | - | - | 0.430 | - | - | - | - |
| Lard | - | - | 0.410 | - | - | - | - |
| Sweets | - | - | - | 0.566 | - | - | - |
| White rice, pasta, groats | - | - | - | 0.542 | - | - | - |
| White bread | - | - | - | 0.529 | - | - | - |
| Oils and margarines | - | - | - | 0.419 | - | - | - |
| Vegetables | - | - | - | - | 0.736 | - | - |
| Fruits | - | - | - | - | 0.700 | - | - |
| Legume-based meals | - | - | - | - | 0.600 | - | - |
| Buckwheat, oats, wholegrain pasta | - | - | - | - | 0.475 | - | - |
| Fruit juices | - | - | - | - | - | 0.765 | - |
| Vegetables or vegetable and fruit juices | - | - | - | - | - | 0.755 | - |
| Water, e.g., mineral and table water | - | - | - | - | - | - | 0.602 |
| Variance explained (%) | 22.35% | 8.06% | 7.27% | 5.89% | 4.52% | 3.72% | 3.27% |
| Total Variance explained (%) | 55.08% | ||||||
| Cronbach’ alpha | 0.842 | 0.804 | 0.734 | 0.696 | 0.682 | 0.811 | - |
| Kaiser’s Measure of Sampling Adequacy: | 0.862 | ||||||
| Dietary Patterns | |||||
|---|---|---|---|---|---|
| Variables | Convenient Pattern 1 n = 109 | Non-Meat Pattern 2 n = 97 | Pro-Healthy Pattern 3 n = 99 | Carnivores Pattern 4 n = 101 | p-Value |
| F1: Highly processed food products | 4.39 a,* | 2.98 b | 1.81 d | 2.66 c | <0.0001 |
| F2: Dairy products | 2.42 d | 3.62 a | 2.77 c | 3.24 b | <0.0001 |
| F3: Meat and meat products | 2.59 c | 1.63 d | 3.59 b | 4.15 a | <0.0001 |
| F4: Different not recommended products | 1.88 d | 3.98 a | 2.64 c | 3.57 b | <0.0001 |
| F5: Vegetables and fruit | 2.63 b | 2.88 b | 4.54 a | 1.97 c | <0.0001 |
| F6: Vegetables and fruit juices | 3.18 a | 2.57 b | 3.12 a | 3.06 a | 0.0096 |
| F7: Water | 3.49 a | 3.37 a | 2.97 b | 2.10 c | <0.0001 |
| Dietary Patterns | |||||||
|---|---|---|---|---|---|---|---|
| Variables | Convenient n = 109 | Non-Meat n = 97 | Pro-Healthy n = 99 | Carnivores n = 101 | p-Value | Total (n) | |
| Age | - | 35.04 b* | 36.67 a | 32.37 c | 33.26 bc | ||
| Gender | Female | 94.5 | 88.66 | 93.94 | 99.01 | 0.0226 | 382 |
| Male | 5.5 | 11.34 | 6.06 | 0.99 | 24 | ||
| BMI categories | Underweight | 5.5 | 6.19 | 4.04 | 7.92 | 0.7069 | 24 |
| Normal weight | 56.88 | 50.52 | 49.49 | 59.41 | - | 220 | |
| Overweight | 24.77 | 31.96 | 34.34 | 23.76 | - | 116 | |
| Obese | 12.84 | 11.34 | 12.12 | 8.91 | - | 46 | |
| Type of work | Unemployed | 22.94 | 15.46 | 18.18 | 23.76 | 0.0397 | 82 |
| Brain work | 60.55 | 73.2 | 54.55 | 56.44 | 248 | ||
| Physical work | 9.17 | 5.15 | 14.14 | 4.95 | 34 | ||
| Standing work | 7.34 | 6.19 | 13.13 | 14.85 | 43 | ||
| Factors with the most substantial impact on food choice | Economical/ financial | 21.1 | 14.43 | 31.31 | 4.95 | <0.0001 | 73 |
| Nutritional values and composition, and ecological origin of the food, bioproducts, and being on a diet | 74.31 | 83.51 | 57.58 | 93.07 | 313 | ||
| Advertisement of a particular food product, a fad for its consumption, gustatory preferences | 2.75 | 1.03 | 4.04 | 0.0 | 8 | ||
| Time/time of meal preparation/food products’ availability | 0.00 | 0.00 | 5.05 | 1.98 | 7 | ||
| I do not make shopping trips/ I do shopping randomly | 1.83 | 1.03 | 2.02 | 0.0 | 5 | ||
| Convenient n = 109 | Non-Meat n = 97 | Pro-Healthy n = 99 | Carnivores n = 101 | p-Value | Total (n) | ||
|---|---|---|---|---|---|---|---|
| Diet targeting thyroid disease in the last two years | Yes | 50.46 | 78.35 | 47.47 | 71.29 | <0.001 | 250 |
| No | 49.54 | 21.65 | 52.53 | 28.71 | 156 | ||
| The current duration of the diet | No diet | 49.54 | 23.71 | 58.59 | 32.67 | <0.001 | 168 |
| Up to 3 months | 22.02 | 20.62 | 21.21 | 30.69 | 96 | ||
| 4–12 months | 20.18 | 19.59 | 9.09 | 18.81 | 69 | ||
| More than 12 months | 8.26 | 36.08 | 11.11 | 17.82 | 73 | ||
| Well-being after the diet (any diet you have followed lately) | Much better | 74.07 | 83.78 | 84.78 | 87.32 | <0.0001 | 203 |
| Hard to assess | 20.37 | 13.51 | 10.87 | 12.68 | 35 | ||
| Worse | 5.56 | 2.7 | 4.35 | 0.00 | 7 | ||
| Having diabetes | No | 97.25 | 95.88 | 96.97 | 98.02 | 0.8461 | 394 |
| Yes | 2.75 | 4.12 | 3.03 | 1.98 | 12 | ||
| Having any cardiovascular disease | No | 95.41 | 94.85 | 96.97 | 96.04 | 0.8935 | 389 |
| Yes | 4.59 | 5.15 | 3.03 | 3.96 | 17 | ||
| Having food allergies | No | 96.33 | 86.6 | 93.94 | 86.14 | 0.0193 | 369 |
| Yes | 3.67 | 13.4 | 6.06 | 13.86 | 37 | ||
| Having food intolerances | No | 83.49 | 67.01 | 78.79 | 73.27 | 0.0377 | 308 |
| Yes | 16.51 | 32.99 | 21.21 | 26.73 | 98 | ||
| Having any intestinal disease | No | 87.16 | 85.57 | 88.89 | 83.17 | 0.6825 | 350 |
| Yes | 12.84 | 14.43 | 11.11 | 16.83 | 56 | ||
| Having kidney failure | No | 100 | 100 | 100 | 100 | - | 406 |
| Yes | 0 | 0 | 0 | 0 | 0 | ||
| Having gout | No | 100 | 100 | 100 | 100 | - | 406 |
| Yes | 0 | 0 | 0 | 0 | 0 | ||
| Having lipid disorders | No | 99.08 | 93.81 | 94.95 | 100 | 0.0213 | 394 |
| Yes | 0.92 | 6.19 | 5.05 | 0 | 12 | ||
| Gastrointestinal complaints * | No | 27.52 | 22.68 | 13.13 | 25.74 | 0.0661 | 91 |
| Yes | 72.48 | 77.32 | 86.87 | 74.26 | 315 | ||
| Nervous system complaints * | No | 22.02 | 16.49 | 10.1 | 12.87 | 0.0962 | 63 |
| Yes | 77.98 | 83.51 | 89.90 | 87.13 | 343 | ||
| Musculoskeletal complaints * | No | 50.46 | 37.11 | 27.27 | 44.55 | 0.005 | 163 |
| Yes | 49.54 | 62.89 | 72.73 | 55.45 | 243 | ||
| Skin complaints * | No | 17.43 | 27.84 | 21.21 | 22.77 | 0.3481 | 90 |
| Yes | 82.57 | 72.16 | 78.79 | 77.23 | 316 |
| Convenient | Non-Meat | Pro-Healthy | Carnivores | p-Value | Total (n) | ||
|---|---|---|---|---|---|---|---|
| Number of meals during the day | 1–2 | 0.92 | 5.15 | 5.05 | 0.99 | 0.1883 | - |
| 3–4 | 81.65 | 72.16 | 78.79 | 72.25 | 313 | ||
| ≥5 | 17.43 | 22.68 | 16.16 | 23.76 | - | ||
| Regularity of meals | Yes | 28.44 | 44.33 | 14.14 | 46.53 | <0.0001 | 135 |
| Yes, some meals | 53.21 | 38.14 | 48.48 | 42.57 | 186 | ||
| No | 18.35 | 17.53 | 37.37 | 10.89 | 85 | ||
| Eating breakfast before leaving the house | Yes, always | 55.96 | 58.76 | 36.36 | 53.47 | 0.0119 | 208 |
| Usually | 43.12 | 35.05 | 56.57 | 41.58 | 179 | ||
| No | 0.92 | 6.19 | 7.07 | 4.95 | 19 | ||
| The frequency of eating between meals | Once/several times a day | 45.54 | 43.3 | 55.56 | 31.68 | 0.0242 | 183 |
| Several times a week/1–2 times a week | 35.78 | 37.11 | 32.32 | 42.57 | 150 | ||
| Never/almost never | 14.68 | 19.59 | 12.12 | 25.74 | 73 | ||
| Salting | Yes, for most meals | 13.76 | 15.46 | 18.18 | 11.88 | 0.7495 | 60 |
| Yes, sometimes | 32.11 | 30.93 | 37.37 | 35.64 | 138 | ||
| No, I do not | 54.13 | 53.61 | 44.44 | 52.48 | 208 | ||
| Sweetening | Yes, 1–2 teaspoons of sugar/honey | 11 | 16.5 | 33.33 | 8.91 | <0.0001 | 70 |
| Yes, I use sweeteners | 1.84 | 8.24 | 8.08 | 5.94 | 24 | ||
| No, I do not or only sometimes | 87.16 | 75.26 | 58.59 | 85.15 | 312 | ||
| Self-assessment of nutrition | Bad or very bad | 25.69 | 23.71 | 63.64 | 11.88 | <0.0001 | 126 |
| Good or very good | 74.31 | 76.29 | 36.36 | 88.12 | 280 | ||
| Self-assessment of nutrition during the week compared to the weekends | No different | 47.71 | 52.58 | 44.44 | 51.49 | 0.5358 | 199 |
| Differs slightly | 37.61 | 39.18 | 43.43 | 41.58 | 164 | ||
| Varies greatly | 14.68 | 8.25 | 12.12 | 6.93 | 43 | ||
| Self-assessment of nutritional knowledge | Inadequate | 11.01 | 8.25 | 23.23 | 4.96 | 0.0002 | 48 |
| Adequate | 24.77 | 17.53 | 28.28 | 20.79 | 93 | ||
| Good | 46.79 | 45.36 | 35.35 | 41.58 | 172 | ||
| Very good | 17.43 | 28.87 | 13.13 | 32.67 | 93 | ||
| Smoking cigarettes or tobacco | Yes | 3.67 | 9.28 | 17.17 | 11.88 | 0.0142 | 42 |
| No | 96.33 | 90.72 | 82.83 | 88.12 | 364 |
| Variables | Convenient | Non-Meat | Pro-Healthy | Carnivores | ||||
|---|---|---|---|---|---|---|---|---|
| * OR (95% CI) p | aOR (95% CI) p | OR (95% CI) p | aOR (95% CI) p | OR (95% CI) p | aOR (95% CI) p | OR (95% CI) p | aOR (95% CI) p | |
| Type of work: | ||||||||
| Unemployed | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Office work | 0.83 (0.48; 1.43) 0.4964 | 0.80 (0.46; 1.39) 0.4351 | 1.79 (1.06; 3.34) 0.0469 | 1.88 (1.03; 3.55) 0.0418 | 0.99 (0.54; 1.81) 0.9732 | 1.03 (0.56; 1.89) 0.9295 | 0.72 (0.41; 1.26) 0.2528 | 0.69 (0.39; 1.22) 0.2044 |
| Physical work | 0.95 (0.4; 2.28) 0.9085 | 0.93 (0.38; 2.24) 0.8663 | 0.77 (0.26; 2.32) 0.6422 | 0.72 (0.23; 2.22) 0.567 | 2.48 (1.05; 5.88) 0.0377 | 2.50 (1.05; 5.98) 0.0389 | 0.42 (0.14; 1.21) 0.106 | 0.45 (0.15; 1.31) 0.1431 |
| Standing work | 0.54 (0.22; 1.32) 0.1762 | 0.49 (0.2; 1.22) 0.1249 | 0.74 (0.27; 2.09) 0.5743 | 0.84 (0.29; 2.39) 0.743 | 1.59 (0.69; 3.68) 0.2753 | 1.75 (0.74; 4.1) 0.2001 | 1.34 (0.61; 2.96) 0.465 | 1.22 (0.54; 2.73) 0.6332 |
| Factors with the most substantial impact on food choice: | ||||||||
| Economical/financial | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Nutritional values and composition, and ecological origin of the food, bioproducts, and being on a diet | 0.76 (0.44; 1.32) 0.33 | 0.75 (0.43; 1.32) 0.3147 | 1.47 (0.78; 2.78) 0.2334 | 1.49 (0.78; 2.85) 0.2317 | 0.30 (0.18; 0.52) <0.0001 | 0.30 (0.17; 0.53) <0.0001 | 5.84 (2.28; 14.94) <0.0002 | 5.96 (2.31; 15.37) <0.0002 |
| Number of meals during the day: | ||||||||
| 1–2 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 3–4 | 4.37 (0.56; 34.35) 0.1609 | 4.42 (0.56; 34.86) 0.1589 | 0.40 (0.12; 1.31) 0.1308 | 0.44 (0.13; 1.47) 0.1812 | 0.47 (0.14; 1.51) 0.2014 | 0.45 (0.14; 1.48) 0.1883 | 3.52 (0.45; 27.7) 0.2314 | 3.18 (0.4; 25.48) 0.2765 |
| ≥5 | 3.37 (0.41; 27.82) 0.2591 | 3.48 (0.42; 28.9) 0.2485 | 0.52 (0.15; 1.82) 0.3072 | 0.59 (0.16; 2.1) 0.4134 | 0.35 (0.1; 1.23) 0.1005 | 0.32 (0.09; 1.18) 0.0866 | 4.63 (0.57; 37.8) 0.153 | 4.56 (0.49; 33.81) 0.1956 |
| Regularity of meals: | ||||||||
| Yes | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes, some meals | 1.52 (0.92; 2.52) 0.1055 | 1.53 (0.92; 2.55) 0.1031 | 0.53 (0.32; 0.89) 0.0152 | 0.52 (0.31; 0.88) 0.0137 | 3.01 (1.58; 5.72) 0.0008 | 3.02 (1.58; 5.77) 0.0008 | 0.56 (0.34; 0.92) 0.022 | 0.56 (0.34; 0.92) 0.0221 |
| No | 1.03 (0.54; 1.96) 0.9228 | 1.06 (0.55; 2.02) 0.8676 | 0.54 (0.28; 0.96) 0.0465 | 0.54 (0.28; 0.94) 0.0437 | 6.66 (3.31; 13.41) <0.0001 | 6.52 (3.21; 13.23) <0.0001 | 0.28 (0.14; 0.58) 0.0006 | 0.27 (0.13; 0.57) 0.0005 |
| Eating breakfast before leaving the house: | ||||||||
| Yes, always | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| I usually do if I have time | 0.86 (0.55; 1.34) 0.5022 | 0.84 (0.53; 1.32) 0.4482 | 0.62 (0.38; 0.98) 0.0429 | 0.60 (0.37; 0.99) 0.0448 | 2.18 (1.35; 3.51) 0.0015 | 2.24 (1.38; 3.65) 0.0012 | 0.87 (0.55; 1.39) 0.5707 | 0.86 (0.55; 1.42) 0.614 |
| I do not eat breakfast | 0.13 (0.02; 0.93) 0.0329 | 0.13 (0.02; 0.97) 0.0463 | 1.22 (0.44; 3.37) 0.6973 | 1.18 (0.42; 3.32) 0.7536 | 2.79 (1.03; 7.57) 0.0443 | 2.93 (1.06; 8.05) 0.0376 | 1.02 (0.35; 2.96) 0.9731 | 1.11 (0.37; 3.30) 0.8517 |
| The frequency of eating between meals: | ||||||||
| Once/several times a day | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Several times a week/1–2 times a week | 0.84 (0.52; 1.36) 0.478 | 0.82 (0.50; 1.34) 0.4304 | 1.06 0,8221 0.8221 | 1.07 (0.64; 1.81) 0.7924 | 0.63 (0.38; 0.94) 0.0426 | 0.64 (0.39; 0.97) 0.0417 | 1.89 (1.13; 3.19) 0.0159 | 1.90 (1.12; 3.24) 0.0182 |
| Never/almost never | 0.67 (0.35; 1.27) 0.2204 | 0.65 (0.34; 1.26) 0.2008 | 1.18 (0.63; 2.21) 0.6021 | 1.08 (0.56; 2.09) 0.8211 | 0.46 (0.23; 0.92) 0.0276 | 0.47 (0.23; 0.95) 0.0367 | 2.61 (1.42; 4.82) 0.0021 | 2.96 (1.56; 5.63) 0.0009 |
| Salting: | ||||||||
| Never/almost never | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes, for most meals | 1.02 (0.51; 2.05) 0.957 | 1.01 (0.50; 2.04) 0.9877 | 0.83 (0.41; 1.7) 0.6151 | 0.86 (0.42; 1.77) 0.6807 | 0.86 (0.44; 1.67) 0.6453 | 0.91 (0.46; 1.79) 0.7857 | 1.41 (0.68; 2.95) 0.3599 | 1.31 (0.62; 2.77) 0.4834 |
| No, I do not | 1.19 (0.62; 2.29) 0.6077 | 1.17 (0.6; 2.28) 0.6363 | 1 (0.52; 1.94) 1 | 1.04 (0.53; 2.04) 0.9195 | 0.63 (0.33; 1.19) 0.1544 | 0.66 (0.35; 1.28) 0.2186 | 1.37 (0.68; 2.77) 0.3843 | 1.27 (0.62; 2.60) 0.5154 |
| Sweetening: | ||||||||
| Yes, 1–2 teaspons of sugar/honey | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes, I use sweeteners | 0.44 (0.09; 2.12) 0.3062 | 0.44 (0.09; 2.12) 0.3022 | 1.69 (0.61; 4.66) 0.3122 | 1.65 (0.59; 4.66) 0.3435 | 0.56 (0.21; 1.48) 0.2421 | 0.56 (0.21; 1.50) 0.2512 | 2.26 (0.71; 7.20) 0.1682 | 2.35 (0.72; 7.69) 0.1573 |
| No, I do not or only sometimes | 2.12 (1.09; 4.12) 0.0276 | 2.13 (1.09; 4.18) 0.0274 | 1.03 (0.56; 1.91) 0.923 | 1.07 (0.57; 2.01) 0.8336 | 0.26 (0.15; 0.44) <0.0001 | 0.26 (0.15; 0.45) <0.0001 | 2.58 (1.23; 5.42) 0.0124 | 2.46 (1.16; 5.22) 0.0186 |
| Having diabetes: | ||||||||
| No | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes | 0.91 (0.24; 3.41) 0.8835 | 1.01 (0.26; 3.86) 0.9943 | 1.62 (0.48; 5.50) 0.4398 | 1.36 (0.38; 4.87) 0.9943 | 1.04 (0.28; 3.90) 0.9595 | 0.91 (0.24; 3.50) 0.888 | 0.59 (0.13; 2.77) 0.5087 | 0.73 (0.15; 3.49) 0.6881 |
| Having any cardiovascular disease: | ||||||||
| No | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes | 1.14 (0.39; 3.32) 0.8075 | 1.14 (0.39; 3.36) 0.8135 | 1.35 (0.46; 3.92) 0.5867 | 1.25 (0.42; 3.74) 0.6904 | 0.65 (0.18; 2.33) 0.5121 | 0.61 (0.17; 2.20) 0.4495 | 0.93 (0.30; 2.91) 0.8956 | 1.09 (0.34; 3.55) 0.882 |
| Having food allergies: | ||||||||
| No | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes | 0.31 (0.11; 0.88) 0.0283 | 0.28 (0.10; 0.82) 0.0203 | 1.84 (0.90; 3.77) 0.0964 | 2.05 (1.09; 4.25) 0.0447 | 0.57 (0.23; 1.42) 0.23 | 0.59 (0.24; 1.46) 0.2505 | 1.97 (1.07; 4.00) 0.0394 | 1.91 (1.03; 3.92) 0.0479 |
| Having food intolerances: | ||||||||
| No | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes | 0.54 (0.30; 0.95) 0.0313 | 0.53 (0.30; 0.93) 0.0281 | 1.81 (1.1; 3.00) 0.0205 | 1.92 (1.15; 3.21) 0.0124 | 0.80 (0.47; 1.39) 0.4346 | 0.83 (0.48; 1.44) 0.507 | 1.20 (0.72; 2.01) 0.4823 | 1.13 (0.67; 1.91) 0.6374 |
| Having any intestinal disease: | ||||||||
| No | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes | 0.89 (0.47; 1.71) 0.7374 | 0.88 (0.46; 1.69) 0.7039 | 1.07 (0.56; 2.06) 0.8341 | 1.07 (0.55; 2.08) 0.8345 | 0.73 (0.36; 1.47) 0.3752 | 0.75 (0.37; 1.51) 0.4153 | 1.38 (0.74; 2.57) 0.3083 | 1.38 (0.73; 2.58) 0.3234 |
| Having lipid disorders: | ||||||||
| No | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Yes | 0.24 (0.03; 1.89) 0.1753 | 0.23 (0.03; 1.85) 0.1673 | 3.33 (1.05; 10.58) 0.0413 | 3.38 (1.04; 11.05) 0.0436 | 2.28 (0.71; 7.35) 0.1678 | 2.09 (0.64; 6.86) 0.2244 | - | - |
| Smoking cigarettes or tobacco: | ||||||||
| Yes | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| No | 3.85 (1.34; 11.06) 0.0122 | 3.91 (1.35; 11.32) 0.0124 | 1.17 (0.54; 2.54) 0.6928 | 1.10 (0.5; 2.43) 0.8068 | 0.43 (0.22; 0.83) 0.0121 | 0.43 (0.22; 0.85) 0.0152 | 0.81 (0.4; 1.65) 0.5591 | 0.83 (0.4; 1.71) 0.608 |
| Self-assessment of nutrition: | ||||||||
| Bad or very bad | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Good or very good | 1.43 (0.87; 2.33) 0.1596 | 1.49 (0.89; 2.52) 0.1309 | 1.61 (0.95; 2.72) 0.0757 | 1.69 (1.04; 2.96) 0.0423 | 0.15 (0.09; 0.24) <0.0001 | 0.13 (0.08; 0.22) <0.0001 | 4.43 (2.32; 8.44) <0.0001 | 4.81 (2.43; 9.51) <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ihnatowicz, P.; Wątor, P.; Gębski, J.; Frąckiewicz, J.; Drywień, M.E. Are Nutritional Patterns among Polish Hashimoto Thyroiditis Patients Differentiated Internally and Related to Ailments and Other Diseases? Nutrients 2021, 13, 3675. https://doi.org/10.3390/nu13113675
Ihnatowicz P, Wątor P, Gębski J, Frąckiewicz J, Drywień ME. Are Nutritional Patterns among Polish Hashimoto Thyroiditis Patients Differentiated Internally and Related to Ailments and Other Diseases? Nutrients. 2021; 13(11):3675. https://doi.org/10.3390/nu13113675
Chicago/Turabian StyleIhnatowicz, Paulina, Paweł Wątor, Jerzy Gębski, Joanna Frąckiewicz, and Małgorzata Ewa Drywień. 2021. "Are Nutritional Patterns among Polish Hashimoto Thyroiditis Patients Differentiated Internally and Related to Ailments and Other Diseases?" Nutrients 13, no. 11: 3675. https://doi.org/10.3390/nu13113675
APA StyleIhnatowicz, P., Wątor, P., Gębski, J., Frąckiewicz, J., & Drywień, M. E. (2021). Are Nutritional Patterns among Polish Hashimoto Thyroiditis Patients Differentiated Internally and Related to Ailments and Other Diseases? Nutrients, 13(11), 3675. https://doi.org/10.3390/nu13113675







