Changes in Dietary Patterns and Clinical Health Outcomes in Different Countries during the SARS-CoV-2 Pandemic
Abstract
:1. Introduction
2. Clinical Pathology of SARS-CoV-2 Infection and COVID-19 Syndrome
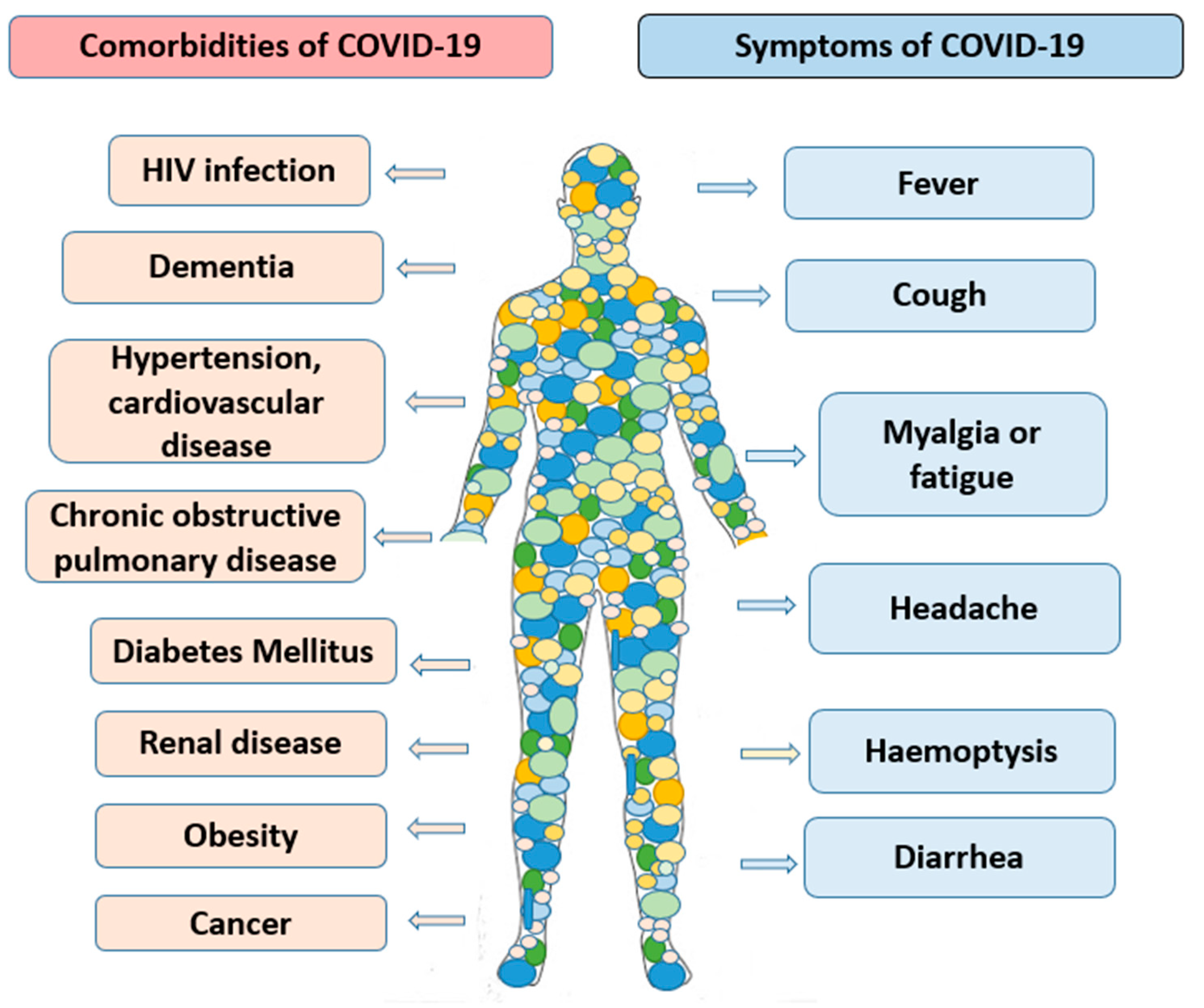
3. COVID-19 and Comorbidities
3.1. Nutrients and Food Bioactive Components Involved in Immune System Stimulation
3.2. The Effects of Isolation, Quarantine, and Lockdowns on Dietary Health
3.2.1. Spain
3.2.2. Italy
3.2.3. France
3.2.4. Greece
3.2.5. Denmark
3.2.6. Poland
3.2.7. China
3.2.8. United States
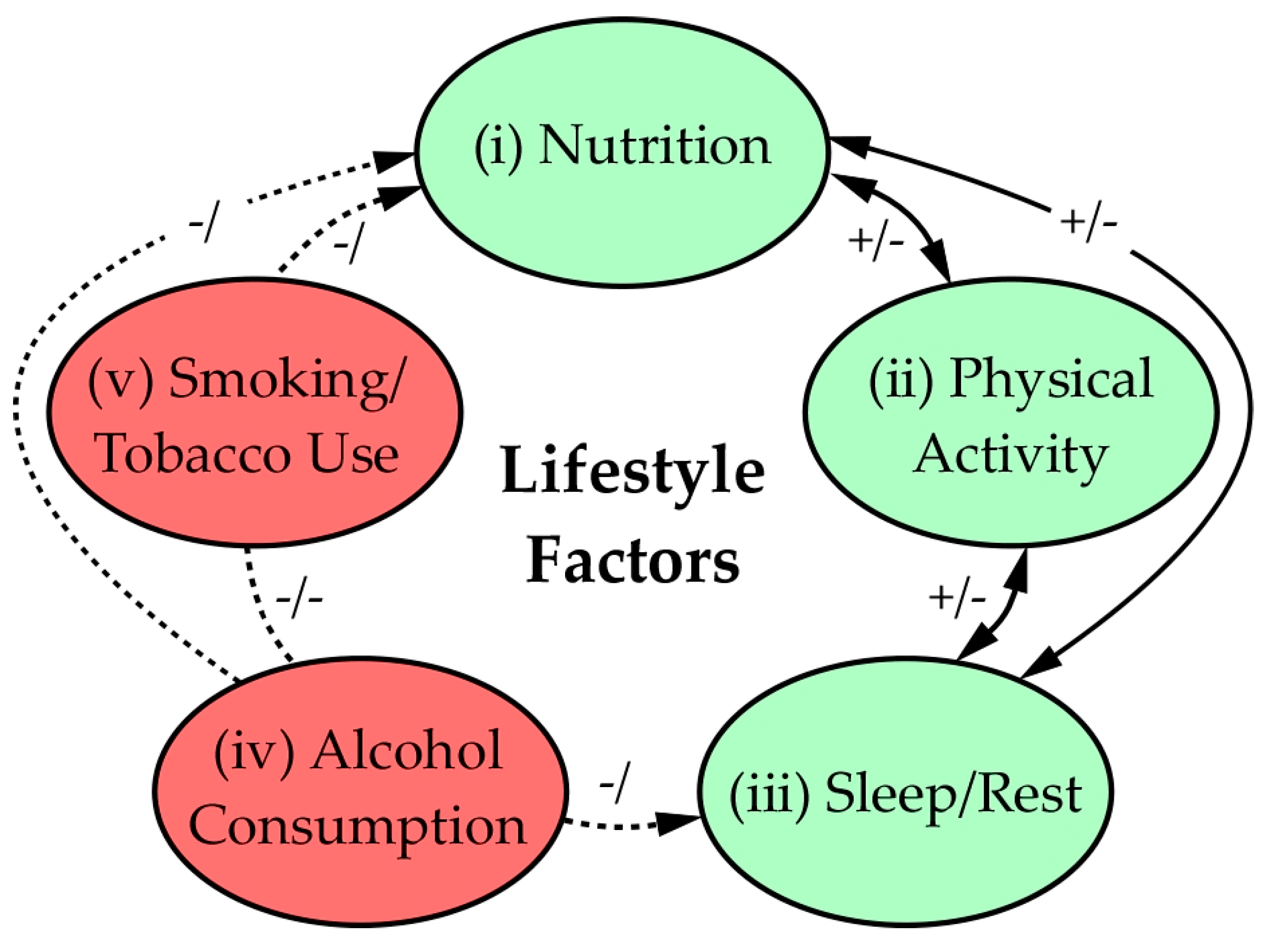
3.3. Importance of Lifestyle in Prevention of COVID-19
3.4. Food Hygiene in COVID-19 Pandemic
3.4.1. Food Security
3.4.2. Food Safety in COVID-19 Pandemic
3.4.3. Personnel Hygiene in Food Industry in COVID-19 Pandemic
3.4.4. Food Retail in COVID-19 Pandemic
3.4.5. Food Delivery Include
3.4.6. Importance of Smart Packaging with Antiviral Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, Y.-C.; Chen, C.-S.; Chan, Y.-J. The outbreak of COVID-19: An overview. J. Chin. Med. Assoc. 2020, 83, 217–220. [Google Scholar] [CrossRef]
- Ng, W.H.; Tipih, T.; Makoah, N.A.; Vermeulen, J.-G.; Goedhals, D.; Sempa, J.B.; Burt, F.J.; Taylor, A.; Mahalingam, S. Comorbidities in SARS-CoV-2 Patients: A Systematic Review and Meta-Analysis. mBio 2021, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Fathi, M.; Vakili, K.; Sayehmiri, F.; Mohamadkhani, A.; Hajiesmaeili, M.; Rezaei-Tavirani, M.; Eilami, O. The prognostic value of comorbidity for the severity of COVID-19: A systematic review and meta-analysis study. PLoS ONE 2021, 16, e0246190. [Google Scholar] [CrossRef]
- Bertuzzi, S.; DiRita, V.J. After COVID-19 We Will Need a New Research System. We Need to Start Planning Now. mBio 2020, 11, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Water, Sanitation, Hygiene, and Waste Management for SARS-CoV-2, the Virus that Causes COVID-19. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-IPC-WASH-2020.4 (accessed on 31 August 2021).
- Cyranoski, D. Bat cave solves mystery of deadly SARS virus—And suggests new outbreak could occur. Nature 2017, 552, 15–16. [Google Scholar] [CrossRef]
- Xiao-Shuang, Z.; Zeng, L.; Yang, X.-L.; Ge, X.-Y.; Zhang, W.; Lin-Fa, W.; Xie, J.-Z.; Dong-Sheng, L.; Zhang, Y.-Z.; Wang, N.; et al. Discovery of a rich gene pool of bat SARS-related coronaviruses provides new insights into the origin of SARS coronavirus. PLOS Pathog. 2017, 13, e1006698. [Google Scholar] [CrossRef]
- Lau, S.K.P.; Lee, P.; Tsang, A.K.L.; Yip, C.C.Y.; Tse, H.; Lee, R.A.; So, L.-Y.; Lau, Y.L.; Chan, K.-H.; Woo, P.C.Y.; et al. Molecular Epidemiology of Human Coronavirus OC43 Reveals Evolution of Different Genotypes over Time and Recent Emergence of a Novel Genotype due to Natural Recombination. J. Virol. 2011, 85, 11325–11337. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Shi, Z.; Yu, M.; Ren, W.; Smith, C.; Epstein, J.H.; Wang, H.; Crameri, G.; Hu, Z.; Zhang, H.; et al. Bats Are Natural Reservoirs of SARS-Like Coronaviruses. Science 2005, 310, 676–679. [Google Scholar] [CrossRef]
- Mulay, A.; Konda, B.; Garcia, G.; Yao, C.; Beil, S.; Villalba, J.M.; Koziol, C.; Sen, C.; Purkayastha, A.; Kolls, J.K.; et al. SARS-CoV-2 infection of primary human lung epithelium for COVID-19 modeling and drug discovery. Cell Rep. 2021, 35, 109055. [Google Scholar] [CrossRef] [PubMed]
- Gryseels, S.; De Bruyn, L.; Gyselings, R.; Calvignac-Spencer, S.; Leendertz, F.H.; Leirs, H. Risk of human-to-wildlife transmission of SARS-CoV-2. Mammal Rev. 2020, 51, 272–292. [Google Scholar] [CrossRef]
- Menachery, V.D.; Yount, B.L.; Debbink, K.; Agnihothram, S.; Gralinski, L.E.; Plante, J.A.; Graham, R.L.; Scobey, T.; Ge, X.-Y.; Donaldson, E.F.; et al. A SARS-like cluster of circulating bat coronaviruses shows potential for human emergence. Nat. Med. 2015, 21, 1508–1513. [Google Scholar] [CrossRef] [PubMed]
- De Backer, C.; Teunissen, L.; Cuykx, I.; Decorte, P.; Pabian, S.; Gerritsen, S.; Matthys, C.; Al Sabbah, H.; Van Royen, K.; The Corona Cooking Survey Study Group. An Evaluation of the COVID-19 Pandemic and Perceived Social Distancing Policies in Relation to Planning, Selecting, and Preparing Healthy Meals: An Observational Study in 38 Countries Worldwide. Front. Nutr. 2021, 7, 621726. [Google Scholar] [CrossRef]
- Czarniecka-Skubina, E.; Pielak, M.; Sałek, P.; Głuchowski, A.; Kobus-Cisowska, J.; Owczarek, T. Use of Food Services by Consumers in the SARS-CoV-2 Pandemic. How the Eating Habits of Consumers Changed in View of the New Disease Risk Factors? Nutrients 2021, 13, 2760. [Google Scholar] [CrossRef]
- Memon, S.; Pawase, V.; Pavase, T.; Soomro, M. Investigation of COVID-19 Impact on the Food and Beverages Industry: China and India Perspective. Foods 2021, 10, 1069. [Google Scholar] [CrossRef]
- Kassir, R. Risk of COVID-19 for patients with obesity. Obes. Rev. 2020, 21, e13034. [Google Scholar] [CrossRef] [Green Version]
- Celorio-Sardà, R.; Comas-Basté, O.; Latorre-Moratalla, M.; Zerón-Rugerio, M.; Urpi-Sarda, M.; Illán-Villanueva, M.; Farran-Codina, A.; Izquierdo-Pulido, M.; Vidal-Carou, M. Effect of COVID-19 Lockdown on Dietary Habits and Lifestyle of Food Science Students and Professionals from Spain. Nutrients 2021, 13, 1494. [Google Scholar] [CrossRef]
- Overview of The 2019 Novel #Coronavirus (2019-nCoV): The #Pathogen of Severe Specific Contagious #Pneumonia (SSCP) (J Chin Med Assoc., Abstract)—ETIDIoH. Available online: https://etidioh.wordpress.com/2020/02/13/overview-of-the-2019-novel-coronavirus-2019-ncov-the-pathogen-of-severe-specific-contagious-pneumonia-sscp-j-chin-med-assoc-abstract/ (accessed on 31 July 2021).
- Chan, J.F.-W.; Yuan, S.; Kok, K.-H.; To, K.K.-W.; Chu, H.; Yang, J.; Xing, F.; Liu, J.; Yip, C.C.-Y.; Poon, R.W.-S.; et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet 2020, 395, 514–523. [Google Scholar] [CrossRef] [Green Version]
- Backer, J.A.; Klinkenberg, D.; Wallinga, J. Incubation period of 2019 novel coronavirus (2019-nCoV) infections among travellers from Wuhan, China, 20–28 January 2020. Eurosurveillance 2020, 25, 2000062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Assiri, A.; Al-Tawfiq, J.A.; Al-Rabeeah, A.A.; Al-Rabiah, F.A.; Al-Hajjar, S.; Al-Barrak, A.; Flemban, H.; Al-Nassir, W.N.; Balkhy, H.H.; Al-Hakeem, R.F.; et al. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: A descriptive study. Lancet Infect. Dis. 2013, 13, 752–761. [Google Scholar] [CrossRef] [Green Version]
- Cockrell, A.S.; Yount, B.L.; Scobey, T.; Jensen, K.; Douglas, M.; Beall, A.; Tang, X.-C.; Marasco, W.A.; Heise, M.T.; Baric, R.S. A mouse model for MERS coronavirus-induced acute respiratory distress syndrome. Nat. Microbiol. 2016, 2, 16226. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. Coronavirus: Covid-19 has killed more people than SARS and MERS combined, despite lower case fatality rate. BMJ 2020, 368, m641. [Google Scholar] [CrossRef] [Green Version]
- Sas, B. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J. Biol. Regul. Homeost. Agents 2020, 33, 9–14. [Google Scholar] [CrossRef]
- Duzgun, S.A.; Durhan, G.; Demirkazik, F.B.; Akpinar, M.G.; Ariyurek, O.M. COVID-19 pneumonia: The great radiological mimicker. Insights Imaging 2020, 11, 118. [Google Scholar] [CrossRef]
- Xie, C.; Ng, M.-Y.; Ding, J.; Leung, S.T.; Lo, C.S.Y.; Wong, H.Y.F.; Vardhanabhuti, V. Discrimination of pulmonary ground-glass opacity changes in COVID-19 and non-COVID-19 patients using CT radiomics analysis. Eur. J. Radiol. Open 2020, 7, 100271. [Google Scholar] [CrossRef]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Guan, W.-J.; Liang, W.-H.; Zhao, Y.; Liang, H.-R.; Chen, Z.-S.; Li, Y.-M.; Liu, X.-Q.; Chen, R.-C.; Tang, C.-L.; Wang, T.; et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: A nationwide analysis. Eur. Respir. J. 2020, 55, 2000547. [Google Scholar] [CrossRef] [Green Version]
- Sheppard, J.P.; Nicholson, B.D.; Lee, J.; McGagh, D.; Sherlock, J.; Koshiaris, C.; Oke, J.; Jones, N.R.; Hinton, W.; Armitage, L.; et al. Association Between Blood Pressure Control and Coronavirus Disease 2019 Outcomes in 45 418 Symptomatic Patients with Hypertension. Hypertension 2021, 77, 846–855. [Google Scholar] [CrossRef]
- Schiffrin, E.L.; Flack, J.M.; Ito, S.; Muntner, P.; Webb, R.C. Hypertension and COVID-19. Am. J. Hypertens. 2020, 33, 373–374. [Google Scholar] [CrossRef]
- Mazucanti, C.H.; Egan, J.M. SARS-CoV-2 disease severity and diabetes: Why the connection and what is to be done? Immun. Ageing 2020, 17, 21. [Google Scholar] [CrossRef] [PubMed]
- Tabrizi, F.M.; Rasmi, Y.; Hosseinzadeh, E.; Rezaei, S.; Balvardi, M.; Kouchari, M.R.; Ebrahimi, G. Diabetes is associated with increased mortality and disease severity in hospitalized patients with covid-19. EXCLI J. 2021, 20, 444–453. [Google Scholar] [CrossRef]
- Chávez-Reyes, J.; Escárcega-González, C.E.; Chavira-Suárez, E.; León-Buitimea, A.; Vázquez-León, P.; Morones-Ramírez, J.R.; Villalón, C.M.; Quintanar-Stephano, A.; Marichal-Cancino, B.A. Susceptibility for Some Infectious Diseases in Patients with Diabetes: The Key Role of Glycemia. Front. Public Health 2021, 9, 559595. [Google Scholar] [CrossRef]
- Erener, S. Diabetes, infection risk and COVID-19. Mol. Metab. 2020, 39, 101044. [Google Scholar] [CrossRef]
- Kumar, A.; Arora, A.; Sharma, P.; Anikhindi, S.A.; Bansal, N.; Singla, V.; Khare, S.; Srivastava, A. Is diabetes mellitus associated with mortality and severity of COVID-19? A meta-analysis. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 535–545. [Google Scholar] [CrossRef]
- Hussain, A.; Bhowmik, B.; Moreira, N.C.D.V. COVID-19 and diabetes: Knowledge in progress. Diabetes Res. Clin. Pract. 2020, 162, 108142. [Google Scholar] [CrossRef]
- Gerstein, H.C.; Thorpe, K.E.; Taylor, D.W.; Haynes, R.B. The effectiveness of hydroxychloroquine in patients with type 2 diabetes mellitus who are refractory to sulfonylureas—A randomized trial. Diabetes Res. Clin. Pract. 2002, 55, 209–219. [Google Scholar] [CrossRef]
- Rekedal, L.R.; Massarotti, E.; Garg, R.; Bhatia, R.; Gleeson, T.; Lu, B.; Solomon, D.H. Changes in glycosylated hemoglobin after initiation of hydroxychloroquine or methotrexate treatment in diabetes patients with rheumatic diseases. Arthritis Rheum. 2010, 62, 3569–3573. [Google Scholar] [CrossRef] [Green Version]
- Coate, K.C.; Cha, J.; Shrestha, S.; Wang, W.; Gonçalves, L.M.; Almaça, J.; Kapp, M.E.; Fasolino, M.; Morgan, A.; Dai, C.; et al. SARS-CoV-2 Cell Entry Factors ACE2 and TMPRSS2 Are Expressed in the Microvasculature and Ducts of Human Pancreas but Are Not Enriched in β Cells. Cell Metab. 2020, 32, 1028–1040.e4. [Google Scholar] [CrossRef]
- Alqahtani, J.S.; Oyelade, T.; Aldhahir, A.M.; Alghamdi, S.M.; Almehmadi, M.; Alqahtani, A.S.; Quaderi, S.; Mandal, S.; Hurst, J.R. Prevalence, Severity and Mortality associated with COPD and Smoking in patients with COVID-19: A Rapid Systematic Review and Meta-Analysis. PLoS ONE 2020, 15, e0233147. [Google Scholar] [CrossRef]
- de Lusignan, S.; Dorward, J.; Correa, A.; Jones, N.; Akinyemi, O.; Amirthalingam, G.; Andrews, N.; Byford, R.; Dabrera, G.; Elliot, A.; et al. Risk factors for SARS-CoV-2 among patients in the Oxford Royal College of General Practitioners Research and Surveillance Centre primary care network: A cross-sectional study. Lancet Infect. Dis. 2020, 20, 1034–1042. [Google Scholar] [CrossRef]
- Increased Susceptibility to Infection in Smokers—Tobacco in Australia. Available online: https://www.tobaccoinaustralia.org.au/chapter-3-health-effects/3-9-increased-susceptibility-to-infection-in-smoke (accessed on 8 October 2021).
- Grundy, E.J.; Suddek, T.; Filippidis, F.T.; Majeed, A.; Coronini-Cronberg, S. Smoking, SARS-CoV-2 and COVID-19: A review of reviews considering implications for public health policy and practice. Tobacco Induced Dis. 2020, 18. [Google Scholar] [CrossRef] [PubMed]
- Paleiron, N.; Mayet, A.; Marbac, V.; Perisse, A.; Barazzutti, H.; Brocq, F.-X.; Janvier, F.; Dautzenberg, B.; Bylicki, O. Impact of Tobacco Smoking on the Risk of COVID-19: A Large Scale Retrospective Cohort Study. Nicotine Tob. Res. 2021, 23, 1398–1404. [Google Scholar] [CrossRef]
- Jiang, H.; Zhou, Y.; Tang, W. Maintaining HIV care during the COVID-19 pandemic. Lancet HIV 2020, 7, e308–e309. [Google Scholar] [CrossRef]
- Inciarte, A.; Gonzalez-Cordon, A.; Rojas, J.; Torres, B.; De Lazzari, E.; De La Mora, L.; Martinez-Rebollar, M.; Laguno, M.; Callau, P.; Gonzalez-Navarro, A.; et al. Clinical characteristics, risk factors, and incidence of symptomatic coronavirus disease 2019 in a large cohort of adults living with HIV: A single-center, prospective observational study. AIDS 2020, 34, 1775–1780. [Google Scholar] [CrossRef] [PubMed]
- Elfiky, A.A. Ribavirin, Remdesivir, Sofosbuvir, Galidesivir, and Tenofovir against SARS-CoV-2 RNA dependent RNA polymerase (RdRp): A molecular docking study. Life Sci. 2020, 253, 117592. [Google Scholar] [CrossRef]
- Etienne, N.; Karmochkine, M.; Slama, L.; Pavie, J.; Batisse, D.; Usubillaga, R.; Letembet, V.-A.; Brazille, P.; Canouï, E.; Slama, D.; et al. HIV infection and COVID-19. AIDS 2020, 34, 1771–1774. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, J.; Akdis, C.; Jutel, M.; Bachert, C.; Klimek, L.; Agache, I.; Ansotegui, I.J.; Bedbrook, A.; Bosnic-Anticevich, S.; Canonica, G.W.; et al. Intranasal corticosteroids in allergic rhinitis in COVID-19 infected patients: An ARIA-EAACI statement. Allergy 2020, 75. [Google Scholar] [CrossRef] [PubMed]
- Conforti, C.; Giuffrida, R.; Dianzani, C.; Di Meo, N.; Zalaudek, I. COVID-19 and psoriasis: Is it time to limit treatment with immunosuppressants? A call for action. Dermatol. Ther. 2020, 33, e13298. [Google Scholar] [CrossRef]
- Torres, T.; Puig, L. Managing Cutaneous Immune-Mediated Diseases During the COVID-19 Pandemic. Am. J. Clin. Dermatol. 2020, 21, 307–311. [Google Scholar] [CrossRef] [Green Version]
- Favalli, E.G.; Ingegnoli, F.; De Lucia, O.; Cincinelli, G.; Cimaz, R.; Caporali, R. COVID-19 infection and rheumatoid arthritis: Faraway, so close! Autoimmun. Rev. 2020, 19, 102523. [Google Scholar] [CrossRef]
- Kennedy, N.A.; Jones, G.-R.; Lamb, C.A.; Appleby, R.; Arnott, I.; Beattie, R.M.; Bloom, S.; Brooks, A.J.; Cooney, R.; Dart, R.J.; et al. British Society of Gastroenterology guidance for management of inflammatory bowel disease during the COVID-19 pandemic. Gut 2020, 69, 984–990. [Google Scholar] [CrossRef]
- Coles, C.E.; Aristei, C.; Bliss, J.; Boersma, L.; Brunt, A.M.; Chatterjee, S.; Hanna, G.; Jagsi, R.; Person, O.K.; Kirby, A.; et al. International guidelines on radiation therapy for breast cancer during the COVID-19 pandemic. Clin. Oncol. 2020, 32, 279. [Google Scholar] [CrossRef] [PubMed]
- Dholaria, B.; Savani, B.N. How do we plan hematopoietic cell transplant and cellular therapy with the looming COVID-19 threat? Br. J. Haematol. 2020, 189, 239–240. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, A.M.; Boddu, P.C.; Patnaik, M.M.; Bewersdorf, J.P.; Stahl, M.; Rampal, R.K.; Shallis, R.; Steensma, D.P.; Savona, M.R.; Sekeres, M.A.; et al. Special considerations in the management of adult patients with acute leukaemias and myeloid neoplasms in the COVID-19 era: Recommendations from a panel of international experts. Lancet Haematol. 2020, 7, e601–e612. [Google Scholar] [CrossRef]
- Lu, X.; Zhang, L.; Du, H.; Zhang, J.; Li, Y.Y.; Qu, J.; Zhang, W.; Wang, Y.; Bao, S.; Li, Y.; et al. SARS-CoV-2 Infection in Children. N. Engl. J. Med. 2020, 382, 1663–1665. [Google Scholar] [CrossRef] [Green Version]
- Hoehl, S.; Rabenau, H.; Berger, A.; Kortenbusch, M.; Cinatl, J.; Bojkova, D.; Behrens, P.; Böddinghaus, B.; Götsch, U.; Naujoks, F.; et al. Evidence of SARS-CoV-2 Infection in Returning Travelers from Wuhan, China. N. Engl. J. Med. 2020, 382, 1278–1280. [Google Scholar] [CrossRef]
- Grasselli, G.; Zangrillo, A.; Zanella, A.; Antonelli, M.; Cabrini, L.; Castelli, A.; Cereda, D.; Coluccello, A.; Foti, G.; Fumagalli, R.; et al. Baseline Characteristics and Outcomes of 1591 Patients Infected with SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA 2020, 323, 1574. [Google Scholar] [CrossRef] [Green Version]
- Lippi, G.; Mattiuzzi, C.; Sanchis-Gomar, F.; Henry, B.M. Clinical and demographic characteristics of patients dying from COVID-19 in Italy vs China. J. Med Virol. 2020, 92, 1759–1760. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Long, Q.-X.; Tang, X.-J.; Shi, Q.-L.; Li, Q.; Deng, H.-J.; Yuan, J.; Hu, J.-L.; Xu, W.; Zhang, Y.; Lv, F.-J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Okba, N.M.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D.; et al. Severe Acute Respiratory Syndrome Coronavirus 2−Specific Antibody Responses in Coronavirus Disease Patients. Emerg. Infect. Dis. 2020, 26, 1478–1488. [Google Scholar] [CrossRef]
- Boechat, J.L.; Chora, I.; Morais, A.; Delgado, L. The immune response to SARS-CoV-2 and COVID-19 immunopathology—Current perspectives. Pulmonology 2021, 27, 423–437. [Google Scholar] [CrossRef] [PubMed]
- Matricardi, P.M.; Negro, R.W.D.; Nisini, R. The first, holistic immunological model of COVID-19: Implications for prevention, diagnosis, and public health measures. Pediatr. Allergy Immunol. 2020, 31, 454–470. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Cheng, Y.; Cheng, G.; Chui, C.H.; Lau, F.Y.; Chan, P.K.S.; Ng, M.H.L.; Sung, J.J.Y.; Wong, R.S.M. ABO Blood Group and Susceptibility to Severe Acute Respiratory Syndrome. JAMA 2005, 293, 1447–1451. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Y.; Huang, H.; Li, D.; Gu, D.; Lu, X.; Zhang, Z.; Liu, L.; Liu, T.; Liu, Y.; et al. Relationship Between the ABO Blood Group and the Coronavirus Disease 2019 (COVID-19) Susceptibility. Clin. Infect. Dis. 2020, 73, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Lu, K.; Pfefferle, S.; Bertram, S.; Glowacka, I.; Drosten, C.; Pöhlmann, S.; Simmons, G. A Single Asparagine-Linked Glycosylation Site of the Severe Acute Respiratory Syndrome Coronavirus Spike Glycoprotein Facilitates Inhibition by Mannose-Binding Lectin through Multiple Mechanisms. J. Virol. 2010, 84, 8753–8764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, Y.; Allen, J.D.; Wrapp, D.; McLellan, J.S.; Crispin, M. Site-specific glycan analysis of the SARS-CoV-2 spike. Science 2020, 369, 330–333. [Google Scholar] [CrossRef]
- Tomaiuolo, R.; Ruocco, A.; Salapete, C.; Carru, C.; Baggio, G.; Franceschi, C.; Zinellu, A.; Vaupel, J.; Bellia, C.; Sasso, B.L.; et al. Activity of mannose-binding lectin in centenarians. Aging Cell 2012, 11, 394–400. [Google Scholar] [CrossRef] [Green Version]
- Boudewijns, R.; Thibaut, H.J.; Kaptein, S.J.F.; Li, R.; Vergote, V.; Seldeslachts, L.; Van Weyenbergh, J.; De Keyzer, C.; Bervoets, L.; Sharma, S.; et al. STAT2 signaling restricts viral dissemination but drives severe pneumonia in SARS-CoV-2 infected hamsters. Nat. Commun. 2020, 11, 5838. [Google Scholar] [CrossRef]
- Kopp, W. Diet-induced hyperinsulinemia as a key factor in the etiology of both benign prostatic hyperplasia and essential hypertension? Nutr. Metab. Insights 2018, 11, 1178638818773072. [Google Scholar] [CrossRef] [Green Version]
- Kopp, W. How Western Diet And Lifestyle Drive The Pandemic Of Obesity And Civilization Diseases. Diabetes Metab. Syndr. Obesity Targets Ther. 2019, 12, 2221–2236. [Google Scholar] [CrossRef] [Green Version]
- Paoli, A.; Gorini, S.; Caprio, M. The dark side of the spoon—Glucose, ketones and COVID-19: A possible role for ketogenic diet? J. Transl. Med. 2020, 18, 441. [Google Scholar] [CrossRef]
- Clemente-Suárez, V.; Ramos-Campo, D.; Mielgo-Ayuso, J.; Dalamitros, A.; Nikolaidis, P.; Hormeño-Holgado, A.; Tornero-Aguilera, J. Nutrition in the Actual COVID-19 Pandemic. A Narrative Review. Nutrients 2021, 13, 1924. [Google Scholar] [CrossRef] [PubMed]
- Lange, K.W. Food science and COVID-19. Food Sci. Hum. Wellness 2020, 10, 1–5. [Google Scholar] [CrossRef]
- Dhok, A.; Butola, L.K.; Anjankar, A.; Shinde, A.D.R.; Kute, P.; Jha, R.K. Role of Vitamins and Minerals in Improving Immunity during Covid-19 Pandemic—A Review. J. Evol. Med. Dent. Sci. 2020, 9, 2296–2300. [Google Scholar] [CrossRef]
- Kumar, P.; Kumar, M.; Bedi, O.; Gupta, M.; Kumar, S.; Jaiswal, G.; Rahi, V.; Yedke, N.G.; Bijalwan, A.; Sharma, S.; et al. Role of vitamins and minerals as immunity boosters in COVID-19. Inflammopharmacology 2021, 29, 1001–1016. [Google Scholar] [CrossRef] [PubMed]
- Suwannasom, N.; Kao, I.; Pruß, A.; Georgieva, R.; Bäumler, H. Riboflavin: The Health Benefits of a Forgotten Natural Vitamin. Int. J. Mol. Sci. 2020, 21, 950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cámara, M.; Sánchez-Mata, M.; Fernández-Ruiz, V.; Cámara, R.; Cebadera, E.; Domínguez, L. A Review of the Role of Micronutrients and Bioactive Compounds on Immune System Supporting to Fight against the COVID-19 Disease. Foods 2021, 10, 1088. [Google Scholar] [CrossRef]
- Di Matteo, G.; Spano, M.; Grosso, M.; Salvo, A.; Ingallina, C.; Russo, M.; Ritieni, A.; Mannina, L. Food and COVID-19: Preventive/Co-therapeutic Strategies Explored by Current Clinical Trials and in Silico Studies. Foods 2020, 9, 1036. [Google Scholar] [CrossRef] [PubMed]
- Shakoor, H.; Feehan, J.; Al Dhaheri, A.S.; Ali, H.I.; Platat, C.; Ismail, L.C.; Apostolopoulos, V.; Stojanovska, L. Immune-boosting role of vitamins D, C, E, zinc, selenium and omega-3 fatty acids: Could they help against COVID-19? Maturitas 2020, 143, 1–9. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, Y.; Tariq, A.; Jiang, X.; Ahmed, Z.; Zhihao, Z.; Idrees, M.; Azizullah, A.; Adnan, M.; Bussmann, R.W. Food as medicine: A possible preventive measure against coronavirus disease (COVID-19). Phytotherapy Res. 2020, 34, 3124–3136. [Google Scholar] [CrossRef]
- Kandeil, A.; Mostafa, A.; Kutkat, O.; Moatasim, Y.; Al-Karmalawy, A.A.; Rashad, A.A.; Kayed, A.E.; Kayed, A.E.; El-Shesheny, R.; Kayali, G.; et al. Bioactive Polyphenolic Compounds Showing Strong Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus 2. Pathogens 2021, 10, 758. [Google Scholar] [CrossRef]
- Khubber, S.; Hashemifesharaki, R.; Mohammadi, M.; Gharibzahedi, S.M.T. Garlic (Allium sativum L.): A potential unique therapeutic food rich in organosulfur and flavonoid compounds to fight with COVID-19. Nutr. J. 2020, 19, 124. [Google Scholar] [CrossRef]
- Shaldam, M.A.; Yahya, G.; Mohamed, N.H.; Abdel-Daim, M.M.; Al Naggar, Y. In silico screening of potent bioactive compounds from honeybee products against COVID-19 target enzymes. Environ. Sci. Pollut. Res. 2021, 28, 40507–40514. [Google Scholar] [CrossRef]
- Chojnacka, K.; Witek-Krowiak, A.; Skrzypczak, D.; Mikula, K.; Młynarz, P. Phytochemicals containing biologically active polyphenols as an effective agent against Covid-19-inducing coronavirus. J. Funct. Foods 2020, 73, 104146. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, I.; Sharma, M.; Mehta, M.; Badyal, S.; Sharma, V.; Sharma, I.; Singh, H.; Sistla, S. Bioactive compounds and probiotics—A ray of hope in COVID-19 management. Food Sci. Hum. Wellness 2021, 10, 131–140. [Google Scholar] [CrossRef]
- Phytochemicals Candidates as Promising Preventives and/or Curatives for COVID-19 Infection: A Brief Review. Available online: https://www.heighpubs.org/hjbm/ibm-aid1019.php (accessed on 31 July 2021).
- Li, S.; Cheng, C.-S.; Zhang, C.; Tang, G.-Y.; Tan, H.-Y.; Chen, H.-Y.; Wang, N.; Lai, A.Y.-K.; Feng, Y. Edible and Herbal Plants for the Prevention and Management of COVID-19. Front. Pharmacol. 2021, 12, 656103. [Google Scholar] [CrossRef] [PubMed]
- Tallei, T.E.; Tumilaar, S.G.; Niode, N.J.; Fatimawali; Kepel, B.J.; Idroes, R.; Effendi, Y.; Alam Sakib, S.; Bin Emran, T. Potential of Plant Bioactive Compounds as SARS-CoV-2 Main Protease (Mpro) and Spike (S) Glycoprotein Inhibitors: A Molecular Docking Study. Scientifica 2020, 2020, 6307457. [Google Scholar] [CrossRef] [PubMed]
- Mattioli, A.V.; Puviani, M.B.; Nasi, M.; Farinetti, A. COVID-19 pandemic: The effects of quarantine on cardiovascular risk. Eur. J. Clin. Nutr. 2020, 74, 852–855. [Google Scholar] [CrossRef] [PubMed]
- Bennett, G.; Young, E.; Butler, I.; Coe, S. The Impact of Lockdown During the COVID-19 Outbreak on Dietary Habits in Various Population Groups: A Scoping Review. Front. Nutr. 2021, 8, 626432. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Quintela, A.; Milton-Laskibar, I.; Trepiana, J.; Gómez-Zorita, S.; Kajarabille, N.; Léniz, A.; González, M.; Portillo, M.P. Key Aspects in Nutritional Management of COVID-19 Patients. J. Clin. Med. 2020, 9, 2589. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.-J.; Rabheru, K.; Peisah, C.; Reichman, W.; Ikeda, M. Loneliness and social isolation during the COVID-19 pandemic. Int. Psychogeriatr. 2020, 32, 1217–1220. [Google Scholar] [CrossRef]
- Pérez-Rodrigo, C.; Citores, M.G.; Bárbara, G.H.; Ruiz-Litago, F.; Sáenz, L.C.; Arija, V.; López-Sobaler, A.; de Victoria, E.M.; Ortega, R.; Partearroyo, T.; et al. Patterns of Change in Dietary Habits and Physical Activity during Lockdown in Spain due to the COVID-19 Pandemic. Nutrients 2021, 13, 300. [Google Scholar] [CrossRef]
- Sánchez-Sánchez, E.; Ramírez-Vargas, G.; Avellaneda-López, Y.; Orellana-Pecino, J.I.; García-Marín, E.; Díaz-Jimenez, J. Eating Habits and Physical Activity of the Spanish Population during the COVID-19 Pandemic Period. Nutrients 2020, 12, 2826. [Google Scholar] [CrossRef]
- Molina-Montes, E.; Uzhova, I.; Verardo, V.; Artacho, R.; García-Villanova, B.; Guerra-Hernández, E.J.; Kapsokefalou, M.; Malisova, O.; Vlassopoulos, A.; Katidi, A.; et al. Impact of COVID-19 confinement on eating behaviours across 16 European countries: The COVIDiet cross-national study. Food Qual. Prefer. 2021, 93, 104231. [Google Scholar]
- Izzo, L.; Santonastaso, A.; Cotticelli, G.; Federico, A.; Pacifico, S.; Castaldo, L.; Colao, A.; Ritieni, A. An Italian Survey on Dietary Habits and Changes during the COVID-19 Lockdown. Nutrients 2021, 13, 1197. [Google Scholar] [CrossRef]
- Grant, F.; Scalvedi, M.; Scognamiglio, U.; Turrini, A.; Rossi, L. Eating Habits during the COVID-19 Lockdown in Italy: The Nutritional and Lifestyle Side Effects of the Pandemic. Nutrients 2021, 13, 2279. [Google Scholar] [CrossRef]
- Di Renzo, L.; Gualtieri, P.; Pivari, F.; Soldati, L.; Attinà, A.; Cinelli, G.; Leggeri, C.; Caparello, G.; Barrea, L.; Scerbo, F.; et al. Eating habits and lifestyle changes during COVID-19 lockdown: An Italian survey. J. Transl. Med. 2020, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Scarmozzino, F.; Visioli, F. Covid-19 and the Subsequent Lockdown Modified Dietary Habits of Almost Half the Population in an Italian Sample. Foods 2020, 9, 675. [Google Scholar] [CrossRef] [PubMed]
- Mascherini, G.; Catelan, D.; Pellegrini-Giampietro, D.E.; Petri, C.; Scaletti, C.; Gulisano, M. Changes in physical activity levels, eating habits and psychological well-being during the Italian COVID-19 pandemic lockdown: Impact of socio-demographic factors on the Florentine academic population. PLoS ONE 2021, 16, e0252395. [Google Scholar] [CrossRef] [PubMed]
- Sofo, A.; Sofo, A. Converting Home Spaces into Food Gardens at the Time of Covid-19 Quarantine: All the Benefits of Plants in this Difficult and Unprecedented Period. Hum. Ecol. 2020, 48, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Marty, L.; de Lauzon-Guillain, B.; Labesse, M.; Nicklaus, S. Food choice motives and the nutritional quality of diet during the COVID-19 lockdown in France. Appetite 2020, 157, 105005. [Google Scholar] [CrossRef]
- Rolland, B.; Haesebaert, F.; Zante, E.; Benyamina, A.; Haesebaert, J.; Franck, N. Global Changes and Factors of Increase in Caloric/Salty Food Intake, Screen Use, and Substance Use during the Early COVID-19 Containment Phase in the General Population in France: Survey Study. JMIR Public Health Surveill. 2020, 6, e19630. [Google Scholar] [CrossRef]
- Cherikh, F.; Frey, S.; Bel, C.; Attanasi, G.; Alifano, M.; Iannelli, A. Behavioral Food Addiction During Lockdown: Time for Awareness, Time to Prepare the Aftermath. Obes. Surg. 2020, 30, 3585–3587. [Google Scholar] [CrossRef] [PubMed]
- Philippe, K.; Chabanet, C.; Issanchou, S.; Monnery-Patris, S. Child eating behaviors, parental feeding practices and food shopping motivations during the COVID-19 lockdown in France: (How) did they change? Appetite 2021, 161, 105132. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, C.; Arija, V.; Aretouli, E.; Tsilidis, K.K.; Bulló, M. Comparing eating behaviours, and symptoms of depression and anxiety between Spain and Greece during the COVID-19 outbreak: Cross-sectional analysis of two different confinement strategies. Eur. Eat. Disord. Rev. 2020, 28. [Google Scholar] [CrossRef]
- Panagiotakos, D.; Kosti, R.I.; Pitsavos, C. How will the way we live look different in the wake of the COVID-19 pandemic? A nutrition survey in Greece. Nutr. Health 2021. [Google Scholar] [CrossRef] [PubMed]
- Grigoriou, E.; Trovas, G.; Papaioannou, N.; Dontas, I.; Makris, K.; Apostolou-Karampelis, K.; Dedoussis, G. Dietary Patterns of Greek Adults and Their Associations with Serum Vitamin D Levels and Heel Quantitative Ultrasound Parameters for Bone Health. Nutrients 2020, 12, 123. [Google Scholar] [CrossRef] [Green Version]
- Giacalone, D.; Frøst, M.B.; Rodríguez-Pérez, C. Reported Changes in Dietary Habits During the COVID-19 Lockdown in the Danish Population: The Danish COVIDiet Study. Front. Nutr. 2020, 7, 592112. [Google Scholar] [CrossRef]
- Janssen, M.; Chang, B.P.I.; Hristov, H.; Pravst, I.; Profeta, A.; Millard, J. Changes in Food Consumption during the COVID-19 Pandemic: Analysis of Consumer Survey Data From the First Lockdown Period in Denmark, Germany, and Slovenia. Front. Nutr. 2021, 8, 635859. [Google Scholar] [CrossRef]
- Błaszczyk-Bębenek, E.; Jagielski, P.; Bolesławska, I.; Jagielska, A.; Nitsch-Osuch, A.; Kawalec, P. Nutrition Behaviors in Polish Adults before and during COVID-19 Lockdown. Nutrients 2020, 12, 3084. [Google Scholar] [CrossRef]
- Sidor, A.; Rzymski, P. Dietary Choices and Habits during COVID-19 Lockdown: Experience from Poland. Nutrients 2020, 12, 1657. [Google Scholar] [CrossRef]
- Yu, B.; Zhang, D.; Yu, W.; Luo, M.; Yang, S.; Jia, P. Impacts of lockdown on dietary patterns among youths in China: The COVID-19 Impact on Lifestyle Change Survey. Public Health Nutr. 2021, 24, 3221–3232. [Google Scholar] [CrossRef]
- Naja, F.; Hamadeh, R. Nutrition amid the COVID-19 pandemic: A multi-level framework for action. Eur. J. Clin. Nutr. 2020, 74, 1117–1121. [Google Scholar] [CrossRef] [Green Version]
- Bin Zarah, A.; Enriquez-Marulanda, J.; Andrade, J.M. Relationship between Dietary Habits, Food Attitudes and Food Security Status among Adults Living within the United States Three Months Post-Mandated Quarantine: A Cross-Sectional Study. Nutrients 2020, 12, 3468. [Google Scholar] [CrossRef] [PubMed]
- Pertuz-Cruz, S.L.; Molina-Montes, E.; Rodríguez-Pérez, C.; Guerra-Hernández, E.J.; de Rangel, O.P.C.; Artacho, R.; Verardo, V.; Ruiz-Lopez, M.D.; García-Villanova, B. Exploring Dietary Behavior Changes Due to the COVID-19 Confinement in Colombia: A National and Regional Survey Study. Front. Nutr. 2021, 8, 644800. [Google Scholar] [CrossRef] [PubMed]
- Skotnicka, M.; Karwowska, K.; Kłobukowski, F.; Wasilewska, E.; Małgorzewicz, S. Dietary Habits before and during the COVID-19 Epidemic in Selected European Countries. Nutrients 2021, 13, 1690. [Google Scholar] [CrossRef] [PubMed]
- Bakaloudi, D.R.; Jeyakumar, D.T.; Jayawardena, R.; Chourdakis, M. The impact of COVID-19 lockdown on snacking habits, fast-food and alcohol consumption: A systematic review of the evidence. Clin. Nutr. 2021. [Google Scholar] [CrossRef] [PubMed]
- Bakhsh, M.A.; Khawandanah, J.; Naaman, R.K.; Alashmali, S. The impact of COVID-19 quarantine on dietary habits and physical activity in Saudi Arabia: A cross-sectional study. BMC Public Health 2021, 21, 1487. [Google Scholar] [CrossRef]
- Huber, B.C.; Steffen, J.; Schlichtiger, J.; Brunner, S. Altered nutrition behavior during COVID-19 pandemic lockdown in young adults. Eur. J. Nutr. 2020, 60, 2593–2602. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Jiang, W.; Zheng, C.; Shao, D.; Liu, Y.; Huang, S.; Han, J.; Ding, J.; Tao, Y.; Li, M. Oral delivery of bacteria: Basic principles and biomedical applications. J. Control. Release 2020, 327, 801–833. [Google Scholar] [CrossRef] [PubMed]
- Serrem, K.; Illés, C.B.; Serrem, C.; Atubukha, B.; Dunay, A. Food safety and sanitation challenges of public university students in a developing country. Food Sci Nutr. 2021, 9, 4287–4297. [Google Scholar]
- Rodríguez-Pérez, C.; Molina-Montes, E.; Verardo, V.; Artacho, R.; García-Villanova, B.; Guerra-Hernández, E.J.; Ruíz-López, M.D. Changes in Dietary Behaviours during the COVID-19 Outbreak Confinement in the Spanish COVIDiet Study. Nutrients 2020, 12, 1730. [Google Scholar] [CrossRef]
- Min, S.; Xiang, C.; Zhang, X.-H. Impacts of the COVID-19 pandemic on consumers’ food safety knowledge and behavior in China. J. Integr. Agric. 2020, 19, 2926–2936. [Google Scholar] [CrossRef]
- Lange, K.W.; Nakamura, Y. Lifestyle factors in the prevention of COVID-19. Glob. Health J. 2020, 4, 146–152. [Google Scholar] [CrossRef]
- Mentella, M.; Scaldaferri, F.; Gasbarrini, A.; Miggiano, G. The Role of Nutrition in the COVID-19 Pandemic. Nutrients 2021, 13, 1093. [Google Scholar] [CrossRef]
- Woods, J.A.; Hutchinson, N.T.; Powers, S.K.; Roberts, W.O.; Gomez-Cabrera, M.C.; Radak, Z.; Berkes, I.; Boros, A.; Boldogh, I.; Leeuwenburgh, C.; et al. The COVID-19 pandemic and physical activity. Sports Med. Health Sci. 2020, 2, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Chick, J. Alcohol and COVID-19. Alcohol Alcohol. 2020, 55, 341–342. [Google Scholar] [CrossRef] [PubMed]
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Al-Rashed, F.; Sindhu, S.; Al Madhoun, A.; Alghaith, A.; Azim, R.; Al-Mulla, F.; Ahmad, R. Short Sleep Duration and Its Association with Obesity and Other Metabolic Risk Factors in Kuwaiti Urban Adults. Nat. Sci. Sleep 2021, 13, 1225. [Google Scholar] [CrossRef]
- Silva, E.D.S.M.E.; Ono, B.H.V.S.; Souza, J.C. Sleep and immunity in times of COVID-19. Revista da Associação Médica Brasileira 2020, 66, 143–147. [Google Scholar] [CrossRef]
- Fidancı, I.; Aksoy, H.; Taci, D.Y.; Fidancı, I.; Başer, D.A.; Cankurtaran, M. Evaluation of the effect of the COVID-19 pandemic on sleep disorders and nutrition in children. Int. J. Clin. Pract. 2021, e14170. [Google Scholar] [CrossRef]
- Kingsbury, J.; Lake, R.; Hewitt, J.; Smit, E.; King, N. Potential for foodborne transmission of Covid-19: Literature review update. Update 2020, 1–78. Available online: https://mia.co.nz/assets/Covid-19/2021-01-14-NZFSSRC-COVID-19-foodborne-transmission-final-Kingsbury.pdf (accessed on 31 August 2021).
- Han, J.; Zhang, X.; He, S.; Jia, P. Can the coronavirus disease be transmitted from food? A review of evidence, risks, policies and knowledge gaps. Environ. Chem. Lett. 2020, 19, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Olaimat, A.N.; Shahbaz, H.M.; Fatima, N.; Munir, S.; Holley, R.A. Food Safety During and After the Era of COVID-19 Pandemic. Front. Microbiol. 2020, 11, 1854. [Google Scholar] [CrossRef] [PubMed]
- Ceylan, Z.; Meral, R.; Cetinkaya, T. Relevance of SARS-CoV-2 in food safety and food hygiene: Potential preventive measures, suggestions and nanotechnological approaches. VirusDisease 2020, 31, 154–160. [Google Scholar] [CrossRef]
- Ceniti, C.; Tilocca, B.; Britti, D.; Santoro, A.; Costanzo, N. Food Safety Concerns in “COVID-19 Era”. Microbiol. Res. 2021, 12, 6. [Google Scholar] [CrossRef]
- Mardones, F.O.; Rich, K.M.; Boden, L.A.; Moreno-Switt, A.I.; Caipo, M.L.; Zimin-Veselkoff, N.; Alateeqi, A.M.; Baltenweck, I. The COVID-19 Pandemic and Global Food Security. Front. Vet. Sci. 2020, 7, 578508. [Google Scholar] [CrossRef]
- Chowdhury, T.; Sarkar, A.; Paul, S.K.; Moktadir, A. A case study on strategies to deal with the impacts of COVID-19 pandemic in the food and beverage industry. Oper. Manag. Res. 2020, 1–13. [Google Scholar] [CrossRef]
- Nakat, Z.; Bou-Mitri, C. COVID-19 and the food industry: Readiness assessment. Food Control 2020, 121, 107661. [Google Scholar] [CrossRef]
- Hossain, S.T. Impacts of COVID-19 on the Agri-food Sector: Food Security Policies of Asian Productivity Organization Members. J. Agric. Sci. Sri Lanka 2020, 15, 116. [Google Scholar] [CrossRef]
- Telukdarie, A.; Munsamy, M.; Mohlala, P. Analysis of the Impact of COVID-19 on the Food and Beverages Manufacturing Sector. Sustainability 2020, 12, 9331. [Google Scholar] [CrossRef]
- Haider, N.; Osman, A.Y.; Gadzekpo, A.; Akipede, G.O.; Asogun, D.; Ansumana, R.; Lessells, R.J.; Khan, P.; Hamid, M.M.A.; Yeboah-Manu, D.; et al. Lockdown measures in response to COVID-19 in nine sub-Saharan African countries. BMJ Glob. Health 2020, 5, e003319. [Google Scholar] [CrossRef]
- Madeira, A.; Palrão, T.; Mendes, A. The Impact of Pandemic Crisis on the Restaurant Business. Sustainability 2020, 13, 40. [Google Scholar] [CrossRef]
- Aday, S.; Aday, M.S. Impact of COVID-19 on the food supply chain. Food Qual. Saf. 2020, 4, 167–180. [Google Scholar] [CrossRef]
- Pexara, A.; Govaris, A. Foodborne Viruses and Innovative Non-Thermal Food-Processing Technologies. Foods 2020, 9, 1520. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, M.F.; Sancho, A.N.; Mendoza, L.M.; Mota, C.R.; Verbyla, M.E. Systematic review and meta-analysis of time-temperature pathogen inactivation. Int. J. Hyg. Environ. Health 2020, 230, 113595. [Google Scholar] [CrossRef] [PubMed]
- Anelich, L.E.C.M.; Lues, R.; Farber, J.M.; Parreira, V.R. SARS-CoV-2 and Risk to Food Safety. Front. Nutr. 2020, 7, 580551. [Google Scholar] [CrossRef] [PubMed]
- Gamble, A.; Fischer, R.J.; Morris, D.H.; Yinda, C.K.; Munster, V.J.; Lloyd-Smith, J.O. Heat-Treated Virus Inactivation Rate Depends Strongly on Treatment Procedure: Illustration with SARS-CoV-2. Appl. Environ. Microbiol. 2021, 87, AEM0031421. [Google Scholar] [CrossRef]
- Lugo-Morin, D.R. Global Food Security in a Pandemic: The Case of the New Coronavirus (COVID-19). World 2020, 1, 13. [Google Scholar] [CrossRef]
- Bakalis, S.; Valdramidis, V.P.; Argyropoulos, D.; Ahrne, L.; Chen, J.; Cullen, P.; Cummins, E.; Datta, A.K.; Emmanouilidis, C.; Foster, T.; et al. Perspectives from CO+RE: How COVID-19 changed our food systems and food security paradigms. Curr. Res. Food Sci. 2020, 3, 166–172. [Google Scholar] [CrossRef]
- Jafri, A.; Mathe, N.; Aglago, E.K.; Konyole, S.O.; Ouedraogo, M.; Audain, K.; Zongo, U.; Laar, A.K.; Johnson, J.; Sanou, D. Food availability, accessibility and dietary practices during the COVID-19 pandemic: A multi-country survey. Public Health Nutr. 2021, 24, 1798–1805. [Google Scholar] [CrossRef] [PubMed]
- Rejeb, A.; Rejeb, K.; Keogh, J.G. Covid-19 and the food chain? Impacts and future research trends. Logforum 2020, 16, 475–485. [Google Scholar] [CrossRef]
- Mouloudj, K.; Bouarar, A.C.; Fechit, H. The Impact of COVID-19 Pandemic on Food Security. Les Cah. du CREAD 2020, 36, 159–184. Available online: https://www.asjp.cerist.dz/en/article/120912 (accessed on 31 July 2021).
- Nosratabadi, S.; Khazami, N.; Abdallah, M.; Lackner, Z.; Band, S.S.; Mosavi, A.; Mako, C. Social Capital Contributions to Food Security: A Comprehensive Literature Review. Foods 2020, 9, 1650. [Google Scholar] [CrossRef]
- Chiwona-Karltun, L.; Amuakwa-Mensah, F.; Wamala-Larsson, C.; Amuakwa-Mensah, S.; Abu Hatab, A.; Made, N.; Taremwa, N.K.; Melyoki, L.; Rutashobya, L.K.; Madonsela, T.; et al. COVID-19: From health crises to food security anxiety and policy implications. Ambio 2021, 50, 794–811. [Google Scholar] [CrossRef]
- COVID-19 and Food Safety: Guidance for Food Businesses: Interim Guidance. Available online: https://www.who.int/publications/i/item/covid-19-and-food-safety-guidance-for-food-businesses (accessed on 31 July 2021).
- Butterick, M.; Charlwood, A. HRM and the COVID-19 pandemic: How can we stop making a bad situation worse? Hum. Resour. Manag. J. 2021, 1–10. [Google Scholar] [CrossRef]
- Dyal, J.W.; Grant, M.P.; Broadwater, K.; Bjork, A.; Waltenburg, M.A.; Gibbins, J.D.; Hale, C.; Silver, M.; Fischer, M.; Steinberg, J.; et al. COVID-19 Among Workers in Meat and Poultry Processing Facilities ―19 States, April 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69. [Google Scholar] [CrossRef] [PubMed]
- Leone, L.A.; Fleischhacker, S.; Anderson-Steeves, B.; Harper, K.; Winkler, M.; Racine, E.; Baquero, B.; Gittelsohn, J. Healthy Food Retail during the COVID-19 Pandemic: Challenges and Future Directions. Int. J. Environ. Res. Public Health 2020, 17, 7397. [Google Scholar] [CrossRef] [PubMed]
- Zuber, S.; Brüssow, H. COVID 19: Challenges for virologists in the food industry. Microb. Biotechnol. 2020, 13, 1689–1701. [Google Scholar] [CrossRef]
- Shahbaz, M.; Bilal, M.; Moiz, A.; Zubair, S.; Iqbal, H.M. Food Safety and COVID-19: Precautionary Measures to Limit the Spread of Coronavirus at Food Service and Retail Sector. J. Pure Appl. Microbiol. 2020, 14, 749–756. [Google Scholar] [CrossRef]
- Al Amin, M.; Arefin, M.S.; Alam, M.R.; Ahammad, T.; Hoque, M.R. Using Mobile Food Delivery Applications during COVID-19 Pandemic: An Extended Model of Planned Behavior. J. Food Prod. Mark. 2021, 27, 105–126. [Google Scholar] [CrossRef]
- Nguyen, T.H.D.; Vu, D.C. Food Delivery Service During Social Distancing: Proactively Preventing or Potentially Spreading Coronavirus Disease–2019? Disaster Med. Public Health Prep. 2020, 14, e9–e10. [Google Scholar] [CrossRef] [PubMed]
- Ouabdesselam, L.; Sayad, A. Food Packaging and COVID-19. Eur. J. Basic Med Sci. 2021, 11, 23–26. [Google Scholar] [CrossRef]
- Kitz, R.; Walker, T.; Charlebois, S.; Music, J. Food packaging during the COVID-19 pandemic: Consumer perceptions. Int. J. Consum. Stud. 2021. [Google Scholar] [CrossRef] [PubMed]
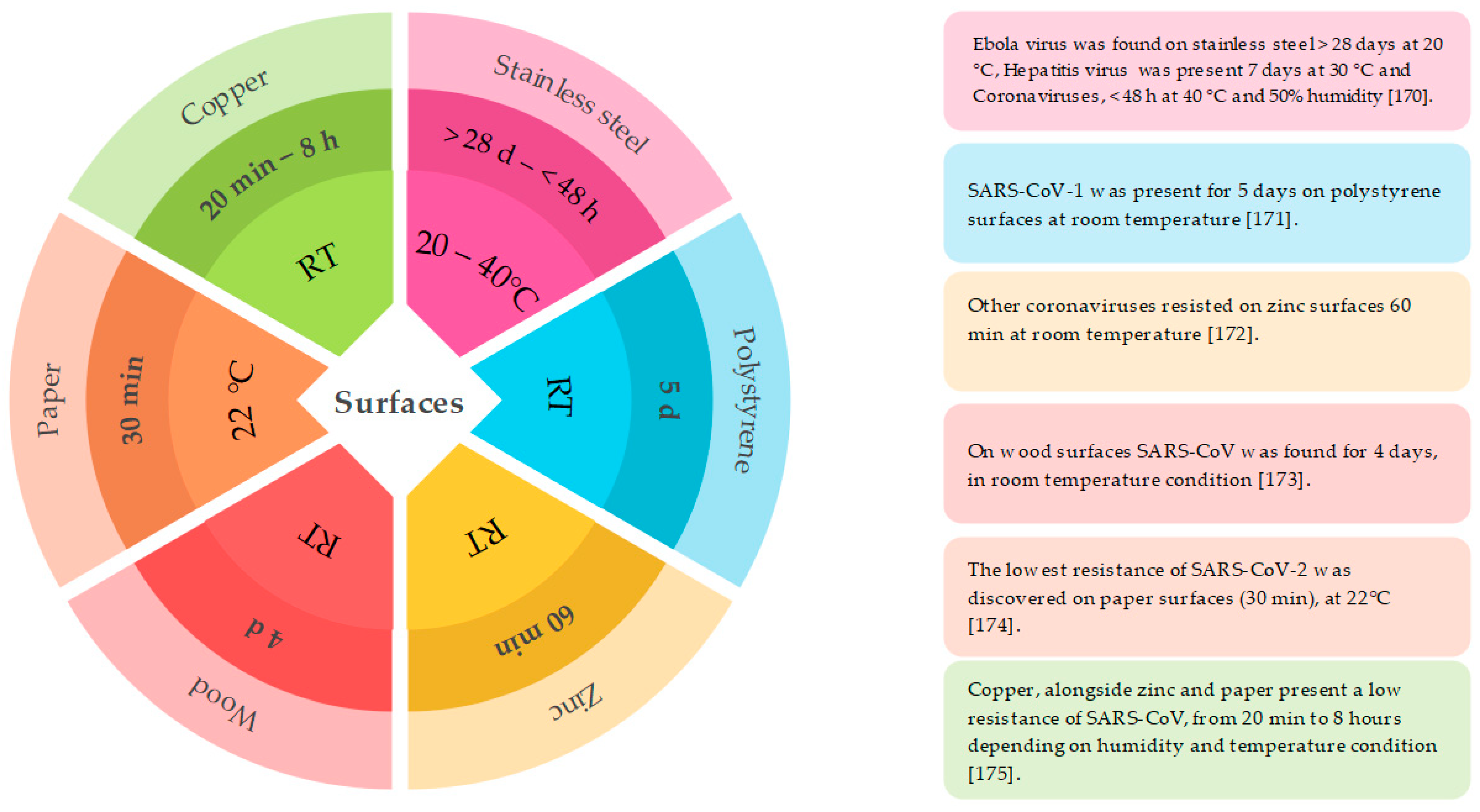
- Riddell, S.; Goldie, S.; Hill, A.; Eagles, D.; Drew, T.W. The effect of temperature on persistence of SARS-CoV-2 on common surfaces. Virol. J. 2020, 17, 145. [Google Scholar] [CrossRef]
- Xue, X.; Ball, J.K.; Alexander, C.; Alexander, M.R. All Surfaces Are Not Equal in Contact Transmission of SARS-CoV-2. Matter 2020, 3, 1433–1441. [Google Scholar] [CrossRef]
- Fiorillo, L.; Cervino, G.; Matarese, M.; D’Amico, C.; Surace, G.; Paduano, V.; Fiorillo, M.T.; Moschella, A.; La Bruna, A.; Romano, G.L.; et al. COVID-19 Surface Persistence: A Recent Data Summary and Its Importance for Medical and Dental Settings. Int. J. Environ. Res. Public Health 2020, 17, 3132. [Google Scholar] [CrossRef]
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020, 104, 246–251. [Google Scholar] [CrossRef] [Green Version]
- Chi, Y.; Wang, Q.; Chen, G.; Zheng, S. The Long-Term Presence of SARS-CoV-2 on Cold-Chain Food Packaging Surfaces Indicates a New COVID-19 Winter Outbreak: A Mini Review. Front. Public Health 2021, 9, 650493. [Google Scholar] [CrossRef]
- Choi, H.; Chatterjee, P.; Coppin, J.D.; Martel, J.A.; Hwang, M.; Jinadatha, C.; Sharma, V.K. Current understanding of the surface contamination and contact transmission of SARS-CoV-2 in healthcare settings. Environ. Chem. Lett. 2021, 19, 1935–1944. [Google Scholar] [CrossRef]
- Saha, N.C. COVID 19—Its Impact on Packaging Research. J. Packag. Technol. Res. 2020, 4, 217–218. [Google Scholar] [CrossRef] [PubMed]

| Vitamins and Minerals | Food Sources | Actions | Role | Reference |
|---|---|---|---|---|
| Vitamin A | Meat, poultry, fish, dairy, eggs | Shows efficiency for immune function and resistance to infection | Immunomodulatory, Anti-inflammatory | [76] |
| Vitamin B1 (thiamine) | Meat, poultry, fish, whole-grains, brown rice dried beans, soybeans, nuts, | Eliminates the SARS-CoV-2 virus by triggering cell-mediated and antibody-mediated immunity | Supports immune response | [77] |
| Vitamin B2 (riboflavin) | Calf liver, fish, nuts, wild rice, dark green leafy vegetables, mushrooms, certain fruits and legumes, beer, yeast, milk, cheese, egg, | Reduces number of pathogens in the blood plasma of COVID-19 patients, and reduce the risk of transfusion–transmission of COVID-19. | Supports immune response | [77,78] |
| Vitamin B3 (niacin) | Meat, liver, beans | Reduces viral infection & stimulates defense mechanisms Reduces neutrophil infiltration in patients with ventilator-induced lung injury | Supports immune response | [77] |
| Vitamin B6 (pyridoxine) | Cereal grains, vegetables (carrots, spinach, peas), milk, potatoes, eggs, cheese, fish, liver, meat, | Relieves COVID-19 symptoms by improving immune response, supporting endothelial integrity, preventing hyper-coagulability & reducing pro-inflammatory cytokines | Supports immune response | [77] |
| Vitamin B12 (cyanocobalamin/cobalamin) | Meat, milk, egg, fish, and shellfish | Essential role in improved immune system function | Supports immune response | [77] |
| Vitamin C | Citrus fruits kiwi, tomato, pineapple, kale, spinach, beef liver, milk, cabbage, broccoli, chicken, oysters, strawberries | Reduces symptom duration Reduces mortality Prevents COVID-19 progression Decreases risk of respiratory failure requiring a ventilator Reduces death rate & dependency on ventilator | Antioxidant, immunomodulatory | [79] |
| Vitamin D | Wild mushroom, fungi, fortified, bread fortified orange juice, milk, eggs, cheese, yogurt, fortified margarine | Improves prognosis in older patients Prevents respiratory infections | Antioxidant, immunomodulatory | [80] |
| Vitamin E | Vegetable oils, Nuts, seeds, avocado, green leafy vegetables, mango, salmon fortified cereals | Antioxidant defense Role in immune response & to reduce viral pathogenicity | Antioxidant, immunomodulatory | [81] |
| Selenium | Whole grains, nuts, mushrooms, dairy products, poultry, cereals, red meat, seafood | Essential for protection against viral infection | Antioxidant, ROS balance in inflammation, immune-cell function | [79] |
| Zinc | Fortified breakfast cereal, nuts, beans, poultry, red meat, whole grains, crustaceans, mollusks | Reduces inflammatory reaction Increases ventilator-free days Organ failure-free days Acute inflammation-free days | Antioxidant, anti-inflammatory, reduces ROS in viral infection | [80] |
| Food Source | Compounds | Biological Activities | Reference |
|---|---|---|---|
| Onion | Quercetin, thiosulfinates, & anthocyanins | Antioxidant | [82] |
| Citrus fruits | Hesperidin | Antioxidant, anti-inflammatory, antiviral | [83] |
| Garlic | Diallyl disulphide, alliin, polyphenols, proteins | Antioxidant, antiviral | [84] |
| Honey | p-coumaric acid, ellagic acid | Antimicrobial, antiviral | [85] |
| Tea Plant | Gallic acid, theaflavin-3,3′-digallate, quercetin, catechins | Antioxidant, antiviral, immunomodulatory | [86] |
| Cranberry | Myricetin | Antiviral | [87] |
| Barberry | Berbamine, berberine | Anticancer | [82] |
| Turmeric | Curcumin | Anti-inflammatory | [83] |
| Soybean | Flavonoids, Isoflavones, phytosterols, saponins & organic acid | Antioxidant | [82] |
| Banana | Bananin | Antiviral | [88] |
| Long pepper, black pepper | Piperine | Anticancer | [82] |
| Plum | Anthocyanins, protocatechuic acid | Antioxidant | [82] |
| Grapes, berries | Quercetin | Antioxidant, anti-inflammatory | [89] |
| Kale | Kaempferol | Anti-inflammatory | [90] |
| Avocado, pistachio, almond | Β-sitosterol | Anti-inflammatory | [89] |
| Mango | Flavonoids, xanthones, phenolic acids, triterpenes | Antioxidant, antiviral | [82] |
| Nuts, seeds | Stigmasterol | Antiviral | [89] |
| Red grape | Resveratrol | Anti-inflammatory | [89] |
| Colombia [119] | Poland [120] | Italy [100] | France [121] | Saudi Arabia [122] | Germany [123] | China [124] | Spain [125] | |
|---|---|---|---|---|---|---|---|---|
| Ate more | ||||||||
| Increase | 45% | - | - | - | - | 40% | 31% | 64% |
| Decrease | 20% | - | - | - | - | 21% | 17% | 36% |
| As before | 35% | - | - | - | - | 39%% | 52% | - |
| Weight gain | ||||||||
| Yes | 22% | 40% | 37% | 38% | 38% | 31% | - | 13% |
| No | 38% | 10% | 52% | 18% | 26% | 16% | - | 47% |
| Unknown | 40% | 50% | 11% | 44% | 36% | 53% | - | 40% |
| Fast food | ||||||||
| Increase | 21% | 8% | - | - | - | - | - | 5% |
| Decrease | 34% | 37% | - | - | - | - | - | 35% |
| As before | 45% | 55% | - | - | - | - | - | 60% |
| Snacking | ||||||||
| Increase | 48% | 30% | 33% | 24% | 45% | - | 25% | 37% |
| Decrease | 22% | 10% | 11% | 18% | 19% | - | 37% | 16% |
| As before | 30% | 60% | 56% | 58% | 36% | - | 38% | 47% |
| Meals out of home | ||||||||
| Increase | - | - | - | - | - | 7% | - | 21% |
| Decrease | - | - | - | - | - | 80% | - | 50% |
| As before | - | - | - | - | - | 13% | - | 29% |
| Alcohol intake | ||||||||
| Increase | 7% | 18% | 16% | 12% | - | - | 26% | 11% |
| Decrease | 18% | 11% | 13% | 12% | - | - | 39% | 57% |
| As before | 22% | 71% | 71% | 39% | - | - | 35% | 32% |
| Never | 53% | - | - | 23% | - | - | - | - |
| Water intake | ||||||||
| Increase | 36% | - | 20% | - | 57% | - | - | - |
| Decrease | 26% | - | 8% | - | 7% | - | - | - |
| As before | 38% | - | 72% | - | 36% | - | - | - |
| Physical activity | ||||||||
| Increase | 23% | - | 36% | 26 | 27% | - | - | 16% |
| Decrease | 48% | - | 11% | 50% | 52% | - | - | 59% |
| As before | 18% | - | 16% | 24% | 21% | - | - | 19% |
| Never | 11% | - | 37% | - | - | - | - | 6% |
| Home cooking | ||||||||
| Increase | 66% | - | - | - | 73% | 96% | 65% | 45% |
| Decrease | 5% | - | - | - | 4% | 4% | 9% | 4% |
| As before | 23% | - | - | - | 23% | - | 26% | 51% |
| Never | 6% | - | - | - | - | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filip, R.; Anchidin-Norocel, L.; Gheorghita, R.; Savage, W.K.; Dimian, M. Changes in Dietary Patterns and Clinical Health Outcomes in Different Countries during the SARS-CoV-2 Pandemic. Nutrients 2021, 13, 3612. https://doi.org/10.3390/nu13103612
Filip R, Anchidin-Norocel L, Gheorghita R, Savage WK, Dimian M. Changes in Dietary Patterns and Clinical Health Outcomes in Different Countries during the SARS-CoV-2 Pandemic. Nutrients. 2021; 13(10):3612. https://doi.org/10.3390/nu13103612
Chicago/Turabian StyleFilip, Roxana, Liliana Anchidin-Norocel, Roxana Gheorghita, Wesley K. Savage, and Mihai Dimian. 2021. "Changes in Dietary Patterns and Clinical Health Outcomes in Different Countries during the SARS-CoV-2 Pandemic" Nutrients 13, no. 10: 3612. https://doi.org/10.3390/nu13103612
APA StyleFilip, R., Anchidin-Norocel, L., Gheorghita, R., Savage, W. K., & Dimian, M. (2021). Changes in Dietary Patterns and Clinical Health Outcomes in Different Countries during the SARS-CoV-2 Pandemic. Nutrients, 13(10), 3612. https://doi.org/10.3390/nu13103612








