Processed Meat Consumption and the Risk of Cancer: A Critical Evaluation of the Constraints of Current Evidence from Epidemiological Studies
Abstract
:1. Introduction
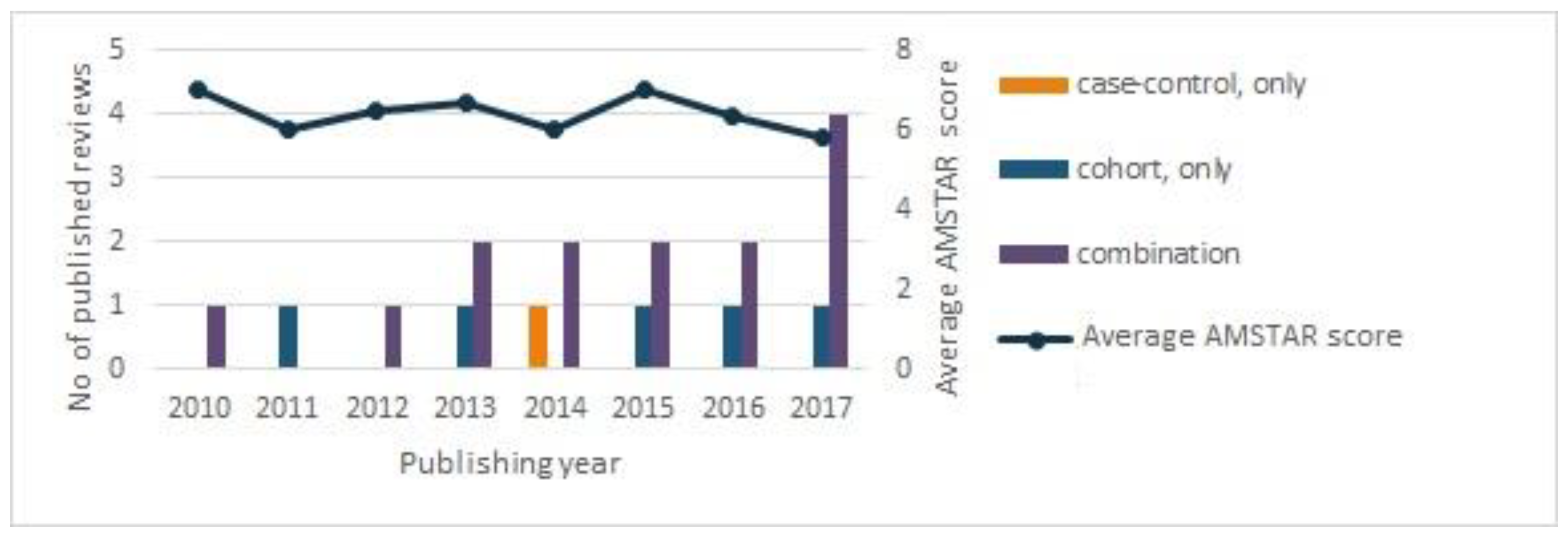
2. Methodological Limitations of Systematic Reviews on Processed Meat and the Risk of Cancer
3. Methodological Limitations of the Primary Studies on Processed Meat and Cancer
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Micha, R.; Khatibzadeh, S.; Shi, P.; Andrews, K.G.; Engell, R.E.; Mozaffarian, D. Global, regional and national consumption of major food groups in 1990 and 2010: A systematic analysis including 266 country-specific nutrition surveys worldwide. BMJ Open 2015, 5, e008705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mejborn, H.; Biltoft-Jensen, A.; Hansen, M.; Licht, T.R.; Olesen, P.T.; Sørensen, I.K. Mechanisms behind Cancer Risks Associated with Consumption of Red and Processed Meat; National Food Institute, Technical Unicersity of Denmark: Søborg, Denmark, 2016. [Google Scholar]
- World Health Organization. Diet, Nutrition and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- European Code against Cancer: IARC/WHO. 2017. Available online: https://cancer-code-europe.iarc.fr/index.php/en (accessed on 13 October 2021).
- International Agency for Research on Cancer. Red Meat and Processed Meat. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; Report No.; International Agency for Research on Cancer: Lyon, France, 2018; Volume 114. [Google Scholar]
- Vieira, A.; Abar, L.; Chan, D.; Vingeliene, S.; Polemiti, E.; Stevens, C.; Greenwood, D.; Norat, T. Foods and beverages and colorectal cancer risk: A systematic review and meta-analysis of cohort studies, an update of the evidence of the WCRF-AICR Continuous Update Project. Ann. Oncol. 2017, 28, 1788–1802. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; U.S. Department of Health and Human Services and U.S. Department of Agriculture: Washington, DC, USA, 2015. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 13 October 2021).
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025, 9th ed.; U.S. Department of Health and Human Services: Washington, DC, USA; U.S. Department of Agriculture: Washington, DC, USA, 2020. Available online: DietaryGuidelines.gov (accessed on 13 October 2021).
- U.S. Department of Agriculture and U.S. Department of Health and Human Services. The Dietary Patterns Subcomittee. Nutrition Evidence Systrematic Review: What Is the Relationship between Dietary Patterns Consumed and Risk of Certain Types of Cancer? 2021. Available online: https://nesr.usda.gov/2020-dietary-guidelines-advisory-committee-systematic-reviews/dietary-patterns-subcommittee/dietary-patterns-cancer (accessed on 13 October 2021).
- Beresford, S.A.; Johnson, K.C.; Ritenbaugh, C.; Lasser, N.L.; Snetselaar, L.G.; Black, H.R.; Anderson, G.T.; Assaf, A.R.; Bassford, T.; Bowen, D.; et al. Low-fat dietary pattern and risk of colorectal cancer: The Women’s Health Initiative Randomized Controlled Dietary Modification Trial. JAMA 2006, 295, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Prentice, R.L.; Caan, B.; Chlebowski, R.T.; Patterson, R.; Kuller, L.H.; Ockene, J.K.; Margolis, K.L.; Limacher, M.C.; Manson, J.E.; Parker, L.M.; et al. Low-fat dietary pattern and risk of invasive breast cancer: The Women’s Health Initiative Randomized Controlled Dietary Modification Trial. JAMA 2006, 295, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [Green Version]
- Teicholz, N. The scientific report guiding the US dietary guidelines: Is it scientific? BMJ 2015, 351, h4962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Public Health England. The Eatwell Guide; Public Health England: London, UK, 2016.
- Nordisk, M. Nordic Nutrition Recommendations 2012: Integrating Nutrition and Physical Activity, 5th ed.; Nordic Council of Ministers: Copenhagen, Denmark, 2014. [Google Scholar]
- Johnston, B.C.; Guyatt, G.H. Causal inference, interpreting and communicating results on red and processed meat. Am. J. Clin. Nutr. 2020, 111, 1107–1108. [Google Scholar] [CrossRef]
- Johnston, B.C.; Zeraatkar, D.; Han, M.A.; Vernooij, R.W.; Valli, C.; El Dib, R.; Marshall, C.; Stover, P.J.; Fairweather-Taitt, S.; Wójcik, G.; et al. Unprocessed Red Meat and Processed Meat Consumption: Dietary Guideline Recommendations From the Nutritional Recommendations (NutriRECS) Consortium. Ann. Intern. Med. 2019, 171, 756–764. [Google Scholar] [CrossRef] [Green Version]
- Lanza, E.; Schatzkin, A.; Daston, C.; Corle, D.; Freedman, L.; Ballard-Barbash, R.; Caan, B.; Lance, P.; Marshall, J.; Iber, F.; et al. Implementation of a 4-y, high-fiber, high-fruit-and-vegetable, low-fat dietary intervention: Results of dietary changes in the Polyp Prevention Trial. Am. J. Clin. Nutr. 2001, 74, 387–401. [Google Scholar] [CrossRef] [Green Version]
- Händel, M.N.; Cardoso, I.; Rasmussen, K.M.; Rohde, J.F.; Jacobsen, R.; Nielsen, S.M.; Christensen, R.; Heitmann, B.L. Processed meat intake and chronic disease morbidity and mortality: An overview of systematic reviews and meta-analyses. PLoS ONE 2019, 14, e0223883. [Google Scholar] [CrossRef] [Green Version]
- Shea, B.J.; Grimshaw, J.M.; Wells, G.A.; Boers, M.; Andersson, N.; Hamel, C.; Porter, A.C.; Tugwell, P.; Moher, D.; Bouter, L.M. Development of AMSTAR: A measurement tool to assess the methodological quality of systematic reviews. BMC Med. Res. Methodol. 2007, 7, 10. [Google Scholar] [CrossRef] [Green Version]
- Shea, B.J.; Hamel, C.; Wells, G.A.; Bouter, L.; Kristjansson, E.; Grimshaw, J.; Henry, D.; Boers, M. AMSTAR is a reliable and valid measurement tool to assess the methodological quality of systematic reviews. J. Clin. Epidemiol. 2009, 62, 1013–1020. [Google Scholar] [CrossRef] [Green Version]
- Guyatt, G.H.; Oxman, A.D.; Schuenemann, H.J.; Tugwell, P.; Knottnerus, A. GRADE guidelines: A new series of articles in the Journal of Clinical Epidemiology. J. Clin. Epidemiol. 2011, 64, 380–382. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, Y.; Song, S.; Song, Y.; Lee, J.E. Consumption of red and processed meat and esophageal cancer risk: Meta-analysis. World J. Gastroenterol. 2013, 19, 1020–1029. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.-C.; Yang, X.; Xu, L.; Zhao, L.-J.; Tao, G.-Z.; Zhang, C.; Qin, Q.; Cai, J.; Ma, J.-X.; Mao, W.-D.; et al. Meat Consumption Is Associated with Esophageal Cancer Risk in a Meat- and Cancer-Histological-Type Dependent Manner. Dig. Dis. Sci. 2014, 59, 664–673. [Google Scholar] [CrossRef]
- Zhao, Z.; Yin, Z.; Zhao, Q. Red and processed meat consumption and gastric cancer risk: A systematic review and meta-analysis. Oncotarget 2017, 8, 30563–30575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, H.; Yang, X.; Zhang, C.; Zhu, C.; Tao, G.; Zhao, L.; Tang, S.; Shu, Z.; Cai, J.; Dai, S.; et al. Red and Processed Meat Intake Is Associated with Higher Gastric Cancer Risk: A Meta-Analysis of Epidemiological Observational Studies. PLoS ONE 2013, 8, e70955. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.; Feng, Q.; Yin, Z.; Shuang, J.; Bai, B.; Yu, P.; Guo, M.; Zhao, Q. Red and processed meat consumption and colorectal cancer risk: A systematic review and meta-analysis. Oncotarget 2017, 8, 83306–83314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Z.; Yin, Z.; Pu, Z.; Zhao, Q. Association Between Consumption of Red and Processed Meat and Pancreatic Cancer Risk: A Systematic Review and Meta-analysis. Clin. Gastroenterol. Hepatol. 2016, 15, 486–493.e10. [Google Scholar] [CrossRef] [Green Version]
- Fang, X.; Wei, J.; He, X.; An, P.; Wang, H.; Jiang, L.; Shao, D.; Liang, H.; Li, Y.; Wang, F.; et al. Landscape of dietary factors associated with risk of gastric cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. Eur. J. Cancer 2015, 51, 2820–2832. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yang, X.X.; Wu, Y.G.; Li, X.Y.; Bai, B. Meat consumption and risk of oral cavity and oropharynx cancer: A meta-analysis of observational studies. PLoS ONE 2014, 9, e95048. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Duan, F.; Zhao, X.; Song, C.; Cui, S.; Dai, L. Red Meat and Processed Meat Consumption and Nasopharyngeal Carcinoma Risk: A Dose-response Meta-analysis of Observational Studies. Nutr. Cancer 2016, 68, 1034–1043. [Google Scholar] [CrossRef]
- Luo, J.; Yang, Y.; Liu, J.; Lu, K.; Tang, Z.; Liu, P.; Liu, L.; Zhu, Y. Systematic review with meta-analysis: Meat consumption and the risk of hepatocellular carcinoma. Aliment. Pharmacol. Ther. 2014, 39, 913–922. [Google Scholar] [CrossRef] [Green Version]
- Quach, P.; El, S.R.; Gomes, J.; Krewksi, D. A systematic review of the risk factors associated with the onset and progression of primary brain tumours. Neurotoxicology 2017, 61, 214–232. [Google Scholar] [CrossRef]
- Esmaillzadeh, A.; Saneei, P.; Willett, W. Red and processed meat consumption and risk of glioma in adults: A systematic review and meta-analysis of observational studies. J. Res. Med. Sci. 2015, 20, 602–612. [Google Scholar] [CrossRef]
- Wallin, A.; Orsini, N.; Wolk, A. Red and processed meat consumption and risk of ovarian cancer: A dose-response meta-analysis of prospective studies. Br. J. Cancer 2011, 104, 1196–1201. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.S.; Wong, M.Y.; Vogtmann, E.; Tang, R.Q.; Xie, L.; Yang, Y.S.; Wu, Q.J.; Zhang, W.; Xiang, Y.B. Meat consumption and risk of lung cancer: Evidence from observational studies. Ann. Oncol. 2012, 23, 3163–3170. [Google Scholar] [CrossRef]
- Händel, M.N.; Rohde, J.F.; Jacobsen, R.; Nielsen, S.M.; Christensen, R.; Alexander, D.D.; Frederiksen, P.; Heitmann, B.L. Processed meat intake and incidence of colorectal cancer: A systematic review and meta-analysis of prospective observational studies. Eur. J. Clin. Nutr. 2020, 74, 1132–1148. [Google Scholar] [CrossRef] [PubMed]
- The Risk of Bias in Non-Randomized Studies-of Interventions (ROBINS-I) Assessment Tool. 2016. Available online: https://www.bristol.ac.uk/population-health-sciences/centres/cresyda/barr/riskofbias/robins-i/ (accessed on 13 October 2021).
- Larsson, S.C.; Wolk, A. Meat consumption and risk of colorectal cancer: A meta-analysis of prospective studies. Int. J. Cancer 2006, 119, 2657–2664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexander, D.D.; Miller, A.J.; Cushing, C.A.; Lowe, K.A. Processed meat and colorectal cancer: A quantitative review of prospective epidemiologic studies. Eur. J. Cancer Prev. 2010, 19, 328–341. [Google Scholar] [CrossRef]
- Chan, D.S.M.; Lau, R.; Aune, D.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Red and Processed Meat and Colorectal Cancer Incidence: Meta-Analysis of Prospective Studies. PLoS ONE 2011, 6, e20456. [Google Scholar] [CrossRef] [Green Version]
- Huxley, R.R.; Ansary-Moghaddam, A.; Clifton, P.; Czernichow, S.; Parr, C.L.; Woodward, M. The impact of dietary and lifestyle risk factors on risk of colorectal cancer: A quantitative overview of the epidemiological evidence. Int. J. Cancer 2009, 125, 171–180. [Google Scholar] [CrossRef]
- Johnson, C.M.; Wei, C.; Ensor, J.; Smolenski, D.J.; Amos, C.I.; Levin, B.; Berry, D.A. Meta-analyses of colorectal cancer risk factors. Cancer Causes Control. 2013, 24, 1207–1222. [Google Scholar] [CrossRef]
- Norat, T.; Lukanova, A.; Ferrari, P.; Riboli, E. Meat consumption and colorectal cancer risk: Dose-response meta-analysis of epidemiological studies. Int. J. Cancer 2001, 98, 241–256. [Google Scholar] [CrossRef]
- Pham, N.M.; Mizoue, T.; Tanaka, K.; Tsuji, I.; Tamakoshi, A.; Matsuo, K.; Wakai, K.; Nagata, C.; Inoue, M.; Tsugane, S.; et al. Meat Consumption and Colorectal Cancer Risk: An Evaluation Based on a Systematic Review of Epidemiologic Evidence Among the Japanese Population. Jpn. J. Clin. Oncol. 2014, 44, 641–650. [Google Scholar] [CrossRef] [Green Version]
- Sandhu, M.S.; White, I.; McPherson, K. Systematic review of the prospective cohort studies on meat consumption and colorectal cancer risk: A meta-analytical approach. Cancer Epidemiol. Biomark. Prev. 2001, 10, 439–446. [Google Scholar]
- Yuan, C.; Spiegelman, D.; Rimm, E.B.; Rosner, B.A.; Stampfer, M.J.; Barnett, J.B.; Chavarro, J.E.; Rood, J.C.; Harnack, L.J.; Sampson, L.K.; et al. Relative Validity of Nutrient Intakes Assessed by Questionnaire, 24-Hour Recalls, and Diet Records as Compared With Urinary Recovery and Plasma Concentration Biomarkers: Findings for Women. Am. J. Epidemiol. 2018, 187, 1051–1063. [Google Scholar] [CrossRef] [Green Version]
- Weismayer, C.; Anderson, J.G.; Wolk, A. Changes in the stability of dietary patterns in a study of middle-aged Swedish women. J. Nutr. 2006, 136, 1582–1587. [Google Scholar] [CrossRef] [Green Version]
- Mishra, G.D.; McNaughton, S.A.; Bramwell, G.D.; Wadsworth, M.E. Longitudinal changes in dietary patterns during adult life. Br. J. Nutr. 2006, 96, 735–744. [Google Scholar]
- Ruder, E.H.; Thiébaut, A.C.M.; Thompson, F.E.; Potischman, N.; Subar, A.F.; Park, Y.; Graubard, B.I.; Hollenbeck, A.R.; Cross, A.J. Adolescent and mid-life diet: Risk of colorectal cancer in the NIH-AARP Diet and Health Study. Am. J. Clin. Nutr. 2011, 94, 1607–1619. [Google Scholar] [CrossRef]
- Gong, C.L.; Zawadzki, N.K.; Zawadzki, R.; Tran, J.; Hay, J.W. Unprocessed Red Meat and Processed Meat Consumption. Ann. Intern. Med. 2020, 172, 637–638. [Google Scholar] [CrossRef]
- Qian, F.; Riddle, M.C.; Wylie-Rosett, J.; Hu, F.B. Red and Processed Meats and Health Risks: How Strong Is the Evidence? Diabetes Care 2020, 43, 265–271. [Google Scholar] [CrossRef] [Green Version]
- Schunemann, H.; Hill, S.; Guyatt, G.; Akl, E.A.; Ahmed, F. The GRADE approach and Bradford Hill’s criteria for causation. J. Epidemiol. Community Health 2011, 65, 392–395. [Google Scholar] [CrossRef]
- Schünemann, H.J.; Tugwell, P.; Reeves, B.C.; Akl, E.A.; Santesso, N.; Spencer, F.A.; Shea, B.; Wells, G.; Helfand, M. Non-randomized studies as a source of complementary, sequential or replacement evidence for randomized controlled trials in systematic reviews on the effects of interventions. Res. Synth. Methods 2013, 4, 49–62. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Händel, M.N.; Rohde, J.F.; Jacobsen, R.; Heitmann, B.L. Processed Meat Consumption and the Risk of Cancer: A Critical Evaluation of the Constraints of Current Evidence from Epidemiological Studies. Nutrients 2021, 13, 3601. https://doi.org/10.3390/nu13103601
Händel MN, Rohde JF, Jacobsen R, Heitmann BL. Processed Meat Consumption and the Risk of Cancer: A Critical Evaluation of the Constraints of Current Evidence from Epidemiological Studies. Nutrients. 2021; 13(10):3601. https://doi.org/10.3390/nu13103601
Chicago/Turabian StyleHändel, Mina Nicole, Jeanett Friis Rohde, Ramune Jacobsen, and Berit Lilienthal Heitmann. 2021. "Processed Meat Consumption and the Risk of Cancer: A Critical Evaluation of the Constraints of Current Evidence from Epidemiological Studies" Nutrients 13, no. 10: 3601. https://doi.org/10.3390/nu13103601
APA StyleHändel, M. N., Rohde, J. F., Jacobsen, R., & Heitmann, B. L. (2021). Processed Meat Consumption and the Risk of Cancer: A Critical Evaluation of the Constraints of Current Evidence from Epidemiological Studies. Nutrients, 13(10), 3601. https://doi.org/10.3390/nu13103601







