Leptin as a Biomarker of Stress: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Data Collection
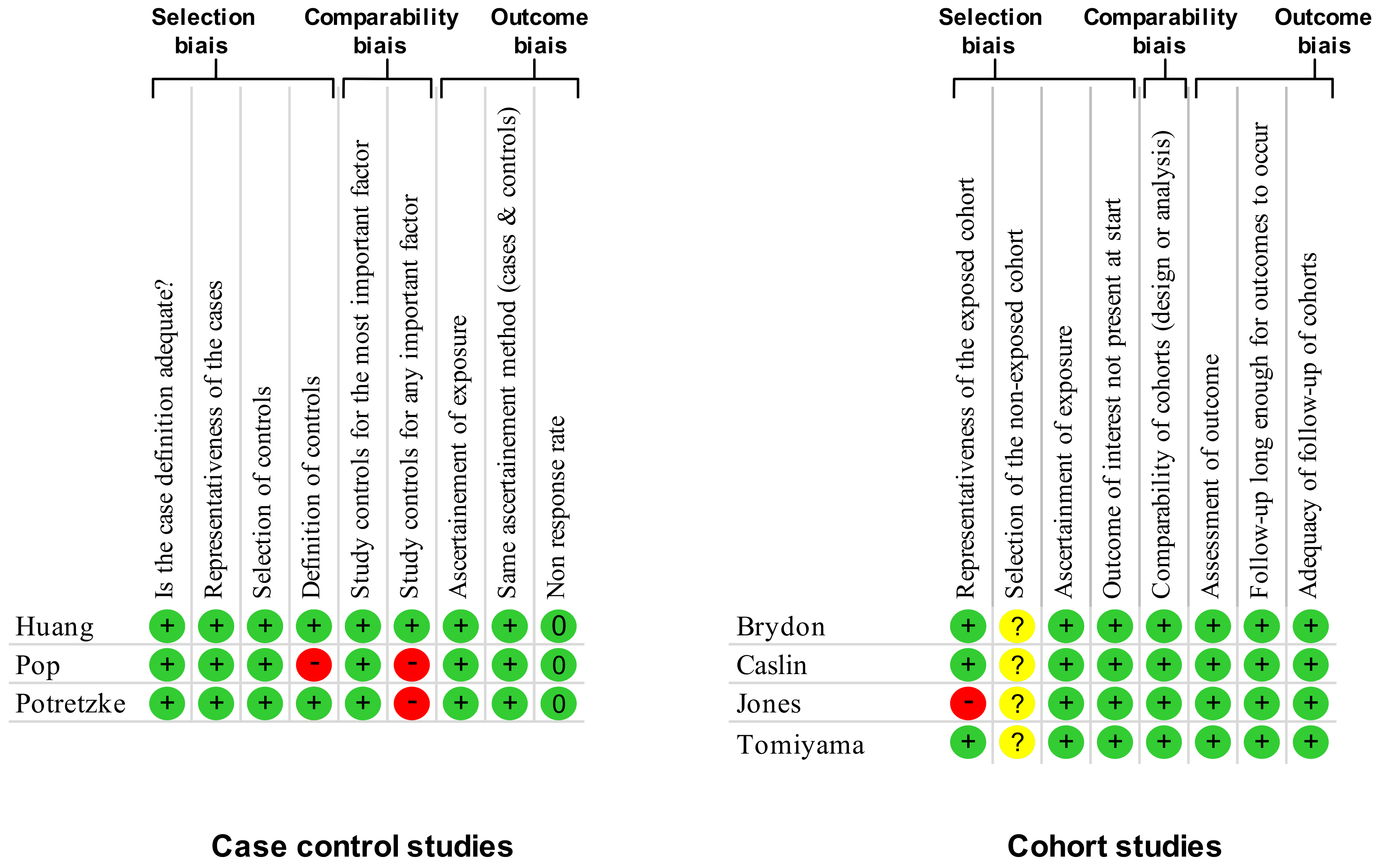
2.3. Quality of Assessment
2.4. Statistical Consideration
3. Results
3.1. Quality of Articles and Study Designs
3.2. Inclusion and Exclusion Criteria of Included Articles
3.3. Population
3.4. Main Outcome of Studies
3.5. Type of Stress
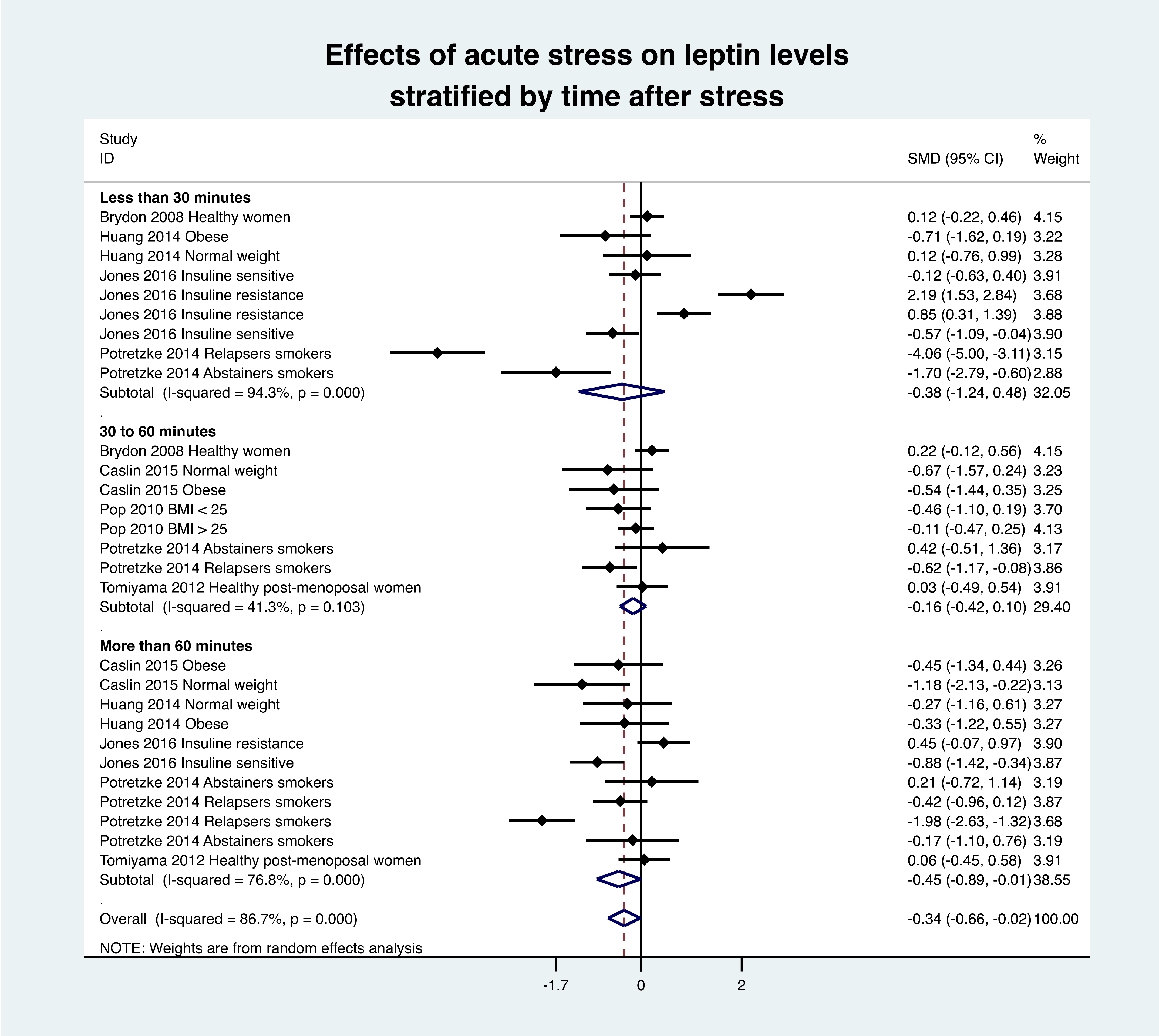
3.6. Time of Procedure
3.7. Method and Time of Sampling
3.8. Meta-Analysis of Leptin Variation
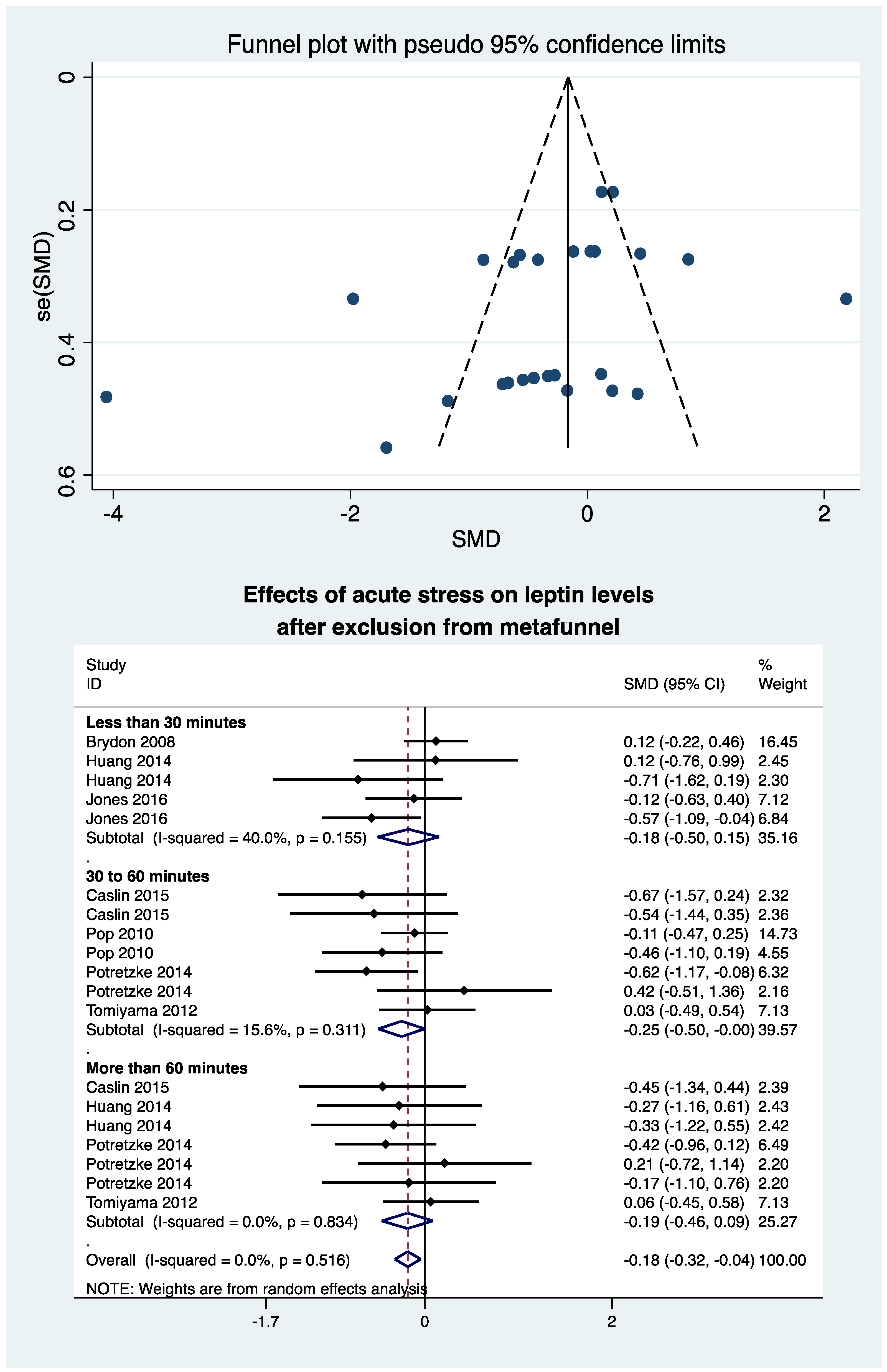
3.9. Metafunnel and Meta-Analysis after Exclusion of Studies Outside of the Metafunnel
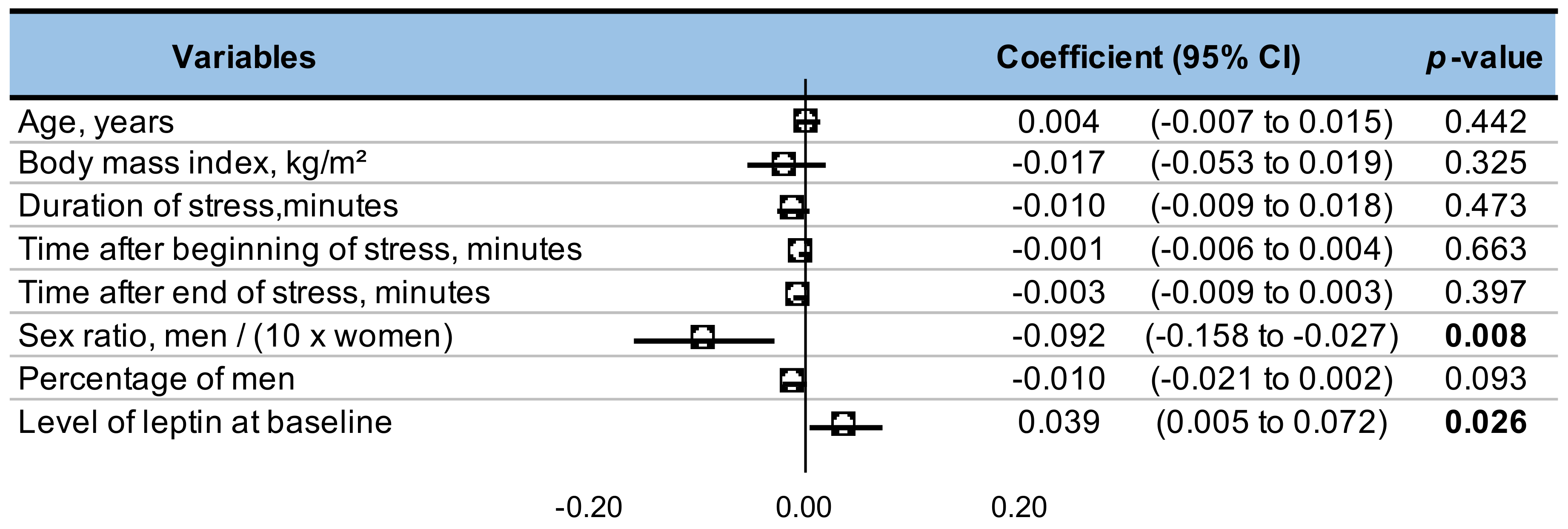
3.10. Metaregressions
4. Discussion
4.1. Leptin as a Biomarker of Stress
4.2. Acute Response to an Acute Stress Intervention
4.3. Impact of the Body Mass Index on Leptin
4.4. Impact of Sex on Leptin Levels
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| PubMed |
| (“leptin”[MH] OR “ob protein”[TW] OR “ob gene product”[TW] OR “obesity factor”[TW] OR “obese gene product”[TW] OR “obese protein”[TW] OR “leptin”[TW]) |
| AND (“Stress, Psychological”[Mesh] OR “Anxiety”[Mesh:NoExp] OR anxie*[TIAB] OR anxious*[TIAB] OR stress*[TIAB] OR mood[TIAB] OR emotion[TIAB]) |
| Filter Language = none |
| Filter Dates = none |
| Web of science (WOS) |
| (leptin OR “ob protein” OR “ob gene product” OR “obesity factor” OR “obese gene product” OR “obese protein”) |
| AND (anxie* OR anxious* OR stress* OR mood OR emotion) |
| Filter Language = none |
| Filter Dates = none |
| Cochrane Library |
| (leptin OR “ob protein” OR “ob gene product” OR “obesity factor” OR “obese gene product” OR “obese protein”) |
| AND (anxie* OR anxious* OR stress* OR mood OR emotion) |
| Filter Language = none |
| Filter Dates = none |
| Embase |
| (leptin OR “ob protein” OR “ob gene product” OR “obesity factor” OR “obese gene product” OR “obese protein”) |
| AND (anxie* OR anxious* OR stress* OR mood OR emotion) |
| Filter Language = none |
| Filter Dates = none |
Appendix B

References
- Graham, N.M. Psychological stress as a public health problem: How much do we know? Community Health Stud. 1988, 12, 151–160. [Google Scholar] [CrossRef]
- Steen, P.B.; Poulsen, P.H.; Andersen, J.H.; Biering, K. Subjective social status is an important determinant of perceived stress among adolescents: A cross-sectional study. BMC Public Health 2020, 20, 396. [Google Scholar] [CrossRef] [Green Version]
- Mathers, C.; Fat, D.M.; Boerma, J.T. The Global Burden of Disease: 2004 Update; World Health Organization, Ed.; World Health Organization: Geneva, Switzerland, 2008; ISBN 978-9241563710. [Google Scholar]
- Wekenborg, M.K.; von Dawans, B.; Hill, L.K.; Thayer, J.F.; Penz, M.; Kirschbaum, C. Examining reactivity patterns in burnout and other indicators of chronic stress. Psychoneuroendocrinology 2019, 106, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Vinkers, C.H.; Kuzminskaite, E.; Lamers, F.; Giltay, E.J.; Penninx, B.W. An integrated approach to understand biological stress system dysregulation across depressive and anxiety disorders. J. Affect. Disord. 2021, 283, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Southwick, S.M.; Vythilingam, M.; Charney, D.S. The psychobiology of depression and resilience to stress: Implications for prevention and treatment. Annu. Rev. Clin. Psychol. 2005, 1, 255–291. [Google Scholar] [CrossRef] [PubMed]
- Dutheil, F.; Aubert, C.; Pereira, B.; Dambrun, M.; Moustafa, F.; Mermillod, M.; Baker, J.S.; Trousselard, M.; Lesage, F.-X.; Navel, V. Suicide among physicians and health-care workers: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0226361. [Google Scholar] [CrossRef] [Green Version]
- Kivimaki, M.; Steptoe, A. Effects of stress on the development and progression of cardiovascular disease. Nat. Rev. Cardiol. 2018, 15, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Chami, R.; Monteleone, A.M.; Treasure, J.; Monteleone, P. Stress hormones and eating disorders. Mol. Cell. Endocrinol. 2019, 497, 110349. [Google Scholar] [CrossRef] [PubMed]
- Tomiyama, A.J. Stress and obesity. Annu. Rev. Psychol. 2019, 70, 703–718. [Google Scholar] [CrossRef] [Green Version]
- Zamkah, A.; Hui, T.; Andrews, S.; Dey, N.; Shi, F.; Sherratt, R.S. Identification of suitable biomarkers for stress and emotion detection for future personal affective wearable sensors. Biosensors 2020, 10, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Condon, E.M. Chronic stress in children and adolescents: A review of biomarkers for use in pediatric research. Biol. Res. Nurs. 2018, 20, 473–496. [Google Scholar] [CrossRef]
- Chojnowska, S.; Ptaszyńska-Sarosiek, I.; Kępka, A.; Knaś, M.; Waszkiewicz, N. Salivary biomarkers of stress, anxiety and depression. J. Clin. Med. 2021, 10, 517. [Google Scholar] [CrossRef]
- García-León, M.Á.; Pérez-Mármol, J.M.; Gonzalez-Pérez, R.; García-Ríos, M.D.C.; Peralta-Ramírez, M.I. Relationship between resilience and stress: Perceived stress, stressful life events, hpa axis response during a stressful task and hair cortisol. Physiol. Behav. 2019, 202, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Münzberg, H.; Morrison, C. Structure, production and signaling of leptin. Metabolism 2015, 64, 13–23. [Google Scholar] [CrossRef] [Green Version]
- Scheja, L.; Heeren, J. The endocrine function of adipose tissues in health and cardiometabolic disease. Nat. Rev. Endocrinol. 2019, 15, 507–524. [Google Scholar] [CrossRef]
- Langendonk, J.G.; Pijl, H.; Toornvliet, A.C.; Burggraaf, J.; Frölich, M.; Schoemaker, R.C.; Doornbos, J.; Cohen, A.F.; Meinders, A.E. Circadian rhythm of plasma leptin levels in upper and lower body obese women: Influence of body fat distribution and weight loss. J. Clin. Endocrinol. Metab. 1998, 83, 1706–1712. [Google Scholar] [CrossRef] [PubMed]
- Gijón-Conde, T.; Graciani, A.; Guallar-Castillón, P.; Aguilera, M.T.; Rodríguez-Artalejo, F.; Banegas, J.R. Leptin reference values and cutoffs for identifying cardiometabolic abnormalities in the spanish population. Rev. Esp. Cardiol. Engl. Ed. 2015, 68, 672–679. [Google Scholar] [CrossRef] [Green Version]
- Triantafyllou, G.A.; Paschou, S.A.; Mantzoros, C.S. Leptin and hormones: Energy homeostasis. Endocrinol. Metab. Clin. N. Am. 2016, 45, 633–645. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-K.; Ahima, R.S. Physiology of leptin: Energy homeostasis, neuroendocrine function and metabolism. Metabolism 2015, 64, 24–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bird, L. Exercise lowers leptin and leukocytosis. Nat. Rev. Immunol. 2020, 20, 2–3. [Google Scholar] [CrossRef]
- Zhang, Y.; Chua, S. Leptin function and regulation. Compr. Physiol. 2017, 8, 351–369. [Google Scholar] [CrossRef] [PubMed]
- Simon, C.; Gronfier, C.; Schlienger, J.L.; Brandenberger, G. Circadian and ultradian variations of leptin in normal man under continuous enteral nutrition: Relationship to sleep and body temperature. J. Clin. Endocrinol. Metab. 1998, 83, 1893–1899. [Google Scholar] [CrossRef]
- Torres, S.J.; Nowson, C.A. Relationship between stress, eating behavior, and obesity. Nutr. Burbank Los Angel. City Calif. 2007, 23, 887–894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Non-Randomised Studies in Meta-Analyses; Ottawa Hospital Research Institute: Ottawa, ON, Canada, 2011; pp. 1–12. [Google Scholar]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Brydon, L.; Wright, C.; O’Donnell, K.; Zachary, I.; Wardle, J.; Steptoe, A. Stress-induced cytokine responses and central adiposity in young women. Int. J. Obes. 2008, 32, 443–450. [Google Scholar] [CrossRef] [Green Version]
- Caslin, H.L.; Franco, R.L.; Crabb, E.B.; Huang, C.J.; Bowen, M.K.; Acevedo, E.O. The effect of obesity on inflammatory cytokine and leptin production following acute mental stress. Psychophysiology 2016, 53, 151–158. [Google Scholar] [CrossRef]
- Huang, C.-J.; Stewart, J.K.; Shibata, Y.; Slusher, A.L.; Acevedo, E.O. Lipopolysaccharide-binding protein and leptin are associated with stress-induced interleukin-6 cytokine expression ex vivo in obesity. Psychophysiology 2015, 52, 687–694. [Google Scholar] [CrossRef]
- Tomiyama, A.J.; Schamarek, I.; Lustig, R.H.; Kirschbaum, C.; Puterman, E.; Havel, P.J.; Epel, E.S. Leptin concentrations in response to acute stress predict subsequent intake of comfort foods. Physiol. Behav. 2012, 107, 34–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, K.T.; Shelton, R.C.; Wan, J.; Li, L. Impact of acute psychological stress on cardiovascular risk factors in face of insulin resistance. Stress Amst. Neth. 2016, 19, 585–592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pop, D.; Bodisz, G.; Petrovai, D.; Borz, B.; Zdrenghea, V.; Zdrenghea, D. The effect of very short duration acute physical exercise upon adiponectin and leptin in overweight subjects. Rom. J. Intern. Med. Rev. Roum. Med. Interne 2010, 48, 39–45. [Google Scholar]
- Potretzke, S.; Nakajima, M.; Cragin, T.; Al’Absi, M. Changes in circulating leptin levels during acute stress and associations with craving in abstinent smokers: A preliminary investigation. Psychoneuroendocrinology 2014, 47, 232–240. [Google Scholar] [CrossRef] [Green Version]
- Katz, A.; Nambi, S.S.; Mather, K.; Baron, A.D.; Follmann, D.A.; Sullivan, G.; Quon, M.J. Quantitative insulin sensitivity check index: A simple, accurate method for assessing insulin sensitivity in humans. J. Clin. Endocrinol. Metab. 2000, 85, 2402–2410. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, E.O.; Webb, H.E.; Weldy, M.L.; Fabianke, E.C.; Orndorff, G.R.; Starks, M.A. Cardiorespiratory responses of hi fit and low fit subjects to mental challenge during exercise. Int. J. Sports Med. 2006, 27, 1013–1022. [Google Scholar] [CrossRef]
- Pop, L.C.; Sukumar, D.; Tomaino, K.; Schlussel, Y.; Schneider, S.H.; Gordon, C.L.; Wang, X.; Shapses, S.A. Moderate weight loss in obese and overweight men preserves bone quality. Am. J. Clin. Nutr. 2015, 101, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef]
- Seoane-Collazo, P.; Martínez-Sánchez, N.; Milbank, E.; Contreras, C. Incendiary leptin. Nutrients 2020, 12, 472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zanchi, D.; Depoorter, A.; Egloff, L.; Haller, S.; Mählmann, L.; Lang, U.E.; Drewe, J.; Beglinger, C.; Schmidt, A.; Borgwardt, S. The impact of gut hormones on the neural circuit of appetite and satiety: A systematic review. Neurosci. Biobehav. Rev. 2017, 80, 457–475. [Google Scholar] [CrossRef] [PubMed]
- Dodd, G.T.; Decherf, S.; Loh, K.; Simonds, S.E.; Wiede, F.; Balland, E.; Merry, T.L.; Münzberg, H.; Zhang, Z.-Y.; Kahn, B.B.; et al. Leptin and insulin act on POMC neurons to promote the browning of white Fat. Cell 2015, 160, 88–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldini, G.; Phelan, K.D. The melanocortin pathway and control of appetite-progress and therapeutic implications. J. Endocrinol. 2019, 241, R1–R33. [Google Scholar] [CrossRef]
- Argiolas, A.; Melis, M.R. Neuropeptides and central control of sexual behaviour from the past to the present: A review. Prog. Neurobiol. 2013, 108, 80–107. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, K.; Kauser, S.; Pritchard, L.E.; Warhurst, A.; Oliver, R.L.; Slominski, A.; Wei, E.T.; Thody, A.J.; Tobin, D.J.; White, A. Proopiomelanocortin (POMC), the ACTH/Melanocortin precursor, is secreted by human epidermal keratinocytes and melanocytes and stimulates melanogenesis. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2007, 21, 1844–1856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandit, R.; Beerens, S.; Adan, R.A.H. Role of leptin in energy expenditure: The hypothalamic perspective. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 312, R938–R947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nwadozi, E.; Ng, A.; Strömberg, A.; Liu, H.-Y.; Olsson, K.; Gustafsson, T.; Haas, T.L. Leptin is a physiological regulator of skeletal muscle angiogenesis and is locally produced by PDGFRα and PDGFRβ expressing perivascular cells. Angiogenesis 2018, 22, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Copas, J. Meta-analysis, funnel plots and sensitivity analysis. Biostatistics 2000, 1, 247–262. [Google Scholar] [CrossRef] [Green Version]
- Tank, A.W.; Wong, D.L. Peripheral and central effects of circulating catecholamines. Compr. Physiol. 2014, 5, 1–15. [Google Scholar] [CrossRef]
- Goessl, V.C.; Curtiss, J.E.; Hofmann, S.G. The effect of heart rate variability biofeedback training on stress and anxiety: A meta-analysis. Psychol. Med. 2017, 47, 2578–2586. [Google Scholar] [CrossRef]
- Jung, Y.-H.; Shin, N.Y.; Jang, J.H.; Lee, W.J.; Lee, D.; Choi, Y.; Choi, S.-H.; Kang, D.-H. Relationships among stress, emotional intelligence, cognitive intelligence, and cytokines. Medicine 2019, 98, e15345. [Google Scholar] [CrossRef]
- Bouillon-Minois, J.-B.; Trousselard, M.; Thivel, D.; Gordon, B.; Schmidt, J.; Moustafa, F.; Oris, C.; Dutheil, F. Ghrelin as a Biomarker of stress: A systematic review and meta-analysis. Nutrients 2021, 13, 784. [Google Scholar] [CrossRef] [PubMed]
- Dutheil, F.; de Saint Vincent, S.; Pereira, B.; Schmidt, J.; Moustafa, F.; Charkhabi, M.; Bouillon-Minois, J.-B.; Clinchamps, M. DHEA as a biomarker of stress: A systematic review and meta-analysis. Front. Psychiatry 2021, 12, 688367. [Google Scholar] [CrossRef]
- Laferrère, B.; Fried, S.K.; Osborne, T.; Pi-Sunyer, F.X. Effect of one morning meal and a bolus of dexamethasone on 24-hour variation of serum leptin levels in humans. Obes. Res. 2000, 8, 481–486. [Google Scholar] [CrossRef] [Green Version]
- Jha, K.P.; Challet, E.; Kalsbeek, A. circadian rhythms in glucose and lipid metabolism in nocturnal and diurnal mammals. Mol. Cell. Endocrinol. 2015, 418 Pt 1, 74–88. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.-Y.; Fu, Y.-R.; Li, X.-H.; Li, Y.-Y.; Bogdan, A.; Touitou, Y. Age-related modifications of circadian rhythm of serum leptin in healthy men. Gerontology 2002, 48, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Isasi, C.R.; Parrinello, C.M.; Jung, M.M.; Carnethon, M.R.; Birnbaum-Weitzman, O.; Espinoza, R.A.; Penedo, F.J.; Perreira, K.M.; Schneiderman, N.; Sotres-Alvarez, D.; et al. Psychosocial stress is associated with obesity and diet quality in Hispanic/Latino adults. Ann. Epidemiol. 2015, 25, 84–89. [Google Scholar] [CrossRef] [Green Version]
- Czeglédi, E. Options for stress management in obesity treatment. Orv. Hetil. 2016, 157, 260–267. [Google Scholar] [CrossRef] [Green Version]
- Myers, M.G.; Leibel, R.L.; Seeley, R.J.; Schwartz, M.W. Obesity and leptin resistance: Distinguishing cause from effect. Trends Endocrinol. Metab. TEM 2010, 21, 643–651. [Google Scholar] [CrossRef] [Green Version]
- Friedman, J.M. Leptin and the endocrine control of energy balance. Nat. Metab. 2019, 1, 754–764. [Google Scholar] [CrossRef]
- Levin, B.E.; Dunn-Meynell, A.A.; McMinn, J.E.; Alperovich, M.; Cunningham-Bussel, A.; Chua, S.C. A New obesity-prone, glucose-intolerant rat strain (F.DIO). Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 285, R1184–R1191. [Google Scholar] [CrossRef] [Green Version]
- Cui, H.; López, M.; Rahmouni, K. The cellular and molecular bases of leptin and ghrelin resistance in obesity. Nat. Rev. Endocrinol. 2017, 13, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Perakakis, N.; Farr, O.M.; Mantzoros, C.S. Leptin in leanness and obesity: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2021, 77, 745–760. [Google Scholar] [CrossRef]
- Majeed, M.; Majeed, S.; Nagabhushanam, K.; Gnanamani, M.; Mundkur, L. Lesser investigated natural ingredients for the management of obesity. Nutrients 2021, 13, 510. [Google Scholar] [CrossRef]
- Montague, C.T.; Farooqi, I.S.; Whitehead, J.P.; Soos, M.A.; Rau, H.; Wareham, N.J.; Sewter, C.P.; Digby, J.E.; Mohammed, S.N.; Hurst, J.A.; et al. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 1997, 387, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Guh, D.P.; Zhang, W.; Bansback, N.; Amarsi, Z.; Birmingham, C.L.; Anis, A.H. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public Health 2009, 9, 88. [Google Scholar] [CrossRef] [Green Version]
- Demerath, E.W.; Sun, S.S.; Rogers, N.; Lee, M.; Reed, D.; Choh, A.C.; Couch, W.; Czerwinski, S.A.; Chumlea, W.C.; Siervogel, R.M.; et al. Anatomical patterning of visceral adipose tissue: Race, sex, and age variation. Obes. Silver Spring Md. 2007, 15, 2984–2993. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Morris, C.J.; Caputo, R.; Wang, W.; Garaulet, M.; Scheer, F.A.J.L. Sex differences in the circadian misalignment effects on energy regulation. Proc. Natl. Acad. Sci. USA 2019, 116, 23806–23812. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Pérez, A.; Sánchez-Jiménez, F.; Maymó, J.; Dueñas, J.L.; Varone, C.; Sánchez-Margalet, V. Role of leptin in female reproduction. Clin. Chem. Lab. Med. CCLM 2015, 53, 15–28. [Google Scholar] [CrossRef]
- Kalaitzopoulos, D.R.; Lempesis, I.G.; Samartzis, N.; Kolovos, G.; Dedes, I.; Daniilidis, A.; Nirgianakis, K.; Leeners, B.; Goulis, D.G.; Samartzis, E.P. Leptin concentrations in endometriosis: A systematic review and meta-analysis. J. Reprod. Immunol. 2021, 146, 103338. [Google Scholar] [CrossRef]
- Palmer, B.F.; Clegg, D.J. The sexual dimorphism of obesity. Mol. Cell. Endocrinol. 2015, 402, 113–119. [Google Scholar] [CrossRef] [Green Version]
- Jenks, M.Z.; Fairfield, H.E.; Johnson, E.C.; Morrison, R.F.; Muday, G.K. Sex steroid hormones regulate leptin transcript accumulation and protein secretion in 3T3-L1 Cells. Sci. Rep. 2017, 7, 8232. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.; Cleary, M.P. The potential role of leptin in tumor invasion and metastasis. Cytokine Growth Factor Rev. 2017, 38, 80–97. [Google Scholar] [CrossRef]
- Atoum, M.F.; Alzoughool, F.; Al-Hourani, H. Linkage between obesity leptin and breast cancer. Breast Cancer Basic Clin. Res. 2020, 14, 1178223419898458. [Google Scholar] [CrossRef] [PubMed]


| Study | Country | Study Design | Population | Stress | Leptin Assessment | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Characteristics | Men | Women | Type | Duration | Time after Stress | Fluid | Technique | |||
| n | n | (Minutes) | (Minutes) | |||||||
| Brydon 2008 | UK | Cohort | Healthy women | 0 | 67 | Mental | 10 | 2 measures: 0 and 45 | Blood | ELISA |
| Caslin 2015 | USA | Cohort | Healthy normal and overweight men | 20 | 0 | Mental | 20 | 2 measures: 30 and 120 | Blood | ELISA |
| Huang 2014 | USA | Case control | Healthy normal and overweight men | 20 | 0 | Mental | 20 | 2 measures: 0 and 60 | Blood | ELISA |
| Jones 2016 | USA | Cohort | Healthy insulin-resistant and insulin-sensitive volunteers | 0 | 60 | Mental (TSST) | 13 | 3 measures: 0, 15, and 90 | Blood | RIA |
| Pop 2010 | Romania | Case control | Healthy normal and overweight volunteers | 42 | 37 | Physical (VO2 max) | 12.5 | 1 measure: 30 | Blood | ELISA |
| Potrezke 2014 | USA | Case control | Healthy smokers (relapsers and abstainers) | 22 | 14 | Mental | 24 | 4 measures: 0, 30, 46, and 76 | Blood | ELISA |
| Tomiyama 2012 | USA | Cohort | Healthy postmenopausal women | 0 | 40 | Mental (TSST) | 10 | 3 measures 0, 50, and 90 | Blood | ELISA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouillon-Minois, J.-B.; Trousselard, M.; Thivel, D.; Benson, A.C.; Schmidt, J.; Moustafa, F.; Bouvier, D.; Dutheil, F. Leptin as a Biomarker of Stress: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3350. https://doi.org/10.3390/nu13103350
Bouillon-Minois J-B, Trousselard M, Thivel D, Benson AC, Schmidt J, Moustafa F, Bouvier D, Dutheil F. Leptin as a Biomarker of Stress: A Systematic Review and Meta-Analysis. Nutrients. 2021; 13(10):3350. https://doi.org/10.3390/nu13103350
Chicago/Turabian StyleBouillon-Minois, Jean-Baptiste, Marion Trousselard, David Thivel, Amanda C. Benson, Jeannot Schmidt, Farès Moustafa, Damien Bouvier, and Frédéric Dutheil. 2021. "Leptin as a Biomarker of Stress: A Systematic Review and Meta-Analysis" Nutrients 13, no. 10: 3350. https://doi.org/10.3390/nu13103350
APA StyleBouillon-Minois, J.-B., Trousselard, M., Thivel, D., Benson, A. C., Schmidt, J., Moustafa, F., Bouvier, D., & Dutheil, F. (2021). Leptin as a Biomarker of Stress: A Systematic Review and Meta-Analysis. Nutrients, 13(10), 3350. https://doi.org/10.3390/nu13103350







