Nutritional Status and Indicators of 2-Year Mortality and Re-Hospitalizations: Experience from the Internal Clinic Departments in Tertiary Hospital in Croatia
Abstract
1. Introduction
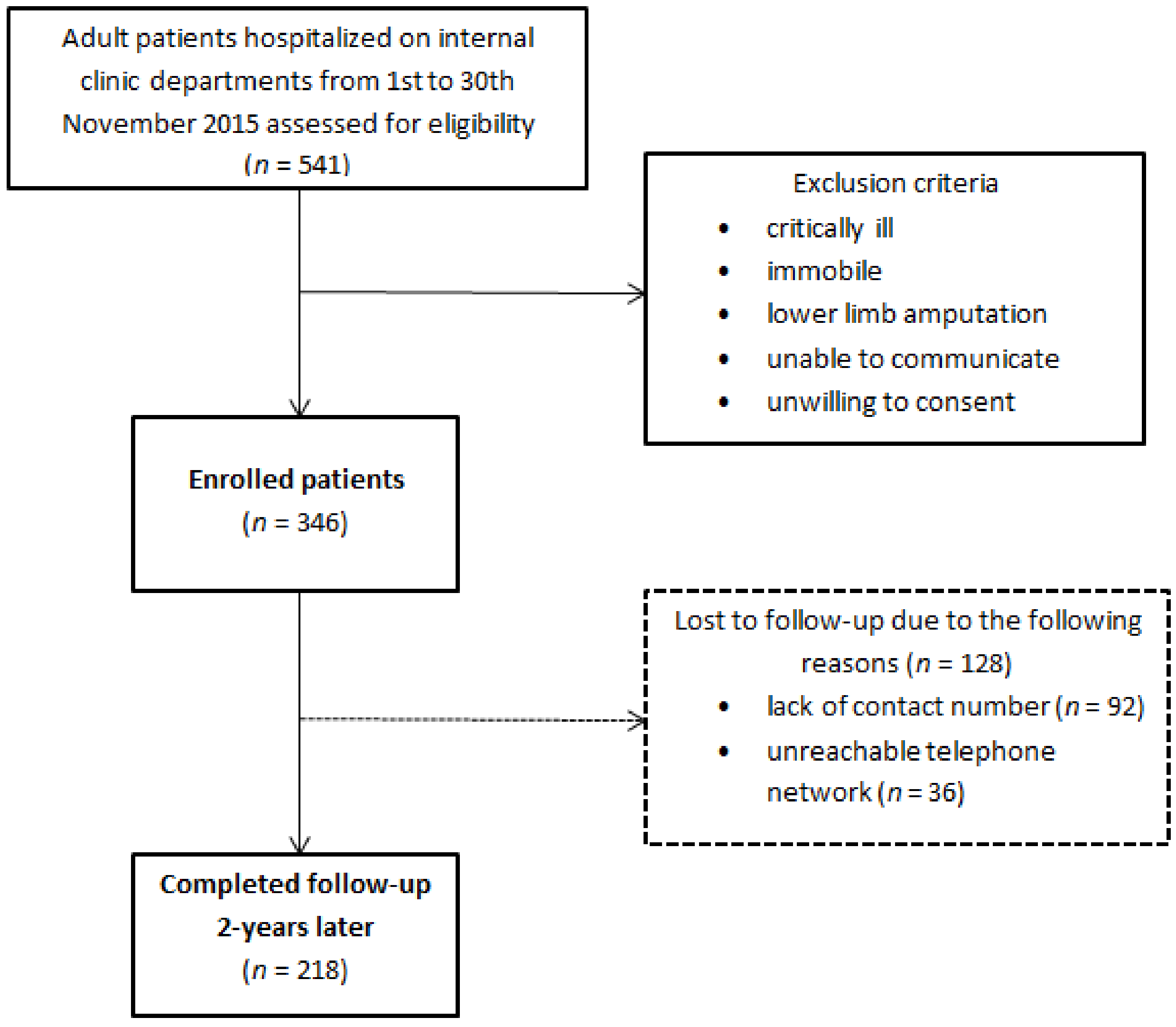
2. Materials and Methods
2.1. Study Design and Setting
2.2. Demographic, Anthropometric, Laboratory, and Medical History Assessment
2.3. Nutritional Assessment
2.4. Follow-Up
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pirlich, M.; Schütz, T.; Kemps, M.; Luhman, N.; Minko, N.; Lübke, H.J.; Rossnagel, K.; Willich, S.N.; Lochs, H. Social risk factors for hospital malnutrition. Nutrition 2005, 21, 295–300. [Google Scholar] [CrossRef]
- Barker, L.A.; Gout, B.S.; Crowe, T.C. Hospital malnutrition: Prevalence, identification and impact on patients and the healthcare system. Int. J. Environ. Res. Public Health 2011, 8, 514–527. [Google Scholar] [CrossRef]
- Lim, S.-L.; Ong, K.C.B.; Chan, Y.H.; Loke, W.C.; Ferguson, M.; Daniels, L. Malnutrition and its impact on cost of hospitalization, length of stay, readmission and 3-year mortality. Clin. Nutr. 2012, 31, 345–350. [Google Scholar] [CrossRef]
- Sharma, Y.; Miller, M.; Kaambwa, B.; Shahi, R.; Hakendorf, P.; Horwood, C.; Thompson, C. Malnutrition and its association with readmission and death within 7 days and 8-180 days postdischarge in older patients: A prospective observational study. BMJ Open 2017, 7, e018443. [Google Scholar]
- Mogensen, K.M.; Horkan, C.M.; Purtle, S.W.; Moromizato, T.; Rawn, J.D.; Robinson, M.K.; Christopher, K.B. Malnutrition, Critical Illness Survivors, and Postdischarge Outcomes: A Cohort Study. J. Parenter. Enter. Nutr. 2018, 42, 557–565. [Google Scholar] [CrossRef]
- Khalatbari-Soltani, S.; Marques-Vidal, P. Impact of nutritional risk screening in hospitalized patients on management, outcome and costs: A retrospective study. Clin. Nutr. 2016, 35, 1340–1346. [Google Scholar] [CrossRef]
- Sanson, G.; Sadiraj, M.; Barbin, I.; Confezione, C.; De Matteis, D.; Boscutti, G.; Zaccari, M.; Zanetti, M. Prediction of early- and long-term mortality in adult patients acutely admitted to internal medicine: NRS-2002 and beyond. Clin. Nutr. 2020, 39, 1092–1100. [Google Scholar] [CrossRef]
- Cederholm, T.; Barazzoni, R.; Austin, P.D.P.; Ballmer, P.P.; Biolo, G.; Bischoff, S.C.; Compher, C.; Correia, M.I.T.; Higashiguchi, T.T.; Holst, M.M.; et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin. Nutr. 2017, 36, 49–64. [Google Scholar] [CrossRef]
- Hersberger, L.; Bargetzi, L.; Bargetzi, A.; Tribolet, P.; Fehr, R.; Baechli, V.; Geiser, M.; Deiss, M.; Gomes, F.; Kutz, A.; et al. Nutritional risk screening (NRS 2002) is a strong and modifiable predictor risk score for short-term and long-term clinical outcomes: Secondary analysis of a prospective randomised trial. Clin. Nutr. 2020, 39, 2720–2729. [Google Scholar] [CrossRef]
- Sun, Z.; Kong, X.-J.; Jing, X.; Deng, R.-J.; Tian, Z.-B. Nutritional Risk Screening 2002 as a Predictor of Postoperative Outcomes in Patients Undergoing Abdominal Surgery: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. PLoS ONE 2015, 10, e0132857. [Google Scholar] [CrossRef]
- Felder, S.; Lechtenboehmer, C.; Bally, M.; Fehr, R.; Deiss, M.; Faessler, L.; Kutz, A.; Steiner, D.; Rast, A.C.; Laukemann, S.; et al. Association of nutritional risk and adverse medical outcomes across different medical inpatient populations. Nutrition 2015, 31, 1385–1393. [Google Scholar] [CrossRef]
- Führ, L.M.; Wazlawik, E.; Garcia, M.F. The predictive value of composite methods of nutritional assessment on mortality among haemodialysis patients. Clin. Nutr. ESPEN 2015, 10, e21–e25. [Google Scholar] [CrossRef]
- Chen, R.; Xing, L.; You, C.; Ou, X. Prediction of prognosis in chronic obstructive pulmonary disease patients with respiratory failure: A comparison of three nutritional assessment methods. Eur. J. Intern. Med. 2018, 57, 70–75. [Google Scholar] [CrossRef]
- Budzyński, J.; Tojek, K.; Czerniak, B.; Banaszkiewicz, Z. Scores of nutritional risk and parameters of nutritional status assessment as predictors of in-hospital mortality and readmissions in the general hospital population. Clin. Nutr. 2016, 35, 1464–1471. [Google Scholar] [CrossRef]
- Jeejeebhoy, K.; Keller, H.H.; Gramlich, L.; Allard, J.P.; Laporte, M.; Duerksen, D.R.; Payette, H.; Bernier, P.; Vesnaver, E.; Davidson, B.; et al. Nutritional assessment: Comparison of clinical assessment and objective variables for the prediction of length of hospital stay and readmission. Am. J. Clin. Nutr. 2015, 101, 956–965. [Google Scholar] [CrossRef]
- Slee, A.; Birch, D.; Stokoe, D. The relationship between malnutrition risk and clinical outcomes in a cohort of frail older hospital patients. Clin. Nutr. ESPEN 2016, 15, 57–62. [Google Scholar] [CrossRef]
- Marco, J.; Barba, R.; Zapatero-Gaviria, A.; Matia, P.; Plaza, S.; García, J.E.L.; Canora-Lebrato, J.; De Casasola, G.G. Prevalence of the notification of malnutrition in the departments of internal medicine and its prognostic implications. Clin. Nutr. 2011, 30, 450–454. [Google Scholar] [CrossRef]
- Rotovnik Kozjek, M.; Milošević, M. Nutritional Recommendations for Patients and the Elderly. Available online: http://www.mz.gov.si/fileadmin/mz.gov.si/pageuploads/javno_zdravje_09/Priporocila_za_prehransko_obravnavo_bolnikov.pdf (accessed on 19 April 2017). (In Slovene)
- Bender, D.V.; Krznaric, Z.; Reiner, Z.; Tomek-Roksandić, S.; Duraković, Z.; Kaić-Rak, A.; Narancić, N.S.; Bosnir, J. Croatian guidelines for nutrition in the elderly, part I. Liječ. Vjesn. 2011, 133, 231–240. [Google Scholar]
- Krznaric, Z.; Bender, D.V.; Kelečić, D.L.; Reiner, Z.; Roksandić, S.T.; Kekez, D.; Pavic, T. Croatian guidelines for nutrition in the elderly, part II—Clinical nutrition. Liječ. Vjesn. 2011, 133, 299–307. [Google Scholar]
- Vrbica, Z.; Vukic Dugac, A.; Popovic Grle, S.; Jalusic Gluncic, T.; Jakopovic, M.; Bival, S.; Krznaric, Z.; Samarzija, M. Recommendations for prevention and treatment of malnutrition in patients with chronic obstructive pulmonary disease. Liječ. Vjesn. 2018, 140, 183–189. [Google Scholar]
- Bašić-Jukić, N.; Radić, J.; Klarić, D.; Jakić, M.; Vujicic, B.; Gulin, M.; Krznarić, Z.; Pavić, E.; Kes, P.; Jelakovic, B.; et al. Croatian guidelines for screening, prevention and treatment of protein-energy wasting in chronic kidney disease patients. Liječ. Vjesn. 2015, 137, 1–8. [Google Scholar]
- Zelic, M.; Bender, D.V.; Kelečić, D.L.; Župan, Ž.; Cicvarić, T.; Maldini, B.; Durut, I.; Rahelić, V.; Skegro, M.; Majerović, M.; et al. Croatian guidelines for perioperative enteral nutrition of surgical patients. Liječ. Vjesn. 2014, 136, 179–185. [Google Scholar]
- Pavic, T.; Ljubičić, N.; Stojsavljević, S.; Krznaric, Z. Nutritional screening model in tertiary medical unit in Croatia. Ann. Nutr. Metab. 2012, 61, 65–69. [Google Scholar] [CrossRef]
- Vranesic Bender, D.; Krznaric, Z.; Colic Baric, I. Assessment of nutritional status of gastroenterology patients in Croatia. Coll. Antropol. 2010, 34, 1329–1334. [Google Scholar]
- Mamic, M.; Kucan, P.; Vukman, D.; Niseteo, T. Prevalence of malnutrition and energy intake in hospitalized children. Paediatr. Croat. 2018, 62, 74–80. [Google Scholar]
- National Health and Nutrition Examination Survey (NHANES). Anthropometry Procedures Manual; CDC: Atlanta, GA, USA, 2015. Available online: https://www.cdc.gov/nchs/data/nhanes/nhanes_13_14/2013_Anthropometry.pdf (accessed on 30 January 2015).
- WHO. Obesity: Preventing and Managing the Global Epidemic; Report of a WHO Consultation; World Health Organization Technical Report Series 894; WHO: Geneva, Switzerland, 2000. [Google Scholar]
- Cederholm, T.; Jensen, G.; Correia, M.; Gonzalez, M.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. J. Cachexia Sarcopenia Muscle 2019, 10, 207–217. [Google Scholar] [CrossRef]
- Després, J.-P.; Lemieux, I.; Prud’Homme, D. Treatment of obesity: Need to focus on high risk abdominally obese patients. BMJ 2001, 322, 716–720. [Google Scholar] [CrossRef]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.T.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar]
- Ashwell, M.; Gunn, P.; Gibson, S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: Systematic review and meta-analysis. Obes. Rev. 2012, 13, 275–286. [Google Scholar] [CrossRef]
- Rasmussen, H.H.; Kondrup, J.; Staun, M.; Ladefoged, K.; Kristensen, H.; Wengler, A. Prevalence of patients at nutritional risk in Danish hospitals. Clin. Nutr. 2004, 23, 1009–1015. [Google Scholar] [CrossRef]
- Fang, S.; Long, J.; Tan, R.; Mai, H.; Lu, W.; Yan, F.; Peng, J. A multicentre assessment of malnutrition, nutritional risk, and application of nutritional support among hospitalized patients in Guangzhou hospitals. Asia Pac. J. Clin. Nutr. 2013, 22, 54–59. [Google Scholar] [PubMed]
- Bozzetti, F.; Mariani, L.; Vullo, S.L.; Amerio, M.L.; Biffi, R.; Caccialanza, R.; Capuano, G.; Correja, I.; Cozzaglio, L.; Di Leo, A.; et al. The nutritional risk in oncology: A study of 1453 cancer outpatients. Support. Care Cancer 2012, 20, 1919–1928. [Google Scholar] [CrossRef] [PubMed]
- Orrevall, Y.; Tishelman, C.; Permert, J.; Cederholm, T. Nutritional support and risk status among cancer patients in palliative home care services. Support. Care Cancer 2009, 17, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J.; Kondrup, J.; Prokopowicz, J.; Schiesser, M.; Krähenbühl, L.; Meier, R.; Liberda, M. EuroOOPS: An international, multicentre study to implement nutritional risk screening and evaluate clinical outcome. Clin. Nutr. 2008, 27, 340–349. [Google Scholar] [CrossRef]
- Borek, P.; Chmielewski, M.; Małgorzewicz, S.; Dębska-Ślizień, A. Analysis of Outcomes of the NRS 2002 in Patients Hospitalized in Nephrology Wards. Nutrients 2017, 9, 287. [Google Scholar] [CrossRef]
- Cawood, A.L.; Elia, M.; Stratton, R. Systematic review and meta-analysis of the effects of high protein oral nutritional supplements. Ageing Res. Rev. 2012, 11, 278–296. [Google Scholar] [CrossRef]
- Milne, A.C.; Potter, J.; Vivanti, A.; Avenell, A. Protein and energy supplementation in elderly people at risk from malnutrition. Cochrane Database Syst. Rev. 2009, 2009, CD003288. [Google Scholar] [CrossRef]
- Beck, A.M.; Holst, M.M.; Rasmussen, H.H. Oral nutritional support of older (65 years+) medical and surgical patients after discharge from hospital: Systematic review and meta-analysis of randomized controlled trials. Clin. Rehabil. 2013, 27, 19–27. [Google Scholar] [CrossRef]
- Stratton, R.J.; Hébuterne, X.; Elia, M. A systematic review and meta-analysis of the impact of oral nutritional supplements on hospital readmissions. Ageing Res. Rev. 2013, 12, 884–897. [Google Scholar] [CrossRef]
- Deutz, N.E.P.; Matheson, E.M.; Matarese, L.E.; Luo, M.; Baggs, G.; Nelson, J.L.; Hegazi, R.; Tappenden, K.A.; Ziegler, T.R. Readmission and mortality in malnourished, older, hospitalized adults treated with a specialized oral nutritional supplement: A randomized clinical trial. Clin. Nutr. 2016, 35, 18–26. [Google Scholar] [CrossRef]
- Mantzorou, M.; Vadikolias, K.; Pavlidou, E.; Serdari, A.; Vasios, G.; Tryfonos, C.; Giaginis, C. Nutritional status is associated with the degree of cognitive impairment and depressive symptoms in a Greek elderly population. Nutr. Neurosci. 2020, 23, 201–209. [Google Scholar] [CrossRef] [PubMed]
| All Subjects n = 346 | Follow-Up n = 218 | Lost to Follow-Up n = 128 | p | |
|---|---|---|---|---|
| Socio-demographic characteristics | ||||
| Age (years); median (IQR) | 67.0 (19.0) | 66.0 (18.0) | 68.0 (21.0) | 0.080 † |
| Gender; n (%) | ||||
| Female | 157 (45.4) | 105 (48.2) | 52 (40.6) | 0.174 * |
| Male | 189 (54.6) | 113 (51.8) | 76 (59.4) | |
| Habits; n (%) | ||||
| Smoking | 72 (20.8) | 43 (19.7) | 29 (22.8) | 0.493 * |
| Anthropometric indices; median (IQR) | ||||
| BMI (kg/m2) | 26.3 (7.0) | 26.4 (7.0) | 26.3 (6.0) | 0.515 † |
| Waist circumference (cm) | 103.0 (18.0) | 102.9 (18.0) | 103.5 (18.0) | 0.963 † |
| WHtR | 0.59 (0.10) | 0.59 (0.10) | 0.60 (0.11) | 0.557 † |
| Forearm circumference (cm) | 29.5 (6.5) | 29.8 (6.5) | 29.5 (7.0) | 0.607 † |
| Forearm skinfold (mm) | 35.0 (12.0) | 39.0 (15.0) | 28.0 (14.0) | <0.001 † |
| Chronic disease; n (%) | ||||
| Hypertension | 215 (62.1) | 130 (59.6) | 85 (66.4) | 0.210 * |
| Diabetes | 101 (29.2) | 64 (29.4) | 37 (28.9) | 0.929 * |
| Cancer | 102 (29.5) | 73 (33.5) | 29 (22.7) | 0.033 * |
| CKD | 71 (20.5) | 57 (26.1) | 14 (10.9) | 0.001 * |
| IBD | 10 (2.9) | 4 (1.8) | 6 (4.7) | 0.117 ** |
| Cirrhosis | 17 (4.9) | 9 (4.1) | 8 (6.3) | 0.378 * |
| Autoimmune disease | 51 (14.7) | 36 (16.5) | 15 (11.7) | 0.224 * |
| Biochemistry; median (IQR) | ||||
| CRP (mg/L) | 11.7 (34.1) | 11.6 (31.8) | 11.8 (46.1) | 0.977 † |
| Glucose (mmol/L) | 6.4 (3.0) | 6.4 (2.8) | 6.3 (3.4) | 0.790 † |
| Creatinine (μmol/L) | 91.0 (55.0) | 95.0 (79.0) | 79.5 (38.0) | <0.001 † |
| eGFR (mL/min) | 69.1 (49.1) | 66.2 (51.5) | 76.7 (48.1) | 0.001 † |
| Nutritional risk assessment; n (%) | ||||
| NRS-2002 ≥ 3 | 133 (38.4) | 82 (37.6) | 51 (40.2) | 0.640 * |
| Nutritional support prior to hospitalization; n (%) | ||||
| None | 229 (66.2) | 128 (58.7) | 101 (78.9) | <0.001 ** |
| Nutritional advice | 7 (2.0) | 6 (2.8) | 1 (0.8) | |
| Vitamin supplement | 60 (17.3) | 47 (21.6) | 13 (10.2) | |
| ONS | 46 (13.3) | 37 (17.0) | 9 (7.0) | |
| Nutritional support included during hospitalization; n (%) | ||||
| ONS | 53 (15.3) | 46 (21.1) | 7 (5.5) | <0.001 * |
| Hospitalization length; median (IQR) | ||||
| Initial hospitalization (days) | 11.0 (7.0) | 11.0 (8.0) | 8.0 (7.0) | 0.006 † |
| NRS-2002 < 3 n = 136 | NRS-2002 ≥ 3 n = 82 | p | |
|---|---|---|---|
| Baseline | |||
| Socio-demographic characteristics | |||
| Age (years); median (IQR) | 62.0 (19.0) | 73.0 (16.0) | <0.001 † |
| Age groups; n (%) | <0.001 * | ||
| <65 years | 77 (56.6) | 23 (28.0) | |
| ≥65 years | 59 (43.4) | 59 (72.0) | |
| Gender; n (%) | 0.208 * | ||
| Female | 61 (44.9) | 44 (53.7) | |
| Male | 75 (55.1) | 38 (46.3) | |
| Habits; n (%) | |||
| Smoking | 33 (24.3) | 10 (12.2) | 0.030 * |
| Anthropometric indices | |||
| BMI (kg/m2); median (IQR) | 28.0 (7.0) | 24.8 (7.0) | <0.001 † |
| BMI category (kg/m2); n (%) | <0.001 ** | ||
| <18.5 | 0 (0.0) | 5 (6.1) | |
| 18.5–24.9 | 42 (30.9) | 39 (47.6) | |
| 25.0–29.9 | 49 (36.0) | 31 (37.8) | |
| 30.0–34.9 | 32 (23.5) | 6 (7.3) | |
| 35.0–39.9 | 10 (7.4) | 1 (1.2) | |
| ≥40 | 3 (2.2) | 0 (0.0) | |
| Undernutrition according to GLIM criteria §; n (%) | <0.001 * | ||
| Yes | 4 (2.9) | 17 (20.7) | |
| No | 132 (97.1) | 65 (79.3) | |
| Waist circumference (cm); median (IQR) | 104.3 (17.0) | 99.5 (18.0) | <0.001 † |
| WHtR; median (IQR) | 0.59 (0.12) | 0.59 (0.11) | 0.026 † |
| WHtR ≥0.5; n (%) | 128 (94.1) | 71 (86.6) | 0.056 * |
| Forearm circumference (cm); median (IQR) | 31.0 (6.0) | 27.0 (5.0) | <0.001 † |
| Forearm skinfold (mm); median (IQR) | 40.0 (14.0) | 35.0 (11.0) | 0.026 † |
| Chronic disease; n (%) | |||
| Hypertension | 74 (54.4) | 56 (68.3) | 0.043 * |
| Diabetes | 42 (30.9) | 22 (26.8) | 0.524 * |
| Cancer | 28 (20.6) | 45 (54.9) | <0.001 * |
| CKD | 31 (22.8) | 26 (31.7) | 0.147 * |
| IBD | 2 (1.5) | 2 (2.4) | 0.633 ** |
| Cirrhosis | 9 (6.6) | 0 (0.0) | 0.015 ** |
| Autoimmune disease | 22 (16.2) | 14 (17.1) | 0.863 * |
| Biochemistry; median (IQR) | |||
| CRP (mg/L) | 9.7 (33.7) | 14.6 (27.6) | 0.440 † |
| Glucose (mmol/L) | 6.4 (3.0) | 6.3 (2.4) | 0.779 † |
| Creatinine (μmol/L) | 93.0 (57.0) | 104.0 (116.0) | 0.208 † |
| eGFR (mL/min) | 72.2 (49.8) | 56.7 (51.1) | 0.006 † |
| eGFR category; n (%) | 0.008 * | ||
| <15.0 | 17 (12.8) | 10 (12.2) | |
| 15.0–29.9 | 8 (6.0) | 13 (15.9) | |
| 30.0–59.9 | 30 (22.6) | 20 (24.4) | |
| 60.0–89.9 | 41 (30.8) | 31 (37.8) | |
| ≥90.0 | 37 (27.8) | 8 (9.8) | |
| Nutritional support prior to hospitalization; n (%) | 0.065 ** | ||
| None | 80 (58.8) | 48 (58.5) | |
| Nutritional advice | 4 (2.9) | 2 (2.4) | |
| Vitamin supplement | 35 (25.7) | 12 (14.6) | |
| ONS | 17 (12.5) | 20 (24.4) | |
| Nutritional support included during hospitalization; n (%) | |||
| ONS | 25 (18.4) | 21 (25.6) | 0.205 * |
| Hospitalization length; median (IQR) | |||
| Initial hospitalization (days) | 10.0 (7.0) | 14.0 (9.0) | 0.001 † |
| Follow-up period | |||
| Percent weight change (%); median (IQR) | +0.06 (7.1) | +4.9 (15.3) | 0.072 † |
| Newly diagnosed chronic disease; n (%) | |||
| Cancer | 17 (12.5) | 3 (3.7) | 0.028 * |
| Hypertension | 4 (2.9) | 1 (1.2) | 0.652 ** |
| Diabetes | 3 (2.2) | 0 (0.0) | 0.293 ** |
| Re-hospitalizations; n (%) | |||
| All re-hospitalizations | 81 (59.3) | 57 (70.4) | 0.109 * |
| One re-hospitalization | 32 (23.7) | 25 (30.9) | |
| Two re-hospitalizations | 19 (14.1) | 12 (14.8) | |
| Three re-hospitalizations | 9 (6.7) | 7 (8.7) | |
| Four or more re-hospitalizations | 20 (14.8) | 13 (16.0) | |
| None | 55 (40.7) | 24 (29.6) | 0.553 * |
| Nutritional support; n (%) | |||
| ONS use | 26 (19.3) | 30 (36.6) | 0.005 * |
| Adverse outcome; n (%) | |||
| Deceased | 32 (23.5) | 35 (42.7) | 0.003 * |
| Age | Hospitalization Duration | BMI | Waist circumference | WHtR | Forearm Circumference | Forearm Skinfold | CRP | eGFR | |
|---|---|---|---|---|---|---|---|---|---|
| NRS-2002 score | 0.471 (<0.001 *) | 0.249 (<0.001 *) | −0.308 (<0.001 *) | −0.188 (0.005) | −0.101 (0.140) | −0.374 (<0.001 *) | −0.112 (0.099) | 0.094 (0.196) | −0.256 (<0.001 *) |
| Age | 0.088 (0.196) | 0.020 (0.770) | 0.207 (0.002) | 0.320 (<0.001 *) | −0.073 (0.284) | 0.069 (0.307) | 0.093 (0.198) | −0.499 (<0.001 *) | |
| Hospitalization duration | −0.023 (0.740) | 0.012 (0.863) | 0.048 (0.482) | −0.121 (0.074) | 0.009 (0.895) | 0.194 (0.007) | −0.084 (0.222) |
| Re-Hospitalization OR (95% CI); p | Death OR (95% CI); p | |
|---|---|---|
| Gender (women are referent group) | 0.51 (0.26–1.00); 0.050 | 0.81 (0.37–1.79); 0.603 |
| Age (<65 yrs is referent group) | 1.56 (0.71–3.46); 0.269 | 1.67 (0.66–4.25); 0.280 |
| Smoking (non-smokers are referent group) | 1.18 (0.49–2.83); 0.710 | 0.50 (0.16–1.61); 0.247 |
| Forearm circumference | 1.00 (0.97–1.03); 0.844 | 0.87 (0.78–0.96); 0.008 |
| NRS-2002 ≥ 3 | 1.57 (0.70–3.53); 0.272 | 0.51 (0.19–1.32); 0.162 |
| CRP | 1.01 (1.00–1.02); 0.032 | 1.00 (0.99–1.01); 0.372 |
| eGFR (≥90.0 is referent) | ||
| <15.0 | 12.49 (1.22–127.61); 0.033 | 1.30 (0.25–6.89); 0.755 |
| 15.0–29.9 | 0.60 (0.14–2.59); 0.495 | 1.02 (0.20–5.29); 0.977 |
| 30.0–59.9 | 1.06 (0.37–3.08); 0.910 | 0.55 (0.15–2.01); 0.361 |
| 60.0–89.9 | 1.26 (0.50–3.21); 0.624 | 0.51 (0.15–1.73); 0.279 |
| ONS during follow-up (didn’t take are referent group) | 2.70 (1.11–6.54); 0.028 | 4.24 (1.80–9.97); 0.001 |
| Diabetes (no is referent group) | 1.78 (0.81–3.90); 0.153 | 3.74 (1.54–9.06); 0.003 |
| Cancer (no is referent group) | 0.69 (0.30–1.60); 0.391 | 5.85 (2.23–15.33); <0.001 |
| CKD (no is referent group) | 1.49 (0.50–4.39); 0.474 | 1.76 (0.52–5.99); 0.366 |
| Re-hospitalized (no is referent group) | - | 0.74 (0.30–1.83); 0.509 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miličević, T.; Kolčić, I.; Đogaš, T.; Živković, P.M.; Radman, M.; Radić, J. Nutritional Status and Indicators of 2-Year Mortality and Re-Hospitalizations: Experience from the Internal Clinic Departments in Tertiary Hospital in Croatia. Nutrients 2021, 13, 68. https://doi.org/10.3390/nu13010068
Miličević T, Kolčić I, Đogaš T, Živković PM, Radman M, Radić J. Nutritional Status and Indicators of 2-Year Mortality and Re-Hospitalizations: Experience from the Internal Clinic Departments in Tertiary Hospital in Croatia. Nutrients. 2021; 13(1):68. https://doi.org/10.3390/nu13010068
Chicago/Turabian StyleMiličević, Tanja, Ivana Kolčić, Tina Đogaš, Piero Marin Živković, Maja Radman, and Josipa Radić. 2021. "Nutritional Status and Indicators of 2-Year Mortality and Re-Hospitalizations: Experience from the Internal Clinic Departments in Tertiary Hospital in Croatia" Nutrients 13, no. 1: 68. https://doi.org/10.3390/nu13010068
APA StyleMiličević, T., Kolčić, I., Đogaš, T., Živković, P. M., Radman, M., & Radić, J. (2021). Nutritional Status and Indicators of 2-Year Mortality and Re-Hospitalizations: Experience from the Internal Clinic Departments in Tertiary Hospital in Croatia. Nutrients, 13(1), 68. https://doi.org/10.3390/nu13010068





