Physical Activity and Low Glycemic Index Mediterranean Diet: Main and Modification Effects on NAFLD Score. Results from a Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Sample Size
2.4. Data Collection
2.5. Randomization and Masking
2.6. Dietary Interventions
2.7. Physical Activity Interventions
2.7.1. Aerobic Activity Program
2.7.2. Combination of Aerobic Exercise and Resistance Training
2.8. Outcome Assessment
2.9. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Variables | Time | Working Arms | ||||||
|---|---|---|---|---|---|---|---|---|
| C a | LGIMD b | PA1 c | PA2 d | PA1 + LGIMD e | PA2 + LGIM f | p-Value | ||
| SBP (Hg mm) g | Baseline | 126.14 (12.14) | 128.86 (17.86) | 123.80 (13.17) | 118.00 (13.22) | 127.04 (13.82) | 122.71 (17.00) | 0.20 |
| 45 days | 121.67 (16.80) | 120.48 (14.31) | 119.50 (10.50) | 116.25 (12.66) | 120.87 (13.03) | 117.08 (13.34) | 0.75 | |
| 90 days | 122.33 (12.37) | 122.78 (12.27) | 119.06 (10.04) | 116.25 (10.88) | 123.61 (12.34) | 118.41 (12.95) | 0.40 | |
| DBP (Hg mm) h | Baseline | 81.59 (8.51) | 82.73 (9.73) | 80.60 (6.18) | 79.50 (6.26) | 82.59 (6.10) | 81.04 (9.32) | 0.73 |
| 45 days | 80.28 (7.37) | 79.76 (8.14) | 79.50 (8.57) | 79.00 (7.88) | 80.00 (7.39) | 77.71 (5.51) | 0.89 | |
| 90 days | 81.67 (8.16) | 77.50 (9.74) | 80.63 (6.80) | 79.69 (6.94) | 81.94 (6.89) | 77.50 (6.50) | 0.29 | |
| BMI i | Baseline | 34.14 (4.97) | 32.91 (4.26) | 32.73 (5.37) | 30.79 (3.16) | 33.07 (4.20) | 34.09 (3.77) | 0.13 |
| 45 days | 32.50 (4.57) | 30.70 (4.22) | 31.87 (4.96) | 30.05 (3.27) | 31.50 (3.61) | 32.19 (3.42) | 0.35 | |
| 90 days | 32.12 (5.11) | 30.14 (4.73) | 31.01 (4.69) | 29.80 (3.53) | 30.89 (3.91) | 31.97 (3.67) | 0.53 | |
| WC (cm) j | Baseline | 106.59 (11.80) | 102.91 (10.98) | 103.84 (8.58) | 99.70 (8.10) | 103.48 (11.89) | 104.46 (12.15) | 0.42 |
| 45 days | 102.11 (12.24) | 97.24 (10.19) | 101.43 (9.71) | 97.32 (6.76) | 97.92 (9.06) | 99.96 (10.20) | 0.42 | |
| 90 days | 101.00 (11.47) | 95.72 (9.69) | 99.41 (8.71) | 97.16 (6.54) | 98.01 (8.91) | 98.91 (10.57) | 0.66 | |
| HC (cm) k | Baseline | 113.59 (11.97) | 110.35 (10.18) | 109.76 (9.54) | 106.78 (7.90) | 107.67 (7.88) | 111.46 (8.04) | 0.15 |
| 45 days | 109.39 (9.80) | 105.29 (8.89) | 106.90 (11.04) | 105.18 (8.23) | 104.68 (7.12) | 107.96 (9.58) | 0.51 | |
| 90 days | 107.31 (11.32) | 104.28 (8.46) | 106.82 (8.61) | 102.68 (7.68) | 103.75 (8.42) | 107.09 (7.52) | 0.42 | |
| FM l | Baseline | 34.21 (12.24) | 31.17 (10.54) | 30.85 (10.68) | 28.07 (7.13) | 31.04 (7.64) | 33.90 (9.16) | 0.31 |
| 45 days | 31.12 (11.31) | 26.84 (9.10) | 29.43 (10.36) | 26.48 (7.76) | 28.49 (6.62) | 29.99 (7.85) | 0.51 | |
| 90 days | 29.82 (12.84) | 24.82 (8.94) | 27.31 (9.34) | 25.46 (7.67) | 26.41 (7.34) | 28.71 (8.57) | 0.60 | |
| FFM m | Baseline | 59.07 (9.99) | 59.27 (9.29) | 60.73 (11.47) | 62.54 (8.72) | 61.71 (11.76) | 62.97 (12.11) | 0.76 |
| 45 days | 57.62 (9.75) | 57.69 (8.64) | 60.71 (10.95) | 61.90 (7.83) | 59.71 (10.64) | 61.44 (10.93) | 0.61 | |
| 90 days | 59.33 (10.15) | 59.17 (9.27) | 60.11 (10.39) | 63.53 (7.60) | 60.19 (11.16) | 62.10 (11.63) | 0.78 | |
| GLUCOSE (mmol/L) | Baseline | 5.33 (0.67) | 5.90 (1.33) | 5.78 (1.70) | 5.41 (0.75) | 5.36 (0.62) | 5.49 (0.67) | 0.77 |
| 45 days | 5.09 (0.40) | 5.77 (1.09) | 5.43 (1.29) | 5.15 (0.63) | 5.20 (0.63) | 5.35 (0.60) | 0.10 | |
| 90 days | 5.10 (0.40) | 5.90 (1.38) | 5.60 (1.53) | 5.28 (1.07) | 5.25 (0.89) | 5.36 (0.53) | 0.26 | |
| TC (mmol/L) n | Baseline | 5.11 (1.06) | 5.11 (1.41) | 5.00 (0.82) | 5.31 (0.93) | 5.25 (1.10) | 5.34 (1.01) | 0.79 |
| 45 days | 5.05 (0.85) | 4.63 (1.13) | 4.97 (0.82) | 5.17 (0.82) | 4.93 (0.88) | 4.73 (1.00) | 0.38 | |
| 90 days | 5.04 (0.69) | 4.94 (1.32) | 5.32 (0.70) | 5.50 (0.92) | 4.95 (0.96) | 4.90 (1.04) | 0.30 | |
| TGL (mmol/L) o | Baseline | 1.19 (0.62) | 1.65 (1.18) | 1.39 (0.99) | 1.39 (0.66) | 1.63 (1.06) | 1.63 (0.89) | 0.78 |
| 45 days | 1.07 (0.52) | 1.45 (0.93) | 1.25 (0.66) | 1.18 (0.74) | 1.32 (1.11) | 1.23 (0.84) | 0.80 | |
| 90 days | 1.08 (0.56) | 1.42 (0.77) | 1.22 (0.62) | 1.38 (0.73) | 1.15 (0.62) | 1.30 (0.88) | 0.73 | |
| HDL-C (mmol/L) p | Baseline | 1.22 (0.34) | 1.18 (0.36) | 1.13 (0.27) | 1.16 (0.30) | 1.14 (0.22) | 1.09 (0.29) | 0.80 |
| 45 days | 1.19 (0.33) | 1.12 (0.36) | 1.06 (0.24) | 1.16 (0.30) | 1.29 (0.78) | 1.04 (0.32) | 0.39 | |
| 90 days | 1.16 (0.33) | 1.24 (0.46) | 1.15 (0.21) | 1.24 (0.27) | 1.21 (0.23) | 1.11 (0.28) | 0.70 | |
| HOMA-IR q | Baseline | 2.93 (1.69) | 2.83 (1.26) | 3.01 (1.68) | 2.51 (1.59) | 3.24 (1.76) | 3.20 (1.36) | 0.81 |
| 45 days | 2.14 (1.15) | 2.59 (1.86) | 2.05 (1.02) | 1.90 (1.20) | 2.05 (0.87) | 1.99 (0.91) | 0.60 | |
| 90 days | 2.06 (1.27) | 2.80 (1.46) | 2.32 (1.20) | 2.33 (2.33) | 2.59 (1.69) | 2.43 (1.38) | 0.65 | |
| HbA1c (mmol/mol) r | Baseline | 0.06 (0.00) | 0.06 (0.01) | 0.06 (0.01) | 0.06 (0.01) | 0.06 (0.00) | 0.06 (0.00) | 0.83 |
| 45 days | 0.06 (0.00) | 0.06 (0.01) | 0.06 (0.00) | 0.06 (0.01) | 0.06 (0.00) | 0.06 (0.01) | 0.85 | |
| 90 days | 0.06 (0.00) | 0.05 (0.00) | 0.06 (0.00) | 0.06 (0.01) | 0.05 (0.00) | 0.06 (0.00) | 0.47 | |
| C a | LGIMD b | PA1 c | PA2 d | PA1 + LGIMD5 e | PA2 + LGIMD f | p-Value | |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | ||
| n | 6 | 5 | 6 | 7 | 10 | 10 | |
| Age (Years) | 47.17 (8.66) | 54.60 (10.83) | 47.00 (7.92) | 46.43 (7.04) | 47.70 (9.01) | 43.50 (7.52) | 0.34 |
| CAP (dB/m) g | |||||||
| Baseline | 270.50 (18.79) | 280.00 (11.90) | 281.50 (15.06) | 277.71 (18.67) | 275.60 (16.27) | 273.30 (16.73) | 0.85 |
| 45 days | 255.00 (86.60) | 266.20 (42.50) | 274.83 (72.58) | 295.86 (53.75) | 249.75 (43.75) | 245.90 (60.18) | 0.62 |
| 90 days | 241.50 (21.27) | 257.00 (100.84) | 292.17 (77.79) | 302.33 (36.84) | 243.14 (21.70) | 231.33 (45.21) | 0.099 |
| HOMA-IR h | |||||||
| Baseline | 1.85 (0.78) | 1.88 (0.71) | 1.81 (0.87) | 1.71 (0.66) | 2.55 (0.77) | 2.49 (0.92) | 0.17 |
| 45 days | 1.39 (0.57) | 1.91 (1.00) | 1.96 (1.07) | 0.98 (0.49) | 1.80 (0.45) | 1.76 (0.54) | 0.21 |
| 90 days | 1.12 (0.32) | 2.09 (0.86) | 1.76 (1.02) | 1.33 (0.73) | 2.57 (2.64) | 1.92 (0.74) | 0.44 |
| FM i | |||||||
| Baseline | 25.70 (11.19) | 25.92 (8.68) | 25.43 (3.17) | 27.66 (3.72) | 32.58 (7.72) | 31.52 (6.33) | 0.21 |
| 45 days | 22.32 (9.45) | 25.46 (8.55) | 23.17 (3.62) | 25.37 (3.82) | 29.38 (6.99) | 27.14 (4.96) | 0.29 |
| 90 days | 20.83 (11.33) | 21.40 (6.85) | 22.05 (2.88) | 24.05 (3.62) | 26.73 (7.27) | 24.50 (4.25) | 0.62 |
| FFM j | |||||||
| Baseline | 55.85 (5.76) | 57.44 (9.47) | 58.98 (14.47) | 64.06 (11.29) | 60.62 (12.94) | 60.58 (11.87) | 0.85 |
| 45 days | 55.82 (4.57) | 54.24 (9.87) | 58.72 (13.60) | 63.89 (10.34) | 58.71 (11.24) | 59.37 (11.05) | 0.70 |
| 90 days | 56.80 (5.24) | 56.10 (12.96) | 59.62 (14.66) | 65.18 (9.99) | 60.13 (12.59) | 61.03 (10.89) | 0.81 |
| BMI k | |||||||
| Baseline | 29.90 (4.20) | 30.38 (4.74) | 29.47 (3.65) | 30.27 (1.78) | 32.77 (3.56) | 32.38 (2.16) | 0.25 |
| 45 days | 28.62 (3.54) | 29.04 (4.94) | 28.57 (3.34) | 29.51 (1.63) | 31.04 (2.98) | 30.44 (1.57) | 0.48 |
| 90 days | 28.42 (3.58) | 27.25 (4.99) | 28.50 (3.33) | 28.83 (2.01) | 30.33 (3.28) | 29.97 (1.44) | 0.62 |
| WC (cm) l | |||||||
| Baseline | 92.17 (7.83) | 98.00 (12.67) | 96.83 (7.36) | 97.14 (6.89) | 99.80 (10.57) | 98.90 (10.06) | 0.74 |
| 45 days | 89.83 (3.13) | 96.00 (14.87) | 93.83 (10.05) | 97.14 (5.96) | 95.50 (10.39) | 96.00 (10.06) | 0.80 |
| 90 days | 89.50 (5.32) | 93.50 (15.67) | 93.33 (10.03) | 98.33 (4.59) | 96.14 (9.03) | 92.78 (6.57) | 0.57 |
| HC (cm) m | |||||||
| Baseline | 104.83 (15.04) | 107.60 (8.02) | 104.17 (5.56) | 107.86 (6.84) | 107.10 (8.62) | 109.70 (7.63) | 0.85 |
| 45 days | 104.00 (12.15) | 103.00 (7.84) | 103.17 (6.21) | 105.57 (10.15) | 104.60 (8.38) | 104.80 (5.31) | 0.99 |
| 90 days | 101.83 (13.47) | 101.00 (8.83) | 102.17 (3.76) | 102.50 (6.80) | 103.00 (11.06) | 102.89 (4.86) | 1.00 |
| GLUCOSE (mmol/L) | |||||||
| Baseline | 5.07 (0.32) | 5.03 (0.37) | 5.01 (0.50) | 5.31 (0.43) | 5.15 (0.46) | 5.15 (0.29) | 0.78 |
| 45 days | 4.97 (0.28) | 5.06 (0.38) | 4.93 (0.59) | 5.23 (0.31) | 5.03 (0.38) | 5.08 (0.49) | 0.84 |
| 90 days | 4.89 (0.38) | 5.27 (0.70) | 5.15 (0.56) | 5.38 (0.59) | 5.04 (0.68) | 5.14 (0.27) | 0.68 |
| TGL (mmol/L) n | |||||||
| Baseline | 1.05 (0.57) | 0.91 (0.34) | 1.05 (0.35) | 1.28 (0.27) | 1.23 (0.74) | 1.82 (1.02) | 0.12 |
| 45 days | 0.94 (0.40) | 0.82 (0.43) | 1.00 (0.41) | 1.09 (0.57) | 1.22 (0.98) | 1.05 (0.62) | 0.91 |
| 90 days | 0.84 (0.16) | 0.83 (0.71) | 1.00 (0.52) | 1.23 (0.38) | 1.10 (0.82) | 1.29 (1.28) | 0.87 |
| HDL-C(mmol/L) o | |||||||
| Baseline | 1.44 (0.47) | 1.49 (0.35) | 1.16 (0.31) | 1.17 (0.15) | 1.18 (0.25) | 1.22 (0.36) | 0.30 |
| 45 days | 1.45 (0.38) | 1.36 (0.41) | 1.10 (0.29) | 1.19 (0.12) | 1.14 (0.20) | 1.19 (0.40) | 0.31 |
| 90 days | 1.36 (0.39) | 1.51 (0.58) | 1.21 (0.22) | 1.20 (0.17) | 1.17 (0.22) | 1.24 (0.35) | 0.60 |
| HbA1c (mmol/mol) p | |||||||
| Baseline | 0.06 (0.00) | 0.06 (0.00) | 0.05 (0.01) | 0.06 (0.00) | 0.06 (0.00) | 0.06 (0.01) | 0.57 |
| 45 days | 0.06 (0.00) | 0.06 (0.00) | 0.05 (0.01) | 0.06 (0.00) | 0.06 (0.00) | 0.06 (0.01) | 0.57 |
| 90 days | 0.06 (0.00) | 0.05 (0.00) | 0.06 (0.01) | 0.05 (0.00) | 0.06 (0.00) | 0.06 (0.01) | 0.94 |
| C a | LGIMD b | PA1 c | PA2 d | PA1 + LGIMD5 e | PA2 + LGIMD f | p-Value | |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | ||
| N | 16 | 18 | 19 | 16 | 17 | 14 | |
| Age (Years) | 52.21 (8.53) | 56.00 (10.39) | 51.93 (9.93) | 46.13 (10.53) | 52.07 (9.90) | 49.07 (11.91) | 0.15 |
| CAP (dB/m) g | |||||||
| Baseline | 378.56 (39.66) | 358.67 (34.39) | 358.37 (33.58) | 359.75 (38.63) | 359.71 (38.34) | 359.57 (40.28) | 0.60 |
| 45 days | 358.73 (56.06) | 309.87 (50.26) | 306.50 (38.53) | 301.27 (69.61) | 289.40 (86.57) | 320.43 (60.87) | 0.13 |
| 90 days | 352.00 (59.79) | 321.38 (75.47) | 285.55 (41.53) | 275.00 (71.38) | 269.58 (69.15) | 329.23 (60.87) | 0.019 |
| HOMA-IR h | |||||||
| Baseline | 3.34 (1.77) | 3.11 (1.26) | 3.41 (1.71) | 2.86 (1.76) | 3.58 (2.02) | 3.79 (1.40) | 0.72 |
| 45 days | 2.45 (1.20) | 3.07 (2.02) | 2.00 (1.05) | 2.25 (1.22) | 2.19 (1.03) | 2.23 (1.18) | 0.50 |
| 90 days | 2.68 (1.29) | 3.23 (1.43) | 2.66 (1.21) | 2.84 (2.71) | 2.60 (1.02) | 2.78 (1.62) | 0.95 |
| FM i | |||||||
| Baseline | 37.86 (11.08) | 32.72 (10.77) | 33.17 (11.98) | 28.26 (8.38) | 30.01 (7.67) | 35.59 (10.64) | 0.14 |
| 45 days | 35.52 (9.68) | 27.76 (9.71) | 31.90 (11.61) | 27.00 (9.12) | 27.85 (6.52) | 32.02 (9.01) | 0.15 |
| 90 days | 35.81 (10.37) | 26.45 (9.82) | 30.17 (10.48) | 26.16 (9.13) | 26.25 (7.67) | 30.98 (9.56) | 0.15 |
| FFM j | |||||||
| Baseline | 60.45 (11.24) | 59.81 (9.45) | 61.47 (10.47) | 61.83 (7.59) | 62.44 (11.33) | 64.67 (12.42) | 0.85 |
| 45 days | 58.53 (11.61) | 59.72 (7.76) | 60.43 (9.60) | 60.97 (6.57) | 60.42 (10.56) | 62.92 (11.01) | 0.91 |
| 90 days | 61.02 (12.45) | 61.20 (7.76) | 60.37 (8.06) | 62.71 (6.45) | 60.22 (10.86) | 62.68 (12.40) | 0.98 |
| BMI k | |||||||
| Baseline | 35.96 (4.18) | 33.61 (3.98) | 34.14 (5.48) | 31.03 (3.67) | 33.27 (4.68) | 35.31 (4.26) | 0.055 |
| 45 days | 34.44 (3.77) | 31.51 (4.01) | 33.26 (5.15) | 30.31 (3.83) | 31.83 (4.09) | 33.44 (3.87) | 0.12 |
| 90 days | 34.59 (4.53) | 31.34 (4.57) | 32.38 (4.88) | 30.28 (4.08) | 31.20 (4.30) | 33.05 (4.09) | 0.28 |
| WC (cm) l | |||||||
| Baseline | 112.00 (7.75) | 104.28 (10.45) | 106.05 (7.84) | 100.81 (8.54) | 105.65 (12.38) | 108.43 (12.26) | 0.051 |
| 45 days | 108.25 (10.18) | 98.93 (7.43) | 104.14 (8.20) | 97.40 (7.30) | 99.53 (8.01) | 102.79 (9.66) | 0.015 |
| 90 days | 107.90 (7.89) | 97.46 (7.14) | 102.73 (6.07) | 96.62 (7.38) | 99.02 (9.05) | 103.15 (10.91) | 0.017 |
| HC (cm) m | |||||||
| Baseline | 116.88 (9.12) | 111.11 (10.78) | 111.53 (9.96) | 106.31 (8.48) | 108.00 (7.67) | 112.71 (8.37) | 0.029 |
| 45 days | 112.08 (7.59) | 106.13 (9.62) | 108.50 (12.79) | 105.00 (7.58) | 104.73 (6.45) | 110.21 (11.39) | 0.26 |
| 90 days | 110.60 (9.00) | 105.15 (8.78) | 109.36 (9.56) | 102.77 (8.32) | 104.15 (7.12) | 110.00 (7.80) | 0.099 |
| GLUCOSE (mmol/L) | |||||||
| Baseline | 5.42 (0.75) | 6.14 (1.40) | 6.02 (1.87) | 5.45 (0.86) | 5.49 (0.68) | 5.73 (0.76) | 0.31 |
| 45 days | 5.15 (0.45) | 6.11 (1.10) | 5.62 (1.50) | 5.11 (0.74) | 5.31 (0.74) | 5.56 (0.61) | 0.056 |
| 90 days | 5.23 (0.37) | 6.24 (1.47) | 5.84 (1.84) | 5.22 (1.27) | 5.37 (0.99) | 5.52 (0.61) | 0.28 |
| TGL (mmol/L) n | |||||||
| Baseline | 1.25 (0.64) | 1.86 (1.26) | 1.50 (1.10) | 1.44 (0.77) | 1.87 (1.16) | 1.48 (0.79) | 0.40 |
| 45 days | 1.13 (0.58) | 1.72 (0.95) | 1.29 (0.71) | 1.23 (0.83) | 1.39 (1.22) | 1.37 (0.97) | 0.62 |
| 90 days | 1.25 (0.67) | 1.70 (0.64) | 1.34 (0.65) | 1.45 (0.87) | 1.18 (0.52) | 1.30 (0.52) | 0.44 |
| HDL-C (mmol/L) o | |||||||
| Baseline | 1.13 (0.25) | 1.10 (0.32) | 1.12 (0.26) | 1.16 (0.35) | 1.12 (0.20) | 1.00 (0.18) | 0.70 |
| 45 days | 1.05 (0.21) | 0.99 (0.25) | 1.05 (0.23) | 1.14 (0.36) | 1.40 (0.99) | 0.92 (0.17) | 0.13 |
| 90 days | 1.02 (0.21) | 1.06 (0.22) | 1.12 (0.22) | 1.26 (0.31) | 1.23 (0.25) | 1.01 (0.18) | 0.046 |
| HbA1c (mmol/mol) p | |||||||
| Baseline | 0.06 (0.01) | 0.06 (0.01) | 0.06 (0.01) | 0.06 (0.01) | 0.06 (0.01) | 0.06 (0.00) | 0.52 |
| 45 days | 0.06 (0.00) | 0.06 (0.01) | 0.06 (0.00) | 0.06 (0.01) | 0.05 (0.00) | 0.06 (0.01) | 0.77 |
| 90 days | 0.06 (0.00) | 0.05 (0.00) | 0.06 (0.00) | 0.06 (0.01) | 0.05 (0.00) | 0.06 (0.00) | 0.37 |
References
- Ludwig, J.; Viggiano, T.R.; McGill, D.B.; Oh, B.J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. 1980, 55, 434–438. [Google Scholar] [PubMed]
- Ratziu, V.; Bellentani, S.; Cortez-Pinto, H.; Day, C.; Marchesini, G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J. Hepatol. 2010, 53, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Osella, A.R.; Diaz Mdel, P.; Cozzolongo, R.; Bonfiglio, C.; Franco, I.; Abrescia, D.I.; Bianco, A.; Giampiero, E.S.; Petruzzi, J.; Elsa, L.; et al. Overweight and Obesity in Southern Italy: Their association with social and life-style characteristics and their effect on levels of biologic markers. Rev. Fac. Cien Med. Univ. Nac. Cordoba 2014, 71, 113–124. [Google Scholar]
- Cozzolongo, R.; Osella, A.R.; Elba, S.; Petruzzi, J.; Buongiorno, G.; Giannuzzi, V.; Leone, G.; Bonfiglio, C.; Lanzilotta, E.; Manghisi, O.G.; et al. Epidemiology of HCV infection in the general population: A survey in a southern Italian town. Am. J. Gastroenterol. 2009, 104, 2740–2746. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, Q.; Wang, T.; Wen, J.; Wang, H.; Zhang, T. Controlled attenuation parameter for assessment of hepatic steatosis grades: A diagnostic meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 17654–17663. [Google Scholar]
- Rockey, D.C.; Caldwell, S.H.; Goodman, Z.D.; Nelson, R.C.; Smith, A.D. Liver biopsy. Hepatology 2009, 49, 1017–1044. [Google Scholar] [CrossRef]
- Grandison, G.A.; Angulo, P. Can NASH be diagnosed, graded, and staged noninvasively? Clin. Liver Dis. 2012, 16, 567–585. [Google Scholar] [CrossRef]
- Lee, S.S.; Park, S.H.; Kim, H.J.; Kim, S.Y.; Kim, M.Y.; Kim, D.Y.; Suh, D.J.; Kim, K.M.; Bae, M.H.; Lee, J.Y.; et al. Non-invasive assessment of hepatic steatosis: Prospective comparison of the accuracy of imaging examinations. J. Hepatol. 2010, 52, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Cesbron-Métivier, E.; Roullier, V.; Boursier, J.; Cavaro-Ménard, C.; Lebigot, J.; Michalak, S.; Calès, P.; Aubé, C. Noninvasive liver steatosis quantification using MRI techniques combined with blood markers. Eur. J. Gastroenterol. Hepatol. 2010, 22, 973–982. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, C.; Becker, U.; Winkler, K.; Christoffersen, P.; Jensen, M.; Henriksen, O. Quantification of liver fat using magnetic resonance spectroscopy. Magn. Reson. Imaging 1994, 12, 487–495. [Google Scholar] [CrossRef]
- Sasso, M.; Beaugrand, M.; De Ledinghen, V.; Douvin, C.; Marcellin, P.; Poupon, R.; Sandrin, L.; Miette, V. Controlled attenuation parameter (CAP): A novel VCTE™ guided ultrasonic attenuation measurement for the evaluation of hepatic steatosis: Preliminary study and validation in a cohort of patients with chronic liver disease from various causes. Ultrasound Med. Biol. 2010, 36, 1825–1835. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Muir, A.J.; Dieterich, D.T.; Falck-Ytter, Y.T. American Gastroenterological Association Institute Technical Review on the Role of Elastography in Chronic Liver Diseases. Gastroenterology 2017, 152, 1544–1577. [Google Scholar] [CrossRef] [PubMed]
- Friedrich-Rust, M.; Romen, D.; Vermehren, J.; Kriener, S.; Sadet, D.; Herrmann, E.; Zeuzem, S.; Bojunga, J. Acoustic radiation force impulse-imaging and transient elastography for non-invasive assessment of liver fibrosis and steatosis in NAFLD. Eur. J. Radiol. 2012, 81, e325–e331. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Park, S.Y.; Kim, S.U.; Jang, J.Y.; Park, H.; Kim, J.K.; Lee, C.K.; Chon, Y.E.; Han, K.H. Discrimination of Nonalcoholic Steatohepatitis Using Transient Elastography in Patients with Nonalcoholic Fatty Liver Disease. PLoS ONE 2016, 11, e0157358. [Google Scholar] [CrossRef] [PubMed]
- Mahady, S.E.; George, J. Management of nonalcoholic steatohepatitis: An evidence-based approach. Clin. Liver Dis. 2012, 16, 631–645. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Abbate, R.; Gensini, G.F.; Casini, A. Accruing evidence on benefits of adherence to the Mediterranean diet on health: An updated systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 92, 1189–1196. [Google Scholar] [CrossRef]
- Galbete, C.; Toledo, E.; Toledo, J.B.; Bes-Rastrollo, M.; Buil-Cosiales, P.; Marti, A.; Guillén-Grima, F.; Martínez-González, M.A. Mediterranean diet and cognitive function: The SUN project. J. Nutr. Health Aging 2015, 19, 305–312. [Google Scholar] [CrossRef]
- Kontogianni, M.D.; Tileli, N.; Margariti, A.; Georgoulis, M.; Deutsch, M.; Tiniakos, D.; Fragopoulou, E.; Zafiropoulou, R.; Manios, Y.; Papatheodoridis, G. Adherence to the Mediterranean diet is associated with the severity of non-alcoholic fatty liver disease. Clin. Nutr. 2014, 33, 678–683. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Salomone, F.; Mlynarsky, L. The Mediterranean dietary pattern as the diet of choice for non-alcoholic fatty liver disease: Evidence and plausible mechanisms. Liver Int. Off. J. Int. Assoc. Study Liver 2017, 37, 936–949. [Google Scholar] [CrossRef]
- Castro-Barquero, S.; Ruiz-León, A.M.; Sierra-Pérez, M.; Estruch, R.; Casas, R. Dietary Strategies for Metabolic Syndrome: A Comprehensive Review. Nutrients 2020, 12, 2983. [Google Scholar] [CrossRef]
- Thoma, C.; Day, C.P.; Trenell, M.I. Lifestyle interventions for the treatment of non-alcoholic fatty liver disease in adults: A systematic review. J. Hepatol. 2012, 56, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Hashida, R.; Kawaguchi, T.; Bekki, M.; Omoto, M.; Matsuse, H.; Nago, T.; Takano, Y.; Ueno, T.; Koga, H.; George, J.; et al. Aerobic vs. resistance exercise in non-alcoholic fatty liver disease: A systematic review. J. Hepatol. 2017, 66, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Piercy, K.L.; Troiano, R.P. Physical activity guidelines for Americans from the US department of health and human services: Cardiovascular benefits and recommendations. Circ. Cardiovasc. Qual. Outcomes 2018, 11, e005263. [Google Scholar] [CrossRef]
- Ryu, S.; Chang, Y.; Jung, H.S.; Yun, K.E.; Kwon, M.J.; Choi, Y.; Kim, C.W.; Cho, J.; Suh, B.S.; Cho, Y.K.; et al. Relationship of sitting time and physical activity with non-alcoholic fatty liver disease. J. Hepatol. 2015, 63, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef]
- Kwak, M.-S.; Kim, D. Non-alcoholic fatty liver disease and lifestyle modifications, focusing on physical activity. Korean J. Int. Med. 2018, 33, 64. [Google Scholar] [CrossRef]
- Misciagna, G.; Del Pilar Diaz, M.; Caramia, D.V.; Bonfiglio, C.; Franco, I.; Noviello, M.R.; Chiloiro, M.; Abbrescia, D.I.; Mirizzi, A.; Tanzi, M.; et al. Effect of a Low Glycemic Index Mediterranean Diet on Non-Alcoholic Fatty Liver Disease. A Randomized Controlled Clinici Trial. J. Nutr. Health Aging 2017, 21, 404–412. [Google Scholar] [CrossRef]
- Franco, I.; Bianco, A.; Dìaz, M.D.P.; Bonfiglio, C.; Chiloiro, M.; Pou, S.A.; Becaria Coquet, J.; Mirizzi, A.; Nitti, A.; Campanella, A.; et al. Effectiveness of two physical activity programs on non-alcoholic fatty liver disease. A randomized controlled clinical trial. Rev. Fac. Cienc. Méd. Córdoba 2019, 76, 26. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- Elia, A. La Rivoluzione Degli Integrali Buoni (Senza L’Aggiunta di Crusca o Fibre) (The Revolution of Good WholeGrains: Without Bran and Fibers Addition); Di Marsico Libri: Bari, Italy, 2010. [Google Scholar]
- Tutino, V.; De Nunzio, V.; Caruso, M.G.; Bonfiglio, C.; Franco, I.; Mirizzi, A.; De Leonardis, G.; Cozzolongo, R.; Giannuzzi, V.; Giannelli, G.; et al. Aerobic Physical Activity and a Low Glycemic Diet Reduce the AA/EPA Ratio in Red Blood Cell Membranes of Patients with NAFLD. Nutrients 2018, 10, 1299. [Google Scholar] [CrossRef] [PubMed]
- Misciagna, G.; Leoci, C.; Guerra, V.; Chiloiro, M.; Elba, S.; Petruzzi, J.; Mossa, A.; Noviello, M.R.; Coviello, A.; Minutolo, M.C.; et al. Epidemiology of cholelithiasis in southern Italy. Part II: Risk factors. Eur. J. Gastroenterol. Hepatol. 1996, 8, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Chiloiro, M.; Caruso, M.G.; Cisternino, A.M.; Inguaggiato, R.; Reddavide, R.; Bonfiglio, C.; Guerra, V.; Notarnicola, M.; De Michele, G.; Correale, M.; et al. Ultrasound evaluation and correlates of fatty liver disease: A population study in a Mediterranean area. Metab. Syndr. Relat. Disord. 2013, 11, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Riboli, E.; Hunt, K.; Slimani, N.; Ferrari, P.; Norat, T.; Fahey, M.; Charrondière, U.; Hémon, B.; Casagrande, C.; Vignat, J.; et al. European Prospective Investigation into Cancer and Nutrition (EPIC): Study populations and data collection. Public Health Nutr. 2002, 5, 1113–1124. [Google Scholar] [CrossRef]
- Cialfa, E.; D’amicis, A.; Leclercq, C.; Quaglia, G.; Sette, S.; Ticca, M.; Tomassi, G. Linee guida per una sana alimentazione italiana. Rome Ist. Naz. Ric. Gli Aliment. Nutr. 2003, 86, 7–94. [Google Scholar]
- Alberti-Fidanza, A.; Fidanza, F. Mediterranean Adequacy Index of Italian diets. Public Health Nutr. 2004, 7, 937–941. [Google Scholar] [CrossRef]
- Cannella, C.P.L. Available from: Storage; National Research Institute for Food and Nutrition (INRAN): Rome, Italy, 2008. [Google Scholar]
- Laukkanen, R.; Oja, P.; Pasanen, M.; Vuori, I. Validity of a two kilometre walking test for estimating maximal aerobic power in overweight adults. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 1992, 16, 263–268. [Google Scholar]
- Physiology, C.S.f.E. The Canadian Physical Activity, Fitness and Lifestyle Approach (CPAFLA): CSEP—Health and Fitness Program’s Health-Related Appraisal and Counselling Strategy; Canadian Society for Exercise Physiology: Ottawa, ON, Canada, 2003. [Google Scholar]
- Hoeger, W.W.; Hopkins, D.R. A comparison of the sit and reach and the modified sit and reach in the measurement of flexibility in women. Res. Q. Exerc. Sport 1992, 63, 191–195. [Google Scholar] [CrossRef]
- Welk, G. Physical Activity Assessments for Health-Related Research; Human Kinetics: Champaign, IL, USA, 2002. [Google Scholar]
- Thompson, D.; Karpe, F.; Lafontan, M.; Frayn, K. Physical activity and exercise in the regulation of human adipose tissue physiology. Physiol. Rev. 2012, 92, 157–191. [Google Scholar] [CrossRef]
- Pate, R.R.; Pratt, M.; Blair, S.N.; Haskell, W.L.; Macera, C.A.; Bouchard, C.; Buchner, D.; Ettinger, W.; Heath, G.W.; King, A.C. Physical activity and public health: A recommendation from the Centers for Disease Control and Prevention and the American College of Sports Medicine. JAMA 1995, 273, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, S.U. Noninvasive monitoring of hepatic steatosis: Controlled attenuation parameter and magnetic resonance imaging-proton density fat fraction in patients with nonalcoholic fatty liver disease. Expert Rev. Gastroenterol. Hepatol. 2019, 13, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Marchesini, G.; Brizi, M.; Morselli-Labate, A.M.; Bianchi, G.; Bugianesi, E.; McCullough, A.J.; Forlani, G.; Melchionda, N. Association of nonalcoholic fatty liver disease with insulin resistance. Am. J. Med. 1999, 107, 450–455. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334. [Google Scholar] [CrossRef]
- Ibrahim, A.A.; Abdelbasset, W.K. The role of physical exercise in treating people with non-alcoholic fatty liver disease. J. Adv. Pharm. Educ. Res. 2020, 10, 64–70. [Google Scholar]
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Zvibel, I.; Goldiner, I.; Blendis, L.; Halpern, Z.; Oren, R. Role of leisure-time physical activity in nonalcoholic fatty liver disease: A population-based study. Hepatology 2008, 48, 1791–1798. [Google Scholar] [CrossRef]
- Keating, S.E.; Hackett, D.A.; Parker, H.M.; O’Connor, H.T.; Gerofi, J.A.; Sainsbury, A.; Baker, M.K.; Chuter, V.H.; Caterson, I.D.; George, J.; et al. Effect of aerobic exercise training dose on liver fat and visceral adiposity. J. Hepatol. 2015, 63, 174–182. [Google Scholar] [CrossRef]
- Bacchi, E.; Negri, C.; Targher, G.; Faccioli, N.; Lanza, M.; Zoppini, G.; Zanolin, E.; Schena, F.; Bonora, E.; Moghetti, P. Both resistance training and aerobic training reduce hepatic fat content in type 2 diabetic subjects with nonalcoholic fatty liver disease (the RAED2 Randomized Trial). Hepatology 2013, 58, 1287–1295. [Google Scholar] [CrossRef]
- Lonardo, A.; Bellentani, S.; Argo, C.K.; Ballestri, S.; Byrne, C.D.; Caldwell, S.H.; Cortez-Pinto, H.; Grieco, A.; Machado, M.V.; Miele, L. Epidemiological modifiers of non-alcoholic fatty liver disease: Focus on high-risk groups. Dig. Liver Dis. 2015, 47, 997–1006. [Google Scholar] [CrossRef]
- Esposito, K.; Giugliano, D. Mediterranean diet and the metabolic syndrome: The end of the beginning. Metab. Syndr. Relat. Disord. 2010, 8, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Gelli, C.; Tarocchi, M.; Abenavoli, L.; Di Renzo, L.; Galli, A.; De Lorenzo, A. Effect of a counseling-supported treatment with the Mediterranean diet and physical activity on the severity of the non-alcoholic fatty liver disease. World J. Gastroenterol. 2017, 23, 3150–3162. [Google Scholar] [CrossRef] [PubMed]
- Franz, M.J.; VanWormer, J.J.; Crain, A.L.; Boucher, J.L.; Histon, T.; Caplan, W.; Bowman, J.D.; Pronk, N.P. Weight-loss outcomes: A systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J. Am. Diet. Assoc. 2007, 107, 1755–1767. [Google Scholar] [CrossRef]
- He, J.; Van de Vijver, F.J.; Dominguez Espinosa, A.; Abubakar, A.; Dimitrova, R.; Adams, B.G.; Aydinli, A.; Atitsogbe, K.; Alonso-Arbiol, I.; Bobowik, M. Socially desirable responding: Enhancement and denial in 20 countries. Cross-Cult. Res. 2015, 49, 227–249. [Google Scholar] [CrossRef]
- Baba, C.S.; Alexander, G.; Kalyani, B.; Pandey, R.; Rastogi, S.; Pandey, A.; Choudhuri, G. Effect of exercise and dietary modification on serum aminotransferase levels in patients with nonalcoholic steatohepatitis. J. Gastroenterol. Hepatol. 2006, 21, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.A.; Sachinwalla, T.; Walton, D.W.; Smith, K.; Armstrong, A.; Thompson, M.W.; George, J. Aerobic exercise training reduces hepatic and visceral lipids in obese individuals without weight loss. Hepatology 2009, 50, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Albright, A.; Franz, M.; Hornsby, G.; Kriska, A.; Marrero, D.; Ullrich, I.; Verity, L.S. American College of Sports Medicine position stand. Exercise and type 2 diabetes. Med. Sci. Sports Exerc. 2000, 32, 1345–1360. [Google Scholar] [PubMed]
- Pollock, M.L.; Franklin, B.A.; Balady, G.J.; Chaitman, B.L.; Fleg, J.L.; Fletcher, B.; Limacher, M.; Piña, I.L.; Stein, R.A.; Williams, M. Resistance exercise in individuals with and without cardiovascular disease: Benefits, rationale, safety, and prescription an advisory from the committee on exercise, rehabilitation, and prevention, council on clinical cardiology, American Heart Association. Circulation 2000, 101, 828–833. [Google Scholar] [CrossRef]
- Hallsworth, K.; Fattakhova, G.; Hollingsworth, K.G.; Thoma, C.; Moore, S.; Taylor, R.; Day, C.P.; Trenell, M.I. Resistance exercise reduces liver fat and its mediators in non-alcoholic fatty liver disease independent of weight loss. Gut 2011, 60, 1278–1283. [Google Scholar] [CrossRef]
- El-Agroudy, N.N.; Kurzbach, A.; Rodionov, R.N.; O’Sullivan, J.; Roden, M.; Birkenfeld, A.L.; Pesta, D.H. Are lifestyle therapies effective for NAFLD treatment? Trends Endocrinol. Metab. 2019, 30, 701–709. [Google Scholar] [CrossRef]
- Katsagoni, C.N.; Georgoulis, M.; Papatheodoridis, G.V.; Panagiotakos, D.B.; Kontogianni, M.D. Effects of lifestyle interventions on clinical characteristics of patients with non-alcoholic fatty liver disease: A meta-analysis. Metabolism 2017, 68, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Arfianti, A.; Pok, S.; Barn, V.; Haigh, W.G.; Yeh, M.M.; Ioannou, G.N.; Teoh, N.C.; Farrell, G.C. Exercise retards hepatocarcinogenesis in obese mice independently of weight control. J. Hepatol. 2020, 73, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Promrat, K.; Kleiner, D.E.; Niemeier, H.M.; Jackvony, E.; Kearns, M.; Wands, J.R.; Fava, J.L.; Wing, R.R. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010, 51, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Vilar-Gomez, E.; Martinez-Perez, Y.; Calzadilla-Bertot, L.; Torres-Gonzalez, A.; Gra-Oramas, B.; Gonzalez-Fabian, L.; Friedman, S.L.; Diago, M.; Romero-Gomez, M. Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis. Gastroenterology 2015, 149, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Asrih, M.; Jornayvaz, F.R. Diets and nonalcoholic fatty liver disease: The good and the bad. Clin. Nutr. 2014, 33, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Mouzaki, M.; Allard, J.P. The role of nutrients in the development, progression, and treatment of nonalcoholic fatty liver disease. J. Clin. Gastroenterol. 2012, 46, 457–467. [Google Scholar] [CrossRef]
- Oeda, S.; Tanaka, K.; Oshima, A.; Matsumoto, Y.; Sueoka, E.; Takahashi, H. Diagnostic Accuracy of FibroScan and Factors Affecting Measurements. Diagnostics 2020, 10, 940. [Google Scholar] [CrossRef]
- Saadeh, S.; Younossi, Z.M.; Remer, E.M.; Gramlich, T.; Ong, J.P.; Hurley, M.; Mullen, K.D.; Cooper, J.N.; Sheridan, M.J. The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002, 123, 745–750. [Google Scholar] [CrossRef]
- Ryan, M.C.; Itsiopoulos, C.; Thodis, T.; Ward, G.; Trost, N.; Hofferberth, S.; O’Dea, K.; Desmond, P.V.; Johnson, N.A.; Wilson, A.M. The Mediterranean diet improves hepatic steatosis and insulin sensitivity in individuals with non-alcoholic fatty liver disease. J. Hepatol. 2013, 59, 138–143. [Google Scholar] [CrossRef]
- Papadaki, A.; Nolen-Doerr, E.; Mantzoros, C.S. The Effect of the Mediterranean Diet on Metabolic Health: A Systematic Review and Meta-Analysis of Controlled Trials in Adults. Nutrients 2020, 12, 3342. [Google Scholar] [CrossRef]
- Zou, T.T.; Zhang, C.; Zhou, Y.F.; Han, Y.J.; Xiong, J.J.; Wu, X.X.; Chen, Y.P.; Zheng, M.H. Lifestyle interventions for patients with nonalcoholic fatty liver disease: A network meta-analysis. Eur. J. Gastroenterol. Hepatol. 2018, 30, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Campanella, A.; Misciagna, G.; Mirizzi, A.; Caruso, M.G.; Bonfiglio, C.; Aballay, L.R.; Vas de Arruda Silveira, L.; Bianco, A.; Franco, I.; Sorino, P.; et al. The effect of the Mediterranean Diet on lifespan. A treatment-effect survival analysis of a population-based prospective cohort study in Southern Italy. Int. J. Epidemiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Montori, V.M.; Guyatt, G.H. Intention-to-treat principle. CMAJ 2001, 165, 1339–1341. [Google Scholar] [PubMed]
- Nseir, W.; Hellou, E.; Assy, N. Role of diet and lifestyle changes in nonalcoholic fatty liver disease. World J. Gastroenterol. WJG 2014, 20, 9338. [Google Scholar]
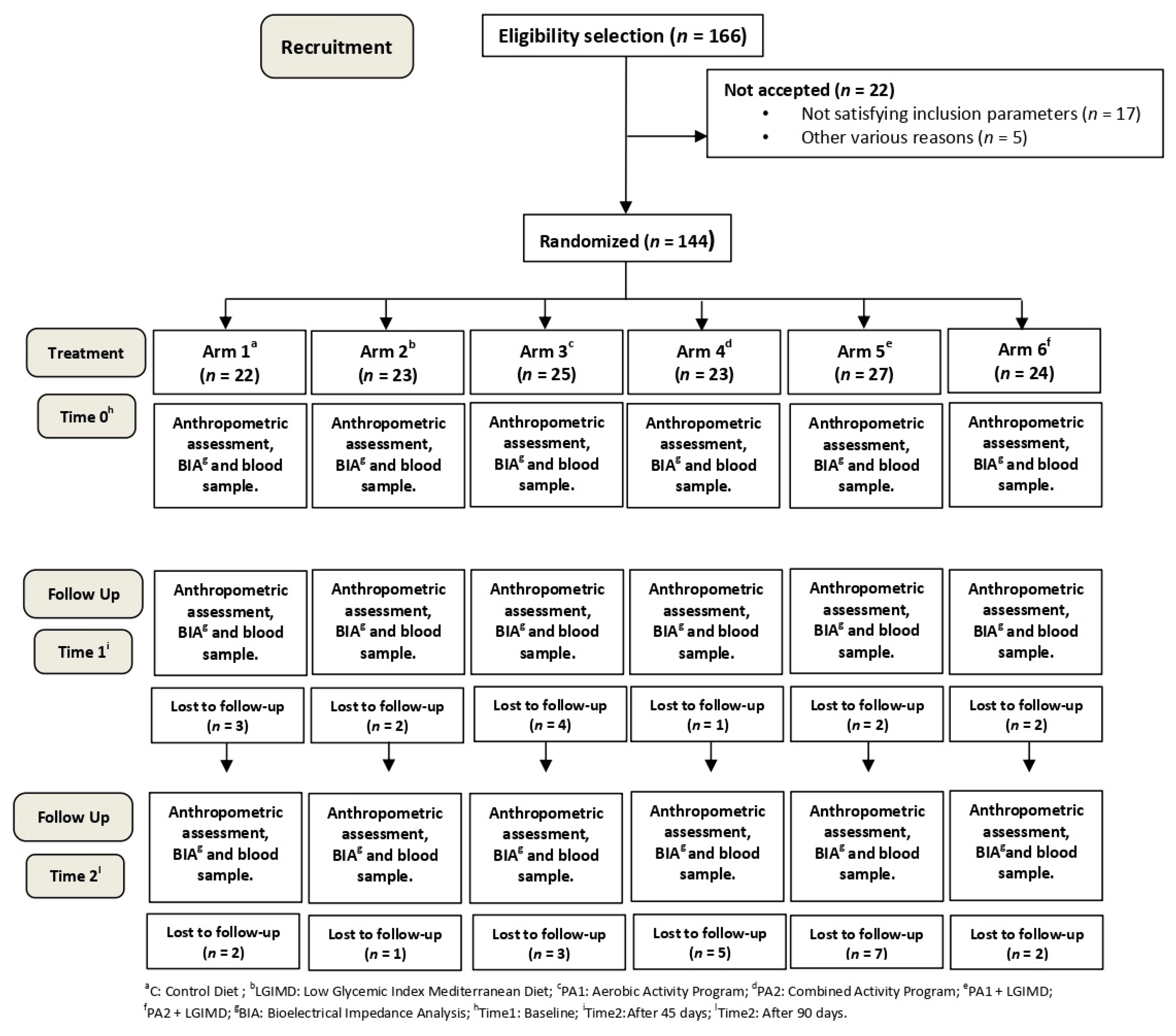
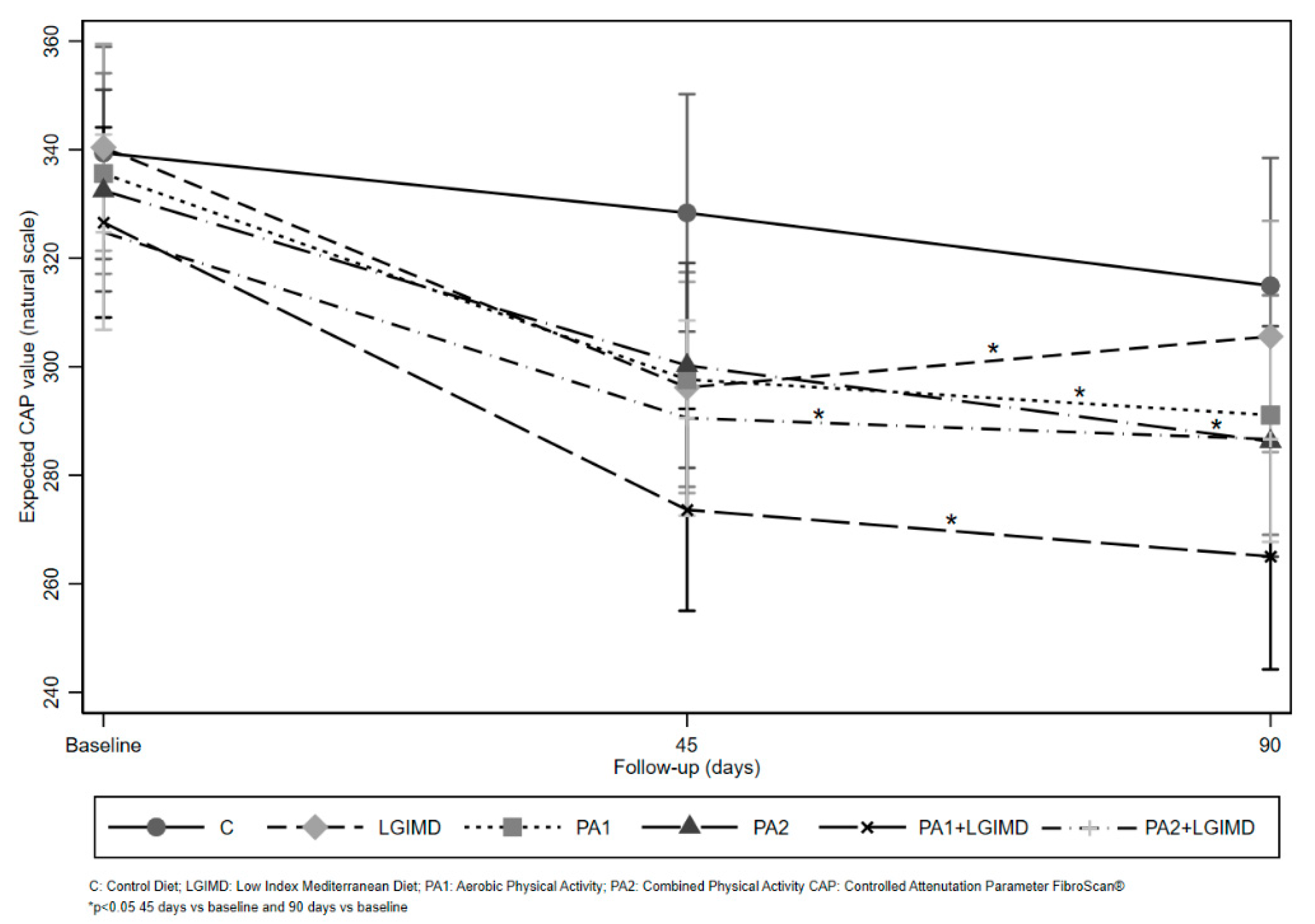
| Variables | Time | Working Arms | ||||||
|---|---|---|---|---|---|---|---|---|
| C a | LGIMD b | PA1 c | PA2 d | PA1 + LGIMD e | PA2 + LGIMD f | p-Value | ||
| N | Baseline | 22 (15.3%) | 23 (16.0%) | 25 (17.4%) | 23 (16.0) | 27 (18.7%) | 24 (16.7%) | |
| Age (Years) | 50.70 (8.67) | 50.74 (1.75) | 50.45 (9.45) | 46.23 (9.39) | 50.32 (9.61) | 46.75 (10.50) | 0.11 | |
| Age (Categorical) | <40 | 1 (6%) | 1 (6%) | 1 (6%) | 5 (28%) | 3 (17%) | 7 (39%) | 0.025 |
| 40–49 | 8 (16%) | 3 (6%) | 10 (20%) | 9 (18%) | 12 (24%) | 8 (16%) | ||
| ≥50 | 11 (17%) | 18 (28%) | 9 (14%) | 8 (12%) | 10 (15%) | 9 (14%) | ||
| Gender | ||||||||
| Male | 11 (12%) | 13 (15%) | 14 (16%) | 17 (19%) | 18 (20%) | 16 (18%) | 0.57 | |
| Female | 11 (20%) | 10 (18%) | 11 (20%) | 6 (11%) | 9 (16%) | 8 (15%) | ||
| CAP (dB/m) g | Baseline | 349.09 (60.28) | 341.57 (45.17) | 339.92 (44.89) | 334.78 (51.01) | 328.56 (52.05) | 323.63 (53.98) | 0.58 |
| 45 days | 322.12 (83.18) | 296.52 (51.10) | 295.10 (50.68) | 299.55 (63.73) | 275.61 (75.84) | 289.38 (70.15) | 0.39 | |
| 90 days | 307.80 (73.10) | 303.72 (81.70) | 287.88 (54.59) | 284.11 (62.22) | 259.84 (57.01) | 289.18 (72.96) | 0.21 | |
| NAFLD h | ||||||||
| Moderate | Baseline | 6 (13.6%) | 5 (11.4%) | 6 (13.6%) | 7 (15.9%) | 10 (22.7%) | 10 (22.7%) | 0.64 |
| 45 days | 3 (7.0%) | 8 (19.0%) | 7 (17.0%) | 10 (24.0%) | 5 (12.0%) | 9 (21.0%) | 0.45 | |
| 90 days | 4 (13.0%) | 3 (9.0%) | 3 (9.0%) | 8 (25.0%) | 5 (16.0%) | 9 (28.0%) | 0.20 | |
| Severe | Baseline | 16 (16.0%) | 18 (18.0%) | 19 (19.0%) | 16 (16.0%) | 17 (17.0%) | 14 (14.0%) | 0.64 |
| 45 days | 13 (21.0%) | 10 (16.0%) | 12 (19.0%) | 10 (16.0%) | 10 (16.0%) | 8 (13.0%) | 0.45 | |
| 90 days | 12 (18.0%) | 11 (17.0%) | 15 (23.0%) | 8 (12.0%) | 10 (15.0%) | 9 (14.0%) | 0.20 | |
| Working Arms | |||||||
|---|---|---|---|---|---|---|---|
| Variables | C a | LGIMD b | PA1 c | PA2 d | PA1 + LGIMD e | PA2 + LGIMD f | p-Value * |
| N | 22 | 23 | 25 | 23 | 27 | 24 | |
| Status | 0.32 | ||||||
| Single | 1 (6%) | 1 (6%) | 1 (5%) | 3 (19%) | 2 (10%) | 3 (16%) | |
| Married | 17 (94%) | 17 (94%) | 18 (90%) | 11 (69%) | 17 (81%) | 16 (84%) | |
| Divorced | 0 (0%) | 0 (0%) | 1 (5%) | 0 (0%) | 2 (10%) | 0 (0%) | |
| Widowed | 0 (0%) | 0 (0%) | 0 (0%) | 2 (13%) | 0 (0%) | 0 (0%) | |
| Study Level | 0.07 | ||||||
| Elementary | 1 (6%) | 2 (11%) | 0 (0%) | 0 (0%) | 1 (5%) | 0 (0%) | |
| Secondary School | 10 (56%) | 8 (42%) | 8 (40%) | 3 (18%) | 4 (19%) | 6 (30%) | |
| High School | 6 (33%) | 7 (37%) | 9 (45%) | 9 (53%) | 8 (38%) | 12 (60%) | |
| Diploma University | 1 (6%) | 1 (5%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | |
| University Degree | 0 (0%) | 1 (5%) | 3 (15%) | 5 (29%) | 8 (38%) | 2 (10%) | |
| Smoking status | 0.80 | ||||||
| Never smoked | 13 (68%) | 10 (50%) | 12 (55%) | 10 (56%) | 17 (71%) | 12 (55%) | |
| Former smoker | 5 (26%) | 6 (30%) | 5 (23%) | 4 (22%) | 4 (17%) | 4 (18%) | |
| Current smoker | 1 (5%) | 4 (20%) | 5 (23%) | 4 (22%) | 3 (13%) | 6 (27%) | |
| Working Arm | Male | Female | |||||
|---|---|---|---|---|---|---|---|
| Test | Baseline | 45 Days | 90 Days | Baseline | 45 Days | 90 Days | |
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | ||
| C a | 2 Walking (sec) * | 97.39 (17.50) | N/A ≠ | 103.09 (12.18) | 14.33 (56.75) | N/A ≠ | 27.69 (37.69) |
| Push-up * | 11.75 (8.81) | N/A ≠ | 13.13 (10.05) | 4.38 (4.24) | N/A ≠ | 9.50 (5.47) | |
| Sit and Reach | −11.00 (10.06) | N/A ≠ | −10.38 (11.11) | −6.25 (10.78) | N/A ≠ | −9.00 (13.61) | |
| LGIMD b | 2 Walking (sec) * | 105.73 (24.97) | N/A ≠ | 100.05 (12.85) | 13.18 (39.20) | N/A ≠ | 39.19 (30.90) |
| Push -up * | 8.92 (7.80) | N/A ≠ | 11.64 (8.50) | 6.38 (5.90) | N/A ≠ | 11.50 (10.01) | |
| Sit and Reach | −9.00 (9.28) | N/A ≠ | −11.36 (6.48) | −11.63 (9.50) | N/A ≠ | −7.00 (8.65) | |
| PA1 c | 2 Walking (sec) * | 87.30 (17.30) | 100.81 (16.10) | 101.88 (9.15) | 27.22 (39.52) | 42.60 (21.97) | 58.46 (26.18) |
| Push-up * | 10.50 (7.86) | 13.75 (10.93) | 15.30 (10.73) | 9.13 (7.08) | 10.50 (7.52) | 15.29 (10.01) | |
| Sit and Reach | −11.21 (8.72) | −10.50 (9.41) | −7.00 (10.26) | −9.25 (10.87) | −6.25 (9.32) | −4.00 (10.49) | |
| PA2 d | 2 Walking (sec) * | 94.08 (10.74) | 96.20 (10.59) | 103.83 (12.16) | 56.29 (20.92) | 63.28 (29.55) | 65.18 (36.41) |
| Push-up * | 11.06 (7.24) | 15.19 (6.72) | 16.67 (7.79) | 6.67 (5.61) | 7.33 (5.20) | 11.50 (2.89) | |
| Sit and Reach | −9.81 (6.65) | −7.81 (7.60) | −4.73 (8.55) | −5.83 (6.55) | −5.83 (5.49) | −7.25 (6.08) | |
| PA1+LGIMD e | 2 Walking (sec) * | 82.90 (12.69) | 95.85 (11.97) | 105.91 (9.26) | 33.47 (30.60) | 42.09 (25.12) | 43.48 (27.77) |
| Push -up * | 9.06 (6.87) | 13.73 (8.56) | 17.17 (9.13) | 14.56 (11.45) | 15.56 (8.75) | 15.25 (9.13) | |
| Sit and Reach | −12.69 (8.10) | −11.53 (8.64) | −9.50 (7.87) | −3.11 (7.77) | −2.67 (7.92) | −2.63 (8.31) | |
| PA2+LGIMD f | 2 Walking (sec) * | 87.26 (16.17) | 102.99 (12.36) | 102.76 (13.39) | 32.24 (26.80) | 50.08 (26.30) | 61.78 (31.95) |
| Push-up * | 7.81 (4.86) | 11.00 (6.78) | 15.14 (8.50) | 11.00 (7.52) | 16.00 (9.90) | 18.25 (7.63) | |
| Sit and Reach | −18.19 (9.23) | −14.38 (10.31) | −13.79 (11.20) | −4.38 (7.85) | −1.63 (6.97) | 0.25 (7.83) | |
| Weeks | PA1 a | PA2 b | LGIMD + PA1 c | LGIMD + PA2 d | |||||
|---|---|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | Male | Female | Male | Female | ||
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | ||
| 3 | Time | 96.5 (9.70) | 100 (0) | 100 (0) | 100 (0) | 97.8 (8.60) | 93.7 (12.40) | 100 (0) | 100 (0) |
| Intensity | 101.6 (3.81) | 100.5 (1.54) | 100.1 (0.61) | 100 (0) | 99.9 (1.22) | 100 (0) | 99.9 (0.45) | 99.4 (2.41) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 4 | Time | 92.4 (15.8) | 89.6 (19.8) | 100 (0) | 100 (0) | 100 (0) | 100 (0) | 100 (0) | 97.4 (9.24) |
| Intensity | 101.5 (3.20) | 100.5 (1.54) | 100.1 (0.61) | 100 (0) | 100.3 (0.63) | 100 (0) | 99.9 (0.51) | 99.4 (2.41) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 5 | Time | 94.4 (12.9) | 95.8 (11.8) | 95.8 (11.4) | 94.4 (13.6) | 100 (0) | 100 (0) | 100 (0) | 94.9 (12.5) |
| Intensity | 101.0 (2.32) | 100.1 (0.28) | 100.1 (0.61) | 100 (0) | 100.6 (1.56) | 100 (0) | 100.1 (0.40) | 98.9 (2.81) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 6 | Time | 94.2 (11.5) | 100 (0) | 93.7 (13.4) | 94.4 (13.6) | 100 (0) | 100 (0) | 100 (0) | 97.4 (9.24) |
| Intensity | 100.5 (3.07) | 100.1 (0.28) | 99.9 (0.41) | 100 (0) | 100.9 (1.84) | 100 (0) | 100.1 (0.40) | 98.9 (2.81) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 7 | Time | 93.0 (13.2) | 92.0 (22.6) | 95.8 (11.4) | 94.4 (13.6) | 97.8 (8.60) | 87.5 (24.8) | 100.0 (0) | 92.3 (20.0) |
| Intensity | 100.6 (1.53) | 100.1(0.28) | 100 (0) | 100 (0) | 100.5 (1.03) | 100.0 (0) | 100.1 (0.40) | 98.9 (2.81) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 8 | Time | 88.2 (19.3) | 91.7 (15.4) | 95.8 (16.7) | 100 (0) | 100 (0) | 83.3 (27.9) | 100 (0) | 97.4 (9.24) |
| Intensity | 100.5 (1.66) | 100.1 (0.28) | 100 (0) | 100 (0) | 100.6 (1.08) | 100 (0) | 100.1 (0.40) | 98.9 (2.81) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 9 | Time | 93.4 (16.1) | 94.8 (11.7) | 93.3 (13.8) | 91.7 (16.7) | 97.4 (9.24) | 88.9 (17.2) | 97.9 (8.33) | 97.4 (9.24) |
| Intensity | 99.6 (1.26 | 100 (0) | 100.2 (0.73) | 100 (0) | 100.1 (1.07) | 100 (0) | 100.2 (0.79) | 98.6 (3.43) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| 10 | Time | 98.4 (5.09) | 84.5 (19.5) | 93.3 (13.8) | 91.7 (16.7) | 97.4 (9.24) | 94.4 (13.6) | 97.9 (8.33) | 94.8 (12.5) |
| Intensity | 99.6 (1.26) | 100 (0) | 100.2 (0.73) | 100 (0) | 100.1 (1.07) | 100 (0) | 100.2 (0.79) | 98.8 (3.62) | |
| Load | N/A * | N/A * | 100 (0) | 100 (0) | N/A * | N/A * | 100 (0) | 100 (0) | |
| Male | Female | ||||||
|---|---|---|---|---|---|---|---|
| 3rd Week | 6th Week | 9th Week | 3rd Week | 6th Week | 9th Week | ||
| Age (Years) | Diet | Median (IQR) | Median (IQR) | Median (IQR) | Median (IQR) | Median (IQR) | Median (IQR) |
| <40 | CD a | 2.3 | 2.3 | 3.5 | 7.8 | 7.8 | 0.9 |
| n | 1 | 1 | 1 | 1 | 1 | 1 | |
| LGIMD b | 7.8 (1.6; 19.1) | 7.8 (1.6; 19.1) | 7.8 (1.6; 19.1) | 4.4 (0.9; 8.7) | 4.4 (0.9; 8.7) | 4.4 (0.9; 8.7) | |
| n | 8 | 8 | 8 | 8 | 8 | 8 | |
| 40–49 | CD a | 4.7 (2.0; 9.4) | 4.7 (2.6; 15.2) | 4.9 (2.6; 15.2) | 2.1 (0.9; 3.3) | 2.1 (0.9; 3.3) | 2.1 (1.6; 3.3) |
| n | 2 | 2 | 2 | 5 | 5 | 4 | |
| LGIMD b | 3.2 (1.3; 5.6) | 3.2 (1.3; 5.6) | 3.2 (1.3; 5.6) | 2.1 (1.1; 3.2) | 2.1 (1.1; 3.2) | 2.1 (1.1; 3.2) | |
| n | 18 | 18 | 18 | 9 | 9 | 9 | |
| ≥50 | CD a | 4.7 (3.4; 12.3) | 5.2 (3.4; 12.4) | 4.7 (3.7; 12.4) | 5.9 (3.6; 9.6) | 5.9 (3.6; 9.6) | 5.9 (3.6; 9.0) |
| n | 8 | 7 | 7 | 5 | 3 | 2 | |
| LGIMD b | 4.7 (3.4; 12.3) | 4.7 (3.4; 12.3) | 4.7 (3.4; 12.3) | 5.9 (3.7; 9.5) | 5.9 (3.7; 9.5) | 5.6 (3.3; 9.5) | |
| n | 30 | 30 | 30 | 17 | 17 | 17 |
| CAP b | β | SE | p-Value | (CI 95%) |
|---|---|---|---|---|
| Working arms | ||||
| C c | 0 | |||
| LGIMD d | −55.81 | 38.61 | 0.150 | (−131.90; 20.29) |
| PA1 e | −166.35 | 38.39 | 0.000 | (−242.01; −90.68) |
| PA2 f | −78.02 | 38.54 | 0.044 | (−153.97; −2.07) |
| PA1+LGIMD g | −94.10 | 38.50 | 0.014 | (−170.87; −19.12) |
| PA2+LGIMD h | −76.37 | 38.45 | 0.048 | (−152.14; −0.60) |
| Time | ||||
| Baseline | 0 | |||
| 45 days | −11.04 | 15.28 | 0.471 | (−41.16; 19.07) |
| 90 days | −24.46 | 16.04 | 0.129 | (−56.07; 7.15) |
| Time * Working arms: | ||||
| (45 days vs. base) C c | −11.04 | 15.28 | 0.471 | (−41.16; 19.07) |
| (90 days vs. base) C c | −24.46 | 16.04 | 0.129 | (−56.07; 7.15) |
| (45 days vs. base) LGIMD d | −44.24 | 14.10 | 0.002 | (−72.03; −16.45) |
| (90 days vs. base) LGIMD d | −34.84 | 14.97 | 0.021 | (−64.34; −5.34) |
| (45 days vs. base) PA1 e | −37.95 | 14.10 | 0.008 | (−65.75; −10.14) |
| (90 days vs. base) PA1 e | −44.50 | 15.01 | 0.003 | (−74.08; −14.92) |
| (45 days vs. base) PA2 f | −32.23 | 13.53 | 0.018 | (−58.88; −5.57) |
| (90 days vs. base) PA2 f | −46.22 | 14.61 | 0.002 | (−75.02; −17.43) |
| (45 days vs. base) PA1+LGIMD g | −52.96 | 13.23 | 0.000 | (−79.03; −26.88) |
| (90 days vs. base) PA1+LGIMD g | −61.56 | 14.23 | 0.000 | (−89.61; −33.50) |
| (45 days vs. base) PA2+LGIMD h | −34.25 | 12.95 | 0.009 | (−59.77; −8.73) |
| (90 days vs. base) PA2+LGIMD h | −38.15 | 13.38 | 0.005 | (−64.53; −11.77) |
| NAFLD a | ||||
|---|---|---|---|---|
| Moderate | Severe | |||
| CAP b | Contrast | (CI 95%) | Contrast | (CI 95%) |
| (45 days vs. base) C c | −15.50 | (−64.02; 33.02) | −8.73 | (−45.28; 27.81) |
| (90 days vs. base) C c | −29.00 | (−77.52; 19.52) | −21.82 | (−61.38; 17.73) |
| (45 days vs. base) LGIMD d | −13.80 | (−66.96; 39.36) | −54.27 | (−85.89; −22.64) ** |
| (90 days vs. base) LGIMD d | −22.65 | (−80.58; 35.28) | −39.13 | (−72.53; −5.73) * |
| (45 days vs. base) PA1 e | −6.67 | (−55.19; 41.86) | −51.36 | (−84.09; −18.62) * |
| (90 days vs. base) PA1 e | 10.67 | (−37.86; 59.19) | −72.77 | (−108.69; −36.85) ** |
| (45 days vs. base) PA2 f | 18.14 | (−26.78; 63.07) | −55.73 | (−87.35; −24.11) ** |
| (90 days vs. base) PA2 f | 18.40 | (−29.24; 66.05) | −77.70 | (−112.16; −43.24) ** |
| (45 days vs. base) PA1 + LGIMD g | −23.75 | (−65.77; 18.27) | −68.53 | (−100.15; −36.91) ** |
| (90 days vs. base) PA1 + LGIMD g | −23.09 | (−37.31; 21.13) | −83.27 | (−117.72; −48.81) ** |
| (45 days vs. base) PA2 + LGIMD h | −27.4 | (−64.99; 10.19) | −39.14 | (−71.87; −6.41) * |
| (90 days vs. base) PA2 + LGIMD h | −46.31 | (−85.43; −7.19) * | −32.57 | (−66.23; 1.09) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franco, I.; Bianco, A.; Mirizzi, A.; Campanella, A.; Bonfiglio, C.; Sorino, P.; Notarnicola, M.; Tutino, V.; Cozzolongo, R.; Giannuzzi, V.; et al. Physical Activity and Low Glycemic Index Mediterranean Diet: Main and Modification Effects on NAFLD Score. Results from a Randomized Clinical Trial. Nutrients 2021, 13, 66. https://doi.org/10.3390/nu13010066
Franco I, Bianco A, Mirizzi A, Campanella A, Bonfiglio C, Sorino P, Notarnicola M, Tutino V, Cozzolongo R, Giannuzzi V, et al. Physical Activity and Low Glycemic Index Mediterranean Diet: Main and Modification Effects on NAFLD Score. Results from a Randomized Clinical Trial. Nutrients. 2021; 13(1):66. https://doi.org/10.3390/nu13010066
Chicago/Turabian StyleFranco, Isabella, Antonella Bianco, Antonella Mirizzi, Angelo Campanella, Caterina Bonfiglio, Paolo Sorino, Maria Notarnicola, Valeria Tutino, Raffaele Cozzolongo, Vito Giannuzzi, and et al. 2021. "Physical Activity and Low Glycemic Index Mediterranean Diet: Main and Modification Effects on NAFLD Score. Results from a Randomized Clinical Trial" Nutrients 13, no. 1: 66. https://doi.org/10.3390/nu13010066
APA StyleFranco, I., Bianco, A., Mirizzi, A., Campanella, A., Bonfiglio, C., Sorino, P., Notarnicola, M., Tutino, V., Cozzolongo, R., Giannuzzi, V., Aballay, L. R., Buongiorno, C., Bruno, I., & Osella, A. R. (2021). Physical Activity and Low Glycemic Index Mediterranean Diet: Main and Modification Effects on NAFLD Score. Results from a Randomized Clinical Trial. Nutrients, 13(1), 66. https://doi.org/10.3390/nu13010066





